Abstract
Quaternary ammonium compounds are antimicrobial chemicals that provide significant public health benefits by controlling bacteria and viruses that cause infections such as colds, flu, and COVID. The benefits of antimicrobial QACs are seen in increased quality-of-life measures, such as reduced time away from work and school and reduced medical costs. As active antimicrobial agents, QACs and QAC-containing consumer and professional products are highly regulated by authorities such as the US Environmental Protection Agency and the European Chemicals Agency. A complete database of guideline safety studies, covering human and environmental health, is available and has been reviewed by multiple regulatory agencies globally. The conclusions of regulatory agencies from these studies indicate no concerns regarding reproductive effects, genotoxicity, carcinogenicity, or other systemic adverse effects. In contrast, some published academic studies provide mixed findings on lipid synthesis, reproductive effects, and asthma. However, many of these studies have been found not to be robust in design and lacked appropriate controls to support conclusions of a clear and direct association with an adverse effect. The hazard data for the QACs show that their primary critical effects are point-of-contact effects such as irritation, which exhibit a threshold and occur in a dose–response manner. This review compares toxicity information for QACs, examines human exposures, and quantifies the risks estimated from QAC product use. QACs are an important cleaning and hygiene tool, and the abundance of safety data generated for them provides assurance they can be safely used in professional and household products.
Keywords
Introduction
Quaternary ammonium compounds (QACs) are a large class of chemicals used in a variety of consumer and industrial products with unique attributes that make them an important public health tool, particularly due to their antimicrobial properties. QACs have a long history of safe use and they are routinely reviewed by competent authorities for safety in the marketplace. The hazard data demonstrate that QACs primarily produce threshold-based point-of-contact effects such as irritation. New reports on QACs continue to emerge, and although some of these studies have raised questions about potential harm to human health and the environment,1,2 other studies comprise an extensive environmental and human health dataset for antimicrobial QACs to support their safe use in a variety of applications.3,4 This review compares toxicity information for QACs, examines human exposures, and quantifies the risks estimated from QAC product use.
There are several QAC sub-classes, and sometimes these sub-classes have very different functions (e.g., herbicidal QACs vs. non-herbicidal QACs). This paper focuses on two main antimicrobial sub-classes: alkyl dimethyl benzyl ammonium chloride (ADBAC) QACs and didecyl dimethyl ammonium chloride (DDAC) QACs. The EPA has designated the term ADBAC to include 19 compounds. They have decided to use alkyl (40% C12, 50% C14, 10% C16) dimethyl benzyl ammonium chloride as the model compound because it has the highest number of active registrations and therefore is expected to be the most representative compound for this case (EPA 2017 work plan). DDAC includes five structurally similar QACs characterized by having a positively charged nitrogen covalently bonded to two alkyl group substituents (at least one C8 or longer) and two methyl substituents.
ADBAC and DDAC QACs are registered active ingredients for antimicrobial products with the US Environmental Protection Agency (EPA) and other regulatory agencies around the world. They are regulated under the EPA Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA), the European Biocidal Products Regulation, Health Canada, and the California Department of Pesticide Regulation. QAC antimicrobial products include algicides, household and institutional sanitizers and disinfectants, food processing sanitizers, hand care products, and process water treatments.
Antimicrobial products, and QACs specifically, play an important public health role in homes, schools, hospitals, and restaurants. Antimicrobial products are defined by the EPA as: “substances or mixtures of substances used to destroy or suppress the growth of harmful microorganisms such as bacteria, viruses, or fungi on inanimate objects and surfaces.” 5
Individual QACs with unique CAS numbers fall within the ADBAC sub-class (19 individual CAS numbers) and DDAC sub-class (5 individual CAS numbers) as part of the EPA FIFRA Antimicrobial Pesticide Registration framework. The FIFRA-registered ADBAC and DDAC substances differ in chain length compared to most other non–FIFRA-registered ADBAC and DDAC substances used for non-antimicrobial applications, such as softeners and cosmetic hair conditioners. These are the most commonly used QACs as active ingredients in antimicrobials and share an extensive mammalian and environmental toxicology database and they are the focus of this manuscript.
Standardized guideline efficacy tests to assess both the bacteriostatic and disinfectant capabilities of products are important to demonstrate microbiological performance of sanitizer and disinfectant products containing QACs. 6 These data are necessary to register a product that will make claims the product can control bacteria or viruses. Testing to show microbiological performance against bacteria typically includes testing against a Gram-negative and a Gram-positive bacterium. For claims against a specific virus, tests must be conducted with the specific organism to demonstrate the product’s efficacy.
The control of infectious agents, such as viruses like SARS-CoV-2 which causes COVID-19 and other contact pathogens is an important task in homes, offices, and healthcare settings. Havill (2013)
7
states that: “Because there is a growing body of evidence that the environment plays a role in the transmission of health care-associated infections (HAIs), more attention is being focused on environmental cleaning and ways of improving the efficacy of the cleaning process to decrease the spread of HAIs in the health care setting.”
Given the many antimicrobial agents available, it is important to select the most appropriate disinfectant for the application. 7
QACs are most appropriate for low-level hard surface disinfection (versus disinfection of medical device applications such as endoscopes and other devices that enter the body) in health care settings and are routinely used to kill fungi, amoebas, mold, many types of microbes and most viruses.
8
They are also used for disinfection in waiting rooms and exam rooms, on patient-care devices like blood pressure cuffs and intravenous pumps, and hard surfaces like bed rails and bedside tables and high-touch surfaces such as doorknobs and light switches. As Weber et al.
9
point out: “In recent years, there has been a growing appreciation that the contaminated surface environment is an important source of pathogens that may colonize or infect patients. For this reason, environmental surfaces in patient rooms are routinely disinfected (e.g., once per day) and disinfected when the patient is discharged from their room (i.e., terminal disinfection)”
QACs are also approved for disinfection of surfaces in business, schools and homes.10–12 They are widely used in the home to sanitize and disinfect various surfaces in the kitchen and bathroom. Interestingly, QACs have been shown to effectively (>95%) remove and/or inactivate contaminants (i.e., multidrug-resistant Staphylococcus aureus, vancomycin-resistant Enterococcus, and Pseudomonas aeruginosa) from computer keyboards following a 5-s contact duration. 13 Common QAC-based household disinfecting wipes, for example, have been demonstrated to kill from 99.9% to 99.999% of pathogenic bacteria and virus on surfaces. 14 Besides disinfection, QACs are noted for their cleaning ability, surface compatibility (pH neutral), low volatility, low odor, and long shelf life. 15
QACs have received increased attention for their anti-viral properties since the emergence of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Following the identification of SARS-CoV-2 as the pathogen responsible for the COVID-19 outbreak by the Centers for Disease Control and Prevention (CDC), in March 2020 the EPA issued List N: Disinfectants for Use Against SARS-CoV-2. 12 As of spring 2021, List N included 551 surface disinfectants that meet the EPA’s criteria for efficacy against SARS-CoV-2. 12 Approximately 80% (n =389) of the disinfectants contained a single active ingredient, with the remaining disinfectants comprising mixtures of two or more active ingredients. Of the single active ingredient disinfectants, approximately 48% contained QACs, highlighting their public health importance during the pandemic.
