Abstract
Introduction
Fatalities from metal fragment wounds are an unfortunate result of armed conflicts. However, technological improvements in armor protection and medical advancements in survival surgery have significantly improved the odds of surviving ballistic injuries. 1 As a result, numerous individuals now live with retained metal fragments in their bodies as a result of their military service. Under the assumption that most embedded metal fragments would be inert, standard surgical guidelines proposed leaving the metal fragment in place except under certain conditions. 2 However, that assumption is not always correct. Although rare, there have been case studies reported in the peer-reviewed scientific literature describing adverse health effects resulting from embedded metal fragments.3–6 Most of these adverse effects were manifested many years after the initial injury.7–9 Another unpredictable consequence of embedded metal fragments is that many metals tend to degrade over time, releasing solubilized metal ions into the body.10–14 This property is not unique to embedded military-relevant metal fragments but has also been found to occur with implanted medical and dental devices as well as from industrial accidents.15–21 For instance, degradation of metal-on-metal hip replacements have resulted in the release of cobalt ions from the cobalt/chromium alloy utilized. 15 Solubilized aluminum from titanium/aluminum/vanadium used in joint prostheses has also been found in urine and serum as has titanium from dental implants.16,17 In some cases, these solubilized metals are excreted in the urine while in other cases, although urine excretion is observed, the metals will also sequester in “target organs” in the body.22,23 This raises the specter of potentially toxic metals localized in sensitive tissues for extended periods of time. Unfortunately, there is a lack of knowledge with respect to the toxicological and biokinetic properties of embedded metal fragments suffered in warfare. To that end, the U.S. Departments of Defense (DoD) and Veterans’ Affairs (DVA) have developed a list of “metals of concern” that require further investigation.24,25 This project is part of a larger collaborative effort with the DVA, the University of Maryland School of Medicine, and the University of Kentucky to study the potential health effects of embedded metals from the DoD and DVA list of “metals of concern.” The overall program is a two-pronged approach using a rodent model developed in our Institute to study embedded metal fragments with a parallel human investigation with military personnel suffering from metal fragment injuries. We have previously reported that metals implanted into rodents for up to 12 months eventually partially solubilize and are found in the urine as well as discrete regions of the brain.26,27 In this study, we have investigated the metal distribution patterns in tissues collected from our metal-implanted Sprague-Dawley rat cohort.
Methods
Animals and animal husbandry
All animal research associated was approved prior to initiation by the Armed Forces Radiobiology Research Institute (AFRRI) Institutional Animal Care and Use Committee (IACUC) under protocol number 2016-05-006. All procedures were conducted in compliance with the Guide for the Care and Use of Laboratory Animals 28 in an Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)-accredited facility. Male Sprague-Dawley (Rattus norvegicus) rats, approximately 30 days old and weighing 75–100 g, were obtained from Envigo (Barrier 208A, Frederick, MD, USA). After arriving, animals were allowed to acclimate for at least two weeks. Rats were pair-housed throughout the study in plastic microisolator cages (23.8 × 45.4 cm) with filter tops. Teklab Sani-Chips (Envigo) were used as bedding and bedding was changed 2–3 times per week. Vivarium rooms were maintained at 21 ± 2°C with 30–70% humidity. A 12:12-h light:dark cycle was maintained with lights on at 0600. Rats were fed a standard rodent chow (Teklad Global Rodent Diet 8604, Envigo) with water available ad libitum.
Experimental design
Previous research in our Institute developed a rodent model for studying the health effects of embedded metal fragments, such as those suffered in a shrapnel wound. 29 We used that model system in this study to investigate the effects of eight military-relevant metals, selected from the DoD and DVA list of “metals of concern,” including tungsten (W), nickel (Ni), cobalt (Co), iron (Fe), copper (Cu), aluminum (Al), lead (Pb), and depleted uranium (DU). Tantalum (Ta) was used as a control for any changes resulting from the surgical procedure or due to the presence of a foreign material in the muscle. Tantalum is considered inert and has been used for implanted prosthetic devices.30–32 Previous studies have shown no differences between naïve and tantalum-implanted rats with respect to body weight gain, mobility, localized metal toxicity, and neoplastic changes in the gastrocnemius tissue surrounding the implanted tantalum pellets.10,29,33,34 Therefore, the total number of rats needed for the study was reduced and the ARRIVE Guidelines met. 35 Animals were randomly assigned to one of the nine metal implantation groups with an n = 8 per metal. The metal groups were conducted in four different cohorts: 1-, 3-, 6-, or 12-months post-implantation surgery (i.e., “Time from Implant”). A total of 288 rats (n = 8 × 9 metal groups × 4 cohorts) were used in the study with the number of animals required per group calculated from the power analysis included with the IACUC protocol to ensure statistical relevance.
Metal pellets (1 mm diameter × 2 mm length) were obtained from Alfa Aesar (Ward Hill, MA, USA) with the exception of DU which was purchased from Aerojet Ordnance (Jonesboro, TN, USA). Prior to implantation, pellets were cleaned and chemically sterilized as previously described. 36
Metal pellet implantation
Metal pellets were surgically implanted bilaterally in the gastrocnemius muscle of rats as previously described.26,27 Briefly, animals were initially anesthetized in an induction chamber using isoflurane (Baxter Healthcare, Deerfield, IL, USA) and then maintained throughout the surgical period using a nose cone with a scavenger/recapture system. The surgical sites were clipped, swabbed with 70% 2-propanol, and finally cleansed with betadine (Purdue Pharma LP, Stamford, CT, USA). Prior to surgery, a prophylactic dose of an analgesic (buprenorphine, 0.05–0.1 mg/kg, s.c., Rickitt and Colman, Hull UK) was administered. Two identification methods were used. A small transponder (Electronic Lab Animal Monitoring System, Bio-Medic Data Systems, Seaford, DE, USA) was injected subcutaneously in the mid-dorsal thoracic region. Each transponder was programmed with a unique animal identification number that can be read with a low-power radio frequency scanner. An ear punch system was used as the second backup identification system in case of transponder failure. Using aseptic technique, a small incision approximately 5 mm in length was made through the skin of each hind leg to expose the gastrocnemius muscle. Each gastrocnemius muscle was implanted with two sterile pellets spaced approximately 1.5 mm apart on the lateral side of the muscle by placing the sterile pellet in a 14–16 gauge needle. Using a specially designed plunger placed inside the needle, the needle was inserted into the gastrocnemius and the plunger depressed forcing the pellet into the muscle. Tissue adhesive (VetBond; 3M Corporation, St. Paul, MN, USA) was used to seal the incision. After surgery, rats were closely monitored until ambulatory. The surgery sites were examined daily for two weeks to assess for signs of inflammation, infection, and local metal toxicity, and after that time, weekly, for the duration of the study.
