Abstract
The effects of exposure of pregnant rats to cadmium (Cd) on the developmental and behavioral outcomes, reproductive functions, and metabolism of their male progeny were evaluated. Rats (Wistar) were injected intraperitoneally with either 0.5 or 5.0 µg Cd/kg body weight from day 12 to day 19 of pregnancy. The male offspring were evaluated for their developmental outcomes and behavioral changes. All developmental and behavioral parameters assessed were comparable among the different groups. All male pups were allowed to develop until 100 days of age and evaluated for reproductive end points. The results revealed that although the body weights and relative weights of liver, brain, kidney, testis, and epididymis were not altered, reproductive parameters, including daily sperm production, epididymal sperm numbers, and concentrations of motile, viable, and hypo-osmotic tail-swelled sperm declined significantly in rats exposed to 0.5 and 5.0 µg Cd during embryonic development. In addition, plasma testosterone levels and activity levels of testicular steroidogenic enzymes also decreased in these rats. In the fertility study, although each male in the 0.5, 5.0 µg, and control groups produced a copulatory plug and impregnated a female, the mean number of implantations and live fetuses was reduced significantly in females mated with rats exposed to 0.5 and 5.0 µg Cd during the prenatal period. The general metabolism of the animals exposed to Cd during embryonic development was comparable with the controls as evidenced by no significant changes in the activity levels of succinate dehydrogenase, lactate dehydrogenase, glucose-6-phosphate dehydrogenase, glutamate dehydrogenase, aspartate aminotransaminase, and alanine aminotransaminases in the liver, kidney, and testis. The results thus suggest that maternal Cd exposure during embryonic development markedly affected the spermatogenesis, steroidogenesis, and fertility potential, but without alterations in the development, behavior, and metabolism.
Introduction
The diverse harmful health effects upon exposure to heavy metals in the environment due to their bio-accumulative and immutable properties are a matter of serious concern and a global issue. Cadmium (Cd) is one of the most toxic metals and is widely distributed in the environment. Cd is used as an anticorrosive in plating metals and other alloys that are valuable in the industry and, as Cd oxide, in storage batteries. 1 With the wide application of Cd-related products in industrialized nations, hazardous exposure to Cd is increasing. Cd has been linked to osteomalacia, hepatotoxicity, renal toxicity, neurotoxicity, as well as infertility and cancer. 2 –4 The toxic action of Cd is documented to be multifactorial. Cd has a long half-life period and resides in the body for about 15–30 years. 5 Due to its protracted half-life and non-degradable properties, Cd accumulates in the human body for a long time even after minimal exposure and has severe toxic effects. Interestingly, pregnant and lactating animals were reported to absorb and retain substantially more dietary Cd than do their non-pregnant counterparts, and Cd can be transferred to fetuses. 6,7 Studies have revealed that Cd can accumulate in the granulosa cells in ovarian follicles and exert serious toxic effects by destroying the cell structure of the ovary and affecting its physiological functions such as hormone synthesis and follicular maturation. 8 –10 Johnson et al. 11 have reported that Cd also accumulates in the uterus and mammary glands. Previous studies have shown that functionally, Cd acts like steroidal estrogen in breast cancer cells as a result of its ability to bind to form a high-affinity complex with the hormone-binding domain of the estrogen receptor. 11 The possible estrogenic activity of Cd could affect the sexual differentiation and maturation of individuals exposed to Cd in utero and during lactation.
It is well-known that perinatal exposure to estrogens and estrogen-like substances causes early onset of puberty and alters mammary gland development in female offspring, 12 –14 reduced testicular size and sperm production, and decreased circulatory testosterone levels and sub-fertility in male progeny. 15,16 Although there is an association between environmental Cd exposure and the increased incidence of reproductive toxicity, 3,17 little attention has been paid to the effect of Cd on maternal-mediated reproductive toxicity. In view of the fact that Cd accumulates in the ovaries and uterus and exhibits estrogen-like activity, the present work was undertaken to gain further knowledge about the effects of exposure to Cd during embryonic development on the reproduction of male progeny at adulthood. In addition, this study broadened its scope by including developing milestones, behavior, and metabolism of male offspring which have not been well characterized following embryonic Cd exposure.
Materials and methods
Animals and treatments
Fifty-one timed-pregnant Wistar rats were obtained from an authorized vendor (Raghavendra agencies, Bengaluru, India) and randomly divided into three groups containing 17 animals in each group. The animals were housed individually in polypropylene cages with sterilized paddy husk as bedding material, in a well-ventilated animal house facility of the Department of Zoology, S.V. University (Tirupati, India) at 25°C ± 2°C with a 12 h photoperiod and relative humidity of 60% ± 10%. The animals were fed on a pellet diet (Sai Durga feeds and foods, Bengaluru, India) and drinking water ad libitum. All the experiments were carried out in accordance with the guidelines of the Committee for the Purpose of Control and Supervision on Experiments in Animals, Government of India. 18 The protocol and animal use were also reviewed and approved by the Institutional Animal Ethical Committee at Sri Venkateswara University, Tirupati, India (regd. no. 438/01/a/CPCSEA dated 17-07-2001) with a resolution no. 10/(i)/a/CPCSEA/IAEC/SVU/PSR dated 08-07-2012.
The rats were injected intraperitoneally with either 0.5 or 5.0 µg Cd/kg body weight from day 12 to day 19 of pregnancy in 100 µL volume. The control rats were injected with 100 µL saline solution. Although humans are principally exposed to heavy metals orally, intraperitoneal injection of substances is a common route of administration in laboratory rodents to test their effects. In the present study, the animals were administered with Cd at doses below the No Observed Effect Level (NOEL) to evaluate whether low doses of Cd exposure during embryonic development induce any alterations in development, behavior, and reproduction. The doses of Cd were selected based on our earlier studies. 19 Besides, similar Cd dosages, route of administration and time schedule were also adopted by Johnson et al. to evaluate the prenatal Cd exposure on female offspring reproduction. 11 The rats were allowed to deliver pups. Within 24 h of birth, the pups were counted, weighed, and redistributed to foster dams (eight pups per dam). All foster dams were saline-treated during pregnancy.
During pregnancy, the dams were observed for clinical signs of toxicity on a daily basis. Cage-side observations included, but were not limited to, behavioral changes (such as, agitation, lethargy, hyperactivity, and cannibalism), neurological changes (such as, convulsions, tremors, muscle rigidity, and hyper-reflexia), and autonomic signs (such as, lacrimation, piloerection, pupil size, appearance of diarrhea, genital swelling, redness and vaginal bleeding, changes in skin and fur, mucous secretions, edema, and unusual respiratory pattern). Additionally, the following data were recorded: pregnancy length, litter size, pup birth weight, and pup weaning weight. Of the pups maintained, the male pups for each group were as follows: control, 58; 0.5 µg Cd, 56; and 5.0 µg Cd, 60. The pups were also observed for bilateral pinna unfolding, fur development, lower and upper incisor eruption, bilateral eye opening, and testis descent into scrotal sac besides behavioral parameters. Male pups were weaned on postnatal day (PND) 22, maintained on normal diet and water, and observed regularly until used for mating at PND 90 and/or necropsied at PND 100.
