Abstract
In most parts of West Africa and other developing countries, herbal medicines are sometimes used by patients concomitantly receiving conventional drugs, which can result in potentially serious adverse effects. This study examined
Keywords
Introduction
Despite significant interventions for disease treatment and control, significant morbidity and mortality cases are recorded in most developing countries prompting locals to try different therapeutic options.
1
For example, it is estimated that at least 90% of reported cases and mortality cases of malaria infection occur in Africa.
2
The burden of disease and the frustration caused by treatment failures, prompts people to try both herbal and conventional therapeutic options.
3
In Nigeria and other parts of West Africa, herbal preparations are used by locals who may feel they are experiencing malaria symptoms, without undergoing necessary laboratory tests.
4,5
However, despite such acclaimed therapeutic efficacy of
Materials and methods
Plant sample collection, identification and extraction
Fresh leaves of
Experimental animals
Thirty apparently healthy male Wistar rats weighing between 92 g and 128 g were obtained from the animal house of the Faculty of Veterinary Medicine, Ahmadu Bello University (ABU), Zaria. They were randomly allocated into six groups of five rats each. They were acclimatized under the same environmental condition, provided feed and water Group 1: Untreated rats administered normal saline (1 ml/100 g) for 10 days. Group 2: Normal saline for 5 days + Group 3: Normal saline for 5 days + Group 4: Group 5: Group 6: Artesunate (10 mg/kg) for 5 days + normal saline for 5 days.
Collection of samples and tissues
At the end of the experiment, the rats were sacrificed humanely by cervical dislocation and blood collected into clean, nonheparinized tubes and allowed to stand for 2 hours before centrifuging at 10,000 × g for 10 minutes to obtain serum. Liver tissues were excised for oxidative stress analysis using standard protocols. Liver and kidney tissues were excised for histological examination.
Serum analysis
Alanine aminotransferase (ALT)
Serum alanine aminotransferase (ALT) in the experimental rats was determined according to a standard method 16 which monitors the amount of pyruvate hydrazine formed with 2,4-dinitrophenylhydrazine. In the reaction mixture, 0.1 ml of diluted sample, phosphate buffer (100 mmol/l, pH 7.4), L-arginine (100 mmol/l) and α-oxoglutarate (2 mmol/l) were put. The mixture was then incubated for 30 min at 37°C. After that, 0.5 ml of 2,4-dinitrophenylhydrazine (2 mmol/l) was added to the reaction mixture and allowed to stand for 20 min at 25°C. Finally, 5.0 ml of NaOH (0.4 mol/l) was added to stop the reaction and absorbance was read against the reagent blank after 5 min at 546 nm. The corresponding ALT concentration was obtained from a standard curve.
Gamma glutamyltransferase (GGT)
Serum gamma glutamyltransferase (GGT) was determined according to a reported method. 17 Diluted sample (0.1 ml) was mixed with 0.5 ml of tris buffer (100 mmol/l, pH 8.25) and incubated at 25°C. Next, a careful shaking of the mixture was done and 0.5 ml of L-γ-glutamyl-3carboxy-4-nitroanilide (2.9 mmol/l) was added. The reaction mixture was mixed, initial absorbance read, and then absorbance values were recorded after 1, 2 and 3 min at 405 nm wavelength. The equation below was used to determine the GGT concentration in the test samples.
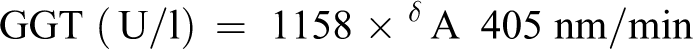
Urea
Urea in serum was determined according to a standard method. 18 Three cuvettes for blank, standard and sample were used. For the blank, 10 µl of distilled water was added to 100 µl of picric acid (35 mmol/l). The cuvettes for standard contained 10 µl of standard and 100 µl of picric acid (35 mmol/l), while the cuvette for sample contained 10 µl of sample and 100 µl of picric acid (35 mmol/l). The three cuvettes were then mixed and incubated at 37°C for 10 min. After that, 2.5 ml of Sodium hydroxide (0.32 mol/l) and Sodium hypochlorite (27 mmol/l) were added to the three cuvettes, mixed and incubated at 37°C for 15 min. Absorbance of sample and standard were then read against the blank at 546 nm. Urea concentration was calculated using the formula below:
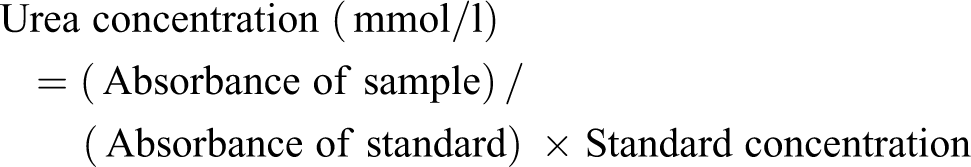
Creatinine
Serum creatinine was determined according to a standard method. 19 1 ml of picric acid (35 mmol/l) and Sodium hydroxide (0.32 mol/l) was pipetted into a cuvette and 0.1 ml of sample was added, mixed and absorbance read after 2 and 4 min. The same was done for standard but without addition of sample. All absorbance was read at 492 nm. Creatinine concentration in mg/dl was calculated as change in absorbance of sample/change in absorbance of standard × standard concentration (mmol/l).