Mode of Action
Knowing the Mode of Action (MOA) of QACs is key to understanding the basis for their efficacy and hazard. The Molecular Initiating Event (MIE) is the interaction of the QAC with a cell membrane. The long alkyl chains of the QAC “permeate into the membrane and disrupt its physical and biochemical properties.” 16 If the QAC is present at a high enough concentration, this MIE can occur with a microbial cell membrane (i.e., a pathogen) or potentially at the point of contact with a mammalian epithelial cell. However, under normal conditions of use, the concentrations that could reach a mammalian epithelial cell are relatively low and therefore may not result in the MIE and subsequent steps leading to toxicity. The potential interaction with mammalian cells notwithstanding, the most important public health impact of the QAC is positive: its antimicrobial action.
Starting with the initial contact of a QAC with a cell membrane, the positively charged nitrogen “head” of the QAC stays at the membrane surface and the hydrophobic carbon chains become immersed in the hydrophobic membrane bilayer. 17 The QAC disrupts the epithelial membrane by altering the configuration of membrane proteins required for structural integrity and/or by disrupting the proteins responsible for catabolism and cellular transport. Under normal conditions, the hydrophobic regions of the phospholipid component of the cell membrane help to moderate the functioning of the proteins present and stabilized by divalent cations such as Ca2+. Cationic QACs, such as benzalkonium chloride (mono-cationic BAC, abbreviated as BAK), can interact with the membrane directly by displacing divalent cations. 17 This interference with the function of the cell membrane may lead to decreased fluidity of the membrane and formation of voids, which may further lead to cell leakage, lysis, and cell death.
Hazards
QACs are registered by the EPA as antimicrobials based on toxicology and efficacy data for an intended use, submitted by the registrants. Most of the data developed to support the registrations of ADBAC/DDAC were sponsored by the registrants and conducted by independent contract research organizations following Good Laboratory Practice (GLP) regulations and procedures. Those standard procedures are typically referred as “guideline” studies, studies that follow strict protocols established by national (e.g., EPA) and international organizations (e.g., OECD) and ensure that the data generated are consistently scientifically robust. Generally, such guideline-driven studies are not published in the scientific literature but submitted to regulatory agencies. However, Luz et al. 4 recently detailed the results of many of the toxicology studies, often by citing the assessments of such data by regulatory agencies. Each of these studies evaluate different human health and environmental endpoints and are reviewed in depth by agency (peer) reviewers, and independently at different agencies (e.g., US, EU, Japan, and California), where typically a decision is rendered each time on whether the study quality is acceptable for risk assessment. A complete analysis is beyond the scope of this article, but the reader is directed to these references.4,18–21 Key points germane to risk assessment and some of the primary references are presented below.
Guideline versus non-guideline studies
Attributes of guideline and non-guideline studies.
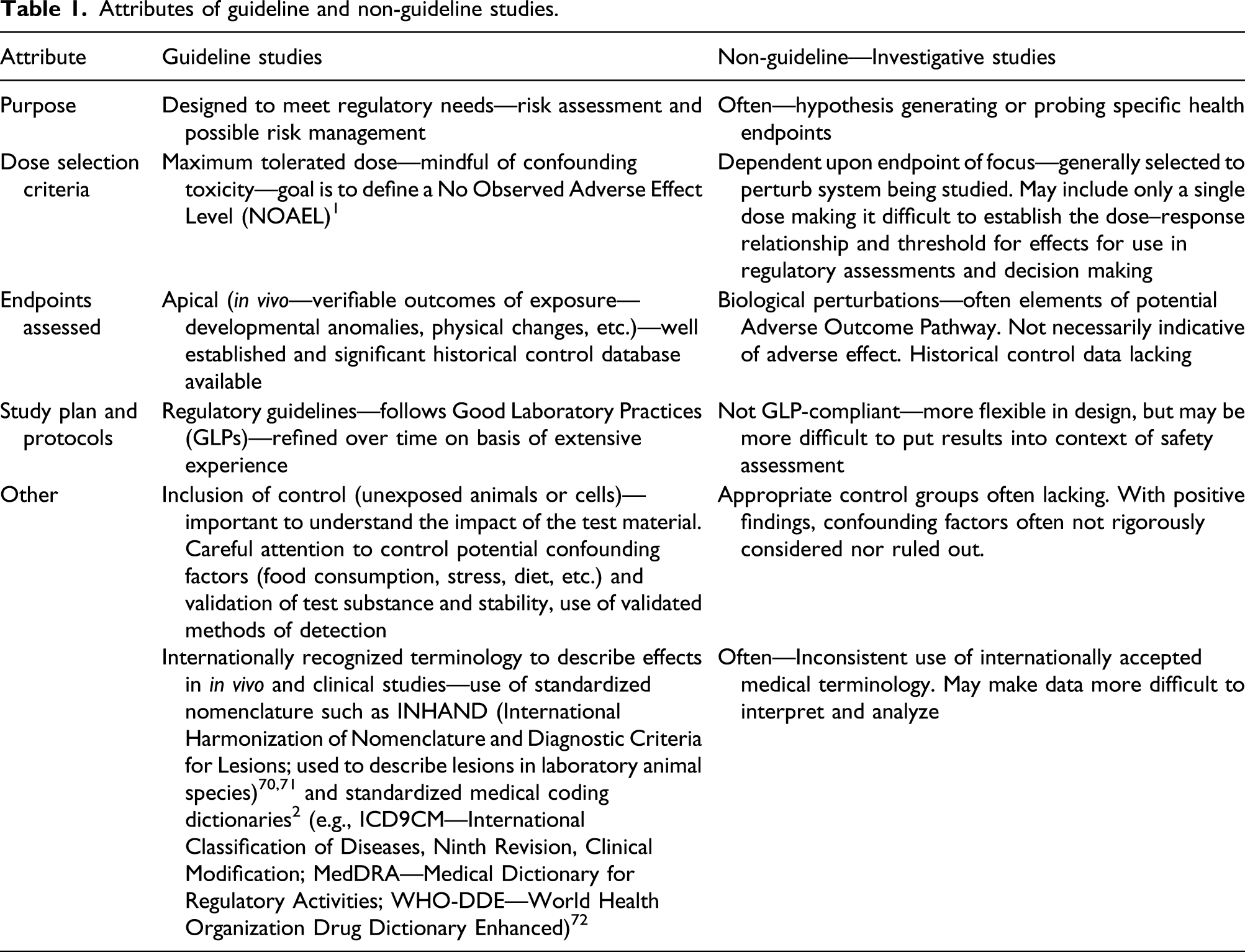
Besides non-guideline whole animal studies, various studies have been reported that focus on subcellular biochemical changes that may be along the “Adverse Outcome Pathway” (AOP) for a chemical resulting from QAC exposure. The question of how to weigh such subcellular changes is worthy of consideration. Generally, the AOP has been a way to identify endpoints to be examined where apical studies are not available. AOPs can be useful to design in vitro test batteries for testing new chemical entities for their ability to perturb the pathways and provide evidence for a likelihood of a particular adverse health outcome to occur. An example of such an approach is the use of the skin sensitization AOP for safety testing of cosmetic ingredients. AOPs conveniently provide a mechanistic underpinning that is pivotal in understating whether the adverse outcomes observed with particular chemical exposures in animal toxicity studies are of relevance to humans.24,25
Both guideline and non-guideline studies have their place in evaluating chemical safety. But, for assessing risk as it is done by regulatory bodies around the world, the non-guideline studies need to be evaluated conservatively and their relevance to risk assessment be carefully considered.
