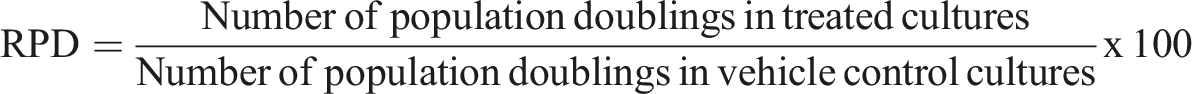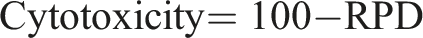Abstract
Background
Alternansucrase is a glucansucrase enzyme that mainly functions in synthesis of glucan-like polysaccharides and can be used for production of novel oligosaccharides and polysaccharides having low glycemic index and prebiotics properties which makes them ideal candidates as dietary fibers and food. From a regulatory perspective, it is necessary to prove the safety of alternansucrase before it can be used in any application as it is a novel enzyme and has not been historically used in food processing.
Objectives
Alternansucrase obtained from Escherichia coli was subjected to toxicological tests to determine its safety for use in various industrial applications.
Design
Toxicity studies were conducted at acute oral and repeated sub-chronic (14 days and 90 days) levels in rats following the OECD guidelines. The genotoxicity studies were conducted using the bacterial reverse mutation test as well as in vitro mammalian cell micronucleus test as per the OECD guidelines.
Results
Alternansucrase did not induce any clinical abnormalities or mortality in rats at the dose level of 2000 mg/kg of body weight in the acute oral toxicity test. A 90-day sub chronic repeated dose oral toxicity study with alternansucrase at and up to dose of 1000 mg TOS/kg of body weight did not show any treatment-related significant toxicological effects on body weight, food consumption, organ weights, hematological and clinical chemistry, or histopathology parameters. Alternansucrase was found to be non-mutagenic up to 5000 µg TOS/plate concentration in the bacterial reverse mutation test. Alternansucrase was determined to be non-clastogenic and non-aneugenic up to the test concentration of 1250 µg TOS/mL during the in vitro mammalian cell micronucleus test.
Conclusion
The lethal dose (LD50) based on this study is greater than 2000 mg/kg body weight, which falls into the category 5 criteria of Globally Harmonized System (GHS). No-Observed-Adverse-Effect-Level (NOAEL) was concluded to be greater than 1000 mg TOS/kg per day. The studies taken together substantiate the safety of alternansucrase enzyme in various food and associated industries. The present study paves a future for safe use of alternansucrase in varied industrial applications.
Introduction
Glycosyltransferase belong to the Glycoside Hydrolase family 70. 1 Alternansucrase (EC No. 2.4.1.140) is a type of glucosyltransferase enzyme that forms a glucan polymer and glucooligosaccharides consisting of mainly alternating α-1,3 and α-1,6 glucosidic bonds. 2 The alternansucrase enzyme aids in the transfer of the glucose residues from sucrose to a sugar acceptor molecule (such as maltose, isomaltose, and so on) with a simultaneous release of a fructose moiety resulting in the formation of a long polymeric glucan called alternan.3,4 This alternan polymer is resistant to degradation by common hydrolytic enzymes that break down starch and other usual carbohydrates2,5,6 and is more water soluble with reduced viscosity. 7 Alternan, thus, finds applications in a wide variety of areas ranging from the food industry to the pharmaceutical and cosmetic industry. The alternans are being promoted as low- or non-caloric food additives or sweeteners, prebiotics, replacements for soluble fibres and bulking agents as well as components of cosmetic products (creams, ointments).2,4–7
Indigenously, alternansucrase enzymes are mainly produced by bacteria belonging to Leuconostoc and Streptococcus genera.2,8 The alternansucrase enzyme was first isolated from the bacterium Leuconostoc mesenteroides NRRL B-1335 (subsequently renamed as L. citreum). 4 Subsequently, the gene responsible for alternansucrase was identified, cloned, and expressed in Escherichia coli. 8
Enzyme safety for human consumption is of prime importance when introducing a new enzyme into the market. Several regulatory authorities require that the safety of the enzyme be demonstrated before approving the sale of that enzyme. Safety of an enzyme can be established in term of safety of the enzyme itself, the safety of the production organism, and safety of the manufacturing process.9–11
Safety of alternansucrase from Leuconostoc spp. has been demonstrated as part of an application made to European Food Safety Authority (EFSA, 2021) for use of alternansucrase in the preparation of sweetening agents and syrups. Since the source organism L. citreum met the criteria for Qualified Presumption of Safety (QPS), no toxicological studies were performed to assess the safety of the enzyme as well. 12 In the present study, the alternansucrase enzyme under study is produced by a genetically modified E. coli strain expressing the gene encoding the alternansucrase enzyme from L. mesenteroides. There are no reported studies on the oral toxicity or genotoxicity safety of the prepared alternansucrase enzyme. In the current study, we report, for the first time, several toxicological tests that were conducted to evaluate and establish the safety of alternansucrase obtained from E. coli. The tests were performed to assess the potential of alternansucrase to cause any kind of acute or prolonged oral toxicity in rats. In addition, the genotoxicity, mutagenic and clastogenic capability of alternansucrase was also examined.
Materials and methods
Test material
Advanced Enzyme Technologies Ltd. provided the alternansucrase enzyme preparation for use in all the studies.
The alternansucrase expressed in E. coli was manufactured according to current Good Manufacturing Practice (cGMP) and the principles of Hazard Analysis and Critical Control Points (HACCP). The manufacturing facility is FSSC 22000 (Food Safety System Certificate) and GMP certified.
The manufacturing process includes four steps, namely, preparation of inoculum, fermentation (seed fermentation and main fermentation), recovery/downstream processing and formulation.
Inoculum was prepared by aseptically transferring the production microorganism from culture vials into an inoculum flask holding the fermentation medium. After sufficient growth, the biomass was transferred to a seed fermenter where the organism grew further in a larger volume under agitation and aeration. Finally, the contents of the seed fermenter were transferred into the main fermenter, where enzyme production occurred. The main fermentation was operated under specified pH, temperature and aeration conditions until sufficient enzyme had been produced. The later recovery steps began with the release of the intracellular enzyme into the fermentation broth by subjecting the fermentation broth to elevated temperature and homogenization process. The resulting fermentation broth was then separated into soluble part containing the enzyme protein and the insoluble medium components and biomass. The soluble part was then subjected to a series of separation, centrifugation and concentration steps at the end of which the food enzyme preparation, devoid of production microorganisms, can be formulated into a commercial preparation.
The test material was a partially purified enzyme preparation, light brown in color and stored at 2-8°C (Figure 1). The chemical and microbiological characterization of the powder was performed by Advanced Enzyme Technologies Ltd. as per the industry guidelines. The total organic solid (TOS) content of the alternansucrase enzyme preparation was 85.18% and the enzyme activity was determined to be 3960 U/g. The ratio of enzyme activity to TOS is an indication of the purity of the enzyme. Alternansucrase powder was suspended in analytical-grade water to form uniform suspensions at desired concentrations based on the study requirements. The formulations were prepared fresh daily according to the study specifications. Alternansucrase powder (test material).
Selection and preparation of animals
Wistar rats (Rattus norvegicus) from Hylasco Biotechnology (India) Pvt. Ltd., Telangana, India, were the animals of choice for the toxicity studies. The number and sex of rats selected for each of the cytotoxicity studies was the minimum required animals as indicated in the respective OECD guidelines in order to comply with the necessary regulatory guidance and to meet the requirement for rational scientific results. The selected female rats for various studies were nulliparous and non-pregnant. The age of the animals at the start of the treatment was 8-9 weeks in the case of acute toxicity study and 6-8 weeks for 14-days and 90-day studies. The weight of the selected animals was within ± 20% of the mean weight of the group.
Groups of two or three animals of similar sex were housed in one cage, in sterilized solid bottom polypropylene cages. The animal room, where experimental animals were housed, was supplied with fresh filtered air with 10 to 15 air changes per hour. The room was air-conditioned with temperatures between 19 to 25°C, relative humidity 30 to 70% and an illumination cycle set to 12 hours light and 12 hours dark. Pelleted rat feed and filtered potable water were provided to all animals ad libitum. Before assignment to any study group, the animals were subjected to a veterinary examination to ensure a good state of health. The animals were acclimatized for a period of 7 to 14 days in the experimental room before the start of the treatment. In case of acute study, the rats were subjected to overnight fasting before dosing in addition to withholding the food for 3-4 hours after dosing. For all the studies, using the Provantis® Preclinical Software, Version 10.2.3 application, the rats were randomly assigned to control and treatment groups.
Methodology for toxicity studies
All the studies were conducted in compliance with the principles of Good Laboratory Practice as outlined in OECD, 1997. 13
Acute oral toxicity study
Acute oral toxicity study of alternansucrase was performed as per OECD Test Guideline 423. 14 In this study, single dose of alternansucrase was orally administered to groups of three female rats (as indicated in the OECD Test Guideline 423) 14 in a stepwise manner. Female rats were selected for this study as they are more susceptible to toxic effects than their male counterparts. The first two steps of the treatment involved administration of 300 mg of alternansucrase/kg of body weight followed by 2000 mg of alternansucrase/kg of body weight in the subsequent two steps by oral gavage. On the day of dosing as well as periodically thereafter, all animals were observed for signs of toxicity and death. A mortality check was done twice a day throughout the observation period of 14 days. The body weights of rats were individually recorded one day before dosing, on the day of dosing and weekly afterward till the end of the treatment period. At the end of 14 days, all animals were humanely euthanized and subjected to a complete necropsy.
