Abstract
The bacterial reverse mutation assay, that is, the Ames test, measures mutations that reverse the inactivation of a gene involved in the synthesis of either histidine in
Introduction
The bacterial reverse mutation assay is commonly known as the Ames test.
1
The Ames test is the most commonly conducted genotoxicity assay. This test detects mutations in either
Analysis of Ames test data
Figure 1 shows a plot of a representative TA98 assay with S9 activation. In Figure 1, the number of TA98 revertants/plate is plotted against the concentration of 6-cyano-1-methyl-2-aminobenzimidazole (6-CN-1-Me-2 AmB) expressed in micrograms/plate (Table 1). The initial slope is calculated as the first derivative of the plotted curve as the concentration of 6-CN-1-Me-2 AmB approaches zero. 2
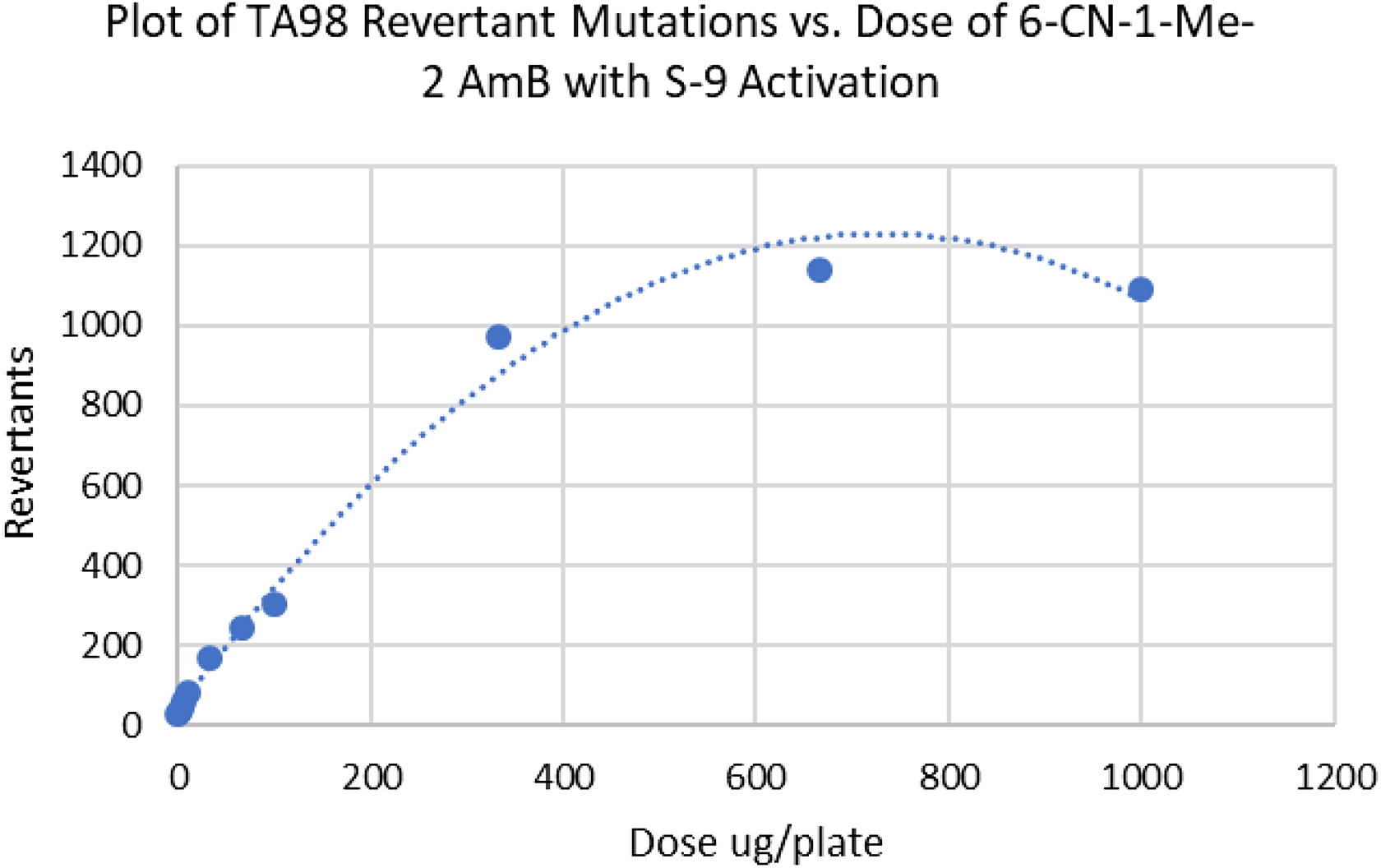
Plot of TA98 revertant mutations versus dose of 6-CN-1-Me-2 AmB demonstrating a bending over of the curve indicative of cytotoxicity. 6-CN-1-Me-2 AmB: 6-cyano-1-methyl-2-aminobenzimidazole.
