Abstract
To understand the effects of endocrine-disrupting chemicals (EDCs), the mechanism(s) by which EDCs exert their harmful effects on humans and their offspring needs careful examination and clarification. Epigenetic modification, including DNA methylation, expression of aberrant microRNA (miRNA), and histone modification, is one mechanism assumed to be a primary pathway leading to the untoward effects of endocrine disruptors. However, it remains unclear whether such epigenetic changes caused by EDCs are truly predicting adverse outcomes. Therefore, it is important to understand the relationship between epigenetic changes and various endocrine endpoints or markers. This paper highlights the possibility that certain chemicals (Cd, As, Pb, bisphenol A, phthalate, polychlorinated biphenyls) reported having ED properties may adversely affect the epigenome. Electronic database sources PubMed, SCOPUS, JSTOR, and the Google Scholar web browser were used to search the literature. The search was based on keywords from existing theories and basic knowledge of endocrine disorders and epigenetic effects, well-known EDCs, and previous search results. Unclear and often conflicting results regarding the effects of EDCs indicate the need for further research to support better risk assessments and management of these chemicals.
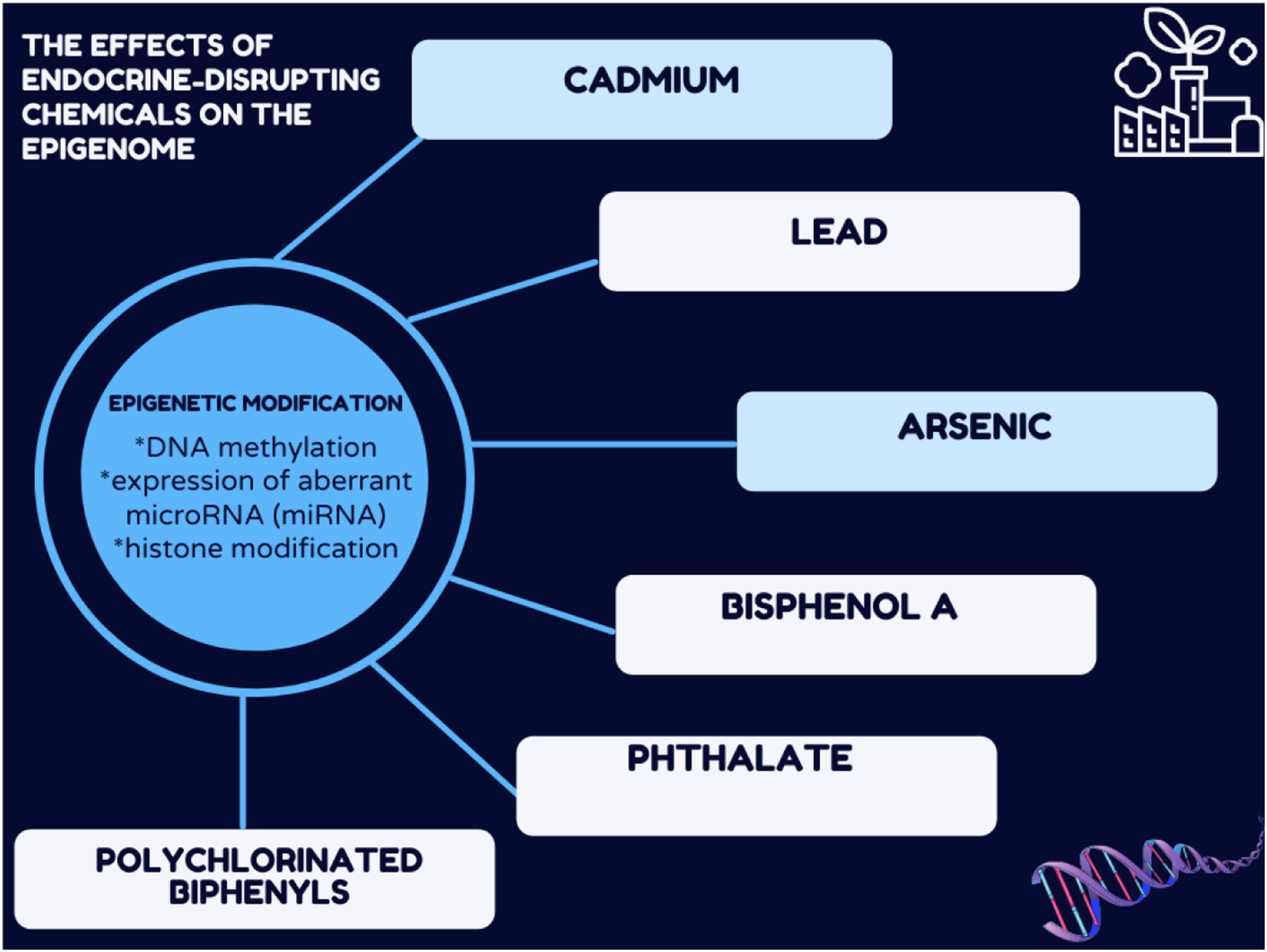
Introduction
Endocrine-disrupting chemicals (EDCs) are defined by the United Nations World Health Organization (UN WHO) as ‘an exogenous substance or mixture that alters function(s) of the endocrine system and consequently causes adverse effects in an intact organism, or its progeny, or (sub)populations.1,2 The topic of EDCs is related to many heated debates. As recently stated in a review by Goumenou et al. (2021), a huge effort has been already made in this field but also is still needed and essential to tackle all hot topics such as low-dose effects of EDCs, critical windows of exposure, nonmonotonic dose-response phenomena, improvement of testing strategies for EDCs identification, appropriateness of the use of threshold or adversity concepts, etc. 3 Furthermore, the introduction the of exposome concept by Wild (2005), as a summation of all exposures and individual experiences over the lifetime, from the moment of conception to advanced age complements the genome, definitely adds another additional layer of complexity to epidemiological studies aiming to derive association between ECDs exposure and particular disease. 4 Another point is disease transmission to future generations and the relations between EDC exposure of ancestors and disease development in future generations.