Determination of tumor necrosis factor-α and interleukin-1β levels using ELISA kits
ELISA kits for tumor necrosis factor-α and Interleukin-1β antibody purchased from Wuhan Fine Biotech Co., Ltd were used to determine cytokine levels in serum. The plate wells were washed twice before commencing the experiment. Dilution buffer (100 µl) was added to the blank wells. Diluted standards (100 µl) and samples (100 µl) were added to the wells, covered and incubated overnight at 37°C for 90 min. The next morning, all reagents were brought to room temperature before the plate was washed twice with 300 µL wash buffer per well. The residual buffer was then blotted out by firmly tapping the plate upside down on absorbent paper. All subsequent washes were performed similarly. Biotin labeled antibody (100 µl) was added to each well to block non-specific binding. The plate was then covered with a seal and incubated at room temperature for an hour, after which the plate was washed three times with wash buffer and 100 µl of conjugate added to the wells. The plate was then covered, incubated at room temperature for 30 min followed by washing with wash buffer. Diluted detection antibody solution (100 µl) was then added to each well, incubated at room temperature for 1 h with shaking, followed by washing with wash buffer. Diluted HRP-streptavidin conjugate (100 µl) was then added to each well, after which the plates was sealed and incubated at room temperature for 30 min and washed five times with wash buffer. TMB substrate solution (90 µl) was then added into each well, covered with seal and incubated at 37°C for 30 min. Finally, the reaction was stopped by adding 50 µl of stop solution to each well. Absorbance was read at 450 nm and corresponding concentration determined from a standard curve.
Oxidative stress
Excised liver and kidney tissues were rinsed in 1.15% KCl and blotted with filter paper. Next, the tissues were weighed, chopped into bits, followed by homogenization in four volumes of the homogenizing buffer (pH 7.4). The homogenate obtained was centrifuged at 10,000 × g using a cold centrifuge. After 10 min, supernatant was collected and stored for further analysis.
Malondialdehyde
Determination of malondialdehyde (MDA) levels in liver tissue homogenates was done according to a reported procedure. 20 The sample (0.4 ml) was mixed with 1.6 ml of Tris-KCl buffer after which 0.5 ml of 30% TCA was added. Next, 0.5 ml of 0.75% TBA was introduced and placed in a water bath for 45 min at 80°C. After allowing to cool, centrifugation was carried out at 3000 × g. The supernatant collected was used to measure absorbance against a reference blank of distilled water at 532 nm. MDA (mmol/gram) in the tissues was calculated using the formula below (molar extinction coefficient = 1.56 × 10−5 m−1cm−1).
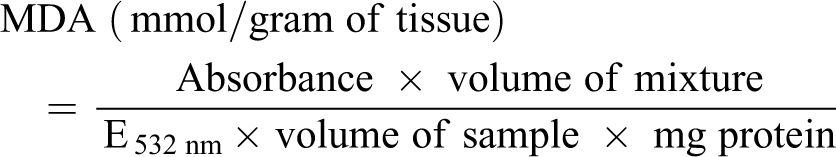
Reduced glutathione
The levels of reduced glutathione (GSH) in the liver tissue homogenates was determined according to a reported method. 21 The sample (1 ml) was deproteinated by adding 4% sulfosalicyclic acid (1 ml). The mixture was then centrifuged at 4000 × g for 5 min. The supernatant obtained (0.5 ml) was then added to 4.5 ml of Ellman’s reagent. The blank was made of diluted precipitating agent (0.5 ml) and Ellman’s reagent (4.5 ml). Absorbance was read at 412 nm to determine reduced glutathione (µg/gram of tissue).
Superoxide dismutase activity
The activity of superoxide dismutase (SOD) in liver tissue homogenates was carried out using a standard protocol.
22
The sample (0.2 ml) was diluted in distilled water (0.8 ml) and 0.2 ml of the diluted sample was added to 0.05 M carbonate buffer (2.5 ml, pH 10.2). The reaction was started by adding freshly prepared 0.3 mM adrenaline (0.3 ml) to the mixture. Absorbance (480 nm) values were recorded every 30 seconds for 150 seconds. Calculation
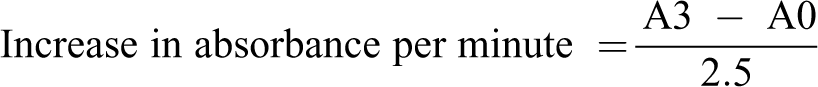
where A0 = absorbance after 30 seconds and A3 = absorbance after 150 seconds

One unit of SOD activity was given as the amount of SOD necessary to cause 50% inhibition of the oxidation of adrenaline.
Catalase activity
The activity of catalase (CAT) in liver tissue homogenates was assayed. 23 The sample (0.2 ml) was mixed with 0.8 ml distilled water to obtain a 1:5 dilution. The rection mixture included 2 ml of H2O2 solution (800 µmoles), phosphate buffer (2.5 ml, pH 7.0) and introduced sample (1 ml). The reaction mixture (1 ml) was introduced into dichromate/acetic acid reagent (2 ml) at an interval of 60 sec.
Catalase activity was determined using a standard curve and the concentration of the remaining H2O2 was extrapolated from the curve.