Behavioral parameters
Three male pups from each mother rat (50 pups from each group) were selected randomly and subjected to the following behavioral tests: cliff avoidance within 30 s (on PND 5), surface righting reflex (on PND 6), negative geotaxis (on PND 7) and ascending wire mesh (on PND 16), open field (on PND 45), and exploratory behavior (on PND 45). Open field behavior was measured for 3 min in a chamber measuring 90 × 90 × 90 cm3 in height; Exploratory behavior was measured for 3 min in a box with a hole board bottom (90 × 90 cm2) containing three equally spaced holes (3 cm in diameter) in the floor. The detailed methodology is published elsewhere. 20,21 All behavioral tests were performed between 08:00 am and 10.00 am. All the behavioral testing procedures were conducted by an investigator blind with regard to the treatment groups.
Fertility studies
Thirty adult virgin female rats (80-day old) showing regular estrus cycle were employed in the fertility test. Ten 90-day-old male rats each from the 0.5, 5.0 µg, and control groups were transferred to a mating cage and cohabited with female rats in proestrus stage (1:1) for 8 days. Females were checked every morning for the presence of copulatory plugs, and vaginal washings were evaluated for the presence of sperm. Rats with positive plugs and/or sperm in vaginal smear were separated from the males and were autopsied on the 18th day of pregnancy to record the number of live fetuses and implantations in both uteri horns. In addition, both ovaries were removed and examined for the number of corpora lutea. Data were analyzed to determine the effect of embryonic exposure to Cd on preimplantation loss (difference between number of corpora lutea and number of implantations/number of implantation sites × 100) and post-implantation loss (difference between number of implantation sites and number of live fetuses/number of live fetuses × 100). In addition to mating (number of mated males/number of paired males × 100), fertility indices (number of pregnant females/sperm positive females × 100), and the conception time, the interval between the first day of cohabitation and the day of presence of vaginal plug and/or sperm in vaginal washing was recorded.
Body and organ weights
Body weight was recorded on PND 100 and weight gain/loss in each group was calculated. The weights of the testis, epididymis, liver, kidney, and brain were recorded for each animal, and the relative weights (weight of the organ/100 g body weight) were calculated.
Daily sperm production
The number of homogenization resistant spermatids in the testis was determined according to the protocol described by Blazak et al. 22 Briefly, the testis was thawed, the capsule was detached and weighed, and the parenchyma was homogenized in 50 mL of ice-cold 0.9% sodium chloride (NaCl) solution containing 0.5% Triton X-100 using a glass-Teflon homogenizer (polytron, PT1600E) at a minimum speed for 1 min. The homogenate was filtered through a metal sieve and the filtrate was used to count the number of homogenization-resistant spermatids using a hemocytometer. Daily sperm production (DSP) was calculated by dividing the total number of spermatids/sperm per testis by 6.1 days, the duration step of 19 spermatids in the seminiferous epithelial cycle. 23
Epididymal sperm analysis
The epididymis was excised at autopsy and placed in a 10 mL vial containing 1.0 mL of physiological saline at 37°C. A suspension of spermatozoa was prepared by mincing with a sterile razor blade on a glass slide in a few drops of physiological saline. A drop of resulting sperm suspension was used for sperm analysis. The numbers of motile, viable, and hypo-osmotic (HOS) tail-swelled sperm were determined according to the World Health Organization laboratory manual. 24
Testosterone assay
Serum level of testosterone was determined by ChemiLuminiscent Immunosorbent assay (CLIA) using a commercial kit, as per manufacturer’s instruction (Master CLIA vast enabled kit). The relative light unit values were measured with Maglumi 600 (Diasystem Scandinavia AB, Sweden). Each sample was run in duplicate and 10% of the total samples were retested randomly. The sensitivity of the assay was 0.025 ng/mL. The intra- and interassay coefficient of variations were 8.5% and 9.4%, respectively.
Assay of steroidogenic enzymes
Activities of 3β-hydroxysteroid dehydrogenase (3β-HSD) (EC.1.1.1.51) and 17β-HSD (EC.1.1.1.61) were measured in the testis according to the method described by Bergmayer. 25 The testicular tissue was homogenized in ice-cold 20 mM Tris-HCl buffer (pH 6.8) with the help of a motor-driven glass Teflon homogenizer on crushed ice for a minute. The microsomal fraction was separated and used for enzyme assays.
The reaction mixture in a final volume of 2.0 mL contained 100 µmol of sodium pyrophosphate buffer (pH 9.0), 0.5 µmol of co-factor (nicotinamide adenine dinucleotide (NAD) for 3β-HSD and nicotinamide adenine dinucleotide phosphate (NADPH) for 17β-HSD), 0.1 µmol of substrate (dehydroepiandrosterone for 3β-HSD and androstenedione for 17β-HSD), and 20 mg equivalent of microsomal protein as enzyme source. The reactions were carried out in a quartz cuvette of 1.0 cm path at 23°C. The absorbance at 340 nm was measured at 10-s intervals for 2 min in an ultraviolet–visible spectroscopy spectrophotometer (Hitachi model: U-2001, Japan) against blank containing all the components except the enzyme source.
Histological evaluation of testes
For histological study, testes were excised from the control and experimental rats. Three- to five-millimeter-thick slices of tissue from the right testis were fixed in Bouin’s solution for 24 h. The fixed specimens were dehydrated in ascending series of alcohol and embedded in paraffin after clearing in xylol. Tissues were sectioned at 6 µm, and the sections were stained with hematoxylin-eosin, and examined with an Olympus phase contrast microscope (model no: BX41TF). 26 Morphometrical measures were performed on five distinct areas and a total of 50 seminiferous tubules were measured in each rat. The diameter of the testicular tubules (basal lamina to basal lamina) and the height of the germinal epithelium (basal lamina to neck of elongated spermatozoa), luminal circumference, and Leydig cell density were determined by projecting the slides at ×50 using a computer-assisted image analysis system (Olympus BX43, Japan).
