Abstract
Introduction
This is an evaluation of the effects of banana peel BP extract on the heavy metals’ mixture HMM mediated oxido-inflammatory effects in the thyroid of female albino rats.
Methods
Five groups (5 female rats/group) were treated as follows for 60 days: Group 1: Deionized water only; Group 2: (Pb, Hg, Mn and Al); Group 3: 200 mg/kg BP extract + HMM; Group 4: 400 mg/kg BP extract + HMM; Group 5: 800 mg/kg BP extract + HMM. On day 60 animals were euthanized, thyroid was harvested and used for, malondialdehyde MDA, nitric oxide NO, antioxidants, tumor necrosis factor alpha (TNF – α), interleukin 6 (IL – 6), Caspase-3, Nuclear factor erythroid 2- related factor 2 (Nrf2), Nuclear factor kappa B (NfkB) and Heme Oxygynase – 1 (Hmox-1) and histopathology.
Results
There was significant bioaccumulation of Pb, Al, Hg and MN; elevated IL-6 and Tnf-α, MDA and NO, caspase-3 and Nrf2, NF-κB and Hmox-1 in the HMM only group in comparison to the control. There was significant (p < 0.05) decrease in SOD, CAT GSH activities in HMM only exposed group in comparison to the control deionized water group, whereas BP co-treatment with HMM significantly (p < 0.05) increased SOD, CAT GSH activities. Co-treatment with BP extract also reversed most of these effects.
Discussion and Conclusions
BP extract may ameliorate HMM -induced thyrotoxicity in female albino rats by blunting oxido-inflammatory activities.
Introduction
Heavy metal exposure in animals and humans has been associated with organ toxicity. 1 The fundamental feature of the deleterious effects of heavy metals is oxidative insult.2,3 Oxidative stress leads to the activation of numerous transducers such as protein kinase C (PKC), and phosphatidylinositol 3 kinase (PI3K) which phosphorylates both Keap1 and Nrf2. 4 On activation, transcriptionally active Nrf2 translocates to the nucleus where it partners with small Maf proteins, Nrf2/Maf heterodimers bind directly to anti-oxidant response elements (AREs) located within promoters of Nrf2 target genes, displacing the transcriptional repressor Bach1. 4 This activation increases the transcriptional expression of Nrf2-inducible genes such as those encoding heme oxygenase-1 (HMOX1), NAD(P)H quinone oxidoreductase (NQO1), and glutathione-S transferase A2 (GSTA2). The products of these genes neutralize reactive oxygen species ROS, biosynthesize glutathione, direct xenobiotic efflux, recycle oxidized proteins and ultimately impair oxidative damage to detoxify the cell.5–9 Chronic oxidative stress triggers apoptosis which prevents cellular degeneration and uncontrollable proliferation. 10 Activation of Nrf2 may therefore mitigate ROS mediated toxicity in the prevention of tissue injury. 11 The thyroid gland is an important regulator of growth, development, and homeostasis of all vertebrates. It plays an integral role in the maintenance of normal body physiological status and is one of the main targets of endocrine disruptors like the heavy metals. 12
Humans employ natural enzymatic and non-enzymatic safeguards against either ROS generation or its detoxification. 13 Among these safeguards are the first line superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX) are indispensable in the entire defence strategy of antioxidants, especially with respect to super oxide anion radical which is exaggerated upon exposure to oxidizing metals. 13 This multifaceted protective machinery may be overburdened by the noxious actions of amplified ROS.14,15 Exogenous supplementation with antioxidant nutraceuticals may augment the amount and upregulate activities of endogenous antioxidants16–19 to serve as credible therapeutic alternatives in combating overelaboration of ROS.15,20,21
Diverse anthropogenic activities especially crude oil exploration in Niger delta, Nigeria have heightened the environmental burden of heavy metals (Pb, Hg, Al and Mn) with concomitant public health challenges demand urgent attention and need to harness available remedies.22–24 It is thought that the phenolic rich banana peel BP extract may be exerting its antioxidant effects by removal of ROS due to the presence of an electron-rich aromatic ring system,25,26 or indirectly by decreasing oxidative stress through modulation of Nrf2 to stimulate antioxidant enzymes. 27 Furthermore, some antioxidants are known to exert anti-apoptotic effect in heavy metal-induced damage via inhibition of oxidative stress due to activated Nrf2 signalling. 28
Given the paucity of data on studies involving female animals, the present study hypothesized that BP extract/natural product may mitigate heavy metal mixture-induced oxidative stress, inflammation, and apoptosis by modulating the Nrf2/Hmox-1 pathway in the thyroid of heavy metal mixture HMM exposed female albino rats.
Materials and methods
Chemicals
Heavy metals such as Lead acetate, Alumnium chloride, Mercury chloride and Manganese dichloride were purchased from Sigma Chemical Co. (St. Louis, MO, USA). Tumor necrosis factor alpha (TNF – α), interleukin 6 (IL – 6), Caspase-3, Nuclear factor erythroid 2- related factor 2 (Nrf2), Nuclear factor kappa B (NfkB) and Heme Oxygynase – 1 (Hmox-1) Rat ELISA Kit were purchased from Elab Science Biotechnology Company, (Beijing, China). All other reagents were of analytical grade and were obtained from the British Drug House (Poole, Dorset, UK).
Collection and identification of banana peel BP
Ripped banana fruits (Musa Cavendish) were purchased from International Institute of Tropical Agriculture (IITA) Ibadan and identified by IITA, (Ibadan, Oyo State), Nigeria.
Preparation of plant extracts
The ripe banana fruit peels were washed and dried in room temperature, pulverized using laboratory blender and sieved. One hundred grams 100 g of BP extract was weighed and 500 mL of deionized water was added, mixed, and filtered with Whatman No. 1 filter paper (GE GmbH, Freiburg, Germany) and stored frozen. 29
Animals and treatments
Twenty five Sprague Dawley female albino rats aged between 6–8 weeks were purchased from the Department of Pharmacology, Animal House, University of Port Harcourt, Rivers State. Animals were housed in standard polypropylene cages under room temperature 25 ± 2°C with a 12-h light/dark cycles throughout the duration of the experiment. Prior to the commencement of the study, the animals were acclimatized for 2 weeks. The ethical approval was obtained from the University of Port Harcourt institutional Centre for Research Management and Development Animal Care and Use Research Ethics Committee (UPH/CEREMAD/REC/18). The experiment was conducted in accordance with the “Guide for the Care of Laboratory Animals” approved by the National Academy of Science (NAS). The animals received standard feed and deionized water ad libitum.
Experimental design
Body, thyroid weight, feed and fluid consumption
The body weight of rats was measured weekly and at the end of the experiment 60th day, while the thyroid weight was recorded directly after the sacrifice on the 60th day.
The absolute weight of thyroid (g) = mean of thyroid weight for each group were taken. The relative weight of thyroid (g/100 g body weight) = mean of thyroid weight for each group/final body weight × 100 were calculated. Feed (g) and fluid consumption (ml) were recorded daily.
