Abstract
Iron (Fe) is an essential trace element required for several physiological processes. It plays important roles in mitochondrial function, synthesis, and metabolism of the neurotransmitter, as well as oxygen transport. However, excess Fe can cause toxicity. Particularly, Fe overload may result in neurotoxicity, contributing to the development and progression of neurodegenerative diseases, although the molecular mechanisms underlying Fe-induced neurodegeneration have yet to be entirely understood. Alternative (non-rodent) experimental models have been pointed as important approaches to elucidate molecular and physiological events mediating Fe-induced pathology. Among such alternative strategies, an advantageous experimental worm-model system, Caenorhabditis elegans (C. elegans), has been used to investigate Fe-induced neurotoxicity and neurodegenerative disorders. Its genome has been fully sequenced, corroborating that it shares significant homology with mammalians, and has approximately 40% of human disease-related genes. As part of this review, we discuss studies using the C. elegans model to study molecular mechanisms such as oxidative stress, mitochondrial dysfunction, disturbed homeostasis, and its potential contribution to the study of metal-induced neurodegenerative diseases such as Parkinson’s disease (PD) and Alzheimer’s disease (AD).
Introduction
Iron (Fe) is a transition metal widely found in the environment. It is an essential element, participating in several biochemical redox reactions that rely on the Fe’s capability to donate or receive electrons, assuming two redox states [ferrous (Fe2+) and ferric (Fe3+)].1,2 Additionally, it is crucial to cellular growth and differentiation, DNA synthesis, mitochondrial function, ATP generation, and neurotransmitter synthesis, metabolism and important enzymes such as hemoglobin, which carry oxygen throughout the body.3,4 Although Fe is essential, Fe overload may cause damage in various organs such as the liver, heart, gastrointestinal tract, lung, and brain, especially under oxygenated conditions. 5 The abnormal Fe accumulation in the brain correlates with several neurogenerative diseases such as Alzheimer’s disease (AD), Parkinson’s disease (PD), and Huntington’s disease (HD). 6
Neurodegenerative diseases are a group of illnesses characterized by abnormal protein deposition in distinct regions of the human brain, leading to progressive dysfunction and loss of function. 7 Indeed, studies have reported that elevated levels of Fe and changes in Fe metabolism in the brain are involved in the pathogenesis of numerous neurodegenerative diseases.8,9 Several factors contribute to Fe overload in the brain, such as aging (where the accumulation of Fe in specific areas of the brain causes neurodegenerative processes to occur) or previous conditions caused by a genetic mutation. For instance, hemochromatosis and aceruloplasminemia increase Fe levels in the brain.6,10 In addition, a mutation in the L-subunit of ferritin, a key protein involved in Fe storage, leads to abnormal aggregates of ferritin and iron in the brain (primarily in the globus pallidus) 11 ; this condition is characterized by severe neurological dysfunction. The mechanism by which Fe contributes to neurodegenerative diseases is not entirely understood. Still, studies have described the involvement of oxidative stress since excessive Fe can generate reactive oxygen species (ROS) via the Fenton and Haber-Weiss reactions. These reactions produce a highly reactive hydroxyl radical ion (OH.), leading to cellular damage in the neuronal cells. 12
Moreover, alterations in mitochondrial Fe homeostasis causes reduced ATP levels and the release of cytochrome c, and mitochondrial fragmentation resulting in altered morphology.13,14 In addition, Fe promotes the aggregation of amyloid beta-peptide and tau, promoting its toxicity in AD patients. 15 It was reported that this metal stimulates the conversion of α-synuclein from the α-helix to the β-sheet conformation in dopaminergic neurons. 16
Animal models are important tools in several field of biomedical research. They may be described as non-human living organisms that show analogy of the physiological behavior or a spontaneous or induced pathological process that can be studied. To study a particular pathology, an animal model should reproduce the principal structures of it. Moreover, these models may be used to identify the onset, the development, the mechanism involved and potential therapeutic targets.17,18