Activities of metabolic enzymes
The liver, testis, and kidney were isolated and immediately stored at −20°C. Tissue homogenates (10% W/V) were prepared in 0.25 M cold-sucrose solution at 4°C in an ice-jacketed glass homogenizer with a motor-driven Teflon-coated pestle. The homogenates were centrifuged at 750 g for 10 min. The pellet obtained was discarded. The supernatant was again centrifuged at 17,000 × g for 20 min and then the pellet was suspended in the homogenizing medium and again centrifuged at 17,000 × g for 20 min. The pellet thus obtained was resuspended in the homogenizing medium and used for the assay of succinate dehydrogenase (SDH) and glutamate dehydrogenase. The activity levels of lactate dehydrogenase, glucose-6-phosphate dehydrogenase, alanine amino transaminase, and aspartate amino transaminase were measured in 105,000 × g supernatant (cytosol). All enzyme assays were made within 2 h after the isolation of tissues.
All the enzyme assays were measured at 37°C with appropriate enzyme and reagent blank using spectrophotometer. The optimal conditions for each enzyme with respect to pH, and substrate and co-factor concentration were established and assays were made under zero-order kinetics after preliminary standardization regarding linearity with respect to time of incubation and enzyme concentration. The optimal assay conditions for individual enzymes 27 were as follows.
SDH (succinate: oxidoreductase; EC 1.3.99.1) was assayed by the method of Nachlas et al. 28 using 100 µmol phosphate buffer (pH 7.4), 40 µmol sodium succinate, 4 µmol INT (2-4 Iodophenyl-3(4-nitrophenyl)-5-phenyl tetrazolium chloride), and 0.5 mL of enzyme source in 2.0 mL volume.
Lactate dehydrogenase (LDH;
Glucose-6-phosphate dehydrogenase (G-6-PDH;
Glutamate dehydrogenase (GDH; glutamate: NAD+ oxidoreductase, EC 1.4.1.3) was assayed by the method of Lee and Lardy. 31 The reaction mixture in a final volume of 2.0 mL contained 100 µmol of phosphate buffer (pH 7.4), 40 µmol sodium glutamate, 4 µmol INT, 0.1 µmol NAD, and 0.5 mL of enzyme source.
Aspartate aminotransaminase (AAT;
Estimation of protein content
Protein content in the enzymes source was estimated by the method of Lowry et al. 33
Statistical analysis of data
Statistical analysis of the data for the male progeny (body weight and organ weight, sperm quality and numbers, serum testosterone concentrations, and enzyme activity levels) was performed using the litter of origin (dam) as the unit. Statistical analysis of the progeny fertility data was carried out using the litter as a unit. The results obtained were expressed as mean ± standard deviation (SD). Statistical significance was assessed by one-way analysis of variance followed by Tukey’s post hoc test, using SPSS software (SPSS for windows, release 17.0.1, 2008 SPSS Inc., Chicago, Illinois, USA). Values of p ≤ 0.05 were considered to be significant.
Results
Behaviors, body weights, and fertility output of dams
No significant changes in lacrimation, urination, respiration, vocalization, postural, or gait abnormalities were observed in any of the control and Cd exposed dams. All the animals were apparently normal and no unusual behaviors (namely, head flicking, head searching, biting, licking, self-mutilation, circling, and walking backwards) were observed in any of the pregnant rats. None of the animals were excluded from the experiment. The body weight gain of dams exposed to either 0.5 or 5.0 µg Cd during pregnancy was comparable with the controls. Similarly, the corrected body weight on GD 21 (body weight of the dams on gestational day [GD] 21 was corrected by subtracting the sum of the weights of the pups on PND 1) in the dams exposed to Cd was also comparable with the control group. No significant changes in pregnancy length and litter size were observed in the rats exposed to Cd when compared to the controls. The birth weight, crown-rump length, viability, and weaning indices of pups exposed to Cd during embryonic development were also comparable with the control pups (data not shown).
Developmental landmarks and behavior
No overt signs were apparent in any pups during the postnatal period. None of the pups in the control and experimental groups died during weaning. The elapsed time for lower and upper incisors eruption, pinna unfolding, eye slit formation, fur development, eye opening, and testes descent in pups delivered to Cd-exposed dams was comparable to the control pups. Time to perform the surfacing righting reflex, cliff avoidance, negative geotaxis movement, and ascending wire mesh were comparable among pups from different groups. Open field and exploratory behavior of rats exposed to Cd during embryonic development was also comparable with the controls (Table 1).
Mean day of appearance of developmental landmarks and behavioral observations in male progeny of 0 (control), 0.5, or 5.0 µg Cd-exposed dams.
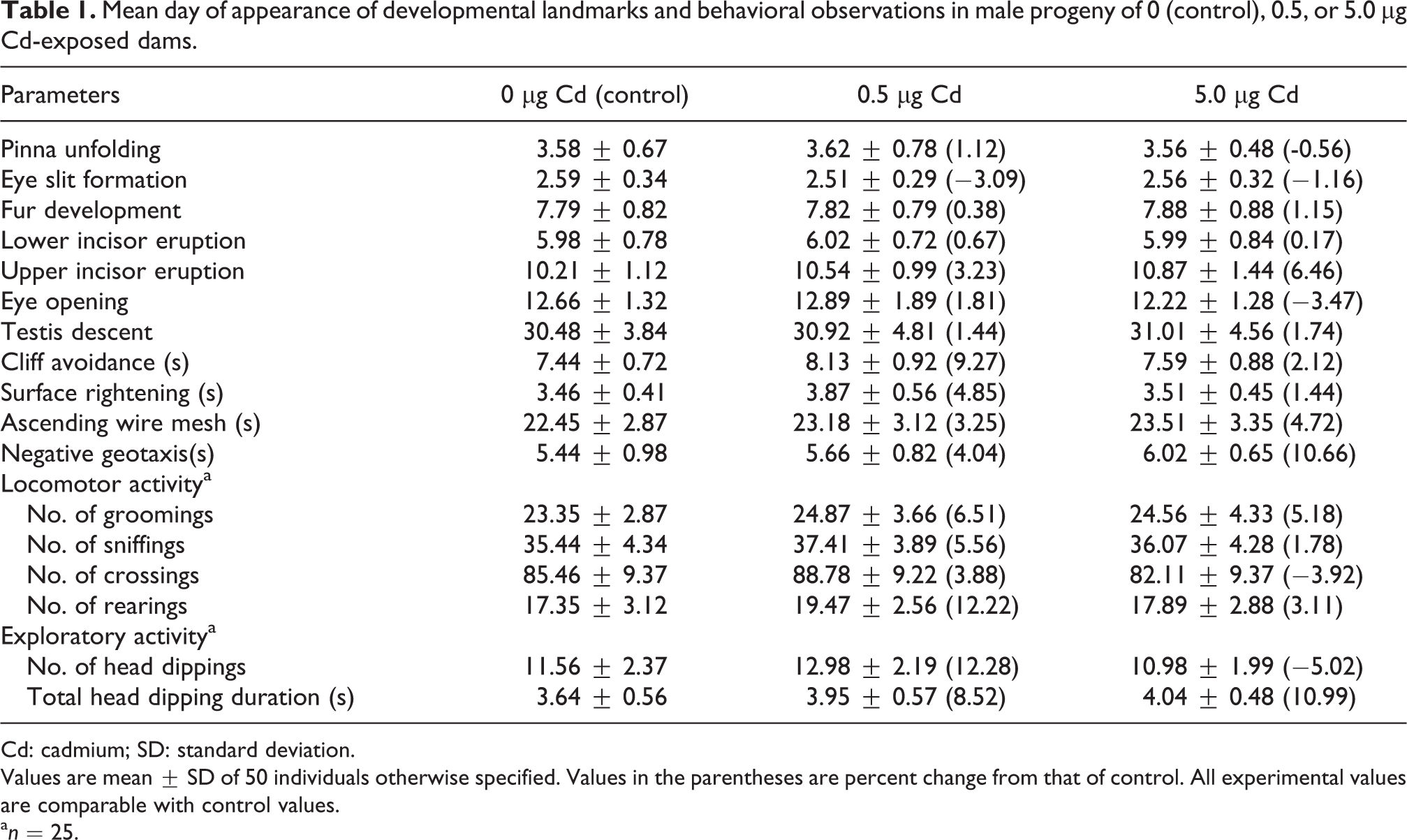
Cd: cadmium; SD: standard deviation.