Tissue collection
Upon reaching their experimental endpoint or when indicated by guidelines approved by the AFRRI IACUC, rats were humanely euthanized under deep isoflurane by exsanguination and confirmatory pneumothorax as per the guidelines of the American Veterinary Medical Association. 37 After euthanasia, a complete gross pathology examination was conducted. A variety of tissues were collected for further examination as has been described in earlier publications.24,25
Sample preparation and metal analysis
Prior to metal analysis, tissue samples were first dried in a muffle furnace (Fisher Scientific, Pittsburgh, PA USA) at 100°C for 24 h. The temperature of the furnace was then ramped to 350°C, at 5°C/min, and the samples held there for 24 h. The furnace temperature was then ramped to 600°C, at 5°C/min, and the samples held there for 48 h. After cooling, the samples were wet-ashed with 5 ml of 70% nitric acid (Optima Ultrapure Grade, Fisher Scientific) and 200 μl of 30% hydrogen peroxide (Semiconductor Grade, Sigma-Aldrich Chemical Co., St. Louis, MO USA) then heated to just below boiling until completely evaporated. The samples were then dry-ashed for 12 h at 600°C in a muffle furnace prior to another cycle of wet-ashing with nitric acid and hydrogen peroxide. After the second run of wet-ashing, the resulting white residue was dissolved in 2% nitric acid and analyzed.
All reagents used were of the highest grade available. Plasticware and other disposables were obtained from ThermoFisher (Pittsburgh, PA, USA). Metal content determined using an inductively coupled plasma-mass spectrometer (Thermo Fisher iCAP RQ/Q with Quantara software, Madison, WI, USA) equipped with a Cetac ASX-560 Autosampler (Cetac Technologies, Omaha, NE, USA). High-pressure liquid argon, 99.997%, was used for the plasma gas. Helium, 99.999%, was used as the CCT gas. Calibration was accomplished with external standards of the appropriate metal standard (SPEX CertiPrep, Metuchen, NJ, USA) in 2% nitric acid. The sample probe was washed with a constant flow of 2% nitric acid to prevent carryover. Quantitative analysis was obtained by reference to the slope of the calibration curve (counts per second/ng per liter) as well as an internal standard. Limit of Detection (LoD)/Limit of Quantitation (LoQ), in ppb, are as follows: Ta—0.012/0.015; W—0.002/0.010; Ni—0.016/0.042; Co—0.001/0.002; Fe—0.541/1.226; Cu 0.026/0.106; Al—0.629/1.716; Pb—0.001/0.009; U—0.001/0.003.
Statistical analysis
Metal concentrations were analyzed by two-way ANOVA using the variables of implanted metal and time from implant followed by Sidak’s multiple comparisons test where each metal implant group within a time from implant is compared back to the corresponding Ta-implanted group value. Analyses were performed using GraphPad Prism Software (version 9.1.2, La Jolla, CA, USA). In all cases, p values<.05 were considered significant.
Results
Kidney metal levels in 1-, 3-, 6-, and 12-month experimental groups.
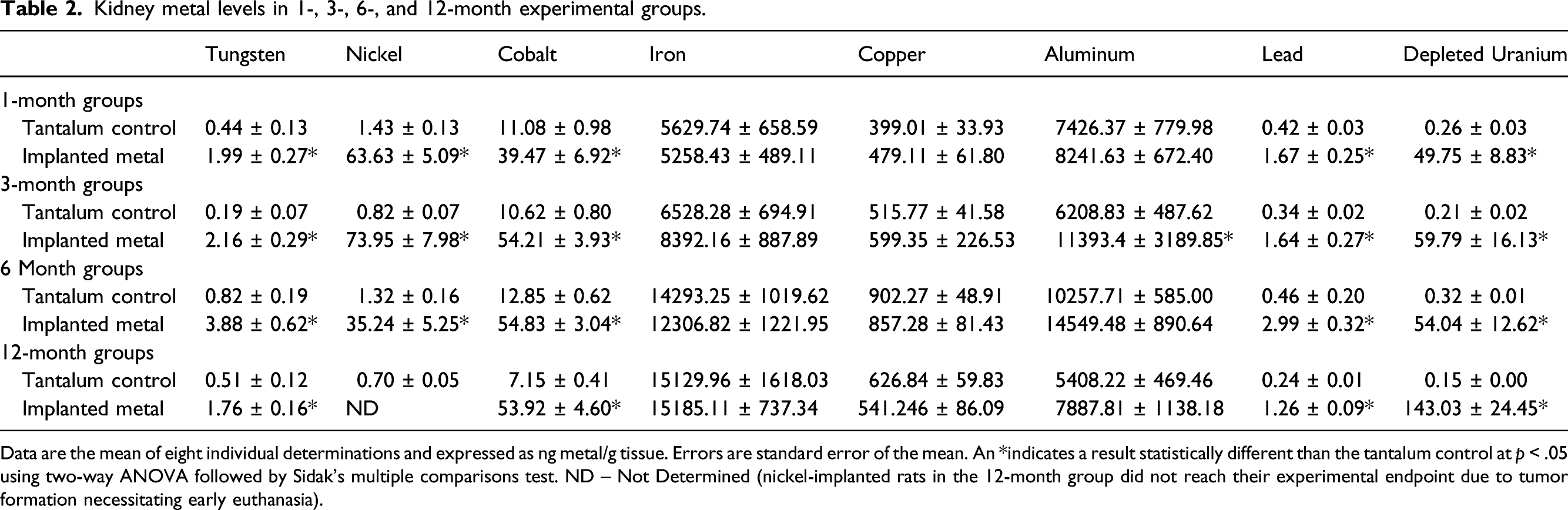
Data are the mean of eight individual determinations and expressed as ng metal/g tissue. Errors are standard error of the mean. An *indicates a result statistically different than the tantalum control at p < .05 using two-way ANOVA followed by Sidak’s multiple comparisons test. ND – Not Determined (nickel-implanted rats in the 12-month group did not reach their experimental endpoint due to tumor formation necessitating early euthanasia).
