Abstract
Gossence™ (galactooligosaccharide; GOS) is a prebiotics and used as one of the major constituents in infant milk formulas that act as a functional food. Gossence is manufactured by Tata Chemicals Ltd, India, through a patented process of biotransformation of lactose. A toxicology study in juvenile rats was carried out to assess the safety profile of Gossence intended for pediatric population. The objective of this study is to assess the potential systemic toxicity of Gossence when administered through gavage at dose levels of 1000, 2000, or 5000/3000 mg/kg/day (equivalent to 1347, 2694, and 6735/4041 mg/kg/day of GOS, respectively) to juvenile Sprague Dawley rats from postnatal day (PND) 4 to PND 52 (i.e. total 49 days of dosing period). A separate group of animals were treated with vehicle (purified Milli Q water) for a similar duration. The following parameters were evaluated during the study period: morbidity/mortality check, clinical signs, body weights, body weight changes, food consumption, functional observational battery, motor activity, postnatal developmental observations, hematology, clinical chemistry, urinalysis, organ weight, gross pathology, and histopathology. During dosing phase, the high-dose group, 5000 mg/kg/day, was reduced to 3000 mg/kg/day (equivalent to 4041 mg/kg/day dose of GOS) from day 16 (PND 19) onward, due to clinical signs of watery feces and yellow color stains at urogenital region and mortality in two animals on day 15 (PND 18) of the study. Time-weighted average dose for 5000 mg/kg/day was equivalent to 3600 mg/kg/day. No further deaths or clinical signs were noticed in animals at 3000 mg/kg/day from day 18 (PND 21) of dosing phase to until terminal euthanization. At the terminal euthanization, there were no test item-related gross changes observed in all surviving rats except for, an increased cecum size in some of the rats at 5000/3000 mg/kg/day, which correlated with the increased weights of cecum with contents during organ weight recording, but this had no correlating light microscopic changes during histological examination. The cecal enlargement was completely recovered following the 14-day recovery period. The no-observedadverse-effect level is 3000 mg/kg/day for Gossence, which is equivalent to 4041 mg/kg/day of GOS in both sexes.
Keywords
Introduction
Prebiotics are nondigestible nutrients, which influence the growth and metabolic activity of beneficial bacteria in the gut flora, leading to health benefits for humans. Hence, these prebiotics are extensively added to dairy products, resulting in long-term consumption among pregnant mothers and young infants. 1,2 Galactooligosaccharides (GOS), also known as oligoglalactosyllactose, oligogalactose, oligolactose, or trans-galactooligosaccharides (TOS), belong to a group of prebiotics. Prebiotics, TOS, have been shown to increase bifidobacteria counts and decrease harmful pathogens like Escherichia coli and Clostridium lituseburense/Clostridium histolyticum in the gut of infants. 5,6 The fermentation products of prebiotics such as acetate, butyrate, and propionate are found to have immunomodulatory effects that lead to increased protection against infections. 7 GOS provide health benefits, by inducing selective proliferation of bifidobacteria and lactobacilli in the guts, which in turn provide resistance against colonization of pathogens, thereby reducing the infections. Also, GOS facilitates to maintain a healthy balance of intestinal flora by producing organic acids, like lactic acid and acetic acid, which in turn increases the acidity of intestine and inhibits the growth of harmful bacteria. As a result, GOS improve the symptoms of diarrhea and constipation. GOS produces short-chain fatty acids, which reduces the risk of cancer, increases absorption of minerals, improves bowel, and controls serum lipid and cholesterol levels. 8 –12 Human breast milk is the natural source of GOS for infants and contains approximately 100 times more GOS than cow’s milk. 4 The infants fed with the human breast milk have bifidobacteria as the predominant microflora. For this reason, manufactured GOS are incorporated as the most common component of infant formulas to strengthen functional attributes associated with breast milk. 3 –5,12 –14 GOS are globally used in various commercial products such as infant formulas, various dairy products, breakfast cereals, beverages, and product related to bakery items. 13
Gossence™ is a GOS (CAS number: 6587-31-1), containing product synthesized by enzymatic action using whole microbial cells of Sporobolomyces singularis MTCC5491 in a manner similar to other GOS products. GOS increases stool frequency and softens feces in young people with mild constipation. 15,16 As a general adverse reaction of indigestible oligosaccharides, diarrhea is a common occurrence due to the indigestibility of the oligosaccharides, and the increase in osmotic pressure depends on the amount of lactose in the oligosaccharides. 16,17
In recent years, juvenile toxicity testing is a method to assess the safety profile for pediatric population by various agencies such as Best Pharmaceuticals for Children Act and the Pediatric Research Equity Act in 2002 and 2003, respectively. 18,19 Juvenile toxicity studies are performed to support the development of drugs intended for use in pediatric patients and to obtain information on the potential different safety profiles from those seen in adults. These studies provide information that is not adequately addressed in general toxicity studies conducted in adult animals and to investigate findings that cannot be adequately or ethically studied in pediatric clinical trials. Juvenile toxicity studies are conducted for drugs that have a novel pharmacological mechanism (first in class) and for drugs that have shown specific adverse effects or toxicity in pediatric population or in juvenile animals in same pharmacological class. 19 –21
In this study, the safety of Gossence (GOS) was evaluated in juvenile Sprague Dawley rats to support pediatric population. The objective of the study was to assess the potential systemic toxicity of Gossence upon 7-week daily gavage administration to juvenile Sprague Dawley rats from postnatal days (PNDs) 4 to 53.
Materials and methods
Test item details
Gossence (GOS) was supplied by Tata Chemicals Limited, produced in their good manufacturing practice (GMP) facility at Sriperumbudur, Tamil Nadu, India. Gossence contains approximately 76.28% of GOS and 23.72% of lactose and others on a dry basis and was stored at room temperature. The details of the physicochemical properties of Gossence (GOS) are summarized in Table 1.
Test material composition and identity.
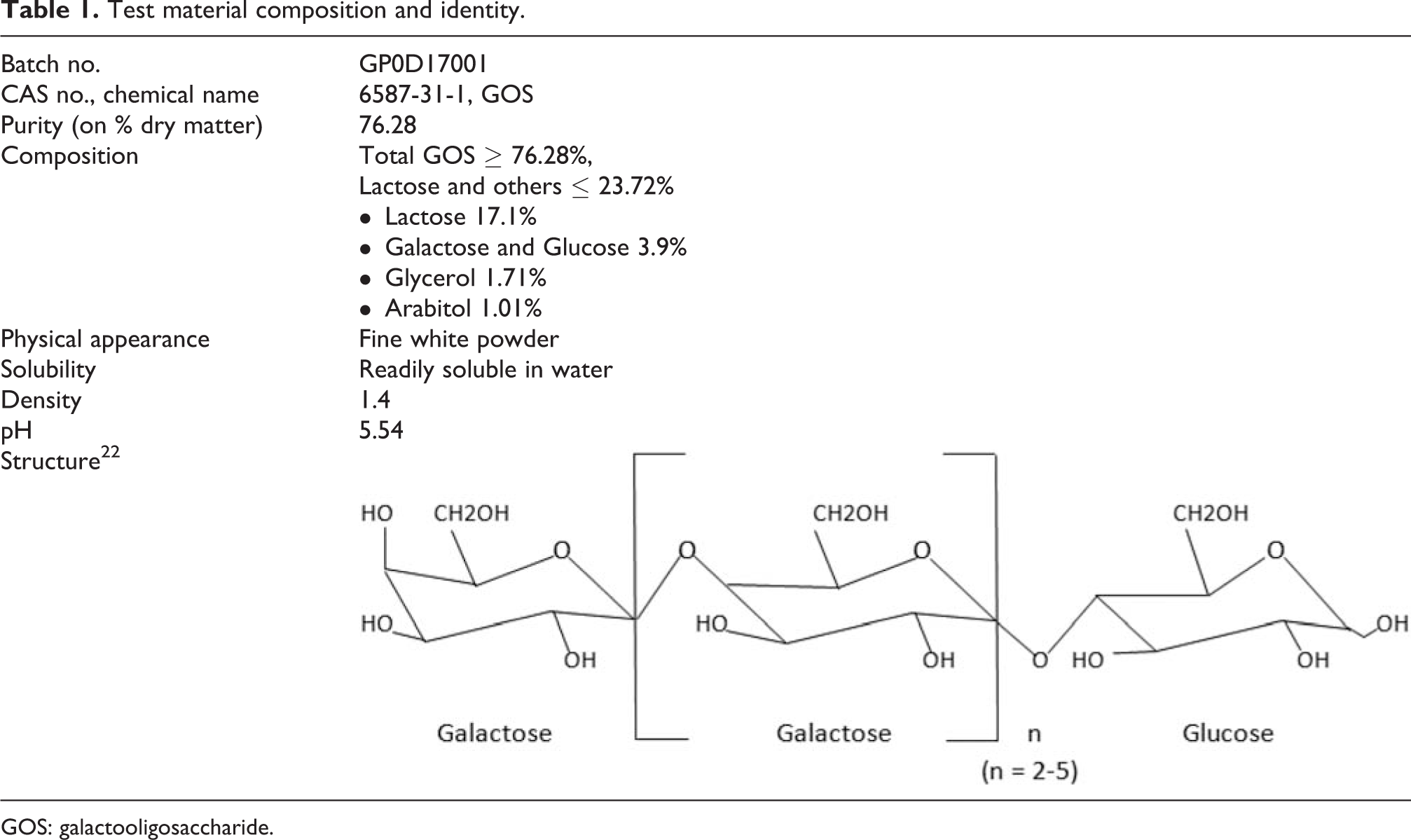
GOS: galactooligosaccharide.