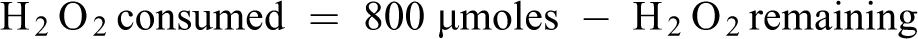
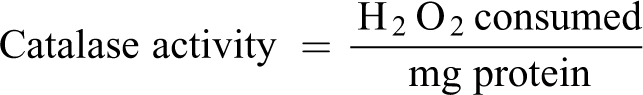
Histological examination
The excised liver and kidney tissues were fixed in 10% buffered formal saline. During tissue processing, the tissues were dehydrated using 100% ethanol, following by clearing using xylene. The tissues were then embedded in paraffin wax. Tissue micro sections (3 µm) were then stained using hematoxylin and eosin and mounted on a mountant according to standard protocol. 24 Histological examination was done using a light microscope at ×400 magnification.
Data analysis
Quantitative data were analyzed using analysis of variance (ANOVA), IBM program (version 20 IBM Corp, Armonk, NY, USA) and expressed as mean ± standard deviation. Duncan Multiple Range Test was used to determine difference between treatment groups. Values of p less than 0.05 (
Results
Alanine aminotransferase (ALT) levels were significantly (
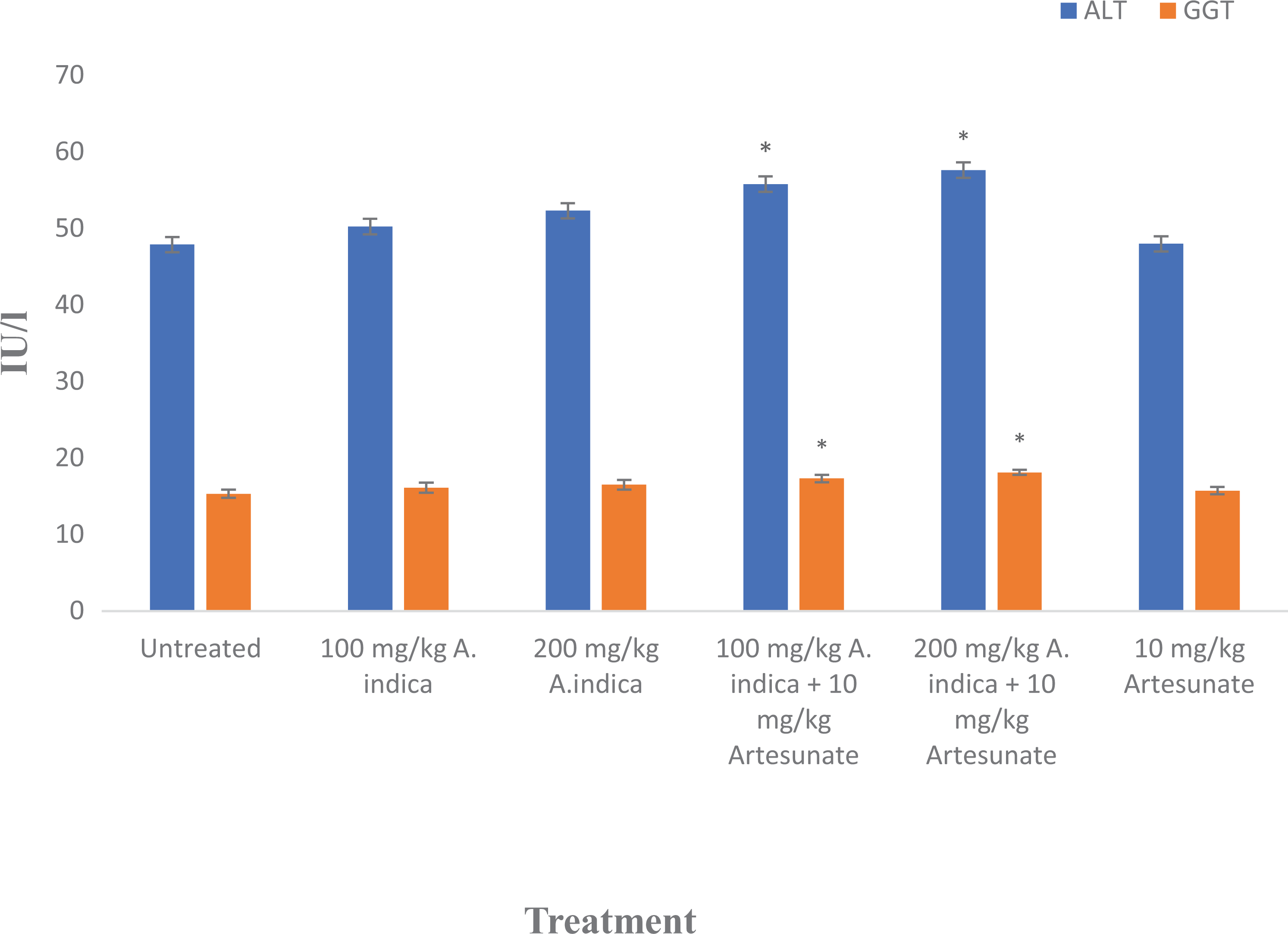
Alanine aminotransferase (ALT) and gamma glutamyltransferase levels in experimental rats. Results are presented as mean ± standard deviation. *Statistical significance (
Urea and creatinine levels were significantly (
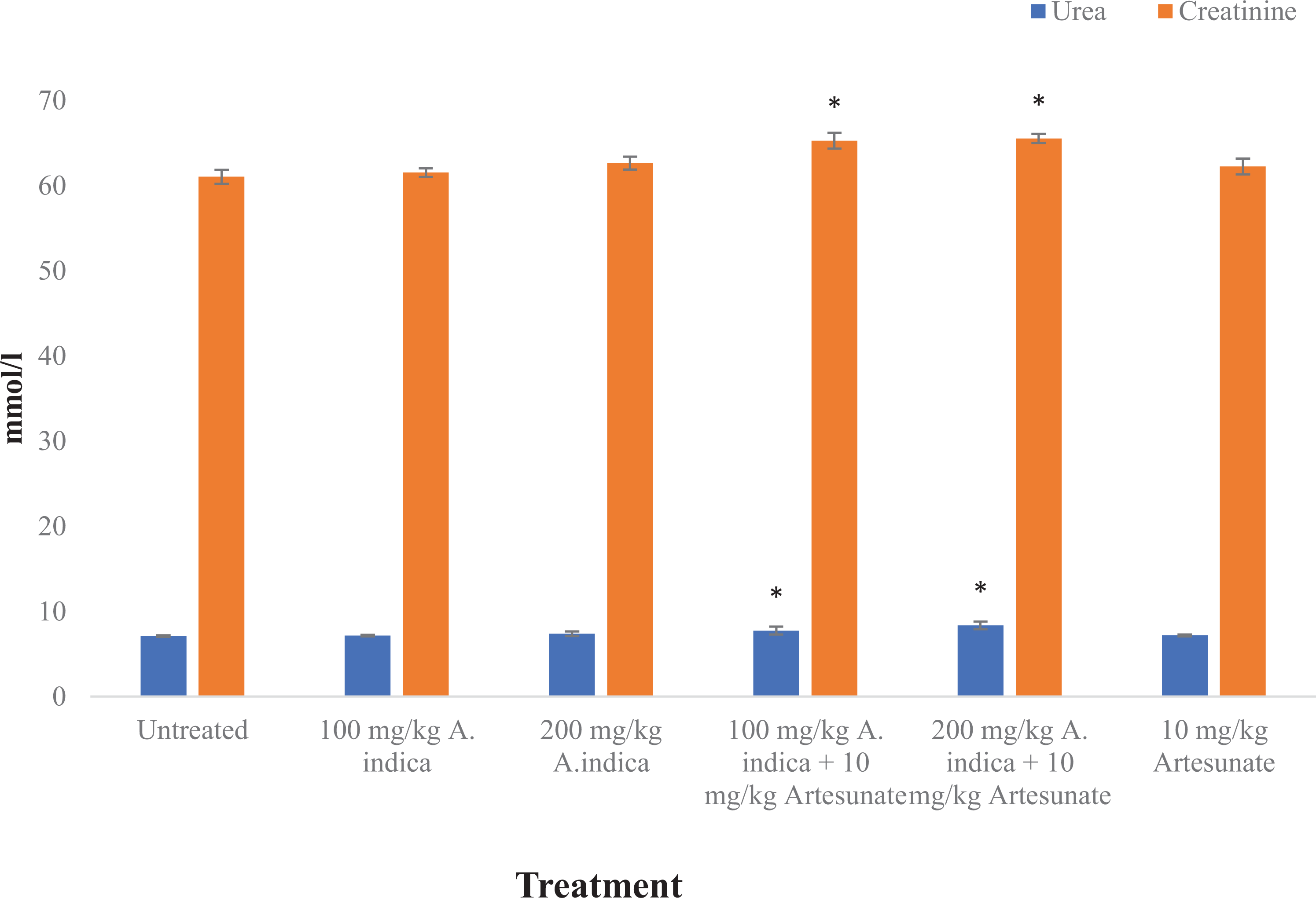
Urea and creatinine levels in experimental rats. Results are presented as mean ± standard deviation. *Statistical significance (
Interleukin 1β and tumor necrosis factor-α levels were significantly (
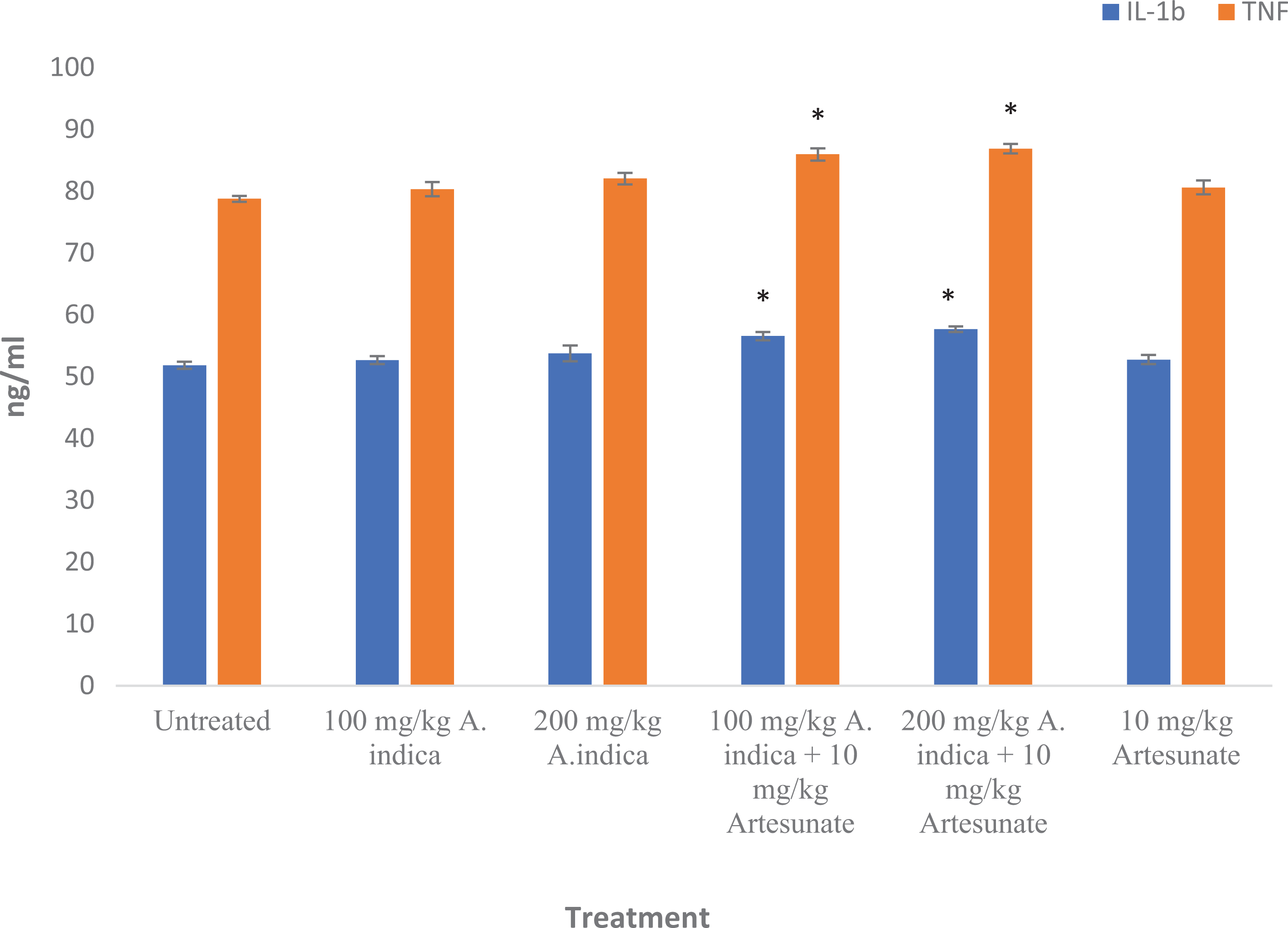
IL-1β and TNF-α levels in experimental rats. Results are presented as mean ± standard deviation. *Statistical significance (
Malondialdehyde levels were significantly (
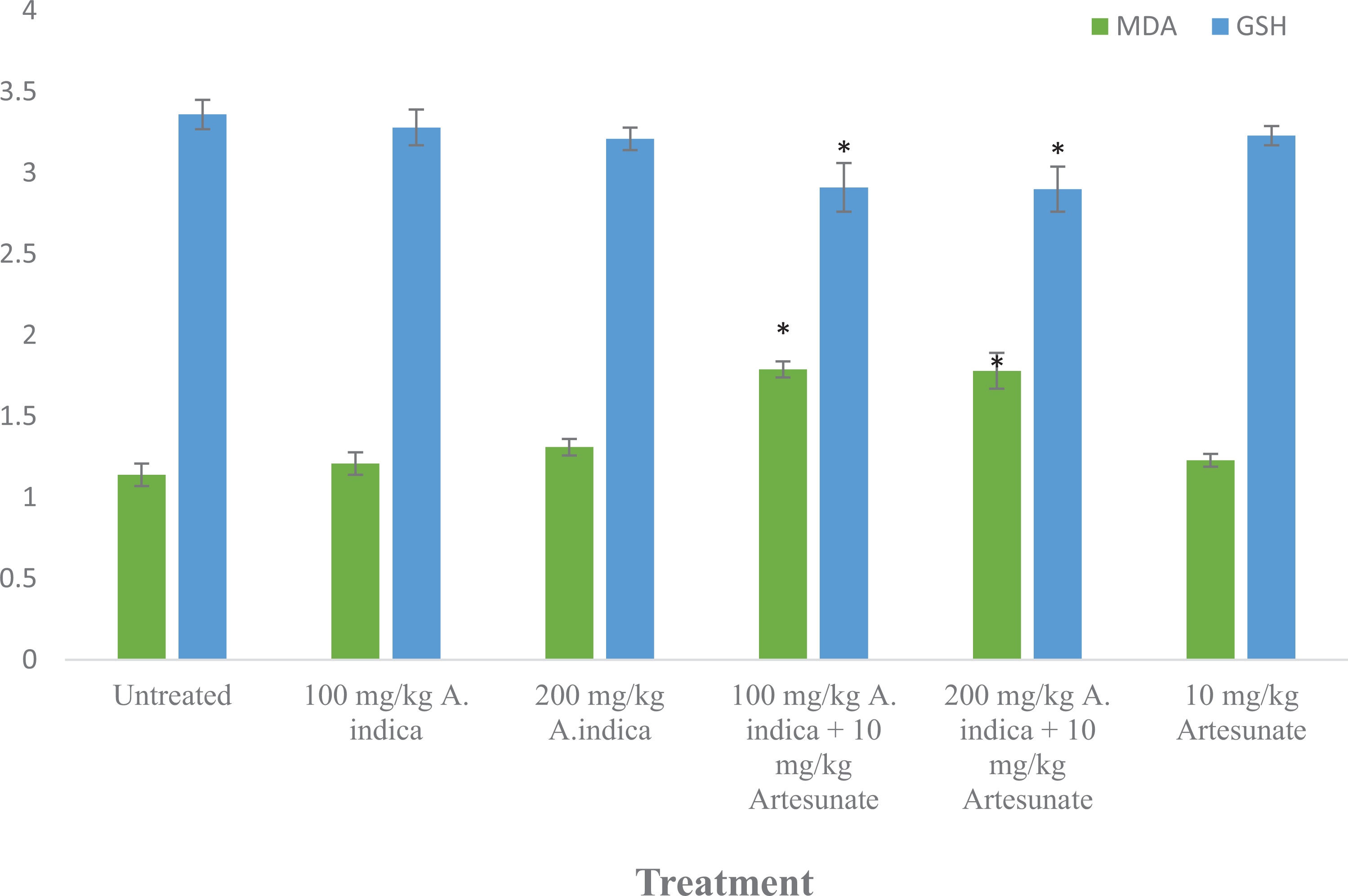