Liver metal levels in 1-, 3-, 6-, and 12-month experimental groups.
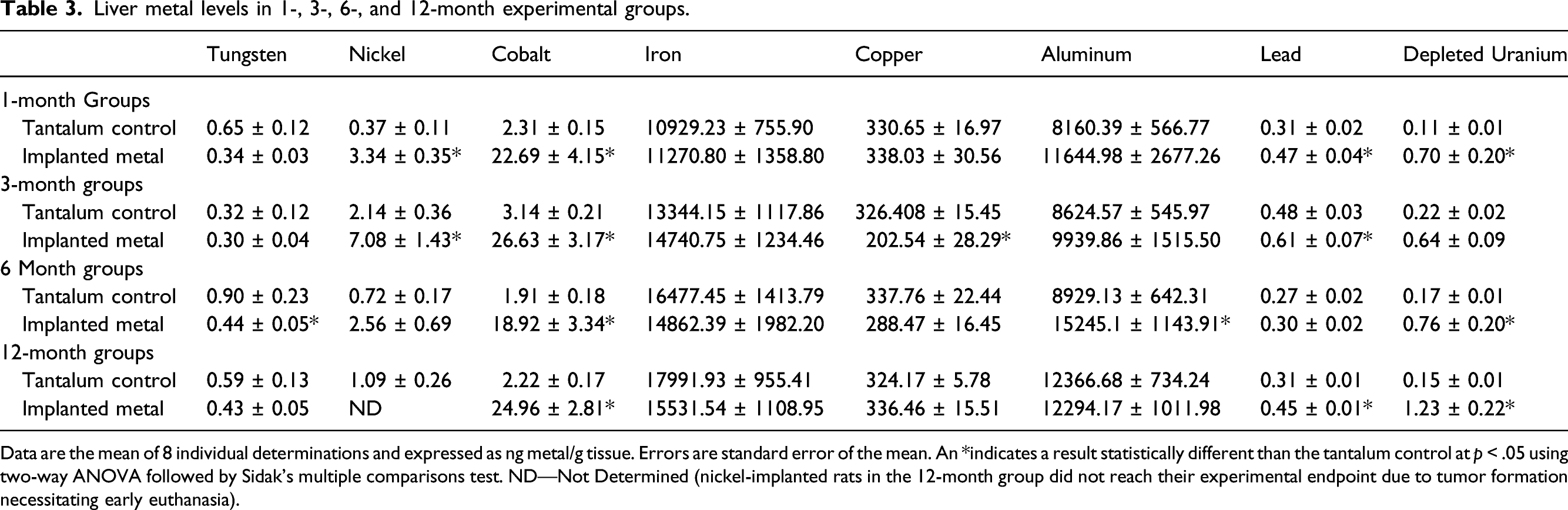
Data are the mean of 8 individual determinations and expressed as ng metal/g tissue. Errors are standard error of the mean. An *indicates a result statistically different than the tantalum control at p < .05 using two-way ANOVA followed by Sidak’s multiple comparisons test. ND—Not Determined (nickel-implanted rats in the 12-month group did not reach their experimental endpoint due to tumor formation necessitating early euthanasia).
Spleen metal levels in 1-, 3-, 6-, and 12-month experimental groups.