Toxicology studies of QACs
Absorption, distribution, metabolism, and excretion studies ADME
Like many other cationic molecules, the absorption of QACs across epithelial membranes is limited. Both the oral and dermal absorption of DDAC and ADBAC is <10%.26,27 An in vitro percutaneous absorption assay with 14C-DDAC showed that only 0.1% of the administered dose was found to penetrate human skin within a 24-h period. Because 9.41% of radioactivity was detected in the dermis and epidermis, as opposed to the receptor fluid in the test apparatus, the European Chemicals Agency (ECHA) conservatively states that dermal absorption in intact human skin does not exceed 10%. 26 Neither ADBAC nor DDAC are readily absorbed from the gastrointestinal tract (<15%) following oral dosing; most is excreted unchanged in the feces. Four major metabolites of each are formed following oxidation of the decyl sidechains, likely mediated by intestinal microbes. 4
Dermal and ocular irritation
Given their surfactant properties, not unexpectedly, both concentrated DDAC and ADBAC can be irritating or corrosive to the skin and eye,18,19 depending on the concentration of the solution. Consumer and professional end-use products are highly diluted with water and typically contain less than 0.3% and 0.5% QAC, respectively, and are much less irritating than the concentrated solutions. Such products are generally labeled: “Caution: Causes moderate eye irritation. Avoid contact with eyes, skin or clothing.” Most diluted end-use products are not labeled as dermal irritants.
Dermal sensitization
Dermal sensitization and photosensitization studies in guinea pigs conducted according to EPA guidelines show that neither ADBAC nor DDAC are dermal sensitizers or photosensitizers.10,11 But several case reports suggest that contact dermatitis (possibly both allergic and non-allergic) can occur following occupational exposure to cleaning products and antimicrobials.28–31 Recently, National Institute for Occupational Safety and Health (NIOSH) scientists showed that DDAC elicited a positive response in the mouse local lymph node assay (LLNA) at concentrations as low as 0.25%. 32 Follow-up studies in mice suggest DDAC may be a “mixed-type responder” having elicited reactions with elements of both IgE and T-cell mediated hypersensitivity. 33 Caution must be exercised when interpreting the results of LLNAs as it has a high false positive rate for strong irritants. This is because non-sensitizing irritants can induce non-specific lymphocyte proliferation.34,35
It is generally accepted that the MIE leading to dermal sensitization in the OECD AOP for dermal sensitization is covalent binding of relatively low molecular weight electrophilic chemical with selected nucleophilic molecular sites of action in skin proteins to form haptens that are antigenic. 36 Given their chemical structures, this is not likely to occur with QACs, reducing the likelihood they are dermal sensitizers.
General toxicity (acute, subacute, sub-chronic, and chronic dosing)
The numerous studies conducted with ADBAC or DDAC show that cellular changes are limited to effects at the point of contact. Short-term or long-term oral toxicity studies in mice, rats, rabbits, and dogs and dermal toxicity guideline studies in rats and rabbits resulted in no adverse cellular effects at distant tissues. Decreases in body weight, body weight gain, and decreased food consumption have been seen at higher doses, but these effects are likely a result of mucosal irritation at the point of contact, leading to reduced food consumption. 4 Repeat-dose studies included histopathology of organs. None of these studies showed evidence of cellular toxicity at sites distant from the point of contact.
Genotoxicity
A full battery of genotoxicity assays including Bacterial Reverse Mutation Test, Mammalian Cell Gene Mutation Test, Mammalian Chromosome Aberration Test, Unscheduled DNA Synthesis Assay, and In Vivo Mammalian Bone Marrow Chromosome Aberration Test have been run on ADBAC and DDAC in compliance with OECD and EPA guidelines. On the bases of these studies, both are considered non-genotoxic.18,19
Carcinogenicity
Multiple lifetime bioassays have been conducted on ADBAC and DDAC in both mice and rats. The EPA18,19 and ECHA26,27 concluded that ADBAC and DDAC are not carcinogenic. The EPA’s Cancer Assessment Review Committee classified ADBAC as Group D “not likely to be carcinogenic to humans” and DDAC as Group E “evidence of non-carcinogenicity for humans.” 37
Developmental and reproductive toxicity
Some of the detailed developmental parameters measured in guideline studies.
