Abstract
The immobility of plants exerted evolutionary selection pressures resulting in the production of thousands of chemical substances thought to function as pesticides against predation by insects and animals. More than 10,000 plant-derived compounds have been isolated with the existence of about 100,000 such compounds postulated. In 1990, Ames et al. reported that 99.99% by weight of the pesticides ingested in a normal human diet are derived from natural plant-based sources. This surprising result raised the question as to whether these natural plant pesticides were toxic to humans. These authors examined a relatively small subset of natural pesticides and determined that their tumorigenicity in rodent cancer bioassays was similar to synthetic pesticides. In this analysis, we used standard United States Environmental Protection Agency programs to estimate the toxicity (T.E.S.T. 4.2) and persistence (EPI Suite 4.1) of a series of high-volume synthetic and natural pesticides. On average, synthetic pesticides were more persistent in the environment than were natural pesticides. This result is consistent with cost, time, and logistical constraints under which farmers apply a limited number of applications of pesticides during a crop cycle. Synthetic and natural pesticides are predicted to possess toxicities including mutagenicity and developmental toxicity. Synthetic pesticides are less often mutagenic.
Keywords
Introduction
DNA evidence suggests that the first plants might have colonized land about 700 million years ago. The first fossil evidence of land plants dates from 510 to 439 million years ago (Ordovician Period) in the form of wind-dispersed spores believed to have been produced by submerged plants that raised their sporangia above the water. 1 Flowering plants, termed angiosperms, are a more recent but still ancient arrival with the first fossil evidence appearing about 125 million years ago during the Lower Cretaceous, with a high degree of diversification by the Middle Cretaceous about 100 million years ago. 2 Insects coevolved with plants with the earliest fossilized insects dating from about 412 million years ago during the Early Devonian Period. However, phylogenetic data suggest that Hexapoda, the largest group of insects, might have evolved about 479 million years ago during the Early Ordovician Period. 3
Pesticides are applied to prevent or control pests, diseases, weeds, and other plant pathogens to ameliorate or eliminate yield losses and maintain high agricultural production quality. Pesticides (both natural and synthetic) are regulated by governments and are premarket tested by employing standardized test methods aimed at achieving minimal impacts on human health and the environment. Under conditions of inappropriate application, unacceptable residue levels can accumulate in water, soil, or ambient air. Contamination from soil leaching, field runoff water, and spray drift, as well as adverse effects on wildlife, fish, plants, and other nontarget organisms can potentially occur. Actualization of potential effects depends on the specific toxicity, quantity and potency of the applied pesticide, safety precautions taken during application, adsorption onto soil colloids, weather conditions prevailing after application, and how long the pesticide persists in the environment. 4 Potential health effects of pesticide exposures are dependent on the biological pathways and organ systems affected. Potential adverse health effects include skin and eye irritation, carcinogenicity (as estimated by rodent cancer bioassay), disruption of endocrine pathways, and interference with neural transmission in the case of some of the organophosphates and carbamates. 5
The immobility of plants exerted evolutionary selection pressures resulting in the production of thousands of chemical substances thought to function as pesticides against predation by insects and animals. In 1990, Ames et al. reported that 99.99% of pesticide exposure experienced by humans (via diet) comes from natural pesticides produced by plants in contrast with trace residues of synthetic pesticides applied to the plants. 6 This contention by Ames et al. is supported by a comprehensive review by Duke. 7 Duke noted that tens of thousands of secondary products of plants have been identified as pesticides. Plant-produced compounds and derivatives with pesticidal activity include 1,8-cineole, cinmethylin, hypericin, and pyrethrins. 7 Plant-produced compounds with insecticidal activity include camphene, nicotine, anabasine, and rotenone. 7 Plant-produced compounds with fungicidal, nematicidal, and rodenticidal activity include pisatin, juglone, alpha-terthienyl, and strychnine. 7 It has been estimated that hundreds of thousands of these secondary plant products exist. The majority of this huge number of chemicals produced by plants “are involved in the interaction of plants with other species—primarily the defense of the plant from plant pests.” 7 Anyone who eats vegetables is exposed to a subset of these natural plant pesticides.
Farmers apply pesticides to improve crop yield. Most pesticides used today are synthetic. However, smaller quantities of both organic and inorganic natural pesticides are also employed. These natural pesticides derive from biochemical, microbial, botanical, or mineral sources.
8,9
Biochemical pesticides, for example pheromones, can disrupt mating behavior thereby controlling insect populations. Bacteria (e.g. δ-aminolevulinic acid), fungi, algae, and naturally occurring viruses or protozoans can be used to produce microbial pesticides. These natural plant pesticides either introduce a pathogen to a particular insect population (e.g. Milky spore),
4
produce a substance toxic to insects, or limit reproduction. A number of plant-derived botanical pesticides have been isolated including nicotine, anabasine, and nornicotine from tobacco; azadirachtin B from neem seeds; linalool from tree bark; rotenone from derris plant; 1,8-cineole from eucalyptus tree; physostigmine from Calabar bean or manchineel tree; pyrethrins, jasmolins, and cinerins from chrysanthemum plant; and
The purpose of this study was to determine whether there are differences in persistence and toxicity between high-volume synthetic and natural pesticides. In this analysis, we used standard United States Environmental Protection Agency (US EPA) programs to estimate the toxicity (Toxicity Estimation Software Tool (T.E.S.T.)) 10 and persistence and bioaccumulation (Estimation Programs Interface (EPI) Suite™ 4.0) 11 of a series of synthetic and natural pesticides. Thirty-four natural plant pesticides and 32 synthetic pesticides were included in this analysis. These subsets of natural and synthetic pesticides were selected because of their relatively high volume as compared with other members of the class, and widely distributed patterns of use. As such, the natural and synthetic pesticides studied herein represent leaders in the respective classes regarding their commercial importance and toxicological relevance to environmental and human health.
