Abstract
Cigarette smoke (CS) is a risk factor contributing to lung remodeling in chronic obstructive pulmonary disease (COPD). COPD is a heterogeneous disease because many factors contribute in varying degrees to the resulting airflow limitations in different regions of the respiratory tract. This heterogeneity makes it difficult to understand mechanisms behind COPD development. In the current study, we investigate the regional heterogeneity of the acute response to CS exposure between large and small airways using in vitro three-dimensional (3D) cultures. We used two in vitro 3D human airway epithelial tissues from large and small airway epithelial cells, namely, MucilAir™ and SmallAir™, respectively, which were derived from the same single healthy donor to eliminate donor differences. Impaired epithelial functions and altered gene expression were observed in SmallAir™ exposed to CS at the lower dose and earlier period following the last exposure compared with MucilAir™. In addition, severe damage in SmallAir™ was retained for a longer duration than MucilAir™. Transcriptomic analysis showed that although well-known CS-inducible biological processes (i.e. inflammation, cell fate, and metabolism) were disturbed with consistent activity in both tissues exposed to CS, we elucidated distinctively regulated genes in only MucilAir™ and SmallAir™, which were mostly related to catalytic and transporter activities. Our findings suggest that CS exposure elicited epithelial dysfunction through almost the same perturbed pathways in both airways; however, they expressed different genes related to metabolic and transporter activities in response to CS exposure which may contribute to cytotoxic heterogeneity to the response to CS in the respiratory tract.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a disease state characterized by poorly reversible, limited airflow that is usually both progressive and associated with an abnormal inflammatory response of the lung. 1 One cause of COPD is chronic exposure to airborne materials such as cigarette smoke (CS), which leads to impaired respiratory function in damaged tissues. 2 –7 Damaged epithelial tissue initiates repair processes including proliferation and redifferentiation until there is complete regeneration of a pseudostratified epithelium. 8,9 These repair processes in airway epithelial tissues are essential for maintaining normal airway function. However, impairment of epithelial repair leads to architectural changes through region-dependent remodeling processes that are associated with a fixed airflow limitation in COPD. 10 –15 For example, airway remodeling such as squamous cell metaplasia, goblet cell hyperplasia, and loss of ciliated epithelial cells are reported in large airways, while goblet cell metaplasia, peribronchiolar fibrosis, and loss of alveolar attachments are described in small airways. 16 –18 Currently, it is not fully understood what factors mostly contribute to airway remodeling heterogeneity in COPD pathogenesis.
Cutting-edge research on regional heterogeneity in the respiratory tract has demonstrated that small airways are more functionally associated with airway remodeling throughout the respiratory tract. 19 Nonetheless, small airways still constitute one of the least understood and challenging areas of the lung due to in vivo inaccessibility. 20 Compared with the large airways, the small airways are difficult to visualize by imaging techniques, and small airway cells are also difficult to sample by bronchoscopic transbronchial biopsy compared with large airways. Therefore, researchers have investigated new methodology to approach these issues. 21,22 This inaccessibility of small airways also makes it difficult to conduct comparative investigations of small airway tissue and other airway tissues in response to external stimulation with airborne materials, including CS. Nonetheless, such understanding is important when considering remodeling processes of damaged tissues throughout the respiratory tract.
Current advances in in vitro cell culture technologies can address these areas of concern. Airway epithelial cells under air–liquid interface (ALI) culture conditions can differentiate into a pseudostratified columnar epithelium with similar cell populations observed in vivo, therefore differentiated three-dimensional (3D) cultures of airway tissues are predicted to be highly relevant with human tissue. 23,24 Furthermore, ALI culture allows direct exposure to airborne materials including CS. 25 Thus, these 3D culture models are useful for investigation of CS effects on proximal and distal lung regions, as well as understanding the contribution of different respiratory tract regions from a mechanistic viewpoint.
Accordingly, in this study, we used two in vitro human airway epithelial 3D culture models, namely, MucilAir™ and SmallAir™ tissues, which are derived from large and small airway epithelial cells, respectively. To focus on regional heterogeneity of the respiratory tract, tissues from a single donor were used to eliminate potential donor-to-donor differences in response to external stimuli. We exposed the tissues to whole CS and examined causal alterations in tissue function and the transcriptome during different periods following the last exposure.
Materials and methods
Cell culture
MucilAir™ derived from human large airway epithelial cells and SmallAir™ derived from human small airway epithelial cells were purchased from Epithelix Sàrl (Geneva, Switzerland). The cultures were composed of epithelial cells from a 71-year-old Caucasian female nonsmoker: MucilAir™ (batch number: MD068001) and SmallAir™ (batch number: SA068001). MucilAir™ and SmallAir™ tissues cultured in 24-well-sized Transwell® inserts (Corning, Corning, New York, USA) were plated into 24-well plates (Corning) with 700 µL of MucilAir™ and SmallAir™ culture medium (Epithelix Sàrl), respectively. Before starting exposure, tissues were acclimatized at 37°C with a 5% CO2 atmosphere for 12–15 days. 26 The medium was changed every 2–3 days.
Whole CS exposure
The 1R6F Kentucky reference cigarette (University of Kentucky, Lexington, Kentucky, USA) was used as a representative conventional combustible cigarette. Before use, cigarettes were conditioned at 22 ± 1°C and 60 ± 3% relative humidity for at least 48 h.
Whole CS exposure was conducted using a Vitrocell® exposure system consisting of a VC 10 smoking robot and Vitrocell® 24/48 module (Vitrocell Systems, Waldkirch, Germany). One 1R6F cigarette was smoked in accordance with the International Organization for Standardization (ISO) Intense smoking regimen: specifically, using the VC 10 smoking robot with a 35-mm butt length (ISO 20778:2018), a 55-mL bell-shaped puff was taken over 2 s and repeated every 30 s with blocking of filter ventilation. The generated mainstream CS was exhausted to a dilution system with 8 s of puff exhaust time, and then mixed with a dilution flow of clean air at a relative humidity of 60%. Dilution flow was controlled with a mass flow controller (Analyt-MTC, Müllheim, Germany) to prepare different concentrations of whole CS. Cumulative dilution flow rates of 4, 2, and 1 L/min correspond to dilution of mainstream CS to concentrations of low, middle, and high, respectively. The smoke concentrations (%; vol/vol with air) used were 9.3% (low), 17.1% (middle), and 29.2% (high). Four 5-min exposures were performed at 60-min intervals to mimic a prototypical CS exposure. Tissues exposed to filtered external air were used as controls. Tissues at 4, 24, 48, and 72 h following the last exposure were subjected to the following analyses. The study design is summarized in Figure 1.