Necropsy and harvesting of thyroid
After 60 days of treatment, animals in each group were euthanized under pentobarbital (50 mg/kg IP) anaesthesia. The thyroid of each rat was harvested, rinsed in cold saline water, weighed and used for both biochemical parameters and heavy metal analyses.
Metal analysis
Twenty milligrams of thyroid were digested using 2 mL of perchloric acid and 6 mL of nitric acid. Afterwards, the samples were kept for 30 min before heating at 105°C until digestion was completed. The solution was made up to 15 mL (final volume) with deionized water. Solar thermo elemental flame Atomic Absorption Spectrometer (Model SG 71906) was used to determine the Lead (Pb), Aluminium (Al), Mercury (Hg) and Manganese (Mn) concentrations Okoye et al. 2021. The limits of detection (LoD) were 0.001 mg/Kg for Aluminium (Al), Mercury (Hg) and Manganese (Mn) and 0.01 mg/Kg for Pb, while the limits of quantification (LoQ) were 0.0033 mg/Kg for Aluminium (Al), Mercury (Hg) and Manganese (Mn) and 0.033 mg/Kg for Pb.
Assessment of oxidative stress markers
The malondialdehyde MDA level (lipid peroxidation marker) was assayed using the procedure of Ohkawa and Ohishi. 32
In this technique, MDA reacts with the chromogenic reagent, 2-thiobarbituric acid (TBA) under acidic medium to produce a pink coloured complex at 532 nm absorbance.
Nitric oxide (NO) level was assayed according to the method of Green et al. 33
Assay of antioxidants
Briefly, GPx activity was assayed according to the method of Rotruck et al. 34 Reduced glutathione (GSH) levels were assessed using the technique illustrated by Jollow et al. 35 CAT activity was evaluated using the technique of Clairborne 36 with slight modification. This technique is premised on the principle that catalase in the sample will split hydrogen peroxide which can be estimated at 240 nm using a spectrophotometer. 36 SOD activity was estimated with the technique previously illustrated by Misra and Fridovich. 37 This technique is based on the principle that at pH 10.2, SOD has the capacity to inhibit the autoxidation of epinephrine.
Enzyme-linked immunosorbent assay
The content of the inflammatory cytokines including tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), heme oxygenase and Hmox-1, apoptotic marker caspase 3, and NFκB, Nrf2 in the homogenized thyroid cell supernatant was detected using commercially available ELISA kits following the manufacturer’s instructions. All experiments were conducted in triplicates.
Assessment of inflammatory markers and transcription factor markers
IL-6, TNF –α, Nrf2, Hmox-1, NFκB and Caspase 3 activities were measured with IL-6, TNF –α, Nrf2, Hmox-1, NFκB and Caspase 3 Activity Assay Kit (Beyotime Institute of Biotechnology, Jiangsu, China) according to the manufacturer’s directions. Thyroid samples were lysed for 15 min on ice. The thyroid homogenate was centrifuged at 16,000 g for 10 min at 4°C.
Briefly, standards or samples were added to the micro-ELISA plate wells and combined with the specific antibody. Then a biotinylated detection antibody specific for rat IL-6 and Avidin-Horseradish Peroxidase (HRP) conjugate were added successively to each micro plate well and incubated. Free components were washed away. The substrate solution was added to each well. Only those wells that contain rat IL-6, biotinylated detection antibody and Avidin-HRP conjugate appeared blue in color. The enzyme-substrate reaction was terminated by the addition of stop solution and the color turned yellow. The optical density (OD) was measured spectrophotometrically at a wavelength of 450 nm ± 2 nm. The OD value was proportional to the concentration of rat IL-6. Concentrations of rat IL-6 in the samples were calculated by comparing the OD of the samples to the standard curve. This procedure was repeated for TNF –α, Nrf2, Hmox-1, NFκB and Caspase 3. NO level was assayed according to the method of Green et al. 33
Histopathologic examinations
After 10% neutral buffered formalin fixed 72 h, thyroid was embedded in paraffin by standard histological method, the tissues were sectioned coronally in 5 μm thickness, and slices were conducted after dewaxing and hydration procedures. The sections were thereafter routinely stained with Hematoxylin-Eosin Stain kit (Vector laboratories, USA) according to the manufacturer’s instructions. The sections of the thyroid were evaluated under a light microscope and photographed with image acquisition parameters settings at 400× throughout the process.
Statistical analysis
All the results were expressed as Mean ± Standard deviation (std). Microsoft Xlstat 2014 was used in performing Analysis of Variance and Tukey multiple comparison pairwise tests to check if the concentration of the biomarkers was significantly (at 5% significant level) different between groups. Pandas was used in obtaining the descriptive statistical parameters (biomarkers and heavy metals mean conc.). Seaborn and Matplotlib were used in plotting all graphs. The data analysis involved performing descriptive statistics (mean and std) on the heavy metals and biomarkers concentration before ANOVA was used to establish if there was significant difference in the concentration of the heavy metals and biomarkers among groups. Pearson R correlation was used to understand the relationship among biomarkers.