The mechanism by which EDCs cause disease transmission to future generations has been hypothesized to include epigenetic modifications.5,6 Epigenetic changes are defined as a long-term change in gene expression that persists even when the initial trigger is no longer present and which does not involve a change in gene sequence or structure. 7 The three major epigenetic modifications are DNA (CpG) methylation, aberrant microRNA (miRNA) expression, and histone modification. Various environmental exposures can affect the epigenome.8,9 However, more work is needed to strengthen the link between exposure-associated epigenetic changes and adverse health outcomes. Exposure to EDCs has been intensively investigated concerning global methylation, while less information is available for gene-specific CpG methylation and miRNA expression. A recent review concluded that perinatal exposure to certain EDCs affects the epigenetic machinery and alters the developmental trajectory of the reproductive and neuroendocrine systems in a sex-specific manner, identifying the female brain as more sensitive to disruption by perinatal EDC exposure than the male brain. 10 This difference could provide a possible explanation for why the female reproductive system is more susceptible to EDCs. Rattan and Flaws (2019) have pointed to epidemiological and experimental evidence of a link between EDCs exposure and adverse ovarian and uterine health outcomes in females across generations. 11
One of the important roles of epigenomes in human health is its potential use as a site of action of drugs. Aberrant epigenetic signaling is known to be an important component of disease development, and the reversibility of epigenetic modifications allows for the development of clinically effective drugs. Epigenetic therapies, which are currently being developed, affect the disruption of DNA methyltransferase or histone deacetylase. The use of epigenetic therapy can have a positive effect on drug delivery, optimize the dosing schedule and improve the effectiveness of existing treatment patterns, especially when it comes to chemotherapy, radiation and immunotherapy. 12
Indeed, epigenetics is considered the link that connects genetics and the environment in shaping endocrine function. 13 Nevertheless, it remains unclear whether such EDC-induced epigenetic changes are genuinely predictive of adverse outcomes. 7 Hence, it is of the utmost importance to understand the link between epigenetic changes and endocrine-related endpoints. This review highlights the possible effects that specific chemicals considered to have ED properties can affect the epigenome.