Repeated dose 14-day oral toxicity study
Five male and five female Wistar rats per group were administered daily with alternansucrase by oral gavage at the doses of 250, 500 and 1000 mg TOS/kg body weight for 14 days as recommended by OECD for preliminary oral toxicity studies. A concurrent control group of rats were administered analytical-grade water. Dose volume was maintained at 5 mL/kg body weight for all groups including control. The rats were examined daily for signs of toxicity, morbidity (including behavioral changes and reactions to treatment) and mortality. Body weights and food consumption were recorded periodically. All animals were sacrificed terminally in a humane manner and were subjected to a complete necropsy. Weights of the organs (kidneys, liver, adrenals, testes, epididymides, uterus including cervix, thymus, spleen, brain, ovaries, heart, prostate + seminal vesicles with coagulating glands) were recorded immediately upon necropsy.
Repeated dose 90-day oral toxicity study
Repeated dose 90-day oral toxicity study of alternansucrase in Wistar rats was performed in compliance with OECD Test Guideline 408. 15 Ten male and ten female Wistar rats (as indicated in the OECD Test Guideline 408) 15 per group were administered daily with alternansucrase by oral gavage at the dose levels of 250 mg TOS/kg, 500 mg TOS/kg & 1000 mg TOS/kg body weight up to day 90 and were sacrificed on day 91 to evaluate its toxicity. A concurrent control group of ten male and ten female rats was also maintained which received analytical grade water at 5 mL/kg body weight for 90 days. Additionally, for assessment of reversibility, persistence, or delayed occurrence of toxicity, two recovery groups of five rats per sex were maintained for further 28 days following the 90-day treatment for control (water) and high dose (1000 mg TOS/kg) treatments.
The rats were examined daily for signs of toxicity, morbidity, and mortality. They were subjected to detailed clinical examination before initiation of the study and weekly thereafter during the treatment period, recovery period and at termination. Just before the administration of alternansucrase, the eyes of rats from all the groups and at the termination of treatment, the eyes of control and high-dose group rats were examined employing an ophthalmoscope. During the last week of treatment, animals were additionally examined for the assessment of sensory reactivity, grip strength and motor activity. Body weights and food consumption were recorded weekly. Laboratory investigations were performed on blood (hematology and clinical chemistry tests) and urine (qualitative, quantitative and microscopy tests) at the termination of the treatment and the end of the recovery period. Vaginal smears were evaluated for female rats of all groups before necropsy for determining oestrus cycle phases both at the termination of the treatment and at the end of the recovery period. All animals were sacrificed terminally in a humane manner and subjected to a detailed necropsy for gross pathology observations and weights of study plan specified organs/tissues were recorded. Histopathological evaluations were performed on all study plan-specified tissues/organs of all the rats of the control and high dose groups.
Bacterial reverse mutation assay
Salmonella typhimurium reverse mutation assay for alternansucrase was performed in compliance with OECD Test Guideline 471. 16 Alternansucrase was evaluated in the Ames test, using the pre-incubation method, to determine its ability to induce reverse mutation at selected histidine loci in five tester strains of S. typhimurium viz. TA1535, TA97a, TA98, TA100 and TA102 in the presence and absence of a metabolic activation system (S9). Based upon the preliminary tests conducted to assess the solubility/precipitation and preliminary cytotoxicity of alternansucrase, the tester strains were exposed to the test item in triplicate cultures at the concentrations of 15, 50, 150, 500, 1500 and 5000 µg TOS/plate. Liver S9, induced in Wistar rats with sodium phenobarbitone and β - naphthoflavone, was used for metabolic activation in the experiments. Analytical-grade water was used as a vehicle. Concurrent positive control groups were also included in the experiments. The positive controls used without the S9 mix were sodium azide (for strain TA1535), 3-methylmethane sulphonate (for strains TA100 and TA102), ICR191 (for strain TA97a) and 4-nitroquinoline-N-oxide (for strain TA98). The positive controls used with the S9 mix were 2-aminoanthracene (for strain TA1535), 2-aminofluorene (for strains TA97a, TA98 and TA100) and danthron (for strain TA102). The exposed bacteria were plated onto a minimal glucose agar medium supplemented with L-histidine. The plates were incubated at 37°C for about 66 to 67 hours, after which the histidine revertant colonies were counted and their frequency was compared with that in the vehicle control group.
In vitro mammalian cell micronucleus test
In vitro mammalian cell micronucleus test of alternansucrase using human lymphoblastoid cell line TK6 was performed in compliance with the OECD Test Guideline 487. 17 This cytogenetic study was performed to evaluate the potential of alternansucrase to cause clastogenic and aneugenic effects in cells, viz. human lymphoblastoid cell line TK6. Based on the results obtained from solubility, precipitation test and preliminary cytotoxicity test, TK6 cells were exposed to alternansucrase at concentrations of 1250, 625 and 312.5 μg TOS/mL with and without metabolic activation system S9 for a period of 3 hours and 24 hours. Liver S9 was obtained by induction in Wistar rats with sodium phenobarbitone and β - naphthoflavone. Triplicate cultures were used at each tested concentration.
In the presence of a metabolic activation system, cells were exposed to alternansucrase for 3 hours whereas, in the absence of a metabolic activation system, exponentially proliferating cells were exposed to alternansucrase for 3 hours and 24 hours. In all these cases, cells were harvested after about 1.5 to 2 cell cycle lengths after the beginning of treatment for cell count measurements. Positive and vehicle controls, both with and without metabolic activation, were tested concurrently. Analytical-grade water was used as a vehicle control. Known micronucleus forming (aneugenic and clastogenic) agents were employed as positive controls which included mitomycin C and vinblastine, at the concentrations of 160 ng/mL and 3.5 ng/mL, respectively, for experiments without metabolic activation system, while cyclophosphamide monohydrate was employed at the concentration of 6.25 μg/mL for experiment with the S9 metabolic activation system.
Cytotoxicity assessment was performed by determining the relative population doubling (RPD) as follows:
At the end of the treatment/recovery period, cells were lysed, and stained and about 10,000 healthy nuclei per group were analyzed. Micronuclei scoring was performed using the flow cytometric method employing Litron-MicroFlow kit as per the manufacturer’s instructions. The percentage of micronucleated events (%MN) was calculated as follows:
Statistical analysis
Using the Provantis® Preclinical Software, Version 10.2.3 application, the rats were randomly assigned to control and treatment groups for the oral toxicity studies. SPSS Version 23.0 or Provantis® Preclinical Software, Version 10.2.3 were employed to perform the statistical analyses. The variance was evaluated at p < .05. The collected data of different groups for various parameters were subjected to a test for homogeneity by Levene’s test and heterogeneous intra-group variances were suitably transformed, wherever required. Any data with heterogeneous intra-group variances, even after log-transformation, were subjected to Kruskal Wallis one-way analysis of variance on ranks, followed by Mann-Whitney U Test as necessary. A one-way analysis of variance (ANOVA) was employed when the data was determined to have homogeneous intra-group variances. When the ‘F' value was significant, Dunnett's multiple comparisons of the means of treated groups with the control group mean was employed as a post-hoc test. For repeated 90-day oral toxicity studies, additionally, an independent t-Test was employed for comparing the data from the two recovery groups. For the in vitro mammalian cell micronucleus test, one-way ANOVA followed by Dunnett's multiple comparisons and Kruskal-Wallis Test followed by Mann-Whitney Test were used for comparison of the frequency of micronucleation events in treatment groups with that observed in the negative control.
Results
Acute oral toxicity study
There was no prior information available on any toxicity tests of alternansucrase, hence, 300 mg/kg body weight was selected as the starting dose as per OECD Test Guideline 423. 14 Administration of rats with 300 and 2000 mg/kg of body weight did not induce any clinical abnormalities or adverse effects on body weights during the observation period. The mortality rate of the rats was zero during the course of the entire study. Upon sacrifice of the rats at the end of 14 days, no gross pathological alterations were observed.
Repeated dose 14-day oral toxicity study
Based on the acute toxicity study results, the doses of 250 mg/kg body weight, 500 mg/kg body weight and 1000 mg/kg body weight were selected for the repeated dose 14-day oral toxicity study. Alternansucrase administration at and up to the dose of 1000 mg/kg of body weight did not lead to mortality in rats. No abnormal clinical signs were observed in either of the sex of the rats throughout the treatment period for any of the administered doses of alternansucrase. The mean body weights of both male and female rats were not adversely affected during the treatment period of 14 days. The body weight of the treated rats did not vary significantly (p < .05) as compared to the body weights of rats in the control group. The average daily food consumption of treated rats was 94-99% of that of the control group rats for all the dosages of alternansucrase and was thus found to be comparable among the treated and control groups of rats. No clinical abnormalities were observed in the treated rats upon examination of various attributes.
Organ weights of rats in control and alternansucrase-treated groups during the repeated dose 14-day oral toxicity study.