Methods
Compilation of natural and synthetic pesticides
A literature search of the databases Google, PubMed (National Library of Medicine), and the Hazardous Substances Data Bank (National Library of Medicine) was conducted. Key search words included the following: natural plant pesticides, natural pesticides, synthetic pesticides, pesticides, and persistent organic pollutants (POPs). This search strategy identified four major publications describing naturally occurring pesticides: Duke, 7 McLaren, 12 Russell, 13 and Mpumi et al. 14 The synthetic pesticides that are identified as POPs are listed within the United Nations Environment Programme, Stockholm Convention (POP chemicals are listed in both Annex A and Annex C) 15 and/or the United Nations Economic Commission for Europe Protocol. 16 Information on the Top 10 pesticides (poundage of active ingredient) was obtained from EPA and was based on Paisley-Jones. 17
Thirty-two synthetic pesticides were included in this analysis. The 32 represent the Top 10 pesticides (poundage of active ingredient) sold worldwide and 22 other popular pesticides used internationally. There are only 8 pesticides common to the set of 32 synthetic pesticides considered in this study, and 514 pesticides previously tested by US EPA 18 (2,4-dichlorophenoxyacetic acid (2,4-D); acetochlor; atrazine; chloropicrin; chlorothalonil; glyphosate; metolachlor; and pentachlorophenol). Seven of the eight common synthetic pesticides are on the Top 10 list (poundage of active ingredient) of pesticides sold worldwide. Of the 32 synthetic pesticides evaluated in this study, 62% are rodent carcinogens as determined by chronic cancer bioassays. 19 Of the Top 10 pesticides by poundage of active ingredient sold worldwide, 30% are rodent carcinogens (Table 1). 17
Information on synthetic and natural pesticides examined in this study.
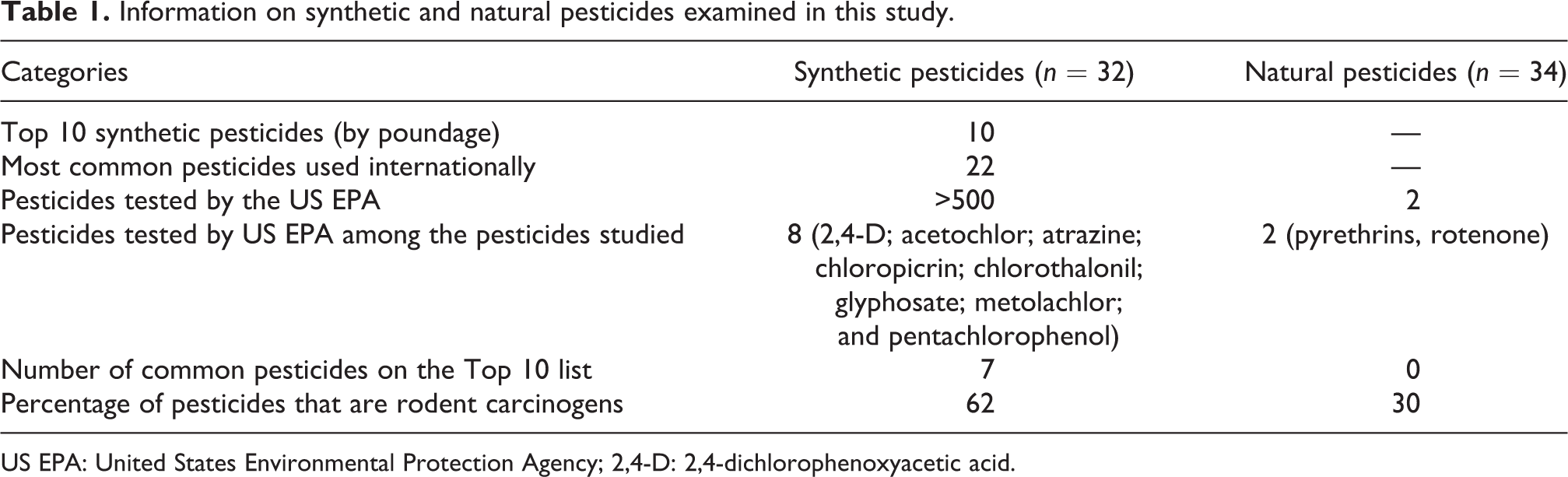
US EPA: United States Environmental Protection Agency; 2,4-D: 2,4-dichlorophenoxyacetic acid.
The 34 natural plant and bacterial extracts analyzed in this study represent the high-volume botanicals and microbial extracts used as commercial pesticides.