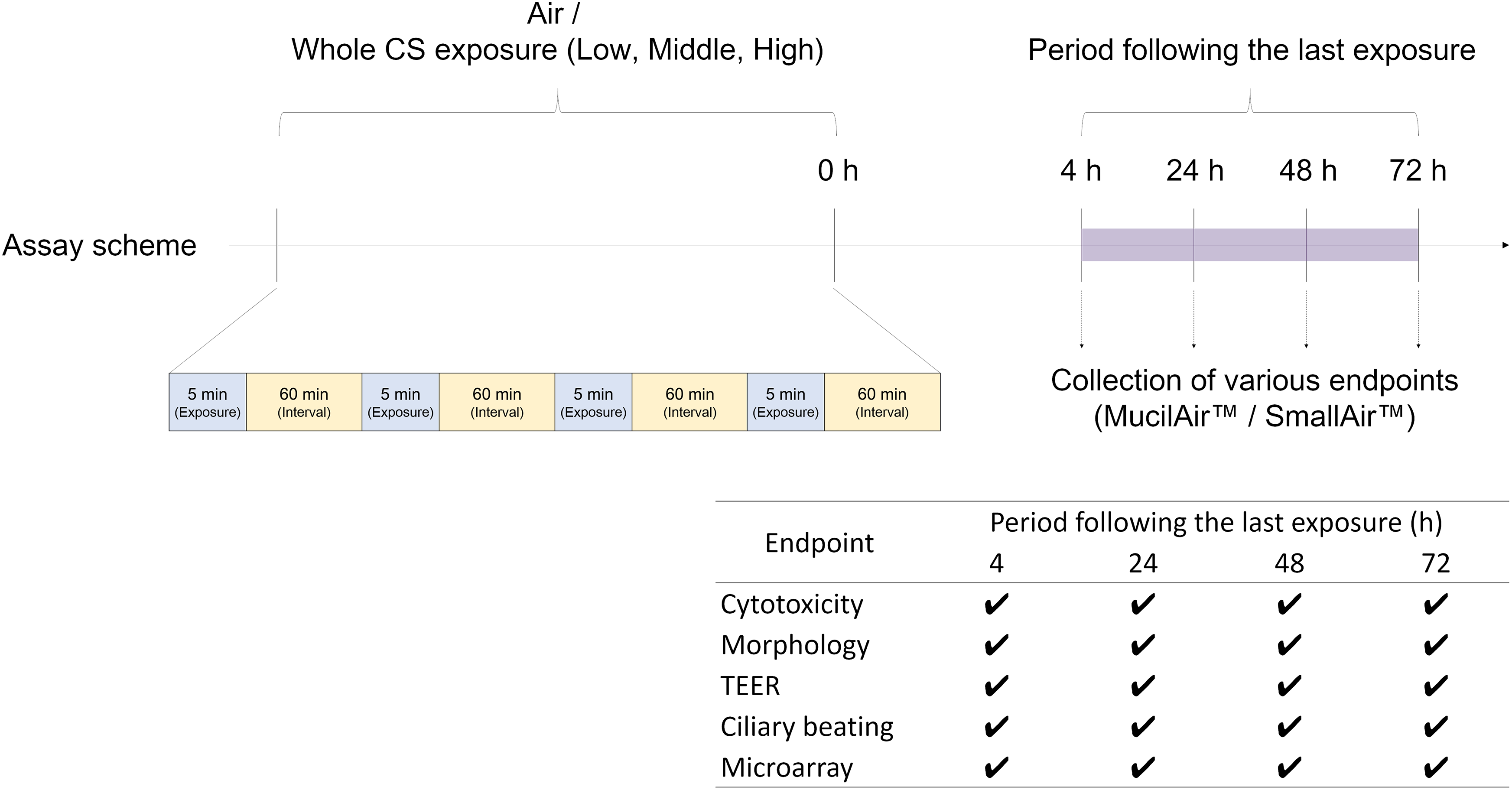
Study design. Four-times repeated exposure was performed daily at 5-min exposure duration and 60-min intervals. Tissues and sampled media at 4, 24, 48, and 72 h following the last exposure were subjected to relevant assays. Measured endpoints are listed in the table. CS: cigarette smoke; TEER: transepithelial electrical resistance.
Dosimetry analysis of delivered nicotine at different dilution flows
To validate the concentration of delivered CS to tissues in different dilution flow (low, middle, and high concentration), we quantified the amount of nicotine delivered to an insert as an index for dosimetry of CS. The 24-well-sized Transwell® inserts were modified for dosimetry of whole CS. Briefly, the microporous membrane was removed from the insert with a scalpel, then an aluminum cap was attached to the bottom of the insert. Next, modified 24-well-sized Transwell® inserts with 320 µL of distilled water were placed in the Vitrocell® 24/48 module and exposed to whole CS (also see Online Supplemental Figure S1). The amount of nicotine in the water was analyzed using an Acquity UPLC system with a photodiode array detector (Waters, Milford, Massachusetts, USA). Separation was achieved by UPLC with a CAPCEL L CORE AQ column (2.1 mm i.d. × 150 mm, 2.7 µm; Shiseido, Tokyo, Japan) and isocratic mobile phase (99% water containing 0.1% formic acid and 1% acetonitrile). The flow rate of the mobile phase was 0.4 mL/min and injection volume was 5 µL. The column temperature was maintained at 40°C. Detection was performed using a photodiode array detector at 260 nm.
Cytotoxicity measurement by adenylate kinase assay
The medium collected during each period following the last exposure was subjected to cytotoxicity analysis by measuring enzymatic activity of adenylate kinase (AK) leaked into the basolateral medium. The medium collected from MucilAir™ or SmallAir™ tissues treated with 1% Triton X-100 for 4 h was used as a positive control (100% cytotoxicity). ToxiLight™ (Lonza, Basel, Switzerland) was used in accordance with the manufacturer’s instructions. The medium was transferred to 96-well plates, then AK detection reagent was added. Luminescence was measured using an Infinite 200 PRO microplate reader (TECAN, Männedorf, Switzerland).
Histology
Tissues were fixed in 4% paraformaldehyde at 4°C and embedded in paraffin. Histological sections (5-µm thickness) were stained with hematoxylin and eosin to evaluate the morphology of the CS-exposed tissues.
Tissue integrity measurements
Transepithelial electrical resistance (TEER) was measured to examine tissue integrity of MucilAir™ and SmallAir™ tissues during each period following the last exposure using Millicell® ERS-2 Epithelial Voltohmmeter (Merck Millipore, Billerica, Massachusetts, USA). The value displayed by the voltohmmeter was multiplied by the insert surface (0.33 cm2) to obtain the resistance value of the total area (Ω × cm2).
Ciliary function measurements
The ciliary beating area (CBA) was measured using the SAVA system (Ammons Engineering, Clio, Michigan, USA) to examine the effect of whole CS on mucociliary clearance capacity. Plates containing MucilAir™ and SmallAir™ tissues were removed from the incubator and placed on a thermoplate (TokaiHit, Shizuoka, Japan) to equilibrate to 37°C before analysis. Ciliary movement (avoiding the insert periphery) was videographed using a 4× objective strength bright-field microscopy, and then the data were analyzed with the SAVA system to calculate the active beating area.
Transcriptomic analysis
To compare the effect of CS exposure on tissues from a mechanism of action viewpoint, we examined the transcriptome of MucilAir™ and SmallAir™ tissues exposed to whole CS. Total RNA was isolated from tissues during each period following the last exposure and purified using the RNeasy® Mini Kit (Qiagen, Hilden, Germany), according to the manufacturer’s instructions. RNA integrity number and quality of RNA was measured with an Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, California, USA), and was deemed appropriate at ≥7.3. Microarray analysis was conducted by Takara Bio., Inc. (Shiga, Japan) using the Human Genome U133 Plus 2.0 array (Affymetrix, Santa Clara, California, USA). Raw data were summarized by GC-Robust Multiarray Average in GeneSpring Version 14.9.1 (Agilent Technologies). Normalized intensity values below the 20th percentile and with coefficients of variation ≥50% were filtered out. The filtered list was analyzed using a moderated t-test with multiple testing correction performed by the Benjamini–Hochberg false discovery rate (FDR) to detect significant differences between the exposure groups and controls. Genes showing significant changes (|log2 fold change| > 1.0, FDR-corrected p < 0.05) were defined as differentially expressed genes (DEGs). Canonical pathway analysis was performed with “Ingenuity Toxicity List Pathways” in Ingenuity® Pathway Analysis software (Qiagen). Pathways related to organs other than airways were eliminated from the analysis. Self-organizing map (SOM) clustering was performed using the GeneSpring algorithm. Gene Ontology (GO) analysis for biological processes was conducted with the GOrilla web tool. 27,28 Transcriptomic data sets used in the current study are available in ArrayExpress (accession number: E-MTAB-8680).
Statistical analysis
The amount of nicotine measured, released AK activity, CBA, and TEER values were expressed as mean ± standard deviation. Data were considered significant at p < 0.05 with Welch’s t-test against each control.
Results
Dosimetry analysis of whole CS
To confirm the relationship between airflow dilution and delivered concentration of whole CS, we first conducted dosimetry analysis by quantifying the nicotine delivered in the modified culture insert (Figure 2(a)). The amount of nicotine consistently increased with a decreasing dilution airflow, and ranged from 0.075 ± 0.003 (low) to 0.143 ± 0.004 µg/insert (high) with high linearity. We also calculated the correlation between the whole CS concentration (%) and the amount of nicotine delivered, which suggested a high correlation (R = 1.0) between these factors (Figure 2(b)).
