Abstract
The essential oil of peppermint and one of its natural constituents, (R)-(+)-pulegone, are approved flavorings added to food worldwide. (R)-(+)-Pulegone and peppermint oil were tested separately in two independent bacterial reverse mutation assays according to Organisation for Economic Co-operation and Development Guideline 471. Both flavorings did not produce any evidence of mutagenicity up to cytotoxic concentrations in either the presence or the absence of exogenous metabolic activation.
Introduction
Herbs of the
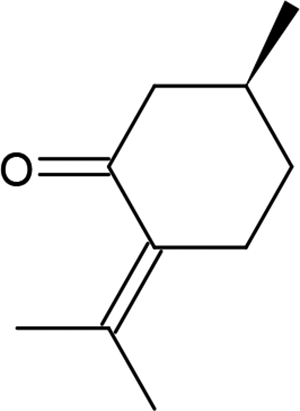
Chemical structure of (R)-(+)-pulegone.
The concern for potential genotoxicity of pulegone was raised following the results of genotoxicity studies conducted by the National Toxicology Program (NTP) in 2011. These studies included three bacterial reverse mutation studies in
Materials and methods
Study compliance
The studies 19,20 were conducted at BioReliance Corporation (Rockville, Maryland, USA) according to Organisation for Economic Co-operation and Development (OECD) Guideline 471 21 and BioReliance standard operating procedures. The studies were also compliant with US FDA Good Laboratory Practice (GLP) Regulations published in 21 CFR Part 58, which is also compatible with OECD Principles of Good Laboratory Practice. 22,23
Test substances
(R)-(+)-Pulegone (IUPAC (5R)-5-methyl-2-propan-2-ylidenecyclohexan-1-one; MW 152.23; CAS No. 89-82-7) and peppermint oil (
Based on solubility testing, ethanol (Sigma-Aldrich, St. Louis, MO) was chosen as the vehicle for dose preparations. The positive controls were all obtained from Sigma-Aldrich with purities of 97.5% and greater. The positive controls for treatments in the absence of metabolic activation were 2-nitrofluorene, sodium azide (SA), 9-aminoacridine, and methyl methanesulfonate. For treatments in the presence of metabolic activation, the positive control was 2-aminoanthracene. All positive controls were diluted in dimethylsulfoxide (Sigma-Aldrich), except SA, which was diluted in sterile water. Positive controls were stored at −10 to −30°C.
Dose preparations
Test substance dilutions and dose preparations were prepared immediately before use and delivered to the test system at room temperature under filtered light. The test substances were determined to be soluble in ethanol up to a concentration of approximately 500 mg/mL. Each test substance was weighed out and diluted in ethanol to a concentration of 100 mg/mL. Because the test solutions were used immediately after preparation, analyses to determine the concentration, homogeneity, and stability of the test substances in the vehicle (ethanol) were not performed in these studies. Although these analyses are not a requirement in the OECD Guideline, this was recorded as a deviation from the laboratory’s GLP.
Test system
The tester strains used were the
Exogenous metabolic activation (S9)
Aroclor 1254-induced rat liver S9 was used as the metabolic activation system. The S9 was prepared from male Sprague-Dawley rats that were injected intraperitoneally with Aroclor™ 1254 (200 mg/mL in corn oil) at a dose of 500 mg/kg, 5 days before sacrifice and was stored at −60°C or colder until use. The S9 mixture prepared for addition was composed of 4 mM of β-nicotinamide-adenine dinucleotide phosphate, 5 mM glucose-6-phosphate, 33 mM potassium chloride, 8 mM magnesium chloride, 100 mM phosphate buffer (pH 7.4), and 10% (v/v) S9 homogenate.
Experimental design
Preparation of tester strains
Overnight cultures were prepared by inoculating from the appropriate frozen permanent stock into a vessel, containing 30–50 mL of culture medium. To assure that cultures were harvested in late log phase, the length of incubation was controlled and monitored. Following inoculation, each flask was placed in a shaker/incubator programmed to begin shaking at 125–175 r/min and incubating at 37 ± 2°C for approximately 12 h before the anticipated time of harvest. Each culture was monitored spectrophotometrically for turbidity and was harvested at a percent transmittance yielding a titer of greater than or equal to 0.3 × 10 9 cells per milliliter. The actual titers were determined by viable count assays on nutrient agar plates.