7,19
The 34 natural pesticides described herein represent only a small fraction of the natural products extracted from plants
20
and bacteria that have been employed as a pesticide, albeit usually under limited circumstances and at relatively low levels. The 1975
Computer programs for estimating persistence, bioaccumulation, and toxicity
Two computer programs were used to develop the tables of data on naturally occurring pesticides and the synthetic pesticides—T.E.S.T. and EPI Suite.
T.E.S.T. (version 4.2) 10 was used to determine the toxicity of several natural pesticides, the Top 10 synthetic pesticides used worldwide, and synthetic pesticides on the Stockholm list of POPs. 15,16 The T.E.S.T. program contains experimental values for the toxicity endpoints. It also can estimate toxicity from molecular structure. T.E.S.T. was developed by the US EPA to allow users to easily acquire experimental data or estimate the toxicity of chemicals using quantitative structure activity relationships (QSARs) methodologies. In QSAR mode, T.E.S.T. estimates the toxicity values and physical properties of organic chemicals based on the molecular structure of the organic chemical entered by the user. T.E.S.T. allows a user to estimate toxicity without requiring any external programs. Users input a chemical to evaluate by drawing into an included chemical sketcher window, entering a structure text file, or importing from an included database of structures. Once entered, toxicity is either provided from experimental data or estimated using one of several advanced QSAR methodologies. The required molecular descriptors are calculated within T.E.S.T. The methodology used for calculations in this study is termed “Consensus.” The Consensus method was shown to achieve the best prediction results as determined by external validation. 10
The endpoints of toxicity contained in, or calculated by, T.E.S.T. 10 are the following:
96 h fathead minnow 50% lethal concentration (LC50).
48 h
Oral rat 50% lethal dose (LD50).
Bioconcentration Factor (BCF)—the BCF data set was compiled by researchers at the Mario Negri Istituto Di Ricerche Farmacologiche. The bioaccumulation factor (BAF) is the ratio of the chemical concentration in fish as a result of absorption via the respiratory surface to that in water at steady state. 10
Developmental Toxicity (DevTox).
Ames Mutagenicity (Mutagenicity).
In all cases, the experimental values for the toxicity endpoints were used when available. These were obtained from US EPA programs including ECOTOX,
24
TETRATOX,
25
ChemID
The EPI Suite is a computer program designed to derive physical/chemical property and environmental fate estimations. 11,24 It was developed by US EPA and the Syracuse Research Corp. (SRC). EPI Suite is a screening-level tool and should not be used if acceptable measured values are available.
For this analysis of natural and synthetic pesticides, the following EPI Suite modules were used: BIOWIN3: BIOWIN™ estimates aerobic and anaerobic biodegradability of organic chemicals using seven different models. Biowin3 (Ultimate Survey Model) and Ready Biodegradability modules were used.
11,24
KOWWIN: KOWWIN™ estimates the log octanol–water partition coefficient, log KOW, of chemicals using an atom/fragment contribution method. Many of the log octanol–water partition coefficients have been experimentally measured. When experimental values were available these were used rather than the calculated log KOW values.
11,24
BCFBAF: BCFBAF™ is the calculated fish BCF and its logarithm using two different methods. The first is a traditional regression based on log KOW plus any applicable correction. The second is the Arnot–Gobas method, which calculates BCF from mechanistic first principles. BCFBAF also incorporates prediction of apparent metabolic half-life in fish and estimates BCF and BAF for three trophic levels.
11,24
Level III Fugacity Model—LEV3EPI™: This program contains a level III multimedia fugacity model and predicts partitioning of chemicals among air, soil, sediment, and water under steady state conditions for a default model “environment.” Additionally, a composite value of persistence is calculated. This composite persistence value was used in the tables of this analysis.
11,24
Mathematical treatments
Hypothesis tests for multiple proportions
Given more than one sample from which proportions are observed, a hypothesis test is usually used to assess whether differences in the proportions are statistically significant. The different samples may be taken from the same population at different times, from the same population with different experimental treatments, or from different groups.