Results
Heavy metal accumulation in the thyroid of rats treated with BP extract following HMM exposure
The effect of BP extract on the concentrations of Pb, Hg. Al, and Mn in the thyroid of HMM exposed female albino rats is shown in Figure 1. There was marked bioaccumulation of Pb, Hg. Al, and Mn in the thyroid after HMM exposure. At 200, 400 and 800 mg/kg, BP extract showed 15.33, 44.02 and 61.89% reduction in Pb; 29.93, 52.30 and 64.71% reduction in Al (Figure 1(a)); 52.87, 71.29 and 79.18% reduction in Hg; and 55.44, 70.34 and 84.99% reduction in Mn concentration in the thyroid respectively (Figure 1(b)). Effect of banana peel on the concenterations of Hg, Mn, Pb and Al in the thyroid of heavy metal mixture exposed female albino rats. Values with different superscripts (a, b, c, d, e) were significantly different from each other (p < 0.05) and those with the same superscripts were not significantly different.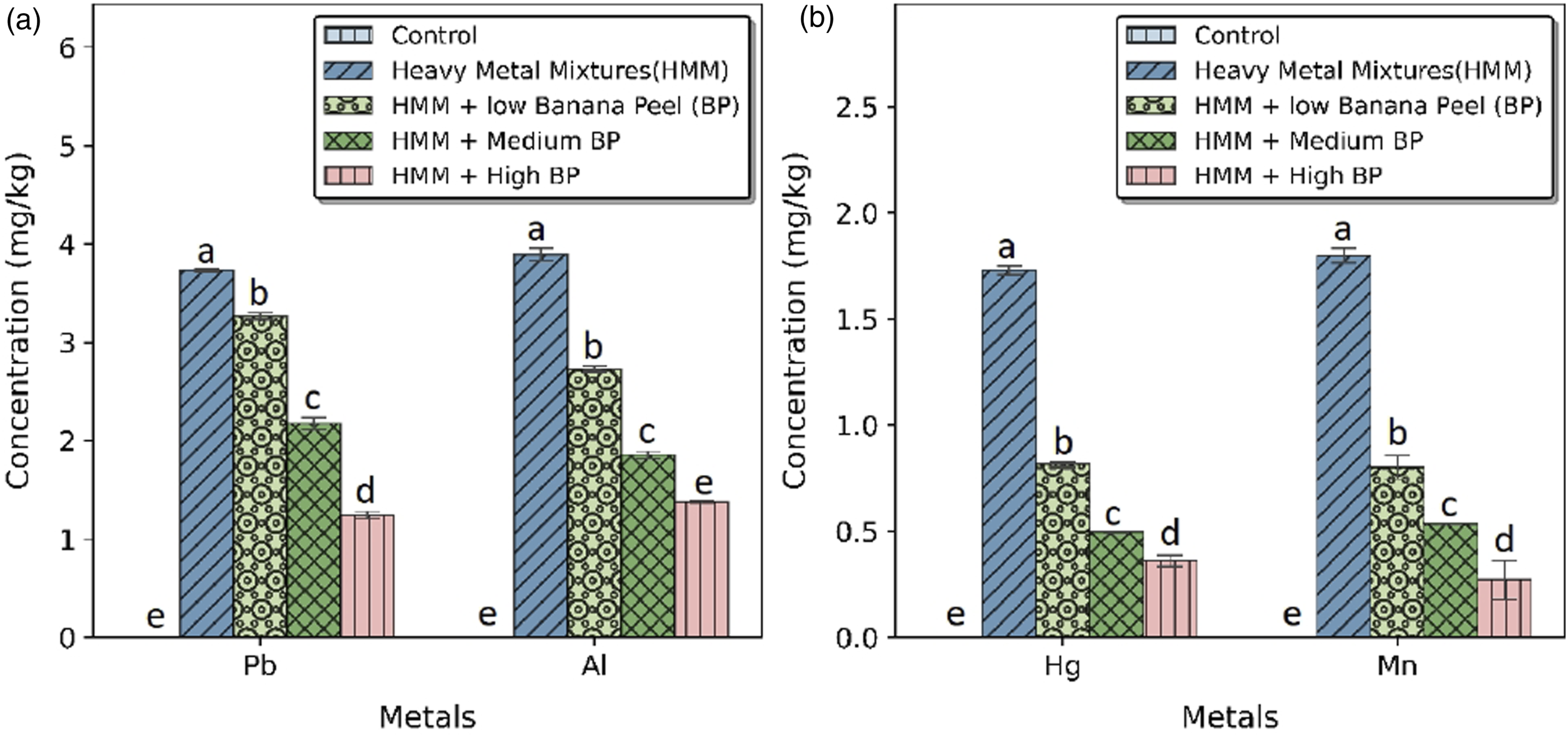
Antioxidant’s profile of rats treated with BP extract following HMM exposure
The effects of BP extract on the thyroidal antioxidant markers (SOD, CAT, GSH and GPx) of HMM exposed female rats are shown in Figure 2. Effect of banana peel on the antioxidant profile in the thyroid of heavy metal mixture exposed female albino rats. Values with different superscripts (a, b, c, d, e) were significantly different from each other (p < 0.05) and those with the same superscripts were not significantly different.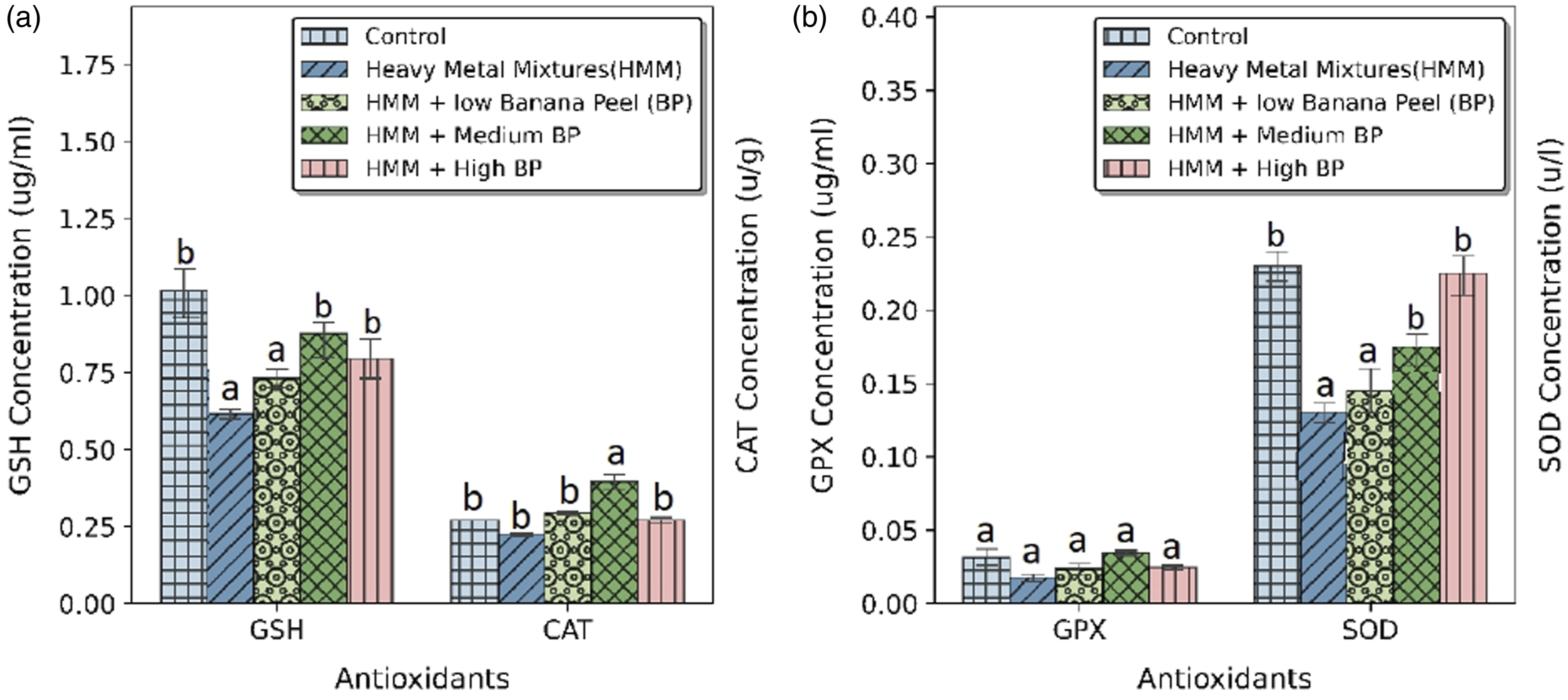
SOD, CAT GSH activities were significantly (p < 0.05) increased by BP extract administration when compared with HMM only treated group (Figures 2(a) and (b)). HMM only exposed rats decreased the antioxidant markers (SOD, CAT, GSH and GPx). There was non-significant difference in the level and activities of GPx between the BP extract treated groups in comparison to the control and the HMM only treated groups.