Compliance statement
The studies were performed in accordance with the regulatory guidelines: Committee for Human Medicinal Products (CHMP), EMEA/CHMP/SWP/169215/2005, “Guideline on the need for Non-clinical Testing in Juvenile Animals of Pharmaceuticals for Pediatrics Indications,” dated January 24, 2008; US Department of Health and Human Services Food and Drug Administration (Centre for Drug Evaluation and Research (CDER)), Guideline for Industry: “Non-clinical Safety Evaluation of Pediatric Drug Products” February 2006; and International Council for Harmonization of Technical Requirement for Pharmaceuticals for Human Use (ICH) Harmonized Tripartite Guideline, “Guidance on Non-clinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals,” M3 (R2); Current Step 4 version, dated June 11, 2009. This study was performed in compliance with the Organization for Economic Co-operation and Development (OECD) Principles of Good Laboratory Practice (GLP) (as revised in 1997), ENV/MC/CHEM (98) 17, OECD, Paris, 1998 (No. 1 in OECD Series on GLP and Compliance Monitoring) concerning Mutual Acceptance of Data in the Assessment of Chemicals [C (81)30(FINAL)] and Council Decision-Recommendation [C (95)8(FINAL)] and as per test facility Standard Operating Procedures prevailing during the study. These principles are equivalent to the US Food and Drug Administration’s (USFDA) GLP Regulations (21 CFR Part 58). All animals were handled humanely with due regard for their welfare and complied with the recommendations of the Association for Assessment and Accreditation of Laboratory Animal Care International and Regulations of Committee for the Purpose of Control and Supervision of Experiments on Animals, Government of India.
Dose formulation preparation and analysis
Potable water, purified with Millipore Elix® 3 water purification system, was used as a vehicle for dose formulation preparation. The stability analysis of GOS in the vehicle was determined in-house at concentrations 100, 200, and 500 mg/mL. GOS was stable up to 7 days at refrigerated (2–8°C) conditions and up to 24 h at room temperature (20–25°C). Since the dose formulation was a clear solution, homogeneity analysis was not performed. The dose formulation was prepared daily and used within the prescribed stability period. Dose formulations prepared for day 1 dosing and toward the end of dosing period (i.e. day 46) were analyzed for major ingredient (4′-galactosyllactose) by validated high-performance liquid chromatography techniques. Gossence (GOS) is a mixture of different forms of oligosaccharides and majorly contains 4′-galactosyllactose with CAS registry number 6587-31-1.
Animals
For juvenile toxicity study, rat is selected as a test system, as it is referred by Test Guidelines (EPA and ICH). Historically, rat has been found to be a suitable model for safety evaluation. Sprague Dawley rat has been accepted and widely used for nonclinical safety studies.
Twenty pregnant female Sprague Dawley rats (gestation days 17–18) were obtained from Vivo Bio Tech, Hyderabad, India, quarantined and acclimated to the experimental room until delivery. Animals were maintained in a controlled environment in the experimental animal room with a temperature range of 19.5–22.8°C, relative humidity range of 53–69% with 12-h light and 12-h dark cycle, and 18–21 fresh air changes per hour. Pregnant females were housed in individual cages during gestation, with litters during lactation period and up to a preweaning day (day 21). The sterilized nesting material (plain white paper shreds) were provided near-term and during lactation period as an enrichment. Autoclaved corncob was used as the bedding.
The pregnant female rats were allowed to litter naturally. The day of delivery was designated as day “0” of lactation (postpartum). Each litter was observed as soon as possible after delivery for the number of pups born, sex of each pup, and presence of any gross abnormalities. Dams (F0 generation dams) were used to maintain F1 generation pups and hence were not considered as a part of the test system.
On day 3 of the postpartum, the size of each litter was adjusted to four pups/sex/litter for selection of main group animals and three pups/sex/litter for selection of recovery group animals by removing extra pups randomly. The extra pups that are not assigned to the study were culled on day 3 of the postpartum (i.e. on the day of litter standardization/ randomization). No pup was cross-fostered during litter standardization, as each dam had enough pups to select appropriate numbers of pups for each dose group.
On PND 3, a total 120 pups (60 males and 60 females) from 16 dams littered on same day were randomly selected for the study and assigned to 4 main groups (G1–G4; 12 pups/sex/group) and 2 recovery groups (G1R and G4R; 6 pups/sex/group), based on body weight stratification method using Pristima version 7.2.0 (Xybion Medical Systems, Lawrenceville, New Jersey, USA). The body weight variation of the pups was slightly exceeded ±20% of the mean body weight (up to ±37% for male pups and up to ±33% for female pups) each sex at the time of randomization. Prior to selecting the pups for study, they were checked for clinical signs and only those pups that are free from health abnormalities were selected for the study.
The unselected pups, which are not assigned to any dose group after randomization were discarded on day of litter randomization (i.e. on PND 3 of the study). Pups were acclimatized to the experimental room for 3 days prior to dose administration. Dams were euthanized postweaning, and the weaned pups were housed two per sex in clean, sterilized polycarbonate rat cages. Animals were fed ad libitum with certified rodent pellet feed (Teklad Certified Irradiated Global 18% Protein Rodent Diet; Envigo, Madison, Wisconsin, USA) and maintenance diet for rat and mice (Gamma Irradiated, Altromin, Germany). Potable water filtered through reverse osmosis was provided ad libitum to all animals, via polycarbonate bottles fitted with stainless steel sipper tubes. Every effort was made to minimize and eliminate pain and suffering of all the animals in the study. The “Form B” for carrying out animal experimentation was reviewed and approved by the Institutional Animal Ethics Committee (IAEC Protocol No: SYNGENE/IAEC/920/03-2018), India.
Study design
A 7-week gavage juvenile toxicity study followed by 14 days of recovery period was conducted in Sprague Dawley rats. The dose levels of 1000 (low), 2000 (mid), and 5000/3000 (high) mg/kg/day of Gossence (equivalent to 1347, 2694, and 6735/4041 mg/kg/day dose of GOS, respectively) were selected for juvenile toxicity study based on the results of a 14-day dose range-finding study conducted at Syngene International Ltd, Bangalore, Karnataka, India, in Sprague Dawley rats. Seven-week duration of dosing for juvenile rats was selected to cover an initial developmental stage of target organs/functions of the intended pediatric population. Moreover, in-house 14-day dose range finding study and 90-day repeat-dose toxicity study were conducted for GOS, and the dosing was initiated at 7-week-old animals (young adult animals). Thus, juvenile toxicity study covered the potential effect of GOS from PND 4 to young adult animals. Gavage was selected as the route of administration, as it is an intended clinical route of administration in pediatric population. The study design is given in Table 2.
