Abstract
Melamine (ME) is a high production volume compound, mainly used for the synthesis of ME-formaldehyde resins. There is an extensive toxicological and epidemiological database on this compound, but the epidemiology has so far not been reviewed. An extensive literature search in PubMed was conducted and relevant ME epidemiology studies were identified and assessed. The epidemiology data nearly entirely consisted of studies on the health effects due to the food tampering catastrophe in China discovered in 2008 where ME was intentionally added to powdered milk products. Most of these studies were aimed at assessing the extent of the catastrophe and the clinical follow-up of affected children. A reliable dose–response relationship could not be derived from the epidemiology studies. However, they do contain several strong data points that can assist in ranging the No Observed Adverse Effect Level (NOAEL) for humans, for children in particular.
Introduction
Melamine (ME) is mainly used for the synthesis of ME-formaldehyde resins for the manufacture of laminates, plastics, coatings, filters, glues or adhesives, dishware, and kitchenware. 1 Global annual production is about 1.2 million tonnes. 2 There is an extensive scientific literature on the toxicology of ME including metabolism, toxicokinetics, short-term and long-term animal toxicity, and carcinogenicity studies and epidemiology studies. ME is regarded as a compound with low acute toxicity. 1 In rats, the target for ME is the urinary system, where calculi can be formed triggering inflammatory reactions and hyperplasia can occur. 3 In humans, the urinary system is also the target organ. The aim of this review is to summarize all available epidemiologic data on ME exposure and health, to provide a solid basis for classification and risk assessment purposes.
Adverse health effects from ME exposure in humans have not been reported in the scientific literature before 2008. In 2008, an epidemic of urolithiasis, or urinary tract stones, in babies and young children was reported in Mainland China. 4,5 It soon became clear that powdered milk products had been tampered with by several producers. ME had been added to make it appear that the protein content of these products was higher. Once the endemic proportions of the catastrophe became obvious, the Chinese government set up health surveillance programs to detect affected children. In total, 22,384,000 children were screened in this program. 6 Although the first reports from this ME tainting date from 2008, it is not clear when it actually started. The first reports of sick babies with discolored urine date from December 2007, and the first child died on May 1, 2008. 7 Food products analyzed between October 2, 2008, and January 31, 2009, showed that other food stuffs than baby food also contained high ME concentrations, for example, liquid milk, yoghurt, and biscuits. 7 ME-contaminated food stuffs were also found in other countries including Hong Kong, Australia, Korea, Taiwan, Thailand, and the United Kingdom.
Some of the pediatric urolithiasis epidemic had spilled over to Taiwan, where a number of urolithiasis cases related to intake of ME-tainted foods were reported as well, due to migration of Taiwanese people between Mainland China and Taiwan. Between September 24 and October 23, 2008, Taiwanese children whose parents were concerned that they might have consumed ME-tainted foods were investigated in the Taipei Hospital and ME-tainted food intake was confirmed. Seven children had urolithiasis of which six had consumed ME powdered milk. 2 In Hong Kong, there was also concern that some of the urolithiasis epidemic might have spilled over and 45,000 children below 12 years of age were clinically examined. Urinary tract stones were identified in 10 children, well within the background prevalence range. 2 World Health Organization (WHO) further estimated that the dietary exposure based on the consumption of ME-tainted infant formula in China ranged from 8.6 to 23.4 mg/kg bw/day, dependent on the age of the infants, which is about 40–120 times the TDI of 0.2 mg/kg bw/day as recommended by WHO.
Urolithiasis can occur when the constitution of urine is such that precipitation and calcification occur. Small calculi can then grow into larger stones, potentially causing damage to the urinary tract lining or even blocking urinary passages. The majority of urolithiasis are kidney stones. In many cases, urolithiasis goes without any symptoms and the stones remain undetected. Once symptoms arise treatment can be implemented, either conservative or in the form of invasive surgery. Recurrence after treatment is quite common, especially if the causes remain unaddressed. Both males and females are affected. Coe et al., for example, mention estimates of 15% in men and 7% in women as lifetime rates in the US population. 8 The prevalence of urolithiasis appears to be rising. Stamatelou et al. reported that the lifetime prevalence in the United States has increased from 3.2% in 1976–1980 to 5.2% in 1988–1994. 9 Pediatric urolithiasis is also quite common, with a similar preponderance for boys, but this gender difference appears to be shifting. 10 Some studies on the other hand report a preponderance of girls with urolithiasis. 11 Bush et al. also report an increase in pediatric stone disease in the United States between 2002 and 2007 but use an inappropriate method to estimate prevalence. Several other studies report increases in pediatric urolithiasis. 11 –13 High childhood urolithiasis prevalences have been reported in China and Taiwan following the ME food tainting catastrophe in these countries. Prevalence rates, in China, following the discovery of the ME food contamination were, for example, 2.51% in Chongqing, 3.61% in Hangzhou, 2.58% in Sichuan, and 2.9% in Beijing (as reported by Lau and Tu 14 ). These are high rates in comparison to prevalences of 0.03–0.6% in Hong Kong 15 or 0.8% in Turkey with its hot and arid climate. 16 Between 2003 and 2007, before the recognition of the ME contamination, the annual admission rate of urinary tract stones, complicated by renal failure, in patients admitted to Beijing’s Children’s hospital varied between 0.05% and 0.45%. 6 In Table 1, a number of urolithiasis prevalence/incidence rates or estimates for children are presented.