Lipid peroxidation and oxidative stress profile of rats treated with BP extract following HMM exposure
The effects of BP extract on the oxidative stress markers (MDA and NO) in the thyroid of HMM exposed female rats are shown in Figure 3. There was non-significant difference in MDA levels of HMM treated rats when compared with the BP treated female rats. BP treatment significantly reduced the NO levels when compared with the HMM and control groups (Figure 3(a)). Co-administration of HMM and BP extracts showed dose dependent reduction in the NO levels when compared with the HMM only-treated group. Effect of banana peel on the MDA and NO in the thyroid of heavy metal mixture exposed female albino rats. Values with different superscripts (a, b, c) were significantly different from each other (p < 0.05) and those with the same superscripts were not significantly different.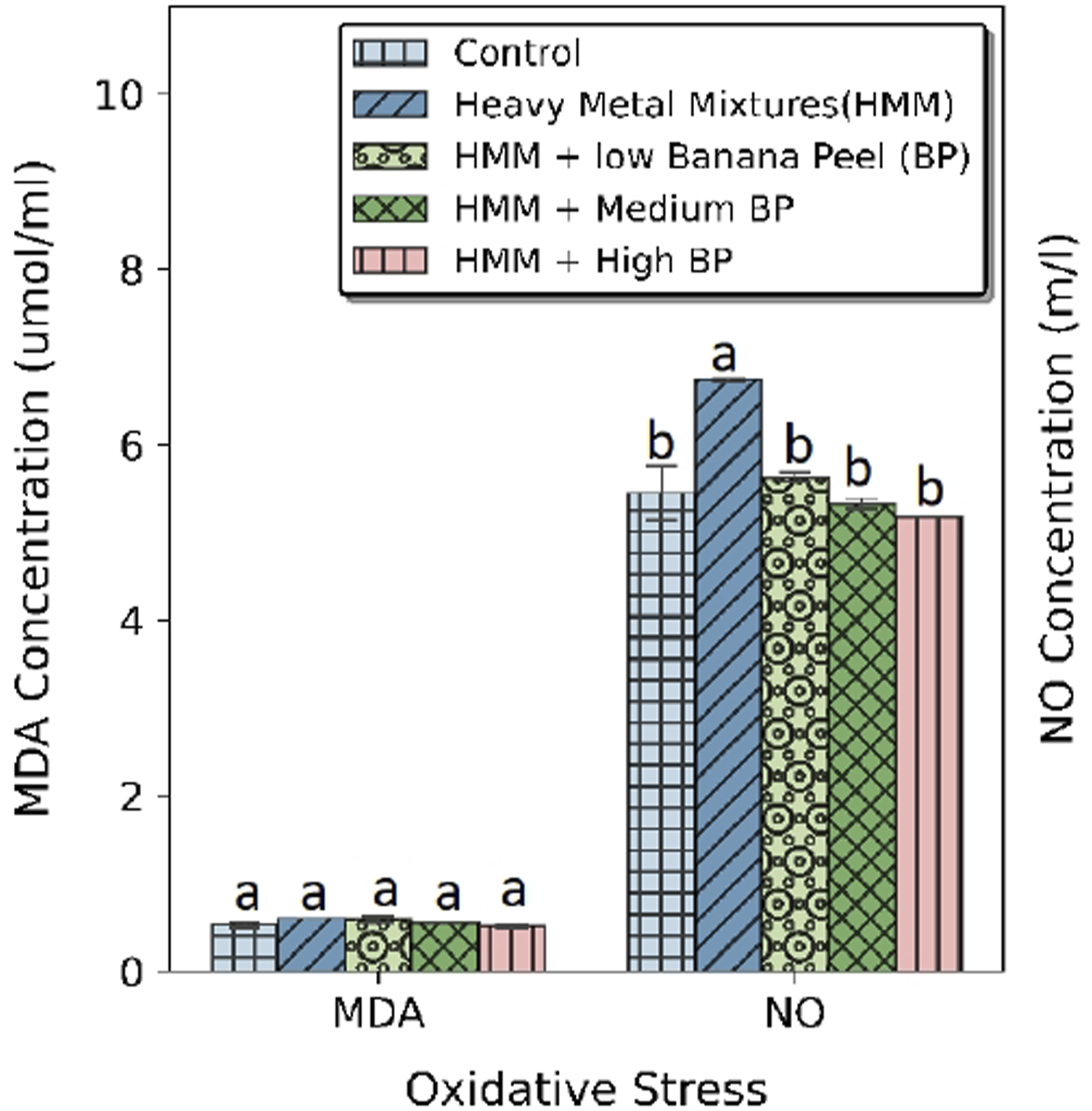
Caspase 3 activation of rats treated with BP extract following HMM exposure
The effects of BP extract on the caspase-3 of HMM exposed female rats are shown in Figure 4. HMM exposure significantly (p < 0.05) showed higher levels of caspase-3 in comparison to 400 and 800 mg/kg BP extract treated groups. The significantly (p < 0.05) decreased level of caspase-3 by BP extract was dose dependent with lowest caspase-3 seen in the 800 mg/kg BP extract treated group (Figure 4). Effect of banana peel on the Caspase-3 in the thyroid of heavy metal mixture exposed female albino rats. Values with different superscripts (a, b, c) were significantly different from each other (p < 0.05) and those with the same superscripts were not significantly different.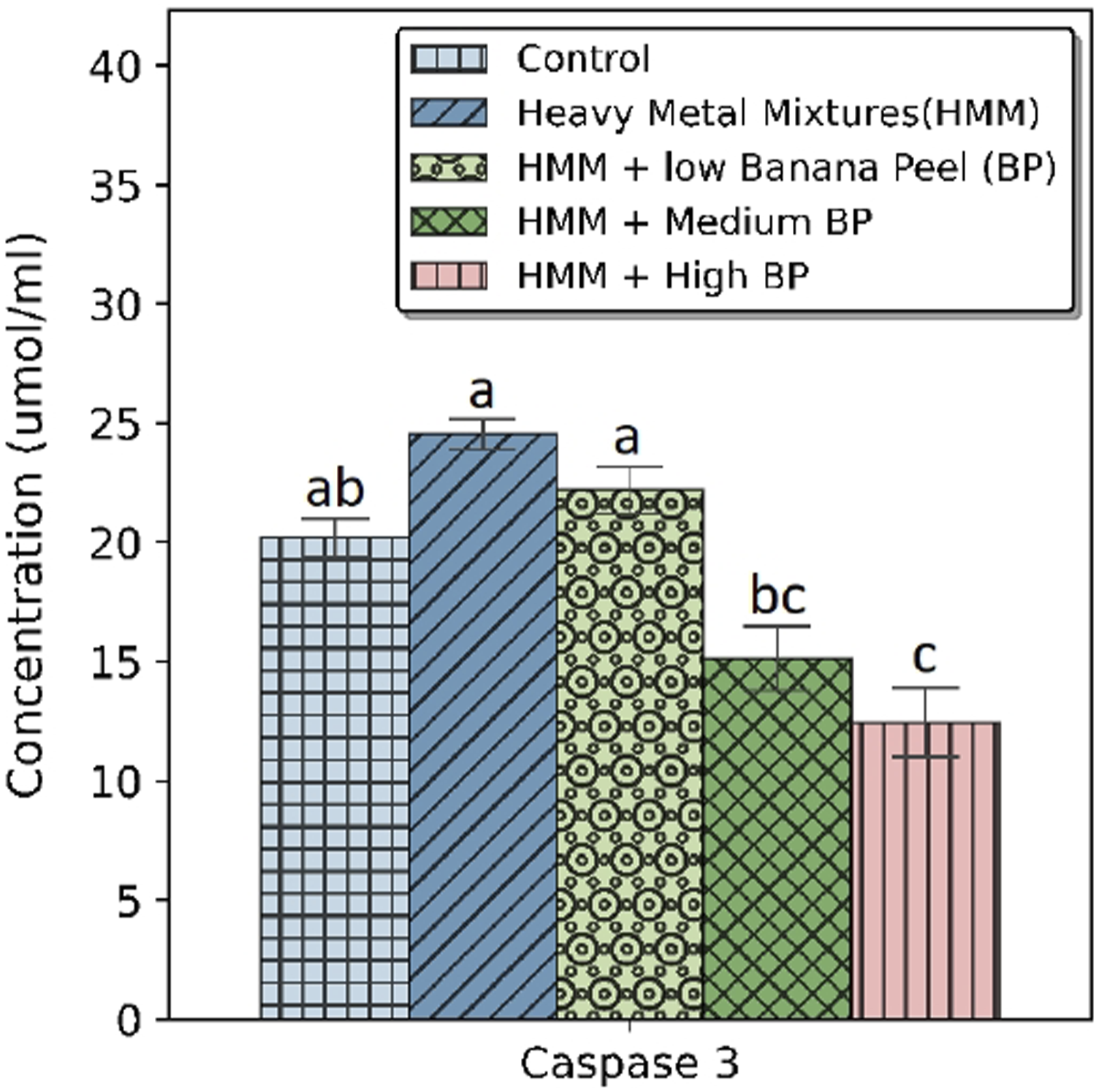