Study design.
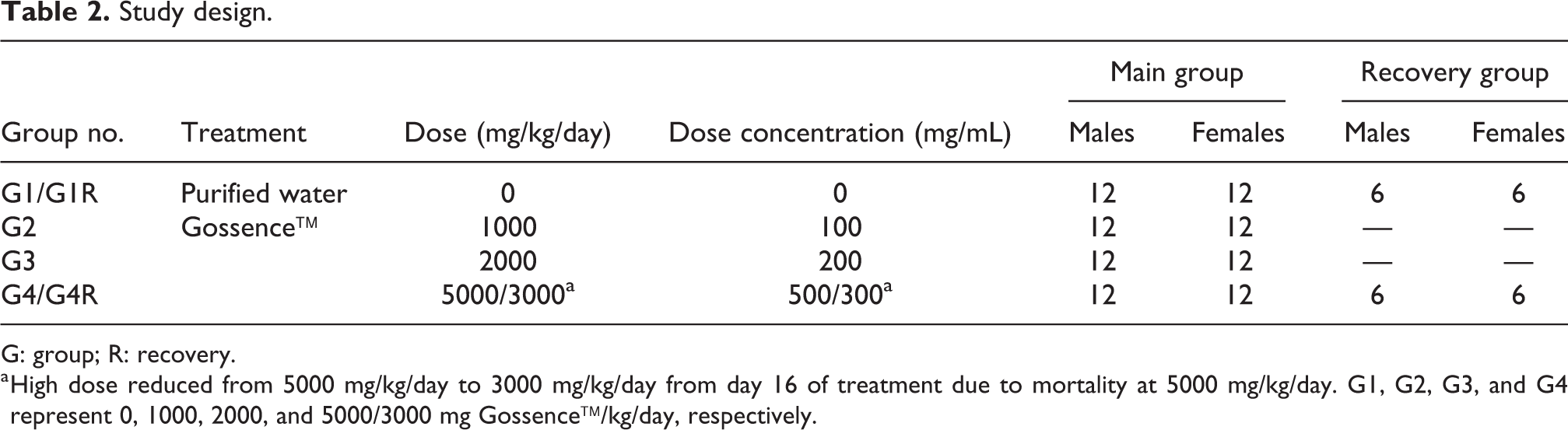
G: group; R: recovery.
a High dose reduced from 5000 mg/kg/day to 3000 mg/kg/day from day 16 of treatment due to mortality at 5000 mg/kg/day. G1, G2, G3, and G4 represent 0, 1000, 2000, and 5000/3000 mg Gossence™/kg/day, respectively.
Pups were dosed once daily for 7 consecutive weeks through gavage using flexible rat intubation cannula (20-gauge Instech (Instech Laboratory Inc., Plymouth Meeting, Pennsylvania, USA) and 18-gauge Harvard (Cadence Inc., Cranston, Rhode Island, USA) oral intubation cannula) at the dose levels of 1000, 2000, and 5000/3000 mg/kg/day from PNDs 4 to 52 (i.e. total 49 days of treatment duration). High dose was reduced to 3000 mg/kg/day Gossence (equivalent to 4041 mg/kg/day dose of GOS) from day 16 (PND 19) onward, due to mortality observed in two animals. Time-weighted average dose of 5000 mg/kg/day was equivalent to 3600 mg/kg/day. Animals in the control group received the vehicle (purified water) for a similar duration. A dose volume of 10 mL/kg was maintained for all the dose groups. The volume administered was calculated for individual animals based on the body weight taken prior to treatment on day 1 (i.e. on PND 4), and thereafter, dose volume was adjusted twice weekly based on the most recent individual body weights. Dose administration was carried out approximately at the same time each day (±3 h).
Parameters evaluated
Clinical observations
All dams and pups were observed once daily (at morning post dose) for clinical signs of toxicity and twice daily (morning prior to dose and evening at post dose) for morbidity and mortality during the dosing phase and recovery period. Detailed clinical examination was done from weaning day (i.e. on PND 21) and at weekly interval thereafter until scheduled necropsy.
Body weights and food consumptions
Individual body weights for all dams were recorded on days 1, 7, 14, and 21of lactation period and for pups on day 1 of littering and twice weekly during dosing and recovery period, until euthanization. Terminal body weights (fasting body weight) were taken on the day of scheduled necropsies, that is, on PND 53 for main group animals and PND 67 for recovery group animals. Feed consumption was recorded twice weekly from day 21 (i.e. from the day of weaning) until euthanization. The cage-wise feed consumption was presented as g/rat/day.
Postnatal developmental landmarks
All pups were observed for the onset of postnatal developmental landmarks like hair growth from PND 5 until bristles appear on the dorsal surface of all pups, incisor eruption from day 7 through the gum of either an upper or lower incisor, ear opening from day 4 for detachment of either the right or left pinna from the side of the head, eye opening from day 10 for any break in the membrane connecting the upper and lower eyelids, balanopreputial separation from day 35 until complete retraction of prepuce from the head of the penis, and vaginal opening from day 28, until complete opening of the vagina.
Ophthalmological examination
Ophthalmological examination was performed toward the end of the dosing period (i.e. on day 44) with an ophthalmoscope (Direct ophthalmoscope, New York City, New York, USA; Welch Allyn) for control and high-dose main group animals. Mydriasis was induced before examination of eyes using a mydriatic agent (1% tropicamide). The low- and mid-dose groups and recovery group animals were not evaluated because there were no GOS-related findings observed in eyes of high-dose group.
Functional observational battery
The neurobehavioral examinations (functional observational battery (FOB)) such as home cage observations, handling observations, open-field observations, sensory observations, neuromuscular observations, and physiological observation (rectal temperature), and motor activity were conducted toward the end of the dosing period (i.e. on days 45 and 46) for all the surviving rats of the main group. FOB was evaluated by Study Director and technicians who were involved in the study from day 1 dosing to until terminal euthanization, thus the neurobehavioral examinations (FOB) were not done by blind observer. The Animal Activity Monitoring System, Opto-Varimex-4 Activity Meter (Columbus Instruments, Columbus, Ohio, USA), was used to monitor the motor activity of rats. Motor activity was evaluated by placing rats individually in motor activity cages for a period of 30 min. The motor activity parameters such as distance traveled, horizontal counts, ambulatory counts, vertical counts, and stereotypes count were evaluated. Order of testing was stratified, for example, first animals of each group followed by second animal and so on to counterbalance groups across the time of day to the maximum extent possible. As there were no treatment-related changes observed in any of the neurobehavioral parameters of the high-dose group (G4), animals of low-dose, mid-dose, and recovery groups were not evaluated further.
Clinical pathology
Terminal blood collection was carried out from all surviving animals of main groups (G1–G4) at the end of dosing period (day 50) and surviving animals of recovery group (G1R and G4R) at the end of recovery period (day 64) for hematology, coagulation, and biochemical evaluations. The animals were fasted overnight (approximately 12–16 h) prior to terminal blood collection. However, water was provided ad libitum to all animals during the period of fasting. Blood samples were collected via retro-orbital plexus puncture under mild isoflurane anesthesia (manufactured by Raman and Weil Pvt. Limited, India, batch number: IF-18007, date of manufacture: July 2018 and date of expiry: September 2022). The hematological, coagulation, and clinical chemistry parameters listed in Table 3 were determined using the ADVIA 2120i fully automated hematology analyzer (Siemens Healthcare GmbH, Henkestr, Erlangen, Germany), fully automated coagulation analyzer, STA Compact (Diagnostica Stago Inc., Parsippany, New Jersey, USA), and Dimension Xpand Plus clinical chemistry system (Siemens Healthcare GmbH, Henkestr, Erlangen, Germany), respectively.
Parameters evaluated in hematology, coagulation, clinical chemistry, and urinalysis.
A/G: albumin/globulin.