The primary literature was considered in this review. Web-based electronic sources of the databases PubMed, SCOPUS, JSTOR, and the web-based search engine Google Scholar were used for the literature search. The search was based on keywords from existing theories and basic knowledge of endocrine-disrupting and epigenetic effects, well-known EDCs, and previous search results. Specific topics or ideas were not targeted; offhand discovery, snowballing, reference chasing, and tracking citations were not included. The authors have highlighted major EDCs, relevant routes of exposure and possible effects of exposure to these chemicals on the epigenome to drive future research. Other significant considerations, e.g. transgenerational or sex-specific effects and potential application in risk assessment, have been considered.
Cadmium
Cadmium (Cd) is a metal without any significant biological function. It is found in the environment as a standard component of the earth’s crust. Still, Cd also occurs as an environmental pollutant due to human activities (batteries, electronic waste, industrial processes, phosphate fertilizers, pesticides). However, the primary route of exposure of the general population to cadmium is through consumption of food and tobacco smoke. 14 The latest data provided by the European Food Safety Authority (EFSA) regarding the intake of Cd is that the intake should not exceed 2.5 μg/kg body mass per week. 15
It has long been known that chronic exposure to cadmium can lead to a variety of disorders of the lungs, kidneys, liver, bones, brain, and other organs. 16 Because of Cd’s ability to alter hormone homeostasis, Cd is classified as an endocrine disruptor. Its metalloestrogenic activity has been confirmed by Silva and colleagues. 17 Cd is a risk factor for the development of insulin resistance, 18 can harm the thyroid gland, 19 and can negatively affect the male reproductive system. 20 Cd has been reported to cause several epigenetic changes, namely DNA methylation, histone modification, and miRNA. These changes can result in altered gene expression and have been observed both in vitro and in vivo. 21
Cd is classified as a known human carcinogen (Group 1) by the Internacional Agency for Research on Cancer (IARC). 22 Cd causes several types of cancer, but the mechanism by which it causes cancer is not entirely understood. However, experimental and epidemiological studies indicate that Cd-induced cancers involve epigenetic modifications. 21 There is an established link between Cd and kidney and prostate cancer, 22 pancreatic cancer, 23 and thyroid cancer, 24 and the role of miRNA is involved in the development of these cancers. A study conducted by Liang et al. (2020) reported a link between epigenetic regulation of signaling pathways such as Wnt and several metabolic pathways. The study supported the findings of previous studies relating to the metabolic properties of normal and cancerous breast tissue. Changes have been associated with faster growth, proliferation, and migration of cancer cells (e.g. faster energy production in the cell). Cadmium-induced epigenome changes are essential in detecting and understanding the cause of breast cancer. Genes such as TXNRD1 and CCT3 have been identified that may be important for both cancer therapy and detection. 25 An understanding of the impact of Cd on the epigenome may well contribute to the improvement of treatment for such cancers.
Several studies have shown that Cd can affect the growth and development of the fetus. Because Cd can accumulate in the placenta, it has been suggested that the accumulation of Cd in the placenta may lead to dysfunctional growth and development of the embryo. The impaired function may be associated with a disorder in DNA methylation. Everson et al. (2018) showed that the presence of Cd in the placenta was associated with DNA methylation, affecting growth processes, metabolism, and the development of inflammation. Genes involved in the immune and inflammatory signaling pathways (RORA, ACOT7, and TNFAIP2) are associated with a child’s size at birth. This study showed that epigenetic regulation of cell growth and inflammatory or immune pathways in the placenta might contribute to cadmium-mediated reproductive toxicity. 26 It has been reported that DNA methylation can lead to changes in sperm levels in mice. 27 Further research, however, is needed to assess the impact of epigenetic changes induced by Cd on future generations.