MDA (mM/gram of tissue) and GSH (µg/gram of tissue) in experimental rats. Results are presented as mean ± standard deviation. *Statistical significance (
There was no significant difference in catalase (except the 100 mg/kg
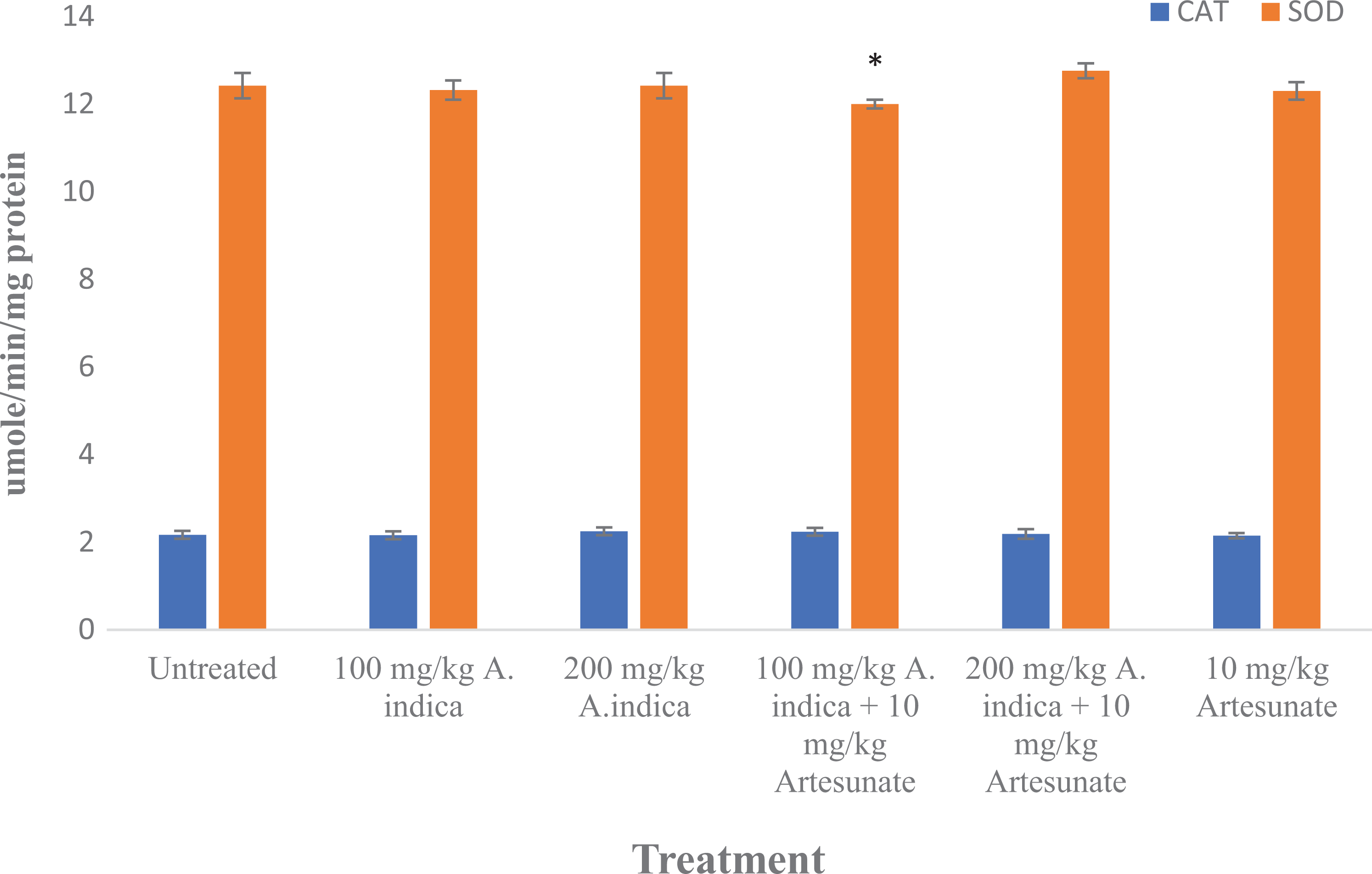
Catalase and superoxide dismutase activity in experimental rats. Results are presented as mean ± standard deviation. *Statistical significance (
Liver histology examination showed slight hepatic necrosis and vascular congestion in the group administered
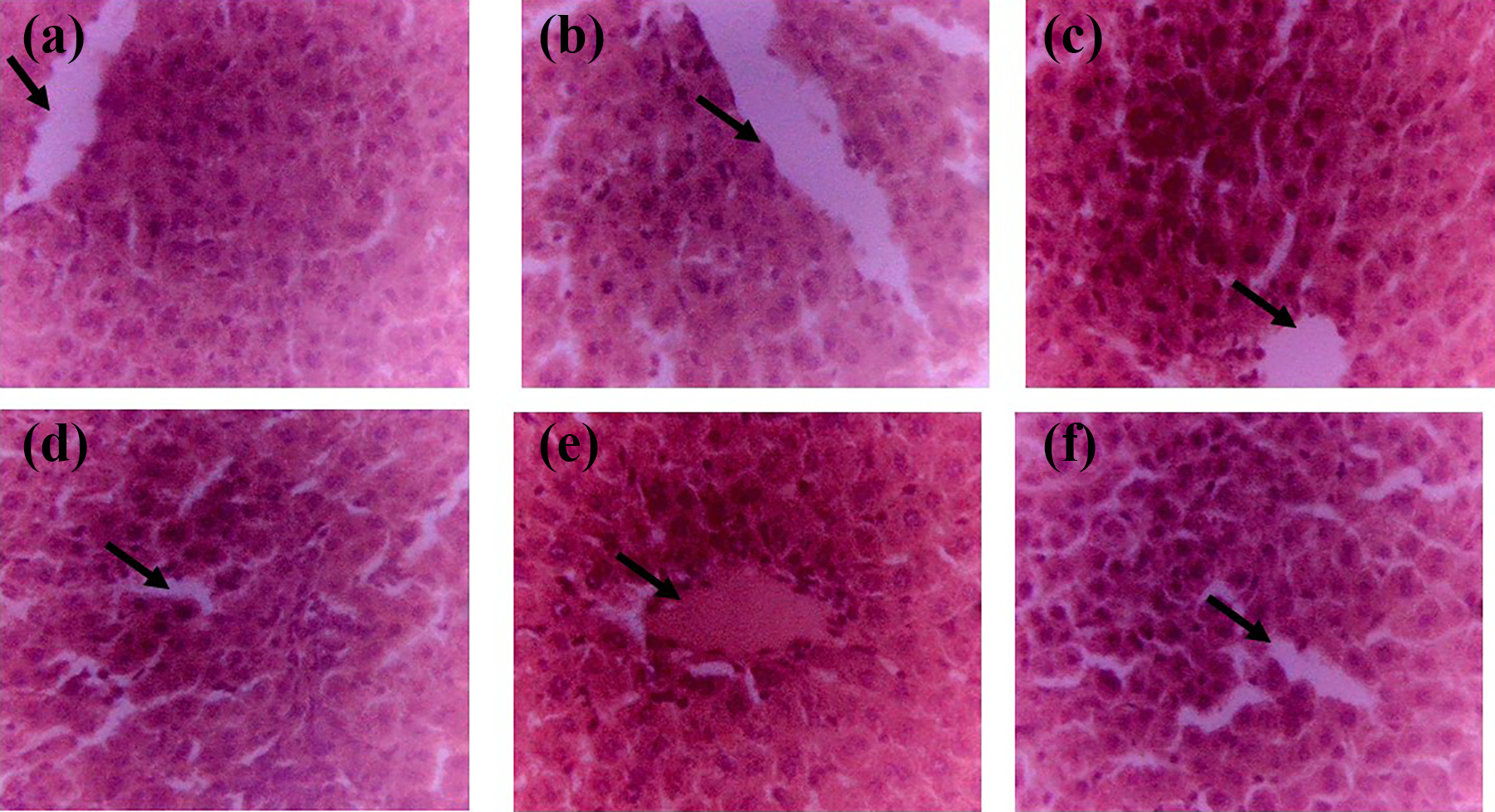
Liver histology of experimental rats (magnification ×400). Liver histology shows normal hepatocytes in the untreated, control group (a). Also, liver histology showed normal features in the group administered 100 mg/kg