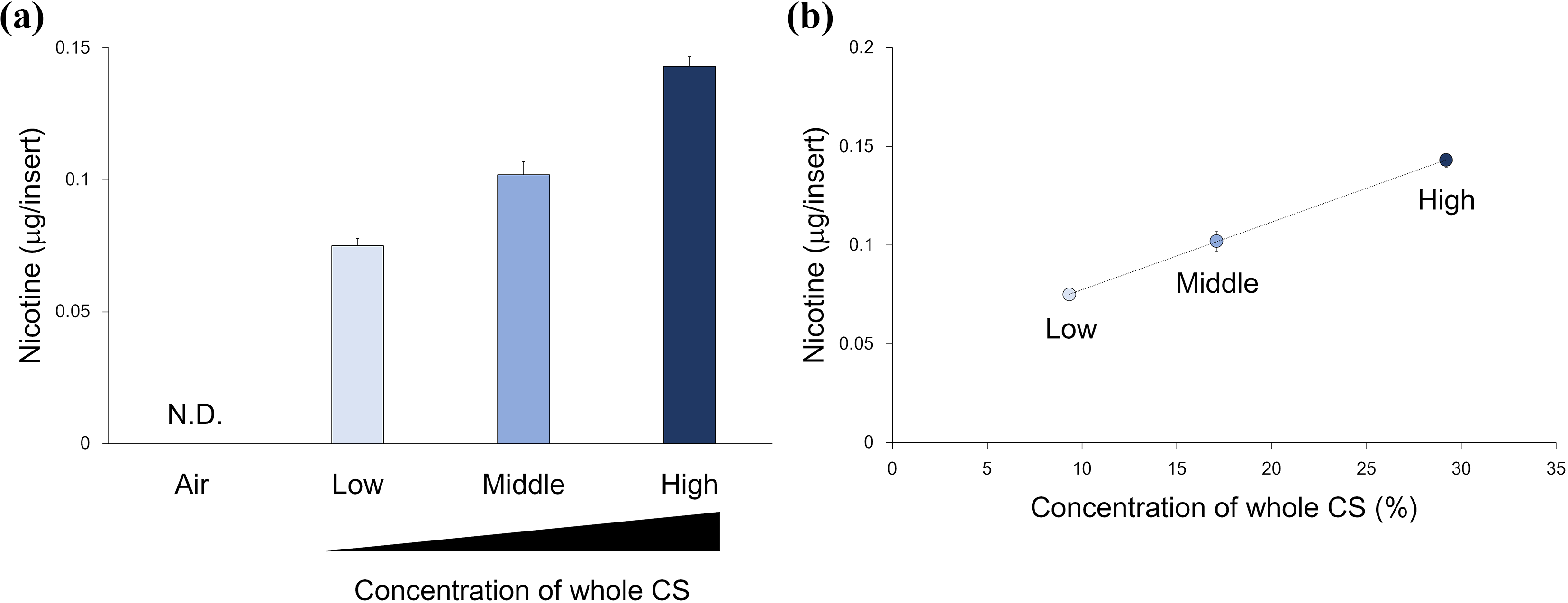
Dosimetry analysis. (a) The calculated amount of nicotine delivered in the trapping solution at different concentrations. Data represent mean ± standard deviation (n = 6). (b) The correlation of the whole CS concentrations (%; vol/vol with the airflow dilution) and nicotine delivery. The correlation coefficient was R = 1.0. Data represent mean ± standard deviation (n = 6). CS: cigarette smoke; ND: not detected.
Cytotoxicity analysis in airway epithelial tissues exposed to whole CS
The cytotoxic effect of whole CS was compared by looking at CS-exposed tissues and to each air-exposed tissue (Figure 3). Statistically significant increases in cytotoxicity were detected in both tissues exposed to any concentration of whole CS for durations longer than 24 h following the last exposure. Marked increases were observed with the middle and high concentrations of whole CS. Furthermore, with the same concentration of whole CS exposure, cytotoxicity was higher in SmallAir™ than MucilAir™ during all periods following the last exposure.
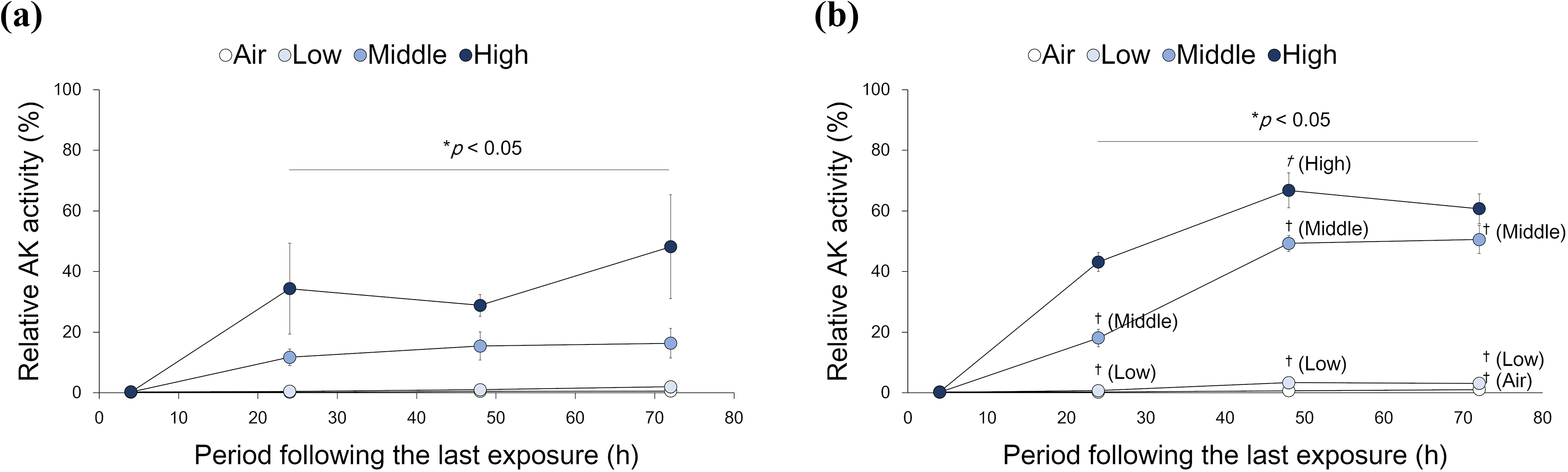
Cytotoxicity assay. Cytotoxicity was determined using media sampled from cultures exposed to whole CS with an AK release assay at various periods following the last exposure and concentrations of exposure. AK levels were normalized relative to the positive control (treated with 1% Triton X-100 for 4 h) and presented as % of cytotoxicity. Each dot indicates mean relative toxicity (%) ± standard deviation in (a) MucilAir™ and (b) SmallAir™ tissues exposed to low, middle, and high concentrations of whole CS. Open circles represent control tissues exposed to air. *Significantly different from air-exposed controls of each tissue at the same period following the last exposure (p < 0.05, Welch’s t-test; n = 6). †Significantly different between MucilAir™ and SmallAir™ tissues at the same concentration and period following the last exposure (p < 0.05, Welch’s t-test; n = 6). The statistical symbols are presented on the side with higher mean AK activity in the comparison. CS: cigarette smoke; AK: adenylate kinase.
Morphology changes in airway epithelial tissues exposed to whole CS
Consistent with the AK cytotoxicity assay, histological analysis showed destruction of both MucilAir™ and SmallAir™ tissues exposed to the middle and high concentrations of whole CS at 24 and 48 h following the last exposure (Figure 4). Interestingly, morphological changes in MucilAir™ tissue exposed to whole CS at the middle concentration appeared to be repaired at 72 h following the last exposure. Meanwhile, SmallAir™ tissue exposed to the middle concentration of whole CS showed only partial reconstruction of the pseudostratified epithelial layer, even at 72 h following the last exposure.