Values are mean ± SD of 50 individuals otherwise specified. Values in the parentheses are percent change from that of control. All experimental values are comparable with control values.
a n = 25.
Body weight and tissue indices of male progeny on PND 100
No significant differences were noted in the body weight gain of the controls and Cd-treated male progeny. The relative weights of the liver, brain, kidney, testes, and epididymis were also comparable between the control and experimental rats (Table 2).
Effect of transplacental exposure to 0 (control), 0.5, or 5.0 µg Cd on body weights (g) and tissue indices (w/w%) in adult male rats.
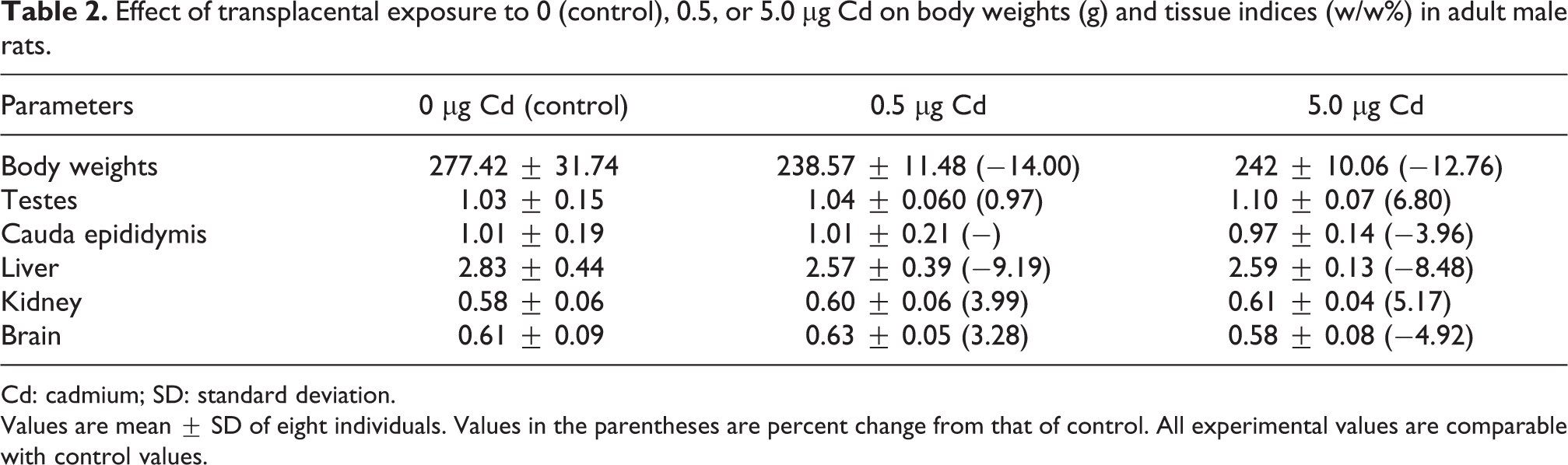
Cd: cadmium; SD: standard deviation.
Values are mean ± SD of eight individuals. Values in the parentheses are percent change from that of control. All experimental values are comparable with control values.
Sperm analysis of progeny
Exposure to Cd during embryonic development significantly decreased the testicular DSP, epididymal sperm count, and percentage of motile, viable, and HOS-tail coiled sperm in adult rats when compared with the control animals (Table 3).
Daily sperm production and epididymal sperm count of motile-, viable-, and HOS tail-coiled sperm in rats exposed to 0 (control), 0.5, or 5.0 µg Cd during embryonic development.
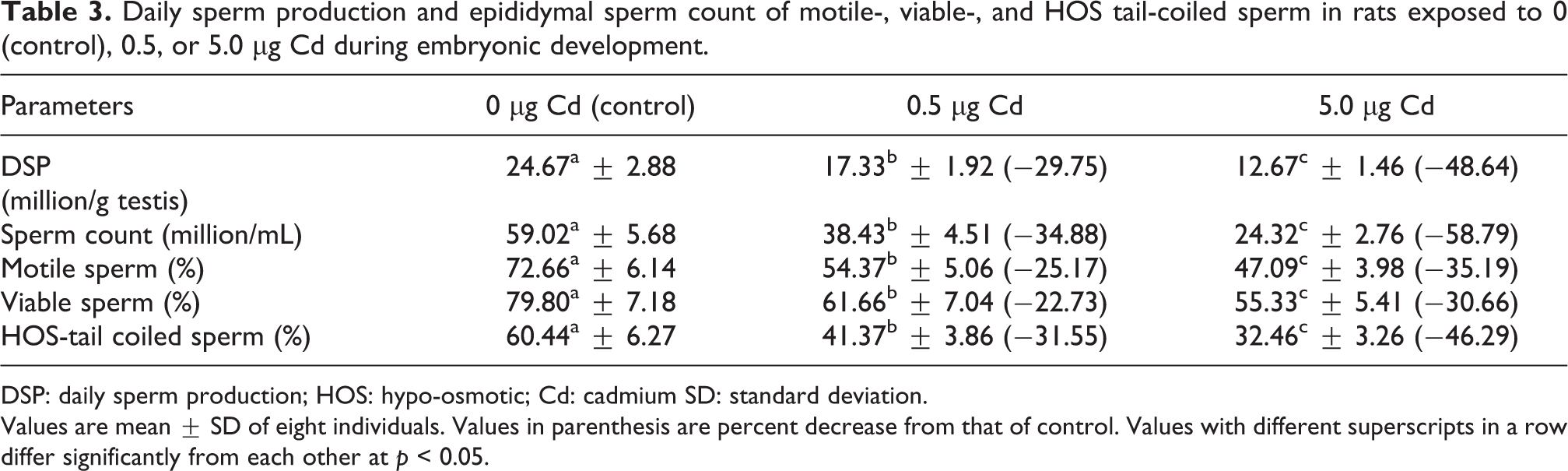
DSP: daily sperm production; HOS: hypo-osmotic; Cd: cadmium SD: standard deviation.