Urinalysis
Urine samples were collected individually from all animals at the end of dosing period (day 50 for main groups) and at the end of recovery period (day 64 for recovery groups) in urine collection tubes. For urine collection, animals were fasted overnight (approximately 12–16 h) in metabolic cages. However, water was provided ad libitum to all animals during the period of fasting. Volume, color, and appearance of urine were recorded manually. Specific gravity was estimated using Refractometer (Opti-Digital Handheld Refractometer, Bellingham Stanley, Kent, UK). Urine parameters were analyzed using a Clinitek Advantus analyzer with Multistix SG Reagent Strips (Siemens Healthcare Diagnostics Ltd, Inc. Tarrytown, New York, USA).
Following hematology, coagulation, clinical chemistry, and urine parameters were evaluated in study.
Gross necropsy, organ weights, and histopathology
All surviving pups were euthanized in a stratified manner by exsanguination under deep isoflurane anesthesia at the end of dosing period (day 50) for main groups and at the end of recovery period (day 64) for recovery groups. Pups thath were not selected for study during litter standardization (day 3 of the postpartum) were discarded without further evaluation. The organs (presented in Table 4), as applicable, from males and females were collected, weighed, and preserved. Paired organs were weighed together. The organ weight was recorded as absolute weight and percentage of body weight and brain weight ratios. All organs were preserved in 10% neutral-buffered formalin, except for testes and eyes with optic nerve. Testes were preserved in modified Davidson’s fixative, while eyes with optic nerves were preserved in Davidson’s fixative.
List of organs collected, weighed, preserved, and examined microscopically.
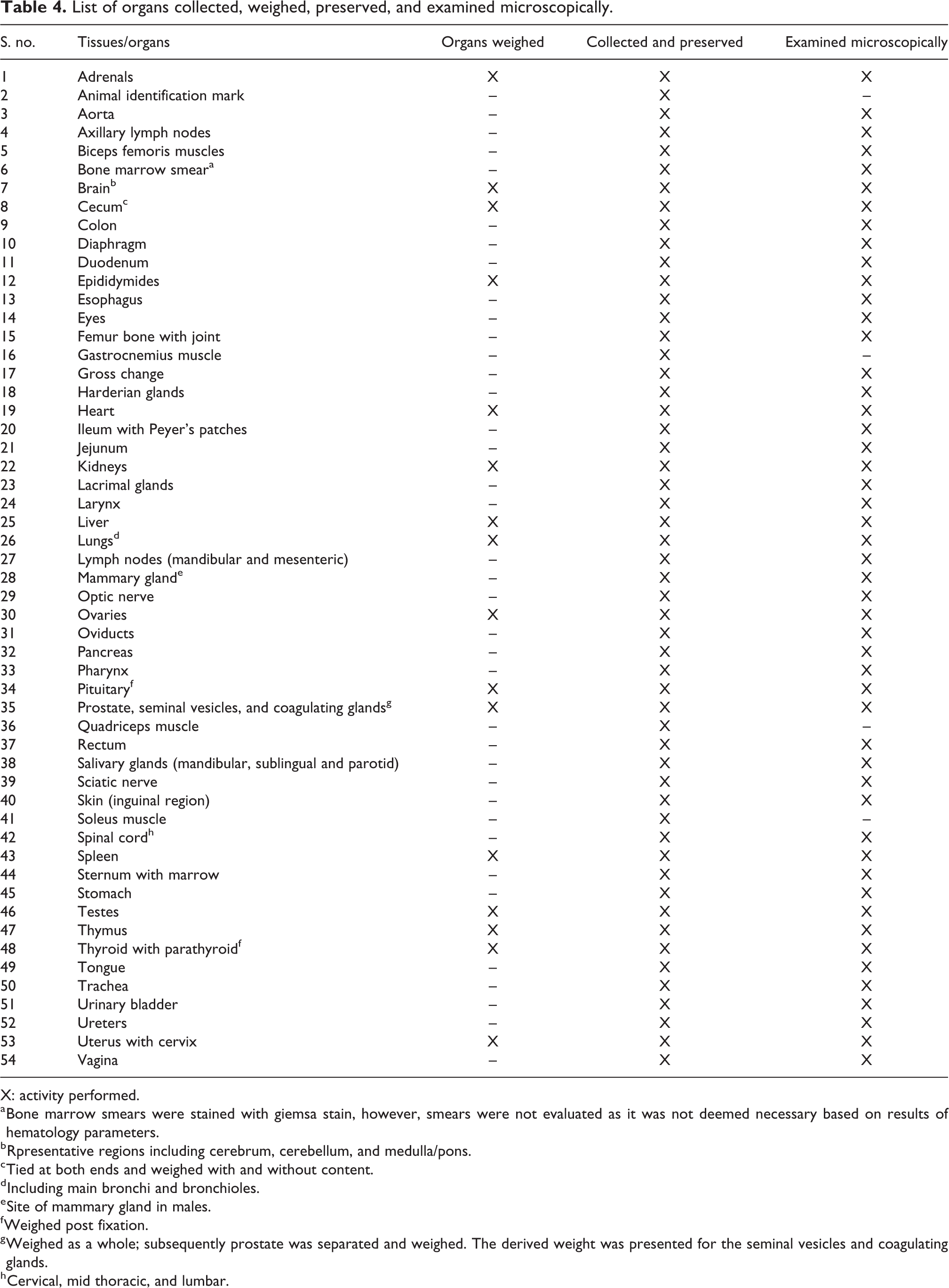
X: activity performed.
a Bone marrow smears were stained with giemsa stain, however, smears were not evaluated as it was not deemed necessary based on results of hematology parameters.
b Rpresentative regions including cerebrum, cerebellum, and medulla/pons.
c Tied at both ends and weighed with and without content.
d Including main bronchi and bronchioles.
e Site of mammary gland in males.
f Weighed post fixation.
g Weighed as a whole; subsequently prostate was separated and weighed. The derived weight was presented for the seminal vesicles and coagulating glands.
h Cervical, mid thoracic, and lumbar.
Histopathology was performed on all preserved organs of vehicle control (G1) and high-dose (G4) groups and for two found dead rats. Gross changes from all rats were also processed and evaluated microscopically. Histopathology was not extended to low-dose (G2) and mid-dose groups (G3), as there were no test item-related changes in high-dose (G4) group. The tissues were processed and embedded in paraffin, sectioned, stained with hematoxylin and eosin, and subjected to histopathological examination. Peer review was performed by an in-house pathologist at Syngene International Limited. The peer review pathologist examined all study plan-specified organs in minimum 30% of animals of each sex of all test article administered groups. The peer review pathologist provided a pathology peer review statement and archived with the study-related material.
Data compilation
The Pristima version 7.2.0 (Xybion Medical Systems) for online data capturing in toxicology studies was used for recording daily observations such as mortality, cage-side clinical signs, detailed clinical signs, body weights, feed input and output, ophthalmological examination, home cage measurements, hand-held measurements, open-field measurements, sensory response measurements, grip strength, hind limb foot splay, rectal temperature, clinical pathology (hematology, coagulation, clinical chemistry, and urinalysis), terminal body weights, organ weights, gross pathology, and histopathology data. All other data/procedures and all postnatal developmental landmark observations were recorded manually and documented in respective standard GLP formats. The Animal Activity Monitoring System, Opto-Varimex-4 Activity Meter (Columbus Instruments) was used to capture the motor activity data of rats.
Statistical analysis
Pristima version 7.2.0 (Xybion Medical Systems) captured data were analyzed using Pristima built-in statistical tests. For comparative statistics, data were evaluated using the Levene test for homogeneity of variances, with significance at 5% level. Data determined to be homogeneous were evaluated for analysis of variance (ANOVA). When ANOVA verified significance at 5% level, pairwise comparisons of each treated group with the control group were calculated using a Dunnett test to identify statistical differences (at 5% level). In cases, wherein, the data were found to be nonhomogeneous, ANOVA was done using suitable transformation. In cases wherein, the homogeneity tests were significant even after transformation, data were evaluated using a Kruskal–Wallis Test for group factor significance. When significance (at 5% level) existed between groups, Dunn’s pairwise comparison was performed. For two group comparisons, data were subjected to Student’s t-test. Statistically significant differences indicated by the aforementioned tests are designated by the superscripts; +/−: significantly higher or lower than the control group/control recovery group.