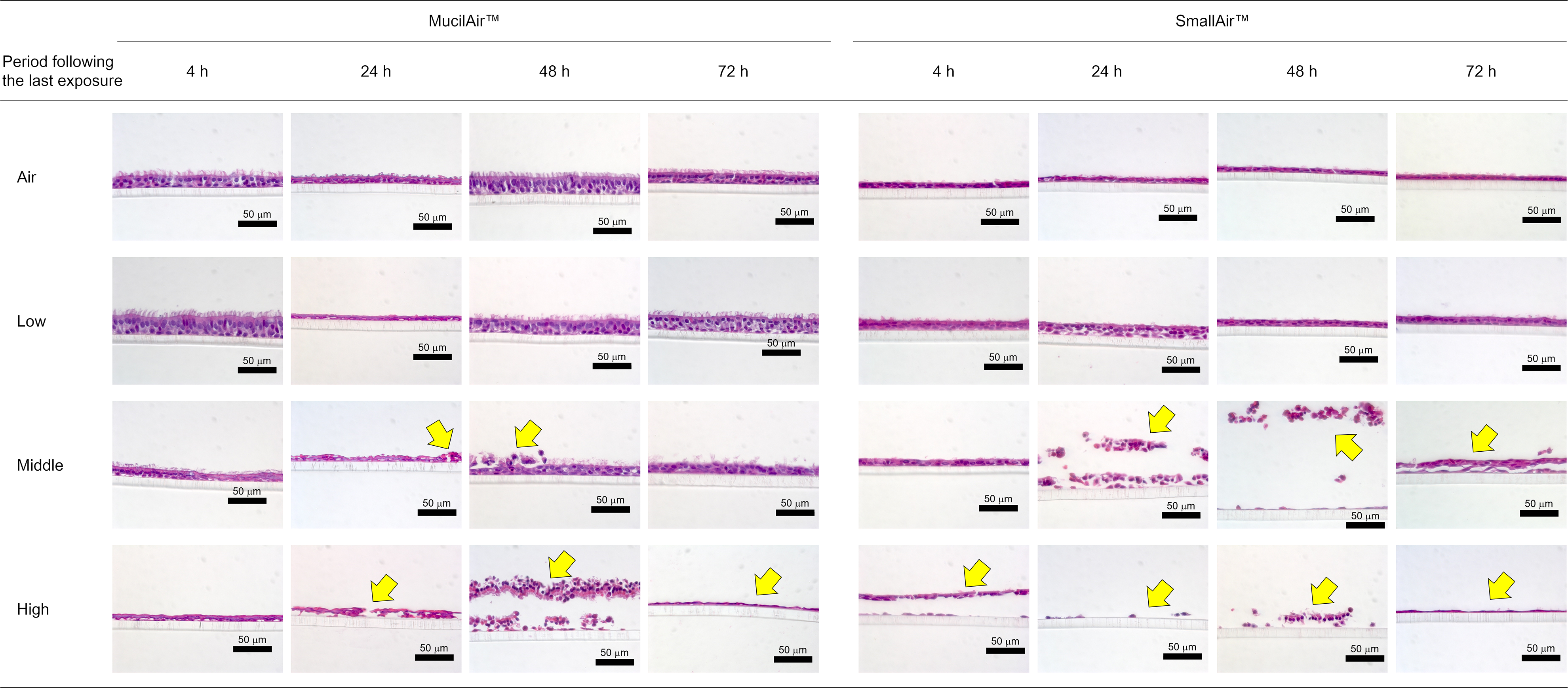
Histological assay. Representative images of tissue sections (MucilAir™ and SmallAir™ tissues) stained with hematoxylin and eosin at various periods following the last exposure and concentrations of exposure. The destruction site is indicated by yellow arrows in the images.
Tissue integrity of airway epithelial tissues exposed to whole CS
Statistically significant decreases of TEER in MucilAir™ tissue were observed at the middle and high concentrations of whole CS exposure starting at 24 h following the last exposure, with a trend toward recovery in the middle concentration at 72 h following the last exposure, but this trend was not observed at the low concentration during any period following the last exposure (Figure 5(a)). Conversely, statistically significant decreases of TEER in SmallAir™ tissue were observed starting at 4 h following the last exposure for the middle and high concentrations, which lacked significant recovery, and a decrease in TEER was also observed for the low concentration at 48 and 72 h following the last exposure (Figure 5(b)). This suggests that SmallAir™ tissue was more sensitive to whole CS than MucilAir™ tissue. In addition, consistent with the histological analysis results, recovery of impaired tissue barrier integrity was only observed in MucilAir™ tissue exposed to the middle concentration of whole CS at 72 h following the last exposure (Figure 5(a)).
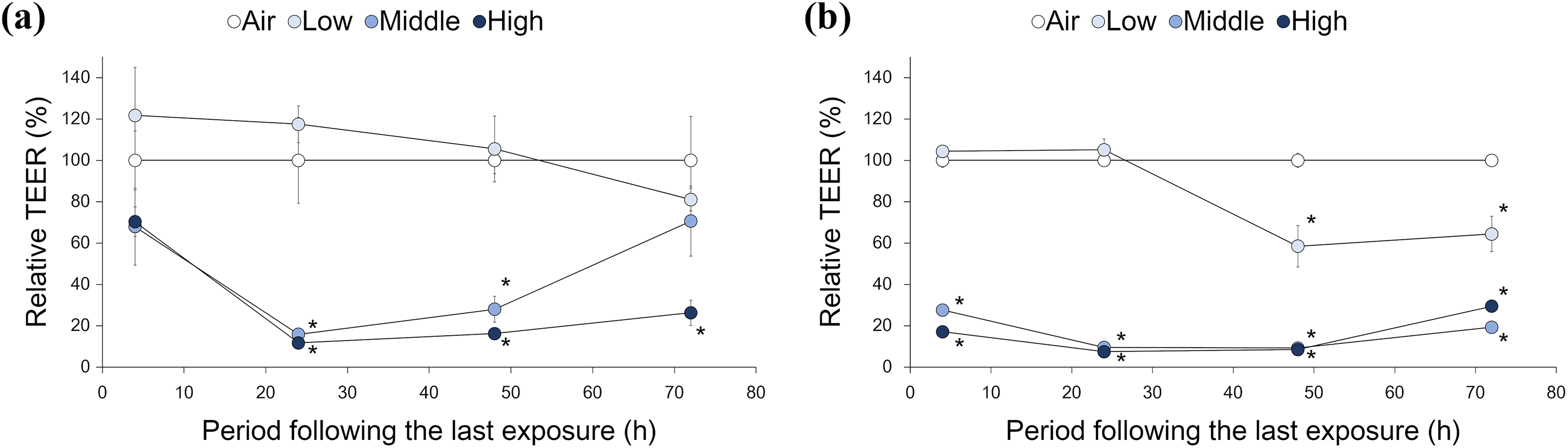
Tissue integrity assay. TEER of MucilAir™ and SmallAir™ tissues was measured at 4, 24, 48, and 72 h following completion of the repeated exposure protocol with low, middle, and high concentrations of whole CS. Each dot indicates mean TEER value relative to each air-exposed tissue at the same period following the last exposure with standard deviations in (a) MucilAir™ and (b) SmallAir™ tissues exposed to low, middle, and high concentration of whole CS. Open circles represent control tissues exposed to air. *Significantly different from air-exposed controls of each tissue at the same period following the last exposure (p < 0.05, Welch’s t-test; n = 3). CS: cigarette smoke; TEER: transepithelial electrical resistance.
Ciliary function of airway epithelial tissues exposed to whole CS
The results in Table 1 showed that the middle and high concentrations of whole CS exposure caused almost complete loss of CBA within the 72 h following the last exposure in both MucilAir™ and SmallAir™ tissues. Differences between MucilAir™ and SmallAir™ tissues were found in tissues exposed to air and low concentration of whole CS. CBA in MucilAir™ tissue at the low concentration remarkably decreased at 24 h following the last exposure (5.7% compared with the control at 24 h following the last exposure), but then gradually recovered by 72 h following the last exposure (89.0% compared with the control at 24 h following the last exposure). Further, air-exposed MucilAir™ tissue showed a slight decrease at 72 h following the last exposure (83.8% compared with the control at 4 h following the last exposure). In contrast, CBA in SmallAir™ tissue exposed to low concentration of whole CS decreased even at 4 h following the last exposure (15.2% compared with the control at 4 h following the last exposure), and did not recover by 72 h following the last exposure (63.1% compared with the control at 72 h following the last exposure). Moreover, a marked decrease in CBA remained in air-exposed SmallAir™ tissue at 72 h following the last exposure (37.0% compared with the control at 4 h following the last exposure).