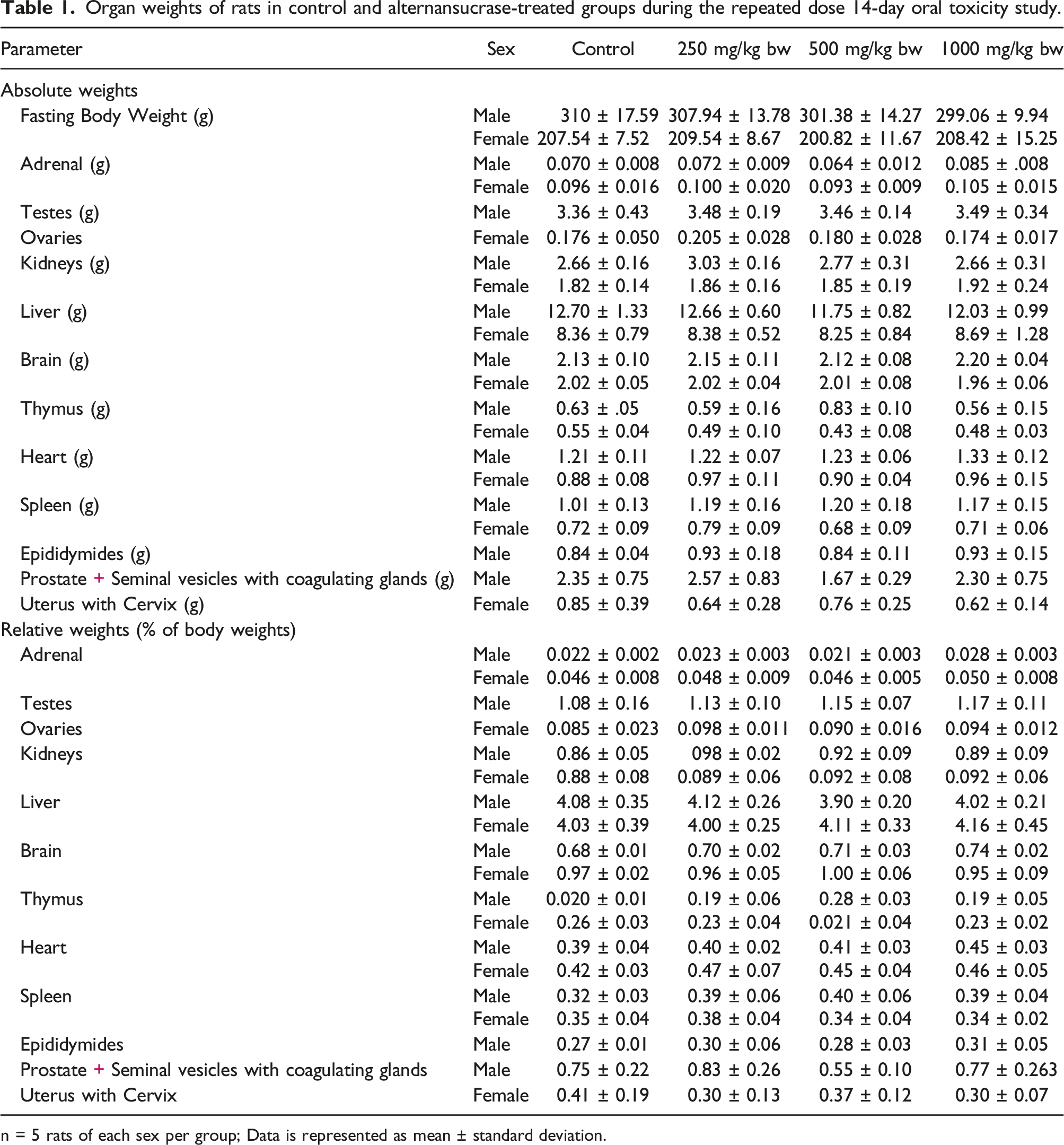
n = 5 rats of each sex per group; Data is represented as mean ± standard deviation.
Alternansucrase, at and up to the dose level of 1000 mg/kg of body weight, did not induce any remarkable and treatment-related gross pathological alterations in any of the tissues of treated rats, as evident in the detailed necropsy examination carried out at the termination of the treatment period. Histopathological examinations were not performed in the absence of any gross pathological changes. As evident from the reported data, no mortality or any kind of adverse effects were observed in the tested rats at all the tested doses of alternansucrase. Based on these observations, the same doses were adopted for the repeated dose 90-day oral toxicity study.
Repeated dose 90-day oral toxicity study
During the treatment and the subsequent recovery period of the repeated dose 90-day oral toxicity study, there was no incidence of mortality or abnormal clinical signs in rats treated at and up to the dose of 1000 mg TOS/kg of rat body weight. No ophthalmic abnormalities were discerned in rats of control groups and high-dose groups rats; hence, ophthalmoscopic examinations were not extended to low-, mid-dose levels and recovery groups.
Functional observations of rats in the 13th week of the repeated dose 90-day oral toxicity study of alternansucrase.
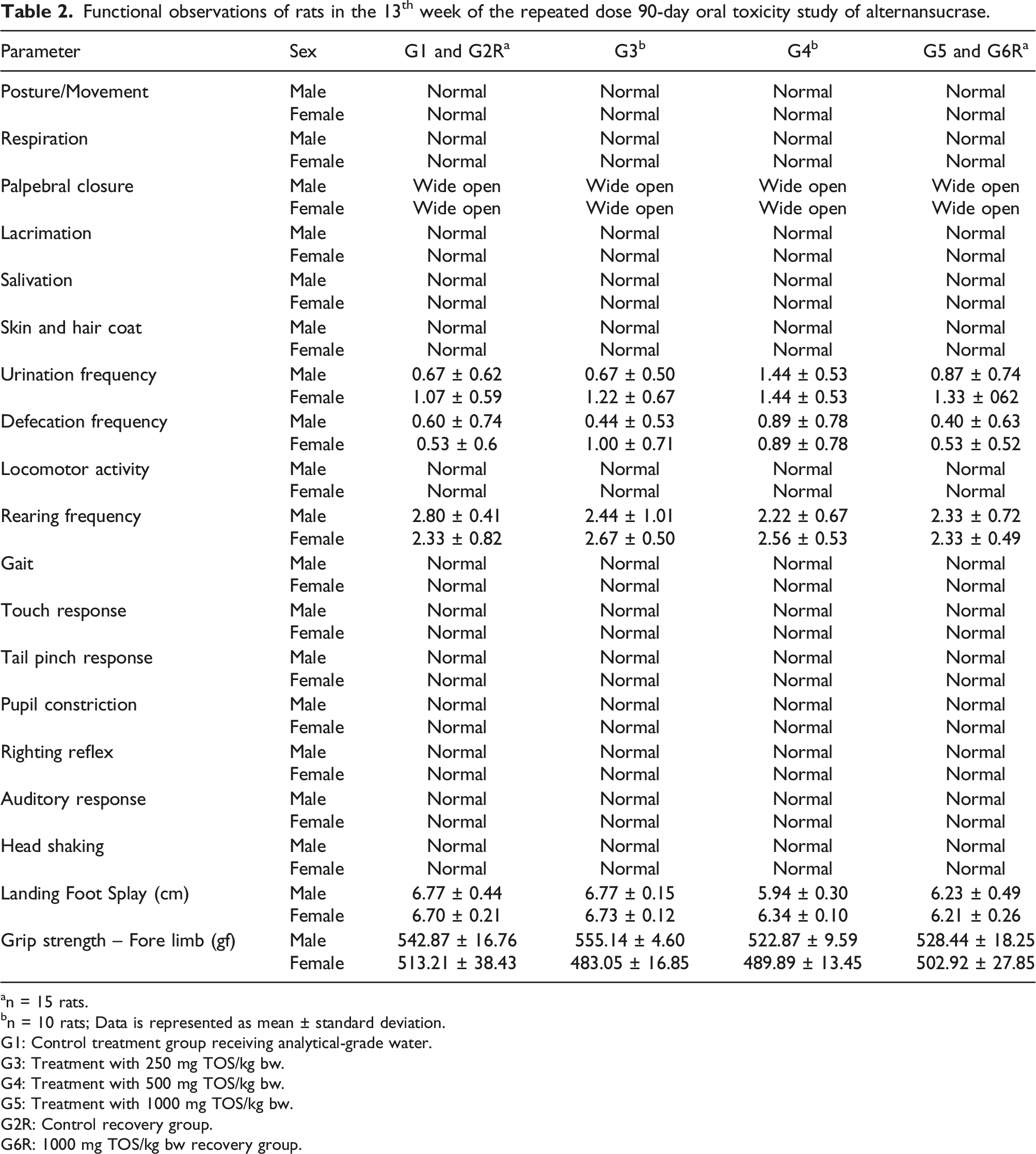
an = 15 rats.
bn = 10 rats; Data is represented as mean ± standard deviation.
G1: Control treatment group receiving analytical-grade water.
G3: Treatment with 250 mg TOS/kg bw.
G4: Treatment with 500 mg TOS/kg bw.
G5: Treatment with 1000 mg TOS/kg bw.
G2R: Control recovery group.
G6R: 1000 mg TOS/kg bw recovery group.