Scoring
The condition of the bacterial background lawn was evaluated for evidence of test substance cytotoxicity by using a dissecting microscope. Precipitate was evaluated after the incubation period by visual examination without magnification. Cytotoxicity, defined as the reduction in the background lawn or the reduction (>50%) of mean revertants, and degree of precipitation were scored relative to the vehicle control plate. As appropriate, colonies were enumerated either by hand or by machine (Sorcerer Colony Counter, Perceptive Instruments, Bury St Edmunds, UK).
Preliminary toxicity experiments
In the preliminary toxicity/range-finding experiments,
Main mutagenicity experiments
Based on the preliminary toxicity experiments, the concentrations selected for the mutagenicity experiments for both test substances were 15.0, 50.0, 150, 500, 1500, and 5000 µg/plate, using approximate half-log spacing. Each of the concentrations were tested in triplicate using the plate incorporation method.
Results
Pulegone mutagenicity experiments
Preliminary toxicity experiment
In the preliminary toxicity/range-finding experiment for pulegone, no precipitate was observed for any of the concentrations tested. Cytotoxicity was observed at 5000 µg/plate in all tester strains in the absence and/or presence of S9, and at 3333 µg/plate in TA100 in the absence and presence of S9 and in TA1535 in the absence of S9. Only moderate toxicity was observed at 5000 µg/plate in most tester strains in the range-finding experiment. Marked cytotoxicity at 5000 µg/plate was observed only in TA100 and TA1535 with S9 and slight or moderate cytotoxicity at the next highest concentration of 3333 µg/plate. The results of the preliminary toxicity experiment are presented in Table 1.
Mean revertant counts with (+S9) and without (−S9) metabolic activation in the preliminary toxicity experiment on pulegone.a
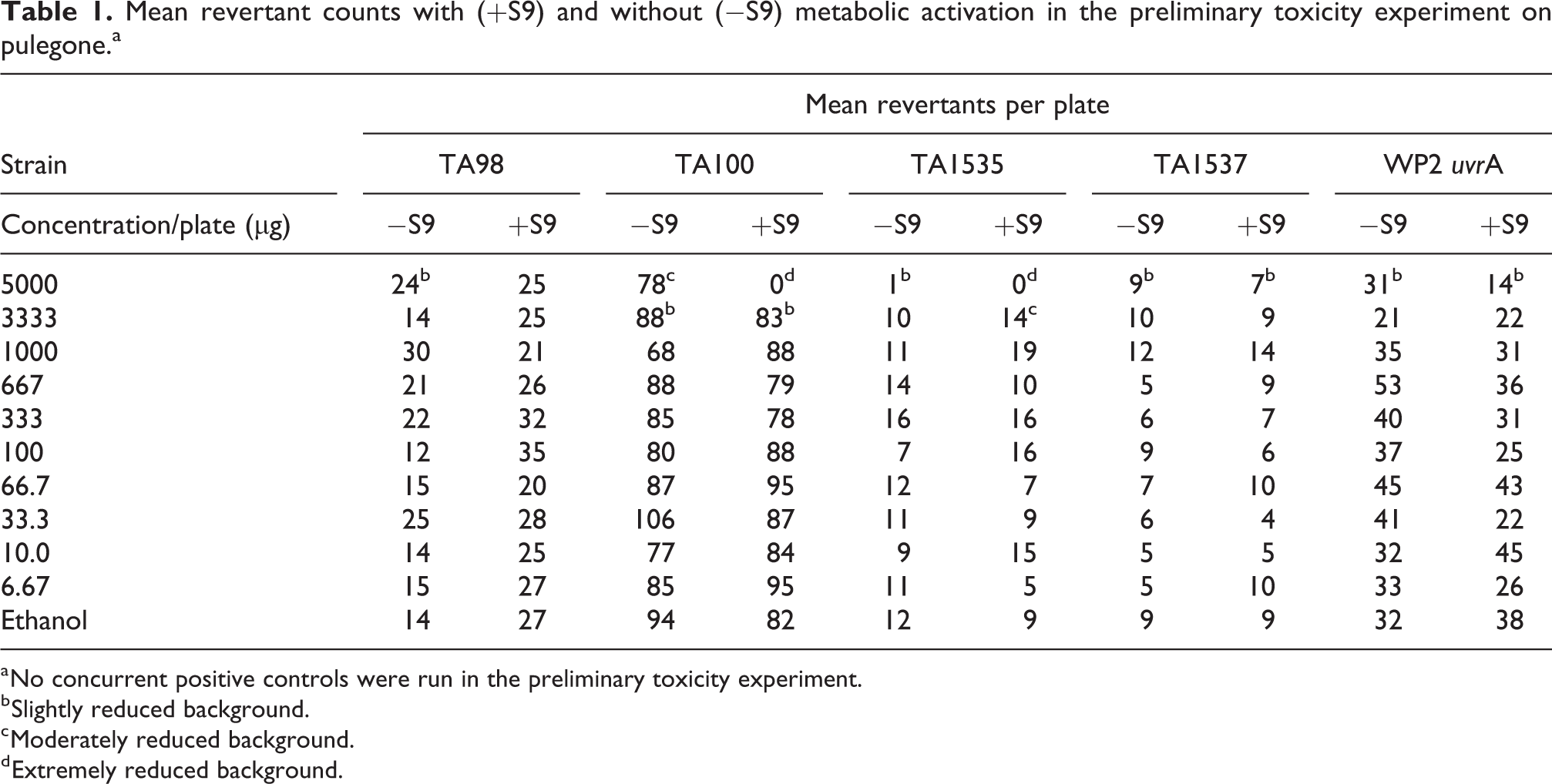
a No concurrent positive controls were run in the preliminary toxicity experiment.
