Abstract
Toxicological effects from chemical interaction may result in weaker or stronger effects. The present study investigated the influence of acute oral co-exposure to dichloromethane (DCM) and ethanol (EtOH) in rats. Four groups of rats were treated for seven consecutive days with corn oil, DCM at 150 mg/kg alone, EtOH at 5 g/kg alone, and both DCM and EtOH, respectively. Subsequently, biomarkers of hepatic and renal functions, cellular antioxidant defense systems, and oxidative stress indices were analyzed in the liver and kidney samples. Results indicated that the significant (p < 0.05) elevations in the biomarkers of hepatic and renal toxicity following exposure of rats to DCM alone and EtOH alone were aggravated in the co-exposure group. Further, the significant reductions in the antioxidant status and the increase in lipid peroxidation in the liver and kidney of rats following exposure to DCM alone and EtOH alone were aggravated in the co-exposure group. Histological alterations of rats treated with DCM alone and EtOH alone were worsened in the co-exposure group. In summary, co-exposure to DCM and EtOH elicited more harmful effects on the liver and kidney than the individual chemical exposure, which is attributable to the intensified oxidative stress in the treated rats.
Introduction
Dichloromethane (DCM) is an organic solvent widely used in industries as an extraction medium, degreaser, lacquer remover, paint stripper, flammability depressant, and aerosols of insecticides. 1,2 The extensive use of DCM has been related to the widespread discharge of DCM and its derivatives into the environment, with potential adverse effects on ecosystems and humans. 3,4 Indeed, DCM is a priority pollutant by the US Environmental Protection Agency owing to its toxicity, persistence, and accumulation in the food chain. 5 Previous studies on DCM revealed that DCM is metabolized mainly in the liver. Specifically, DCM undergoes two main metabolic pathways, namely the microsomal oxidation and cytosolic pathway. DCM is converted to carbon monoxide and carbon dioxide via oxidative dechlorination reaction in the microsomes, whereas the cytosolic pathway involves the reaction with glutathione (GSH) leading to the production of carbon dioxide but not carbon monoxide. 6,7
Occupational exposure to DCM is frequent and reportedly causes both acute and chronic toxicity. 6,8,9 Previous studies have classified DCM as carcinogenic in humans due to carcinogenicity observed in liver and lung of B6C3F1 mice. 10 –12 Also, epidemiologic studies on DCM provided additional data, which have raised concerns due to its associations with non-Hodgkin lymphoma and multiple myeloma. 13,14 Although the hepatotoxicity of DCM has been reported, the exact biochemical mechanisms by which it elicits its toxic effects on the liver remains poorly understood. Besides, there is paucity of information on its adverse effects on the kidney.
Moreover, consumption of alcohol among adolescent and young adult in the population continues to be a major public health concern globally. 15,16 Unnecessary alcohol intake is common during adolescence as a result of exuberance, ease of access, and lack of legal consequences. African countries have been ranked among the highest consumers of alcohol per capita. 17,18 Previous studies have demonstrated that alcohol consumption elicits oxidative damage in several organs including liver, kidney, brain, and testes with associated morphological and functional changes. 19 –21 Individual occupationally exposed to DCM 22,23 from varied household, and personal care products containing DCM 23,24 may indulge in binge or social ethanol (EtOH) consumption, which may result in toxic chemical mixture interaction systemically. Although co-exposure to DCM and alcohol is conceivable, 6 there is no detail information in the literature on the influence of their co-exposure on hepatorenal indices, albeit a growing public health concern on the need to identify and to characterize the risk associated with exposure to chemical mixtures. 25 Toxicological effects resulting from chemical–chemical interaction may cause a synergistic or antagonistic/competitive effect. 26 An earlier study demonstrated that EtOH pretreatment enhances DCM-mediated carboxyhemoglobin formation in treated rats. 27 Consequently, considering the role of cellular antioxidant defense mechanisms of the liver and kidney, there is a need to elucidate the impact of co-exposure to DCM and EtOH.
The present study aimed at investigating the nature of toxicity following co-exposure to DCM and EtOH in pubertal male Wistar rats by assessing the biomarkers of hepatic and renal toxicity, antioxidant enzyme activities, and histopathological lesions in the liver and kidney of the treated rats.
Materials and methods
Chemicals
EtOH (190% proof), DCM, epinephrine, hydrogen peroxide (H2O2), thiobarbituric acid (TBA), 1-chloro-2,4-dinitrobenzene (CDNB), GSH, and 5′, 5′-dithiobis-2-nitrobenzoic acid (DTNB) were purchased from Sigma Chemical Co. (St Louis, Missouri, USA). All other chemicals were of analytical grade and were purchased from the British Drug Houses (Poole, Dorset, UK).
Animal care
Forty pubertal male Wistar rats (6 weeks old, 132 ± 5 g) obtained from the Experimental Animal Unit, Faculty of Veterinary Medicine, University of Ibadan, Nigeria, were used for this investigation. The animals were housed in plastic cages situated in a well-ventilated rat house, provided rat chow and water ad libitum, and subjected to natural photoperiod of 12-h light/12-h dark. All the animals adequately cared for following the conditions stated in the “Guide for the Care and Use of Laboratory Animals” prepared by the National Academy of Science (NAS) and published by the National Institute of Health. Also, the study was executed in accordance with the US NAS guidelines and authorization by the University of Ibadan Ethical Committee.
Experimental protocol
Following 1 week of acclimatization, the animals were randomly assigned to four groups of 10 rats each and were treated by gavage 7 consecutive days as follows: Group 1: Control rats orally treated with 2 ml/kg of corn oil. Group 2: Rats orally treated with 150 mg/kg DCM dissolved in corn oil. Group 3: Rats orally treated with 5 g/kg EtOH (190% proof). Group 4: Rats orally treated with the mixture of DCM (150 mg/kg) and EtOH (5 g/kg).