Values are mean ± SD of eight individuals. Values in parenthesis are percent decrease from that of control. Values with different superscripts in a row differ significantly from each other at p < 0.05.
Steroidogenic marker enzymes and serum testosterone levels in male progeny
Significant reduction in the activities of 3β-HSD and 17β-HSD was observed in the testis of rats exposed to either 0.5 or 5.0 µg Cd during embryonic development compared to the activities of control rats (Table 4). The circulatory levels of testosterone also decreased significantly in the male progeny of 0.5 or 5.0 µg Cd-treated rats (Table 4).
Effect of transplacental exposure to 0 (control), 0.5, or 5.0 µg Cd on testicular 3β- and 17β-hydroxysteroid dehydrogenase activity levels and serum testosterone levels in adult male rats.
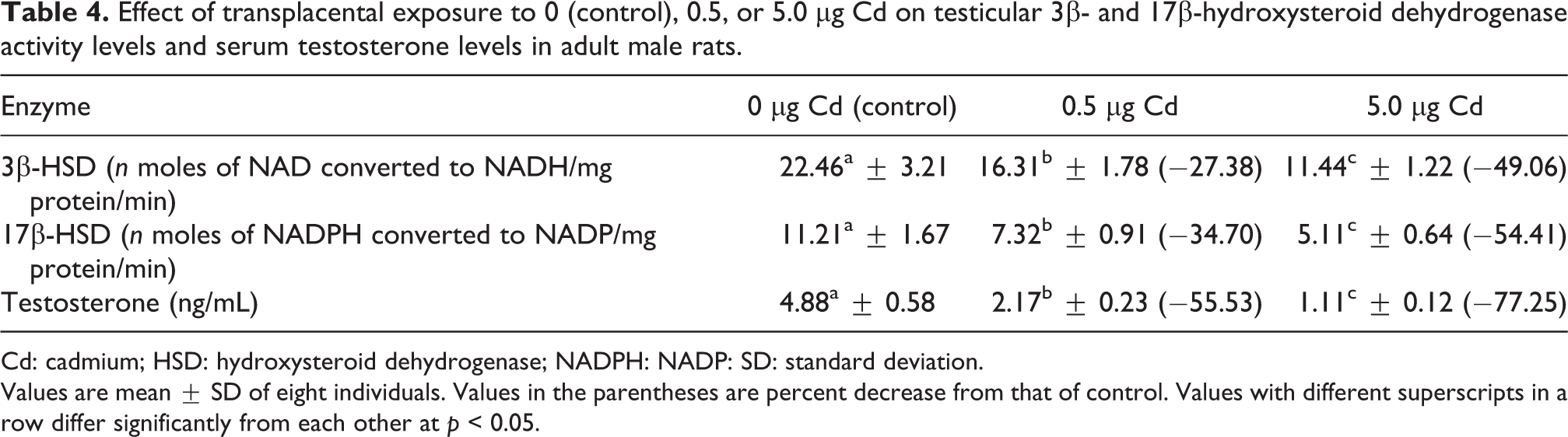
Cd: cadmium; HSD: hydroxysteroid dehydrogenase; NADPH: NADP: SD: standard deviation.
Values are mean ± SD of eight individuals. Values in the parentheses are percent decrease from that of control. Values with different superscripts in a row differ significantly from each other at p < 0.05.
Histomorphology of testis of progeny
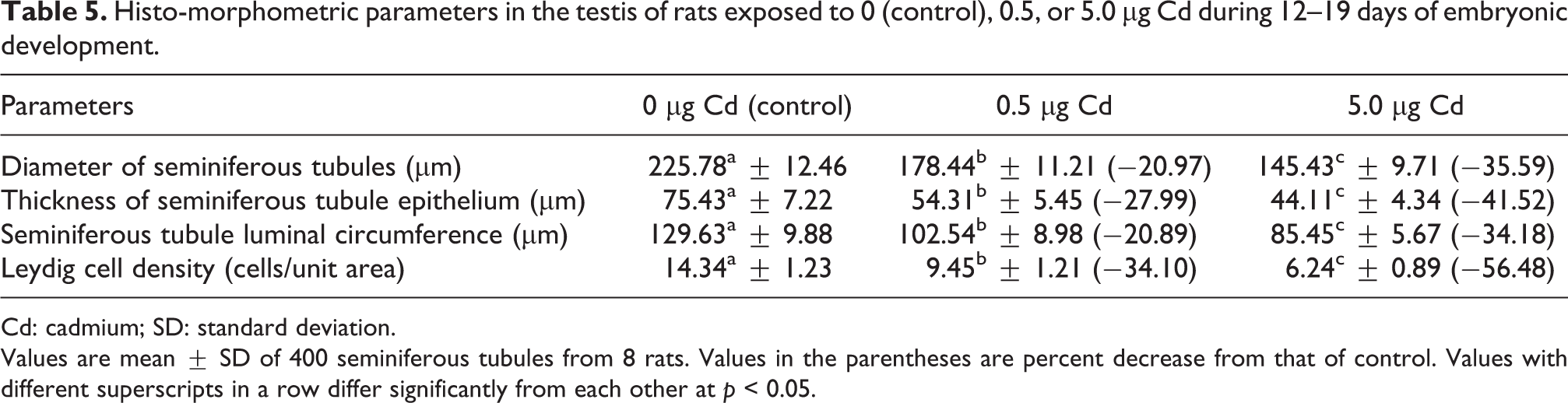
Histological examination of the testes of control rats revealed normal spermatogenesis along with thick stratified germinal epithelium containing proliferating germ cells and narrow lumen filled with spermatozoa (Figure 1(a)). The seminiferous tubules appeared to be uniform in size and shape; between the tubules, Leydig cells were found in the interstitial tissue. Conversely, significant reduction in the size of tubules with enlarged intertubular spaces and reduced number of sperms in the tubule was observed in the testes of rats exposed to Cd during embryonic development (Figure 1(b) and (c)), though the changes were not uniform as both affected and normal seminiferous tubules were observed in same section. The diameter of seminiferous tubules was significantly (p < 0.01) smaller in testes of rats exposed to Cd during embryonic development compared to the control rats (Table 5). Furthermore, marked reductions were noted in the Leydig cell density, height of germinal epithelium, and luminal circumference in the seminiferous tubules of testis of rats exposed to 0.5 or 5.0 µg Cd during embryonic development compared with the control rats (Table 5).