b Slightly reduced background.
c Moderately reduced background.
d Extremely reduced background.
Main mutagenicity experiment
The results of the main mutagenicity experiment on pulegone are presented in Table 2. No precipitate was observed in any of the concentrations tested. The highest test concentration for the main study was maintained at 5000 μg/plate, since only slight to moderate toxicity was observed at the next highest test concentration in most strains in the range-finding experiment. In the main experiment, marked cytotoxicity was observed at 5000 µg/plate only in TA100 without and with S9 and in TA1535 with S9 and only moderate cytotoxicity in all other tester strains in the absence and presence of S9. No cytotoxicity was observed at the next highest test concentration (1500 µg/plate) except for slight cytotoxicity in TA100 in the absence of S9. No positive mutagenic responses were observed with any of the tester strains in either the presence or absence of S9 activation.
Mean revertant counts with (+S9) and without (−S9) metabolic activation in the main mutagenicity experiment on pulegone.
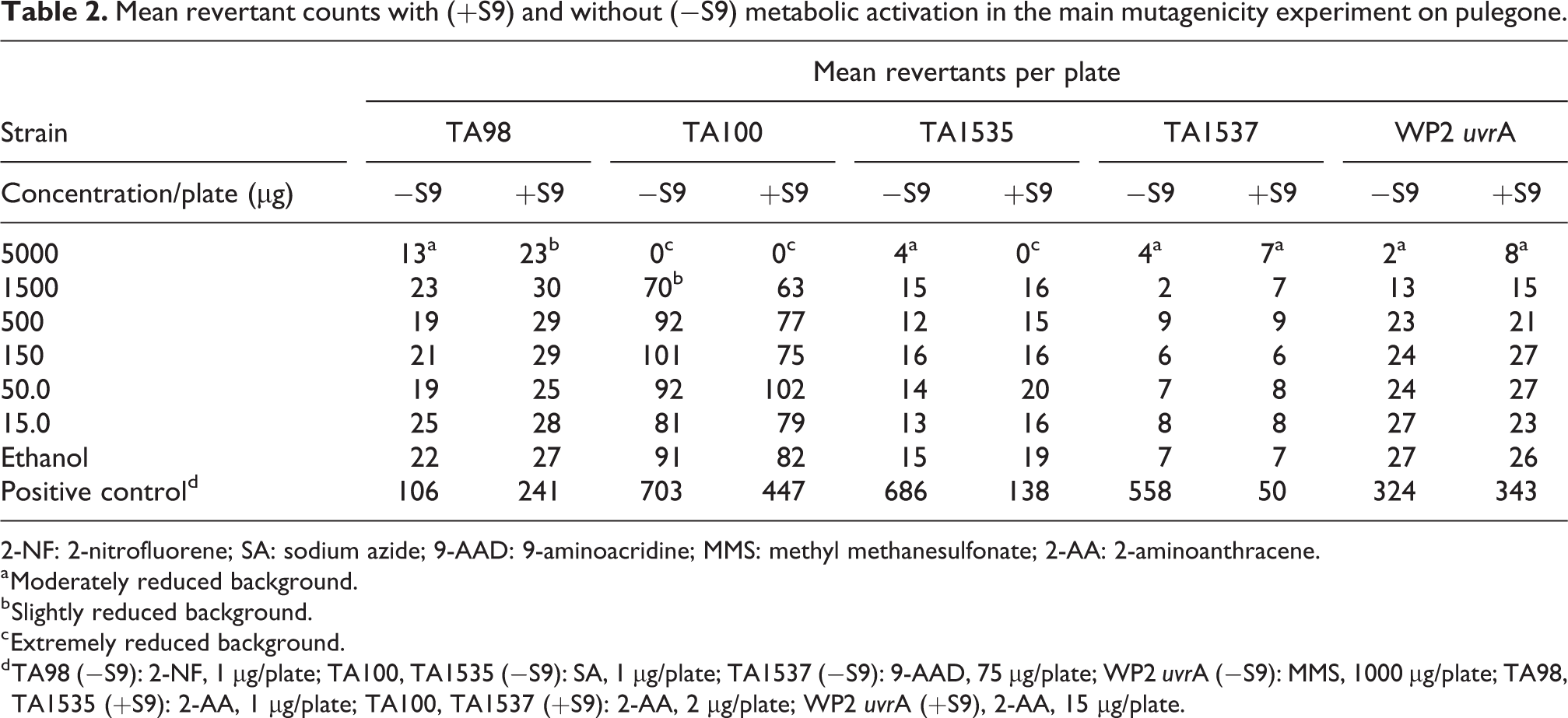
2-NF: 2-nitrofluorene; SA: sodium azide; 9-AAD: 9-aminoacridine; MMS: methyl methanesulfonate; 2-AA: 2-aminoanthracene.
a Moderately reduced background.
b Slightly reduced background.