The doses of DCM (150 mg/kg) and EtOH (5 g/kg) established in the present investigation were chosen based on the results from a pilot study in our laboratory and previously published data. 15
Twenty-four hours after the last treatment, the rats were weighed and the blood was collected from retro-orbital venous plexus into plain tubes, before they were sacrificed by cervical dislocation. Subsequently, the serum samples were obtained by centrifugation of the clotted blood at 3000 g for 10 min. The serum samples were stored frozen at −20°C until use for the analysis of liver and kidney function indices.
Evaluation of liver and kidney function biomarkers
Analysis of serum activities of aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma glutamyl transferase (GGT), and alkaline phosphatase (ALP) as well as urea and creatinine levels was done using available commercial kits from Randox Laboratories Limited (UK).
Evaluation of liver and kidney antioxidant status
The liver and kidney samples of the rats were homogenized in 50 mM Tris–HCl buffer (pH 7.4) containing 1.15% potassium chloride. The homogenates were thereafter centrifuged at 12,000 g for 15 min at 4 °C to obtain the supernatant which was used for biochemical estimations. Hepatic and renal protein concentration was assayed according to the method described by Bradford. 28 Superoxide dismutase (SOD) activity was assayed according to the method described by Misra and Fridovich, 29 whereas catalase (CAT) activity was assayed using hydrogen peroxide as a substrate according to the method described by Clairborne. 30 GSH level was assayed according to established method. 31 Glutathione peroxidase (GPx) activity was assayed according to Aber et al. 32 while glutathione-S-transferase (GST) activity was assayed according to the method of Lowe. 33 Lipid peroxidation (LPO) was quantified as malondialdehyde (MDA) according to the established method. 14,34
Microscopic examinations of liver and kidney
Liver and kidney samples from the rats in each group were processed for histology according to standardized procedure. 35 Succinctly, the samples were fixed using 10% phosphate buffered formalin (PBF) for 3 days. The samples were embedded in a paraffin after dehydration procedures. Subsequently, sections of 4–5 µm were cut using a microtome and stained with hematoxylin and eosin (H&E). All the slides were coded before examination under a light microscope (Leica DM 500, Germany) and photomicrographs taken using a digital camera (Leica ICC50 E, Germany) by a pathologist.
Statistical analysis
One-way analysis of variance (ANOVA) to compare the experimental groups followed by Bonferroni’s post hoc to identify significantly different groups was done using the GraphPad Prism 5 software (Version 4; GraphPad Software, La Jolla, California, USA). The value of p < 0.05 was considered statistically significant.
Compliance with ethical standards
The study was conducted after approval by the University of Ibadan Ethical Committee on the use of Laboratory animal for experimental purposes.
Results
Effects of administration of DCM and EtOH, singly and in combination, on body weight gain and relative organ weights of rats
The body weight gain and the relative liver and kidney weights of rats orally treated with DCM and EtOH singly or in combination for seven consecutive days are presented in Table 1. Alone, DCM or EtOH did not significantly (p > 0.05) affect the body weight gain and the relative liver and kidney weights, whereas rats co-treated with DCM and EtOH showed significant (p < 0.05) reduction in the body weight gain with concomitant significant increase in the relative organ weights compared with the control.
Body weight gain and relative organ weights of rats after treatment with DCM alone, EtOH alone, and both DCM and EtOH for 7 consecutive days.
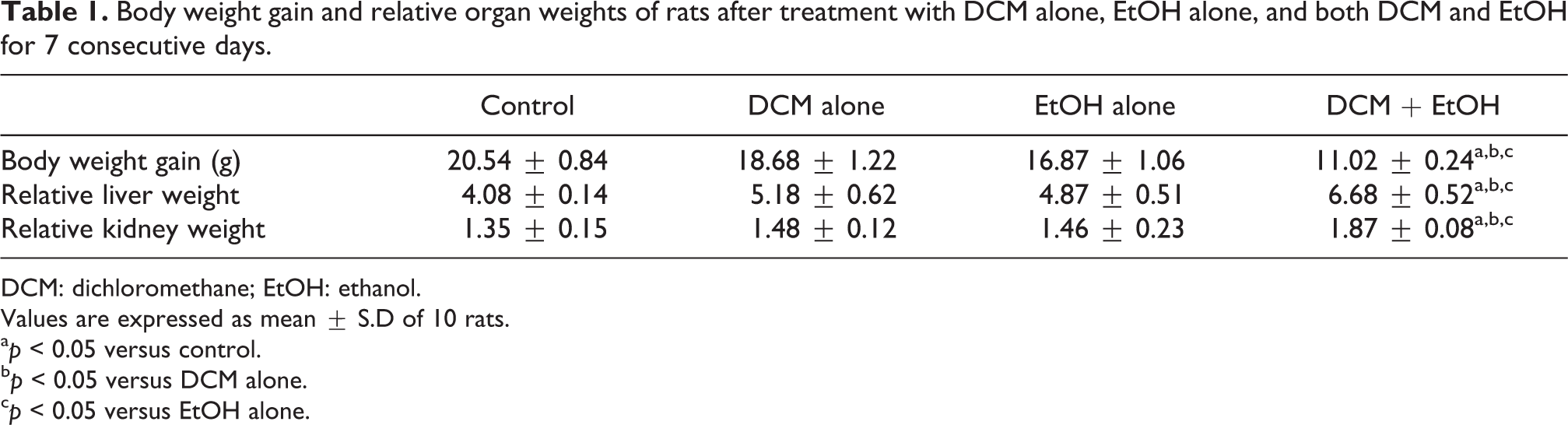
DCM: dichloromethane; EtOH: ethanol.