Detected beating area of tissues exposed to whole 1R6F smoke.a
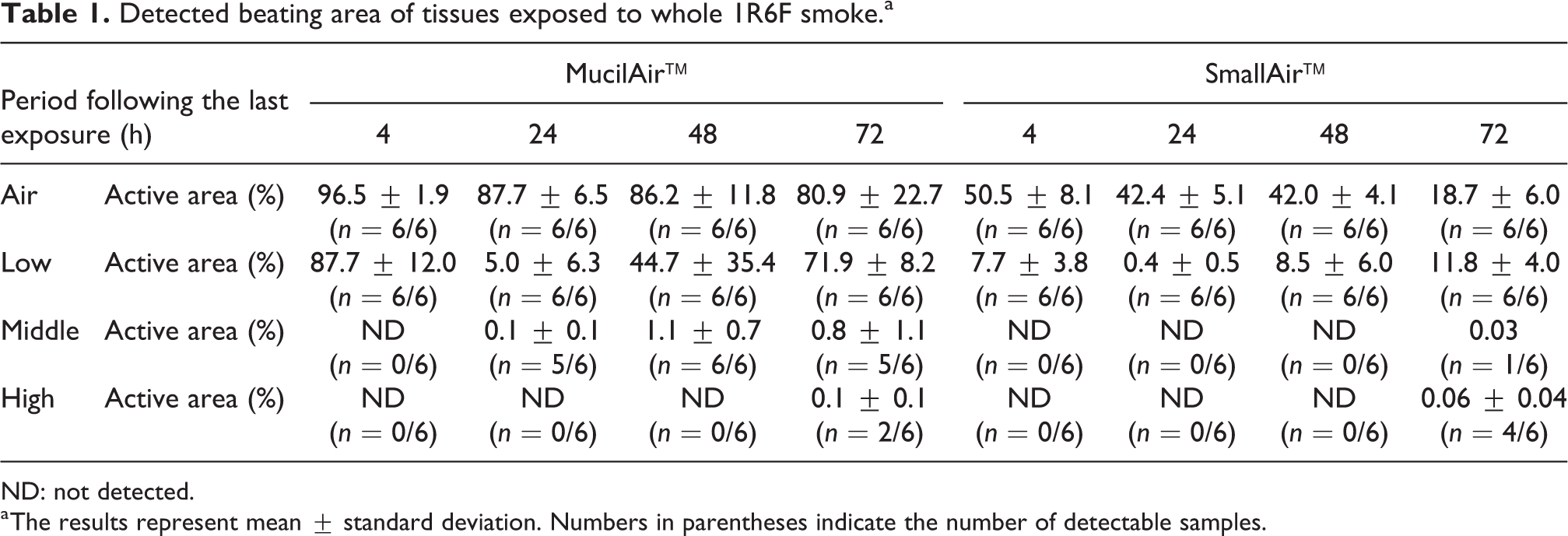
ND: not detected.
a The results represent mean ± standard deviation. Numbers in parentheses indicate the number of detectable samples.
Acute transcriptomic changes and related canonical pathways in airway epithelial tissues exposed to whole CS
We compared the effect of whole CS exposure on the transcriptome of both MucilAir™ and SmallAir™ tissues by analyzing concentration- and time-dependent alterations in number of DEGs. The number of DEGs in MucilAir™ tissue exposed to whole CS reached a maximum at 24 h following the last exposure and decreased thereafter for all concentration (Figure 6(a)). A similar trend was also observed in SmallAir™ tissue exposed to the low concentration of whole CS. However, different from MucilAir™ tissue, the number of DEGs continuously increased in SmallAir™ tissue exposed to the middle and high concentrations of whole CS during the 72 h following the last exposure (Figure 6(b)).
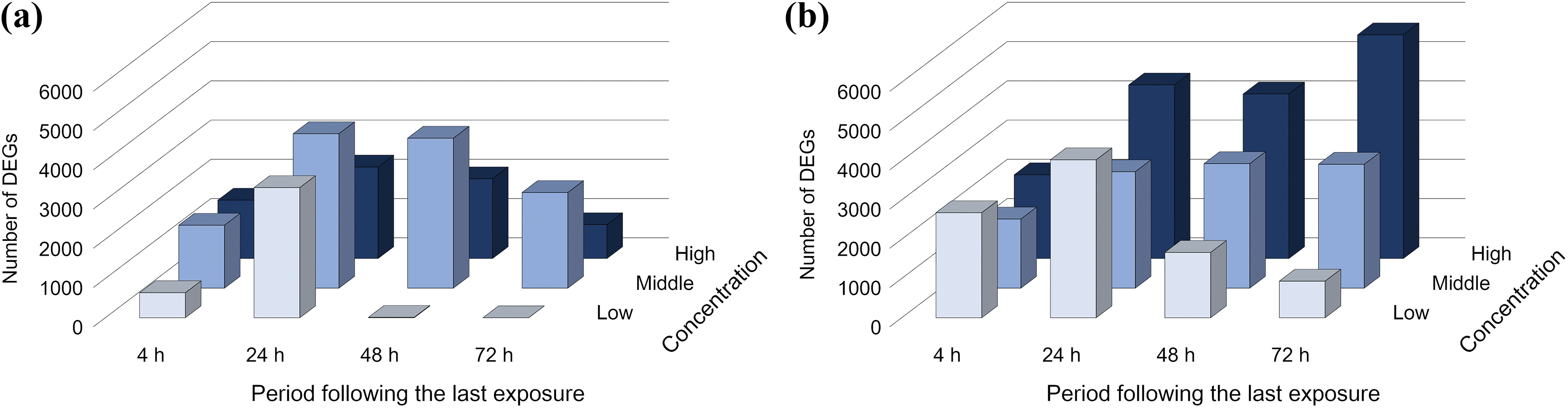
Comparison of differential gene expression. The number of DEGs of (a) MucilAir™ and (b) SmallAir™ tissues exposed to whole CS at any concentration within 72 h following the last exposure. Genes showing significant changes (|log2 fold change| > 1.0, FDR-corrected p < 0.05) were defined as DEGs. CS: cigarette smoke; DEG: differentially expressed gene; FDR: false discovery rate.
To examine the biological processes associated with these identified DEGs, we then performed canonical pathway analysis. The related canonical pathways and their activity states mostly overlapped between MucilAir™ and SmallAir™ tissues exposed to whole CS, except for “Cancer Drug Resistance by Drug Efflux” (Figure 7). Although the activation state of each pathway was almost same between MucilAir™ and SmallAir™ tissues exposed to whole CS, the number of significantly perturbed pathways was higher in the SmallAir™ tissue than the MucilAir™ tissue. Regarding MucilAir™ tissue exposed to the low concentration of whole CS at 48 and 72 h following the last exposure, the activities of listed canonical pathways were not predicted because only a few DEGs were detected in these conditions.
