Abstract
A safety assessment for β-galactosidase derived from
Introduction
The
Although
Materials and methods
Test material
The test article is GODO-FAL, produced by GODO Shusei Co., Ltd, Japan (GODO-FAL). The production process of GODO-FAL involves the fermentation of
Chromosome aberration test in cultured Chinese hamster cells
A mammalian chromosome aberration test was performed in CHL/IU cells derived from the lung of a female Chinese hamster in accordance with OECD 473 (2014) and in compliance with Good Laboratory Practice (GLP). The CHL/IU cells were purchased from DS Pharma Biomedical Co., Ltd (Osaka, Japan) and used at passage numbers from 14 to 22. The cells were negative for mycoplasma. The cells were cultivated in a 60-mm culture plate in a carbon dioxide (CO2) humidified incubator set at 5% CO2 and 37°C. The culture medium for this assay was Eagle’s minimal essential medium (MEM) liquid medium (lot no: DSG7016; Wako Pure Chemical Industries, Ltd., Osaka, Japan) with 10% inactivated fetal bovine serum (lot no: AZM197211; HyClone) and 1% penicillin/streptomycin (lot no: 1786393; GIBCO, Grand Island, New York, USA).
A cell growth inhibition test was conducted according to OECD 474, with the following concentrations: 125, 250, 500, 1000, and 2000 µg/mL, to determine the dosages used for the main study. Cell growth inhibition was measured by measurement of cell proliferation rate (relative population doubling (RPD)). RPD was calculated by the following formula:
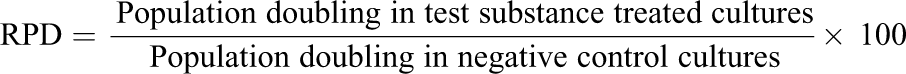
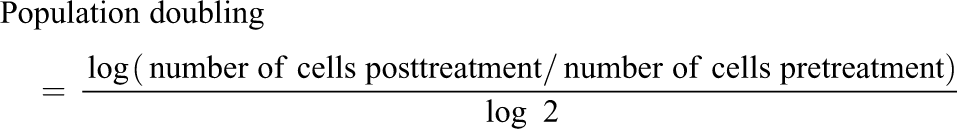
For both the cell proliferation assay and the main chromosome aberration assay, 5 mL of cell suspension at 4 × 103 cells/mL was seeded to a plate and cultured for 3 days.
Results of the cell growth inhibition test are shown in Supplemental Table S1. No cell growth inhibition exceeding 50% was observed in any test substance treatment groups; therefore, 2000 µg/mL was selected as the highest concentration and a total of four concentrations were prepared using a common ratio of 2.
Short-term tests with and without S9 metabolic activation were conducted after 6 h of treatment with GODO-FAL. Rat liver S9 was produced by Oriental Yeast Co., Ltd (Tokyo, Japan), and stored at −80°C until use. After 6 h of treatment, the cells were washed with Dulbecco’s phosphate buffer saline (pH 7.1), and 5 mL of fresh culture medium was added to the plate. The cells were further cultured for 18 h. Continuous treatment tests with and without S9 metabolic activation were conducted after 24 h of treatment with GODO-FAL. Sterile water was used as the negative control. The positive control without metabolic activation was 0.05 µg/mL mitomycin C (MMC; lot no: 577AEE; Kyowa Hakko Kirin Co., Ltd, Tokyo, Japan). The positive control with metabolic activation was 5.0 µg/mL cyclophosphamide (CP; lot no: MKBS0021V; Sigma-Aldrich Inc., St. Louis, Missouri, USA) added to culture medium at 1% volume. Two hours prior to the preparation of specimens, the cultures were treated with 0.2 µg/mL colcemid (GIBCO, Grand Island, New York, USA). The cells were separated with trypsin solution and then centrifuged. The collected cells were resuspended in hypotonic solution (0.075 M potassium chloride), and then Carnoy’s fixative (methanol:acetic acid, 3:1) was added. Fixing procedures were repeated three times. The fixed cells were dropped onto a slide and air-dried and stained with 2% Giemsa solution. Duplicate slides were prepared for each plate.
Analysis for chromosome aberration was performed in three test substance concentrations from the highest concentration. For structural chromosome aberrations, 300 well-spread metaphase cells in total per concentration (150 metaphase cells per plate) were observed under a microscope at a magnification of 1000. For numerical aberrations, 400 well-spread metaphase cells in total per concentration (200 metaphase cells per plate) were observed under a microscope at a magnification of 200. The chromosome aberrations were classified as shown subsequently. The cells with structural aberrations excluding gap (−gap) and including gap (+gap) were separately totaled. The frequency of the cells with structure aberrations excluding gap (−gap) was used for evaluation of chromosome aberrations.
Results would be considered positive for chromosomal aberrations if the test substance-treated samples were statistically significantly increased (the
Micronucleus test
The micronucleus test was performed using an OECD-compliant protocol (OECD 474, 2014) in 8-week old Crl: CD(SD) male rats in compliance with GLP. Six animals each in the test substance groups given negative control (water for injection) 500, 1000, or 2000 mg/kg daily of GODO-FAL for 2 days via oral gavage. The positive control (CP; Sigma-Aldrich Inc., St. Louis, Missouri, USA, lot no: MKBS0021V, 20 mg/kg) was administered via one intraperitoneal injection on the second day. All animals were observed for clinical signs daily, and body weights were measured on administration day and on the day of euthanization. Animals were terminated and specimens collected 18–24 h after the final administration of the test substance. Bone marrow cells in the femur were washed with fetal bovine serum. Excess serum was removed from the bone marrow cells, and the cells were smeared onto three slides/animal. The cell-smeared specimens were dried at room temperature, fixed with methanol for 4 min, and stained with 0.007% acridine orange stain. The slides were washed twice with phosphate buffer solution (1/15 M, pH 6.8) and allowed to dry. Two specimens per animal were observed with a fluorescence microscope at a magnification of 1000 at random; 4000 immature erythrocytes/animal were examined, and the frequency of micronucleated immature erythrocytes was calculated; 1000 erythrocytes/animal were observed and the ratio of immature erythrocytes was also calculated.
The Kastenbaum and Bowman statistical analysis method was used to evaluate the frequency of micronuclei between the negative control group and test substance and positive control groups. Dunnett’s test was used to evaluate differences in body weight and frequency of immature erythrocytes between the negative control group and other test substance groups.
Reverse mutation test (Ames assay)
The reverse mutation test (Ames assay) was performed in accordance with OECD 471 (1997) and compliance with GLP. (2-(2-Furyl)-3-(5-nitro-2-furyl) acrylamide (Wako Pure Chemical Industries, Ltd; lot no: SAE0315), sodium azide (Wako Pure Chemical Industries, Ltd., Osaka, Japan; lot no: JPG7700), 9-aminoacridine, (Sigma-Aldrich; lot no: BCBK1177V), and 2-aminoanthracene (Wako Pure Chemical Industries, Ltd., Osaka, Japan; lot no: DCK3519) were used as positive controls. Each positive control was dissolved in dimethyl sulfoxide (DMSO). All strains of
The mutagenicity of GODO-FAL was determined using the preincubation method, with and without metabolic activation (S9, as described in the chromosome aberration test). GODO-FAL was diluted in water at 313, 625, 1250, 2500, and 5000 µg per plate incubated with strains of
Two statistical analyses of Dunnett’s multiple comparison method (one-side test) and linear regression method were used.