c Extremely reduced background.
d TA98 (−S9): 2-NF, 1 µg/plate; TA100, TA1535 (−S9): SA, 1 µg/plate; TA1537 (−S9): 9-AAD, 75 µg/plate; WP2
Peppermint oil mutagenicity experiments
Preliminary toxicity experiment
In the preliminary toxicity/range-finding experiment for peppermint oil, no precipitate was observed for any of the concentrations tested. Cytotoxicity was observed at 3333 and 5000 µg/plate in all tester strains in the absence and presence of S9. Marked cytotoxicity was observed at 5000 and 3333 μg/plate for TA100 and TA1535 without S9 and in TA100, TA1535, and TA1537 with S9. Moderate to slight cytotoxicity was observed at 3333 µg/plate in TA1537 without S9 and in WP2
Mean revertant counts with (+S9) and without (−S9) metabolic activation in the preliminary toxicity experiment on peppermint oil.a
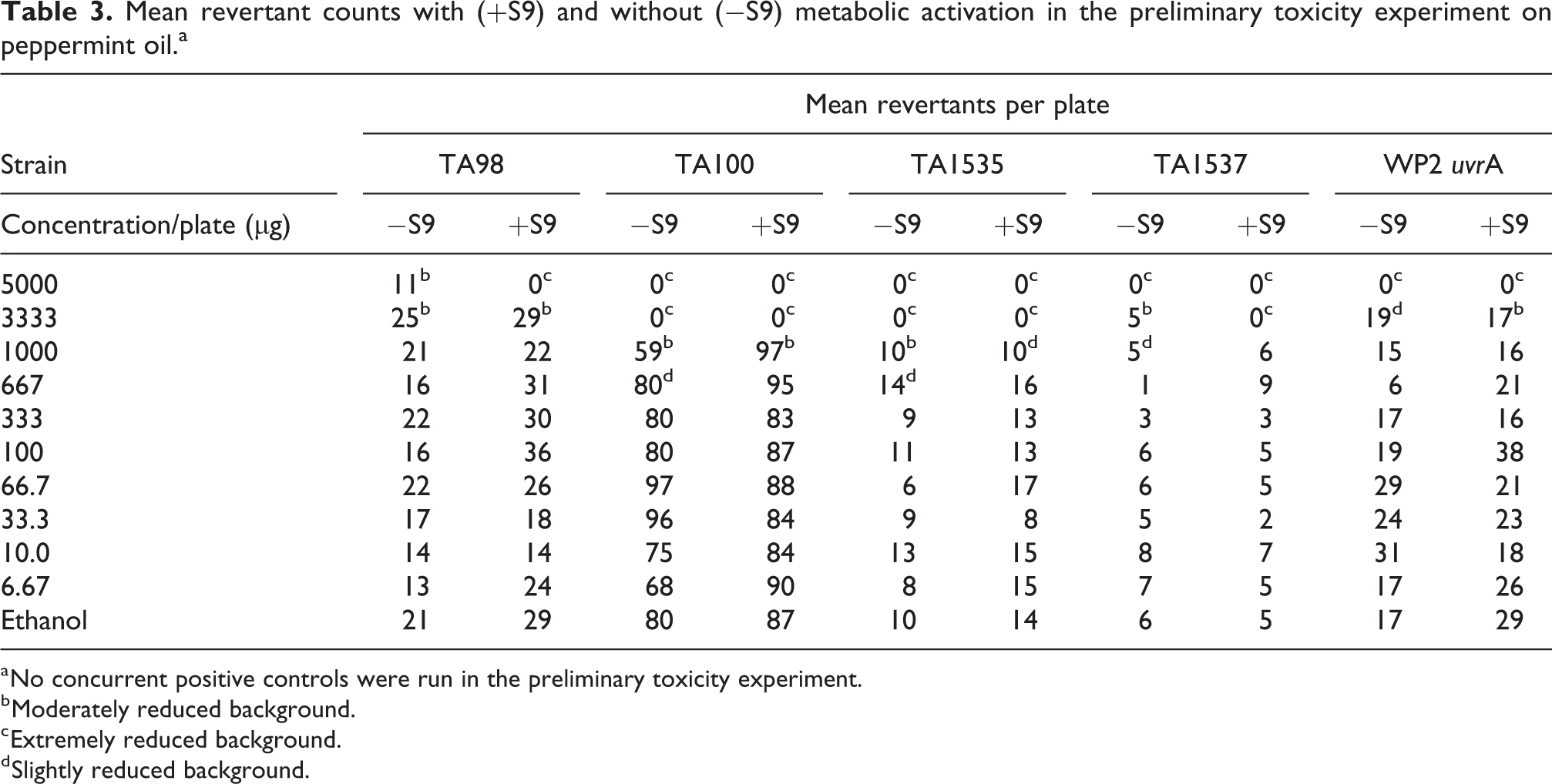
a No concurrent positive controls were run in the preliminary toxicity experiment.
b Moderately reduced background.
c Extremely reduced background.
d Slightly reduced background.
Main mutagenicity experiment
The results of the main mutagenicity experiment on peppermint oil are presented in Table 4. No precipitate was observed in any of the concentrations tested. Marked cytotoxicity was observed at 5000 µg/plate in all strains and tested conditions. Moderate cytotoxicity was observed at 1500 µg/plate in TA100 and TA1535 in the absence and presence of S9 and in TA1537 in the presence of S9. Slight cytotoxicity was observed at 1500 µg/plate in TA98 and TA1537 in the absence of S9 and in WP2