Results
Stability and dose confirmation analysis
The stability analysis of GOS in vehicle (purified water) was determined in-house at concentrations 100, 200, and 500 mg/mL. Dose formulation analysis performed at 100, 200, and 500 mg/mL concentrations for day 1, and fresh dose formulations at 100, 200, and 300mg/mL concentration prepared separately for day 46 were analyzed for active ingredient of GOS by validated high-performance liquid chromatography technique in vehicle. Results of days 1 and 46 analyses were found to be within the acceptance limits of the nominal concentration (% recovery: 90–110%) and the percent relative standard deviation was found to be less than 10% of GOS, as represented in Table 5. The vehicle control samples did not show any test item concentration, indicating no contamination during study period.
Dose formulation analysis results.a
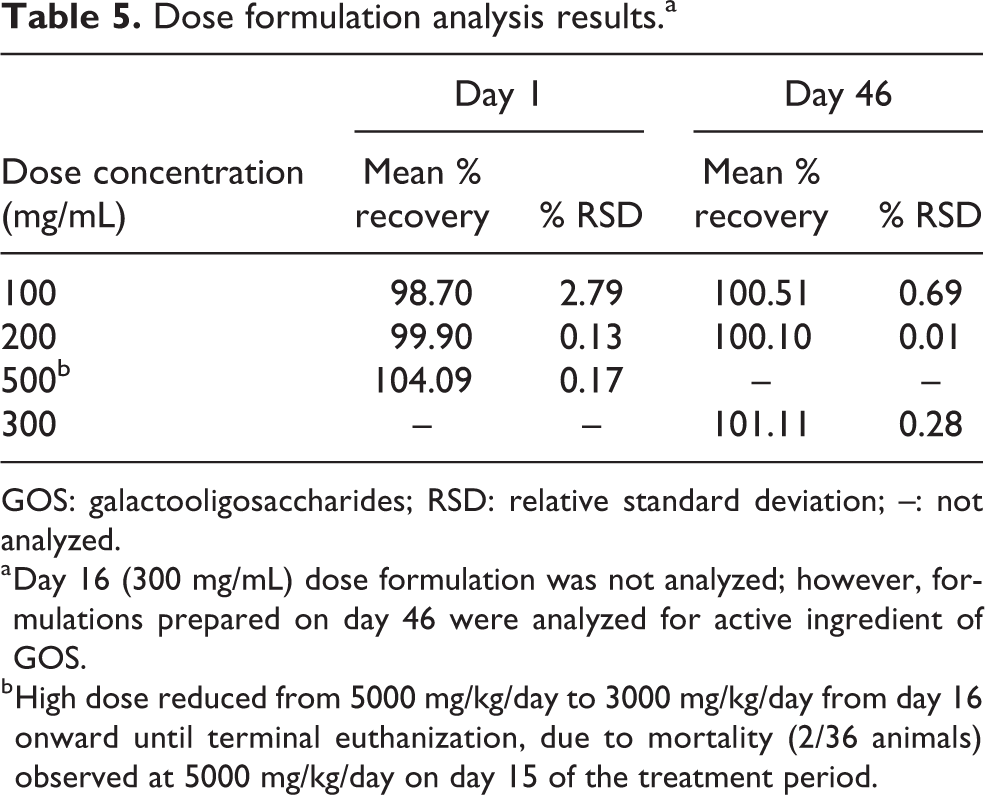
GOS: galactooligosaccharides; RSD: relative standard deviation; –: not analyzed.
a Day 16 (300 mg/mL) dose formulation was not analyzed; however, formulations prepared on day 46 were analyzed for active ingredient of GOS.
b High dose reduced from 5000 mg/kg/day to 3000 mg/kg/day from day 16 onward until terminal euthanization, due to mortality (2/36 animals) observed at 5000 mg/kg/day on day 15 of the treatment period.
Mortality, clinical signs, and ophthalmological examination
At 1000 and 2000 mg/kg/day, there were no clinical signs of toxicity or mortality observed till terminal euthanization. At 5000 mg/kg/day, clinical signs of watery feces and yellow color stains at urogenital region were observed in 13 of 34 animals (9 males and 4 females) from days 15 to 17 (PND 18–20) of the dosing phase. Two deaths occurred (1 male and 1 female) on day 15 of the dosing phase (PND 18). Hence, for the high-dose group, the dose was reduced from 5000 mg/kg/day to 3000 mg/kg/day from day 16 of the dosing phase. No further deaths or clinical signs were noticed in animals at 3000 mg/kg/day from day 18 of dosing phase to until terminal euthanization. Gross observation at necropsy in two dead animals was characterized by the presence of yellowish liquid contents in the intestine (both in small and in large intestine). Exact cause of death could not be determined based on gross and histomorphological evaluations. There were no test item-related changes noticed in ophthalmoscopy in any dose group.
Body weights
Statistically significant decrease in mean body weights at 5000 mg/kg/day on day 4 (−14% to −16%) and day 7 (−12%) and a significant reduction in mean body weight gain on days 1–4 (−34% to −35%) was noticed in both male and female pups of high-dose recovery group during treatment period, when compared to concurrent vehicle control group. A nonsignificant decrease in mean body weights was observed on day 11 (−6% to −7%) and day 14 (−6% to −8%) of the treatment period, when compared to concurrent vehicle control group.
However, after the reduction of high dose from 5000 mg/kg/day to 3000 mg/kg/day from day 16 onward, no further decrease in body weight or body weight gain was noticed from day 18 of dosing phase to until terminal sacrifice.
There were no test item-related changes in mean body weight, and body weight gain was observed in both sexes at 1000 and 2000 mg/kg/day when compared to concurrent vehicle control group till terminal euthanization (Figures 1 and 2).
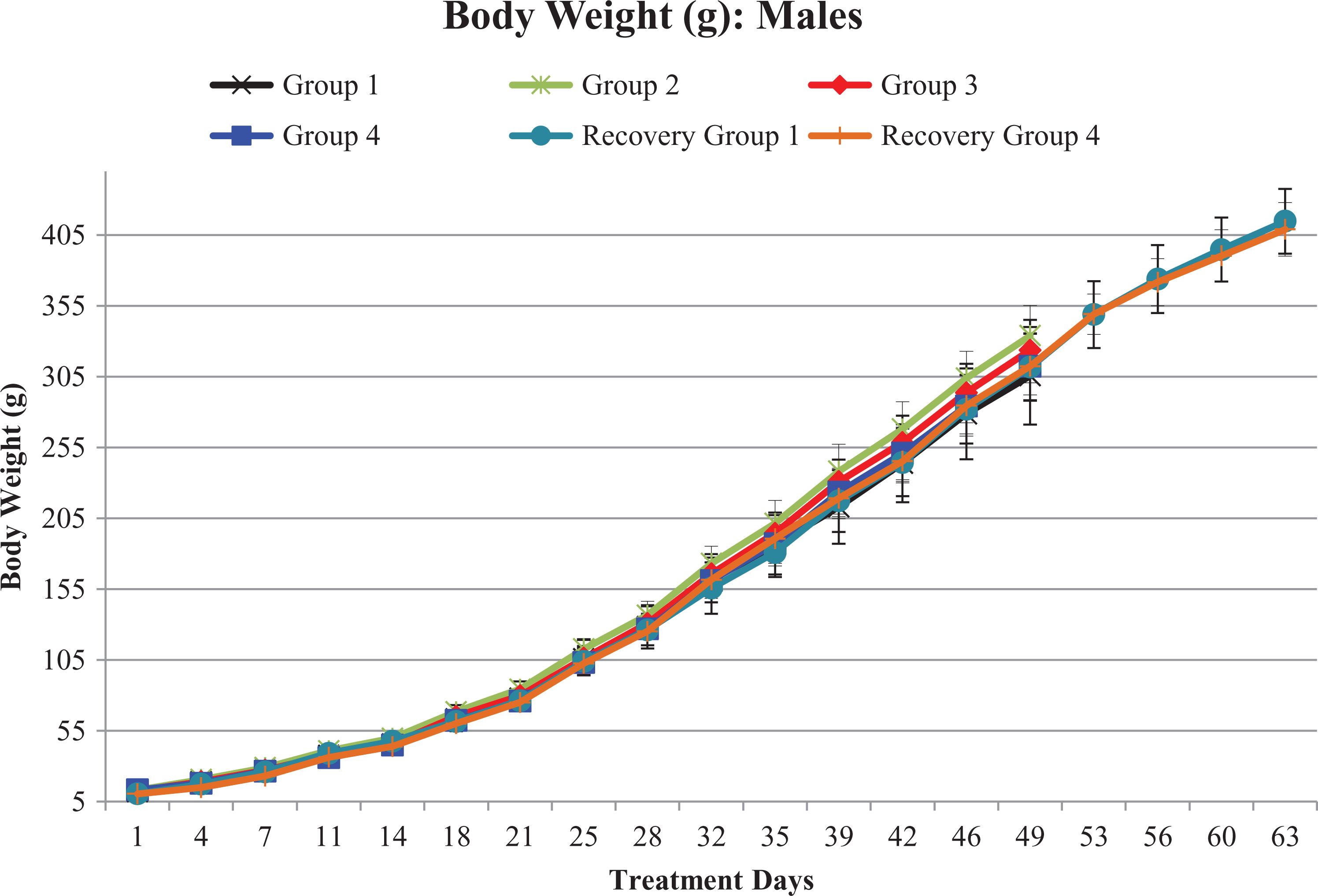
Growth curves of males in terms of mean body weight (g): G1/G1R, G2, G3, and G4/G4R represent 0, 1000, 2000, and 5000/3000 mg of Gossence™/kg/day, respectively. From days 50 to 63, treatment-free recovery period.