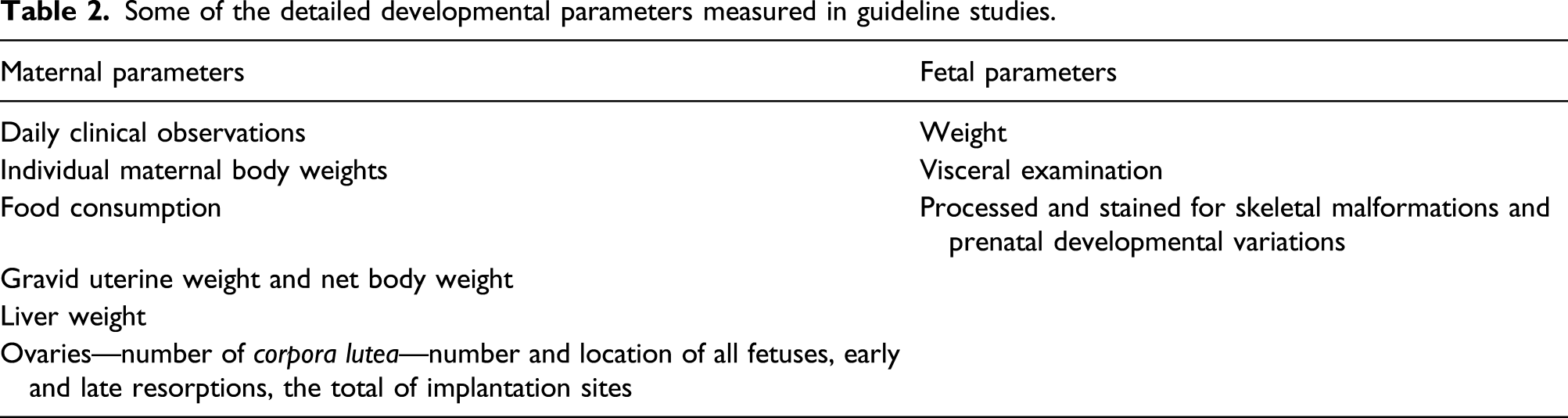
Based on the available data, the EPA 11 concluded there was no evidence of developmental toxicity following prenatal dosing of ADBAC to rats or rabbits. In a two-generation reproduction and fertility study in rats, the only effect noted was reduced pup body weight during lactation and postweaning, and only at the highest dose tested. 11 Similar to ADBAC, EPA 10 concluded there was no evidence of developmental toxicity following prenatal dosing of DDAC to rats or rabbits. In a two-generation reproduction and fertility study of DDAC in rats, the only effect noted was reduced pup body weight and weight gain, also only at the highest dose tested. 10 ECHA concluded from these studies that neither compound “affected reproduction or development at doses that were not toxic to the mother.”26,27 These results are expected, given that QACs exert their effects at the point of contact, rather than be absorbed and affect distant organs in the body leading to lasting systemic effects.
Non-Guideline Studies
Aside from the studies reported above, which followed regulatory guidelines and followed GLPs, several other investigations have been reported. They are non-guideline, non-GLP studies, usually conducted at academic centers. They were not designed to produce results that lend themselves to regulatory review and application to human health risk assessment. Three non-guideline studies of QACs have been published, two in mice and one in rats, that show no reproductive and no developmental toxicity.40–42
More recently, Melin et al.43,44 and Hrubec et al. 45 conducted non-guideline developmental toxicity and reproductive toxicity studies on a mixture of ADBAC and DDAC that reported adverse effects on fetal development and fertility in mice, a non-standard species not part of any regulatory safety assessment program. These results conflict with those studies in rats and rabbits on which the EPA10,11 based their assessments. A detailed discussion of the drawbacks of the recent reports is in letters to the journal’s editor46,47 and responses from the authors.48,49 These studies were also reviewed by Luz et al. 4 and Hostetler et al. 38 Salient points are presented below.
Melin et al. 44 described a decrease in fertility in both male and female mice exposed to a disinfectant concentrate containing approximately 6.8% ADBAC and 10.1% DDAC (dose levels of 0, 60, and 120 mg/kg/day for 2 weeks). The primary concern expressed by Hostetler 47 and Luz et al. 4 is that the effects observed in the offspring are likely due to excessive maternal toxicity rather than a specific effect on development. Melin et al. 44 reported clear evidence of significant toxicity: “Clinical signs initiating euthanasia included: partial closure of eyelids (squinting), reduced activity, ataxia, kyphosis, hypothermia, rapid breathing and dyspnea, and cyanosis.” Because of such indications of excessive toxicity at these high dose levels, 1 out of 10 dams in the 60 mg/kg/day treatment group and 4 out of 10 dams in the 120 mg/kg/day treatment group had to be euthanized. As will be discussed below, these doses are greater than those that humans receive. For example, 120 mg/kg/day is almost 4000× as high as the average daily US exposure to ADBAC and DDAC combined (0.0303 mg/kg/day).10,11
Similarly, Melin et al. 43 administered a combined ADBAC/DDAC dose via the diet to breeding pairs of mice. Exposure to 120 mg/kg/day of the mixture (the only dose tested) resulted in a decrease in the number of corpora lutea after 8 weeks of exposure, a decrease in the number of mid-gestational embryos after 8 weeks of exposure, and a decrease in the number of estrus cycles after 2 weeks of exposure. Clouding the interpretation of this study was the lack of reporting on commonly used measures of maternal health in GLP-compliant studies: body weight, clinical signs, and food consumption. The dose used (120 mg/kg/day) was shown in Melin et al. 44 to cause significant maternal toxicity. Measurements of maternal toxicity in developmental studies is a critical piece of information to determine whether developmental effects observed were due to a test material or associated with maternal stress and subsequent effects on the fetuses.
Hrubec et al.
45
reported two studies of note. The first one is observational in which mating pairs of CD-1 mice and SD rats were unknowingly exposed to unquantified levels of ambient ADBAC and DDAC following use of disinfectants containing ADBAC and DDAC in the university’s vivarium. The incidence of neural tube defects (NTDs) on gestation days (GD) 10 or 11 increased in offspring from dams in the treated rooms versus those in untreated rooms.
3
It is interesting to note that the authors point out that: “Despite the NTDs observed in GD 10 embryos, we did not see exencephaly in late gestation fetuses. This was unexpected as NTDs are not lethal to the developing fetus.”
Further: “It is also possible that repair process is occurring during gestation that reduces or eliminates the NTDs in GD 18 fetuses. Further studies are needed to determine if some or all of the NT lesions observed at GD 10 close over time, or whether affected fetuses die later in gestation.”
45
No information is available on the levels of ADBAC or DDAC present or on other chemicals that may have been present. This is a serious limitation of this study and makes the assessment of cause and effect attributable to the QAC impossible. It also needs to be noted that it is very uncommon to evaluate mice so early in gestation and so there is no reference point to determine whether that effect could be a common background effect in mice that is naturally repaired by the time mice reach their full gestational term. This is an important point as the effect observed is concerning, but there is a significant lack of evidence to clearly indicate that it is a persistent, test material effect.