The mean body weights of male rats treated at and up to the dose of 500 mg TOS/kg and female rats at and up to the dose of 1000 mg TOS/kg did not differ significantly (p < .05) from those of the control group rats as shown in Figure 2. A statistically significant decrease (7% to 8%) in mean body weight gain was observed in male rats treated at the dose of 1000 mg TOS/kg body weight. The average daily food consumption per rat per day, computed over the period of 90 days of treatment and further 28 days of recovery, for all rats were between 96% and 102% of that in control rats. Body weights of rats during the 90-day repeated dose 90-days oral toxicity study of alternansucrase (a) Body weights of male rats and (b) Body weights of female rats. 15 animals were tested in groups G1 and G5, 10 animals were tested in groups G3 and G4 and 5 animals were tested in groups G2R and G6R. G1: Control treatment group receiving analytical-grade water; G3: Treatment with 250 mg TOS/kg bw; G4: Treatment with 500 mg TOS/kg bw; G5: Treatment with 1000 mg TOS/kg bw; G2R: Control recovery group; G6R: 1000 mg TOS/kg bw recovery group. Data is represented as mean ± standard deviation.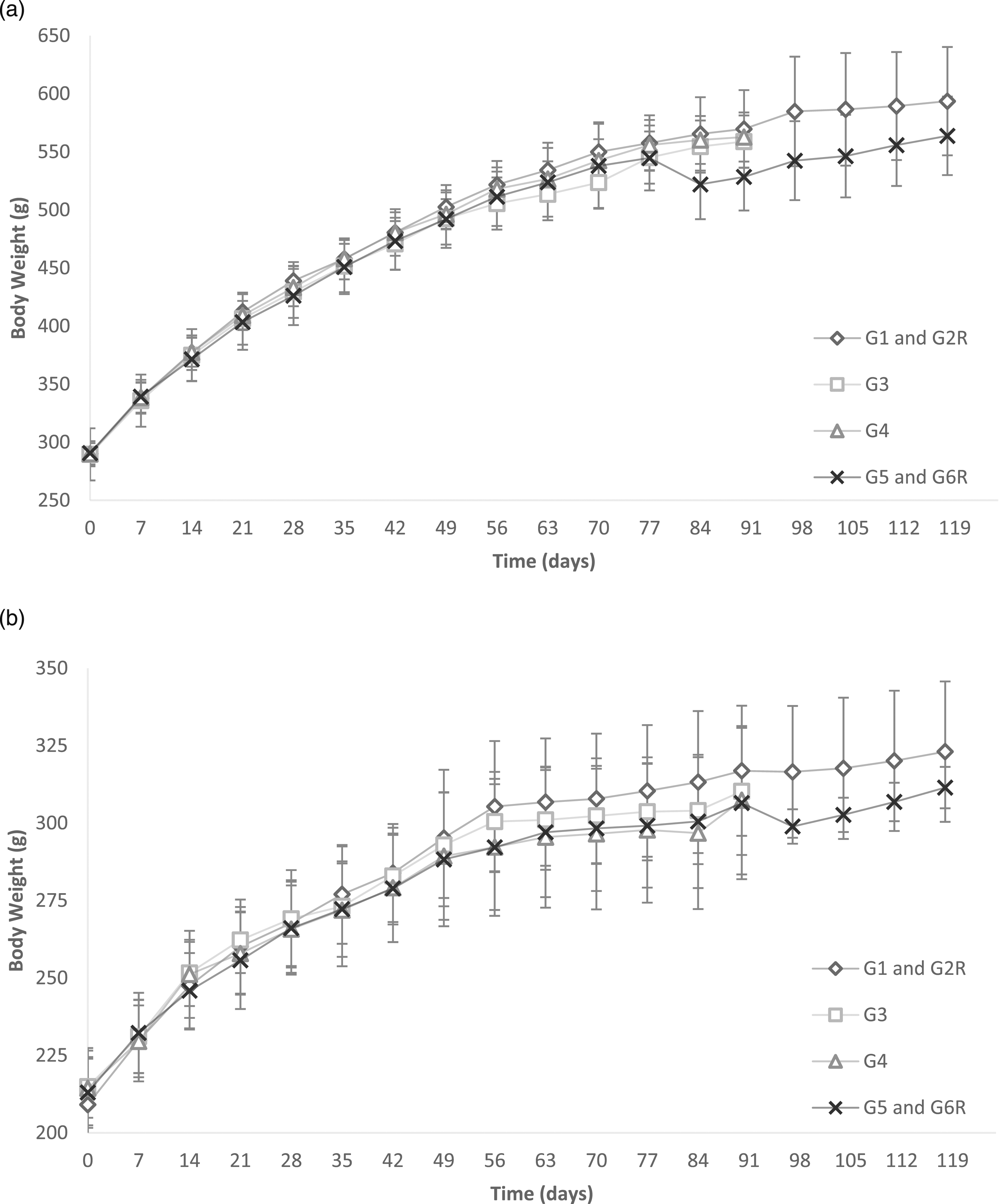
Hematology and coagulation data of rats at the end of the treatment and recovery period, respectively, during the repeated dose 90-day oral toxicity study of alternansucrase.
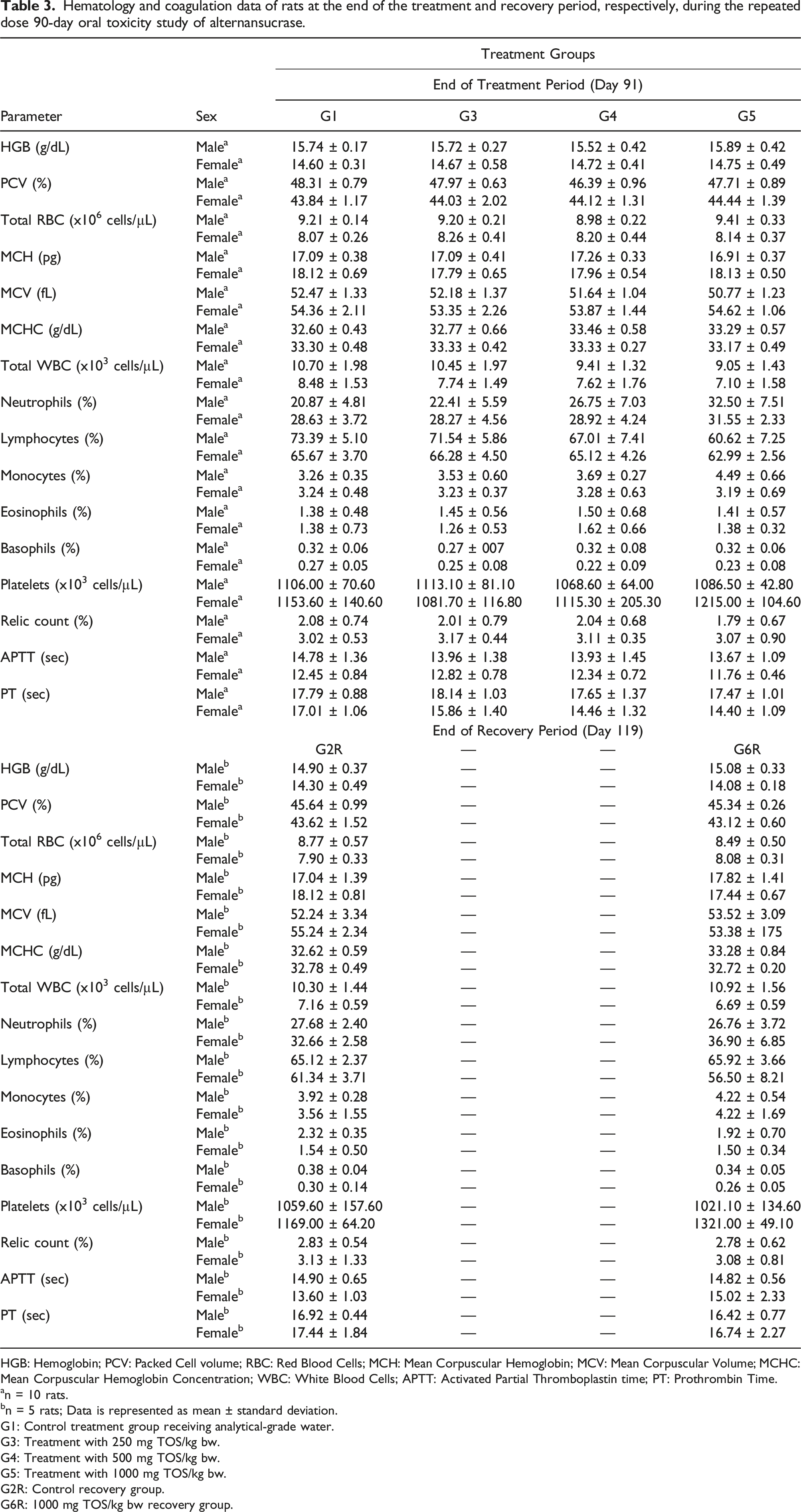
HGB: Hemoglobin; PCV: Packed Cell volume; RBC: Red Blood Cells; MCH: Mean Corpuscular Hemoglobin; MCV: Mean Corpuscular Volume; MCHC: Mean Corpuscular Hemoglobin Concentration; WBC: White Blood Cells; APTT: Activated Partial Thromboplastin time; PT: Prothrombin Time.
an = 10 rats.
bn = 5 rats; Data is represented as mean ± standard deviation.
G1: Control treatment group receiving analytical-grade water.
G3: Treatment with 250 mg TOS/kg bw.
G4: Treatment with 500 mg TOS/kg bw.
G5: Treatment with 1000 mg TOS/kg bw.
G2R: Control recovery group.
G6R: 1000 mg TOS/kg bw recovery group.
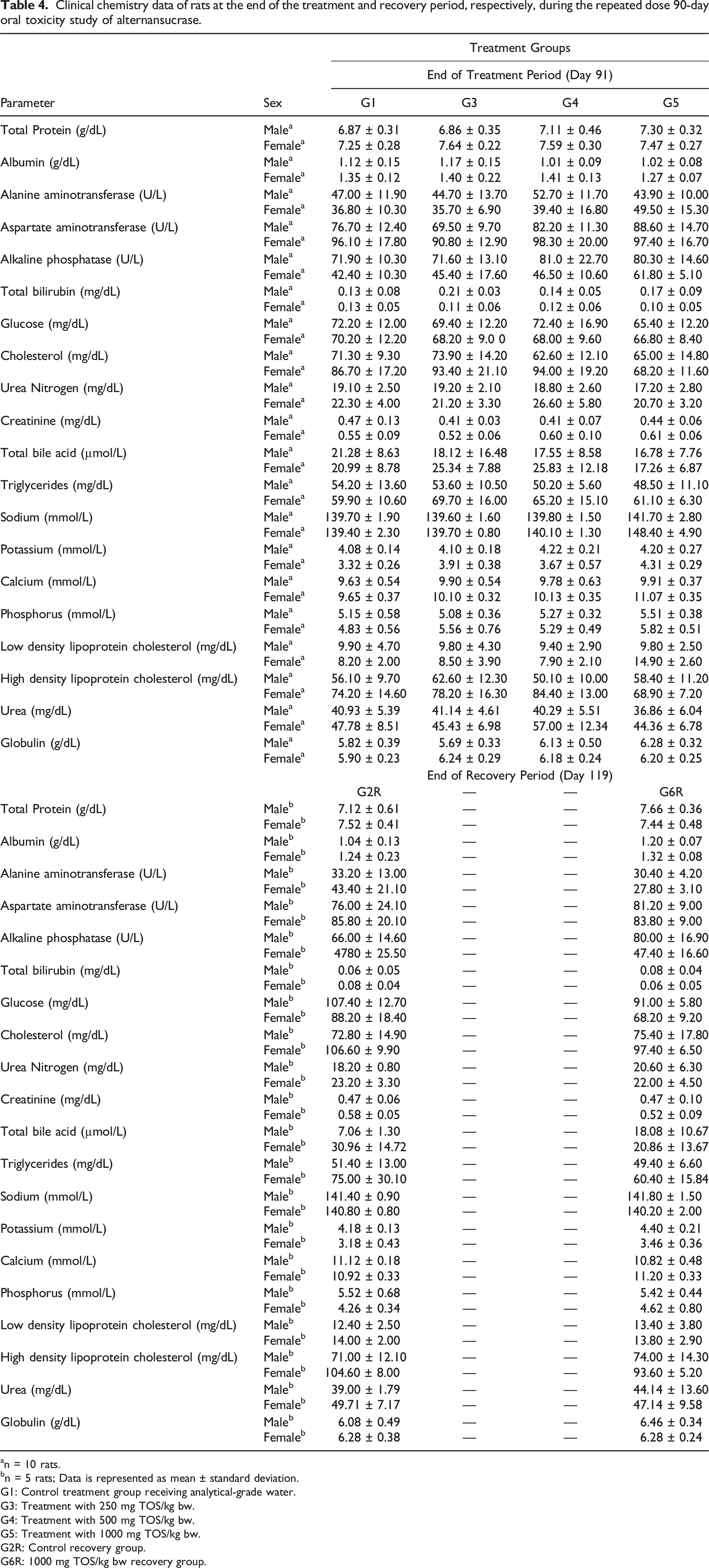
Clinical chemistry data of rats at the end of the treatment and recovery period, respectively, during the repeated dose 90-day oral toxicity study of alternansucrase.
an = 10 rats.
bn = 5 rats; Data is represented as mean ± standard deviation.