The number of revertant colonies for each bacterial strain and concentration in the dose-finding study and main study was compared with that of the negative control in both the presence and the absence of metabolic activation, and statistically significant difference in the number of revertant colonies between those two groups was analyzed by multiple comparison method (
Acute oral toxicity study
This test was performed in accordance with OECD 420 (2001) and compliance with GLP. Five 5-week-old female Sprague–Dawley (SD) rats were acclimatized and monitored for abnormalities and clinical signs for 3 weeks. Each animal was housed individually. The rats were provided feed and water ad libitum, except for fasting the evening before administration of GODO-FAL; 2000 mg/kg GODO-FAL was administered by oral gavage and the rats were then monitored for 14 days. Mortality and clinical signs were observed before administration; 30 min; and 1, 2, 4, and 6 h after administration once a day for the 14 days following administration. Body weight was monitored on days 0 (before administration), 1, 2, 4, 7, and 14. Animals were euthanized and necropsies performed on day 14.
Twenty-eight-day repeated oral toxicity study
This test was performed in accordance with OECD 407 (2008), with the following exceptions: detailed functional observations were not recorded, and the following organs were collected but not subjected to histopathology: spinal cord, eye, thyroid, trachea, gonads (testis and ovaries), accessory sex organs (uterus and cervix, epididymides, prostate + seminal vesicles with coagulating glands), vagina, urinary bladder, peripheral nerve, skeletal muscle, bone, and bone marrow. Twenty male and 20 female 6-week old SD rats were acclimatized and monitored for abnormalities and clinical signs for 10 days prior to GODO-FAL administration. An ophthalmologic examination was also performed during the acclimatization period. Each animal was housed individually. The rats were provided feed and water ad libitum.
The rats were divided into groups of five animals/sex for each dose of GODO-FAL: 0, 500, 1000, and 2000 mg/kg/day administered by oral gavage for 28 days. Mortality and clinical signs were observed twice daily (before and after administration of GODO-FAL) and before necropsy. Body weights were recorded on the first day of GODO-FAL administration and weekly during the administration period. Rats were weighed the day of necropsy, and this body weight measurement was used for calculation of the relative organ weight. Food consumption was measured (food intake per day) by the amount of food given and food remaining. Ophthalmologic examinations were performed during week 4 of dosing. Urine was collected using a urine funnel during week 4 of dosing, and the following parameters were analyzed: pH, protein, glucose, ketone bodies, urobilinogen, bilirubin, occult blood, sediments, color, volume, specific gravity, sodium (Na), potassium (K), and chloride (Cl).
All animals were fasted the night before euthanization and necropsy. Blood was collected from the abdominal aorta of all animals under isoflurane anesthesia at necropsy after the measurement of body weight, and the following hematological parameters were analyzed: red blood cell count, white blood cell count, hematocrit value, hemoglobin content, mean corpuscular hemoglobin, mean corpuscular volume, mean corpuscular hemoglobin concentration (MCHC), reticulocyte count, platelet count, prothrombin time (PT), activated partial thromboplastin time, and differential leukocyte count. The following clinical chemistry parameters were also analyzed from serum collected at necropsy: aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, lactate dehydrogenase, γ-glutamyl transpeptidase, glucose, total cholesterol, triglyceride, phospholipid, total protein, albumin, albumin/globulin ratio (A/G), urea nitrogen, creatine, total bilirubin (T.Bil.), Na, K, Cl, inorganic phosphorus, and calcium (Ca). At necropsy, the organs were weighed and fixed as described subsequently. The pituitary gland and thyroid gland were fixed in 