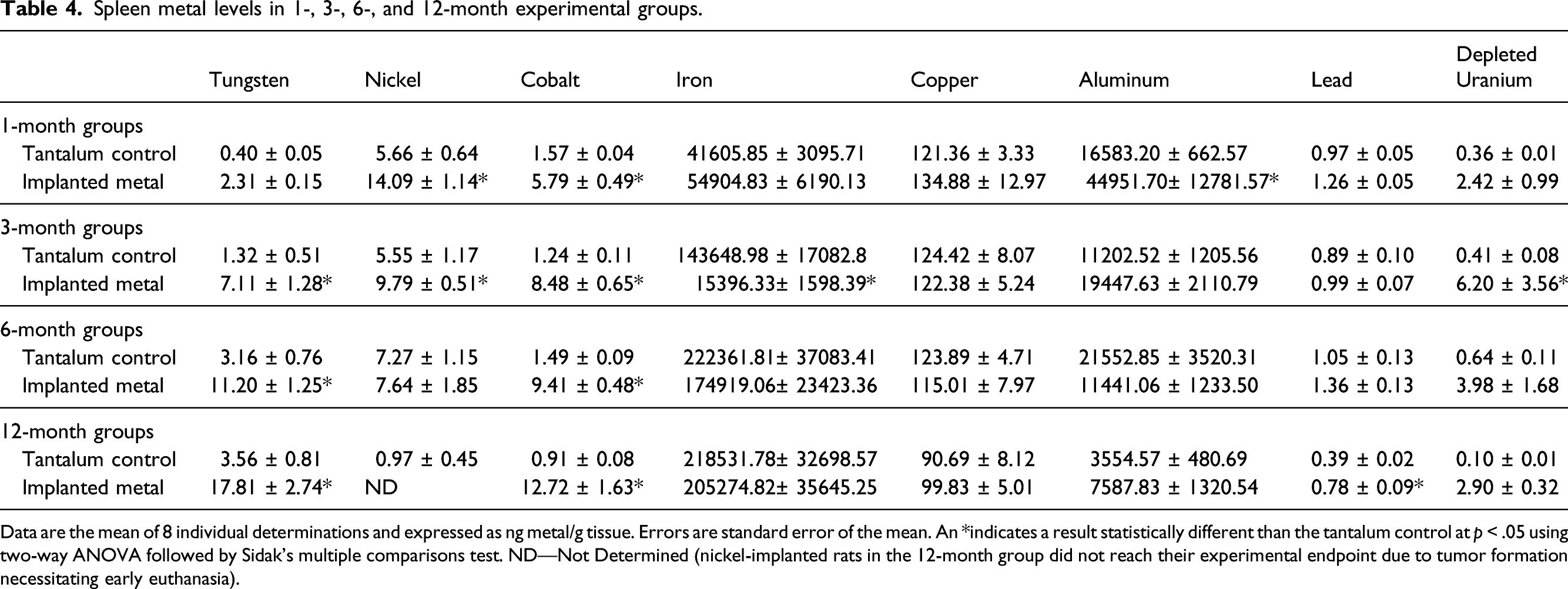
Data are the mean of 8 individual determinations and expressed as ng metal/g tissue. Errors are standard error of the mean. An *indicates a result statistically different than the tantalum control at p < .05 using two-way ANOVA followed by Sidak’s multiple comparisons test. ND—Not Determined (nickel-implanted rats in the 12-month group did not reach their experimental endpoint due to tumor formation necessitating early euthanasia).
Testes metal levels in 1-, 3-, 6-, and 12-month experimental groups.
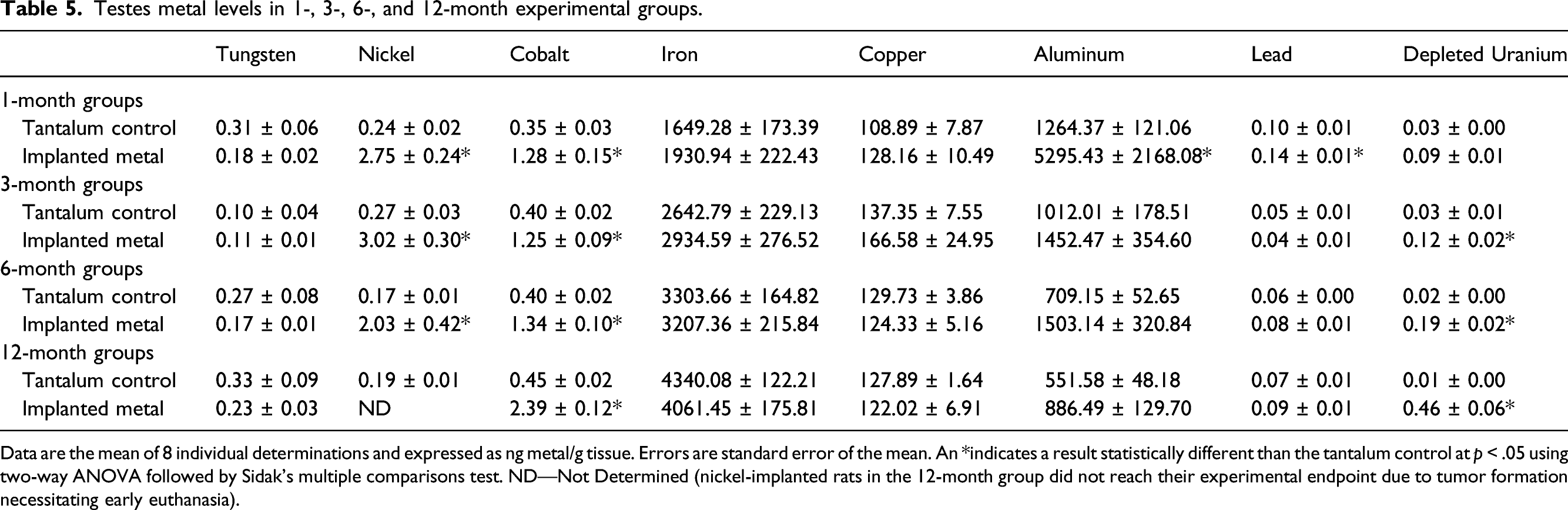
Data are the mean of 8 individual determinations and expressed as ng metal/g tissue. Errors are standard error of the mean. An *indicates a result statistically different than the tantalum control at p < .05 using two-way ANOVA followed by Sidak’s multiple comparisons test. ND—Not Determined (nickel-implanted rats in the 12-month group did not reach their experimental endpoint due to tumor formation necessitating early euthanasia).
Gastrocnemius metal levels in 1-, 3-, 6-, and 12-month experimental groups.