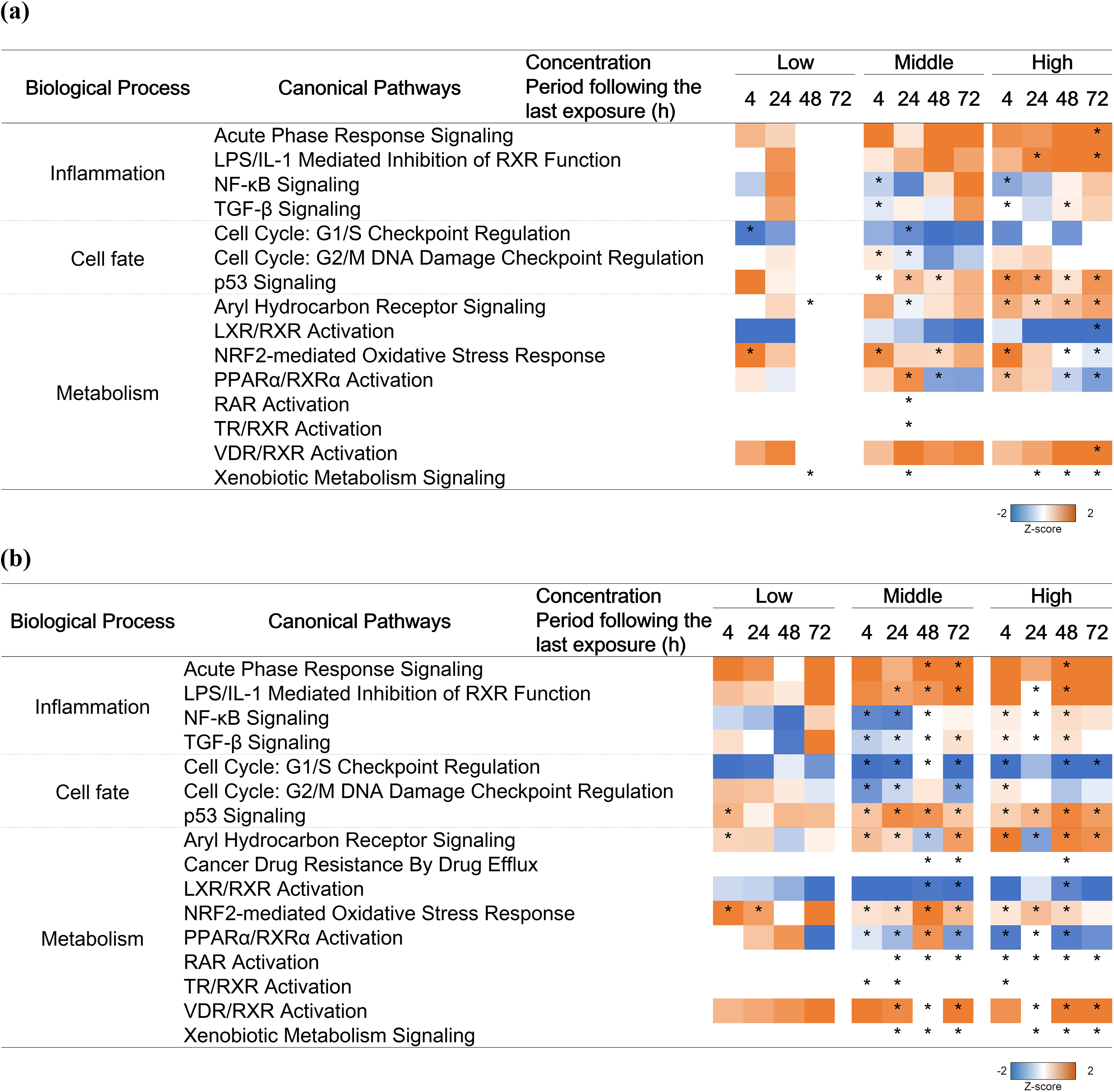
Canonical pathway analysis. The results of canonical pathway analysis for DEGs of (a) MucilAir™ and (b) SmallAir™ tissues at any concentrations within 72 h following the last exposure. The z-score is a statistical measure to infer likely activation state (orange) or inactivation state (blue) of each canonical pathway. *Significantly perturbed pathways in at least one condition are listed (FDR-corrected p < 0.05). DEG: differentially expressed gene; FDR: false discovery rate.
Comparison of gene alterations between large and small airway epithelial tissues exposed to whole CS
We further analyzed transcriptomic data using a SOM clustering algorithm to compare global gene alterations within 72 h following the last exposure between MucilAir™ and SmallAir™ tissues exposed to whole CS. For each tissue, the darkest cell indicates the most distinctive gene cluster (Figure 8(a), circled in red line). Most were considered to include concentration-dependent upregulated genes (see Online Supplemental Figure S2A). The opposing sides of cells were clustered as a theoretical countertrend to the distinct clusters (Figure 8(a), circled in red dotted line). Indeed, these clusters mostly included concentration-dependent downregulated genes (Online Supplemental Figure S2B). Among the genes in both clusters (circled by a line and dotted line), only 29% of genes overlapped (Figure 8(b)).
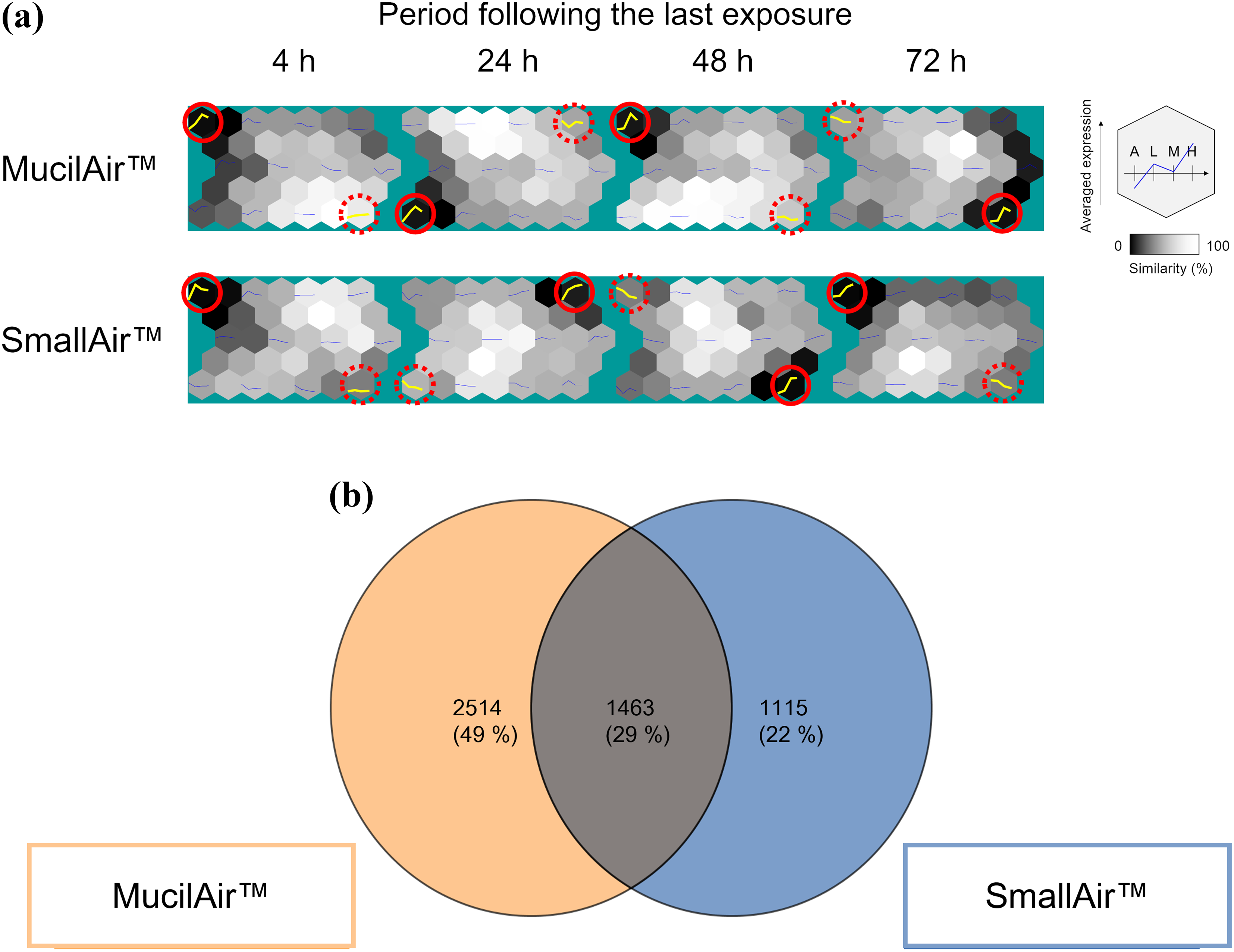
SOM analysis. (a) SOM clustering was performed with transcriptome data in MucilAir™ and SmallAir™ tissues exposed to whole CS. The U-Matrix view displays clusters arranged in a 2D hexagonal grid such that similar clusters are physically closer to each other in the grid. Cells in the grid are of two types: nodes and non-nodes. Nodes and non-nodes alternate in the grid. Clusters are only associated with nodes. Each node displays the average expression profile of included genes at the same concentration and mapped along durations following the last exposure. This average expression profile is plotted in blue and yellow. The purpose of non-nodes is to indicate similarity between neighboring nodes: if a non-node between two nodes is very bright, this indicates that the two nodes are similar. If the non-node is comparatively darker, then the two nodes are different. Furthermore, the shade of a node reflects its similarity to its neighboring nodes. Thus, cells circled in red lines were identified as the most distinct clusters, and the opposing sides of cells circled in dotted red lines were determined as countertrend clusters. (b) Venn diagram of genes identified by SOM analysis for all periods and concentrations (circled clusters of each MucilAir™ and SmallAir™ tissue in Figure 8(a)). A: air-exposed group; L: group exposed to low concentration; M: group exposed to middle concentration; H: group exposed to high concentration; SOM: self-organizing map; CS: cigarette smoke; 2D: two-directional.