Arsenic
Arsenic (As) is a toxic element widely present in the earth’s crust. The primary use of arsenic is in alloys of lead (batteries and ammunition) and semiconductor electronic devices. Arsenic trioxide has been used to produce pesticides and treat wood products; however, these applications, for the most part, have been phased out following the recognition of the toxicity of arsenic. People are most often exposed to arsenic by consuming polluted water, a severe problem for public health, especially in areas such as Bangladesh. In 2009, the Scientific Panel on Contaminants in the Food Chain (CONTAM Panel) within EFSA adopted the opinion that the provisional tolerable weekly intake (PTWI) established by the Joint FAO/WHO Expert Committee on Food Additives (JECFA), which was 15 μg/kg body mass, was not adequate. Adverse effects have been found to occur when exposed to lower doses than those suggested by JECFA. The CONTAM Panel concluded that the Benchmark dose lower confidence limit (BMDL01) is between 0.3 and 8 μg As/kg body mass per day. 28
Due to arsenic exposure, health problems such as kidney and liver disorders, neurological problems, skin problems, and reproductive and cardiovascular toxicity can occur. In addition, arsenic has been reported to have mutagenic, genotoxic, and carcinogenic properties. 29 Because arsenic stimulates the formation of DNA adducts and cross-links, 30 its genotoxicity stems primarily from the ability of arsenic to produce reactive oxygen species. 31 Similar to Cd, As acts through DNA methylation, histone modification, and miRNA. Exposure to this metalloid has also been reported to affect telomerase function, potentially leading to cancer or cell death. 32
According to its IARC classification, the inorganic compounds of As are Group 1 human carcinogens. Chronic exposure to arsenic can act on oncogene or tumor suppressor genes because of the action of arsenic on miRNA, and As has been linked to lung, skin, bladder, and several other types of cancer. 33 miR-182-5p is suppressed in the presence of arsenic, which contributes to the overexpression of hypoxia factors, and the overexpression of these factors has been suggested to contribute to the carcinogenic potential of arsenic. 34 As is involved in miRNA carcinogenesis through its effect on the regulation of the p53 gene. As affects miR-200-b, which can lead to reduced transcription of the p 53 gene. At the same time, growth factors such as TNF-alpha are released. 35 Zhou and Xi (2018) have reported a link between changes in histone levels, such as acetylation and methylation, with cancer development. 36 A small number of studies have suggested that histone phosphorylation may contribute to this effect. For example, arsenic can induce histone three phosphorylation, which increases the expression of specific oncogenic proteins. 37
Other harmful effects can occur because of the action of As on the epigenome. One example is the relationship between placental DNA methylation and arsenic exposure, especially LYRM2 methylation. These results need to be evaluated further to better explain the placenta’s role in protecting the embryo/fetus from arsenic exposure. 38 Wallace and colleagues (2020) have recently reported a link between miRNA expression changes and neurodegenerative diseases (Alzheimer’s and Parkinson’s disease). 39
Lead
Lead (Pb) is found naturally in the earth’s crust. Significant sources of lead in the environment are from mines and metal smelters. Numerous health disorders occur following lead exposure,40,41 including diseases of the nervous, hematopoietic, reproductive, and gastrointestinal systems. According to IARC, inorganic lead is a Group 2A carcinogen, probably carcinogenic to humans. 42 The U.S. Food and Drug Administration (FDA) has set a maximum daily intake of lead in food, the Interim Reference Level (IRL). The IRL is 3 μg/day for children and 12.5 μg/day for adults. 43
Exposure to Pb can lead to changes in the epigenome. The influence of Pb on the epigenetic regulation of genes that affect the development of neurons has been investigated by Masoud et al. (2016) who reported the effect of Pb on the expression of genes encoding specific miRNAs. Their study showed that exposure to lead at an early age could adversely affect miRNA expression, which has been linked to the overexpression of neurotoxic proteins and the potential development of Alzheimer’s disease. 44 A case-control study conducted in China suggested a link between increased miRNA21 expression, urine Pb levels, and the occurrence of microalbuminuria, which suggested that miRNA21 may be a mediator in the development of lead-induced albuminuria. 45 Pb has also been reported to increase the expression of certain forms of miRNA and decrease other miRNAs. Li et al. (2015) suggested that Pb interacts with the placenta via miRNA, adversely affecting development and cognitive function. 46
Bisphenol A
Bisphenol A (BPA) was first synthesized in 1891, but it was not until 1936 that it was reported to have estrogenic activity. 47 In 2013, the global production of BPA was approximately 6.7 million tons. 48 Its use in plastics, food packaging, and toys has resulted in ubiquitous and long-term human exposure to BPA. Bisphenol A is one of the most thoroughly investigated endocrine-disrupting chemicals. Fortunately, BPA is rapidly metabolized into non-bioactive metabolites, such as glucuronide conjugates, with a short half-life of approximately 4–5 h. The U.S. Environmental Protection Agency (EPA) has set the safe limit for BPA exposure at 50 μg/kg body mass daily. In comparison, the European Food Safety Authority (EFSA) had a lower limit of 4 μg/kg body mass daily. 49 However, in February 2022 the EFSA Panel on Food Contact Materials, Enzymes, and Processing Aids recommended a limit of 0.04 ng BPA/kg/day.