Mean revertant counts with (+S9) and without (−S9) metabolic activation in the main mutagenicity experiment on peppermint oil.
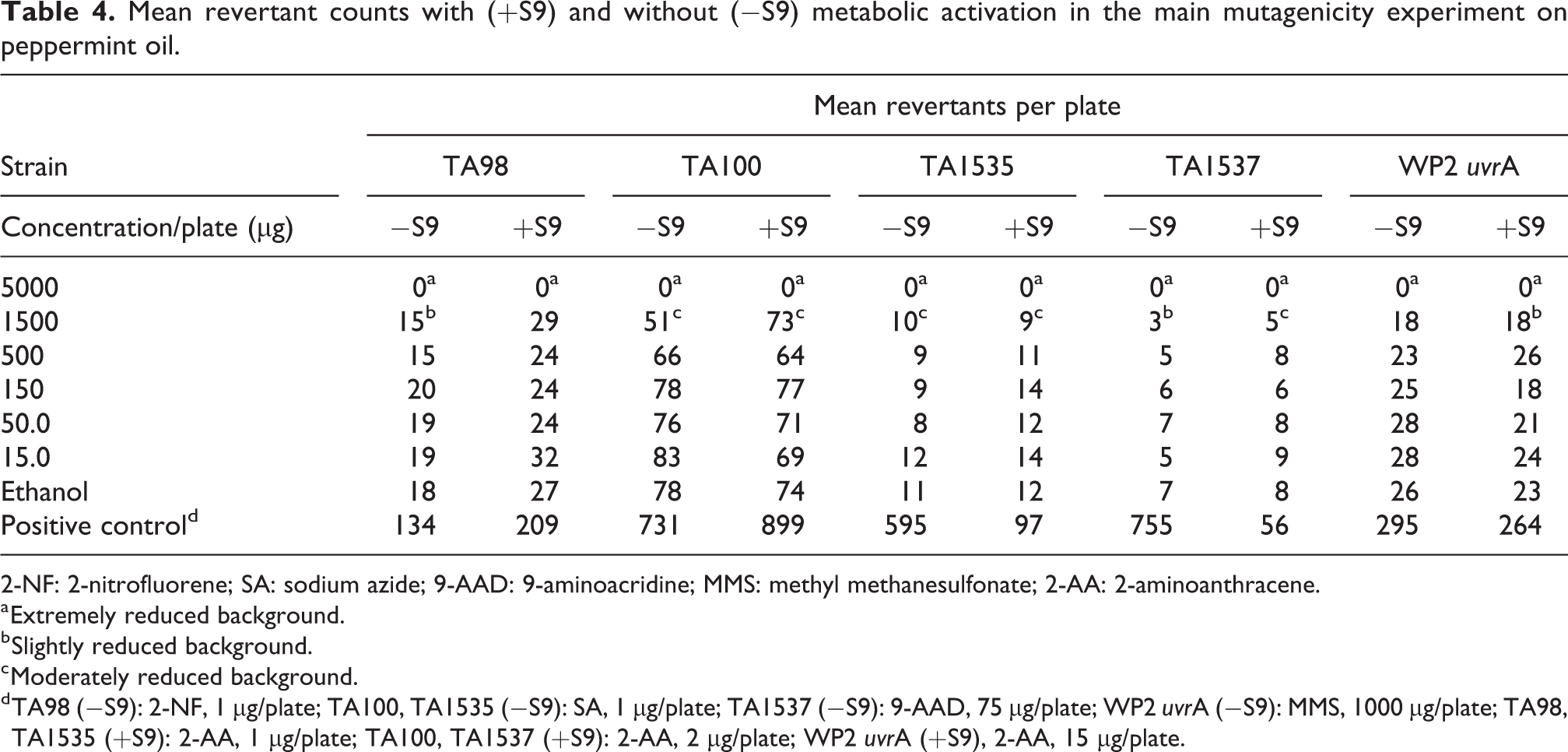
2-NF: 2-nitrofluorene; SA: sodium azide; 9-AAD: 9-aminoacridine; MMS: methyl methanesulfonate; 2-AA: 2-aminoanthracene.
a Extremely reduced background.
b Slightly reduced background.
c Moderately reduced background.
d TA98 (−S9): 2-NF, 1 µg/plate; TA100, TA1535 (−S9): SA, 1 µg/plate; TA1537 (−S9): 9-AAD, 75 µg/plate; WP2
Discussion and conclusion
Herbs of the
The NTP genotoxicity testing for pulegone included three bacterial reverse mutation studies and an in vivo micronucleus induction study in B6C3F1 mice. In the first NTP bacterial reverse mutation study conducted at an independent contract laboratory,
In the in vivo micronucleus assay in male and female B6C3F1 mice conducted by the NTP, pulegone did not increase the frequency of micronucleated peripheral blood erythrocytes when administered at dose levels of 9.375, 18.75, 37.5, 75, or 150 mg/kg bw/day by gavage for 3 months. 