To determine the biological processes of clustered genes in each MucilAir™ and SmallAir™ tissue, we then performed GO enrichment analysis for the biological process category. GO analysis showed a hierarchical GO tree that included the enriched GO terms with a directed acyclic graph in each alphabetically ranked gene list. The GO terms at the top of the hierarchy were as follows: “catalytic activity,” “molecular function regulator,” and “transporter activity” in commonly regulated genes; “catalytic activity,” “binding,” “molecular function regulator,” and “transporter activity” in genes regulated only in MucilAir™; “catalytic activity” and “transporter activity” in genes regulated only in SmallAir™ (enlarged in Figure 9(a)). We also compared the functional category of proteins encoded by regulated genes. In all cases, the top three categories were same, enzyme (33.7–39.0%), transcription regulator (20.6–22.0%), and transporter (9.3–12.7%) (Figure 9(b)). Full-size images are available in Online Supplemental Figures S4 to 6 (Figure S4: Common genes, Figure S5: Genes only in MucilAir™, Figure S6: Genes only in SmallAir™).
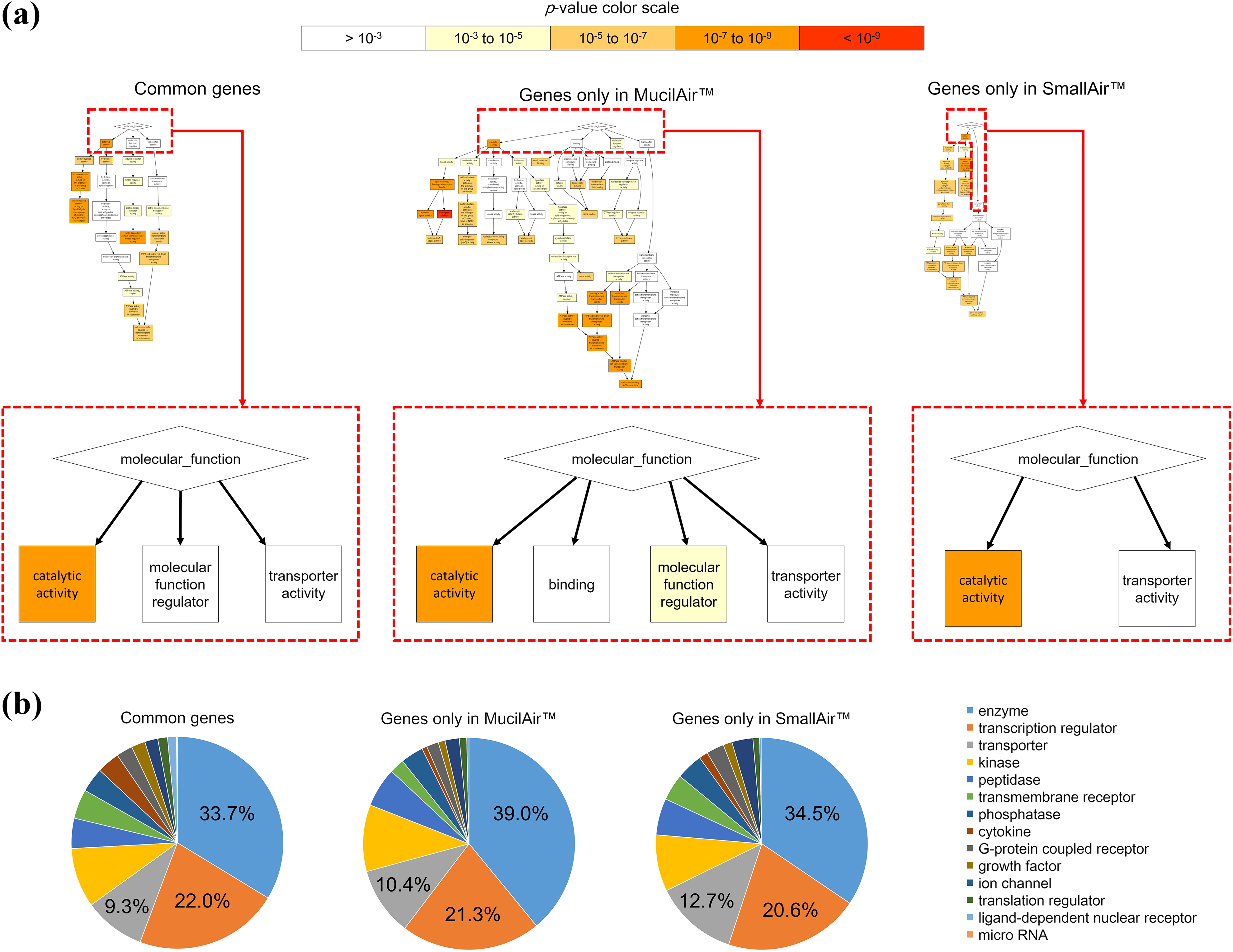
Function analysis of identified genes. (a) Chart shows enriched GO terms upon the category of biological functions for commonly perturbed genes in both MucilAir™ and SmallAir™ tissues exposed to whole CS (left), and regulated in only MucilAir™ (middle) or SmallAir™ (right) tissue exposed to whole CS. The gathering point of enriched GO terms are enlarged. Sensitivity was adjusted by changing p-value threshold to p < 10−5. (b) Pie chart represents function categories of proteins encoded by commonly perturbed genes in both MucilAir™ and SmallAir™ tissues exposed to whole CS (left), and regulated in only MucilAir™ (middle) or SmallAir™ (right) tissue exposed to whole CS. CS: cigarette smoke; GO: Gene Ontology.
Discussion
The aim of the current study was to examine regional heterogeneity of large and small airway epithelial tissues in the acute response to external stimuli. For this purpose, we exposed MucilAir™ and SmallAir™ tissues (which are recognized as human-relevant large and small airway epithelial in vitro cultures, respectively) to whole CS. Toxicological evaluation showed that whole CS exposure causes more severe and rapid damage to SmallAir™ tissue than MucilAir™ tissue, as deemed by higher cytotoxicity (Figure 3), more severe morphological destruction (Figure 4), and lower tissue integrity (Figure 5) in SmallAir™ tissue at any concentration of whole CS exposure. Furthermore, tissue integrity and morphology of MucilAir™ tissue generally recovered over 72 h following the last exposure to CS at the low and middle concentrations, while SmallAir™ tissue did not recover from the damage at any concentration. Similarly, impairment of ciliary function was also more sensitively and rapidly reduced in SmallAir™ tissue than MucilAir™ tissue. Further, no recovery of ciliary function was found in SmallAir™ tissue, even at the low concentration of whole CS exposure. These results are consistent with a previous report by Baskoro et al., in which they demonstrated higher susceptibility of small airway epithelial cells than large airway epithelial cells to CS extract in submerged culture conditions. 29 Our transcriptomic analysis also highlighted differences in susceptibility to CS: the number of DEGs in MucilAir™ tissue showed a decrease from 24 h to 72 h following the last exposure at all concentrations, whereas SmallAir™ tissue showed a time-dependent increase in number of DEGs at middle and high concentrations of whole CS exposure (Figure 6). The SmallAir™ tissues exposed to middle and high concentrations of whole CS showed morphological destruction even at 72 h following the last exposure (Figure 4); therefore, the increase in number of DEGs might reflect the status of morphological changes in the SmallAir™ tissues. Altogether, small airways are more vulnerable than large airways to acute CS exposure, which partially reflects that the small airways are prone to pathology. 30 –32
In canonical pathway analysis with DEGs at each condition, we found that pathways identified as significantly activated or inactivated mostly overlapped, and trend of directional consistency (z-score) during the period following the last exposure was also similar (Figure 7). Moreover, the number of significantly perturbed pathways was higher in the SmallAir™ tissue than the MucilAir™ tissue exposed to whole CS, and the transient canonical pathway perturbations disappeared only in the MucilAir™ tissue exposed to the low concentration of whole CS at 72 h following the last exposure. This effect was consistent with other biological analyses and suggested that the transcriptomic analysis was sensitive for detection of airway epithelial tissue impairment. The predicted canonical pathways included well-known CS-inducible biological processes, that is, inflammation (e.g. nuclear factor-κB signaling 33 ), cell fate (e.g. p53 signaling 34 ), and metabolism (e.g. aryl hydrocarbon receptor signaling 35,36 and nuclear factor erythroid 2-related factor 2-mediated oxidative stress 37 –39 ). Although the drug efflux-related pathway was predicted as significantly perturbed in only SmallAir™ tissue exposed to whole CS (at the middle concentration at 48 and 72 h following the last exposure, and the high concentration at 48 h following the last exposure), the expression profiles of genes encoding ATP-binding cassette transporters within the pathway were mostly similar between MucilAir™ and SmallAir™ tissues (Online Supplemental Figure S3). Altogether, the canonical pathway analysis suggests that key biological processes are elicited through similar canonical pathways in both MucilAir™ and SmallAir™ tissues.