G1: Control treatment group receiving analytical-grade water.
G3: Treatment with 250 mg TOS/kg bw.
G4: Treatment with 500 mg TOS/kg bw.
G5: Treatment with 1000 mg TOS/kg bw.
G2R: Control recovery group.
G6R: 1000 mg TOS/kg bw recovery group.
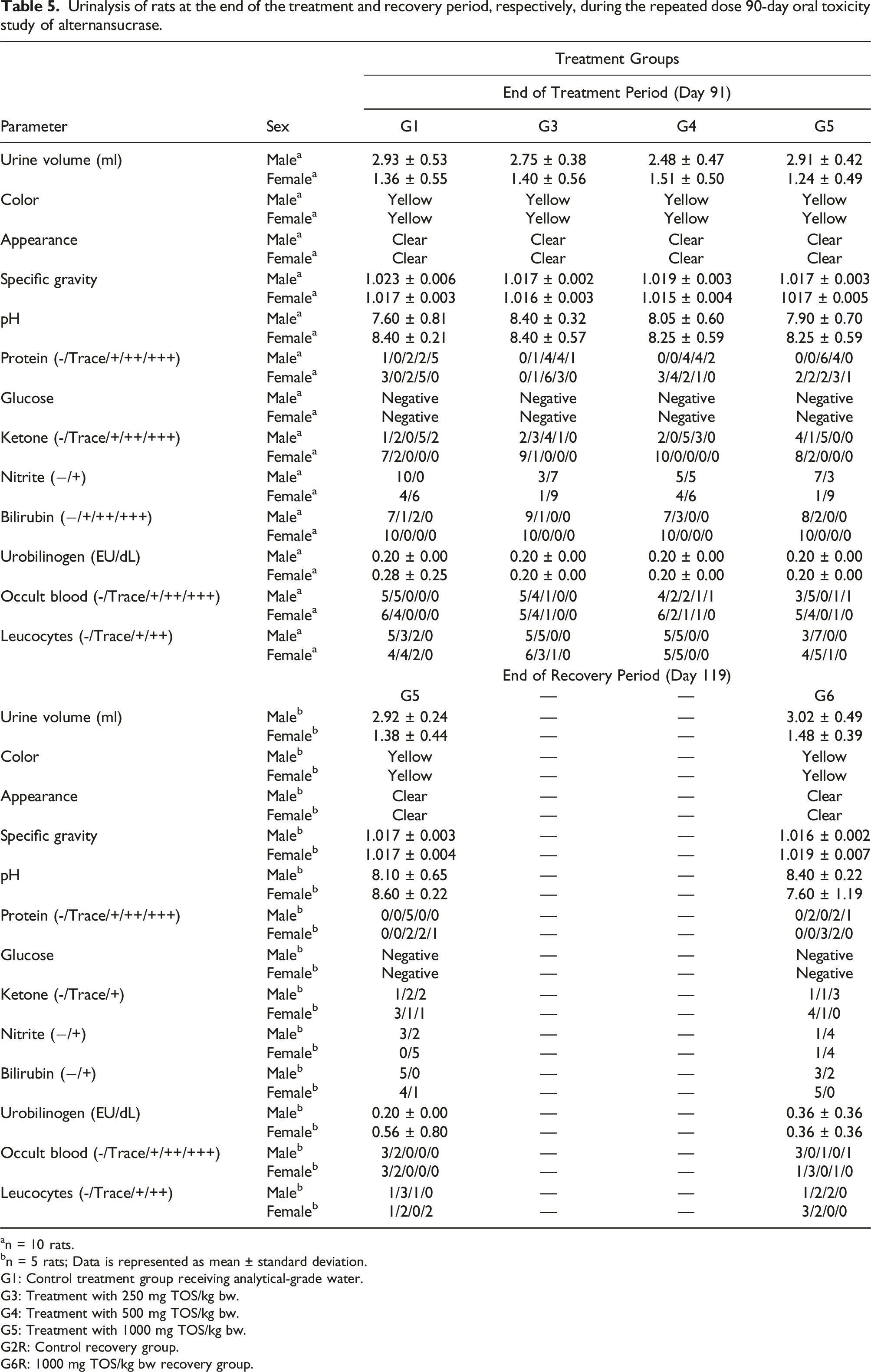
Urinalysis of rats at the end of the treatment and recovery period, respectively, during the repeated dose 90-day oral toxicity study of alternansucrase.
an = 10 rats.
bn = 5 rats; Data is represented as mean ± standard deviation.
G1: Control treatment group receiving analytical-grade water.
G3: Treatment with 250 mg TOS/kg bw.
G4: Treatment with 500 mg TOS/kg bw.
G5: Treatment with 1000 mg TOS/kg bw.
G2R: Control recovery group.
G6R: 1000 mg TOS/kg bw recovery group.
Absolute organ weights of rats at the end of the treatment period and recover period, respectively, for the repeated dose 90-day oral toxicity study of alternansucrase.
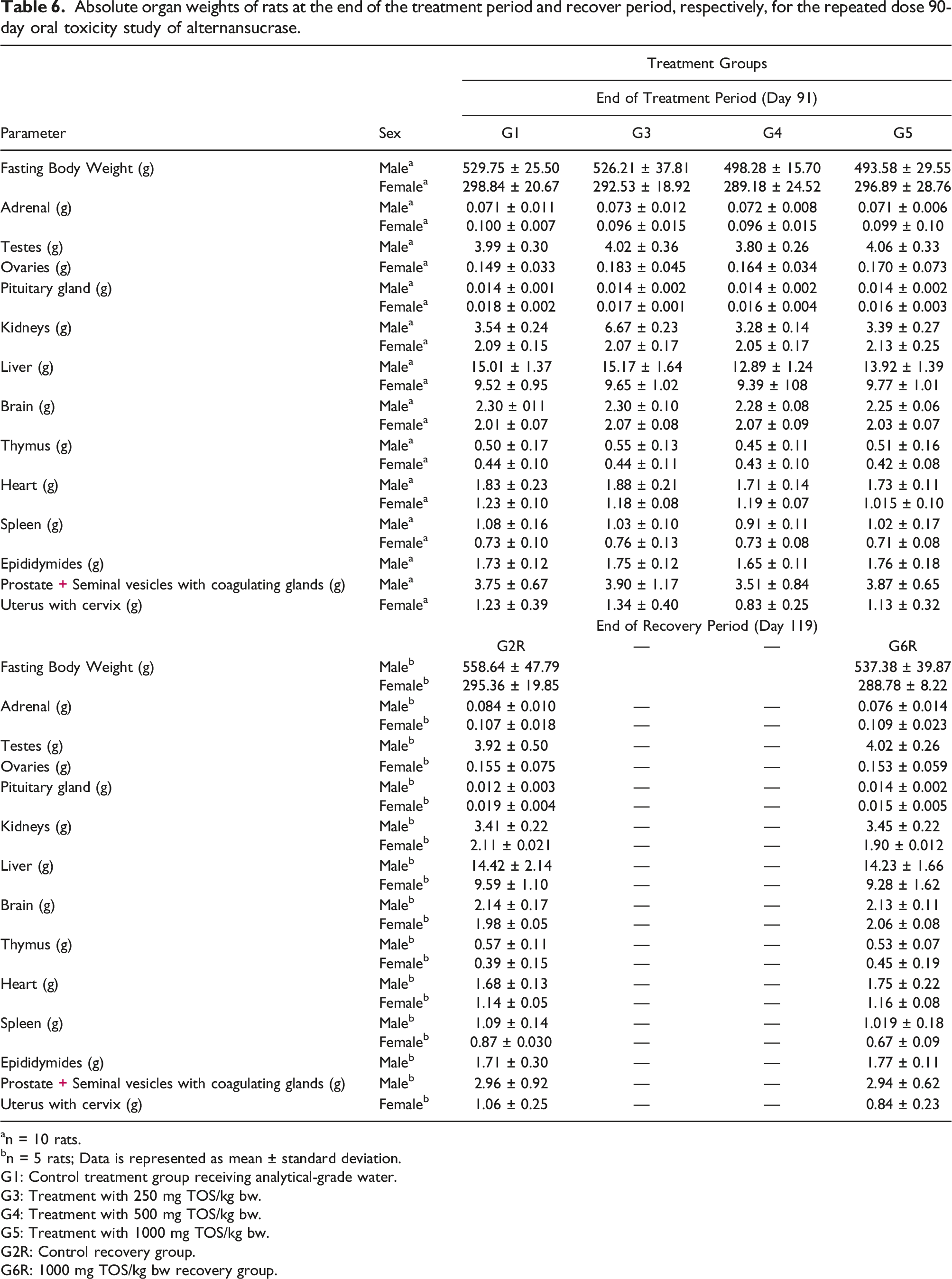
an = 10 rats.
bn = 5 rats; Data is represented as mean ± standard deviation.