The methods described in the following text are among those designed according to the “classical approach to hypothesis testing.” In the “classical approach,” an exact assumption is made about the population’s characteristics, usually but not always, restricted to an assumption about the value of a population parameter such as the true proportion, mean or standard deviation. This assumption is called the null hypothesis and is so named because it is usually an assumption of either no difference or no change. A test statistic is chosen that will have an exact distribution, or at least be close to the same distribution, when the null hypothesis is true. The probabilities of the values of the test statistic that are less likely than the value calculated from the sample(s) are summed to obtain what is termed the
Commonly used tests for two proportions
Given two samples of sizes Pooled test
The null hypothesis is
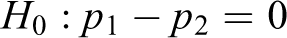
The formula for the pooled test statistic comparing two proportions is
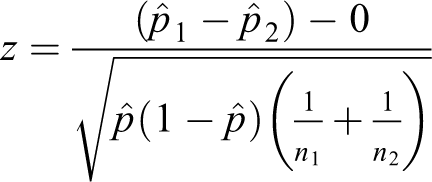
where
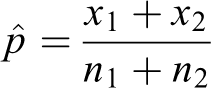
The standard error is
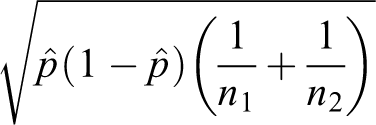
Unpooled test
The null hypothesis is