TA98 Ames data on the mutagen 6-CN-1-Me-2 AmB.

6-CN-1-Me-2 AmB: 6-cyano-1-methyl-2-aminobenzimidazole.
Initial slopes of Ames test data are determined using a special case of the parameterization of Stead et al. 3 according to the method of Myers et al. 2 This procedure has several advantages including that data need not be discarded if they become nonlinear due to toxicity and mathematical transformations of the data are unnecessary. 4 The quality of the fit of the statistical model to the data (goodness-of-fit) was evaluated using the “extra sum of squares” principle of Draper and Smith. 5 The last three data points in the dose–response curve in Figure 1 display the bending over of the curve indicative of cytotoxicity, although 6-CN-1-Me-2 AmB could be considered as only moderately cytotoxic in comparison with the examples to follow. 6
In a quantitative structure–activity relationship (QSAR) study conducted on a series of quinolines, Morton from our group at that time developed a statistical model to simultaneously consider the cytotoxicity that can occur at higher mutagen doses.
7
Morton’s statistical model analyzed the results from a TA100
The model that was used, that is,
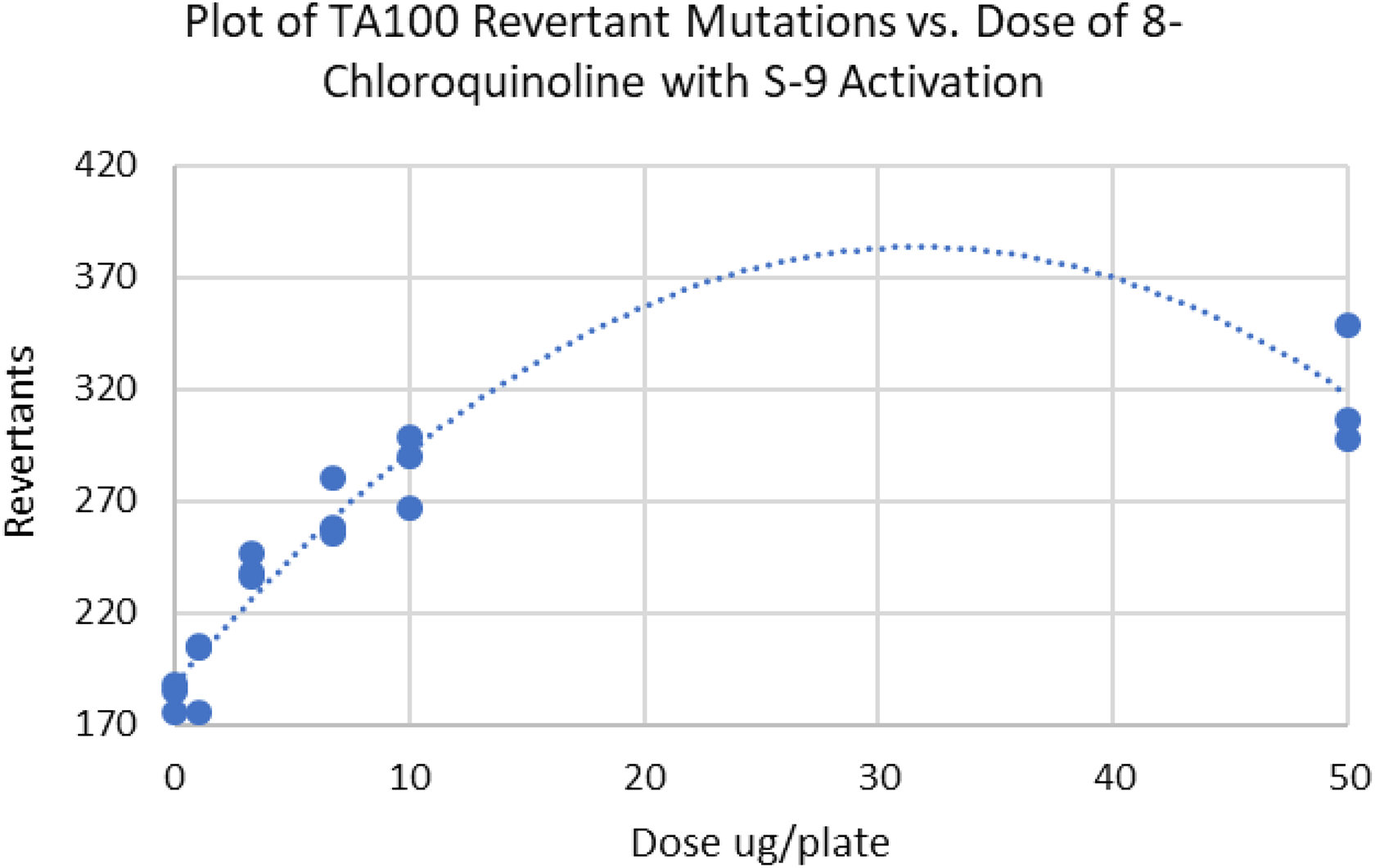
Plot of TA100 revertant mutations versus dose of 8-chloroquinoline demonstrating good fit. Each compound was assayed three times.
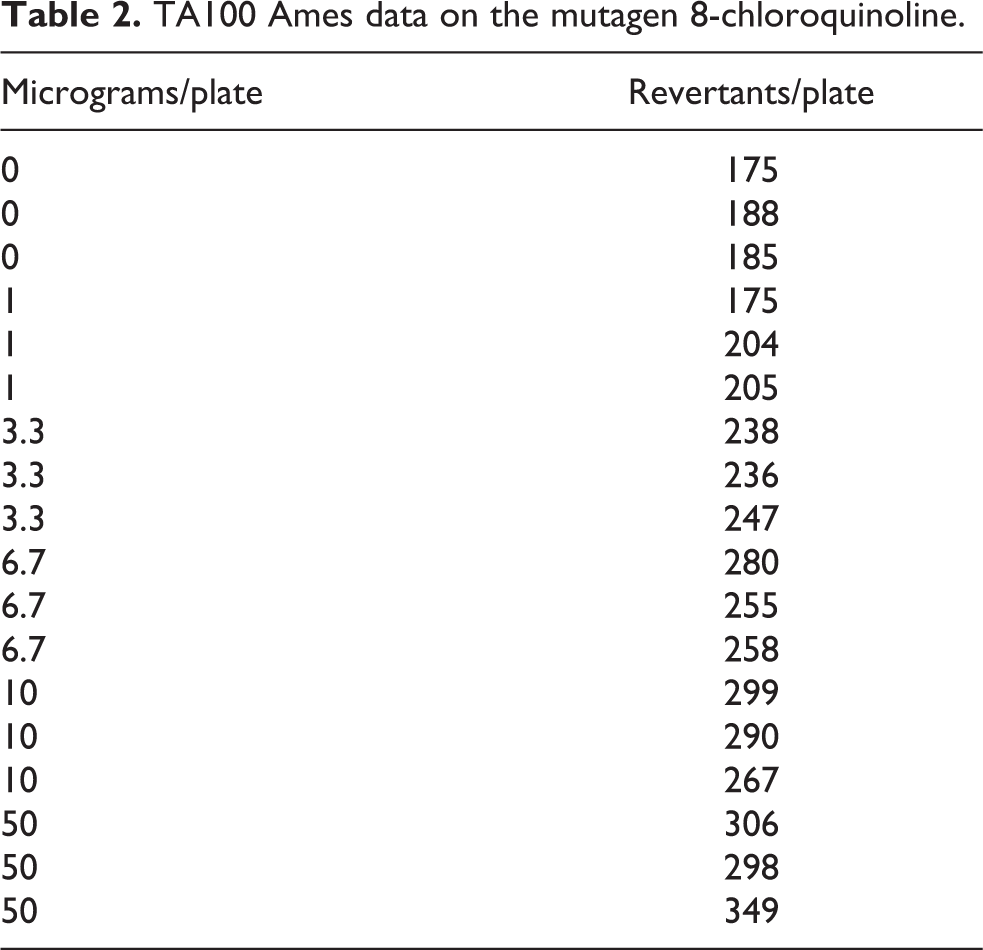
TA100 Ames data on the mutagen 8-chloroquinoline.