HMM caused non-significant (p > 0.05) increase on the caspase-3 when compared with control.
Pro-inflammatory markers of rats treated with BP extract following HMM exposure
The effects of BP extract on the thyroidal pro-inflammatory markers (IL-6 and TNFα) of HMM exposed female rats are shown in Figure 5. HMM exposure resulted in a significant increase in TNF –α and IL - 6 levels in the thyroid when compared with the control. However, co-treatment with BP extract significantly decreased the TNF – α and IL – 6 levels in comparison to both the control and HMM groups in a dose dependent manner. HMM only exposed rats had significant increase the thyroidal pro-inflammatory markers (IL-6 and TNFα) when compared with control (Figure 5). Effect of banana peel on the Pro-Inflammatory markers Interleukin-6 (IL-6) and tumor necrosis factor (TNF-α) in the thyroid of heavy metal mixture exposed female albino rats. Values with different superscripts (a, b, c) were significantly different from each other (p < 0.05) and those with the same superscripts were not significantly different.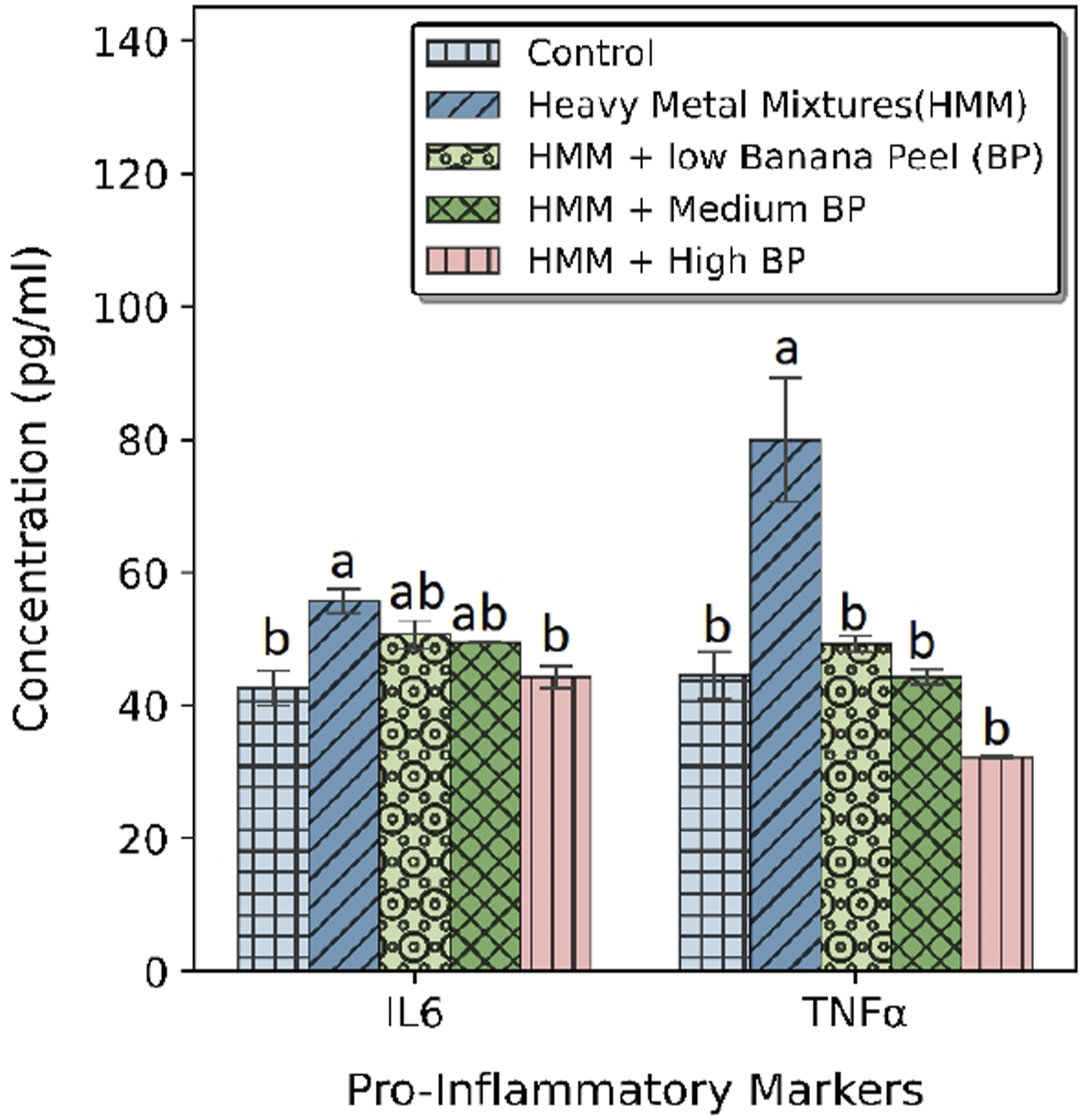
Nrf2, NFκB and Hmox-1 activation in the thyroid of rats treated with BP extract following HMM exposure