Childhood urolithiasis incidence/prevalence rates as reported in the open literature, based on children presenting themselves with physical complaints or blood in urine, not based on screening programs in the general population, if not stated otherwise.
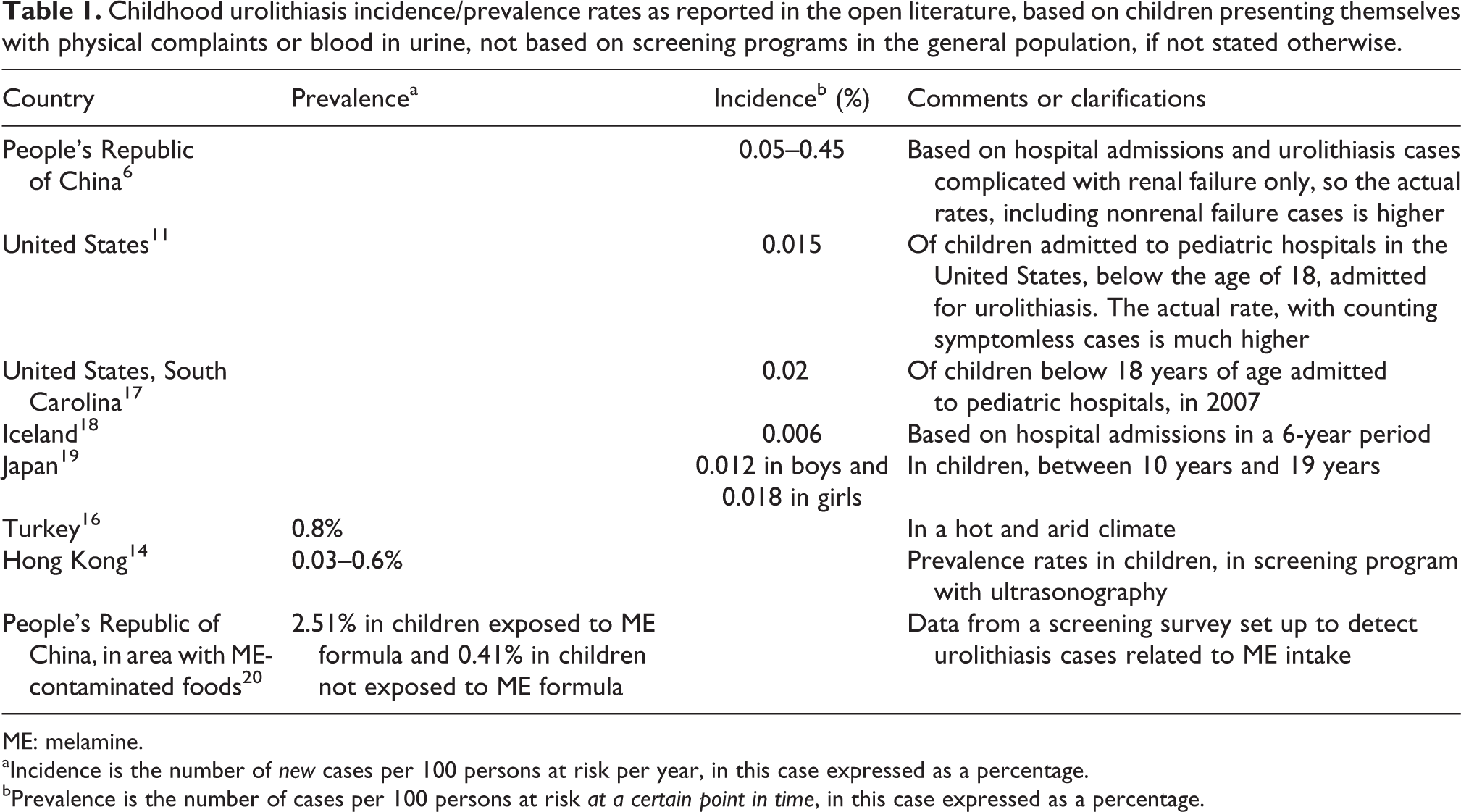
ME: melamine.
aIncidence is the number of
bPrevalence is the number of cases per 100 persons at risk
Several risk factors for pediatric urolithiasis have been reported in the literature. Many cases of urolithiasis are associated with metabolic disorders such as hypercalciuria, hypocitraturia, and cystinuria. 21 Hypercalciuria as a trait of the patient occurs in about 25% of the pediatric urolithiasis cases in the United Kingdom, while cystinuria accounts for about 10% of the cases. Low fluid intake has been found to be associated with nephrolithiasis. 17,23,24 With this respect, it is noteworthy that warm regions such as the Middle East, India, and Pakistan report higher incidence rates. 25 High intake of salt has been reported to increase urinary stone formation. 26,27 In addition, it is thought that obesity leads to a decreased urinary pH, which increases the risk for uric acid stone formation. However, there is no consensus about this. 28 Recurrence of pediatric urolithiasis is frequent. Recurrence rates of 40% 21 and even 70% 29 have been reported.