Kidney histology examination showed slight tubular necrosis and lymphocyte hyperplasia in the group administered
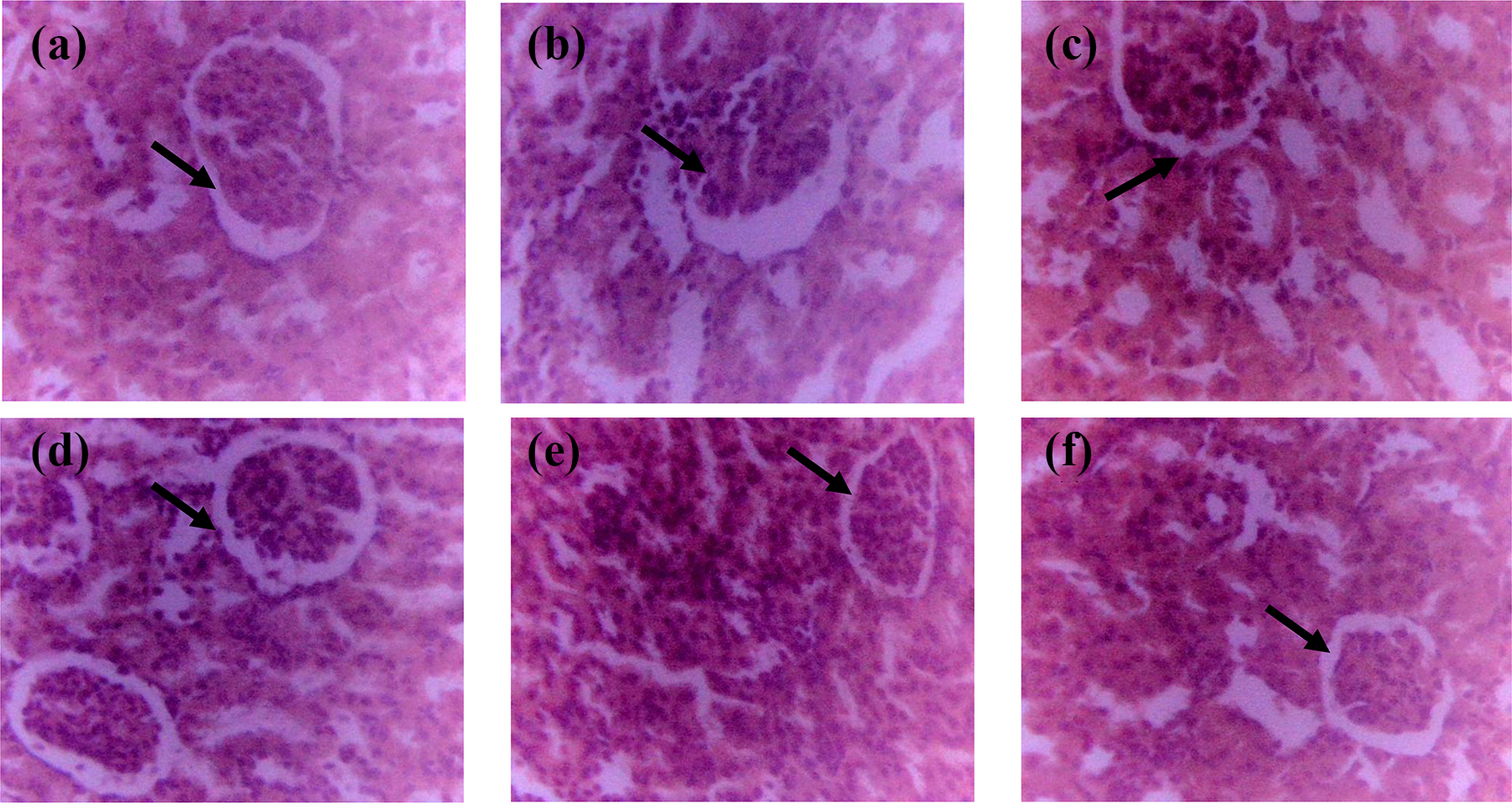
Kidney histology of experimental rats (magnification ×400). Kidney histology showed normal tubules and glomerulus in the untreated, control group (a) as well as the group administered 100 mg/kg
Discussion
Reports of resistance or treatment failure has been reported for some endemic diseases among patients in West Africa and other developing countries.
25
This challenge makes people in such areas prone to the use of multiple approaches in the search for therapy.
6
As a result, some people habitually use both traditional herbal preparations and conventional drugs.
6
Artesunate is a semi-synthetic derivative of artemisinin while