Values are expressed as mean ± S.D of 10 rats.
a p < 0.05 versus control.
b p < 0.05 versus DCM alone.
c p < 0.05 versus EtOH alone.
Biomarkers of hepatic and renal function in pubertal rats co-exposed to DCM and EtOH
To evaluate the impact of co-exposure to DCM and EtOH, the rats were orally administered DCM and EtOH for seven consecutive days after which the functional indices of the liver and kidney were monitored. Figure 1 shows the results of the hepatorenal indices in pubertal rats treated with DCM and EtOH singly or in combination. The serum activities of ALT, AST, ALP, and GGT were significantly (p < 0.05) increased in rats administered with DCM alone and EtOH alone. The marked elevations in these biomarkers of toxicity were more pronounced in rats co-treated with DCM and EtOH. Serum ALT activity was significantly raised by 50, 55, and 65%, whereas serum AST activity was significantly raised by 46, 54, and 63% in rats treated with DCM alone, EtOH alone and co-exposure group, respectively, when compared with the control. Also, serum ALP activity was evidently raised by 49, 56, and 65%, whereas serum GGT activity was markedly raised by 41, 31, and 54% in rats treated with DCM alone, EtOH alone, and co-exposure group, respectively, when compared with the control. Further, the renal function indices, namely urea and creatinine levels, were significantly elevated in rats administered with DCM alone and EtOH alone, whereas they were intensified in the rats co-treated with DCM and EtOH. Serum creatinine level was raised by 46, 30, and 63%, whereas urea level was raised by 56, 28, and 70% in rats treated with DCM alone, EtOH alone, and co-exposure group respectively, when compared with the control.
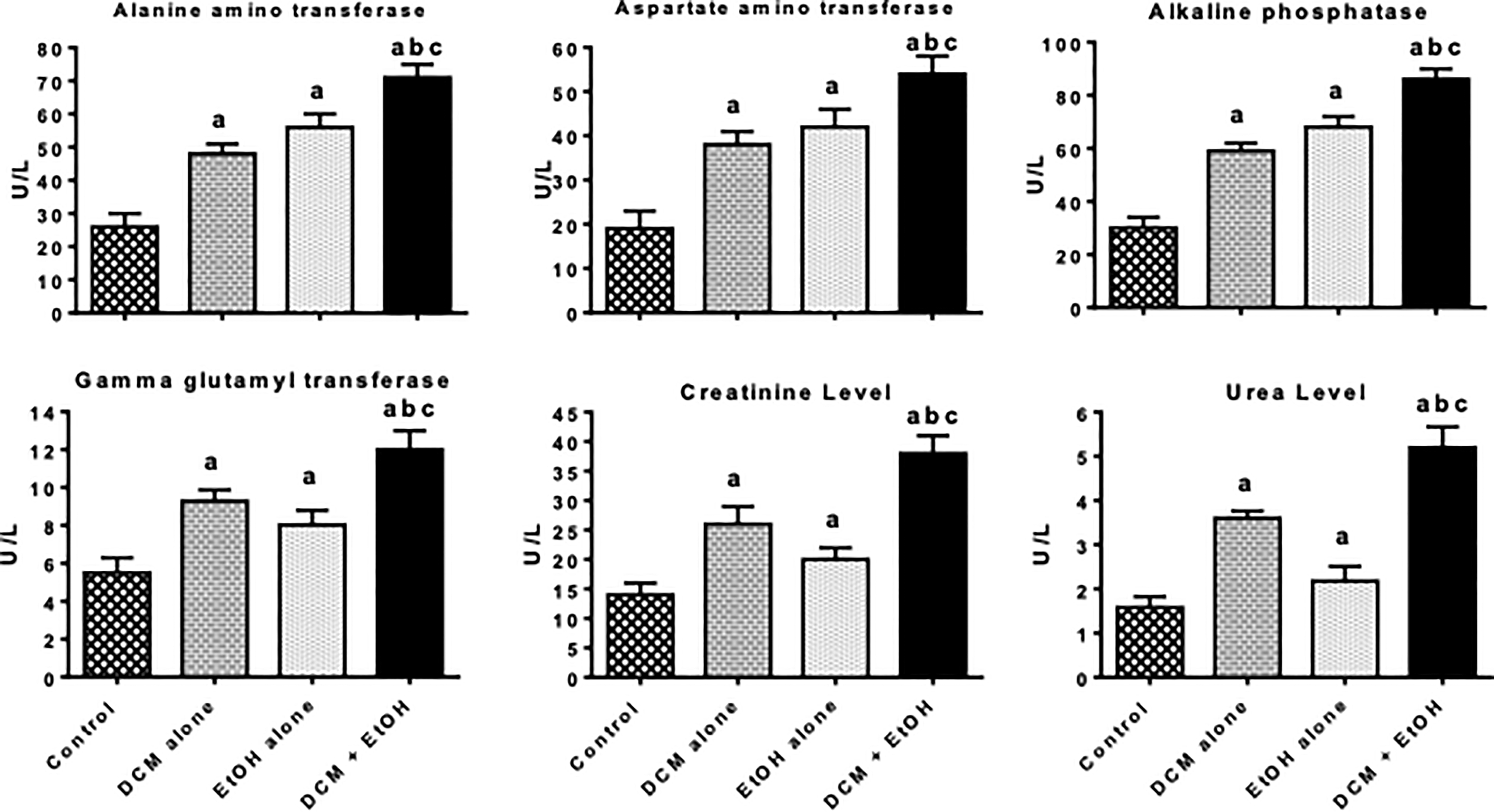
Hepatic and renal functional indices in pubertal rats following oral treatment with DCM alone, EtOH alone, and both DCM and EtOH for seven consecutive days. Each bar represents mean ± SD of 10 rats; a p < 0.05 versus control; b p < 0.05 versus DCM alone; c p <0.05 versus EtOH alone. DCM: dichloromethane; EtOH: ethanol; SD: standard deviation.