Literature review methods
To find relevant epidemiology studies on ME exposure and human health, several literature searches were made in PubMed, in March 2018 and again in May 2018 and July and August, with combinations of “melamine” AND “exposure” and “melamine” AND “urolithiasis” and melamine AND “kidney.” In total, 667 journal articles were found and the abstracts were reviewed to determine whether the journal article was an epidemiology study. Reference lists of journal articles on epidemiology studies were further searched for relevant publications. In total, 36 epidemiology studies on ME intake and urolithiasis were identified; 26 studies were conducted in Mainland China, 6 in Hong Kong, and 4 in Taiwan; 5 studies were prospective cohort studies of ME-exposed children, 3 were case–control studies, 14 were cross-sectional studies, 8 were prospective follow-up studies on pediatric urolithiasis cases (prognostic studies), and 6 were case series. Three of the 36 were on adults. In addition, a systematic review of patient prognosis was identified. The literature review further focused on the studies with larger sample size, those that contained information on ME exposure and on studies that included information on urolithiasis risk at low ME exposure levels. These journal articles were further critically reviewed regarding their methodology and informativeness, keeping in mind the ultimate aim of the review to determine the threshold of ME intake at which a urolithiasis risk can be expected. While this literature review did not include human or animal subjects, the study was not reviewed by an ethical review board or ethical committee.
Urolithiasis and ME intake
As already mentioned, in 2008, a urolithiasis outbreak among newborns and young children was reported in China and it soon became clear that a contributory factor was intentional ME adulteration of powdered milk. A large public health survey of 22,384,000 Chinese children was set up to detect and treat cases of pediatric urolithiasis to minimize later health effects and complications. A substantial number of clinical and epidemiological studies was conducted among the Chinese children affected populations to further assess the related health risks and prognosis. Similar studies were also conducted in Taiwan and Hong Kong after the discovery of tampered powder milk there. Initially, epidemiology studies were of a cross-sectional character. Later, follow-up studies on urolithiasis cases were conducted and reported, aimed at investigating prognosis.
Following the recognition of the endemic nature of the ME contamination of foods, Chen et al. screened 3976 children who had ingested ME-tainted milk formula and were presented to the Linyi People’s hospital in the Shandong province of China. 30 In these 3976 children, 63 cases of nephrolithiasis and/or hydronephrosis were identified, giving a prevalence estimate of 1.6%. For 49 of these 63 children, ME intake was estimated by multiplying the daily amount of formula ingested by the ME concentration of that specific brand. Since other food stuffs had also been contaminated with ME, to an extent that could not be quantified, the study group was further restricted to those children who had only ingested the formulas with known ME concentrations. Four of 14 of these subjects with stones had ingested less than 0.2 mg/kg bw per day indicating that the WHO recommended TDI would not be safe. However, this conclusion is rather doubtful for several reasons. First, ME intake concentrations were merely estimates based on self or parent reported food quantities. Second, the sample size of four cases is too small to draw firm conclusions. Thirdly and most importantly, the design of this study does not allow drawing such conclusions because an appropriate comparison group of healthy subjects is lacking: kidney stones are very common with lifetime prevalence up to 10%. Thus, it is to be expected that in a population of 3976 children, several non-ME-related cases of nephrolithiasis would be observed. It is likely that the four cases with ME exposure below the WHO-recommended Total Daily Intake (TDI) are background cases unrelated to ME exposure. This becomes even more plausible because the entire group of 3976 screened infants were selected because of their probable ingestion of ME containing formula and subsequent health complaints. In conclusion, the lack of a proper comparison group, the improper study design, and the uncertainties involved in the exposure assessment process impose severe limitations on this study to such a great extent that no firm conclusions should be drawn about urolithiasis risk at low ME exposure levels. A similar study by Guan et al. on ME intake and urolithiasis was published in the New England Journal of Medicine in 2009 31 and triggered a commentary by Langman, who did not limit his comments to the study by Guan, but also addressed the paper by Wang et al. from Taiwan. 32 In addition to the shortcomings already mentioned above, Langman pointed to another serious bias in these studies due to the higher risk of urolithiasis in premature children as compared to term children. Langman also noted that most Chinese children with urolithiasis potentially associated with ME-contaminated food intake were relatively asymptomatic and lacked hematuria or leukocyturia, indicating the renal damage was not present. Langman also pointed to statement by the American Society of Pediatric Nephrology that recommends a conservative treatment of nephrolithiasis children with previous exposure to ME-contaminated food, because many of the ME-related stones had been shown to easily pass after hydration.
Premature children have a substantially increased risk for urolithiasis. 22 Since premature children are more likely to receive additional nutrition, for example, milk products, a statistical but noncausal association between urolithiasis and ME intake must occur.