Serum alanine aminotransferase (ALT) and gamma glutamyl transferase (GGT) are used as markers of liver damage, as damage to hepatocytes during xenobiotic exposure can cause increased serum levels of ALT and GGT.
28
The elevated levels of ALT and GGT in the animals administered
Elevated levels of serum cytokines including interleukin 1β (IL-1β) have been associated with impaired renal clearance and increased mononuclear phagocyte production.
31
Increased serum levels of tumor necrosis factor-α (TNF-α) have been associated with development of liver injury due to activation of Kupffer cells.
32
Thus, elevated levels of IL-1β and TNF-α in the group administered
Malondialdehyde (MDA) levels are used as a marker of oxidative stress, because MDA is a product of lipid peroxidation.
33
Glutathione is a tripeptide with antioxidant capacity, with cellular levels indicative of antioxidant potency.
34
The relatively higher levels of malondialdehyde and relatively lower levels of glutathione in the group administered
Conclusion
Collectively, the results from the present study suggest some form of liver and kidney damage in the experimental rats subjected to the sequential administration of
Footnotes
Acknowledgment
The authors wish to acknowledge the assistance of Mr Bamidele and Mr Shailong for histological examination and biochemical analysis, respectively.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was applied for and obtained (Reference number: ABUCAUC/2018/004) from Ahmadu Bello University Ethical Committee in accordance with the ethical standards of the institutional and national research committee in line with the 1964 Helsinki Declaration and later amendments.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