Antioxidant status of the liver and kidney of pubertal rats co-exposed to DCM and EtOH for seven consecutive days
Figures 2 to 4 depict the effects of treatment with DCM and EtOH, singly and in combination, on antioxidant defense system and oxidative stress indices in liver and kidney of rats. The administration of DCM alone and EtOH alone caused a significant decrease in the activities of antioxidative enzymes, such as SOD, CAT, GPx, and GST, as well as nonenzymatic antioxidant GSH level in the liver and kidney of the treated rats when compared with the control. The decrease in these antioxidant parameters was intensified in the co-exposure group when compared with DCM alone and EtOH alone. However, the administration of DCM alone and EtOH alone resulted in a significant elevation in the level of MDA, a biomarker of LPO, in the liver and kidney of the treated rats when compared with the control. In comparison with rats administered with DCM alone or EtOH alone, the marked increase in the MDA level was intensified in the rats co-administered with DCM and EtOH.
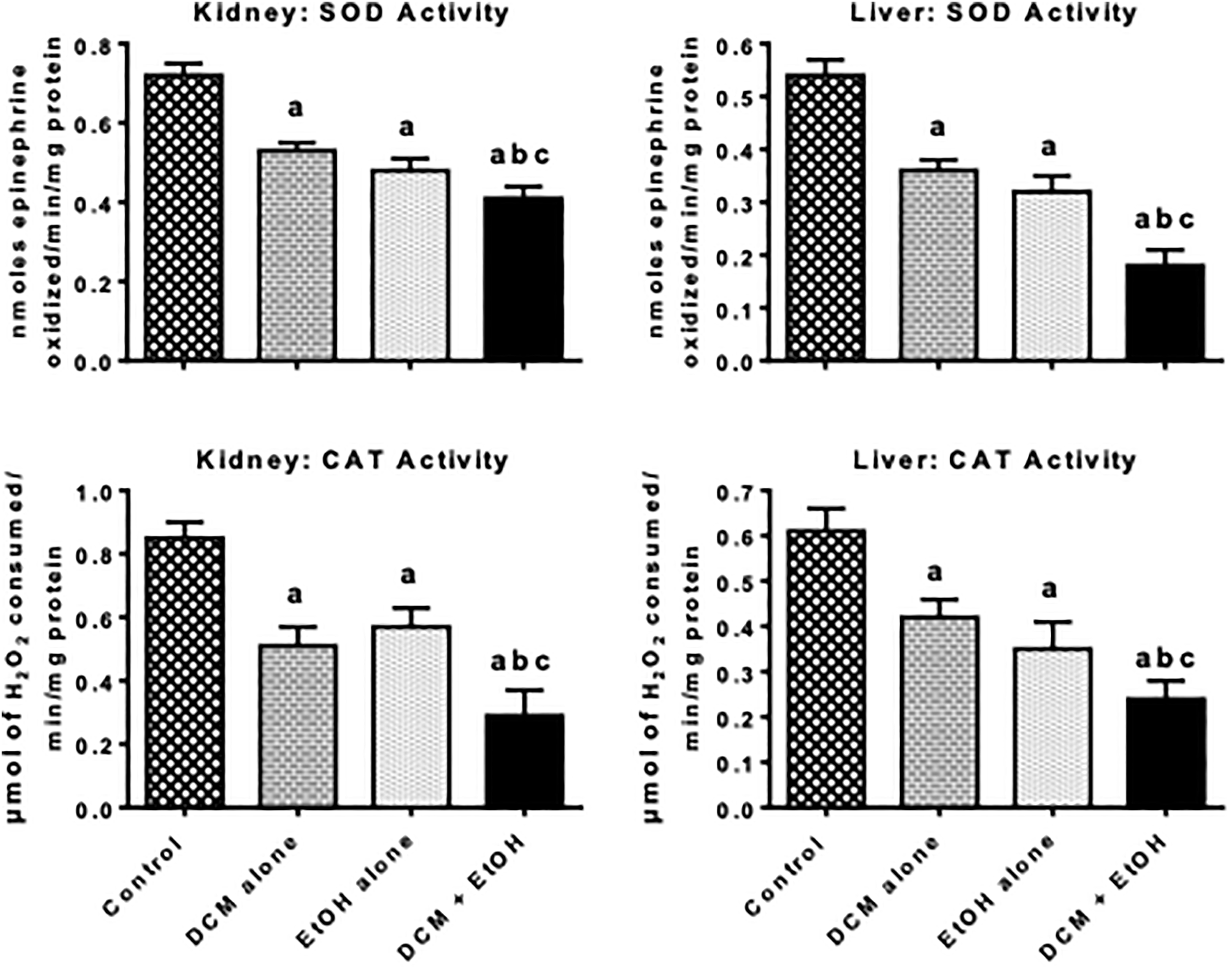
Activities of SOD and CAT in liver and kidney of pubertal rats following oral treatment with DCM alone, EtOH alone, and both DCM and EtOH for seven consecutive days. Each bar represents mean ± SD of 10 rats; a p < 0.05 versus control; b p < 0.05 versus DCM alone; c p < 0.05 versus EtOH alone. DCM: dichloromethane; EtOH: ethanol; SD: standard deviation; CAT: catalase; SOD: superoxide dismutase.