10% neutral-buffered formalin and then weighed on the day after necropsy. Paired organs were weighed together. The following organs were weighed and fixed: brain, heart, thymus, spleen, lung (including bronchus), submandibular glands, liver, kidneys, prostate, seminal vesicle, testes, epididymis, ovaries, uterus, pituitary gland, thyroid glands (including parathyroid gland), and adrenal glands. The eyes, including the optic nerve, were fixed in Davidson’s fixative, the testes were fixed in Bouin fixative, and other organs and tissue were fixed in 10% neutral-buffered formalin. Bone tissues were decalcified with 10% formic acid formalin. For all rats of the control and high dose groups, the following fixed organs were embedded, thin sectioned, stained with hematoxylin and eosin (H-E), and examined microscopically: brain, heart, thymus, spleen, lung, liver, kidney, adrenal gland, stomach, duodenum, jejunum, ileum, cecum, and colon. Statistical analysis for homogeneity of variance was performed by Bartlett’s test (significant level: 5%). Dunnett’s multiple comparison test (significant level: 5%, two-tailed) was used for homogeneous data, and Steel’s multiple comparison test (significant level: 5%, two-tailed) was used for heterogeneous data.
Subchronic toxicity study in rats
A 90-day subchronic toxicity study was performed in 6-week old male and female SD rats in compliance with GLP. The study was performed in compliance with OECD 408 (1998), with the exception that only one dose of GODO-FAL was used in addition to the control (water). Rats were housed individually during quarantine, acclimation, and the study period. Prior to dosing, the rats were randomized by body weight into two groups (
Results
Chromosome aberration test
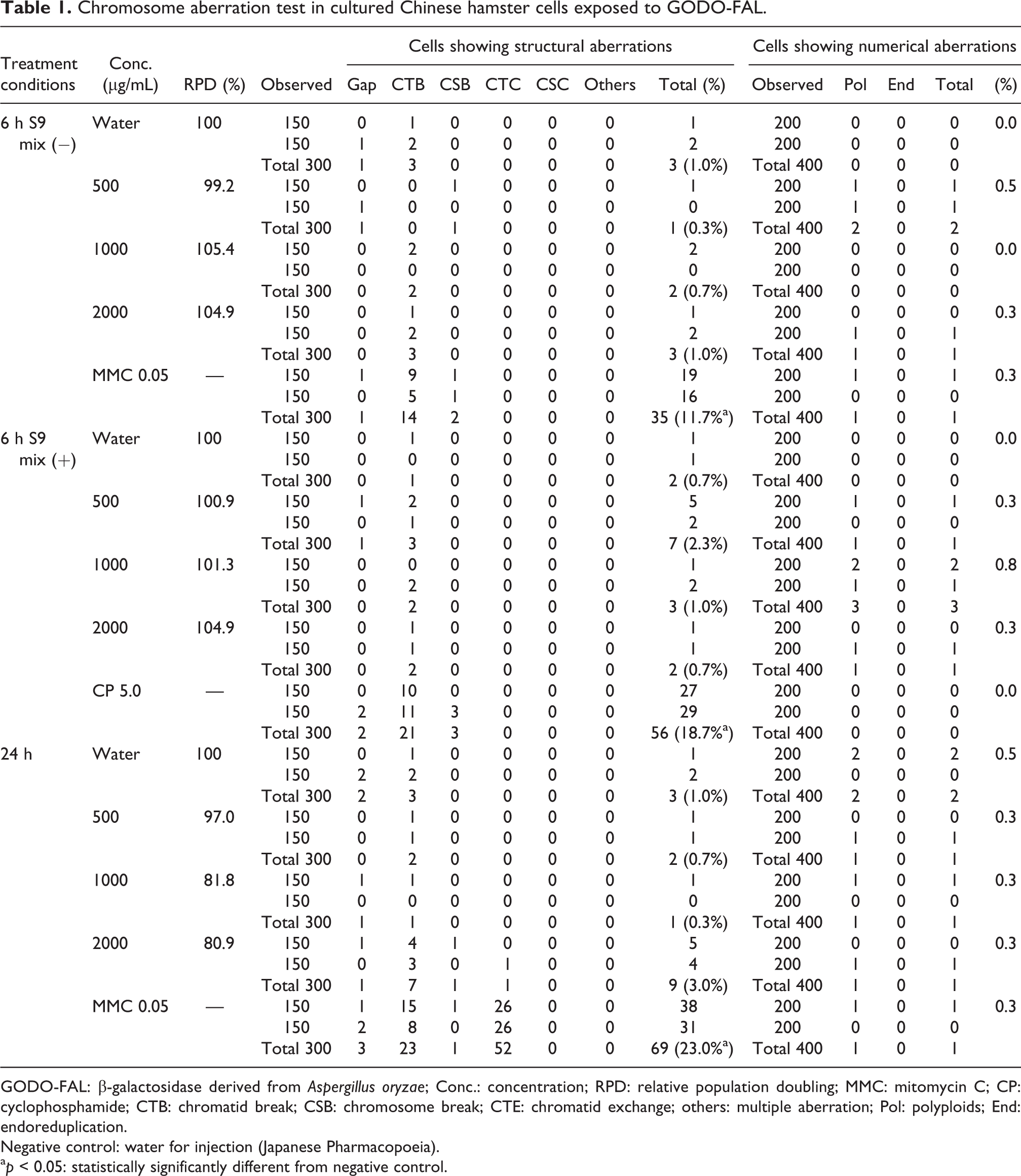
Exposure to GODO-FAL did not inhibit cell growth of CHL/IU cells at concentrations up to 2000 µg/mL, the highest dose used (see Online Supplemental Table S1). No statistical difference was observed between the negative control and any concentration of GODO-FAL in the frequencies of cells with structural aberrations and numerical aberrations in the 6-h or 24-h (Table 1) treatments. The frequencies of cells with structural aberrations in all positive controls were statistically increased compared with the negative controls, demonstrating the validity of the assay.
Chromosome aberration test in cultured Chinese hamster cells exposed to GODO-FAL.
GODO-FAL:
Negative control: water for injection (Japanese Pharmacopoeia).
a
The main chromosome aberration test was performed with short-term treatments with and without metabolic activation, and 24-h continuous treatment with 500, 1000, and 2000 µg/mL GODO-FAL. The frequencies of cells with structural aberrations and numerical aberrations in the GODO-FAL–treated cells were not statistically significantly different than the negative control. In contrast, the positive controls for the 6 and 24 h treatments with and without S9 activation were statistically significantly increased compared to the negative control. Based on these results, GODO-FAL did not induce chromosomal aberrations under the study conditions.
In vivo micronucleus assay
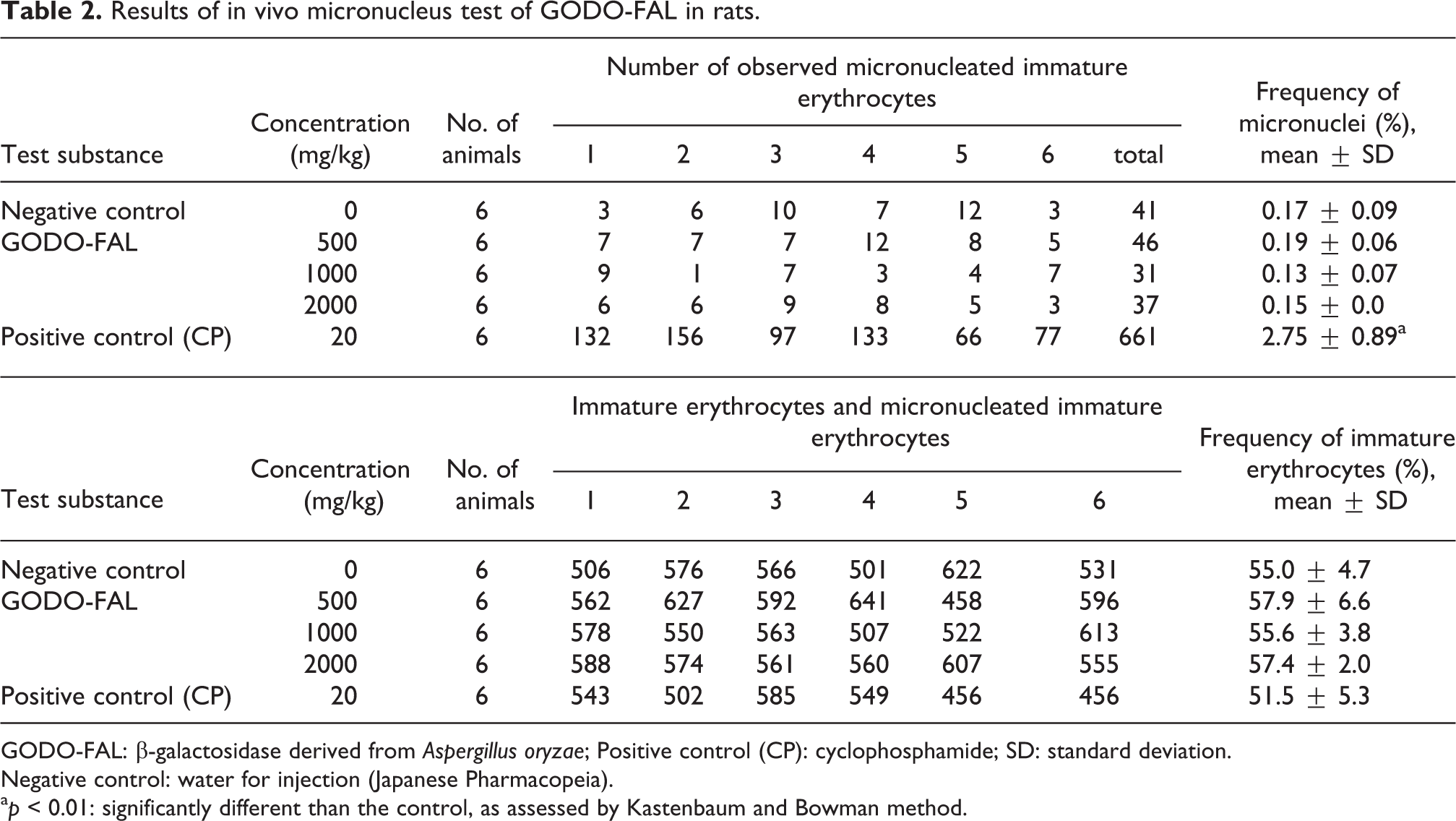
No abnormal clinical signs and no significant body weight changes were observed in any of the rats orally administered GODO-FAL for the in vivo micronucleus test. Clinical sign and body weight results are shown in Supplemental Tables S3 and S4. The frequency of micronuclei in GODO-FAL administered groups was not statistically different from the negative control (Table 2) and within the range of the background data of the negative control (see Supplemental Table S5). Conversely, the rats treated with the positive control had statistically increased frequency of micronuclei. The frequency of immature erythrocytes to total erythrocytes was not statistically different among the GODO-FAL groups compared to the negative control (Table 2), suggesting that bone marrow toxicity did not occur in this assay. Thus, GODO-FAL did not induce micronucleus formation under the study conditions.