Growing evidence suggests Fe’s involvement in neurodegenerative diseases, and therefore studies to clarify and provide a better understanding of the mechanisms involved are required. In this context, alternative models in vivo and in vitro are essential approaches to provide new insights on neurodegenerative damage induced by Fe. Caenorhabditis elegans (C. elegans) is an advantageous experimental worm-model system. C. elegans is a non-parasitic nematode, the first multicellular organism to have its genome fully sequenced. The worm has a small size (∼1.5 mm adult), short lifespan with a rapid life cycle, where worms after hatching show different larval stages (L1, L2, L3, L4) until adulthood. C. elegans hermaphrodites self-fertilize and produce ∼300 offsprings, enabling easy maintenance in the lab with low cost. Due to its translucent body, reporter gene probes as green fluorescent protein (GFP) can be inserted and used to analyze cell morphology, protein expression, neurodegeneration in specific neurons, to name a few. C. elegans genes are evolutionarily conserved, sharing homology to mammalian systems, and these nematodes express approximately 40% of human disease-related genes.19–22
Recent studies have shown in C. elegans model have addressed the mechanism by which Fe may enter in the brain, its role in neurodegenerative diseases caused by metal overload and potential treatment or chelator therapy.13,23 Indeed, C. elegans has orthologs proteins of humans that are highly conserved and participate in Fe metabolism, such as the divalent metal transporter 1 (DMT-1), ferritin (FTN-1, FTN-2), and ferroportin (FPN-1.1, FPN-1.2, FPN-1.3). 24 Moreover, the nematode has been used to evaluate dyshomeostasis and toxicity induced by Fe addressing several important parameters such as lifespan, brood size, body bend frequency, oxidative stress, ATP levels, chemotaxis plasticity, neuronal viability, to name a few.25,26 In this present review, we addressed new insights on Fe toxicity and its involvement in neurodegenerative diseases, using C. elegans as an alternative and complementary model system.
C. elegans as an alternative animal model to study Fe imbalance and ferroptosis observed in Parkinsonism
Mitochondria play a key role in Fe homeostasis as they confer critical compartmentalization to regulate its cellular levels strictly. Fe in excess provokes a fragmented mitochondrion due to an excessive fission, event associated with neuronal death triggered by oxidative stress, while its deficiency induces a fussed/aggregated mitochondrion. 27 Importantly, Fe’s redox properties allow efficient electron transfer in the mitochondria, which translates into energy supply for biologic reactions. However, as mentioned, these Fe reactive properties can also promote ROS generation, damaging intracellular systems at high concentrations. Thus, in the presence of O2, Fe is continuously cycling between Fe2+ and Fe+3. Nearly 65% Fe is bound to hemoglobin as Fe2+, the 10% in myoglobin (as Fe2+), cytochromes (Fe2+ and Fe3+), and other proteins; the remaining 25% is present in transferrin, which transports Fe in the plasma, or bound to storage proteins such as ferritin (in the cells) and hemosiderin, while only trace amounts of the metal are free in plasma. 28 With age and under conditions of Fe overload, mitochondrial redox-active Fe concentration (free Fe) rises, generating hydroxyl radicals (OH.), though the potential mechanisms are still not known.
The Fe redox reaction mediates the generation of other free radicals, such as those generated from non-enzymatic dopamine (DA) oxidation in the dopaminergic cells. This leads to the production of semiquinones and H2O2 at the level of the mitochondrial Complex I of the electron transfer chain (revised in Carocci et al. 2018
12
). Notably, the mitochondrial insult induced by different environmental and endogenous toxic agents can initiate and lead to Fe release from non-toxic sites (as the Fe-bounded proteins), which can trigger and accelerate the progression of neurodegenerative diseases. The Fe-S clusters present in mitochondrial Complex I and III can be attacked by ROS, such as superoxide anion O2- releasing free Fe that participates in the Fenton reaction triggering a general pro-oxidant cycle. The pair Fe2+/Fe3+ can stimulate membrane lipid peroxidation, which can intensify oxidative stress, contributing to mitochondrial death through the formation of the mitochondrial permeability pore.