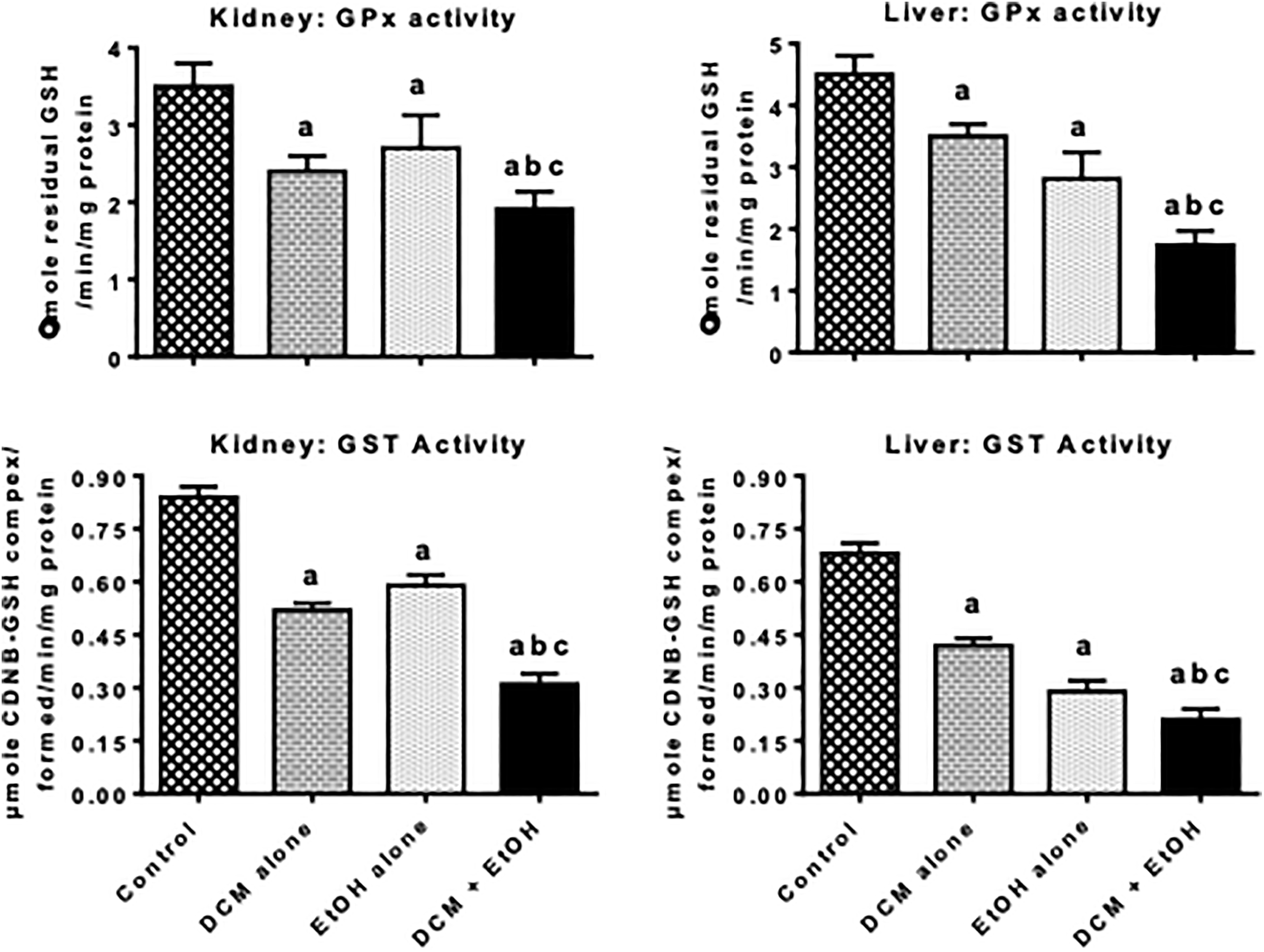
Activities of GPx and GST in liver and kidney of pubertal rats following oral treatment with DCM alone, EtOH alone, and both DCM and EtOH for seven consecutive days. Each bar represents mean ± SD of 10 rats; a p < 0.05 versus control; b p < 0.05 versus DCM alone; c p < 0.05 versus EtOH alone. GPx: glutathione peroxidase; GST: glutathione-S-transferase; DCM: dichloromethane; EtOH: ethanol; SD: standard deviation.