Results of in vivo micronucleus test of GODO-FAL in rats.
GODO-FAL:
Negative control: water for injection (Japanese Pharmacopeia).
a
Reverse mutation test (Ames assay)
No precipitation or growth inhibition was observed in the GODO-FAL treated group. No statistically significant increase in the number of revertant colonies was observed in any of the GODO-FAL treatment groups compared to the negative control. The numbers of revertant colonies in the positive control were twice or more than those of the negative control in all bacterial strains in both the presence and the absence of metabolic activation, demonstrating the validity of the assay (Table 3). Based on these results, GODO-FAL was not mutagenic under the present study conditions.
GODO-FAL bacterial reverse mutation test.
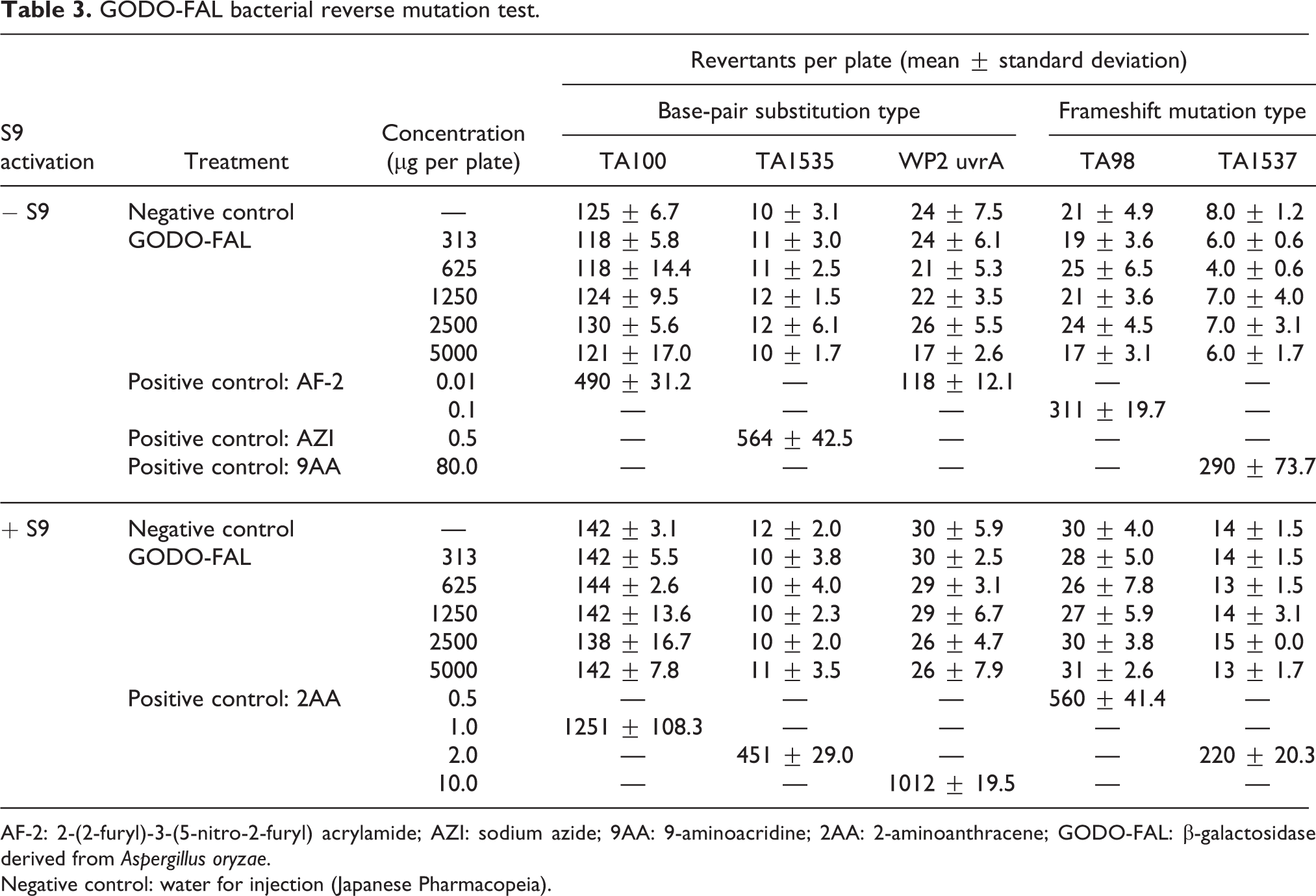
AF-2: 2-(2-furyl)-3-(5-nitro-2-furyl) acrylamide; AZI: sodium azide; 9AA: 9-aminoacridine; 2AA: 2-aminoanthracene; GODO-FAL:
Negative control: water for injection (Japanese Pharmacopeia).
Acute toxicity study
No deaths were observed after single oral administration of GODO-FAL at 2000 mg/kg in five female SD rats. No abnormal clinical signs were observed in any rats during the observation period. A decrease in body weight was observed in two rats on day 2 and one rat on day 7 (see Supplemental Table S6). These decreases were slight and may have been GODO-FAL administration related. No abnormalities were found during necropsy and gross pathology. Accordingly, the lethal dose of GODO-FAL in rats was determined to be over 2000 mg/kg.
Twenty-eight-day repeated oral toxicity study
In a 28-day dose-range finding study, 0 (control), 500, 1000, and 2000 mg/kg GODO-FAL was given to male and female SD rats for 28 days. No deaths or abnormalities were observed in any groups during the dosing period. A small, but statistically significant, decrease in food consumption was observed in males fed GODO-FAL at 1000 mg/kg/day on days 27–28 of the study (23 g compared to 28 g in the control). This change was not considered treatment related because there was no dose relationship and no decrease in food consumption was observed in the female rats. Additionally, there were no differences in body weights in the 1000 mg/kg fed male rats compared to control or to other treatment groups (data not shown). No differences in body weight were observed in female rats during the study.
Decreases in total urine excretion of Na, K, and Cl were observed in males at 500 and 2000 mg/kg during the last week of treatment. Decreases in total urine excretion of K and Cl were observed in males at 1000 mg/kg. These decreases were very slight and since no histopathological abnormality was observed in the kidney, and similar observations were not found in the female treatment groups, these changes were not considered to be related to treatment or toxicologically significant. No other differences were observed in urinalysis between groups.
Hematology parameters showed a statistically significant decrease in neutrophils (8.4% compared to 16.3% of leukocytes in the control,
Clinical chemistry results noted increases in gamma-glutamyl transpeptidase (0.5 U/L compared to 0.3 U/L in the control) and Cl (112 mEq/L compared to 110 mEq/L in the control) in males administered 2000 mg/kg/day GODO-FAL. These changes were very slight and were therefore not considered to have toxicological significance. No changes were noted in females from any treatment group.
One male administered 2000 mg/kg/day of GODO-FAL had an enlarged spleen and a small prostate. One male administered 500 mg/kg/day of GODO-FAL had an enlarged right-side adrenal gland. These gross pathology findings were considered incidental. No gross pathology abnormalities were found in any of the female groups.