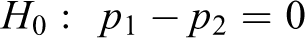
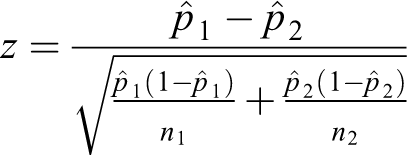
Chi-squared statistic
The Chi-squared (
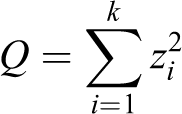
is distributed according to the
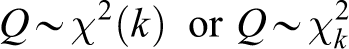
The
p Values from binomial tail probabilities
Given two samples of sizes
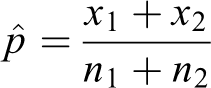
that is
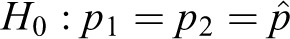
The test statistic is simply the outcome (
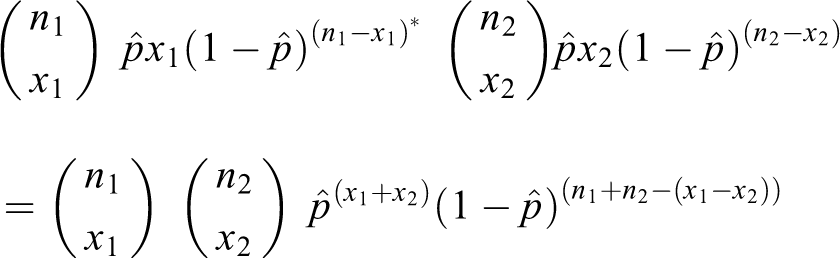
The
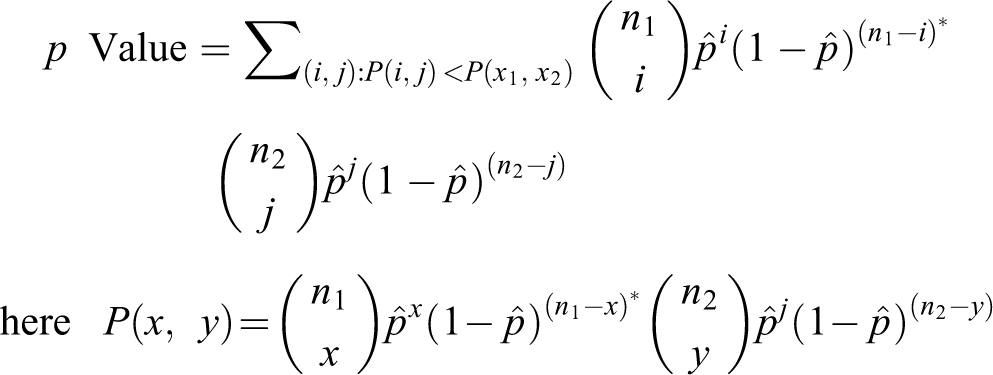
This summarizes to
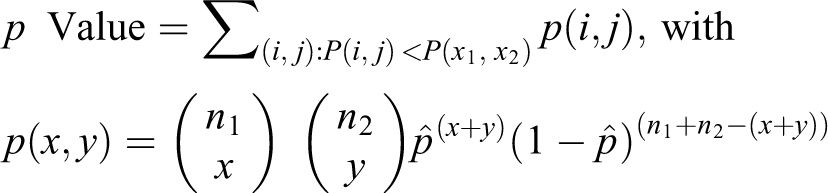
Table 15 (Online Supplement) shows the statistics on the comparison of characteristics for all natural and synthetic pesticides studied.
Results
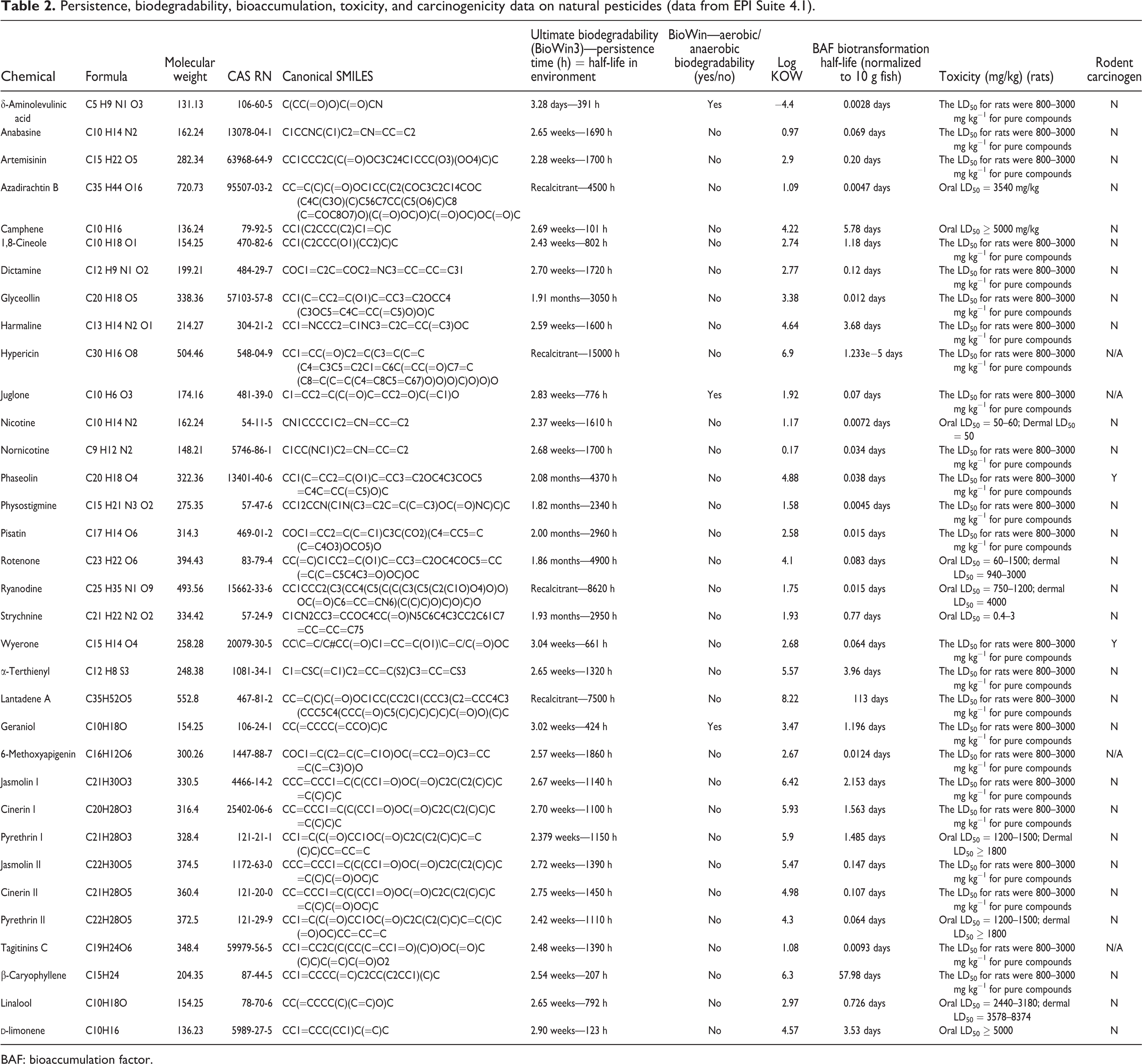
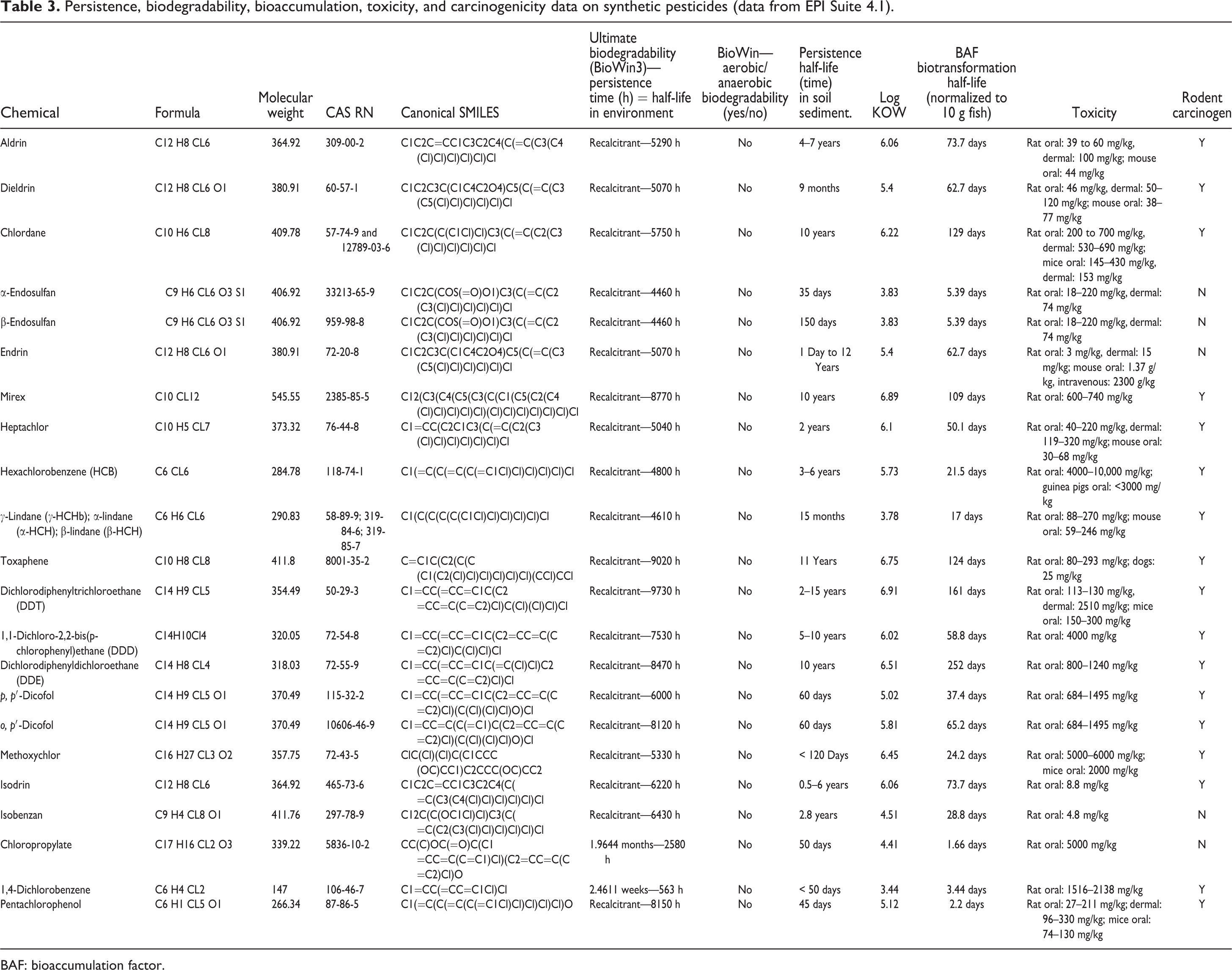
Tables 2 to 4 summarize the persistence, biodegradability, bioaccumulation, toxicity and carcinogenicity data on the 34 relatively high-volume natural pesticides, the 22 high-volume synthetic pesticides, and the Top 10 commercial pesticides sold worldwide. With the exception of rodent carcinogenicity data available on the Internet, the data in Tables 2 to 4 were derived from EPI Suite 4.1.