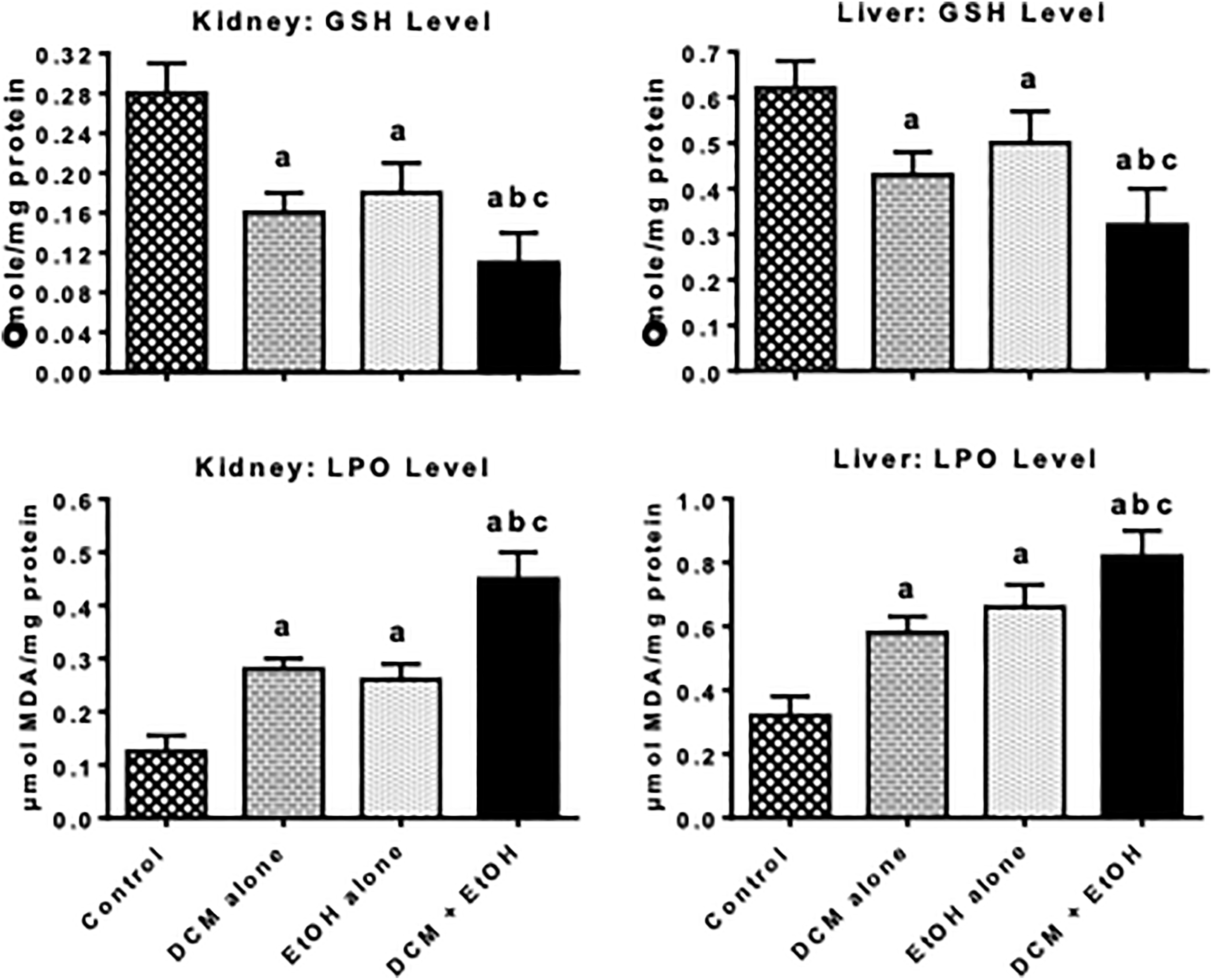
Activity of GSH level and lipid peroxidation (LPO) in liver and kidney of pubertal rats following oral treatment with DCM alone, EtOH alone and both DCM and EtOH for 7 consecutive days. Each bar represents mean ± SD of 10 rats; a p < 0.05 versus control; b p < 0.05 versus DCM alone; c p < 0.05 versus EtOH alone. GSH: glutathione; DCM: dichloromethane; EtOH: ethanol; SD: standard deviation.
Histopathological lesions in liver and kidney of rats co-treated with DCM and EtOH
The representative photomicrographs of the liver and kidney from the experimental rats are presented in Figures 5 and 6. The results from the microscopic examination of the liver and kidney indicated that the architectures of liver and kidney of control rats were structurally and functionally normal (Table 2). However, kidneys of rats treated with DCM alone revealed mild disseminated congestion (green arrow), whereas EtOH alone-treated rats showed focal area of congestion (red arrow). However, the kidney of rats co-treated with DCM and EtOH showed multifocal area of marked inflammation (black arrow) as well as tubular necrosis (yellow arrow). Moreover, the liver of rats treated with DCM alone showed marked disseminated periportal infiltration by inflammatory cells (black arrow) and mild disseminated congestion (yellow arrow), whereas EtOH alone-treated rats showed mild focal congestion (green arrow) and periportal infiltration by inflammatory cells (black arrow). However, the liver of rats co-administered with DCM and EtOH showed marked disseminated congestion (green arrow), focal area of portal fibrosis (blue arrow), and focal area of periportal infiltration by inflammatory cells (black arrow).