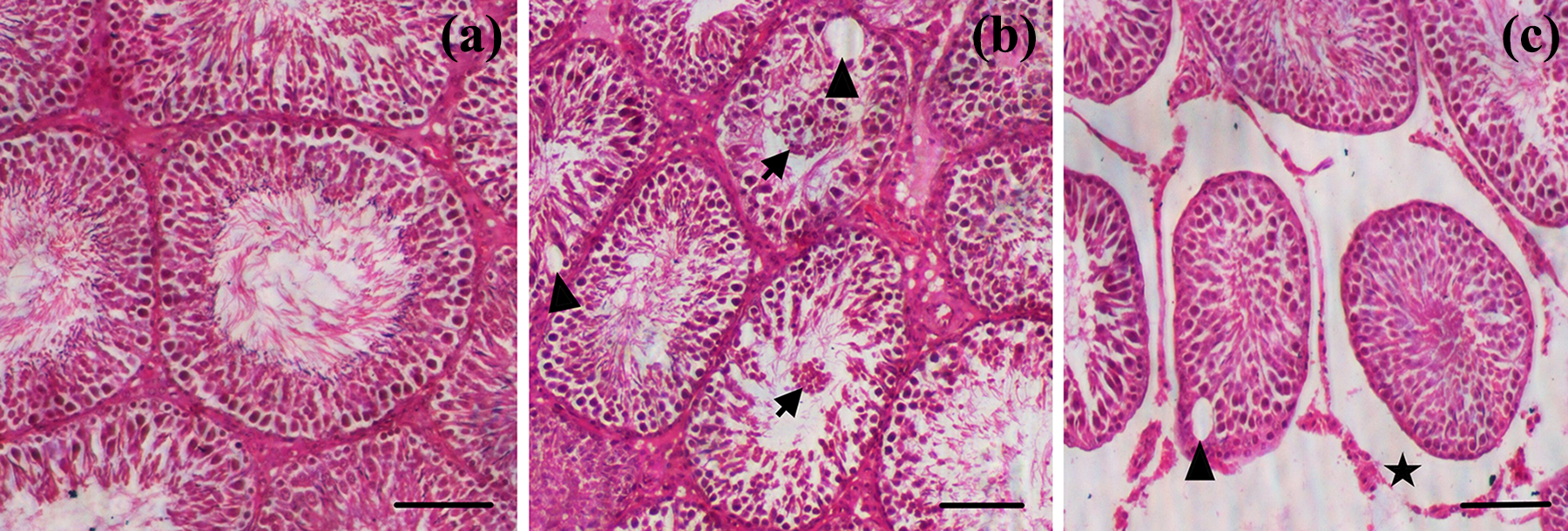
Hematoxylin and eosin-stained sections of rat testis: (a) testis of control rat showing normal appearance of the seminiferous tubules and (b and c) rats exposed to 0.05 or 5.0 µg Cd during embryonic development, respectively, showing the hypertrophied epithelium projecting into the lumen (arrows), presence of intra-epithelial vacuoles (arrow heads), and large intertubular spaces (*). Scale bar = 50 µm. Cd: cadmium.
Histo-morphometric parameters in the testis of rats exposed to 0 (control), 0.5, or 5.0 µg Cd during 12–19 days of embryonic development.
Cd: cadmium; SD: standard deviation.
Values are mean ± SD of 400 seminiferous tubules from 8 rats. Values in the parentheses are percent decrease from that of control. Values with different superscripts in a row differ significantly from each other at p < 0.05.
Fertility studies of male progeny
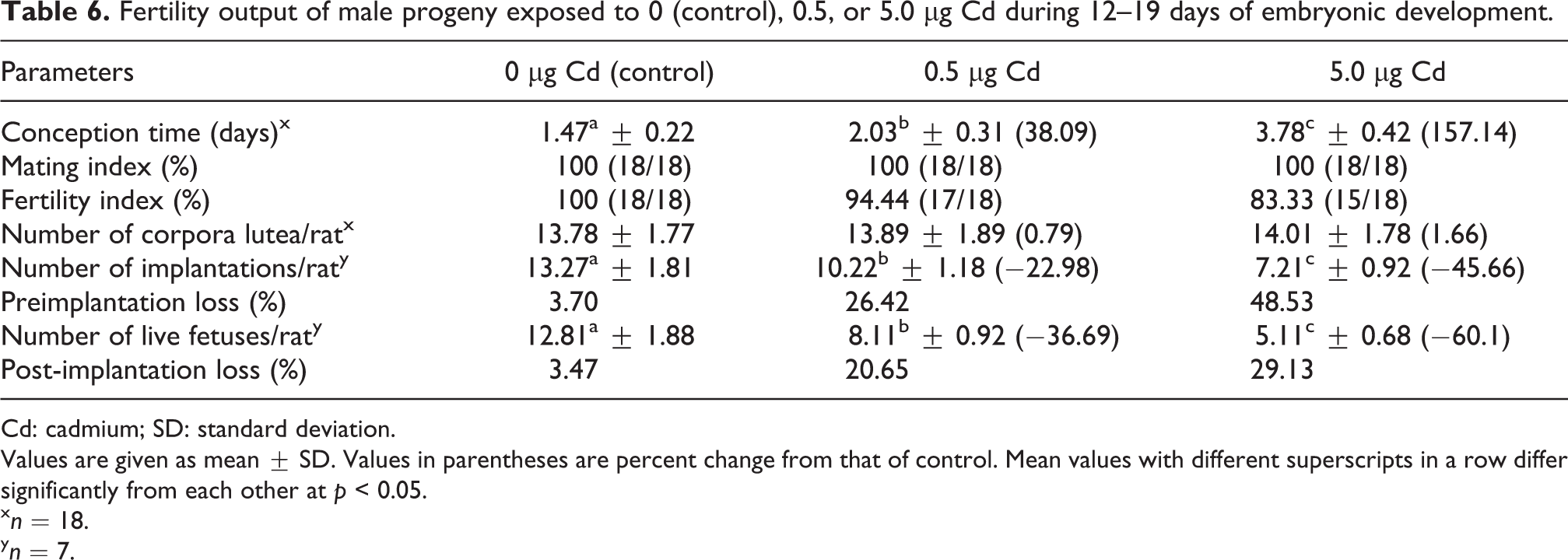
Although all females mated with males in the control, 0.5, or 5.0 µg Cd-exposed groups showed vaginal plugs (mating index 100%), the fertility index was decreased in the 5.0 µg Cd-exposed group (83.33%; Table 6). Significant delay in conception time was observed in females mated with males exposed to 0.5 or 5.0 µg Cd during embryonic development when compared with controls. The mean number of corpora lutea was comparable among all the groups. The mean number of implantations and live fetuses per dam was decreased with an increase in pre- and post-implantation losses in females mated with males exposed to 0.5 or 5.0 µg Cd during embryonic development (Table 6).