The absolute and relative weights of the thymuses in females fed 500 mg/kg/day GODO-FAL were increased compared to the controls (data not shown). This increase was not considered treatment related as there were no changes in organ weights observed in male rats fed GODO-FAL, no dose-dependent relationship was observed, and there was no corresponding histopathology.
Histopathological examination in male rats fed 2000 mg/kg/day GODO-FAL for 28 days noted focal mononuclear cell infiltration in the liver. This finding was very slight and also observed in the control group and was not considered to be treatment related nor was it toxicologically significant. No other histopathological abnormalities were observed.
Based on these results, GODO-FAL was administered at 2000 mg/kg. Only one dose level was evaluated consistent with OECD repeated-dose study protocol guidance that allows for a limit test at one dose level if no observed adverse effects are produced at a level equivalent to 1000 mg/kg of body weight per day in other studies, and no toxicity is expected based on existing data for other related compounds.
Subchronic toxicity study
To evaluate the toxicity of GODO-FAL, doses of GODO-FAL at 0 (control) and 2000 mg/kg were given to SD rats for 90 days. No significant differences in body weights in males or females (Figure 1) or food consumed (Table 4) between the GODO-FAL and control groups were found during the dosing period. No differences in urinalysis parameters were observed between control and treated male or female rats (see Supplemental Tables S7 to S10).
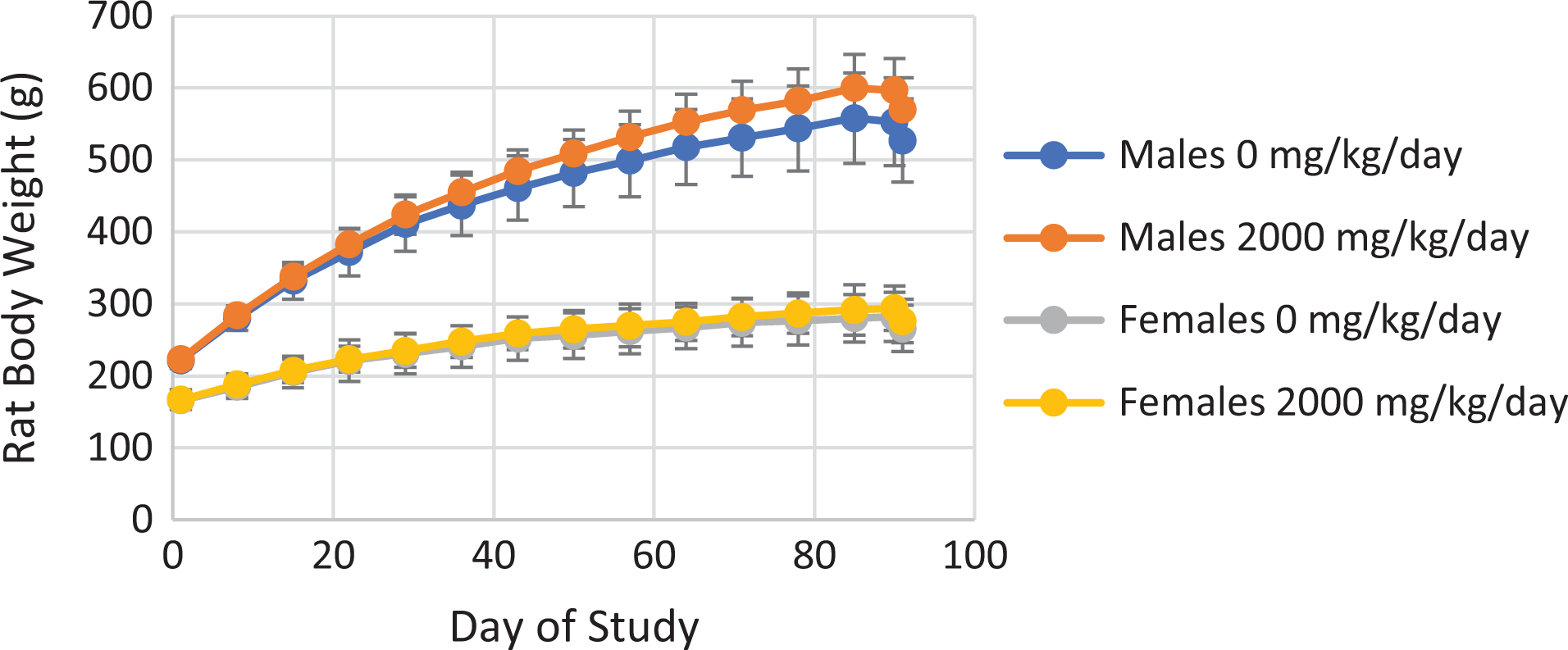
Rat body weight during subchronic toxicity study, treated with 0 or 2000 mg kg−1 day−1 GODO-FAL. Each point is the average of five rats/group with the standard deviations shown.
Food consumption (g/day) in the 90-day subchronic toxicity study.
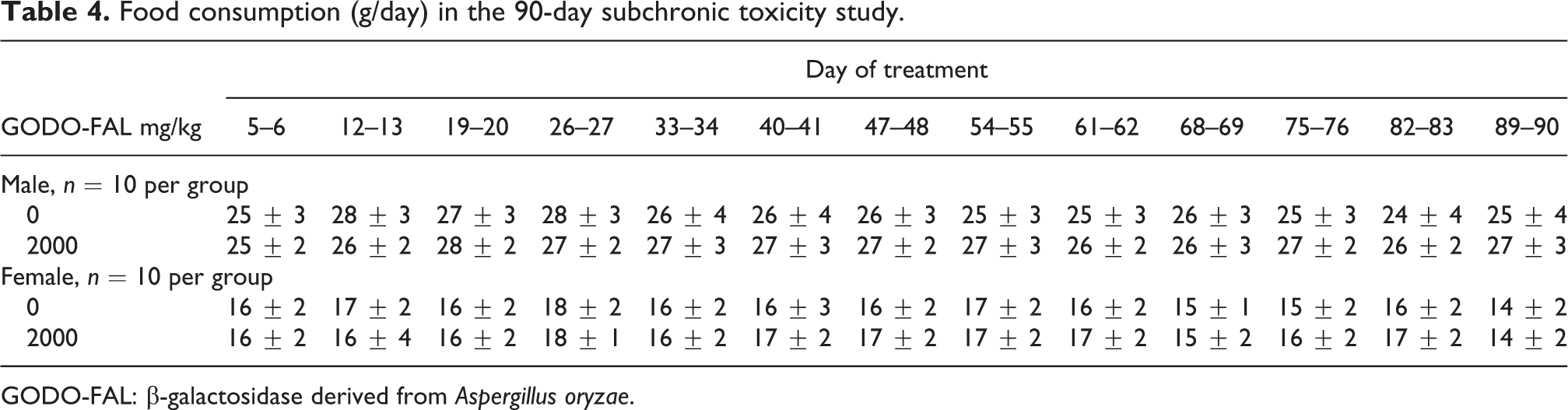
GODO-FAL:
Hematology analysis noted a decrease in PT in males, a decrease in MCHC, and increase in the absolute and relative numbers of reticulocytes (Ret) in females administered 2000 mg/kg/day GODO-FAL (Table 5). The PT in males fed GODO-FAL was statistically decreased when compared to the control (13.6 ± 0.7 vs. 14.5 ± 0.7 in the control) but was still within the historical control data range (mean: 14.4 ± 1.7, range: 11.5–21.5). MCHC was statistically decreased in females fed GODO-FAL (37.5 ± 0.5 vs. 38.1 ± 0.5) but was within the normal historical control data range for SD rats (mean: 37.3 ± 0.8, range: 35.4–39.1). Absolute and relative numbers of Ret were statistically increased in females fed GODO-FAL (absolute Ret in test group was 26.68 ± 4.10 vs. 22.06 ± 5.11 in the control; relative Ret in test group: 3.34 ± 0.56 vs. 38.1 ± 0.5 in the control) but was considered within normal historical control data ranges (mean absolute Ret: 25.30 ± 4.33, range: 15.94–39.55; mean relative Ret: 3.00 ± 0.52, range: 1.88–4.62). Although statistically different from control, these differences were very slight, not toxicologically significant and not considered test article related as the changes are within the historical control data for the testing facility.