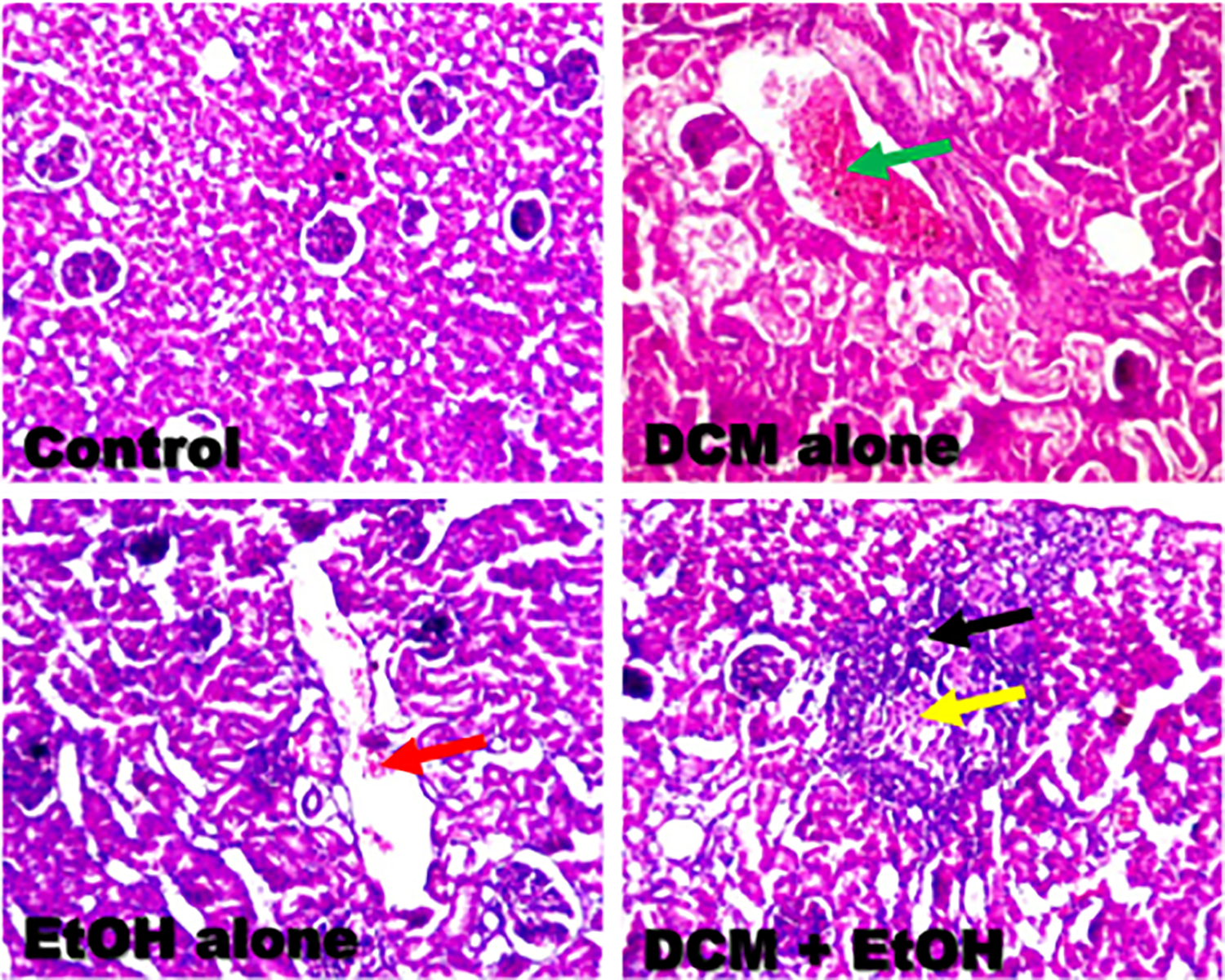
Representative photomicrographs of kidney from control, DCM alone, EtOH alone, and co-exposure groups. Kidney of control rats were structurally and functionally normal. Kidney of rats treated with DCM alone showing mild disseminated congestion (green arrow), whereas EtOH alone-treated rats showing focal area of congestion (red arrow). Kidney of rats co-exposed to DCM and EtOH showing multifocal area of marked inflammation (black arrow) as well as tubular necrosis (yellow arrow). Original magnification: ×200. DCM: dichloromethane; EtOH: ethanol.
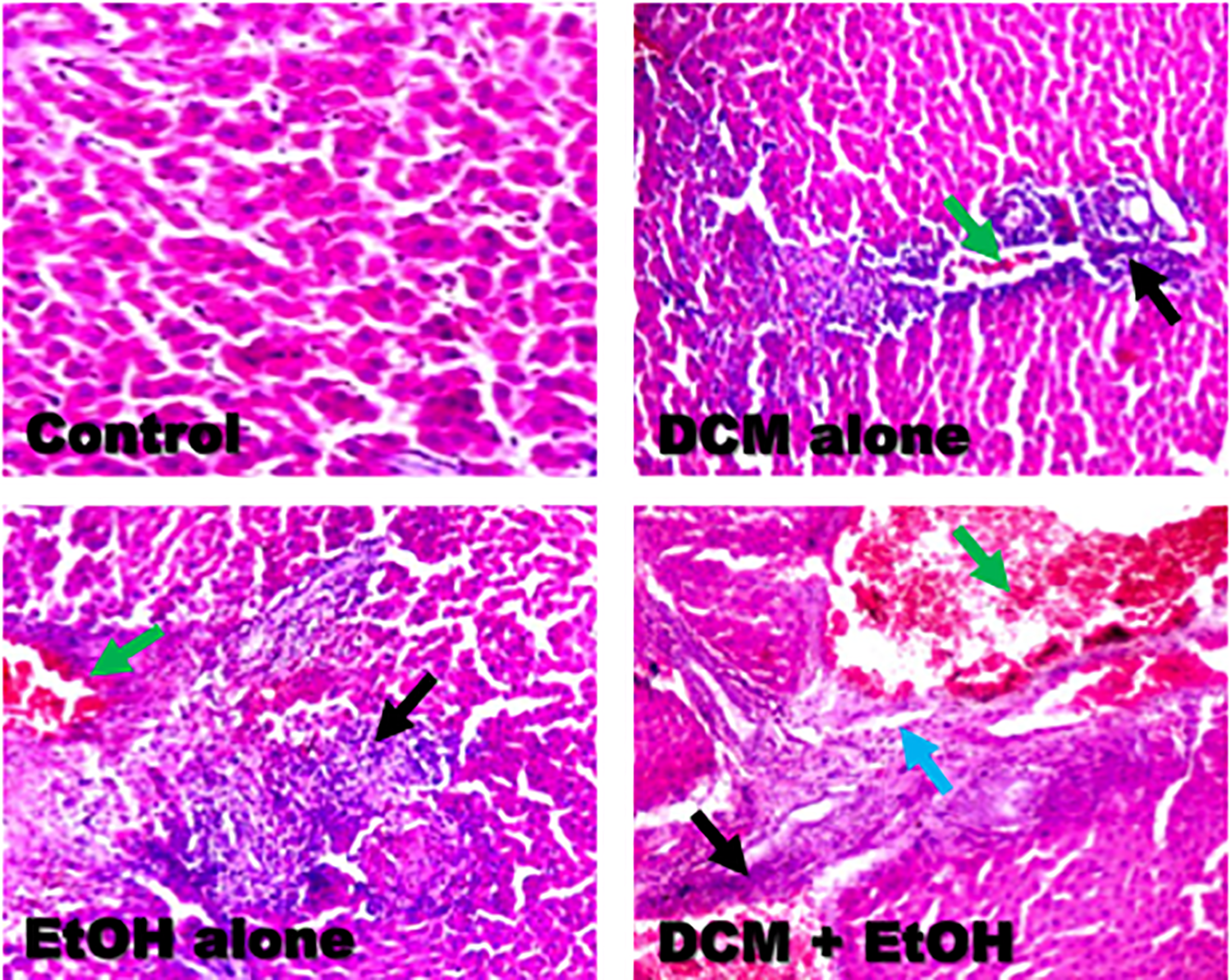
Representative photomicrographs of liver from control, DCM alone, EtOH alone, and co-exposure groups. Liver of control rats showing normal morphology. Liver of rats treated with DCM alone showing marked disseminated periportal infiltration by inflammatory cells (black arrow) and mild disseminated congestion (yellow arrow). EtOH alone-treated rats showing mild focal congestion (green arrow) and periportal infiltration by inflammatory cells (black arrow). Liver of rats co-exposed to DCM and EtOH showing marked disseminated congestion (green arrow), focal area of portal fibrosis (blue arrow) and focal area of periportal infiltration by inflammatory cells (black arrow). Original magnification: ×200. DCM: dichloromethane; EtOH: ethanol.
Incidence of lesion in liver and kidney of rats treated with DCH and EtOH.

DCM: dichloromethane; EtOH: ethanol; L: liver; K: kidney.
Discussion
Several previous investigations have demonstrated the need to characterize the role of chemical mixtures in living organisms to assist in establishing a focused technique in evaluating the risk associated with human populations exposed to them. 36 Environmental contaminants may interact in numerous ways resulting in antagonistic, synergistic, or additive effects. 37 The co-exposure to DCM and EtOH is possible. Hence, investigation into the potential effects of their co-exposure is of great significance to human risk assessment. The present investigation intended to characterize the effects of co-exposure to DCM and EtOH in pubertal rats. The relative organ weight represents a good indicator of atrophy. 38 A decrease in the relative organ weight might be due to atrophy and an increase may be considered due to inflammation or metabolic adaptation. 25 The present investigation revealed that the body weight gain and relative organ weights were not significantly affected in rats treated with either DCM alone or EtOH alone. However, there were apparent adverse effects in the rats co-exposed to DCM and EtOH as evidenced in the marked reduction in the body weight gain with concomitant significant increase in the relative liver and kidney weights. The reduction in weight gain signifies abnormality in the general metabolic condition of the animals, whereas an increase in relative organ weight related to inflammation in the co-exposed rats.