Persistence, biodegradability, bioaccumulation, toxicity, and carcinogenicity data on natural pesticides (data from EPI Suite 4.1).
BAF: bioaccumulation factor.
Persistence, biodegradability, bioaccumulation, toxicity, and carcinogenicity data on synthetic pesticides (data from EPI Suite 4.1).
BAF: bioaccumulation factor.
Persistence, biodegradability, bioaccumulation, toxicity, and carcinogenicity data on Top 10 pesticides worldwide (data from EPI Suite 4.1).
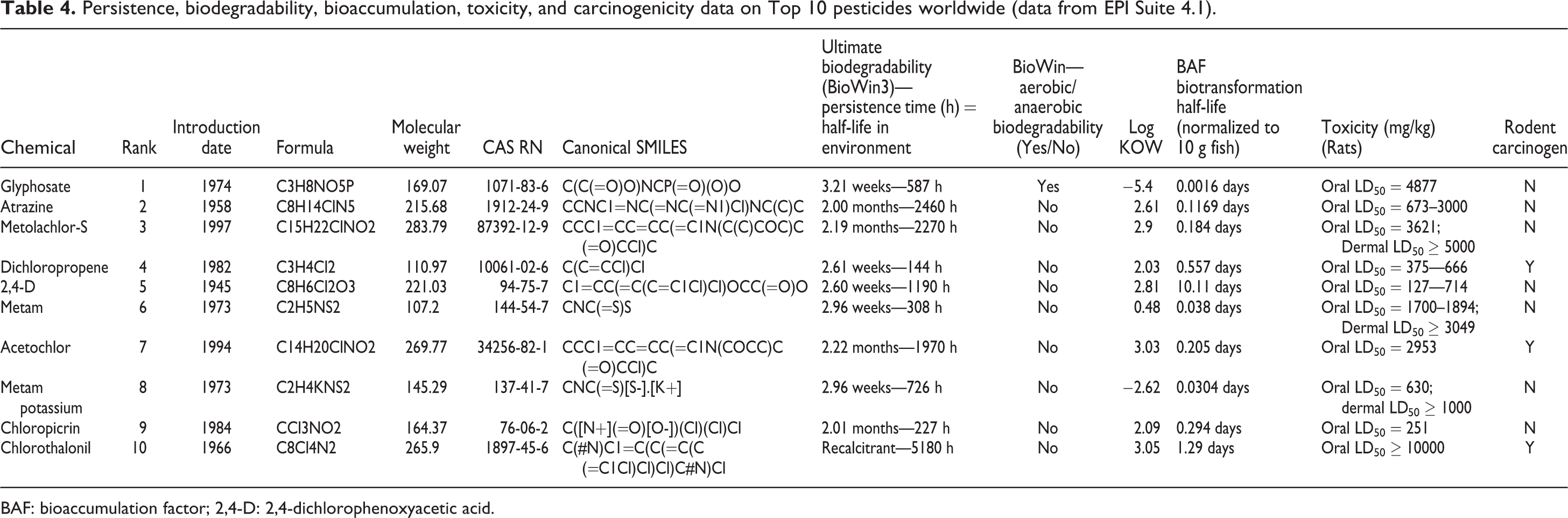
BAF: bioaccumulation factor; 2,4-D: 2,4-dichlorophenoxyacetic acid.
Table 4 summarizes the persistence, biodegradability, bioaccumulation, toxicity, and carcinogenicity data on the Top 10 synthetic pesticides sold worldwide. The maximum, minimum, and average persistence for the Top 10 synthetic pesticides were 5180, 144, and 1506 h. The average persistence time for the 34 natural pesticides was statistically significantly lower than for the 32 synthetic pesticides (
Table 2 summarizes the persistence, biodegradability, bioaccumulation, toxicity, and carcinogenicity data on the 34 natural pesticides. For the 34 natural pesticides, the maximum persistence time (half-life in the environment) was 15,000 h, the minimum persistence time was 101 h, and the average persistence time 2402 h.
Table 3 summarizes the persistence, biodegradability, bioaccumulation, toxicity, and carcinogenicity data on the 22 high-volume synthetic pesticides. The maximum, minimum, and average persistence for these 22 synthetic pesticides were 9730, 144, and 4579 h.