29
This evidence supports the possibility that mitochondrial lipid oxidation could be a critical step in ferroptosis. Thereby, this recently characterized cellular death pathway is initiated by a disruption in the antioxidant defenses dependent on glutathione and glutathione peroxidase 4 (GPx4), which results in lipid peroxidation and eventual cell death.30–32 The antiporter glutamate/cystine (xCT) exports cellular glutamate in interchange with extracellular cystine. Once inside the cell, cystine is converted into cysteine, a precursor of the endogenous antioxidant glutathione, a process that is inhibited by erastin, a classic ferroptotic agent. Under homeostatic conditions, the ferroptotic signal is ended by GPx4
33
as well as by the ferroptosis suppressor protein 1 (FSP1)-coenzyme Q10 (CoQ10)-NAD(P)H,
34
and (iii) GTP cyclohydrolase-1 (GCH1)-tetrahydrobiopterin/dihydrobiopterin (BH4/BH2)
35
pathways. Of note, GPx4 needs glutathione as a cofactor, while its inactivation, either by glutathione depletion or directly by inhibitors, induces lipid peroxidation/oxidative stress and eventually cell death.
36
Conversely, deferoxamine, although it does not cross biological membranes, can prevent cellular death by trapping the excess of Fe. Hence, despite the physiological role and the mechanisms that govern ferroptosis-induced cellular death are not thoroughly described, four processes are known to be critical: (1) Fe accumulation; (2) glutathione depletion; (3) lipid peroxidation, and (4) the insertion of highly oxidizable phospholipids containing polyunsaturated fatty acids (PUFA) into the membranes, increasing oxidative stress.37,38 The Figure 1 shows the pathways in ferroptosis. The overall scheme demonstrating pathways involved in ferroptosis. Elevation of intracellular free iron may result from increased iron absorption through transferrin receptor (TfR)-dependent and divalent metal transporter 1 (dmt-1)-dependent mechanisms, as well as degradation of intracellular ferritin through NCOA4-mediated ferritinophagy. Moreover, mitochondrial oxidative stress was also shown to increase iron release from mitochondria, thus contributing to elevated labile iron pool. Mitochondrial dysfunction also results in electron leakage and superoxide generation at Complex I and III. Superoxide dismutation by both mitochondrial and cytoplasmic superoxide dismutase (SOD) results in increased production of hydrogen peroxide (H2O2). The latter is detoxified by GPX in a reaction using reduced glutathione (GSH) as a cofactor. GSH is synthetized by