G1: Control treatment group receiving analytical-grade water.
G3: Treatment with 250 mg TOS/kg bw.
G4: Treatment with 500 mg TOS/kg bw.
G5: Treatment with 1000 mg TOS/kg bw.
G2R: Control recovery group.
G6R: 1000 mg TOS/kg bw recovery group.
Relative organ weights of rats at the end of the treatment period and recovery period, respectively, for the repeated dose 90-day oral toxicity study of alternansucrase.
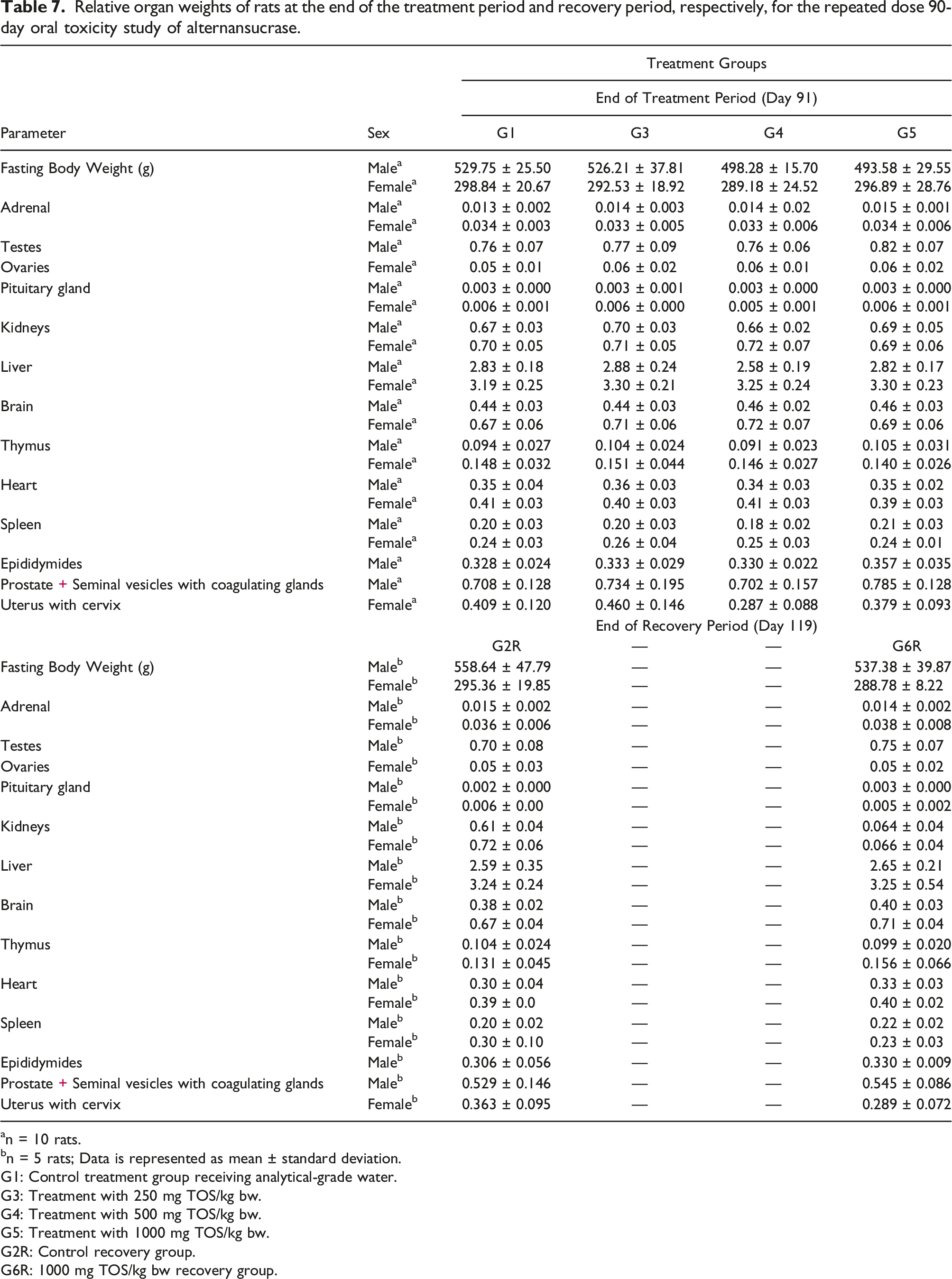
an = 10 rats.
bn = 5 rats; Data is represented as mean ± standard deviation.
G1: Control treatment group receiving analytical-grade water.
G3: Treatment with 250 mg TOS/kg bw.
G4: Treatment with 500 mg TOS/kg bw.
G5: Treatment with 1000 mg TOS/kg bw.
G2R: Control recovery group.
G6R: 1000 mg TOS/kg bw recovery group.
Thyroid hormone levels and organ weight of rats at the end of the treatment and recovery period during the repeated dose 90-day oral toxicity study of alternansucrase.
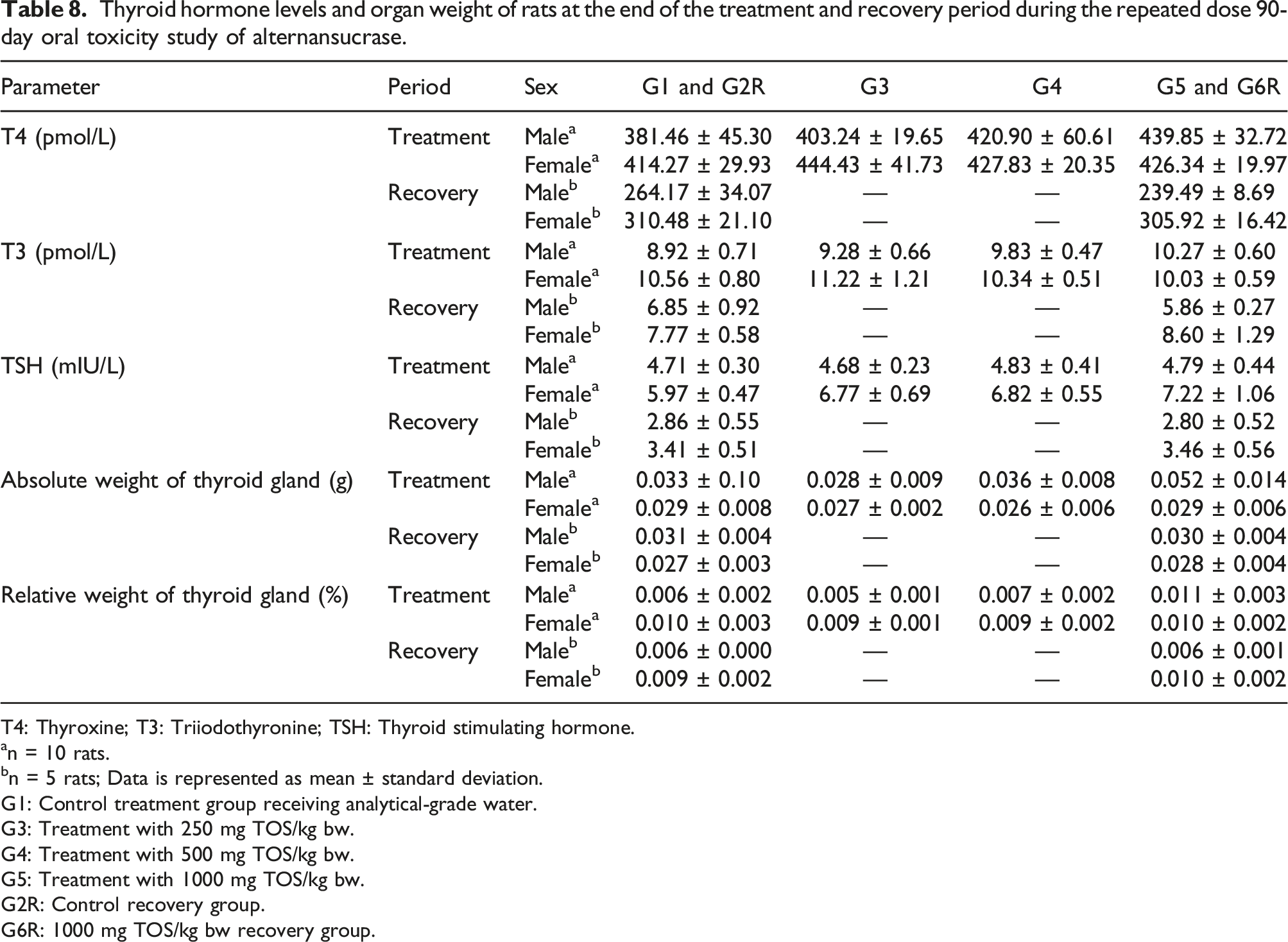
T4: Thyroxine; T3: Triiodothyronine; TSH: Thyroid stimulating hormone.
an = 10 rats.
bn = 5 rats; Data is represented as mean ± standard deviation.
G1: Control treatment group receiving analytical-grade water.
G3: Treatment with 250 mg TOS/kg bw.
G4: Treatment with 500 mg TOS/kg bw.
G5: Treatment with 1000 mg TOS/kg bw.
G2R: Control recovery group.
G6R: 1000 mg TOS/kg bw recovery group.
Bacterial Reverse Mutation Test
A preliminary cytotoxicity study carried out to determine the concentrations to be used during the main study indicated that alternansucrase was non-cytotoxic to the tested strains of S. typhimurium at any of the concentrations in the presence and absence of metabolic activation.
Histidine revertant colonies of S. typhimurium when exposed to various concentrations of alternansucrase during the reverse bacterial mutation assay.