He et al. conducted a cross-sectional survey in Hangzou China of 15,577 infants and children. 33 ME exposure was estimated by means of food intake questionnaires. Ultrasonography was used to detect urolithiasis; 88.6% of the cases were below 36 months of age. In the total screened population, 3.61% had urolithiasis identifiable with ultrasonography. In infants aged between 6 months and 12 months, the urolithiasis prevalence was 6.21%. The authors note that many of the children who participated in the survey were already suspect of having urinary symptoms and the reported prevalence rates are likely to be an overestimation of the true prevalence. It was further noted that the capital of Hangzhou has a large migrant population with a lower socioeconomic status, who are more likely to consume foods with ME concentrations up to 2563 mg/kg, because the ME-tainted formulas were less expensive than the untainted formulas.
In Yuanshi County, China, a similar survey was conducted by Liu et al. also using ultrasonography screening to detect cases of urolithiasis and hydronephrosis. 34 Liu et al. screened 7933 children and found 48 cases of urolithiasis, a prevalence rate of 0.3%, a much lower rate than reported by He. The study population was selected by means of the county birth registry and thus is more complete and representative than the population studied by He. ME intake was estimated through food intake questionnaires obtained through interviewing the mothers. The estimated median daily ME intake of the cases was 116 mg/day.
Similarly, Li et al. analyzed the combined data from a survey on children’s health and nutrition status and a study on nephrolithiasis in Beijing 35 ; 1788 children with nephrolithiasis were diagnosed in Beijing hospitals. A comparison group of 7064 children without stones was formed by means of using the data from a survey of children’s health and feeding status in the same geographic area. Next, ME intake was estimated by multiplying the ME concentrations in various milk formulas and the self, or parent report data on powdered milk consumption. Again, no efforts were made to validate the exposure estimates. The bias caused by the existing association between extra nutrition more frequently given to premature children and their intrinsic higher risk for urolithiasis is also present in this study. Next, there is considerable concern about the entirely different data collection schemes for cases and controls. Cases were diagnosed in hospitals and controls were participants in a general health survey. The participation rate in the survey was 15.8% (6498 of an estimated 41,000), compared to a participation rate in the cases of 68.6%. Furthermore, cases were recruited from a defined set of hospitals in Beijing, whereas the controls were taken from a survey that included subjects from outside the catchment areas of these hospitals. It is inappropriate to select cases from a different source population than the controls. This methodological error entirely invalidates the study.
In Shanghai, a population screening program with additional case follow-up was conducted by Gao et al. 36 ; 8335 children aged 6 or less with a history of consuming ME-contaminated milk powder were included in the study. Ultrasonography was used to detect urolithiasis. An estimate of ME intake was made by means of food questionnaires. At baseline, 105 children with urolithiasis were found, yielding a prevalence rate of 1.26%. The urolithiasis prevalence was highest in the highest exposed group. The authors do not describe however how the population was selected and it is quite likely that children with urinary complaints were more likely to participate, leading to an overestimation of the true prevalence.
In two townships of Wuwei city in Gansu, China, a similar screening survey was carried out by Shi et al. 37 A total of 2733 children of 3 years or younger were screened by ultrasonography. Village doctors requested the parents or guardians of children to participate and questionnaire information was used to assess ME-contaminated food intake. Of the ultimately 2186 eligible screened infants, 362 were found to have urolithiasis, including two bladder stones, a prevalence of 16.6%. The authors attribute the high prevalence of urolithiasis in their study to the high consumption of infant formula contaminated with ME, the high ME contamination in the particular brand of infant formula, a more sensitive screening criterion of any calculus ≥2 mm as urolithiasis, and the inclusion criterion of children of 3 years of age or younger.
The ME contamination catastrophe in mainland China triggered concern in adjacent Hong Kong and Taiwan. Lam et al. reported on a population survey carried out in Hong Kong. The major outpatient clinics in Hong Kong were designated as screening centers where residents could walk in to be clinically examined. 38 –40 Children aged 12 years or less with presumed consumption of ME-contaminated foods were eligible for screening. Ultrasonography and urine analysis were used to detect urinary tract abnormalities. Of the 3170 children screened, one case with a renal calculus was detected, a prevalence of 0.03% well within the range of the normal background prevalence. Seven children were suspected of having renal deposits but no formal stones. The estimated ME intake of the eight children with some form of renal stones or deposits was between 0.01 mg/kg/day and 0.21 mg/kg/day. The authors concluded that the ME contamination in Hong Kong was so low that it had not resulted in any noticeable adverse effect on the children.
Following these cross-sectional screening type of studies, clinicians set up follow-up studies on urolithiasis cases to assess the longer-term health impacts. Many of these follow-up studies have been conducted, for example, Shen et al., 41 Shang et al., 42 Dai et al., 43 Zou et al., 44 He et al., 45 and Chang et al. 46 A systematic review by Wang et al. covering 26 studies on the clinical profile and prognosis of ME-induced urolithiasis concluded that there were inconsistencies regarding the clinical features and prognosis of the reported cases. 47 Overall, however, patients had small calculi and could recover without surgical intervention. Kidney abnormalities remained in about 8% of the urolithiasis patients at 12-month follow-up. 47 Follow-up studies on urolithiasis patients with a history of consuming ME-contaminated foods are helpful to the clinician treating or monitoring these patients, but bear no information relevant to risk assessment or the identification of threshold or exposure limits.