Hematology results from 90-day subchronic toxicity study.
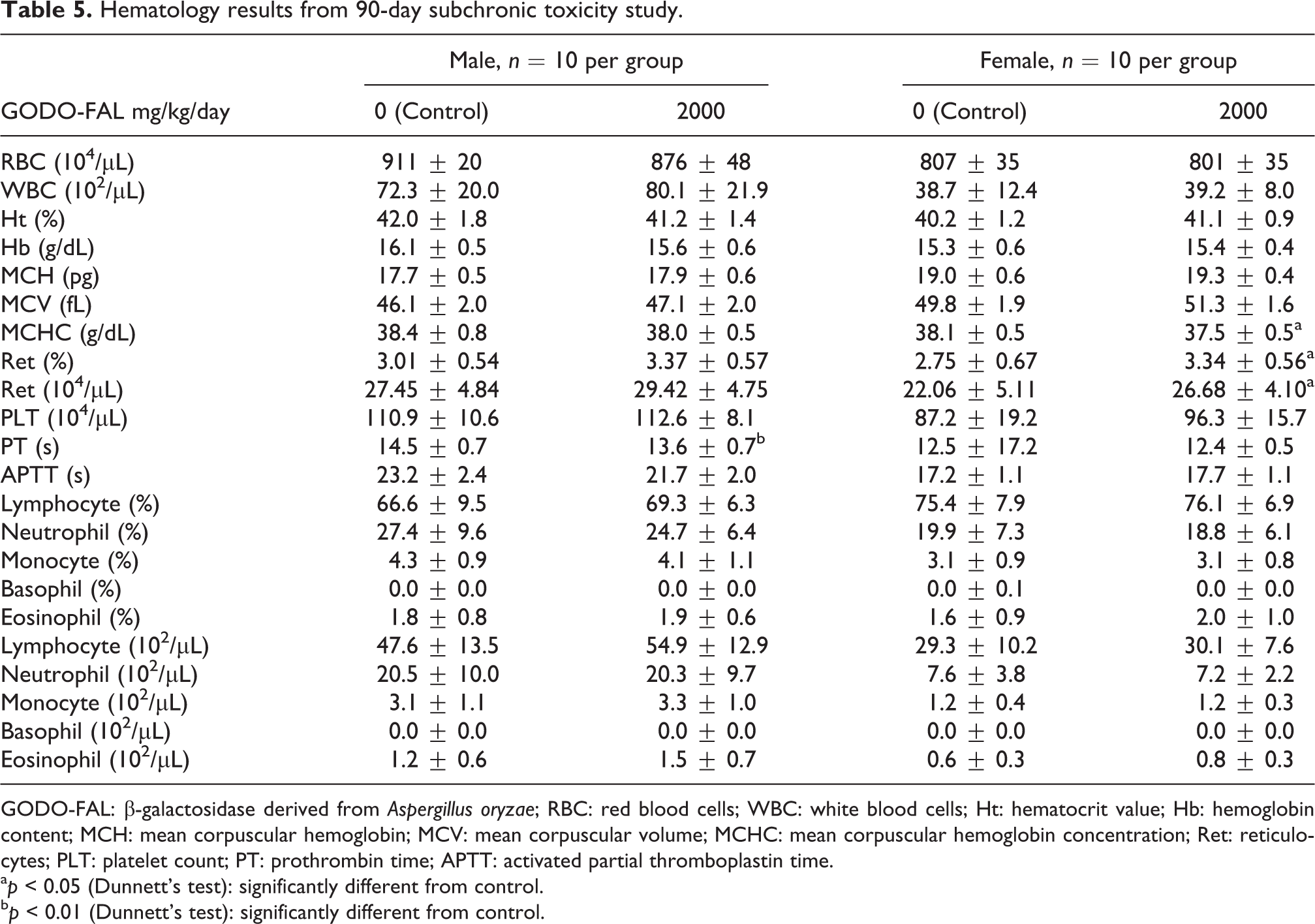
GODO-FAL:
a
b
Clinical chemistry results noted statistically significant increases in serum Ca in males, A/G, and T.Bil. in females fed 2000 mg/kg/day GODO-FAL (Table 6). The serum Ca in males fed GODO-FAL was statistically increased when compared to the control (10.1 ± 0.3 vs. 9.7 ± 0.3 in the control) but was still within the historical control data range (mean: 9.6 ± 0.4, range: 8.9–11.6). The A/G ratio in females fed GODO-FAL was statistically increased when compared to the control (0.78 ± 0.06 vs. 0.72 ± 0.04 in the control) but was still within the historical control data range (mean: 0.73 ± 0.07, range: 0.6–0.94). The females fed GODO-FAL also had a statistical increase in T.Bil. compared to control animals (0.10 ± 0.02 vs. 0.07 ± 0.01 in the control). This result was within the historical control data range (mean: 0.08 ± 0.02, range: 0.04–0.17). However, these changes were not considered related to administration of GODO-FAL because these changes were within the historical data for control animals at the testing facility.
Clinical chemistry results from the 90-day subchronic toxicity study.