γ-glutamylcysteine synthetase and glutathione synthetase from precursor cysteine, that is transported into the cell in the form of cystine using antiporter glutamate/cystine (Xc−). Accumulating H2O2 and increased LIP result in participation of Fe2+ in Fenton reaction resulting in generation of highly reactive hydroxyl radical (HO•) that induces lipid (L) peroxidation with the formation of lipid radical (L•), lipid peroxyl radical (LOO•), and lipid hydroperoxide (LOOH) ultimately leading to ferroptosis. GPX4 is also capable of terminating lipid peroxidation by LOOH decomposition. Therefore, inhibition of both Xc- and GPX4 is considered as a critical event in promoting ferroptosis.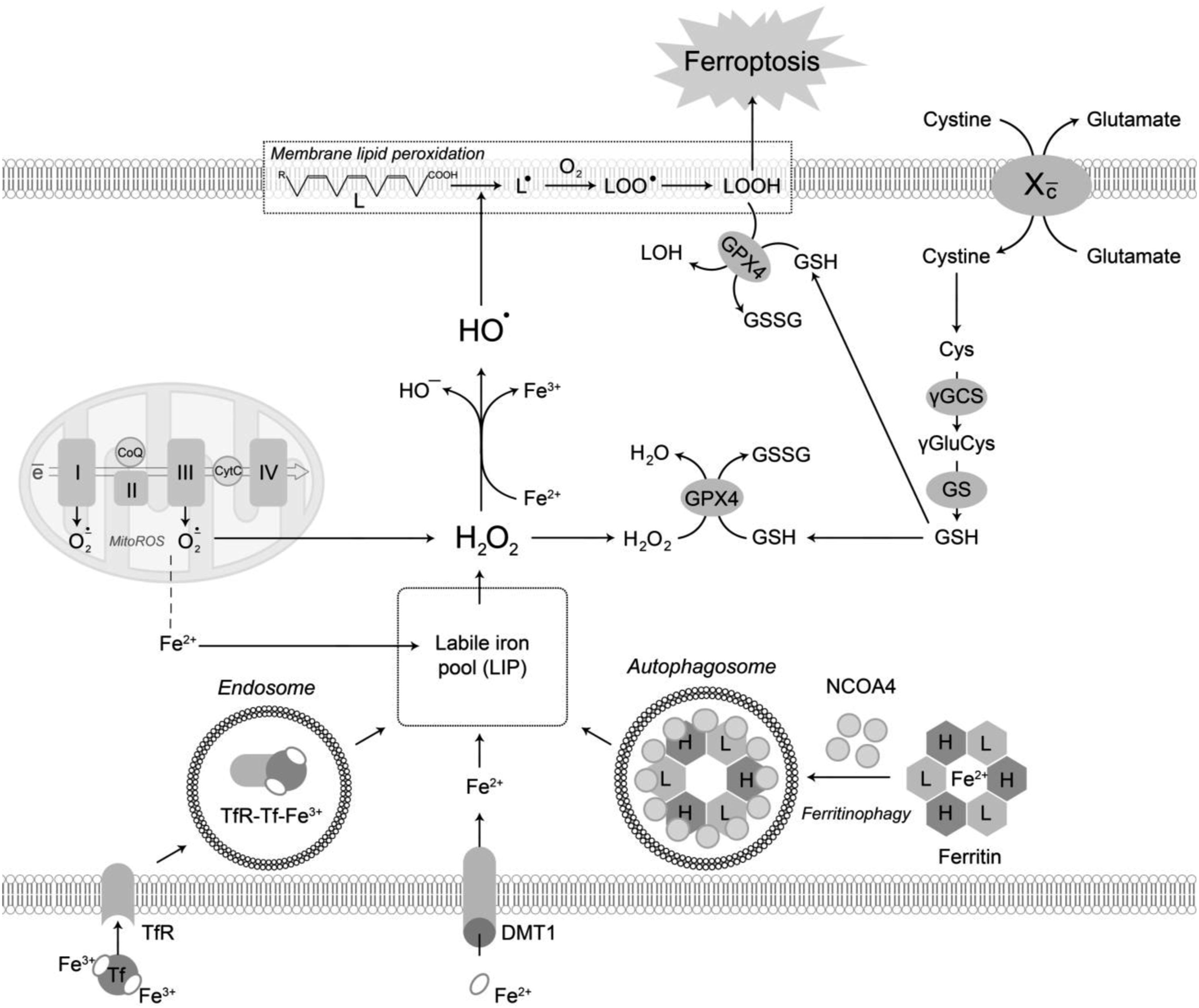
C. elegans expresses orthologous genes of the principal components of the pathways that regulate Fe homeostasis. 24 Fagundez et al. 2015 reported that Fe exposure resulted in motor damage at the dopaminergic and cholinergic neuronal functionality level, accompanied by an increase in ROS production and a reduction in life expectancy. 39 Moreover, ferroptosis was shown to be modulated by inhibitors, ferritin genetic manipulation, by NADPH oxidase and GPxs, as well as by the dietary co-supplementation with oleic acid. This demonstrates the multiple parallels between mammalian and nematode-induced ferroptosis. 40 In other studies in C. elegans, it has been reported that glutathione depletion is coupled to an elevation of ferrous Fe, with both events happening in adult life to “imprint” the cells to ferroptosis. Moreover, C. elegans also possesses glutathione and other systems related to the redox balance such as GPx, glutathione-S-transferases, glutaredoxins, thioredoxins, and peroxiredoxins. 41 Finally, blocking ferroptosis by inhibiting lipid peroxidation or limiting Fe retention, age-related cell death decreases while the length of life markedly increases 42 as opposed to an accelerated aging process after gene deletion of the four GPxs expressed in the nematode. 43
One of the characteristics of aging is mitochondrial dysfunction. It is proposed that Complex I regulates aging by at least two mechanisms: (1) ROS-dependent, which leads to mitochondrial DNA damage, and (2) ROS-independent by the control of the NAD+/NADH relationship. The control of the levels of these cofactors will allow the regulation of glycoxidative and lipooxidative damage and the activation of sirtuins, NADH-dependent deacetylases involved in longevity. 