The effects of BP extract on the thyroidal NFκB, Nrf2 and Hmox-1 of HMM exposed female rats is shown in Figure 6. The 200 mg/kg BP extract group showed significantly (p < 0.05) higher Hmox-1 level in comparison to the control and HMM only groups (Figure 6(a)). HMM only exposure caused increase in Nrf2 level. This increase was significant (p < 0.05) in comparison to control, and BP extract treated groups. The 200 mg/kg BP extract group showed higher Nrf2 level in comparison to the control groups (Figure 6(b)). There was significant increase in the NFκB level of the HMM only group when compared with the control group and 400 and 800 mg/kg BP extract groups. The 400 and 800 mg/kg BP extract groups showed significant (p < 0.05) decrease in the NFκB levels. HMM only exposed rats had significant (p < 0.05) increase the thyroidal Hmox-1, Nrf2 and NFκB when compared with control. Effect of banana peel on the Transcription factors (NFκB, and Nrf2) and Hmox-1 in the thyroid of heavy metal mixture exposed female albino rats. Values with different superscripts (a, b, c) were significantly different from each other (p < 0.05) and those with the same superscripts were not significantly different.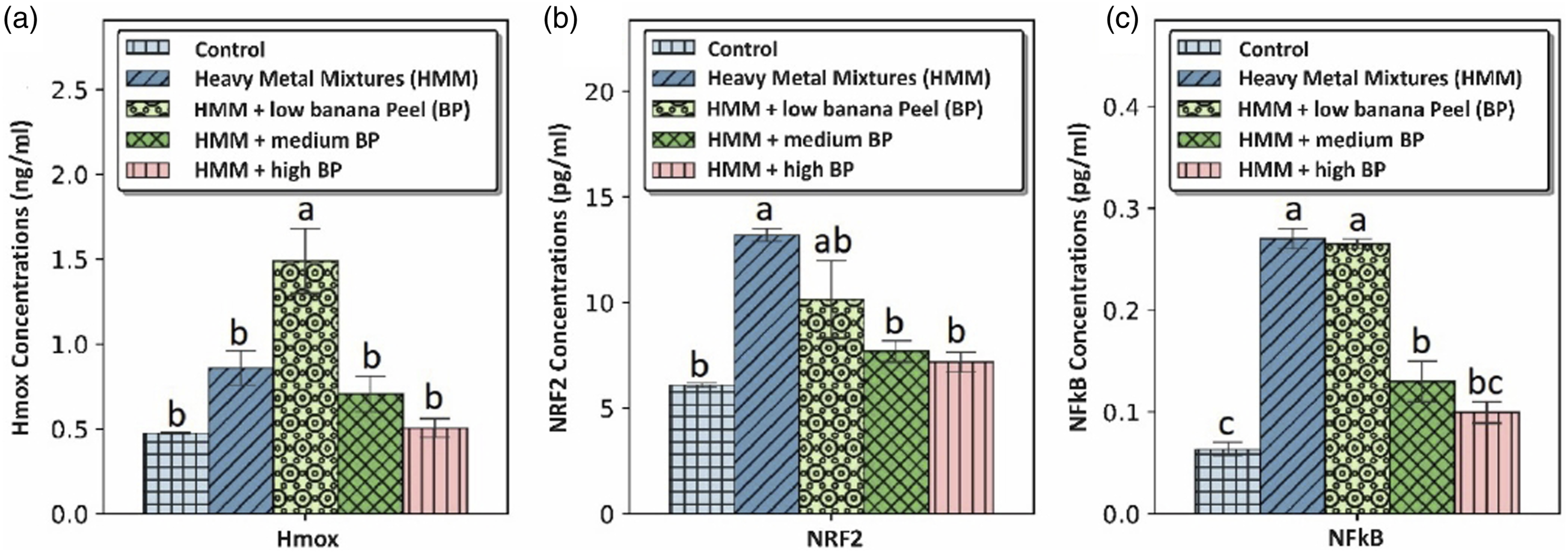
Relationship between biomarkers using Pearson R correlation
Relationship between oxidative stress, antioxidants, pro-inflammatory marker, apoptotic marker and transcription factors in the thyroid.
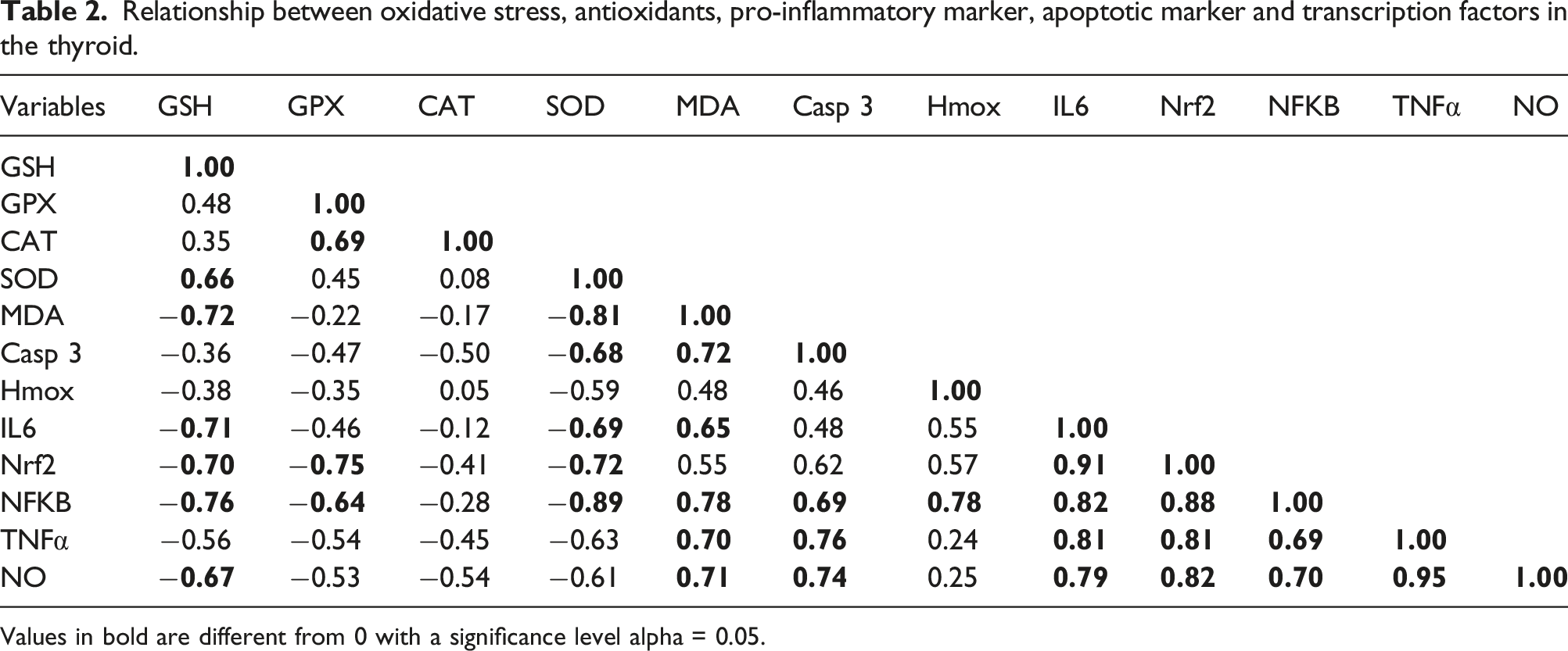
Values in bold are different from 0 with a significance level alpha = 0.05.