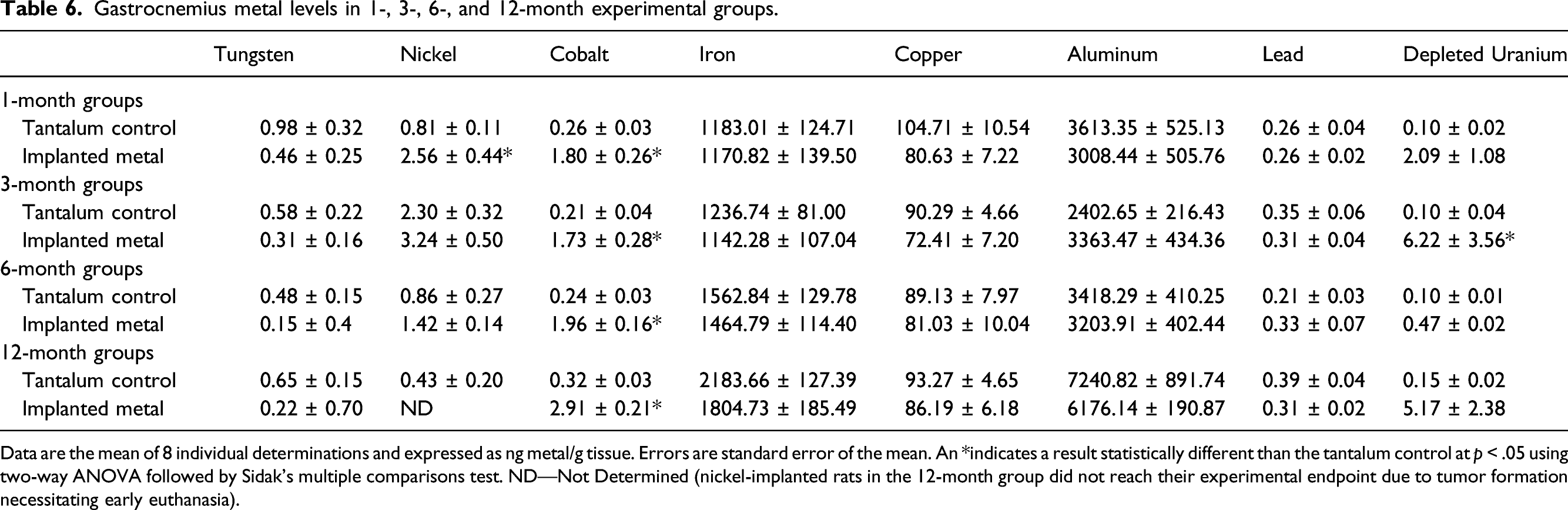
Data are the mean of 8 individual determinations and expressed as ng metal/g tissue. Errors are standard error of the mean. An *indicates a result statistically different than the tantalum control at p < .05 using two-way ANOVA followed by Sidak’s multiple comparisons test. ND—Not Determined (nickel-implanted rats in the 12-month group did not reach their experimental endpoint due to tumor formation necessitating early euthanasia).
Lung metal levels in 1-, 3-, 6-, and 12-month experimental groups.
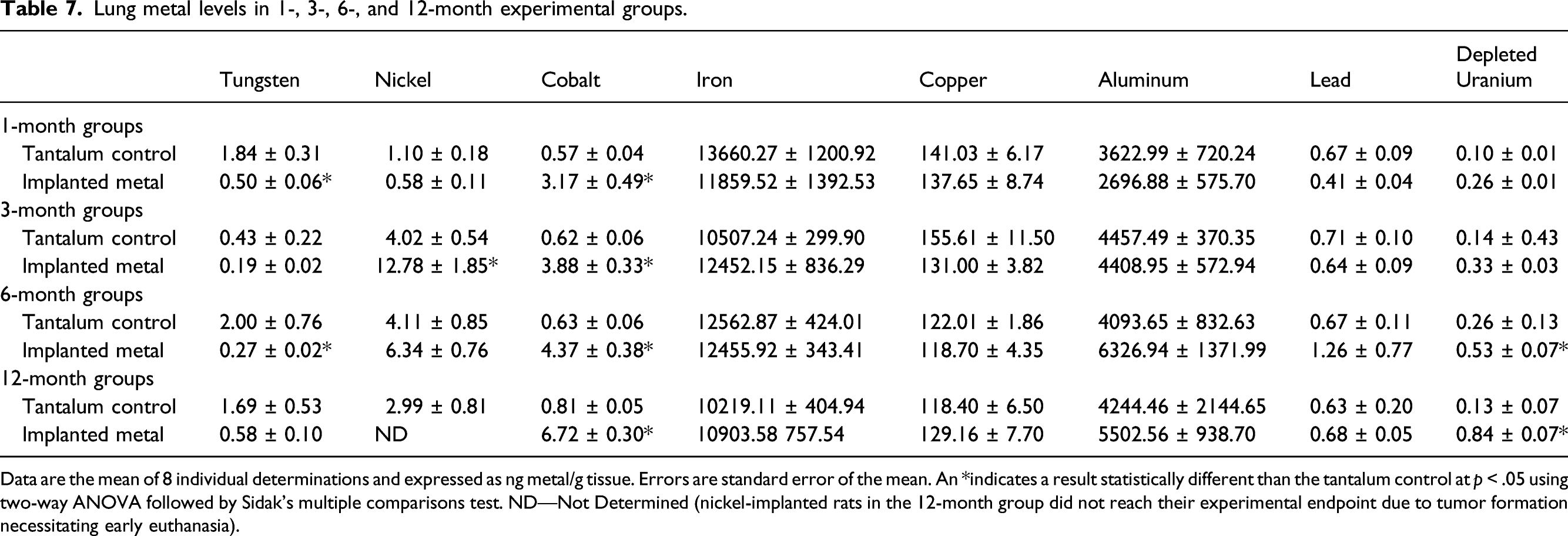
Data are the mean of 8 individual determinations and expressed as ng metal/g tissue. Errors are standard error of the mean. An *indicates a result statistically different than the tantalum control at p < .05 using two-way ANOVA followed by Sidak’s multiple comparisons test. ND—Not Determined (nickel-implanted rats in the 12-month group did not reach their experimental endpoint due to tumor formation necessitating early euthanasia).
Tibia metal levels in 1-, 3-, 6-, and 12-month experimental groups.