Fertility output of male progeny exposed to 0 (control), 0.5, or 5.0 µg Cd during 12–19 days of embryonic development.
Cd: cadmium; SD: standard deviation.
Values are given as mean ± SD. Values in parentheses are percent change from that of control. Mean values with different superscripts in a row differ significantly from each other at p < 0.05.
x n = 18.
y n = 7.
Tissue metabolic enzymes of male progeny
No significant changes in the activity levels of SDH, lactate dehydrogenase, glucose-6-phosphate dehydrogenase, glutamate dehydrogenase, alanine aminotransferase, and aspartate aminotransferase were observed in the liver, kidney, and testis of rats exposed to 0.5 or 5.0 µg Cd during embryonic development when compared with the controls (Figures 2 to 4).
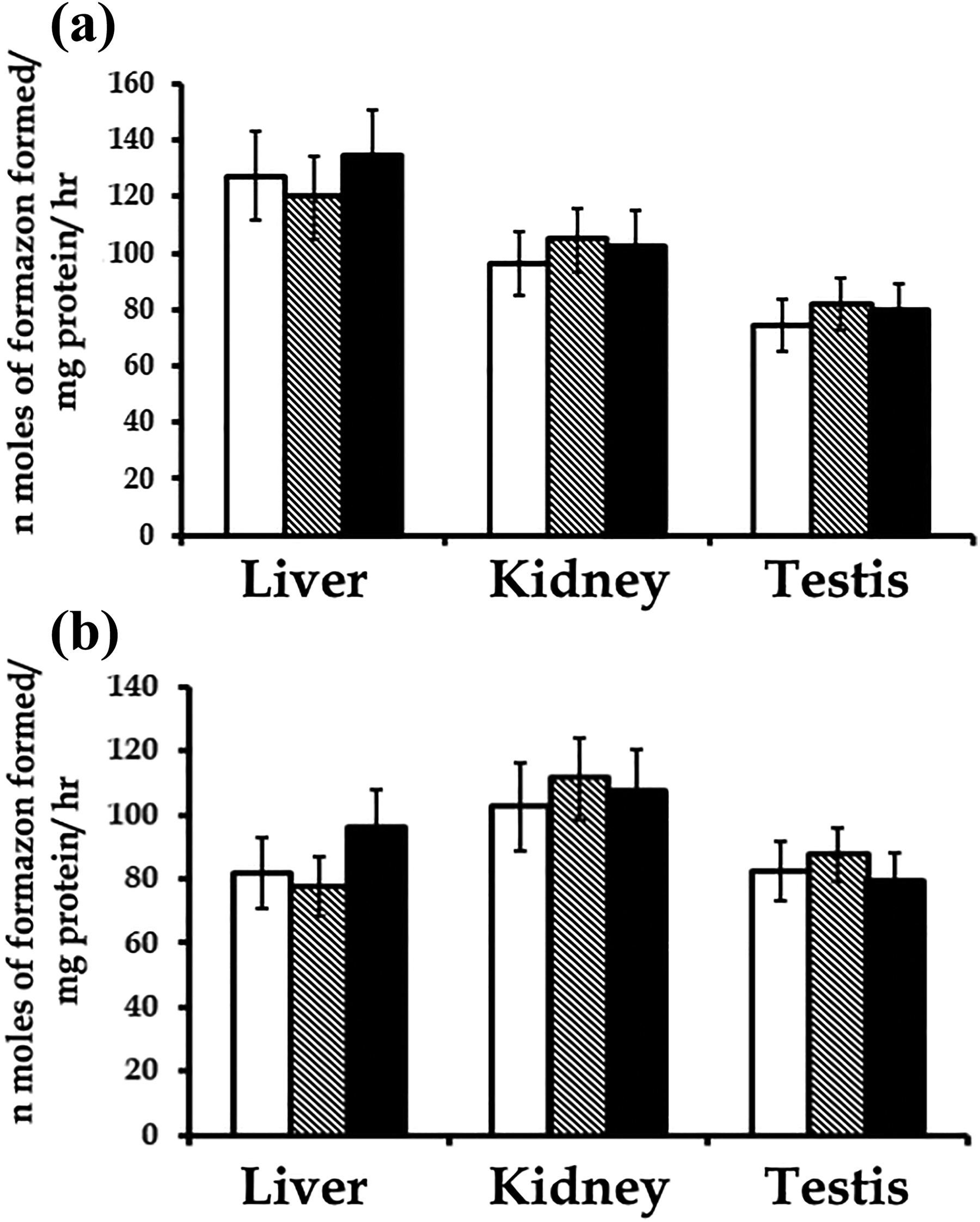
Activity levels of: (a) succinate dehydrogenase and (b) lactate dehydrogenase in the liver, kidney, and testis of rats exposed to 0 (control) (open bars), 0.5 (striped bars), and 5.0 µg Cd (solid bars) during embryonic development. Bars are represented as mean ± SD of 8 individuals. Mean values between groups are comparable. Cd: cadmium; SD: standard deviation.
Activity levels of: (a) glucose-6-phosphate dehydrogenase and (b) glutamate dehydrogenase in the liver, kidney, and testis of rats exposed to 0 (control) (open bars), 0.5 (striped bars), and 5.0 µg Cd (solid bars) during embryonic development. Bars are represented as mean ± SD of 8 individuals. Mean values between groups are comparable. Cd: cadmium; SD: standard deviation.
Activity levels of (a) aspartate amino transaminase and (b) alanine amino transaminase in the liver, kidney, and testis of rats exposed to 0 (control) (open bars), 0.5 (striped bars), and 5.0 µg Cd (solid bars) during embryonic development. Bars are represented as mean ± SD of 8 individuals. Mean values between groups are comparable. Cd: cadmium; SD: standard deviation.