Only 3 of the 34 natural pesticides were biodegradable, that is, δ-aminolevulinic acid, juglone, and geraniol. None of the 22 synthetic pesticides were biodegradable. Only 1 of the Top 10 synthetic pesticides were biodegradable (glyphosate). The difference in tendency to biodegrade between the 34 natural pesticides and 32 synthetic pesticides was not statistically significant (
For the natural pesticides, the maximum log KOW was 8.22, the minimum −4.4, and the average 3.41 (Table 2). The log KOW for the 22 high-volume synthetic pesticides had a maximum, minimum, and average value of 6.91, 3.44, and 5.47, respectively (Table 3). The log KOW for the Top 10 synthetic pesticides had a maximum, minimum, and average value of 3.05, −5.4, and 1.10, respectively (Table 4). On average, the 32 synthetic pesticides were more lipophilic than the 34 natural plant pesticides, although this difference was not statistically significant (
The BAF (biotransformation half-life normalized to 10 g fish) for the natural pesticides ranged from 113 days to 0.00001233 days, with an average of 5.83 days (Table 2). The BAF for the high-volume pesticides ranged from 252 days to 1.66 days, with an average of 62.2 days (Table 3). The BAF for the Top 10 synthetic pesticides ranged from 10.1 days to 0.0016 days, with an average of 1.28 days (Table 4). The BAF of the 32 synthetic pesticides was statistically significantly higher than the comparable value of the 34 natural plant pesticides (
The toxicity in rats measured as the oral LD50 ranged from 5000 mg/kg to 0.4 mg/kg, with an average of 989 mg/kg on the basis of the minimum dose for the 34 natural pesticides (Table 2). The toxicity in rats measured as the oral LD50 ranged from 5000 mg/kg to 3 mg/kg, with an average of 1044 mg/kg on the basis of the minimum dose for the 22 high-volume pesticides (Table 3). The toxicity in rats measured as the oral LD50 ranged from 10,000 mg/kg to 127 mg/kg, with an average of 2521 mg/kg on the basis of the minimum dose for the Top 10 pesticides (Table 4). The toxicity as measured by the oral LD50 for rats was not statistically different for natural and synthetic pesticides (
Only two of the 34 natural pesticides were rodent carcinogens (phaseolin and wyerone) (7%) (Table 2). Seventeen of the 22 high-volume synthetic pesticides were rodent carcinogens (77%) (Table 3). Only 3 of the Top 10 synthetic pesticides were rodent carcinogens (acetochlor, chlorothalonil, and dichloropropene) (30%) (Table 4). The overall percentage for the 32 synthetic pesticides for rodent carcinogenicity was 63% with the comparable value for the 34 natural pesticides at only 7%. This difference was statistically significant (
Table 5 (Online Supplement) shows toxicity data from fathead minnow,
Table 6 (Online Supplement) shows toxicity data from fathead minnow,
Table 7 (Online Supplement) shows toxicity data from fathead minnows,
In Tables 8, 9, and 10 (Online Supplement), toxicity data from fathead minnow,
Table 12 (Online Supplement) provides an overview of the average values of the calculations for the 34 natural pesticides and the 32 synthetic pesticides. Statistical significance of the data is noted at the 95% confidence level. The persistence of the natural pesticides is significantly shorter than for the synthetic pesticides (
Discussion
Persistence
Thirty-four natural plant pesticides and 32 synthetic pesticides were included in this analysis. On average, synthetic pesticides lasted longer in the environment than natural plant pesticides. The median persistence for synthetic pesticides was 5055 h (210.6 days) with an average persistence of 4578.9 h (190.8 days). In contrast, the median persistence for natural plant pesticides was 1600 h (66.7 days), with an average persistence of 2402.5 h (100.1 days). The increased persistence of the synthetic pesticides is statistically significant (
Application of synthetic pesticides and herbicides to agricultural fields is a precise, highly technical, expensive, and time-consuming process. 32 The number of applications required is specific to the soil type and fertility, rainfall, erosion and weathering, field slope and runoff pathways, potency toward the intended pests, and crop type. 33 –39 US EPA and the European Union promulgate regulations toward minimization of pesticide use via programs of Integrated Pest Management. 40,41
Agro-scientists and farmers, and the biological evolution of plants, share several of the same goals but operate on different timescales. The goal of maximizing crop yield per unit cost has incentivized the design of fast-acting synthetic pesticides. 42 In contrast, although natural plant pesticides applied to agricultural crops can be effective, they are generally slower acting. 43,44 This kinetic difference is not unexpected as a particular natural plant pesticide is usually found in a vegetable along with a large number of other natural plant pesticides, for example, broccoli is known to contain approximately 40 different plant pesticides. 6 Therefore, most natural plant pesticides did not evolve as standalone deterrents to insect or animal predation, with several notable exceptions, for example, highly toxic nicotine in the tobacco plant. 45,46
Toxicity
Using the T.E.S.T. program, the oral rat LD50 (mg/kg bodyweight) of the 34 natural plant pesticides and 32 synthetic pesticides were either directly obtained from experimental data or predicted. The median LD50 for the synthetic pesticides is 176.9 mg/kg. This level of toxicity falls within Category II (moderately toxic) (Table 13, Online Supplement). 47,48 The mean LD50 for the synthetic pesticides is 974.3 mg/kg, which falls within Category III (slightly toxic). 47,48 The median LD50 for the natural pesticides is 569.6 mg/kg, which falls within Category III (slightly toxic). The mean LD50 for the natural pesticides is 1431.9 mg/kg (Category III/slightly toxic). While the median and mean LD50s of the synthetic pesticides are lower (more toxic) than comparable values for the natural pesticides, neither type of pesticide is notably toxic as measured by oral rat LD50 values. 47,48