Kong et al. identified 2119 Hong Kong school children participated in a survey on milk and milk products consumption to identify children with a high likelihood of having consumed these products. 48 Five hundred and two subjects were invited to provide urine aliquots for ME content analysis. The ME/creatinine ratio ranged from undetectable to 1467 µg/mmol. After an average duration of 23.5 months of follow-up, 47 subjects with high ME urine content were examined with for ultrasonography. One subject declined. The 46 children, with a median urine albumin concentration of 0.70 µg/mmol, had a normal renal function and no cases of urolithiasis were detected.
The ME-tainted food tampering in Mainland China has mainly affected babies and young children and not the adult population. The explanation for this difference is that the food tampering mainly occurred in baby milk formulas, consumed by babies and that these formulas constituted a large proportion of their daily food consumption.
Urolithiasis at low doses
The aforementioned epidemiology studies were queried for data on quantitative dose–response relationships and particularly on risks at low exposure. Only a limited number of publications provide data on exposure level and urolithiasis risk in the form of a dose–response or stratification into exposure/dose categories. Since ME contamination was different between Mainland China, Taiwan, and Hong Kong, with Hong Kong being only marginally affected, the results are presented for these three areas.
Chinese studies with low-dose data
The ME food contamination in China resulted in very high ME intakes, mainly by newborns and young children. Jia et al. estimated that ME daily intake by Chinese exposed children far exceeded the WHO-recommended TDI of 0.2 mg/kg bw/day. 49 The median ME intake was estimated to be 23.4 mg/kg bw for babies up to 3 months of age, with a maximum of 110.2 mg/kg bw/day and a median of 8.6 mg/kg bw/day for babies of 24 months of age, with a maximum of 40.3 mg/kg bw/day as a result of digesting the highest contaminated Sanlu baby formula. Chen et al. studied 3976 Chinese children who had ingested ME-containing milk foods. 30 From these, 63 had urolithiasis as confirmed by ultrasonography with estimated ME intakes ranging from 0.01 to 62.67 mg/kg bw. However, since no data on exposure in the healthy subjects were collected, no comparisons with respect to exposure between cases and controls can be made. Urolithiasis cases in the low exposure range can simply be background cases since urolithiasis is quite prevalent. Similarly, Sun et al. screened 2235 children fed with ME-tainted formula. 50 They identified 79 cases of urolithiasis and compared the estimated daily intakes with those of 103 healthy controls. Average estimated ME intake of the urolithiasis cases was 5.17 mg/kg bw per day with an average duration of 12.53 months as compared to 2.38 mg/kg bw per day with an average duration of 8.65 months in the controls. Unfortunately, they did not analyze their data in any dose–response format, so no conclusions can be drawn about low-dose impact. Li et al. compared the estimated ME intake between 683 urolithiasis cases and 6498 control children. 35 The cases were selected from a list of 1788 urolithiasis children from patient lists at Beijing hospitals, after they were visited by the researchers and a food questionnaire was taken. The authors provide no information on how these 683 cases were selected from the initial list of 1788 urolithiasis cases. They also provide no information on how the 6498 controls were selected from the survey of children’s health and feeding status. In fact, no selection criteria for final inclusion into the study were specified in their publication and no justification for rather drawing samples than using the entire populations is given. The proper procedure to select the urolithiasis cases would have been to examine the 39,729 subjects enrolled in the survey on children’s health and feeding to identify urolithiasis cases. The methodology followed by the researchers has resulted in a comparison between two haphazard samples selected from two entirely different populations. The applied exposure assessment was limited to the infant formulas containing ME, whereas later analyses uncovered that a wide range of foods were contaminated with ME. Thus, it is likely that the ME estimates used in this study are an underestimation of true ME intake. The average ME intake of 2.38 mg/kg bw/day in the group without urolithiasis is about 10 times higher than the WHO-recommended TDI, which clearly demonstrates the severity of the situation.
Taiwan studies with low-dose data
The ME-tainted food outbreak spilled over to Taiwan as many Taiwanese frequently migrate to Mainland China and back. In addition, ME-tainted food products had been imported from China to Taiwan, and several epidemiological studies were conducted to assess the health consequences for the Taiwanese population. Wang et al. reported one such study 51 ; 1222 Taiwanese children were examined by ultrasonography and a dietary history of ME-contaminated food was taken. ME intake was expressed by the ME content in ppm of the consumed product. All subjects were classified into three groups: control group: <0.05 ppm; low exposure group: 0.05–2.5 ppm; and high exposure group: >2.5 ppm. The cutoff point of 2.5 ppm was taken because it is the Food and Drug Administration recommended safety level. In total, 14 urolithiasis cases were detected. The relative risk of 61 in the high exposure group demonstrates that high ME intake was present. The low exposure group (0.05–2.5 ppm) had a relative risk of 1.13 (95 confidence interval: 0.19–6.76) comparable with background prevalence rates. Liu et al. analyzed a series of 309 urolithiasis patients from Taiwanese hospitals and measured markers of renal tubular injury. 52 Urolithiasis cases were recruited from three Taiwanese hospitals, between November 2010 and January 2015. Urinary ME concentrations in urolithiasis cases were significantly higher than controls from a previous study.