Although canonical pathway analysis based on identifying DEGs with uniform thresholds of fold change and statistical significance is a standard procedure to compare transcriptomic alterations, it also has limitations. There is the possibility of overlooking small differences in whole gene expression and amplitude of gene expression changes. To counter this, SOM, one of the classification algorithms, can cluster subjected genes with similar expression patterns, therefore it likely complements the result of canonical pathway analysis through extraction of biologically useful information. 40 Indeed, we identified distinct gene clusters that mostly included concentration-dependent up- and downregulated genes in MucilAir™ and SmallAir™ tissues for all periods following the last exposure (Figure 8(a) and Online Supplemental). Interestingly, only 29% of these genes overlapped between MucilAir™ and SmallAir™ tissues, suggesting that individual gene alterations in response to CS exposure were quite different between MucilAir™ and SmallAir™ tissue. Overall, we found that these genes were mostly related to catalytic, enzymatic, and transporter activities (Figure 9). Generally, large airways are defined as >2 mm in diameter and consisting of a high density of ciliated cells, while small airways are defined as <2 mm in diameter and consisting of a high amount of club cells. 30 Club cells are the principal site of xenobiotic metabolism by the P450 system in the lung, 41 and are also the regional progenitor cells that repair damaged tissue. 42 Emura et al. also suggested that club cells are the most sensitive lung cell population because of their capacity for metabolic activation of exogenous toxic compounds. 43 Hukkanen et al. suggested that nonuniform distribution of xenobiotic activation and detoxification enzymes in the lung is one of the causes for cell-specific toxicity. 44 In addition, Bosquillon reported regional heterogeneity in transporter expression in the human lung. 45 Together, although additional studies focusing on the metabolic activity and transporter function are still needed to further understand the functional differences between large and small airway tissues, these findings suggest that differences in metabolic activity and transporter expression of each type of cell should contribute to vulnerability to whole CS between MucilAir™ and SmallAir™.
Overall, our results provide informative insight into understanding regional heterogeneity in the respiratory tract exposed to CS. We believe that using a single donor is important to address these issues, because cell and tissue characteristics are donor-specific due to varied lifestyles, medical histories, and even genetic backgrounds. However, further investigation is still needed because we used only one donor in this study. It is therefore essential to determine whether our conclusions can be applicable to other donors. Moreover, we exposed MucilAir™ and SmallAir™ tissues to whole CS for four 5-min periods over approximately 4.5 h to mimic the actual daily smoking condition of smokers as far as possible. Because CS can have acute and eventually chronic effects, it is also important to evaluate the chronic exposure effect via a repeated long-term exposure study. Moreover, when considering extrapolation of the current in vitro results to individuals, we should take into account the composition of CS reaching different regions of the respiratory tract. Several prediction models have been reported in which the deposition of CS in the pulmonary tree was estimated. 46 –49 However, the estimated deposition fraction varied because of the model structure (e.g. inclusion of cloud effects, mixing with air 50 ). In addition, some reports have shown differences in deposition between the right and left lung lobes. 51 Although the cells used in our study were from the upper airway, the donor site in the respiratory tract was not available, and the site is donor-specific. Therefore, dose extrapolation under in vitro condition to the actual human condition should be further investigated in the next step. Nonetheless, cumulative damage followed by impaired epithelial repair following acute exposures to CS on healthy tissue may be a key process leading to lung dysfunction by habitual exposure. Therefore, studies with longer periods of repeated exposure should provide further insight into determining the effects of CS on each region of the respiratory tract.
Conclusions
The current study suggested that the small airway is more susceptible to the toxic effects of whole CS than the large airway as assessed by the epithelial cell structure and functions as well as transcriptomic alterations. The CS exposure elicited common biological processes through almost the perturbation of the same canonical pathways in both large and small airways; however, there were also differences in dominantly regulated genes by CS exposure because regional heterogeneity in metabolic and transporter activities was also noted. We believe that our results provide informative insights to examine the complex mechanism of CS-inducible heterogeneous respiratory diseases.
Supplemental material
Supplementary_Figure_S1_(600dpi) - Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures
Supplementary_Figure_S1_(600dpi) for Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures by Kazushi Matsumura, Takeshi Kurachi, Shinkichi Ishikawa, Nobumasa Kitamura and Shigeaki Ito in Toxicology Research and Application
Supplemental material
Supplementary_Figure_S2_(600dpi) - Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures
Supplementary_Figure_S2_(600dpi) for Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures by Kazushi Matsumura, Takeshi Kurachi, Shinkichi Ishikawa, Nobumasa Kitamura and Shigeaki Ito in Toxicology Research and Application
Supplemental material
Supplementary_Figure_S3 - Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures
Supplementary_Figure_S3 for Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures by Kazushi Matsumura, Takeshi Kurachi, Shinkichi Ishikawa, Nobumasa Kitamura and Shigeaki Ito in Toxicology Research and Application
Supplemental material
Supplementary_Figure_S4_Common_genes - Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures
Supplementary_Figure_S4_Common_genes for Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures by Kazushi Matsumura, Takeshi Kurachi, Shinkichi Ishikawa, Nobumasa Kitamura and Shigeaki Ito in Toxicology Research and Application
Supplemental material
Supplementary_Figure_S5_Genes_only_in_MucilAir - Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures
Supplementary_Figure_S5_Genes_only_in_MucilAir for Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures by Kazushi Matsumura, Takeshi Kurachi, Shinkichi Ishikawa, Nobumasa Kitamura and Shigeaki Ito in Toxicology Research and Application
Supplemental material
Supplementary_Figure_S6_Genes_only_in_SmallAir - Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures
Supplementary_Figure_S6_Genes_only_in_SmallAir for Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures by Kazushi Matsumura, Takeshi Kurachi, Shinkichi Ishikawa, Nobumasa Kitamura and Shigeaki Ito in Toxicology Research and Application
Supplemental material
Supplementary_file_1_(revised) - Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures
Supplementary_file_1_(revised) for Regional differences in airway susceptibility to cigarette smoke: An investigational case study of epithelial function and gene alterations in in vitro airway epithelial three-dimensional cultures by Kazushi Matsumura, Takeshi Kurachi, Shinkichi Ishikawa, Nobumasa Kitamura and Shigeaki Ito in Toxicology Research and Application
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are employees of Japan Tobacco Inc., and have no competing interests with respect to the research, authorship, and/or publication of this article.
Ethics approval
All procedures in the current study were performed in accordance with approval from the local research ethics committees at Japan Tobacco Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