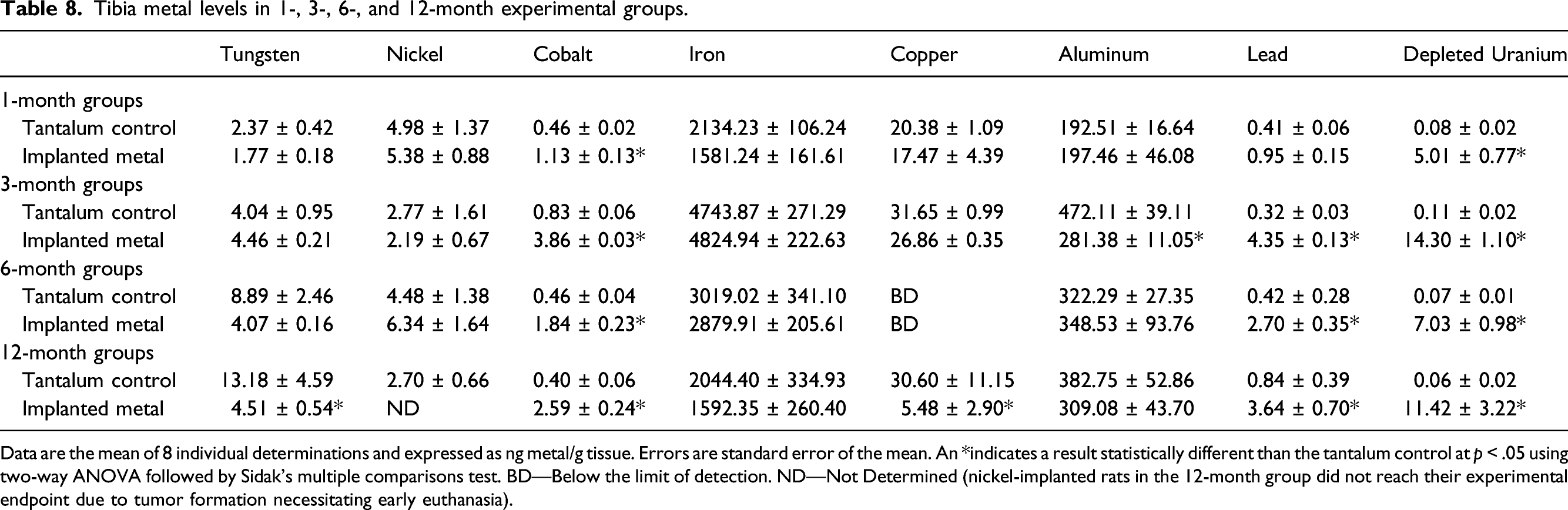
Data are the mean of 8 individual determinations and expressed as ng metal/g tissue. Errors are standard error of the mean. An *indicates a result statistically different than the tantalum control at p < .05 using two-way ANOVA followed by Sidak’s multiple comparisons test. BD—Below the limit of detection. ND—Not Determined (nickel-implanted rats in the 12-month group did not reach their experimental endpoint due to tumor formation necessitating early euthanasia).
Fibula metal levels in 1-, 3-, 6-, and 12-month experimental groups.
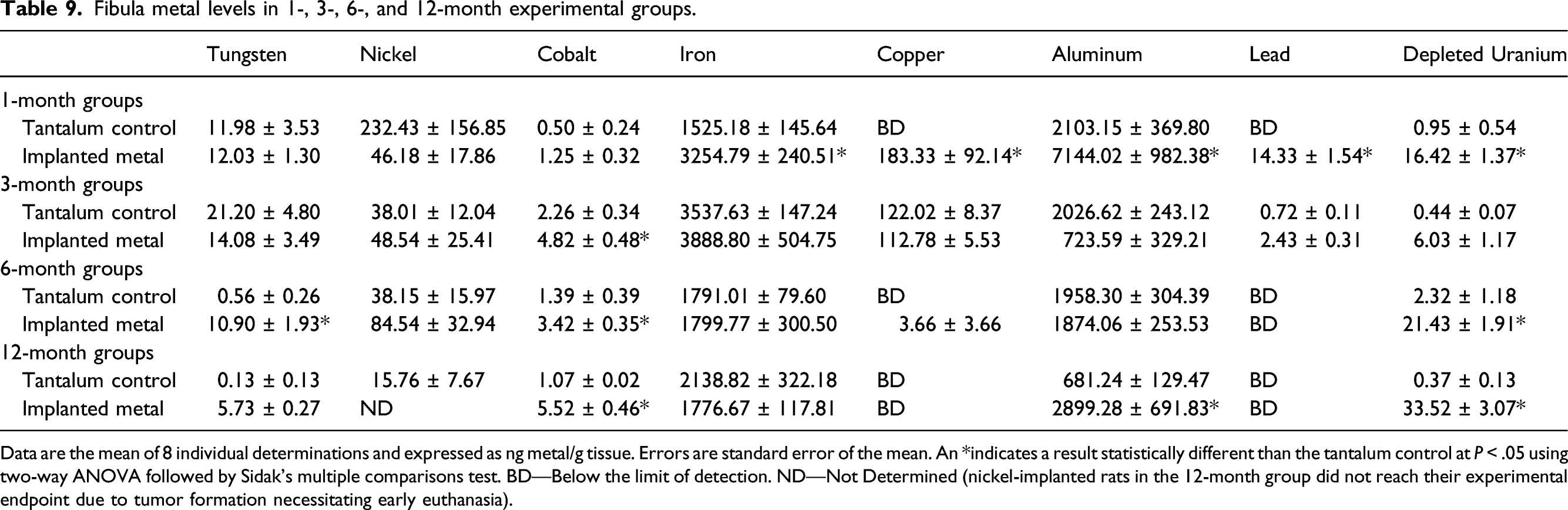
Data are the mean of 8 individual determinations and expressed as ng metal/g tissue. Errors are standard error of the mean. An *indicates a result statistically different than the tantalum control at P < .05 using two-way ANOVA followed by Sidak’s multiple comparisons test. BD—Below the limit of detection. ND—Not Determined (nickel-implanted rats in the 12-month group did not reach their experimental endpoint due to tumor formation necessitating early euthanasia).