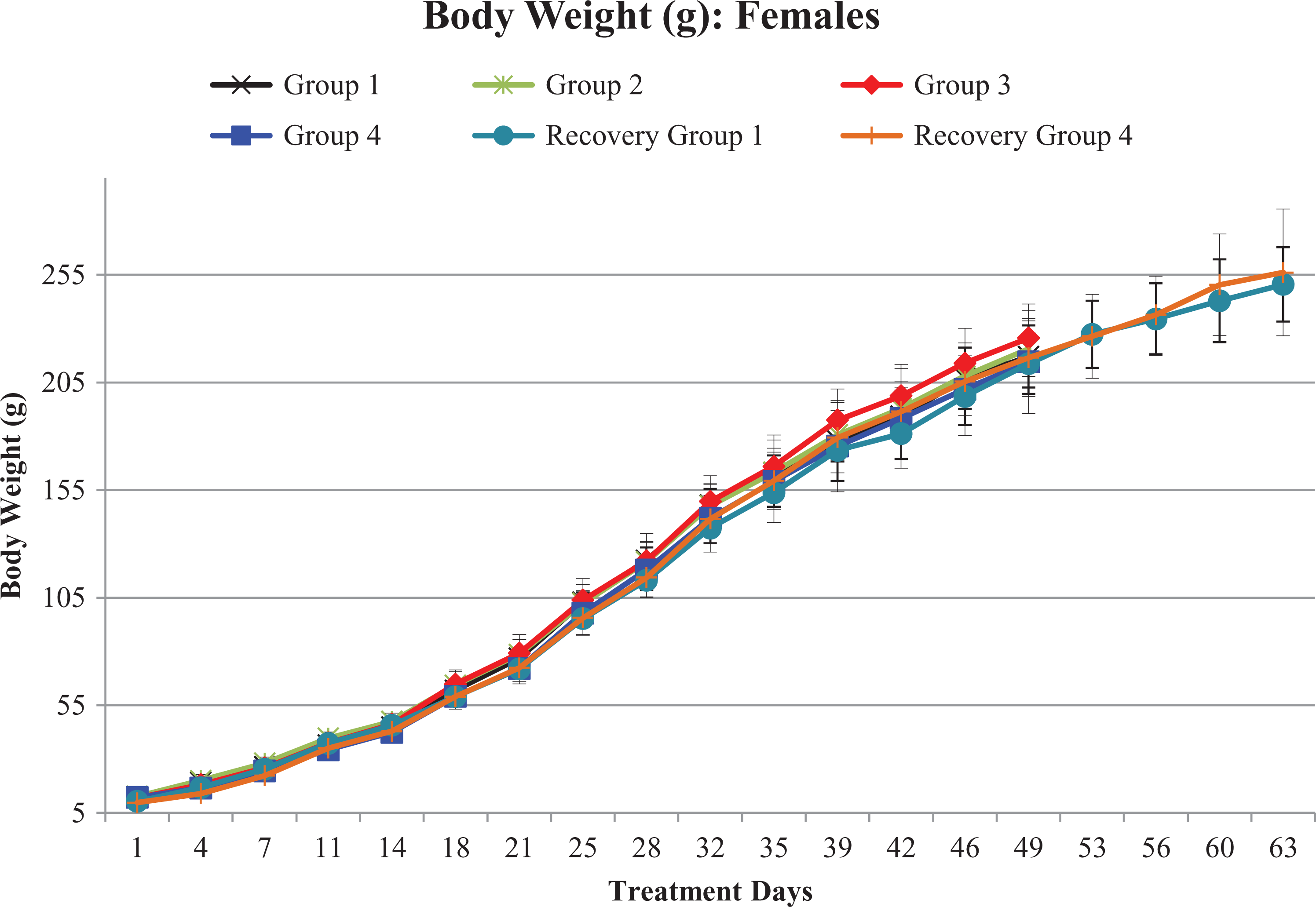
Growth curves of females in terms of mean body weight (g): G1/G1R, G2, G3, and G4/G4R represent 0, 1000, 2000, and 5000/3000 mg of Gossence™/kg/day, respectively. From days 50 to 63, treatment-free recovery period.
Food consumption
There were no test item-related changes in mean food consumption observed in both sexes up to 5000/3000 mg/kg/day and was comparable to concurrent vehicle control group till terminal sacrifice (Figures 3 and 4).