Hong Kong studies with low-dose data
The spread of ME-contaminated foods in Hong Kong was much less severe than in Mainland China. As such, the studies conducted in Hong Kong provide more reliable information on urolithiasis risk at low levels than those from Mainland China or Taiwan, because the probability of misclassification of high exposures into low exposure categories is much less likely. Lam et al. reported on a population survey carried out in Hong Kong. The major outpatient clinics in Hong Kong were designated screening centers where residents could walk in to be clinically examined. 38 –40 Children aged 12 years or less with presumed consumption of ME-contaminated foods were eligible for screening. Ultrasonography and urinalysis were used to detect urinary tract abnormalities. Among the 3170 children screened, 1 case with a renal calculus was detected, a prevalence of 0.03% well within the range of the normal background prevalence. The estimated ME intake of the single case of urolithiasis and seven additional persons with suspect deposits was between 0.01 mg/kg/day and 0.21 mg/kg/day. The authors concluded that the ME contamination in Hong Kong was so low that it had not resulted in any noticeable adverse effect on the children. This study provides evidence that ME concentrations at or below 0.21 mg/kg/day is not associated with a urolithiasis risk. A similar study by Kong et al. in Hong Kong, already described in the preceding section, consisted of a survey among 2119 Hong Kong school children and a follow-up of 46 children with high ME intake. 48 No cases of urolithiasis were detected and the urinary albumin/creatinine ratio in the high exposed group was the same as in the children with no or low urinary ME concentrations. The authors concluded that in Hong Kong school children, of which 46 had a median urinary ME/creatinine ratio of 13.21 µg/mmol, no indications of renal damage were present, implying that exposure had been below the effect level.
Urolithiasis and low-dose data in adults
Wu et al. published a preliminary investigation on adult patients with urolithiasis. 53 Twenty-two cases with calcium urolithiasis and 11 with uric acid urolithiasis were compared with a matched group of 22 healthy subjects. Cases were diagnosed between 2003 and 2007 at the Kaohsiung Medical University Hospital in Taiwan. Controls were selected at a later stage. Controls had a median urinary concentration of 0.06 µg/mmol creatinine as compared to 0.14 µg/mmol creatinine in the calcium urolithiasis cases and 0.50 µg/mmol creatinine in the uric acid urolithiasis cases. The authors conclude that the study shows that low ME levels play a role in urolithiasis. There are several shortcomings in this study. First, it is well-known that urinary ME is a poor measure of past ME intake if any at all. In rats, 90% of administered ME is excreted within 24 h with a half-life of about 3 h. 54 Urinary ME levels thus may be an indicator of ME intake in the previous 24 h, a time frame that bears no relevance to urolithiasis formation. In humans, renal stones are formed over longer time periods preceding hospital visits by many months or years. Second, the sample size is quite small, but more importantly, it is difficult to accept that being southern Taiwan’s largest medical center with 1712 beds and 12 departments that only 33 new adult urolithiasis cases are diagnosed in a period of 4 years. The group must represent a preselected sample from a larger cohort of new urolithiasis cases and they might have been selected on the grounds of their ME urinary levels or suspected ME intake. Thirdly, the control group was selected at a later stage than the cases, after the discovery of ME-tampered foods. It is quite likely that the controls, included at that later stage, had lower urinary ME levels, because they had avoided consuming ME-contaminated foods. Later, the case–control study on adult urolithiasis that was conducted by Wu was expanded by Liu et al. 55 Two hundred and eleven adults with urolithiasis and a matched control group were selected to investigate the association with ME intake. Cases were diagnosed by ultrasonography or radiography in the Kaohsiung Medical Center. Controls were selected from subjects who received routine health screening at the same hospital. Urine samples were taken and ME concentrations determined by mass spectrometry. An association between urinary ME concentration and urolithiasis was observed, among other risk factors such as low fluid intake, smoking, betel quid chewing, and uric acid and calcium concentrations. Since urine samples were collected at diagnosis, the study essentially is of a cross-sectional nature. ME is rapidly excreted and therefore urine samples taken at diagnosis should not be regarded as representative for the period relevant for the etiology of the observed urolithiasis cases. The authors claim that their study provides further support for the need for a further reduction of the WHO TDI of 0.2 mg/kg per day but do not provide the data supporting that claim, given that this extension of their earlier work suffers from the same shortcomings. In addition, this study suffers from several other shortcomings. First, the authors state that the cases and controls were taken from an earlier series consisting of 354 cases and 354 controls, but they fail to provide the selection criteria or how this further selection was conducted. Second, urine samples were collected at time of diagnosis and given the high excretion rate as described earlier they only provide information on exposure immediately prior to diagnosis, not during the stone formation period. The ME-contaminated food scandal was discovered in 2008 and exposure occurred earlier, whereas the urine samples were collected after its discovery, after public health measures were taken and ME-containing foods were taken off the market. Thus, the ME concentrations measured in the urine samples are a serious underestimation of past exposure if they are representative at all and the claim that low-level ME exposure has resulted in urolithiasis is unfounded. It is more likely that the urolithiasis cases examined in this study have been exposed to far higher ME levels during the ME food tampering period. This likelihood is further supported by the detection of ME in nine of nine stones analyzed for their chemical composition. ME has not only been demonstrated to be present in baby powdered milk but also in other food stuffs. 7 Maximum concentrations, even exceeding those of the infant formulas, were reported for powdered milk products and nondairy creamers. These critical points were also made by Garcia Lopez and Quereda who seriously questioned the relevance of the study conducted by Liu et al. for determining the true lower exposure ranges. 56 Essentially, these two Taiwanese studies provide no reliable evidence for the claim that the study subjects had been exposed to low ME levels only, since exposure was measured by means of urinary ME concentrations that only provide information on ME exposure during the last 24 h, 48 h at most. Urinary ME levels are inappropriate measures of past exposure.