44 Mitophagy involves mitochondria degradation through an autophagy-mediated process (autophagocytosis), a lysosomal mechanism that is genetically regulated in cells and functions to remove and recycle dysfunctional components. 45 During aging, mitophagy decreases in efficiency, contributing to age-related disorders in mammals and leading to mitochondrial accumulation in C. elegans. In this respect, mitophagy induction, NAD+ supplementation, Fe chelation, and mitochondrial proteins suppression promote the health and life expectancy in C. elegans.46,47 In addition, and in concordance with an abnormal mitochondrial dynamic, Fe accumulation has been described as another typical feature of aging, concurrently with the pathogenesis of age-associated neurodegenerative disorders increasing ROS generation by the Fenton reaction, facilitating toxic protein aggregation, inducing cell death and inflammation, all causally related to its adverse effects on the CNS. 45 However, its temporal relationship with neurodegeneration must be clarified, because it could play a vital role in the progression yet be unrelated to the initiation of these diseases.37,48,49 Of particular importance, aging-dependent Fe accumulation has already reported in C. elegans and such accumulation was paralleled by a decrease in the levels of GSH, 42 which represents a trigger event leading to ferroptosis. Notably, these same authors observed that ferrostatin 1 (a ferroptosis inhibitor 30 ; and targeting the accumulation of late life iron (using the Fe-chelating agent salicylaldehyde isonicotinoyl hydrazone) resulted in a marked increase in C. elegans lifespan. 42 This study was pioneer in showing a potential relationship between iron accumulation, ferroptosis and decreased lifespan/healthspan.
Preliminary evidence has demonstrated that ferroptosis inhibition can decrease the incidence of PD. Dopaminergic neurons are inherently vulnerable to ferroptosis, a fact that may explain their increased susceptibility to neurodegeneration. 37 Iron accumulation in the brain is observed in the substantia nigra pars compacta in PD. Furthermore, in dopaminergic neurons, ferric ions are quickly reduced to ferrous ions by cytotoxic by products of dopamine oxidation. 50 Neurodegeneration observed in these conditions could be related to the inability of these neurons to manage Fe overload and mitochondrial dysfunction. In this way, Fe and DA form a toxic pair provided the pro-oxidant characteristics of each of these two compounds. Both Fe2+ and Fe+3 can directly bind α-synuclein, but the ferric form would be most potent in accelerating fibrils formation and aggregation.