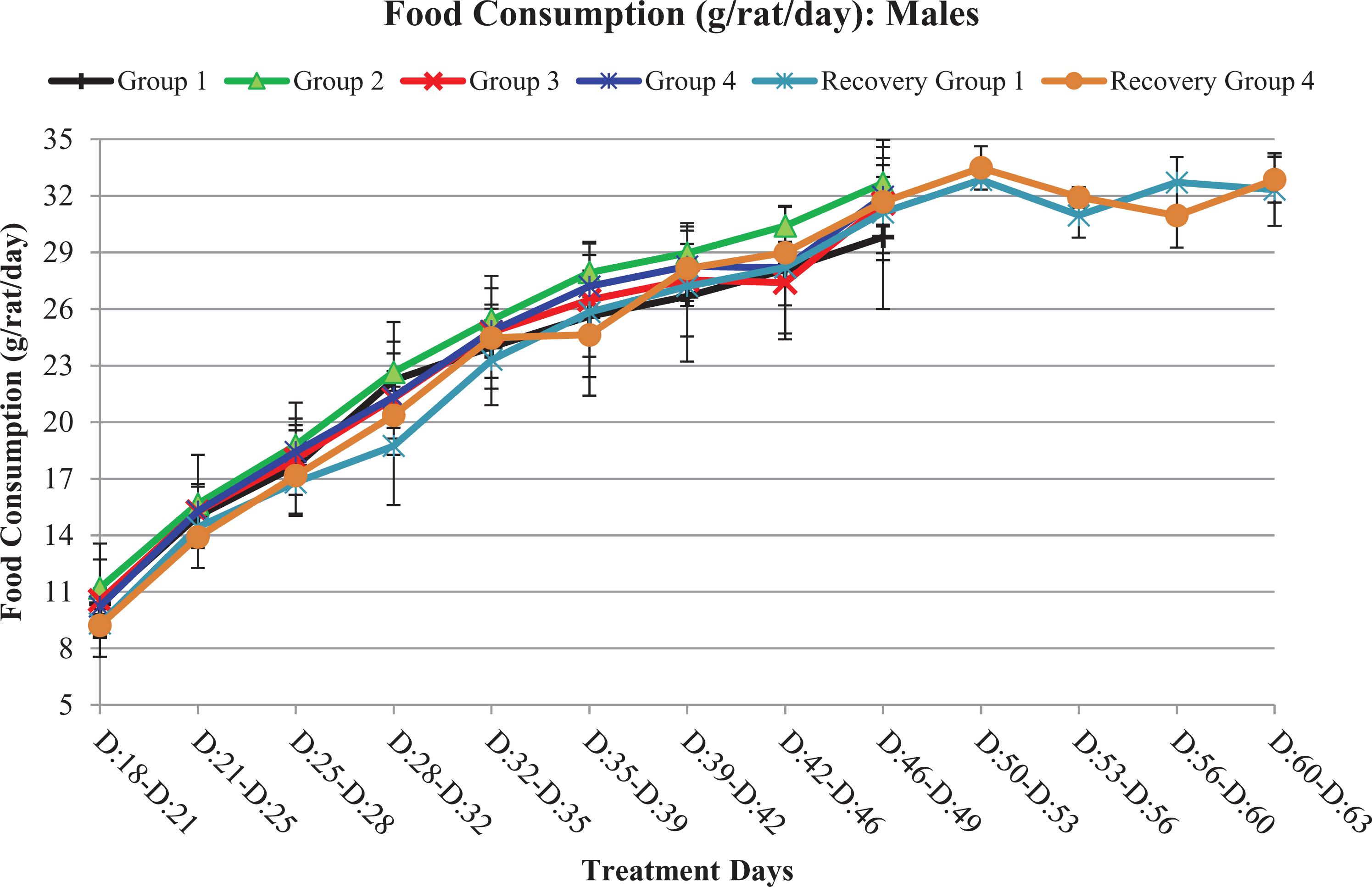
Food consumption curves of males in terms of mean food consumption: G1/G1R, G2, G3, and G4/G4R represent 0, 1000, 2000, and 5000/3000 mg of Gossence™/kg/day, respectively. From days 50 to 63, treatment-free recovery period.
Food consumption curves of females in terms of mean food consumption: G1/G1R, G2, G3, and G4/G4R represent 0, 1000, 2000, and 5000/3000 mg of Gossence™/kg/day, respectively. From days 50 to 63, treatment-free recovery period.
Postnatal developmental landmarks
There were no GOS-related changes in either, on the day of acquisition of different developmental landmarks (hair growth, incisor eruption, ear opening, eye opening, balanopreputial separation, and vaginal opening) during postnatal growth or in the percentage of pups acquiring these parameters on a particular day when compared to the vehicle control group. Also, there was no treatment-related effect observed in the mean body weight on the day of acquisition of balanopreputial separation in males and vaginal opening in females when compared with the control counterparts.
FOB test and motor activity
There were no GOS-related changes observed in FOB (home cage, handling, open field, and sensory observations), neuromuscular observations (grip strength and landing foot splay), and motor activity parameters when compared with the vehicle control group.
Clinical pathology and urinalysis
There were no test item-related changes in hematology, coagulation, clinical chemistry, and urine analysis parameters in the study.
Necropsy, organ weights, and histopathology
Grossly, increased size of cecum was observed at 5000/3000 mg/kg/day (6 of 12 males and 2 of 12 females) and 2000 mg/kg/day (2 of 12 males and 1 of 12 females) in terminal euthanized animals, which was correlated with increased weights of the cecum (with contents) during organ weight recording but had no correlating light microscopic changes. The cecal enlargement completely recovered following the 14-day recovery period. The cecal enlargement was considered a compensatory and adaptive response to the administration of GOS. 1 The weights of the cecum without luminal contents were not affected, indicating that increased weights were primarily due to the luminal contents. There were no other test item-related changes noticed in any other organs collected and weighed, and no gross changes and histological findings were observed in any of the tissues examined in the study. The changes in mean cecum weights with content are summarized in Table 6.
Gossence™ (GOS)-related mean changes in cecal weights.a
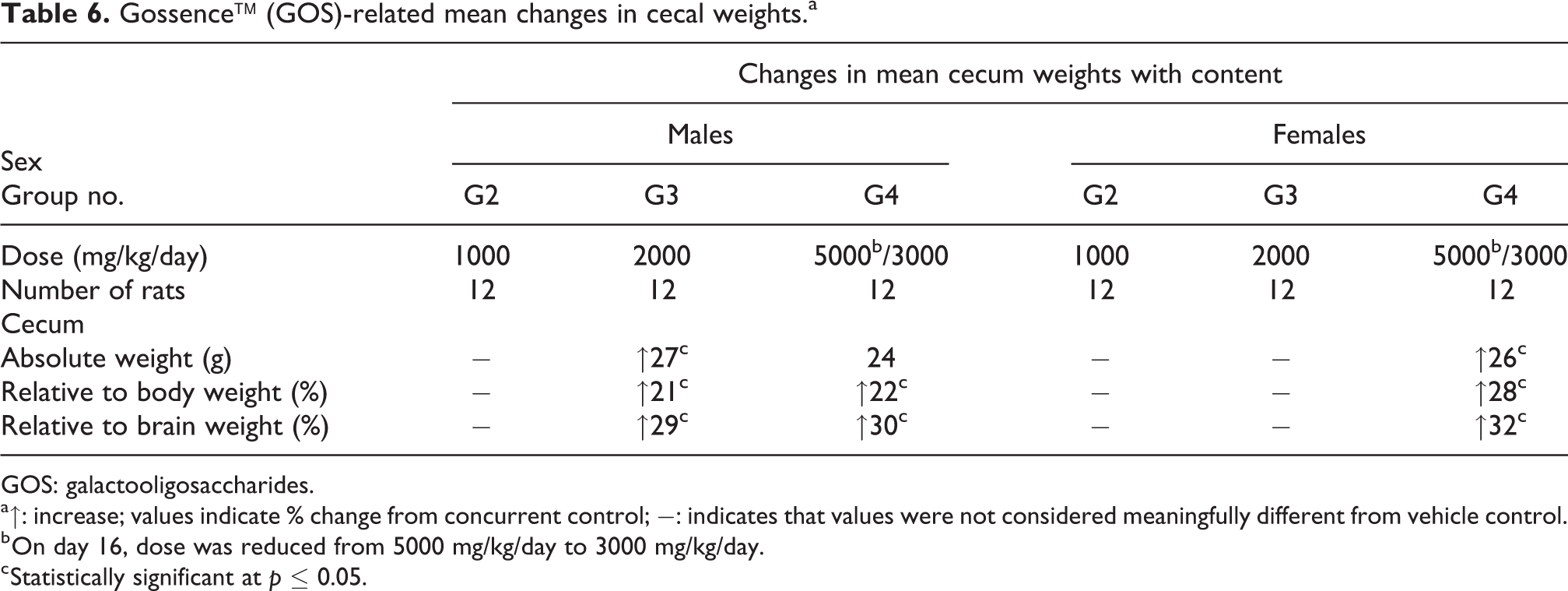
GOS: galactooligosaccharides.
a↑: increase; values indicate % change from concurrent control; −: indicates that values were not considered meaningfully different from vehicle control.
b On day 16, dose was reduced from 5000 mg/kg/day to 3000 mg/kg/day.
c Statistically significant at p ≤ 0.05.
Discussion
Juvenile toxicity study was conducted to evaluate potential systemic toxicity of Gossence (GOS) when administered daily via gavage to juvenile Sprague Dawley rats from PND 4 at dose levels of 1000, 2000, or 5000/3000 mg/kg/day for a period of 7 consecutive weeks, followed by 14-day recovery period. Dosing was initiated on PND 4 of the pups as it is the earliest possible initiation time for gavage administration in juvenile rats based on practical feasibility and to mimic the earliest age of the intended pediatric population.