As a follow-up to the study above, mating pairs of mice were administered 0, 60, or 120 mg/kg/day of a combined ADBAC/DDAC formulation for 8 weeks (Hrubec et al. 42 ). Exposure to the combined ADBAC/DDAC formulation resulted in an increase in NTDs in GD 10 mouse embryos (120 mg/kg/day), a decrease in fetal and placental weight in GD 18 mouse embryos (60 mg/kg/day), and an increase in late gestation resorptions (120 mg/kg/day).
As noted by Hostetler et al., 38 Hostetler, 46 Hrubec et al. 45 failed to consider potential confounding factors which may be responsible for the observed developmental effects. Neural tube closure is one example of a dynamic process that can be delayed by maternal stress from handling and transporting bred mice, environmental conditions resulting in hyperthermia, low humidity, noise, and odors during critical development stages. As with the Melin et al.43,44 studies, these studies conducted by Hrubec et al. 44 did not include common measures of maternal health. The high dose of 120 mg/kg/day is comparable to levels overtly toxic in other studies.
Some authors speculate that the mixture of ADBAC and DDAC may have synergistically contributed to the observed developmental effects even though there is no evidence to support this hypothesis.45,50 The EPA and other regulatory authorities accept that ADBAC and DDAC behave via a similar mechanism and each compound does not produce systemic toxicity beyond irritation and associated effects at the site of exposure. For example, dermal irritation is observed across multiple test species following topical application and, likewise, gastrointestinal irritation is observed following oral administration. In such situations, and given the mechanism of action, synergy is unlikely.
The non-guideline studies are notable for what is omitted. For example, the role of maternal toxicity in producing the reported developmental effects is not given sufficient consideration. Likewise, using mice for such studies is not in accord with customary regulatory testing programs because scant historical data are available to establish baseline responses for developmental and reproductive endpoints. Based on these points and on our review of the studies cited above, the evidence of developmental or reproductive toxicity in non-guideline studies is insufficient to receive consideration in the risk assessment for ADBAC and DDAC.
Subcellular effects
Effects of QACs on subcellular functions.

The studies cited above are difficult to fit in the framework of human health risk assessment. Many endpoints measured could be considered key events along a pathway leading to an adverse outcome in an individual. As mentioned, the “Adverse Outcome Pathway—AOP” is a useful concept to understand modes of action by chemicals. The OECD states that an AOP: “organises existing knowledge concerning biologically plausible and empirically supported links between molecular-level perturbation of a biological system
4
and an adverse outcome at a level of biological organisation of regulatory concern. This approach supports the use of a mode (and/or mechanism) of action basis for understanding the adverse effects of chemicals and other stressor.”
51
The above notwithstanding, it does not seem appropriate to focus on perturbations in key events along an AOP when dealing with chemicals such as ADBAC and DDAC with extensive GLP-conducted in vivo toxicology studies that have directly characterized dose–response relationships in potential adverse outcomes. While these and follow-up studies on subcellular effects deserve consideration, the effects reported per se should not be treated as adverse and be incorporated into risk assessments.
Epidemiology studies: occupational asthma
Evaluating a possible causal relationship between exposure and occupational asthma in any workplace is daunting. Evidence-based evaluations of the literature require careful consideration of the strengths and limitations of the studies. There are reports in the literature of work-related asthma associated with exposure to cleaning agents and disinfectants, and some of these reports relate to using QACs.52–55 Gonzalez et al. 56 reported that nursing professionals exhibited a significantly higher risk of reported physician-diagnosed asthma and, for registered nurses, of nasal symptoms than administrative staff working in the healthcare sector. Yet, aerosol spray use, which is the primary means of respiratory exposure since QACs are not volatile, was not associated with increased risk. Also, asthma cases had no association of IgE antibodies to QACs. The highest risk was associated with tasks involving dilution of QAC-containing products by manual mixing leading Gonzalez et al. 56 to attribute this to possible exposure to repeated peaks of concentrated products “known to be strong respiratory irritants”—even though, due to their non-volatility, such actions are unlikely to result in respired aerosols. Commenting on this study, Heederik 57 concluded that “initiatives are needed in particular to improve education and labeling of products and to reduce exposure to disinfectants and cleaning agents.”
Other reports suggest no such relationship. Dumas et al. 58 assessed the relationship between asthma and exposure to cleaning products in hospital workers. No association was observed between cleaning and disinfecting tasks and asthma in men or in women. Likewise, using a cross-sectional population study, Mirabelli et al. 59 showed no association between healthcare professionals-reported asthma and disinfection use.
In response to the reports described above, the NIOSH formed the Cleaning and Disinfecting in Health Care (CDHC) Working Group, consisting of experts in inhalation toxicology, industrial hygiene, epidemiology, and infection control. This group published an article
60
that discussed the potential hazards of the chemicals used for cleaning and disinfection, including but not limited to QACs, and how those hazards could be reduced by a better understanding of the efficacy of cleaning and disinfecting products and procedures. The article also noted that asthma symptoms or exacerbations have been associated with using disinfectant sprays. Following the report of the CDHC Working Group, Weber et al.
9
critically reviewed the literature on the association between low-level disinfectant use and dermatitis and respiratory symptoms (e.g., asthma). They included data from their examination of the medical records for an occupational health clinic serving the employees of the University of North Carolina (UNC) Hospital. During the study period (2003–2012), UNC Hospital employees had over 144 million person days of possible on-the-job exposures to chemicals of all sorts. Injuries or illnesses caused by chemical exposures were uncommon. Overall, 70 of the 128 chemical exposures were caused by a known germicide (i.e., antiseptic, high-level disinfectant, or low-level disinfectant), only 18 of these were attributed to QACs. Thus, QAC were associated with approximately 1.25 adverse effects per 100 million person days of workplace exposures, an extremely low rate. These data combined with their literature review led the authors to conclude: “The scientific evidence does not support that using low-level disinfectants by health care personnel is an important risk for the development of asthma or contact dermatitis.”
9
In response to this article, a letter to the editor of the publishing journal was written by Pechter and Rosenman. 61 They contended that the conclusion of Weber et al. (2016) 9 was not supported by the occupational health clinic data or the literature review. They stated that over 40 articles have documented the association of cleaning products, and specifically disinfectants used in hospitals, with asthma. In response to Pechter and Rosenman, 61 Weber and Rutala 62 disagreed with many issues and criticisms raised. Weber’s response discusses the substantial morbidity and mortality associated with HAIs and how daily disinfection can reduce HAIs. They stated that most of the literature is focused on the risks of asthma from high-level disinfectant uses and there are fewer studies on low-level disinfectant uses. In addition, they cited that the 40 articles mentioned in the letter were not based on clinical trials or prospective cohort studies.