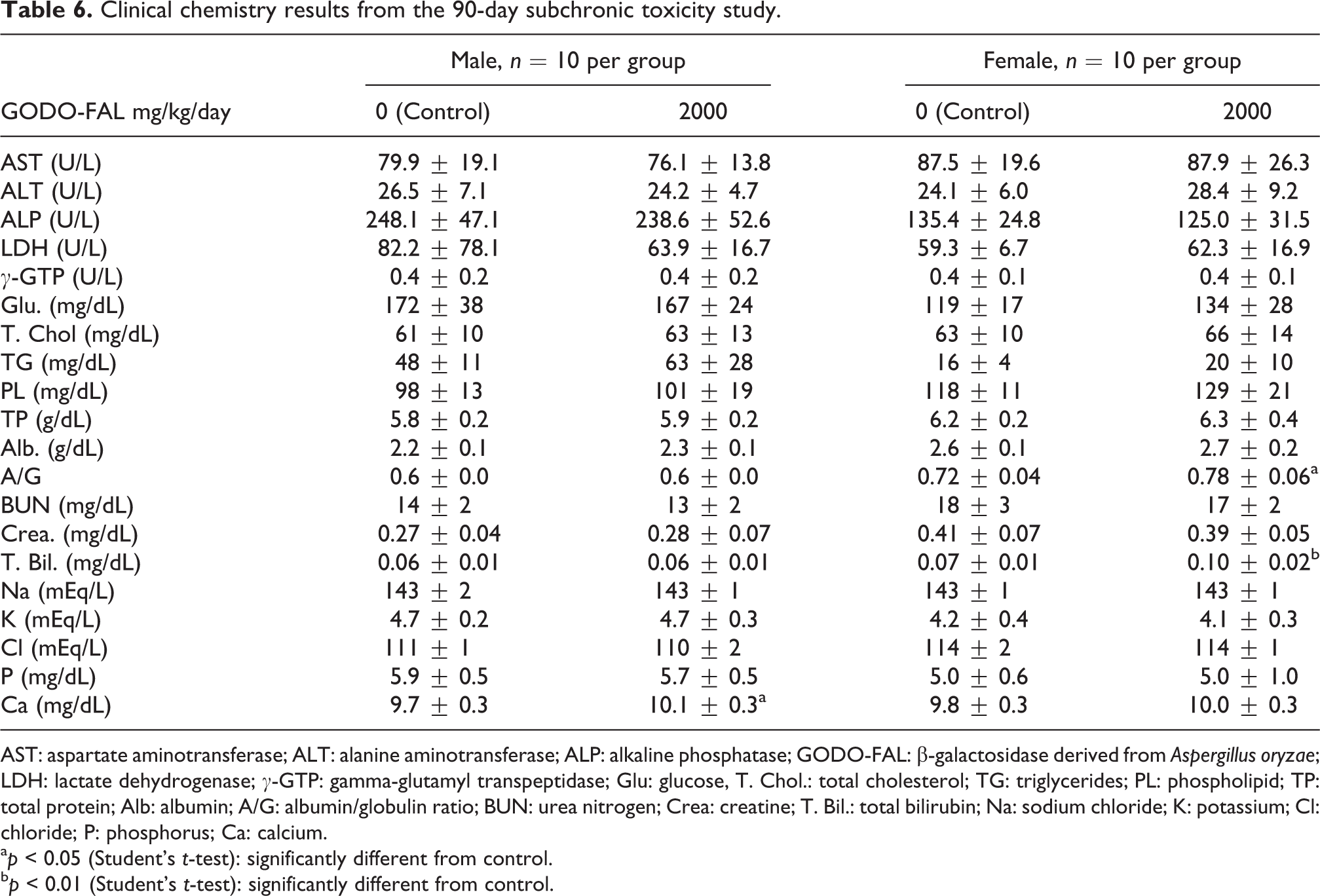
AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; GODO-FAL:
a
b
A dilated pelvis in the right kidney and cyst cervix in the uterus were observed upon gross pathology in one female administered 2000 mg/kg/day GODO-FAL for 90 days. These findings were considered to be incidental, as it was only observed in one animal and was therefore not test substance related.
A decrease of relative weight in the brain, testes, epididymis, and seminal vesicle in males and a decrease of relative weight in the kidney in females were observed in GODO-FAL–fed groups. Although these decreases were statistically significant, the decreases in organ weights were very small and well within the historical control data for SD rats from this facility measured over the preceding 5 years of studies and there was no corresponding histopathology. The relative brain weights in males fed GODO-FAL were statistically decreased when compared to the control (0.38 ± 0.03 vs. 0.41 ± 0.04 in the control) but were still within the historical data range (mean: 0.41 ± 0.4, range: 0.3–0.52). The GODO-FAL–fed males had a statistical decrease in relative testes weight (0.59 ± 0.04 vs. 0.64 ± 0.06 for controls), but this decrease was within the historical controls (mean: 0.64 ± 0.7, range: 0.45–0.80). Similarly, a statistical decrease in the relative weight of epididymides was observed between the GODO-FAL fed males and the controls males (0.21 ± 0.012 vs. 0.24 ± 0.019 in the control). This difference was within the range of expected epididymides relative weight (mean: 0.26 ± 0.034, range: 0.18–0.36). The relative weight of the seminal vesicles in the GODO-FAL–fed males was statistically smaller than the control (0.31 ± 0.054 vs. 0.37 ± 0.040 in the control) but still within historical data (mean: 0.38 ± 0.061, range: 0.25–0.61). The kidney relative weights in females fed GODO-FAL were statistically decreased when compared to the control (0.60 ± 0.02 vs. 0.65 ± 0.05 in the control) but were still within the historical control data range (mean: 0.66 ± 0.5, range: 0.56–0.76). None of these observed changes in relative organ weights were considered to be test substance-related as the changes were minimal, within the historical control data range for SD rats from 2012 to 2017 at the test facility, and there were no corresponding differences in the histopathology for any of these organs (Table 7).
Absolute and relative organ weight results in 90-day subchronic toxicity study.
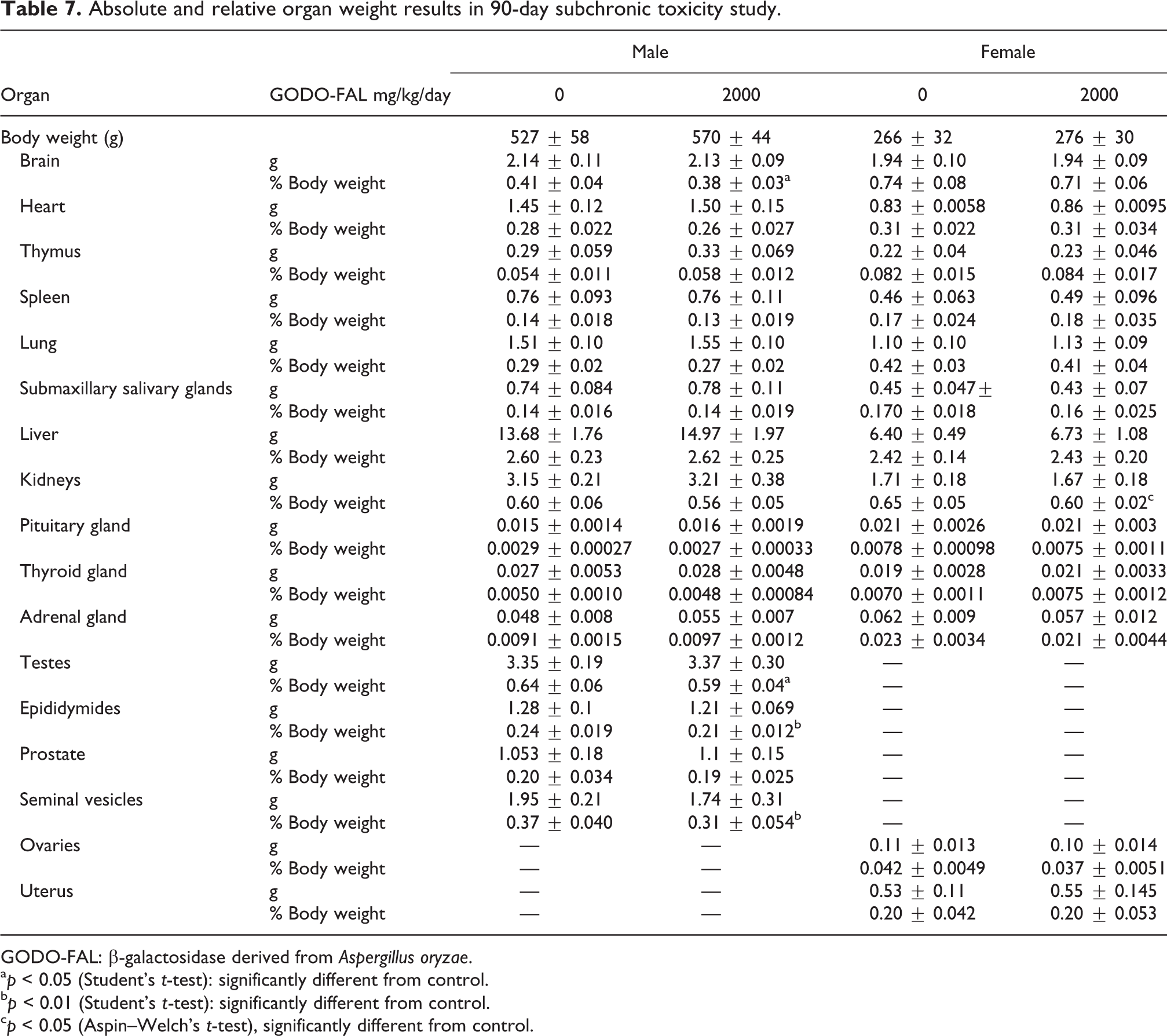
GODO-FAL:
a
b
c
Histopathological analysis found some slight (grade 1) pathological findings in the heart, liver, pancreas, and prostates in both treated and untreated males (Table 8). One female in the treated group had marked dilation of the right kidney, which was noted in the gross pathological findings (Table 8). These findings were considered incidental and not test substance related.