At 1000 and 2000 mg/kg/day, there were no clinical signs of toxicity or mortality; at 5000 mg/kg/day, clinical signs of watery feces and yellow stains at urogenital region were observed in 13 of 34 animals from days 15 to 17 (PND 18–20) of the dosing phase. Two deaths occurred on day 15 of the dosing phase (PND 18). The exact cause of death could not be determined based on gross and histomorphological evaluation. Thus, the dose for the high-dose group was reduced from 5000 mg/kg/day to 3000 mg/kg/day on day 16 of the dosing phase. No further deaths or clinical signs occurred after reducing the dose.
Decreased body weight and body weight gain were noticed in pups during days 4–14 of the dosing phase at 5000 mg/kg/day, upon reduction of dose to 3000 mg/kg/day from day 16 onward, body weight profile returned to normal and comparable to concurrent the vehicle control until euthanization. There were no test item-related changes in ophthalmology and food consumption at any dose levels. FOB revealed no test item-related effects on home cage, handling, open-field, sensory, neuromuscular, and physiological observations up to the highest dose of 5000 mg/kg/day, which was consistent with the published literature on juvenile rat toxicity studies conducted on similar class of test compounds with different chemical compositions and method of synthesis. 18,23 There were no test item-related effects on the onset of postnatal developmental landmarks (hair growth, incisor eruption, ear opening, eye opening, balanopreputial separation, and vaginal opening) at any administered dose levels. 18,23 Increased size of the cecum at 5000/3000 mg/kg/day (male 6/12, female 2/12) and 2000 mg/kg/day (male 2/12, female 1/12) was the only gross observation, which correlated with the increased weights of ceca only with contents but had no correlating light microscopic changes, suggesting that increased weights of cecum was only due to increased cecal contents. The cecal enlargement completely reversed following 14-day recovery period, indicating reversibility of the change. These changes were considered as non-adverse, compensatory, or adaptive response to the administration of GOS. 1 The cecum enlargement is consistent with other reports for the same class of compounds; various studies had been reported that consumption of pectin and nondigestible oligosaccharides can cause histological changes in cecum in rats. 2,24 The cecum is an area of bacterial fermentation and growth. Cecal hypertrophy can occur due to trophic effects of short fatty acids which are produced during bacterial fermentation after an excessive amount of nonabsorbed carbohydrate and dietary fiber enter the cecum and colon. 17
However, the current study shows novelty from the previously published literature 18,23 in the following aspects: (1) test item in the present study is different in chemical composition and in method of synthesis; (2) high dose selected for the current study is 5000 mg/kg/day, which was reduced to 3000 mg/kg/day due to mortality; (3) a reversible, non-adverse, compensatory, or adaptive response of cecal enlargement was noticed in the present study at 2000 and 5000/3000 mg/kg/day; and (4) no-observed-adverse-effect level (NOAEL) is considered as 3000 mg/kg/day for Gossence, which is equivalent to 4041 mg/kg/day of GOS.
Observation for additional end points such as skeletal examinations, assessment for central nervous system effects (learning and memory test, evaluation for neuropathology), evaluation of reproductive performance (i.e. estrous cycle, sperm count, motility, morphology), and assessment for functional immunity was not calculated for the study, as it was not warranted based on the available literature on the GOS. GOS is considered to be Generally Recognized as Safe in humans. 25
Based on the review of results and weight of evidence, it was concluded that Gossence (GOS) has no effect on the growth and development of juvenile rats of ages day 4 to 7 weeks post birth.
Further available literature on one-generation reproduction toxicity study with novel GOS in adult rats through gavage showed no changes in any reproductive parameters, including growth and development of pups up to 2000 mg/kg/day in both the sexes. 23
Similarly, in-house 90-day repeat dose toxicity study conducted for Gossence in adult rat reveals that Gossence (GOS) was found to be well tolerated at 5000 mg/kg/day and did not cause any mortality, or clinical signs, and changes in body weights, feed consumption, clinical pathology, and histopathology. NOAEL was found to be 5000 mg/kg/day for Gossence, which is equivalent to 6735 mg/kg/day of GOS. 22
The study results of Gossence in juvenile rats will be useful in providing safety information necessary to design and to monitor the necessary parameters in clinical trials of pediatric population.
Conclusion
In conclusion, repeated dose gavage administration of Gossence (GOS) from PNDs 4 to 53 at 1000 or 2000 mg/kg/day (equivalent to 1347 and 2694 mg/kg/day of GOS, respectively) did not result in any adverse findings in male and female juvenile Sprague Dawley rats. The high dose (5000 mg/kg/day) of Gossence resulted in two mortalities. Thus, 5000 mg/kg/day dose of Gossence exceeded the maximum tolerated dose in juvenile rats. However, upon reducing the dose to 3000 mg/kg/day, the dose was well tolerated with no adverse changes. Therefore, NOAEL is considered as 3000 mg/kg/day for Gossence, which is equivalent to 4041 mg/kg/day of GOS.
Estimated dietary consumption of GOS from intended food use is 8 g of GOS/L in the reconstituted or ready-to-drink product per GRAS Exemption Claim for Galacto-Oligosaccharides (USFDA). 25
Voluntary addition GOS, alone or in combination with fructo-oligosaccharides, to infant foods and formulated supplementary foods for young children was in the range of 0.12–0.8 g/100 mL (1.6 g/serve) or 0.8 g/100 g, 26 respectively. In the current study, the arrived NOAEL is 3000 mg/kg/day for Gossence, which corresponds to approximately 1000 mg/kg/day of human equivalent dose (HED) for an infant with an average weight of 4 kg body weight, per conversion of animal dose to HED based on body surface area 27 [HED = animal dose in mg/kg × (animal weight in kg/human weight in kg) × 0.33]. Thus, the current NOAEL (3000 mg/kg/day of Gossence™) is three times more than the HED.
Supplemental Material
Supplemental Material, Additional_Supplementary_Data - Juvenile toxicity study of Gossence™ (galactooligosaccharides) in Sprague Dawley rats
Supplemental Material, Additional_Supplementary_Data for Juvenile toxicity study of Gossence™ (galactooligosaccharides) in Sprague Dawley rats by Manish Jain, Moninder Kaur, Deepika Pandey Tiwari, Chandrashekara Vishwanath, Nataraju Javaregowda, Govind Chandrayan, Prabhakar Y Bhoite, Mohan Krishnappa and Ashok Kumar Dubey in Toxicology Research and Application
Supplemental Material
Supplemental Material, Histology_of_Cecum_Images - Juvenile toxicity study of Gossence™ (galactooligosaccharides) in Sprague Dawley rats
Supplemental Material, Histology_of_Cecum_Images for Juvenile toxicity study of Gossence™ (galactooligosaccharides) in Sprague Dawley rats by Manish Jain, Moninder Kaur, Deepika Pandey Tiwari, Chandrashekara Vishwanath, Nataraju Javaregowda, Govind Chandrayan, Prabhakar Y Bhoite, Mohan Krishnappa and Ashok Kumar Dubey in Toxicology Research and Application
Supplemental Material
Supplementary_files - Juvenile toxicity study of Gossence™ (galactooligosaccharides) in Sprague Dawley rats
Supplementary_files for Juvenile toxicity study of Gossence™ (galactooligosaccharides) in Sprague Dawley rats by Manish Jain, Moninder Kaur, Deepika Pandey Tiwari, Chandrashekara Vishwanath, Nataraju Javaregowda, Govind Chandrayan, Prabhakar Y Bhoite, Mohan Krishnappa and Ashok Kumar Dubey in Toxicology Research and Application
Footnotes
Authors’ note
MJ, AD, DP, and CV contributed to the conception or design of the work. MJ and MK were involved in the analysis of GOS. CV, NJ, and GC contributed to the data collection. CV, NJ, GC, PB, and MKr contributed to the data analysis and interpretation. CV, NJ, and GC drafted the article. All authors critically reviewed and approved the manuscript.
Acknowledgements
The authors are thankful to the management of Tata Chemicals Limited and Syngene International Limited.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are (or were) employees of Tata Chemicals or Syngene International.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported solely by Tata Chemicals Limited with mutual agreement with Syngene International.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