The significant pathways for BPA endocrine disruption involve ER and PPARγ. 50 The timing of BPA exposure and the developmental phase of the exposed organism are of significant importance. 51 As with many EDCs, the earlier the exposure during development, the more permanent and far-reaching are the effects. 49 For example, UDP-glucuronosyltransferase (UGT), the primary enzyme responsible for BPA conjugation and excretion, displays altered isoform expression during pregnancy. Consequently, with maternal BPA elimination capacity altered, the fetus is potentially more vulnerable to BPA exposure. Additionally, the lack or reduced fetal BPA elimination, possibly from a poor expression of liver UGT in the fetus, may be another factor contributing to increased fetal vulnerability to epigenetic changes induced by BPA. 50
Prenatal exposure to BPA increased hypermethylation of CAPS2, TNFRSF25, and HKR1 genes by promoting hypermethylation of relevant cytosine-phosphate-guanine (CpG) sites, often resulting in obesity.52,53 There is evidence that even low BPA levels can affect the epigenome and that the effect is sex-specific. A cross-sectional study of prenatal BPA exposure suggested that DNA hypermethylation, a primary epigenetic mechanism, was the predominant effect of BPA exposure among male infants. In contrast, the female infants mainly showed DNA hypomethylation. 54 Another study suggested that male rats exposed to BPA expressed the free fatty acid uptake gene Fat/Cd36 more than females or controls. At the same time, the expression of genes involved in triglyceride synthesis and β-oxidation, such as Dgat, Agpat6, Cebpα, Cebpβ, Pck1, Acox1, Cpt1a, and Cybb, was decreased. These changes subsequently led to an increase in hepatic steatosis in male rats.
The exposure to BPA also led to the hypermethylation of the gene for carnitine palmitoyl transferase 1A (CPTA1), an enzyme enabling long-chain fatty acids to enter the mitochondria and further promoting their oxidation by transferring acyl groups, originating from fatty acids, from coenzyme A to carnitine. 55 Further evidence supporting the hypothesis that BPA favors epigenetically induced metabolic disorders, indicating the promotion of obesity and possibly insulin resistance following BPA exposure, was highlighted in a study by Junge et al. (2018). These authors investigated epigenetic changes induced by BPA in infant cord blood. Their results were confirmed in experimental animals and on human adipocyte-derived mesenchymal stem cells. The human mesoderm-specific transcript (MEST) promoter was the target affected by BPA. This gene codes α/β hydrolases, a family of enzymes linked to obesity, adipocyte size, and preadipocyte proliferation, and was found to be hypomethylated, which could result in altered expression and could contribute to the onset of the metabolic disorder. 56
Although BPA is a commonly studied EDC, only a few studies on its transgenerational effects were found in the literature. One study, concerning the exposure of pregnant rats to BPA, concluded that exposure to the chemical promoted infertility in male descendants of female rats exposed to BPA during pregnancy. The androgen receptor expression, ERβ, steroid receptor co-regulator 1, and nuclear co-repressor protein were diminished in male rats.57,58 The effect was transgenerational, affecting phenotypes of the F1-F3 generations. When interpreting the results of such studies on females, it is essential to keep in mind – if F0 exposure took place during pregnancy, F1 ova (which would become F2) were also exposed. To be genuinely transgenerational, it has to affect at least the F3 generation. 59 Interestingly, BPA was also found to decrease DNA methylation in rats, involving enzymes such as PDE type 4 variant 4 and coat color.60,61
Phthalates
Phthalates and phthalate esters are liquid plasticizers used to produce floor tiles, vinyl flooring, wall coverings, furniture upholstery and mattresses, textiles, personal care products, household supplies, toys, pacifiers, teethers, nipples, and medical equipment. 62 Within a decade of their initial use in the 1920s, the use of phthalates and phthalate esters was widespread with the expansion of polyvinyl chloride plastics. Phthalates are usually not chemically bound to the plastic, making them easier to absorb, which is why they are often detected in human urine, serum, and breast milk. The daily exposure to di (2-ethylhexyl)phthalate (DEHP) is estimated to be between three (3) and 30 μg/kg. 49 According to EFSA, the tolerable daily intake (TDI) for DEHP is 50 μg/kg body mass/day. 63