11 In another recent in vivo study, an essential oil containing 65% pulegone was tested in a bone marrow micronucleus assay in rat and a modified comet assay in blood lymphocytes at dietary concentrations of 70, 260, and 460 mg/kg bw/day (equivalent to 45.5, 169, and 299 mg pulegone/kg bw/day, respectively) and was negative for induction of micronuclei in bone marrow erythrocytes and for DNA damage in blood cells. 13
Previous genotoxicity studies also support the lack of genotoxicity for the primary metabolite of pulegone, menthofuran,
14
–16
and for peppermint oil,
12,17,18
including a lack of mutagenicity in various
When these experiments are considered collectively, the only positive finding for pulegone was found in the third reverse mutation study conducted by the NTP, a finding inconsistent with the other genotoxicity assays conducted by NTP, as well as all other genotoxicity and mutagenicity assays.
11
–13
The validity and significance of the positive result that was reported with pulegone in
Footnotes
Author contributions
Mses. Harman and Lu and Drs Bastaki and Taylor researched, compiled, and interpreted available study data and drafted the manuscript. Drs Aubanel, Bialk, Choi, Demyttenaere, Diop, Etter, Han, Krammer, Schnabel, and Wilson reviewed and provided comments on the content and interpretation of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the International Organization of the Flavor Industry.