Histopathological findings in the 90-day subchronic toxicity study.a
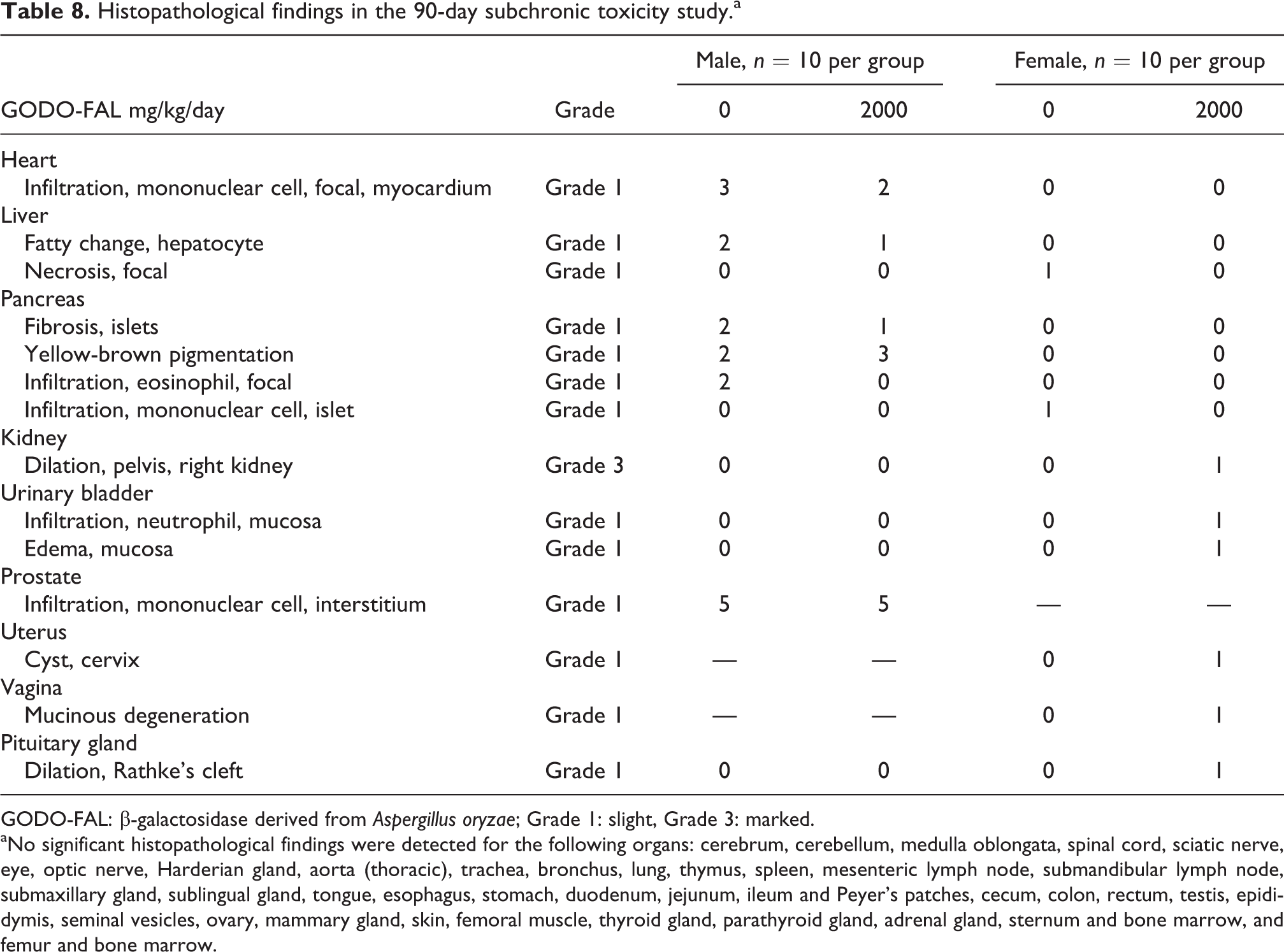
GODO-FAL:
aNo significant histopathological findings were detected for the following organs: cerebrum, cerebellum, medulla oblongata, spinal cord, sciatic nerve, eye, optic nerve, Harderian gland, aorta (thoracic), trachea, bronchus, lung, thymus, spleen, mesenteric lymph node, submandibular lymph node, submaxillary gland, sublingual gland, tongue, esophagus, stomach, duodenum, jejunum, ileum and Peyer’s patches, cecum, colon, rectum, testis, epididymis, seminal vesicles, ovary, mammary gland, skin, femoral muscle, thyroid gland, parathyroid gland, adrenal gland, sternum and bone marrow, and femur and bone marrow.
No significant treatment-related toxicologically changes were found in male or female rats administered 2000 mg/kg/day GODO-FAL. In conclusion, the NOAEL of GODO-FAL was determined to be at least 2000 mg/kg/day under the present study conditions.
Discussion
Although
GODO-FAL was non-mutagenic, non-clastogenic, and has a NOAEL of at least 2000 mg/kg/day (TOS 206 mg/kg/day) in a 90-day toxicology rat study. The results of these studies of GODO-FAL confirm the safety of this
Supplemental material
Supplemental_Tables - Toxicological assessment of β -galactosidase shows no adverse effects in vivo and in vitro
Supplemental_Tables for Toxicological assessment of
Footnotes
Acknowledgment
The authors acknowledge Kiyomi Miyauchi (Godo Shusei Co., Ltd) for enzyme analysis.
Author contributions
Jennifer Symonds and Claire Kruger contributed equally to the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jennifer Symonds and Claire Kruger are employees of Spherix Consulting Group, Inc. Spherix Consulting Group, Inc. is a private company that was mandated and financed by GODO Shusei Co., Ltd to write this manuscript. Tomohiro Fujita, Shouhei Aoki, and Kazuma Shiota are employees of GODO Shusei Co., Ltd, the company which mandated and funded the studies presented in this manuscript and preparation of this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