The most complete assessment of the occupational asthma question was undertaken by LaKind and Goodman.
63
Among issues they grappled with were as follows: 1. Quantifying exposure to non-volatile QACs relative to the myriad of other chemicals (many of which are volatile); 2. Distinguishing between developing asthma and eliciting a response from pre-existing asthma; 3. Establishing the role of irritation in the reported responses.
With these in mind to guide their assessment, LaKind and Goodman (2019) 63 reported their review of 18 investigations, including population studies and both case reports and challenge studies of individual patients and assessed the association between QACs and asthma occurrence in various subject groups. Interestingly, they noted that “studies of occupational groups with the highest potential for QAC exposure (e.g., cleaners and farmers) do not consistently report increased incidence of asthma attributable to QACs.” They cautiously concluded that the data could not distinguish the effects of the QACs from other possible exposures in the workplace associated with cleaning—both chemical and biological (e.g., potent allergens such as pet dander, mites, and pollen).
Hazard summary and toxicity benchmarks
The available data confirm that the primary critical effects associated with the QACs are point-of-contact effects, such as dermal and ocular irritation, which follow a dose–response. Even for inhalation exposures, no cellular toxicity at distant sites was observed following QAC inhalation, even at high concentrations. The available findings reported in the non-guideline studies are not robust enough to be used for human hazard identification or risk assessment.
For quantitative risk assessment for dietary exposure to antimicrobials, consistent with the practice of the EPA and other regulatory bodies, we identified a No Observed Adverse Effect Level (NOAEL) from the most sensitive animal toxicology study. This value is often called the Point of Departure (POD) for risk assessment. The appropriate PODs for ADBAC and DDAC for chronic exposure dietary are 44 mg/kg/day and 10 mg/kg/day, respectively.10,11 Neither value is based on systemic organ toxicity. Rather, they are based on decreased body weight and body weight gain in a chronic/oncogenicity rat study (ADBAC) and based on increased clinical signs in both males and females (emesis and soft/mucoid feces) and decreased total cholesterol in females in a chronic dog study (DDAC). The effects are likely a result of the irritating nature of repeated oral ingestion of the QAC on the gastrointestinal tract at relatively high doses.
With dermal exposures, the PODs for ADBAC and DDAC (for all durations) of dermal exposure are 20 mg/kg/day (80 μg/cm2) and 2 mg/kg/day (8 μg/cm2) respectively.10,11 For ADBAC, 20 mg/kg/day is the highest dose tested before irritation became significant at day 43 in a 90-day dermal toxicity study in rats. No adjustment was needed for risk assessment. With DDAC, a 90-day dermal toxicity study in rats showed a NOAEL = 2 mg QAC/kg/day (8 μg/cm2) based on increased clinical and gross findings (erythema, edema, exfoliation, excoriation, and ulceration).
Regarding inhalation exposures, Dotson et al. (2020) 64 used a weight-of-evidence approach to derive three potential occupational inhalation exposure limits (OELs) for QACs based on irritant toxicity, developmental and reproductive toxicity, and modified existing health-based exposure limits. They concluded that the selected OEL of 0.1 mg/m3 was considered protective of all identified potential adverse health outcomes.
Exposure
Dermal
The Antimicrobial Exposure Assessment Task Force II (AEATF II) generated extensive data on dermal and inhalation exposure to antimicrobials during actual use. 65 These data provide very relevant occupational exposure data for scenarios where QACs are commonly used. These exposure data are used by the EPA to conduct dermal and inhalation risk assessments for active ingredients, such as QACs.
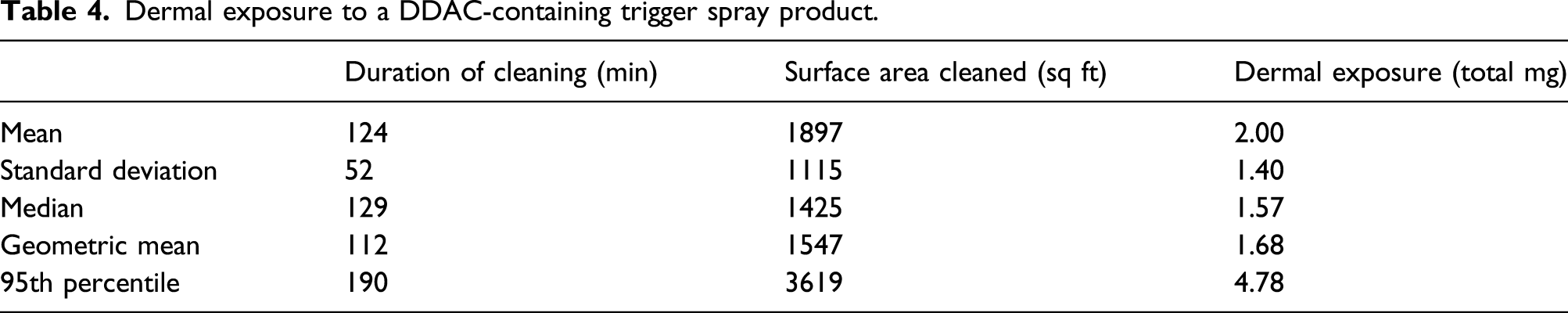
The AEATF II trigger spray exposure study used a Lemon QAC product. 5 The trigger spray and wipe scenario used a 32-ounce trigger spray bottle to spray surfaces and wipe them. Workers cleaned various horizontal and vertical surfaces such as walls, toilets and stall walls, hand railings, shelves including underneath shelves, and counter tops using up and down, side-to-side, and circular motions consistent with their routine cleaning procedures.
Dermal exposure to a DDAC-containing trigger spray product.