Femur metal levels in 1-, 3-, 6-, and 12-month experimental groups.
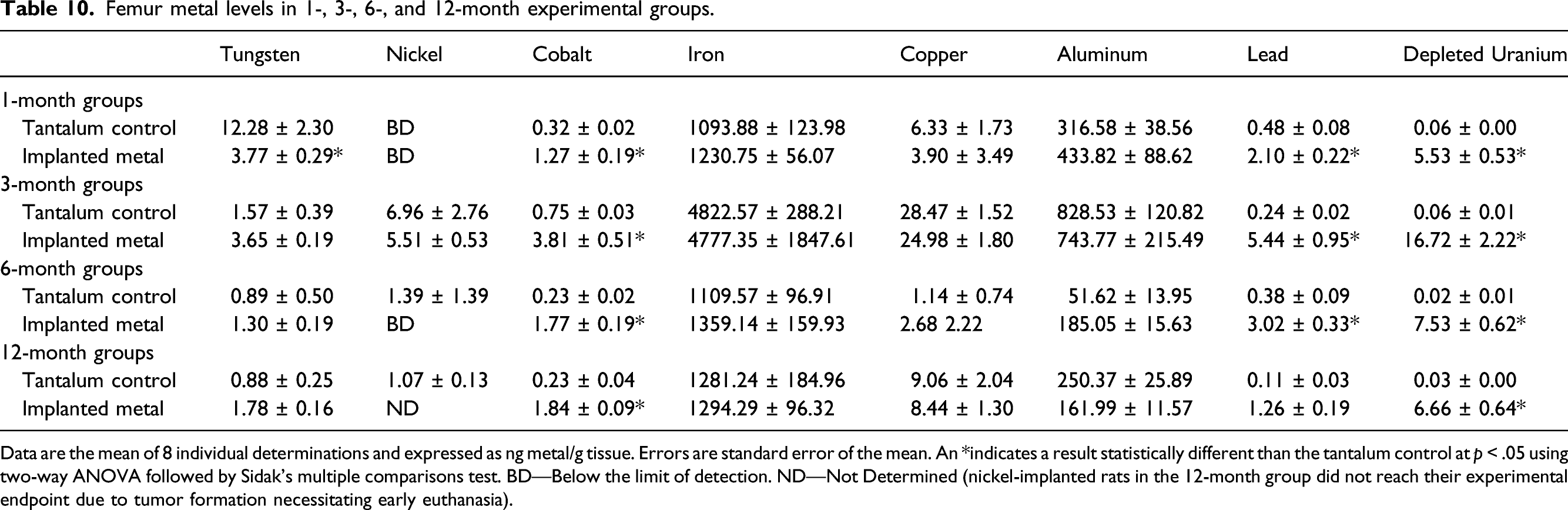
Data are the mean of 8 individual determinations and expressed as ng metal/g tissue. Errors are standard error of the mean. An *indicates a result statistically different than the tantalum control at p < .05 using two-way ANOVA followed by Sidak’s multiple comparisons test. BD—Below the limit of detection. ND—Not Determined (nickel-implanted rats in the 12-month group did not reach their experimental endpoint due to tumor formation necessitating early euthanasia).
Discussion
Embedded metal fragment injuries are an inevitable consequence of armed conflict. Advances in weapon design, as well as the use of improvised explosive devices, now mean that there is theoretically no limit on what metals could be found in an embedded fragment wound. In most cases, the toxicological and carcinogenic potential of many of these metals are not known. Thus, the short- and long-term health effects of these types of injuries are an emerging medical issue. As a result, the United States DoD and DVA developed a list of “metals of concern” with respect to embedded fragment wounds.24,25 Using a rodent model designed to investigate the effects of embedded metal fragments, we selected several metals from these lists to assess the associated health risks. Herein we report on the tissue distribution patterns of these metals at 1-, 3-, 6-, and 12-months post-implantation.
Earlier studies on embedded metal fragments suggest that, over time, many can solubilize with the released metals excreted in the urine and, in some cases, deposited in specific tissues.10,12–14,34,36,38 We had previously reported that metals selected from the DoD/DVA list of “metals of concern” when implanted into the gastrocnemius muscle of laboratory rats also eventually solubilized and were excreted in the urine. 26 In addition, some of the solubilized metals were capable of crossing the blood-brain barrier and depositing in discrete regions of the brain. 27 In this study, with the exception of iron, copper, and aluminum, all of the implanted metals were found at significantly elevated levels in the kidney. This is not surprising considering that excretion is the primary route of elimination once the metals solubilize. The extent of distribution of metals in the kidney also varies over time. For example, tungsten and lead levels tend to increase over time with a peak at 6-months post-implantation followed by a decrease at 12-months post-implantation. Nickel levels in the kidney peak at 3-months post-implantation before dropping at 6-months post-implantation possibly due to tumor formation around the implanted pellet limiting metal solubilization. Cobalt levels in the kidney also peak at 3-months post-implantation but remained constant for the remainder of the experimental period while depleted uranium levels in the kidney continued to increase over time. As reported earlier, histopathological examination of the kidney did not reveal any observable metal-induced damage. 26 In fact, histopathology did not identify any metal-associated tissue lesions with the exception of rhabdomyosarcoma formation at the pellet implantation sites in the nickel and cobalt groups.27,39
Not all implanted metals localized to all tissues at significant levels. In fact, implanted copper, iron, and aluminum did not result in significantly higher levels in any of the tissues analyzed, possibly because the small number of pellets implanted did not significantly add to the levels found normally in the body. In addition, the implanted copper pellets were discovered to move from the gastrocnemius muscle and extrude through the skin by 3-months post-implantation, so it is not unexpected that no significantly high levels of copper were identified in any of the tissues. 39 Other metals were only found in a limited number of tissues. For example, tungsten was found at significant levels only in spleen, while nickel was elevated in both liver and testes. On the other hand, cobalt, lead, and depleted uranium were found in a wide variety of tissues including liver, spleen, testes, lung, tibia, fibula, and femur. Tungsten and cobalt levels in spleen exhibited a similar pattern with tissue levels of both consistently increasing over the duration of exposure. Conversely, nickel levels peaked at 1-month and depleted uranium levels at 6-months post-implantation before both decreasing. Nickel showed similar accumulation patterns in liver, testes, gastrocnemius, and lung with a peak early after implantation (1 to 3 months) followed by a decline. There were no significant levels of nickel in any of the bone samples. Cobalt levels in liver, testes, gastrocnemius, and lung also showed a constant pattern of accumulation with a peak early after implantation with the tissue levels of the metal remaining relatively constant throughout the experimental period. Lead accumulation was not found in any significant amounts over time in gastrocnemius, testes, or lung; and in liver reached peak accumulation by 3-months before decreasing. A similar pattern was observed for lead in bone. For most tissues, depleted uranium accumulation tended to rise throughout the experimental period.