Using the T.E.S.T. program, the Ames
The
Chronic bioassays conducted in rats or mice for the purpose of assessing the tumorigenic or carcinogenic potential of a chemical are very expensive and time-consuming. 54,55 The majority of chemicals evaluated in these resource-intensive bioassays are commercial products being tested either as part of product stewardship or regulatory compliance. 56,57 As most natural plant pesticides are not commercial products, relatively few of the more than 10,000 identified have been tested for carcinogenic potential. 58 –62 Ames et al. 6 noted that up through 1990, 1052 chemicals had been tested in at least one species in chronic cancer tests. Of the 1052 chemicals, 52 were natural plant pesticides. 58 –61 Of the 52 natural plant pesticides tested, 27 were carcinogenic, that is, 27/52 (52%). However, the natural plant pesticides considered by Ames et al. 6 are found in commonly eaten vegetables and do not include the 34 commercial natural plant pesticides applied to crops and considered in this analysis. It is possible that the selection process that resulted in wide use of the 34 natural plant pesticides used in commerce took carcinogenic potential into consideration thereby resulting in only 2/28 being reported as rodent carcinogens.
Over the last 33 years (1985–2018), the US EPA has evaluated over 500 pesticides for carcinogenic potential of pesticide chemicals via the US EPA’s Pesticide Program (US EPA 2018). 18 The latest report was issued in late 2018. 18 The list includes the chemical name, CAS RN, PC code (a unique chemical identifier used by the EPA Office of Pesticide Programs), EPA human cancer classification, and report date. In the 2018 report, approximately 520 pesticides were listed. Of these, 514 possessed sufficient information for analysis. Table 14 (Online Supplement) shows the relationship between the human cancer classification and data collected from rodent carcinogenicity testing. Of the 514 pesticides for which data were available, 36% were carcinogenic to rodents. 18
The 32 synthetic pesticides in this analysis represent the Top 10 (poundage of active ingredient) pesticides sold worldwide 17 and 22 other popular pesticides used international. 15,16 Eight pesticides are common to the set of 32 synthetics in this study and the 514 synthetics tested by the US EPA, that is, 2,4-D, acetochlor, atrazine, chloropicrin, chlorothalonil, glyphosate, metolachlor, and pentachlorophenol. Seven of these pesticides are on the Top 10 (poundage of active ingredient) list of pesticides sold worldwide. The overall rodent carcinogen percentage of 62% for the 32 synthetic pesticides (Top 10 plus the 22 high-volume) is notably higher than the 30% rate reported for the just the Top 10 synthetic pesticides sold worldwide. It is possible that the relatively low rate of rodent carcinogenicity has been a factor in elevating this subset of pesticides into the Top 10 positions by poundage of active ingredient used. 63,64
In addition to an increase in predicted or measured Ames mutagenicity, the T.E.S.T. program predicts a statistically significant increase in the probability that the 34 natural plant pesticides will induce developmental toxicity than the 32 synthetic pesticides. However, QSAR is generally considered as less reliable in predicting effects on developmental toxicity than in predicting Ames mutagenicity. 65 –67
Conclusions
In 1990, Ames et al. evaluated the toxicological significance of exposures to synthetic pesticides as compared with naturally occurring pesticides. They calculated that 99.99% (by weight) of the pesticides ingested in the American diet are chemicals that plants produce to defend themselves. Of the natural pesticides tested in high-dose animal cancer tests, about half were rodent carcinogens found in many common foods. They concluded that natural and synthetic chemicals are equally likely to be positive in animal cancer tests. Based on the relative mass of ingested material, synthetic pesticides represented an insignificant health risk as compared to the risks posed by natural pesticides.
In conclusion, the current study suggests that persistence in the environment is higher for synthetic as compared with natural pesticides. Similarly, rodent carcinogenicity is also higher in synthetic pesticides. Acute toxicity as determined by LD50 values in rats did not differ between synthetic and natural pesticides. Natural pesticides display significantly higher developmental toxicity and mutagenicity as compared with synthetic pesticides. Based on these findings and the much higher level of ingestion of natural pesticides (via food ingestion), health risks to the general human population from natural plant pesticide exposure would be expected to be greater than risks expected from exposure to synthetic pesticides on average. However, individual fruits and vegetables should always be washed prior to ingestion to remove any potentially harmful organic material, or synthetic pesticide residues that might be extant at higher than average values.
Supplemental material
Supplemental_Material_for_TOR_20 - A comparison of the persistence, toxicity, and exposure to high-volume natural plant-derived and synthetic pesticides
Supplemental_Material_for_TOR_20 for A comparison of the persistence, toxicity, and exposure to high-volume natural plant-derived and synthetic pesticides by Carr J Smith and Thomas A Perfetti in Toxicology Research and Application
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