Low-dose data: Occupational exposure
A single epidemiologic study in an occupational setting, with potential exposure to ME and renal effects, was found in the conducted literature searches. In 2015, Wu et al. published the results of a study on renal injury in 44 ME tableware manufacturing workers and a control group consisting of 105 workers from a ship building company in Taiwan. 57 The authors reported to have found that some markers of early renal injury were correlated with urinary ME levels. The markers were NAG and β2-MG. However, other markers of renal injury were similar in both groups and no abnormal clinical renal function parameters such as serum BUN, creatinine, uric acid glomerular filtration rate (eGFR), and creatinine clearance rate were similar in both groups. The most problematic aspect in this cross-sectional study, as in many other studies on ME exposure, is that the authors rely on ME urine levels as proxy for past ME exposure. This is particularly problematic since 3 years earlier ME-contaminated foods were detected in many countries including Taiwan, with ME exposures far exceeding the levels encountered in the tableware factory. It is much more likely that the early markers of renal injury are related to the ME food contamination 3 years earlier.
Persistence of urolithiasis
It is well-known that urolithiasis in general has a high recurrence rate, both for children as well as in adults. In the United States, recurrence rates of pediatric urolithiasis have been estimated at 70%. 29 More recent recurrence estimates are in the range of 40%. 58 Reason for these high recurrence rates is that many of the risk factors for urolithiasis are inherited metabolic abnormalities such as hypercalciuria, hyperoxaluria, cystinuria, and hyperuricosuria. 21 Only a few factors can be influenced, for example, increasing daily fluid intake. Thus, even after a successful treatment, either by conservative treatment or by surgical removal of stones, new stones can present themselves after several months or years. Since the underlying triggers of stone formation remain present in the patient, it is to be expected that new stones will be formed (see e.g. Zisman et al. 59 ). As described earlier, many follow-up studies of pediatric urolithiasis patients with ME intake have been published. In 2013, a systematic review and meta-analysis of the then available follow-up studies was published by Wang et al. 47 The systematic review included 26 studies with a combined number of 2164 patients with kidney abnormalities, 94.4% with urinary calculi. The combined percentages of patients with stones ≤10 mm (small calculi) was 95.5%, based on 13 studies that reported these data. Pooled recovery rates were 67.1% after 1 month, 76.3% at 3 months, 85.4% at 6 months, and 92.3% after 12 months following diagnosis. The authors concluded that the children with urolithiasis following high ME intake in general had small stones and were asymptomatic or mildly symptomatic. Eight percent of the children diagnosed with urolithiasis had kidney abnormalities 1 year after diagnosis. Some populations of urolithiasis patients with high ME intake have been followed for longer periods. For example, Chang et al. conducted a 5-year follow-up of 207 pediatric urolithiasis patients. 46 At 5 years of follow-up, residual stones were observed in 17/198 = 8.6% of the patients; stones already present 5 years ago but not washed out. This persistence figure of 8.6% is quite low as compared to persistence rates of 40% reported for children with urolithiasis unrelated to ME exposure. Since no recurrence of stones was observed, the authors concluded that there is no medium-term risk for stone formation in these urolithiasis patients with high past ME intake. This study shows that once ME intake is eliminated or strongly reduced, no new kidney stone formation from past ME intake occurs. Stone persistence strongly depends on their composition. Stones formed after ME intake can easily be pulverized to smaller particles, are softer, and have a less solid texture. Once fragmented, the parts will be flushed out through the urinary system. The epidemiology studies in China and Taiwan clearly show that once ME-containing foods were taken off the market and ME intake was eliminated there were no further reports of new cases. Patients identified in the screening surveys did not suffer from newly formed stones. Renal damage from the stones of course is persistent.