Exposure to phthalates has been reported to have various untoward effects on the ovary, uterus, anterior pituitary gland, reproductive cycle, fertility in females, and steroidogenesis. Phthalates are also known for their weak affinity for estrogen receptors and induction of peroxisome proliferator-activated receptor (PPARβ) in adipose tissue. 49 The hypothesis that some of these adverse effects are realized at least partly through the PPAR pathway is supported by the fact that stressors, such as the PPARγ antagonist T0070907, have a significantly stronger effect when administered after prior exposure to phthalates. Lee et al. (2017) explained the impact of phthalates in low doses as a “first hit” against the fetal endocrine system that renders them vulnerable to “second hits” in adulthood, represented by a PPARγ antagonist. Furthermore, retinoid X receptors were associated with decreased aldosterone levels in their study, complementing the hypothesis that the effect was realized through PPARs. Interestingly, the phthalate “first hit” also affected the Kcnk5 potassium channels, and their deregulation was observed in animal adrenal glands. 64
Lu et al. (2020) identified several differentially methylated CpG sites associated with phthalate urine concentrations. Many of the sites were linked to genes associated with metabolic syndrome, hypertension, obesity, type 2 diabetes, insulin resistance, and other glycemic traits. 65 Physical traits such as larger waist circumference, higher BMI, or thicker skinfolds, were suggested to be associated with or predict metabolic syndrome or disorders linked to the syndrome in adults, 66 adolescents,67–69 and children (McCArthy 2007, Liang et al. 2015).70,71 At the same time, adipogenesis was reported by Gore and his colleagues. 49
Phthalates were also found to disrupt oocyte development and maturation and have been linked to differentially methylated regions of genes associated with metabolic processes and early growth and tumorigenesis.72,73 Although the effects of EDCs are often more pronounced when the exposure occurs early in life, epigenetic changes following phthalate exposure are not exclusive to exposure during early development. Differentially methylated thyroid receptor-interacting protein 6 (TRIP6) gene promoters were found in peripubertal children exposed to phthalates. 74
Wei et al. (2020) suggested that phthalates could contribute to the onset of metabolic disorders by modulating microRNAs (miRNAs). di (2- Ethylhexyl) phthalate was used to trigger insulin resistance in mice. The methylation of the Dnmt3a-dependent promoter and long non-coding RNAs (lncRNAs) downregulated miR-17 in skeletal muscle cells. The authors proposed that miR-17 then disrupted the Keap1-Nrf2 redox system and activated Txnip in response to the oxidative stress, which then upregulated miR-200a, targeting directly the 3′ untranslated region (UTR) of Insr and Irs1 genes, which could consequently disrupt insulin signaling and hinder glucose uptake. To support this hypothesis, the authors found that miR-17 overexpression resulted in miR-200a silencing and mitigated insulin resistance in the exposed mice. 75 An association of phthalates with miRNAs targeting mRNAs linked to processes characteristic for adverse events, such as angiogenesis, apoptosis, and proliferation of connective tissues, was found in human subjects. 76 Another group has suggested that EDCs could realize their effects through mechanisms involving epigenetic changes of mitochondrial DNA (mtDNA). 77
The transgenerational effects of phthalates were also considered in a recent murine study. The authors found that prenatal exposure to phthalates resulted in changes in the epigenetic regulation of several genes, including Dmnt1, Tet1, and Tet2, spanning several generations, with some encompassing the F1-F3 generations. 78 The increasing number of studies on the epigenetic effects of phthalates in recent years warrants a better understanding of these effects and a better rationale for phthalate regulation in the future.