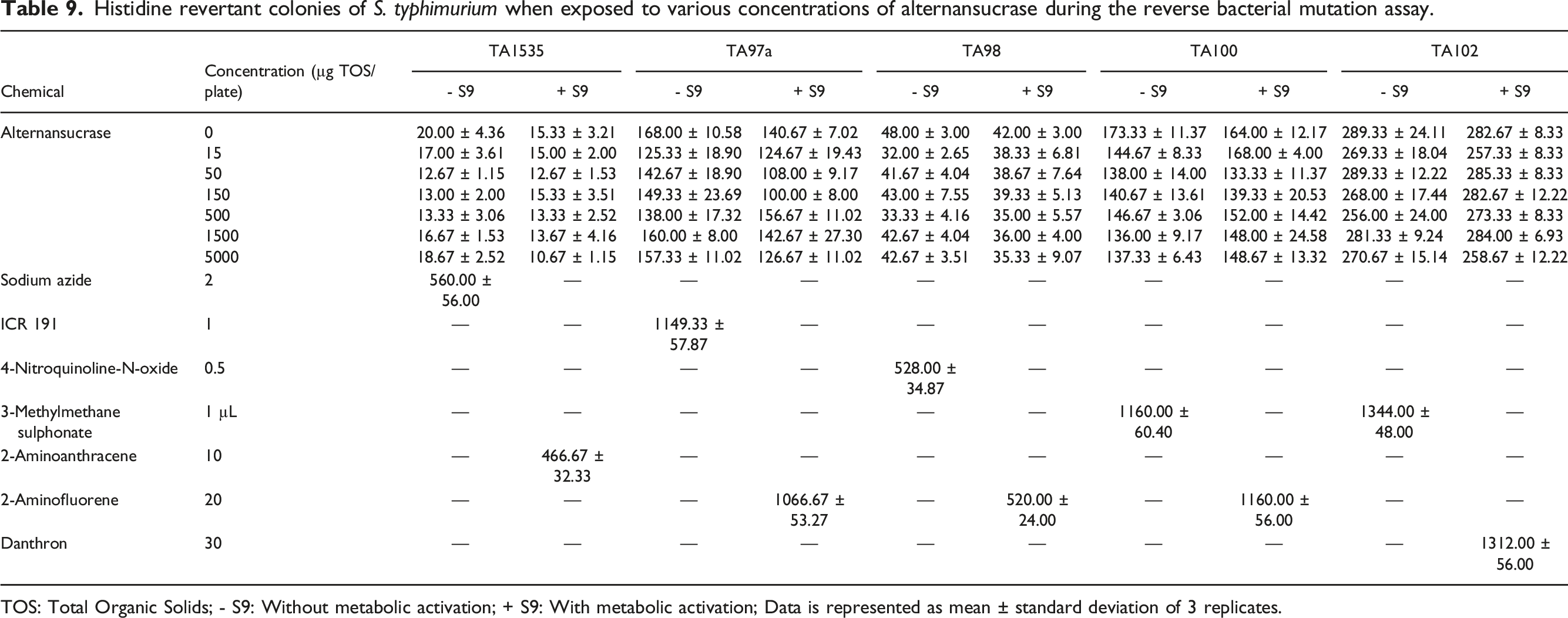
TOS: Total Organic Solids; - S9: Without metabolic activation; + S9: With metabolic activation; Data is represented as mean ± standard deviation of 3 replicates.
Plate counts for the spontaneous histidine revertant colonies in the negative control were found to be within the frequency ranges expected from the laboratory historical control data and compared well with the range reported in the literature. Concurrent positive controls demonstrated sensitivity of the assay with and without metabolic activation as the frequencies of the histidine revertant colonies were significantly higher than those in the negative control.
In vitro Mammalian cell micronucleus test
Incidence of micronuclei and cytotoxicity data for in vitro mammalian cell micronucleus test of alternansucrase with and without metabolic activation.
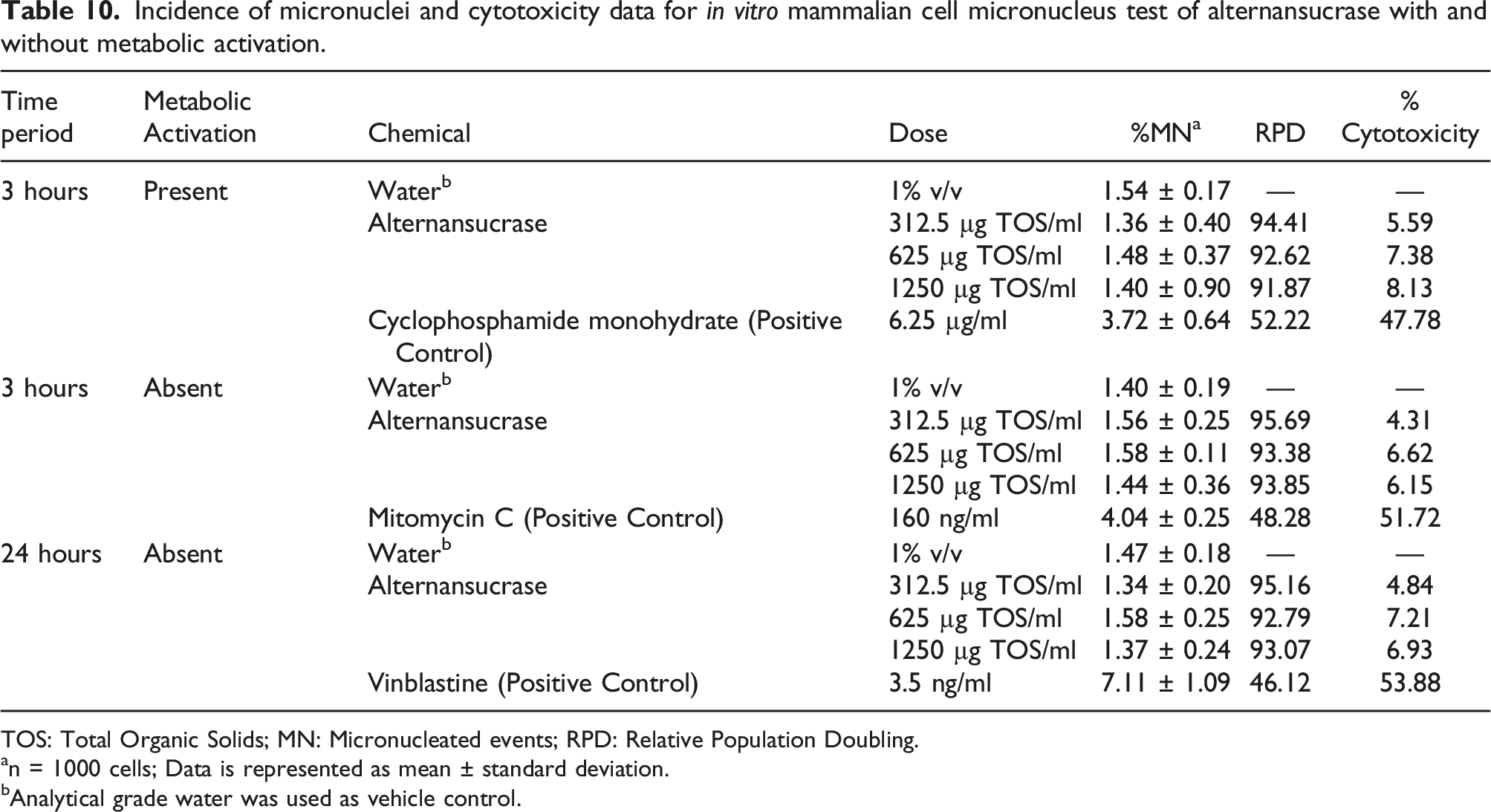
TOS: Total Organic Solids; MN: Micronucleated events; RPD: Relative Population Doubling.
an = 1000 cells; Data is represented as mean ± standard deviation.
bAnalytical grade water was used as vehicle control.
Less than 10% cytotoxicity was observed in TK6 cell line treated with alternansucrase. Likewise, the percentage of micronuclei formation in alternansucrase-treated lymphocytes was comparable to that of the control whereas the percentage of micronuclei for the positive control was significantly higher as compared to the control. Thus, the comparison of the percentage incidence of micronuclei for each of the conducted treatments, either with or without the metabolic activation system, did not reveal any significant and dose-related increase at any of the concentration levels in the cells treated with alternansucrase.
The sensitivity of the test system and activity of the S9 mix were demonstrated in the positive control group. The positive controls, viz. directly acting clastogen mitomycin C, directly acting aneugen vinblastine and indirectly-acting clastogen cyclophosphamide, induced a significant increase in frequencies of micronuclei over the concurrent controls, which validated the test method.
Discussion
Alternansucrase is being increasingly explored for use in dietary supplements and other human nutrition applications. Previously, safety evaluation of alternansucrase from a natural source (L. citreum) has shown the enzyme product to be free from any antimicrobial activity and allergenicity potential. 12 However, no toxicological studies have been ever reported for alternansucrase produced by any micro-organism. To establish the safety, alternansucrase from E. coli was subjected to a series of toxicological tests in compliance with GLP and OECD guidelines. The results from the studies discussed here would help establish the safety of alternansucrase enzyme for human consumption and add to the available knowledgebase of this interesting enzyme.
In the assessment and evaluation of toxic characteristics of any substance, one of the initial steps is the determination of acute toxicity by an oral route which would provide information on health hazards likely to arise due to overdosage in man. The acute oral toxicity study of alternansucrase was performed to rank and classify alternansucrase according to the Globally Harmonized System for the classification of chemicals. In principle, the acute toxicity class method is not intended to allow the calculation of LD50 value, but it does allow for the determination of defined exposure ranges where lethality is expected since the death of a proportion of the animals is still the major endpoint of the test. Based on a stepwise process, information was obtained on the acute toxicity of alternansucrase to enable its classification. The absence or presence of alternansucrase-dependent mortality of the dosed rats at a given step would help determine the dose to be gavaged at the subsequent steps. Since no mortality of rats was observed at all the tested doses of alternansucrase, it was classified in GHS Category 5 or unclassified for the obligatory labeling requirement for oral toxicity. The corresponding LD50 cut-off value for alternansucrase was determined to be 5000 mg/kg body weight.