Dietary
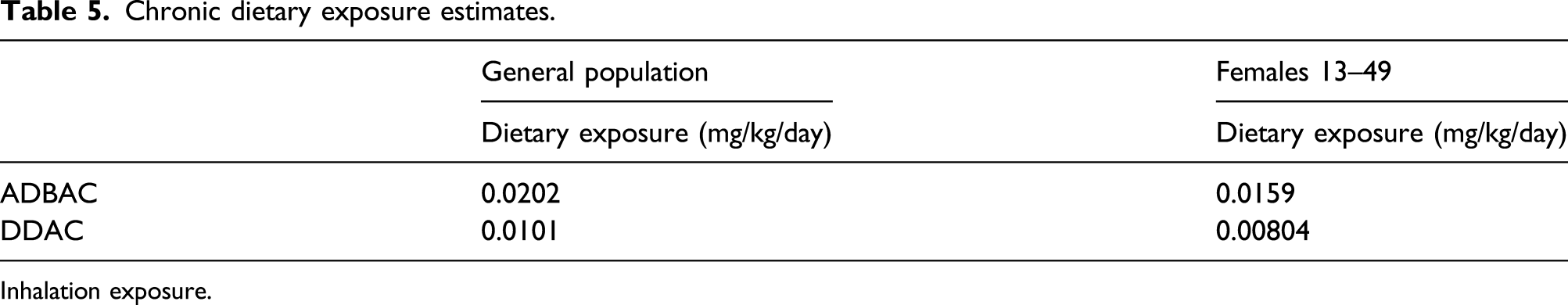
As part of EPA’s Registration Review in 2006, they conducted comprehensive exposure assessments for ADBAC and DDAC.18,19 EPA recently updated these to reflect current uses and provide the exposure data for the risk characterization.10,11
Table 5 summarizes dietary exposure estimates for these exposure scenarios: 1. ADBAC—Use of ADBAC in residential areas without a potable water wash (4900 ppm) and 2. DDAC—Tier 3—Use of DDAC in residential areas (15,000 ppm; with 60% removal from potable water rinse, and 0.89–44.3% transfer from hard surfaces to food). Chronic dietary exposure estimates. Inhalation exposure.
Inhalation exposure
QACs are not volatile, so inhalation exposures may come from inhalation of aerosol spray droplets or from particles containing QACs such as house dust. Vincent et al. 66 measured airborne levels of DDAC in working atmospheres in Lyautey Hospital (Strasbourg, France) during the disinfection cleaning. All DDAC samples were lower than the detection limit of the analytical method by ion chromatography (equivalent to 28 μg/m3). They concluded that “under the standard conditions of use of the disinfecting solutions, the insignificant volatility of DDAC would not seem to be able to contaminate the indoor hospital atmosphere during the disinfection process.” 66 The most significant limitation of this study is that details of the cleaning process and nature of the products (liquid, trigger spray, wipe, etc.) was not provided. Moreover, samples were not taken in the breathing zone of the applicator of the product.
Inhalation exposure estimates for DDAC (mg/m3).

Biomonitoring
Recently, biomonitoring of antimicrobial QACs has been reported. Low levels of QACs were measured in the blood of volunteers. 50 Biomonitoring offers an integrated measure of exposure from all potential sources, including food, dermal exposure, inhalation of sprays, or house dust. However, QAC biomonitoring data provide limited utility for risk assessment since QACs are direct acting, point-of-contact irritants only and not systemic toxicants. 4 And the CDC emphasizes that “The presence of an environmental chemical in people’s blood or urine does not mean that it will cause effects or disease. The toxicity of a chemical is related to its dose or concentration, in addition to a person’s individual susceptibility. Small amounts may be of no health consequence, whereas larger amounts may cause adverse health effects.” 67
Risk characterization
The risk to humans from a chemical is a function of both hazard and the exposure that a human receives. We are exposed to many chemicals with some degree of inherent toxicity. But, because of a low level of exposure, limited bioavailability, and/or the body’s ability to detoxify and eliminate many of these chemicals, the risk is low or essentially nonexistent. The hazard data for the QACs show that the primary critical effects associated with the QACs are point-of-contact effects such as irritation, which exhibit a threshold and occur in a dose–response manner.
The starting point for risk characterization for dermal and dietary exposures is the calculation of a Margin of Exposure (MOE), defined as the ratio of a NOAEL obtained from animal toxicology studies to the predicted or estimated human exposure level or dose. This was calculated for dermal and dietary exposures. For gauging the acceptability of a given MOE, a Level of Concern (LOC) is determined by considering the uncertainty factors (UF) associated with using the available data to estimate risk to humans. Generally, default UFs of 10× for interspecies differences and 10X for intraspecies differences are used. However, because QACs are direct acting irritants with toxicity at the point of contact in the gastrointestinal tract, considerations such of absorption, distribution, metabolism, and excretion characteristics are not likely to have a significant effect on the response among the human population. Therefore, the toxicokinetic component of the interspecies extrapolation UF can be reduced to 1×, leaving 3× for interspecies toxicodynamic differences, and the default 10× intraspecies variation UF can be reduced to 3×. The resulting LOC is 3× 3 which is usually rounded to 10. Thus, an exposure scenario with a MOE >10 should be viewed as posing essentially no risk.
For inhalation exposures, we calculated MOEs based on exposure estimates (Table 6), defined as the ratio of the potential exposure to the substance (indicated by airborne exposure level) and the level at which no adverse effects are expected in humans, that is, the OEL as developed by Dotson et al. (2020). 64 Because the OEL already incorporated UF, the LOC for inhalation is MOE < 1.
Dermal
The exposure component of the risk equation comes from the AEATF II study cited above. 65 Although this was conducted with a DDAC-only containing product, it is reasonable to assume that an analogous ADBAC-containing product would produce similar results. Thus, we will estimate risks for ADBAC and DDAC using these exposure data, but we will use ADBAC- and DDAC-specific hazard data for the hazard component of the risk calculation.