Discussion
Adverse maternal uterine environment, including exposure to chemical/physical stresses during pregnancy, can “program” persistent changes in several physiological systems and behaviors, probably via epigenetic mechanisms. In the present study, pregnant rats were administered with Cd at doses below the NOEL in order to evaluate whether low doses of Cd induce any developmental, behavioral, metabolic, and reproductive anomalies. The NOEL for Wistar rat was reported as 2.6 mg/kg body weight/day. 34 In this study, pregnant rats were injected intraperitoneally with 0 (control), 0.5, or 5.0 µg Cd/kg body weight during the critical period of reproductive organs development (days 12–19 of pregnancy) and dams and male progeny were evaluated. Maternal observations revealed that selected doses of Cd did not cause any adverse effects to dams, as evidenced by their behavior, body weight gain, pregnancy duration, and their fecundity suggesting that the treatment had no effect on the fertility output of dams. 19 The litter size, birth weight, and survival rate of pups, developmental landmarks (including descent of testis into scrotal sac) are comparable between control and in utero Cd-exposed rats indicating no general toxicity to dams and progeny. Furthermore, developmental and behavioral parameters of pups exposed to Cd during embryonic development were comparable with the controls indicating the absence of overt toxicity. Conversely, meta-analysis of data revealed that exposure to higher levels of Cd during the prenatal period is associated with impaired cognitive development in children. 35
The present data reveals that the weight of liver, brain, kidney, testis, and epididymis in adult rats exposed to either 0.5 or 5.0 µg Cd during embryonic development was also comparable with the controls, suggesting that the general metabolism was also unaltered. Furthermore, the activity levels of lactate dehydrogenase, an enzyme localized at a strategic point between glycolysis and Krebs’ cycle, and SDH and glucose-6-phosphate dehydrogenase representing Kreb’s cycle and HMP shunt pathway, respectively, in liver, testis, and kidney of the control and experimental rats were also comparable. Besides, the activity levels of glutamate dehydrogenase, which catalyzes the oxidative deamination of glutamate, providing α-ketoglutarate to citric acid cycle and alanine- and aspartate transaminases function at the metabolic junction between carbohydrate and protein metabolism by interconverting the strategic compounds such as α-ketoglutarate, pyruvate, oxaloacetate, alanine, and aspartate and often used as the biochemical index of stress in liver, kidney, and testis were also not significantly changed, indicating that the metabolic activity of tissues is in order. Similarly, no evidence of Cd toxicity in the liver or kidney and no effect on the body weight were observed in rats exposed to Cd in utero. 11
Conversely, in utero exposure to Cd caused a reduction in DSP and epididymal sperm numbers indicating decreased spermatogenesis. The treatment also had an adverse effect on the number of motile, viable, and HOS-tail swelled sperm in the cauda epididymis. Impaired sperm motility may result in infertility due to the failure of sperm to reach the site of fertilization as well as their ability to penetrate zona pellucida. Furthermore, nonuniform degenerative changes were noticed in the seminiferous tubules in the testis of in utero Cd-exposed rats with both affected and normal tubules in some sections; the affected tubules showed diverse degenerative changes such as loosening of germinal epithelium, mixing of spermatids of different stages of spermatogenesis, and occurrence of multinucleated giant cells. Marked reductions in the diameter of the seminiferous tubules and in the height of the germinal epithelium in testes of in utero Cd-exposed rats suggest an adverse effect on spermatogenesis. 36 It is well documented that spermatogenesis is a testosterone-dependent process. 37 The observed impairment of spermatogenesis may reflect reduced bioavailability or production of androgen in the rats exposed to Cd during embryonic development. The data also reveal a decrease in the activity levels of 3β-HSD and 17β-HSD in the testis of experimental rats, associated with significantly reduced serum testosterone levels, indicating a possible inhibition of androgen synthesis. Thus, it is reasonable to assume that the suppression of spermatogenesis in rats exposed to Cd in utero in this study is caused due to deficiency of testosterone.
The final reproductive end point that was tested was the ability of male exposed to Cd in utero to sire offspring. All the females that were cohabited with males from the control and exposed to 0.5 or 5.0 µg Cd in utero had copulatory plugs, suggesting that libido was not affected in rats after exposure to 0.5 or 5.0 µg Cd during embryonic development, though there was a delay in conception time. Conversely, additional observations of fewer implantations and higher implantation loss in females mated with males exposed to Cd in utero suggest compromised sperm fertility. Probably the observed decline plasma testosterone level in rats exposed to Cd during embryonic development played a role in decreasing the density and deteriorating quality of sperm, resulting in suppressed reproduction.
Previous studies have shown that Cd has potent estrogen-like activity in vivo and functionally acts like steroidal estrogen as a result of its ability to form a high-affinity complex with the hormone-binding domain of the estrogen receptor. 11 In utero exposure of Sprague-Dawley rats to Cd mimicked the effects of estrogen by inducing early onset of puberty and an increase in the epithelial area and the number of terminal end buds in the mammary gland of female offspring. 11 Although estrogen and estrogen receptors are essential for normal male fertility, exposures to exogenous estrogen mimics during the perinatal period induce structural and functional changes in the male reproductive tract and inhibit proliferation of precursor Leydig cells during the neonatal period, thus altering the steroidogenic capacity of adult testis. 38 Similar to our results, male rats treated neonatally/perinatally with estrogens or estrogen mimics exhibited decreased androgen production, compromised sperm fertility and reduced fecundity. Estrogen treatment during the perinatal period induces infertility in male Sprague-Dawley rats, which is long-term, probably permanent, and cannot be reversed by testosterone substitution in adult rats. 39,40 Our previous studies have shown that administration of progesterone during pregnancy suppressed the reproduction in male progeny of mice at adulthood. 41 Wistar rats treated neonatally with different doses of diethylstilbestrol showed a decrease in testis weight and germ cell volume and an increase in apoptotic index of germ cells at adulthood, implying a long-lasting effect of neonatal exposure to estrogens. 42 –44
In conclusion, this study provides compelling evidence of altered reproductive functions, including reduced steroidogenesis and spermatogenesis and fertility output in adult Wistar rats that were exposed to Cd in utero without eliciting detectable anomalies in development, behavior, and general metabolism. These findings will be significant because pregnant women are indiscriminately exposed to a gamut of xenoestrogens including heavy metals, and such an adverse uterine environment may be more prone to higher incidence of reproductive disorders in the next generation, though the individuals seem to be apparently normal.
Footnotes
Authors’ note
This work is a part of the PhD thesis of KS at The Department of Zoology, Sri Venkateswara University, Tirupati, India.
Author contributions
PSR was involved in the conceptualization, data analysis, methodology, and original draft preparation; KS was in charge of project administration, performed the experiments, data analysis, and validation; and BPG was involved in data analysis and validation, technical assistance, and editing. The manuscript was drafted by PSR, BPG, and KS and was approved by all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