Histology of the thyroid of rats treated with BP extract following HMM exposure
The histology of representative rats from each group used in this study are shown in Figure 7(a)–(e). The photomicrograph of the thyroid gland tissue of a rat in the control group showed normal thyroid follicles and normal follicular cells (FC), Follicular cells colloid (FCC), Connective tissues (CT) and Para follicular cells (PFC) (Figure 7(a)), while the toxicity control (HMM alone) showed increased cellularity (IC) of thyroid follicles with monomorphic cells forming small follicular structures that are filled with colloid (Figure 7(b)). The test groups co-treated with low dose BP and HMM showed increased cellularity of thyroid gland, chronic inflammatory cells (CIC), thyroid colloid and thyroiditis (Figure 7(c)), while medium dose BP and HMM showed collections of CIC with C-Cells hyperplasia (CCH) and increased cellularity (ICC) (Figure 7(d)) and groups treated with high dose BP and HMM showed thyroid colloid (TC), irregular, anisotropic calcium oxalate crystal and ruptured thyroid follicles (RTF) causing granulomatous reaction and thyroiditis (Figure 7(e)). (a): Photomicrograph of the thyroid gland tissue of rats in the control group. Group 1 (Control): (H&E ×400). Micrograph of normal thyroid gland with inactive thyroid follicles (ITF) containing colloid bounded by simple squamous epithelium. There is visible parafollicular cells (PFC) or C-cells. There is also a connective Tissue Septa (CTS) which divides the gland into globules. (b): Photomicrograph of the thyroid gland tissue of rats administered HMM only. Group 2 HMM only (H&E ×400). Micrograph showing extensive inflammatory cell infiltration with Atrophic Inactive Follicles (black arrows) and Active follicles (yellow arrows). There is decreased number of thyroid follicles which are generally atrophic and are often devoid of celluloid. The follicular epithelial cells are transformed into their degenerated state termed Hurthle cells (also called as Askanazy cells, or oxyphil cells or, oncocytes). (c): Group 3 HMM treated with BP 200 mg/kg (H&E ×400). Micrograph showing involution stage of thyroid follicle; there are large distended inactive follicles (arrows) bounded by colloid and lined by flattened follicular epithelium. (d): Group 4 HMM treated with BP 400 mg/kg (H&E ×400). Micrograph showing involution stage of thyroid follicle with extensive inflammatory cell infiltration; there are large distended inactive follicles (arrows) by colloid. (e): Group 5 HMM treated with BP 800 mg/kg (H&E ×400). Micrograph showing fatty degeneration of active follicles (arrows) with moderate inflammatory cell infiltration of the interstitium.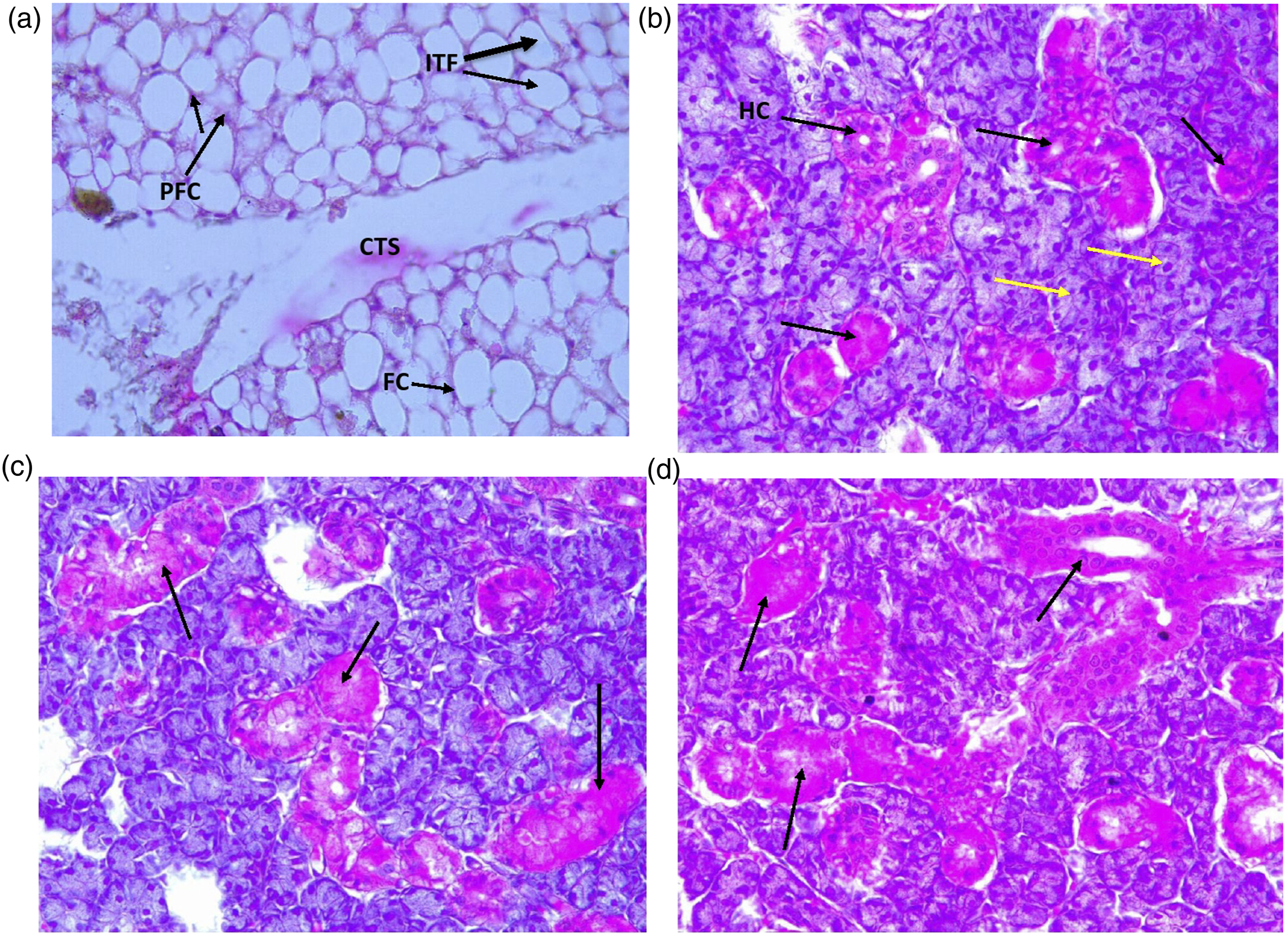
Discussion
This study has demonstrated an appreciable attenuation of heavy metal-induced dysfunction of the thyroid, oxidative stress, proinflammation and apoptosis, by BP extract, suggestive of ameliorative and beneficial effects of BP extract. Concurrent exposure to different pollutants can trigger the toxicity of individual components. 38 This study has demonstrated that exposure to HMM provoked oxido-inflammatory process in the thyroid gland in Sprague Dawley female rats. 39
This study has demonstrated that the thyroid gland can sequester heavy metals. The thyroid gland cloistered substantial amounts of Pb, Al, Hg and Mnma which caused histopathological alterations. Treatment with BP extracts caused significant reduction of the accumulated Pb, Al, Hg and Mn in the thyroid of female rats. This reduction of thyroidal Pb, Al, Hg and Mn accumulation was remarkable and suggest that BP extracts could have enhanced the excretion of the absorbed metals evinced by high fecal matter Pb, Al, Hg and Mn levels (Table not shown) which agree with previous findings of Anyanwu et al. 40
Nrf2 can be induced injuriously by ROS 41 or innocuously by natural antidotes.42,43 On exposure to oxidants or nutraceuticals, the cysteine moieties on the Keap1-Nrf2 complex experience cellular redox changes, which lead to conformational changes of Keap1, nuclear translocation of Nrf2, and synthesis of electrophile response element or antioxidant response element ARE 44 and eventual induction of transcription of Phase II antioxidant enzymes. In this study, BP extract boosted hmox-1 and blunted Nrf2 expressions in the thyroid of heavy metal mixture - exposed rats, suggesting that the attenuation of the pernicious Nrf2 expression may be one of the main protective mechanisms of BP extract.