If the response shows curvature, the model will try to fit that curvature with the exponential term and the curvature would be interpreted as cytotoxicity. The statistical model used in this study to describe the simultaneous mutation and bacterial cell killing processes may not be appropriate in certain cases where the biological responses follow a different pattern from those assumed. When the enzymes involved in the S9 metabolic activation of parent compounds to mutagenic metabolites follow saturation kinetics and their concentration becomes a rate-limiting factor, the measured mutagenic rate would slow. In this particular case, the model would incorrectly interpret the slowing mutagenic rate as cytotoxicity. Experienced Ames assay laboratories are aware of this possibility and try to use adequate concentrations of S9, although some underestimates of mutagenicity due to this mechanism probably do occur. 7
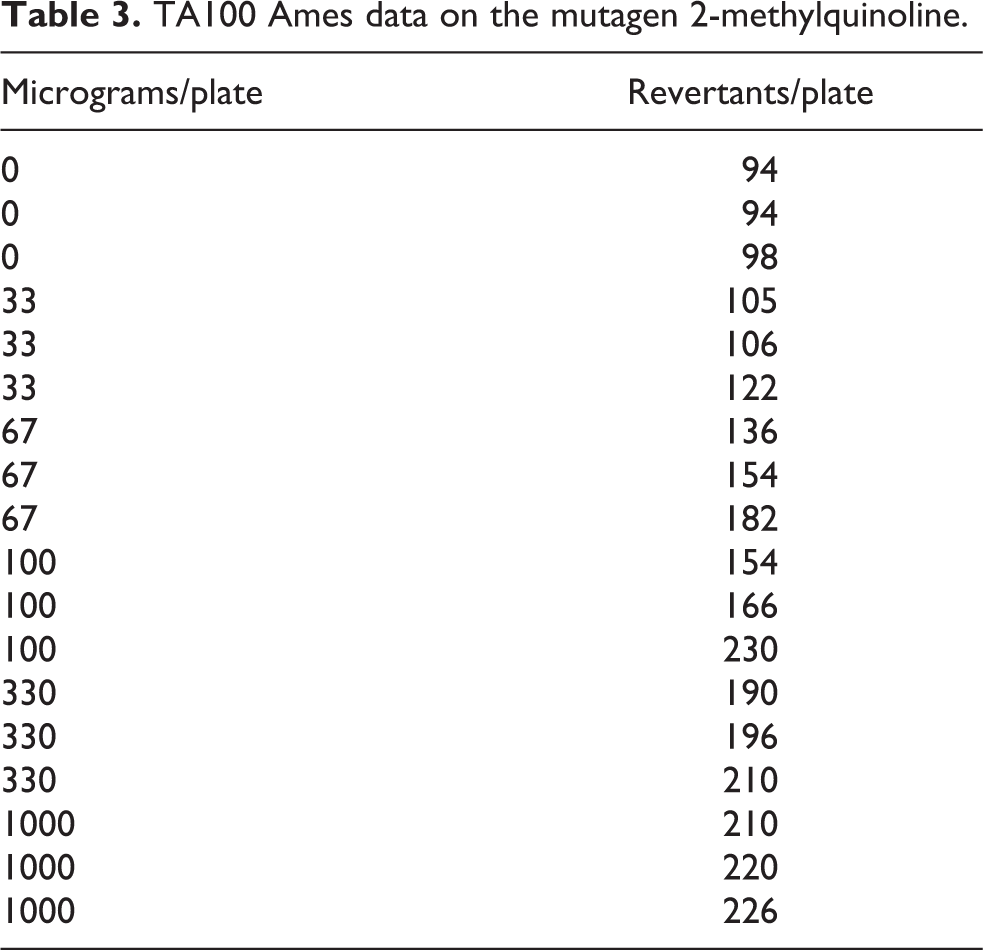
It should be noted that an incorrect interpretation by the mutagenicity calculation model can occur when the concentration of mutagen must reach a “threshold” value before the compound becomes cytotoxic. In this case, the curvature detected after the threshold will cause the initial slope estimate to incorrectly show the effect of some toxicity below the threshold value (Figure 3, Table 3). An additional, though probably not common case would be anti-mutagenic compounds that would be protective at low doses. In this situation, a plot of the “revertant mutations” versus “dose of quinoline” curve might assume a “J” shape. 7