Risk calculations for intermediate term dermal exposures. 7
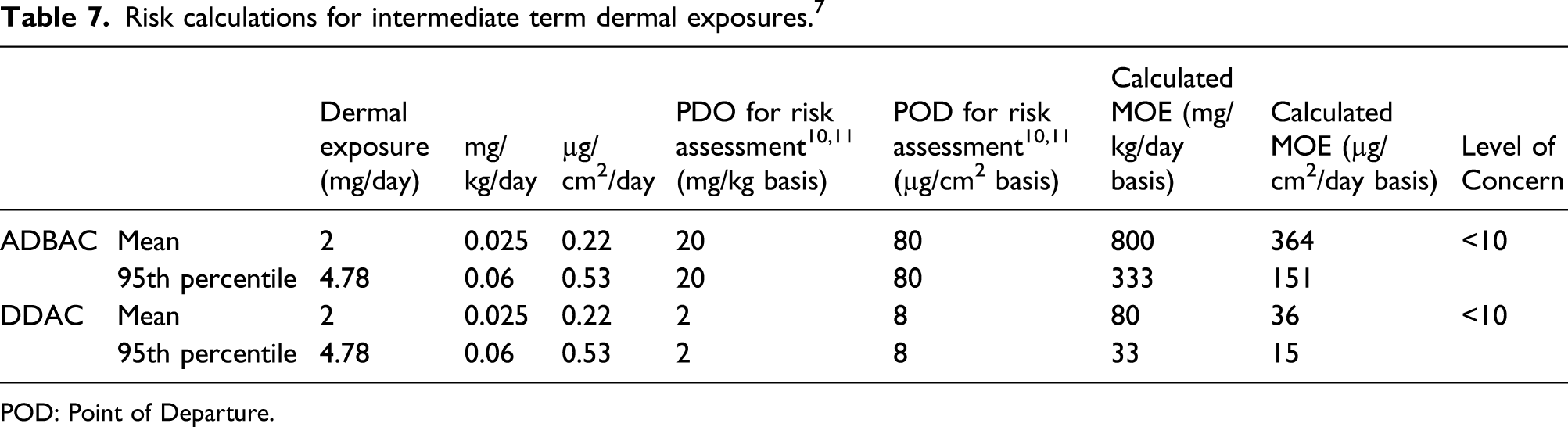
POD: Point of Departure.
Dietary
As mentioned above, the EPA conducted aggregate assessments for consumer exposures to ADBAC and DDAC from food, drinking water, and residential uses as part of their Reregistration Program.18,19 Using these data, they determined that the human health risks from these combined exposures were within acceptable levels. As part of their assessment, EPA included consideration of the available information on the special sensitivity of infants and children.
New risk assessments are being undertaken by the EPA as exposure assessments are updated (Table 5). Screening level dietary assessments have been done for both ADBAC and DDAC as part of developing the work plans. The EPA risk estimates associated with chronic dietary exposure for residential uses are well within the acceptable range (data not presented).10,11
Representative dietary risk assessment for residential use of ADBAC and DDAC.
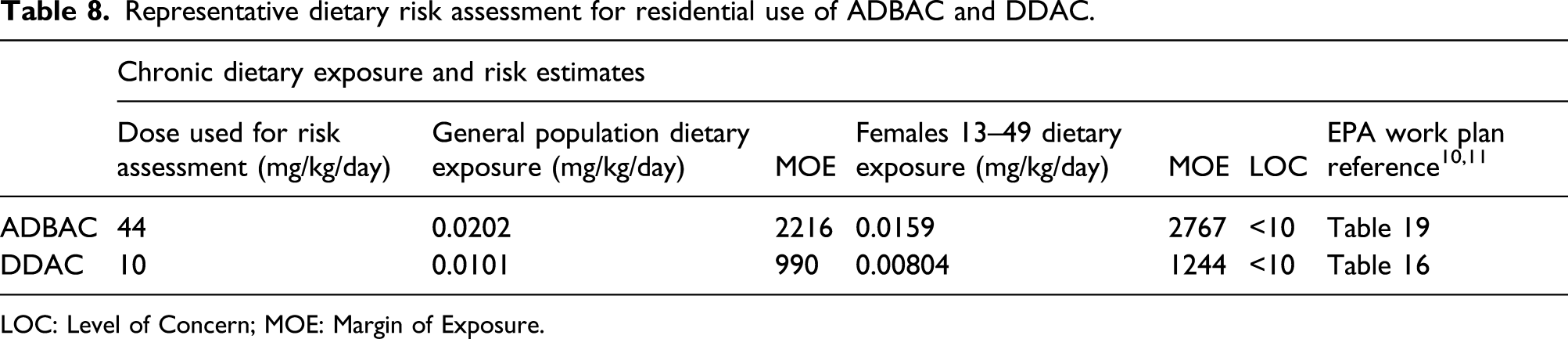
LOC: Level of Concern; MOE: Margin of Exposure.
Inhalation
Representative inhalation risk assessment for use of ADBAC and DDAC in trigger spray product (based on OEL of 0.1 mg/m3)—MOE Level of Concern <1.
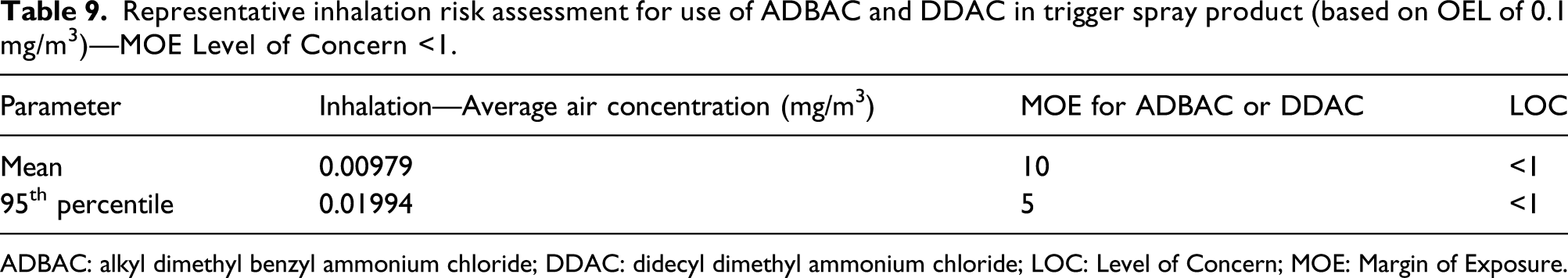
ADBAC: alkyl dimethyl benzyl ammonium chloride; DDAC: didecyl dimethyl ammonium chloride; LOC: Level of Concern; MOE: Margin of Exposure.
Conclusion
Both laboratory studies and real-world situations show that QACs are an essential tool to kill a variety of common pathogenic microbes in hospitals, schools, and restaurants and at home. The public health benefits of controlling bacteria and viruses that cause illnesses such as colds, flu and COVID-19 are significant.
These clear benefits continually need to be weighed against possible risks to human health. QACs have been used safely for many decades. The extensive scientific data show that human exposures and the associated risks are exceedingly low. Despite this conclusion, QACs remain under regulatory and NGO pressure primarily due to sweeping conclusions about safety drawn from recent studies, many of which have significant limitations. Given the benefits of QACs as antimicrobials, carefully conducted, chemical- and use-specific risk assessments should inform regulatory action, and these risk assessments should be based on well-conducted, reliable studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Quat Residue Group (QRG) under the auspices of Ignite Solutions (associated with the Household and Commercial Products Association) [grant number not applicable].