Emerging evidence points-out that α-synuclein, in turn, plays a role in Fe homeostasis, reducing Fe3+ to Fe2+ due to its ferrireductase activity, thus increasing intracellular Fe2+. 51 In summary, a cyclic association between α-synuclein and Fe appears to exist, in which α-synuclein induces Fe accumulation and Fe induces the protein aggregation. These reactions give rise to a sequence of events in which α-synuclein causes mitochondrial dysfunction, which in turn results in Fe accumulation and higher protein aggregation culminating in hydroxy-radical-mediated damage. 52 In this respect, deferiprone (a Fe chelator approved in the USA and Europe that prevents intracellular Fe overload) and N-acetylcysteine (a precursor of glutathione synthesis) were able to rescue LUHMES cells from erastin-induced toxicity. 53 Moreover, the increased toxicity induced by erastin secondary to increase DA levels was also rescued by ferroptosis inhibitors. Thus, Fe is essential for biological processes under physiological conditions, although it contributes to a pro-oxidant environment in the cells. In the pathologic stage of PD, elevated Fe induces oxidative damage, accelerates DA-mediated toxicity, and aggravates α-synuclein dysfunction promoting its expression and aggregation. The defects in vesicular DA incorporation induced by α-synuclein increases free DA, while the overexpressed protein increases its ferrireductase activity and Fe ferrous levels. At the same time, DA promotes α-synuclein aggregation while the metabolic products react with Fe, manifested in PD as a vicious circle between Fe, dopamine, and α-synuclein. 54 In summary, the combination of mitochondrial dysfunction, free Fe accumulation, and oxidative damage in dopaminergic neurons will initiate cellular death by ferroptosis in aged animals.
C. elegans as a model to study Alzheimer diseases induced by Fe
C. elegans is a powerful genetic model to explore the molecular mechanisms, identify changes in pathways and detect proteins that are involved in neurotoxicological responses and neurodegenerative diseases such as AD, which may be induced by toxic metals, such as mercury and cadmium and also by essential metals such as manganese and Fe, to name a few.55,56 Indeed, AD is the major neurodegenerative disease and some hallmarks such as deposition of Aβ plaques and hyperphosphorylated tau protein creating neurofibrillary tangles may be found. The Aβ plaques are formed by cleavage of amyloid precursor protein (APP) via α-, β-, and γ-secretase enzymes to produce APP fragments including Aβ monomers, which may aggregate, leading to impaired neuronal function. In mammals, APP can be cleaved by an amyloidogenic pathway where β-secretase enzyme cleaves APP, releasing the sAPPβ fragment that is successively cleaved by γ-secretase enzyme liberating Aβ to lumen. Interestingly, the C. elegans genome encodes an ortholog APP- related gene, apl-1, which is cleaved by α and γ-secretase enzymes, but not by β-secretase and therefore Aβ monomers are not formed.57,58 To overcome this limitation, a C. elegans strain model was developed. The CL2006 strain expresses Aβ minigene constitutively by the muscle-specific unc-54 promoter, where Aβ clone was chromosomally integrated with rol-6 phenotypic marker.59,60 This strain was used to investigate the role of oxidative stress on Fe homeostasis disruption. The authors reported that Fe concentration in CL2006 strain was significantly higher than in wild type C. elegans N2, suggesting that Aβ enhances the Fe content, which is a source of redox-generated free radicals, 61 as previous studies demonstrate that CL2006 strain showed more than 2.5-fold higher endogenous ROS levels than wild type. 62 Moreover, the higher Fe levels found in CL2006 strain when compared with wild type also may be observed in patients with mild cognitive impairment and preclinical AD that showed increased Fe levels and deposition in brain regions such as cortex and cerebellum when compared with control individuals. 63
Indeed, it was reported that homeostatic balance changed in CL2006 worms, since that Fe concentration in this strain is higher when compared with wild type and increased ROS accumulation. It was correlated with paralysis behavior in the CL2006 strain. 64 To investigate the mechanism by which Fe can induce ROS formation related to ongoing AD, some Fe transporters to mitochondria were studied. In this line, Mitoferrin-1 and mitoferrin-2 are two proteins in mammals responsible for Fe transportation from the cytoplasm to mitochondria, controlling Fe homeostasis in mitochondria. 65 In C. elegans, there is only mitoferrin protein, an ortholog of human mfn-1, and is involved with the lifespan of the worms and associated with mitochondria Fe regulation. 66