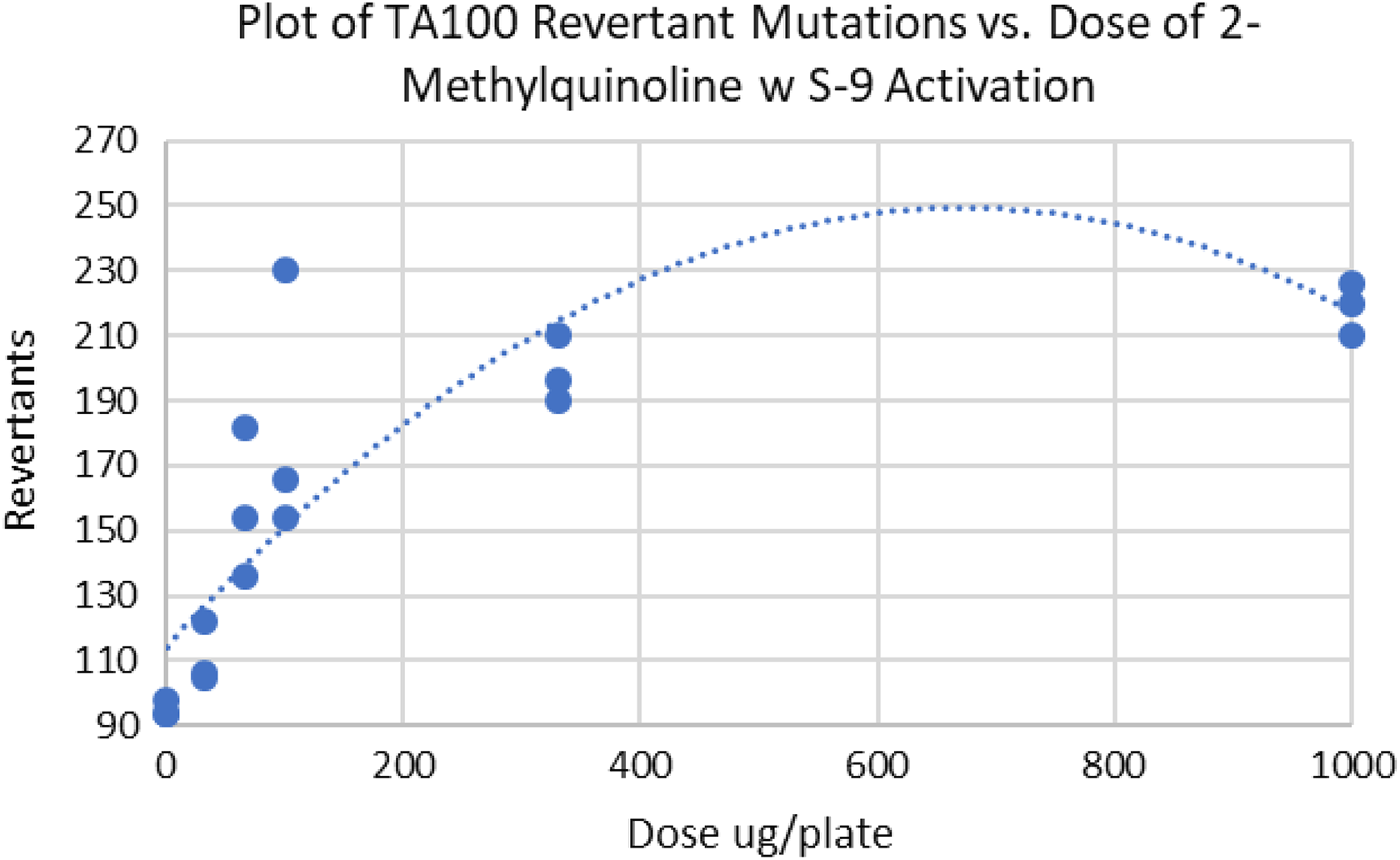
Plot of TA100 revertant mutations versus dose of 2-methylquinoline demonstrating inadequate fit because at high doses the compound is not as cytotoxic as early trends suggest.
TA100 Ames data on the mutagen 2-methylquinoline.
Discussion
For regulatory purposes, Ames test results are usually considered as either positive or negative without consideration as to the calculated potency of the test agent. The “two-fold rule” is the most widely used method for nonstatistical analysis of Ames test results. 9,10 For the simple case where a positive response is considered as twice the background mutation rate, 9,10 cytotoxicity is normally indicated by the partial or complete absence of a bacterial background lawn. 1 Similarly, cytotoxicity is also indicated by a substantial dose-related reduction in revertant colony counts as compared with a particular Ames’ laboratory’s historical controls under comparable experimental conditions. 1 However, for situations in which the potency of an Ames mutagen is relevant, the considerations described previously should be noted. 6,7
QSAR is a method first developed by Corwin Hansch and Toshio Fujita in 1964 to predict the biological activity of a chemical from its structure.
11
Common biological activities considered are pharmacological efficacy or toxicity.
12
The Hansch Method for QSAR analysis entails the use of physical organic chemistry principles to select a set of structurally similar molecules termed congeners.
13