As noted earlier, embedded metal fragments, especially those suffered in wartime, were originally considered inert and were left in place. We now know that this is not the case and that certain metals can solubilize and be transported to tissues far from the original injury. This metal solubilization and transport is not limited to war wounds but has also been identified as occurring in medical implants such as metal-on-metal hip replacements. For example, although elevated levels of cobalt and chromium have been found in the serum and urine of patients with failed hip replacements, there have also been instances where the joint failure led to cobalt-induced cardiotoxicity and significantly high levels of cobalt in the heart muscle. 40
As described above, with the exception of nickel and cobalt, no overt adverse effects resulting from the implanted metals were seen in the tissues of the experimental rats. It should be noted that the amount of metal implanted into the experimental rats is similar to that reported being surgically excised from a wounded veteran. 12 Additional research from this project is currently investigating tissue biomarkers and histopathological assessment of the rodent tissues; therefore, it may be possible that the amount of metal implanted is not high enough to result in substantial adverse health effects. However, metal deposition in tissues is not without risks. Standard toxicological and carcinogenic concerns aside, metals are also involved in the production of reactive oxygen species through the Fenton and Haber-Weiss reactions.41,42 These reactive oxygen species can initiate a cascade of toxic effects leading to lipid peroxidation as well as protein and DNA damage. Metals have also been shown to induce epigenetic and gene expression changes.43,44 Investigations using gastrocnemius muscle isolated from our implanted rat cohorts has demonstrated that the embedded metals induced transcriptional changes in the tissue surrounding the pellet and changed urinary miRNA patterns.39,45 This raises serious concerns as to the long-term effects of metals translocated from embedded fragments to other tissues. As shown previously, certain metals can cross the blood-brain barrier 27 and as demonstrated in this report, also cross the blood-testes barrier and were also found in bone. The effect of these metals on gene expression in these sensitive tissues requires further investigation as does the pathways involved in transporting the metal from the embedded fragment to these tissues. Research on the biokinetics of embedded material suggests that multiple routes are required to account for the clearance of easily solubilized metals as well as any particulate material. 46 A variety of factors will affect the rate of solubilization and translocation including the chemical and physical properties of the embedded fragment and the induction of inflammation in the tissue and the recruitment of macrophages and other immune system cells to the damaged area. 22 It may be possible to assess serum and urinary oxidative stress biomarkers to identify the onset of adverse health effects resulting from the embedded metal, an area we continue to investigate.
With the realization that not all embedded metal fragments can be considered “inert,” the current conundrum with respect to the management of these injuries is how to balance the long-term health risks with the morbidity that can sometimes occur as a result of extensive surgery to remove the fragments. Current guidance still leans toward leaving embedded fragments in place unless they are easily accessible or may present a life-threatening health issue in the future. 47 However, with the virtually limitless number of metals and metal mixtures that may be found in embedded fragment wounds, the lack of a knowledge base with respect to long-term health effects, especially solubility and translocation properties, put health care professionals at a disadvantage when designing treatment strategies for dealing with these types of injuries.
Footnotes
Acknowledgements
The authors would like to thank Raisa Marshall, Anya Fan, and William Danchanko for their expertise in the pellet implantation surgeries, animal welfare checks, and tissue collection.
Author contributions
Conceptualization, JFK; methodology, VBV, JFH, JFK; software, VBV, JFH, JFK; validation, VBV, JFH, JFK; formal analysis, VBV, JFH; investigation, VBV, JFH, JFK; resources, JFK; data curation, VBV, JFH, JFK; writing—original draft preparation, JFK; writing—review and editing, VBV, JFH, JFK; visualization, VBV, JFH, JFK; supervision, JFK; project administration, JFK; funding acquisition, JFK. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project described was supported by the grant Health Effects of Blast Injuries and Embedded Metal Fragments (W81XWH-16-2–0058) from the Congressionally Directed Medical Research Program (CDMRP) Peer-Reviewed Medical Research Program.
Ethical approval
All procedures involving animals were (a) conducted with maximal possible well-being of the rats, (b) approved by the AFRRI Institutional Animal Care and Use Committee (IACUC) prior to the start of the study under protocol 2016-05-006, and (c) performed in compliance with the guidelines set forth in the Guide for the Care and Use of Laboratory Animals in an Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC)-accredited facility.
Data availability statement
All data supporting the results described are provided within the manuscript.