This study also investigated the effects of BP extract on oxidative stress and apoptosis of HMM -exposed thyroid. Oxidative stress, marked by lopsided pro-oxidation machinery, resulted in thyroid damage via amplification of lipid peroxidation and decrease of antioxidant enzyme activity. 45 This study showed that BP extract decreased the MDA (lipid peroxidation product and biomarker of oxidative stress) level but increased antioxidant enzyme activities (SOD and CAT) of thyroid of rats, exposed to HMM.46,47 BP extract may not only scavenge free radicals but may also improve antioxidant enzyme activity to resist oxidative stress as seen in other poly phenolic nucleophilic compounds.48,49
The increase in caspase-3 activity in the HMM-exposed rats which suggest the activation of the thyroidal apoptotic pathway is in agreement with other studies that have implicated the involvement of caspase 3 dependent cascades in the heavy metal induced apoptosis in different tissues.50–54 On the contrary, BP tended to abrogate the HMM-induced apoptosis.
Thyroids possess an antioxidant enzymatic machinery which hinder generation of free radicals and their attendant deleterious effect. The first line of enzymatic antioxidant defense system (super oxide dismutase SOD) responsible for the catalytic conversion of superoxide radicals to less innocuous molecule H2O2, which is eventually converted to water by CAT. 55 Exhaustion or saturation of this mechanism activates the second line of antioxidant enzyme defense, predominantly mediated by GPx. 56 GPx are cytoprotective against oxidative stress and lipid peroxidation, detoxification of H2O2 and lipid peroxides into less reactive species, using reduced glutathione GSH as a substrate. 57
In mammals, the overexpression of NO following exposure to any xenobiotic has been reported to damage the antioxidant system.58,59 Elevated levels of ROS and NO have been established to trigger diverse inflammatory response.58,59 BP extract markedly decreased the NO levels in the thyroid, supportive of cytoprotective and antioxidant mechanism. In the present study HMM mediated upregulation of pro-inflammatory cytokines IL-6 and TNF-α in the thyroid were significantly decreased by both the BP extract treatment. According to Diez et al., 60 TNF – α plays a vital role is the expression of other cytokines involved in thyroid disorders, so it could have also triggered the over expression of IL – 6 and other cytokines in HMM treated rats as have been reported by previous researchers. 61 Plant-Derived Food Grade Substances PDFGS have consistently demonstrated anti-inflammatory activities. 62
Inflammatory response is usually amplified by oxidative stress through the modulation of associated genes.63,64 Oxidative stress provokes the activation of numerous transducers and redox sensitive transcription factor. The amplification of thyroidal nuclear transcription factor (NF-kB) following exposure to HMM is an evidence of oxidative stress mediated inflammation which has been antagonized by BP extract. 65
The rats in the control group showed normal histological configuration in the thyroid section. The follicles at the boundary of the gland had few small peripheral vacuoles and were bigger than the ones at the central follicle with more colloids. 66 The small and centrally dispersed follicles are responsible for the secretion of thyroid hormone while the follicles at the periphery predominantly serve as a pool of old hormones. 67 In consonance with earlier reports on thyroid histology, the thyroid sections of the rats in HMM treated group showed histological alterations in comparison to the control group. The observed increase in the cellularity of thyroid follicles with monomorphic cells forming small follicular structures that are filled with colloid were in agreement with previous works on metals induced hypothyroidism. 68 In the lumen, numerous follicles become degenerated with exfoliated epithelial cells, coalesce, collapse with distorted walls. Also, dark pyknotic nuclei and the cytoplasmic vacuolations observed in the follicular cells were in affirmation with those reported by Aboul-Fotouh et al. 68 The observation that the BP extract did not show visible protection against HMM mediated histopathological alterations in the thyroid of female rats may be due to the duration of the present study will require further investigation.
Taken together, heavy metals accumulated in the thyroid, elicited histoarchitectural distortions, alter cytokine expression with upregulation of the pro-inflammatory markers. 69 These cascades were mitigated by BP extract.
In conclusion, BP extract may provide appreciable protective effect against HMM -induced toxicity by inhibiting oxidative stress and apoptosis associated with modulation of the pernicious expression of Nrf2. These results may hold promise as a new strategy for protecting against heavy metal-induced toxicity, create a use for BP which is hitherto considered a waste to boost circular economy.
Footnotes
Acknowledgements
Boma Eddie-Amadi of World Bank Africa Centre of Excellence in Oilfield Chemicals Research (ACE-CEFOR), University of Port Harcourt received tuition waiver from CEFOR, Mr Moses, Department of Anatomy, University of Port Harcourt anchored the dissection and tissue processing, and Engineer Nelson Oyenike handled data analyses.
Author Contributions
BFE-A bench work, analyses of data, drafting of manuscript; ANE bench work and supervision; CNO bench work; AnC and AlC drafting and revision of manuscript, OEO analyses of data, manuscript writing and conceptualization.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The ethical approval was obtained from the University of Port Harcourt institutional Centre for Research Management and Development Animal Care and Use Research Ethics Committee (UPH/CEREMAD/REC/18).