The set of congeners is usually selected by gathering a group of chemicals, for example, 10–20, each sharing a common molecular backbone. Side groups are attached to the common molecular backbone to provide a wide range of lipophilicities (hydrophobicities). Concomitantly, side groups that either withdraw or release electrons into the molecular backbone are attached to provide a wide range of electronic parameters.
13
The constructed or selected series of structural congeners is then subjected to a particular test of biological activity, for example, the Ames
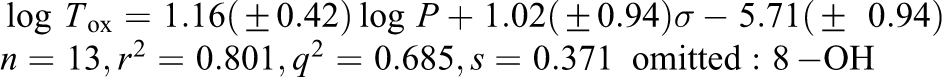
Log
The Morton statistical model was published in 1997.
7
Published studies reporting the development of QSARs with cytotoxicity as the primary end point are common.
11
–14
In contrast, the simultaneous consideration of mutagenicity and cytotoxicity in a congeneric series of molecules subjected to the Ames
It is noteworthy that the QSAR equations for the mutagenicity and cytotoxicity of quinolines demonstrate very different relationships. 7,20 –23 For the mutagenicity of these quinolines, both hydrophobic and steric interactions appear to be important. In contrast, the cytotoxicity is mainly affected by increasing hydrophobicity and by the addition of electron-withdrawing substituents to the quinoline ring. A large number of different chemicals are routinely subjected to assay in the Ames test. When these Ames assays display significant cytotoxicity in addition to the primary endpoint of mutagenicity, further mechanistic data could be garnered from also quantitating the cytotoxicity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