In summary, urolithiasis consisting of small stones can be washed out through the urinary tract or they can become trapped in the tubuli and cause renal damage. When the stones have become too large to pass though the urinary passages, the subject must be treated, first conservatively by advising to increase fluid intake and increasing urinary pH. This also applies to urolithiasis associated with high ME intake. Once high ME intake is stopped, no further stone formation as a result of past ME intake is to be expected. ME-associated stones are composed of uric acid and ME. Background stones usually are of the oxalate type. ME-containing stones are of a different texture, are more easily crushed, and dissolve faster than non-ME-related stones.
Discussion
In newborns and children under the age of 2, urolithiasis is rarely diagnosed. Urolithiasis prevalence rates between 0.03% and 0.6% have been reported in Hong Kong and of 0.8% in Turkey with its hot and arid climate. In a screening survey in children living in Mainland China, the prevalence of urolithiasis was reported to be 0.41% in non-ME-exposed children and 2.51% in children with intake of ME-tainted formula. 20 Unrelated to ME intake, most cases are associated with metabolic disorders such as hypercalciuria, hypocitraturia, and cystinuria, although some additional factors such as fluid intake, salt intake, and to an extent urinary pH and therefore obesity have been extensively reported in the literature. The incidence of adult as well as childhood urolithiasis has been reported to be increasing as a result of our changing nutritional habits.
Several epidemiologic studies provided data on the dose–response relationship between daily ME intake and urolithiasis prevalence or data relevant to it. Some of these, including Li, applied flawed methodologies. However, some epidemiologic studies provide several anchoring points for the dose–response relationship between daily ME intake and urolithiasis: The urolithiasis incident was restricted to Mainland China, but there was concern that it had spilled over to Hong Kong, which triggered several investigations there. These Hong Kong studies found no urolithiasis and concluded the prevailing ME intake had not caused any health effects.
14,39
Daily ME intake in Hong Kong was reported to be between 0.01 mg/kg/day and 0.21 mg/kg/day. This range therefore must be considered as being an exposure level below the No Observed Adverse Effect Level (NOAEL), how far it is below the NOAEL cannot be determined from these data. The only epidemiology study conducted in Mainland China for which the data were analyzed in a dose–response format is unreliable and has serious methodological shortcomings and should not be used for standard setting or for the derivation of dose–response descriptors.
35
Sun et al. compared ME intake between 79 urolithiasis cases and 103 non-urolithiasis cases.
50
Mean estimated daily ME intake in the cases was 5.17 mg/kg/day as compared to 2.38 mg/kg/day in the controls with a standard deviation of 3.39 mg/kg/day, suggesting that the latter dose is not associated with urolithiasis risk. In Taiwan, Wang et al. conducted a case–control study of urolithiasis and estimated ME intake.
51
They reported no increase in urolithiasis in the group, with estimated daily intake of food products containing ME between 0.05 ppm and 2.5 ppm.
The observations described above all demonstrate that at low ME intake the incidence of urolithiasis is not above the background incidence as reported in nonexposed populations. The highest ME daily intake in these observations with no urolithiasis is 2.38 mg/kg/day with a standard deviation of 3.39 mg/kg/day which can be taken as a no adverse effect level. In the epidemiology studies on children with ME intake and urolithiasis included in this review, there is no reliable indication that there is a urolithiasis risk below or in the range of 2.38 mg/kg/day and a standard deviation of 3.39 mg/kg/day.
There are no other publications or reports in the open scientific literature that ME intake can induce urolithiasis in humans other than those from the food tampering incident in Mainland China and the spillover to Taiwan. There is no evidence that ME intake other than this incident has led to urolithiasis in humans. Of the many epidemiological studies conducted following the ME food tampering incident, several studies provide data on the daily dose, the urolithiasis cases had been exposed to although mostly from food questionnaires. Some of the studies suffer from methodological shortcomings, but others provide relatively reliable information. The Hong Kong studies provide more reliable information on low-dose effects because the population had not experienced the high doses that were endemic in Mainland China and spilled over to Taiwan. WHO estimated ME intake in children who were fed tainted formula as being 40–120 times the recommended TDI of 0.2 mg/kg bw/day. The highest daily ME intake with no increase in urolithiasis reported in the reliable studies has been reported by Sun et al. which was 2.38 mg/kg/day in the controls free of urolithiasis, with a standard deviation of 3.39 mg/kg/day. 50 None of the epidemiologic studies with sound methodology provide evidence that at or below this level a risk of increased urolithiasis exists and consequently they provide no evidence that the WHO-estimated TDI is unsafe.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The manuscript titled: “Urolithiasis and exposure to Melamine: A review of the epidemiological literature” has received funding from the Melamine REACH Consortium. It was prepared solely by Dr GMH Swaen, as documentation submission for Melamine. The funding agency provided a part of the literature reviewed, and Dr Swaen conducted several PubMed searches to complete the available literature and prepared the manuscript. The funding agency did not interfere with the work conducted. They provided some suggestions and clarifications, but the manuscript was conceived by Dr Swaen himself.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This review was funded by the Melamine REACH Consortium.