Based on the acute toxicity study results, the doses of 250 mg/kg body weight, 500 mg/kg body weight and 1000 mg/kg body weight were selected for the repeated dose 14-day oral toxicity study. The purpose of this study was to assess the toxicological profile of alternansucrase and as a preliminary study to the repeated dose 90-day oral toxicity study. The results of this study would provide information on target organs and the possibilities of cumulative toxicity. The statistically insignificant changes in the organ weights of rats (besides for heart) during this study were not considered as alternansucrase-related due to lack of dose dependency and their comparability to the historical control group data at the test facility. Since none of the selected doses induced any abnormal clinical manifestations or mortality, the doses of 250 mg/kg, 500 mg/kg and 1000 mg/kg were opted for repeated dose 90-day oral toxicity study of alternansucrase.
The repeated dose 90-day oral toxicity study was executed to establish information on the possible accumulation and reversibility of toxic effects of alternansucrase as well as to estimate the No-Observed-Adverse-Effect-Level (NOAEL) to determine the safety criteria of alternansucrase for human exposure.
In this study, no test item-associated abnormal clinical signs were noted which could be suggestive of motor dysfunction. In addition, variations in the functional observations did not display any dose-dependency leading to the conclusion that the decrease in forelimb grip strength & landing foot splay were incidental. All the functional observations taken together did not reveal any neurotoxic potential of the enzyme alternansucrase. The statistically significant decrease in mean body weight gain observed in male rats treated at the dose of 1000 mg TOS/kg body weight was slight (7% to 8%) and considered toxicologically insignificant due to a lack of dose-dependency.
The observations of variations in the hematology parameters in the rats were incidental as they were found to be not dose-dependent for the male rats and were not observed in the main groups for the female rats. Although a dose-dependent decrease in the MCV of male rats and in prothrombin time of female rats was noticed, these were determined to be toxicologically insignificant as the observed values were within the historical control range at the test facility. A statistically significant change was observed in the differential white blood cell count only at the highest dose administered and was found to be reversible and non-adverse as ascertained by the return to normalcy during the recovery period. Most of the differences in the clinical chemistry parameters reported after treatment and after the recovery period were recognized to be not dose-dependent and were deemed incidental. The increase in alkaline protease content of female rats was observed to be dose-dependent and significant but was inferred to be toxicologically insignificant since no correlated changes were noted in the other tested blood parameters. Although a dose-dependent increase in sodium and calcium contents of female rats was also observed, it was deduced to be incidental as well as the reported values were within the historical control range recorded at the test facility. The changes in the urine pH and urobilinogen of rats were deduced to be statistically insignificant whereas an incidental decrease in the specific gravity of urine of the male rats was determined to be not dose dependent.
The statistically significant changes observed in weight of liver, kidneys and uterus with the cervix at the end of the recovery period were considered as incidental and not to be treatment-related due to their comparability to historical control values or due to lack of dose dependency amongst treatment groups. The increase in thyroid hormones was considered a test item-related effect. Moreover, in this study, there were no abnormal clinical signs suggestive of any dysfunction of thyroids. At necropsy there were no gross pathological findings in the thyroid and the histopathological evaluations of the thyroid in both sexes did not reveal any abnormalities. Also, reversal was observed in thyroxine (T4), triiodothyronine (T3) and thyroid stimulating hormone (TSH) levels and absolute and relative thyroid gland weights at the end of the recovery period. Hence the increase in thyroid hormone levels and absolute and relative weights of the thyroid gland were considered non-adverse.
Based on these findings and observations, the No-Observed-Adverse-Effect-Level (NOAEL) of alternansucrase following oral administration for 90 days was 1000 mg TOS/kg body weight.
A notable limitation of the toxicological studies carried out in rats is the extrapolation of the results obtained to humans. 18 These extrapolations include inter-species extrapolations as well as dose related extrapolations. The limitation can be attributed to the fact that humans and rats are different mammalian species and the results obtained in rat models may not necessarily be directly extrapolated to humans. Another limitation of these tests is that the tests are carried out by providing high doses of the test time over a short time period. The safety evaluation of the test item in these conditions may or may not be applicable if the test item is consumed in low doses but over a large period. Nonetheless, there is enough historical evidence to suggest that the safety evaluations carried out in rats have been applied to humans with no adverse effects. Inclusion of in vitro tests for toxicity analysis has paved way to circumvent some of these limitations.
Mutagenic nature of the test item alternansucrase was evaluated using the bacterial reverse mutation test. The objective of the bacterial reverse mutation assay was to evaluate the potential of the alternansucrase to induce point mutations which revert the mutations already present in the tester strains of Salmonella typhimurium and to restore the functional capability of bacteria to synthesize an essential amino acid, viz. histidine. The study would provide information on genotoxic hazards likely to arise from exposure to alternansucrase. Alternansucrase was determined to be non-mutagenic under the conditions of the study in the tested strains of Salmonella typhimurium at all concentrations.
The in vitro mammalian cell micronucleus test was performed to evaluate the potential of alternansucrase to induce the formation of micronuclei or small membrane-bound DNA fragments in the cytoplasm of interphase cells. These micronuclei usually originate from acentric fragments (chromosome fragments lacking a centromere) or whole chromosomes that are unable to migrate to the poles during the anaphase of cell division. The assay was thus used to detect the clastogenic and aneugenic activity of alternansucrase. The test was carried out using the TK6 human lymphoblastoid cell line as the test system as this was a highly validated system. It has been reported to be stable (p53 competent, karyotypically stable and DNA-repair proficient), permanent cell type with a modal chromosome number of 47, cell cycle time of 13-15 hours. This cell line has also been reported to produce fewer misleading positive results than other cell lines. Based on the observed results, it was concluded that alternansucrase did not induce a significant increase in the incidence of micronuclei over the tested range which indicated that, under the test conditions, alternansucrase was found to be non-clastogenic and non-aneugenic.
A significant limitation of the in vitro tests is that these tests prove the cytotoxic effect by detecting the surviving cells. These tests do not indicate any tissue-specific or interactive cytotoxic effects as well as any other mild to moderate effect that may not be inhibitory to cell survival but may affect its functioning. 18
The toxicological studies reported here indicate that alternansucrase can be considered safe for use in food/feed applications. Safety evaluation of alternansucrase obtained from Leuconostoc citreum, as reported in EFSA, 2021, shows that this enzyme gives rise to no safety concerns for use in food applications. Toxicological studies were not conducted in this case as the production organism was QPS i.e., Qualified Presumption of Safety, and therefore is considered safe. 12 Similar toxicological studies were conducted for another glucosyltransferase enzyme, namely, 1,4-α-glucan 6-α-glucosyltransferase, from Paenibacillus alginolyticus. 19 The enzyme was subjected to a battery of toxicological studies and was found to be non-mutagenic and did not induce chromosomal aberrations. 90-day oral toxicity study of 1,4-α-glucan 6-α-glucosyltransferase, no significant dose-dependent toxicological effects were observed. 19 The authors concluded the NOAEL for this enzyme to be 24 mg TOS/kg of body weight. This is much lower that the NOAEL reported for alternansucrase in the present study, indicating that the enzyme evaluated in these studies is safer that other similar naturally occurring glucosyltransferase.
Conclusion
In summary, five different toxicological studies were performed on alternansucrase from E. coli to establish its safety. No morbidity was observed in rats in the acute and repeated oral-toxicity studies. Alternansucrase was classified in Globally Harmonized System (GHS) category 5 or unclassified and the LD50 cut-off value was determined to be 5000 mg/kg body weight based on the reported acute oral toxicity study data. Alternansucrase, at a maximum dose up to 2000 mg/kg body weight, did not lead to any abnormalities in the tested Wister rats. Alternansucrase had a No-Observed-Adverse-Effect-Level (NOAEL) of 1000 mg TOS/kg body weight in rats, the highest dose administered to the rats during the sub-chronic repeated dose 90-day oral toxicity study.
At the genotoxic level, alternansucrase enzyme preparation is non-mutagenic to tested Salmonella typhimurium strains TA1535, TA97a, TA98, TA100 and TA102 up to the test concentration of 5000 µg TOS/plate. Micronucleus test preformed with human lymphoblastoid T6 cell lines proved that alternansucrase has no cytogenetic or aneugenic effect at concentrations of 1250, 625 and 312.5 μg TOS/ml with and without metabolic activation system (S9). Overall, we conclude that alternansucrase can be considered safe to be used as food enzyme/food processing agent in various industrial applications. The reported study is a landmark study to further strengthen the safety assessment ladder for alternansucrase enzyme preparation.
Supplemental material
Supplemental material - Safety evaluations of alternansucrase enzyme expressed in Escherichia coli shows No adverse effects in-vivo and in-vitro
Supplemental material for Safety evaluations of alternansucrase enzyme expressed in Escherichia coli shows No adverse effects in-vivo and in-vitro by Ankit Rathi, Harshada Chandan, Chaitali Vira and Mukul P Pore in Toxicology Research and Application.
Footnotes
Acknowledgments
The authors would like to thank the Mr. V. L. Rathi, Mr. M. M. Kabara and Dr. Abhijit Rathi for their technical guidance and support for this study.
Author contributions
AR and HC were involved in the conceptualization of study and project administration. MP was the study director and was responsible methodology development and investigation. All authors (AR, HC, MP and CV) were responsible for formal analysis of the study. CV was responsible for writing the original draft manuscript. All authors (AR, HC, MP and CV) were responsible for critical evaluation (review and editing) of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Advanced Enzyme Technologies Ltd. funded the studies carried out at Intox.
Consent for publication
All the authors have agreed to submission of this manuscript for peer review and publication.
Ethical statement
Data availability statement
Supplemental material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.