A recent study using CL2006 and GMC101 strains demonstrates that knockdown of mitoferrin-1 could extend the lifespan of both strains. 13 Moreover, down-regulation of mitoferrin-1 decreased the paralysis rate and increased AD progression in this C. elegans model. The authors also reported that down-regulation of mitoferrin-1 changes mitochondrial Fe content, which shows decreased, affecting mitochondria Fe metabolism that results in an up-regulation of the transcription of mitochondrial iron-sulfur protein ferritin-related genes leading to decrease of mitochondria ROS. Therefore, mitoferrin-1 may be involved in mitochondrial Fe homeostasis where its down-regulation can decrease the Fe toxicity in C. elegans. 13
Studies have used the C. elegans model to investigate possible therapeutic agents and chelation Fe to treat neurodegenerative diseases such as AD. In addition, natural phytochemical compounds are described to induce prevention and promising molecules for the development of drugs to treat or prevent neurodegenerative diseases.67–69 In this line, honokiol, a biphenyl neolignan present in the plant Magnolia officinalis, 70 shows anti-inflammatory, neuroprotective, and anti-oxidative properties. This compound was used to evaluate its effect against Aβ-induced toxicity using a transgenic C. elegans model that expresses Aβ42. It was observed that honokiol inhibited the Aβ42 pathway aggregation and honokiol-treated worms exhibited a delayed time-dependent paralysis. Besides, this compound has the ability to chelate Fe, which is a potential clinical strategy to AD therapy. 71
Fe chelators are used to disrupt metal-Aβ interaction and decrease Aβ formation. In this regard, some Fe chelators and their conjugates were used to investigate the possible mitigation of Fe-induced neurotoxicity. The first chelator used to treat Fe overload conditions was desferrioxamine (DFO), a siderophore secreted by the bacteria Streptomyces pilosus. In addition, conjugates with aromatic amines, which inhibited acetylcholinesterase (AChE) activity was developed to obtain DFO conjugates. 72 Recently, Carvalho et al. (2021) reported that new DFO conjugates with aniline (DFOANI), benzo-sulfanilamide (DFOBAN), 2-naphthalenamine (DFONAF) and 6-quinolinamine (DFOQUN) potentiate the inhibition of β-amyloid peptide aggregation and repress AChE activity when compared to DFO. Moreover, behavioral tests in C. elegans showed reversal of Fe-induced motor impairment by DFOQUN, with greater efficacy compared to DFO. The authors suggested that conjugation Fe chelator may be a promising approach for mitigating Fe-induced AD. 73
Conclusion
Iron has vital roles in several physiological functions, especially in the brain. Nevertheless, overexposure to this metal leads to neurotoxic effects and neurodegenerative disorders. In this regard, studies using the C. elegans model have provided a better understanding on the molecular mechanisms and the pathophysiological interrelationship between Fe exposure and neurodegenerative disorders. However, some limitations should be considered regarding the use of C. elegans, given its cellular and anatomical differences, absence of circulatory system and nonexistence of specific organs, such as skin, heart, kidneys, and lung. Furthermore, shortage of males in wild type population and the difficult to identify behaviors of relevance to study psychiatric disorders are important limitations.74,75
Recently, the C. elegans model has been used to investigate new molecular mechanisms such as ferroptosis, which is involved in PD etiology. In addition, Fe transport regulators and proteins involved in neurodegenerative disorders are also mechanisms that may be investigated in the C. elegans model. Moreover, research on new therapeutic targets and new substances with efficacy in treating PD and AD are necessary and may identify novel targets in ameliorating Fe-induced neurotoxicity. Overall, the findings summarized in this review warrant further investigation using the C. elegans model to clarify the molecular mechanisms on Fe-induced neurodegenerative disease with future extrapolation to mammalian systems.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Environmental Health Sciences (NIEHS) R01ES10563 and R01ES07331 to MA.