Polychlorinated biphenyls
Although no longer commercially available, polychlorinated biphenyls (PCBs) are still present in the environment due to their long-standing persistence. For dioxin-like PCBs, EFSA has recommended a tolerable weekly intake of 2 pg TEQ/kg body mass. 79 The active transport of the PCBs across the placenta 80 can result in in utero exposure, in addition to a potential release from lipid storage during pregnancy. 81 PCBs have been reported to interact with various receptors and enzyme systems, and their potential endocrine-disrupting properties have been recently reviewed. 82 The results from a prospective cohort of children across the United States conducted by the National Children’s Study did not establish a relationship between placental PCB levels and methylation in the placenta. 83 A study conducted by Li and colleagues, however, did suggest a positive association between specific PCB congeners and miR-1537 expression. 46 Walker and colleagues have reported that methylation of the enzyme DNMT in the brain was altered by early-life exposure to PCBs.10,84 It has also been shown that the DNA methylation status is affected differently by non-dioxin-like and dioxin-like PCBs: the former favoring hypomethylation, the latter resulting in more methylation. 85 Studies in rats have suggested that miRNAs may be influenced by perinatal exposure to PCBs. 86 The totality of the evidence points toward an epigenetic mechanism underlying the ED properties of the PCBs.
Risk Assessment Considerations
Making use of data on epigenetic changes in risk assessment was suggested in 2014, although at the time, the information concerned toxic metals traditionally researched in toxicology, such as arsenic, cadmium, chromium, lead, and mercury. 87 In 2017, a group of authors further developed this idea, considering EDCs, BPA and di (2-ethylhexyl)phthalate (DEHP), concepts like adverse outcome pathways (AOPs), evidence of causality, and potential application of information in hazard identification, dose-response assessment, and risk characterization. 88 EFSA published a summary report on epigenetics and risk assessment the same year, aiming to identify the role of epigenetics in food safety assessment. The report stated that more work was needed to translate limited experimental data that is available into action relevant to public health. Evaluating data in the context of modes of action (MOAs) and AOPs was identified as the critical step to linking epigenetic modifications to actual alterations in gene expression. Also, the possible hereditary nature of epigenetic changes was identified as highly relevant. 89 To support research on causality, studies examining content of toxic substances in tissues have been conducted recently, focusing on EDCs such as cadmium or epigenetic mechanisms such as DNA methylation or those modulated by miRNAs.90–92 Before epigenetic data is used in risk assessment, another key point to consider is that epigenetic mechanisms can overlap and intertwine and that the totality of epigenetic-related effects could differ from the sum of individual modifications should be considered.88,93 Although the present knowledge on epigenetics and EDCs is limited, thus preventing its use in risk assessment at the moment, recent advancements have paved the way for future experimental studies that could result in information needed to apply this knowledge in real-world risk assessment.
Conclusion
The adverse effects of toxic metals as endocrine disruptors appear to involve their interaction with the epigenome. Defining the precise mechanism by which Pb and the heavy metals act on the epigenome leading to harmful effects, will require more research in the clinic and at the bench. Although there has been undeniable advancement in understanding BPA, phthalates, and BCBs, many aspects of exposure, especially tissue- and organ-specific effects, mechanistic data, and the translation of the conclusions from animal to human remain largely unexplored. A clearer interpretation of the diverse and at times conflicting results surrounding EDCs is warranted and further research is needed to ensure better risk assessments, EDC management, and regulation for legacy EDCs with a rational approach to the introduction of new replacement chemicals that are not endocrine disruptors.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The work of AB and DM was supported by the Science Fund of the Republic of Serbia, PROMIS, DecodExpo (6066532), The work of AAT was supported by the Russian Ministry of Science and Higher Education (0856-2020-0008).