Aspartate and alanine aminotransferases are localized in the peripotal hepatocytes and are reliable marker enzymes of early acute hepatic damage because their blood activities markedly increase following loss of hepatocyte structural integrity and leakage. 39 Further, the assessment of GGT, which is localized in the plasma membrane, is recognized as the most sensitive enzymatic indicator of hepatobiliary diseases and by extension changes in renal function. Similarly, an assessment of ALP, an ectoenzyme in the plasma membrane of the bile duct, 39 reveals the condition of hepatobiliary system and the movement of bile into the small intestine. The intensification in the serum activities of aminotransferases (AST and ALT) observed in rats co-exposed to DCM and EtOH reflects the worsening of their individual toxic effects on hepatic structure and function. Moreover, the intensification in the serum activities of membrane bound enzymes (GGT and ALP) in rats co-exposed to DCM and EtOH signifies an additive effect and a compromise in the membrane structural integrity in the treated rats. In addition, the present investigation revealed that co-exposure to DCM and EtOH caused a significant increase in the renal functional indices specifically, urea and creatinine when compared to rats administered DCM alone and EtOH alone. Biochemically, an elevation in the serum urea level indicates decreased reabsorption at the renal epithelium, whereas elevation in the serum creatinine level signifies renal dysfunction in the glomerular filtration rate. 40
Antioxidant enzymes, namely SOD, CAT, GPx, and GST as well as nonenzymatic antioxidant including GSH, play an important role in the maintenance of normal cellular function by protecting the cells against radical-mediated damage. Hepatic and renal SOD scavenges cytotoxic superoxide radicals by accelerating its conversion to H2O2 whereas CAT acts on the H2O2 and converts it to water and oxygen. The marked decrease in the activities of SOD and CAT was intensified in the liver and kidneys of rats co-exposed to DCM and EtOH signifies an additive inhibition of their defensive activities against cellular oxidative damage in the rats. Moreover, GSH is a key cellular sulfhydryl molecule, which plays a pivotal role in the regulation of the cellular redox state. Hence, a decrease in the cellular GSH level is well-known to indicate induction oxidative stress. 41 In the cell, the major function of GPx is to eliminate noxious soluble H2O2 and alkyl H2O2, whereas GST eliminates toxic electrophilic molecules using of GSH as the substrate. 42 Thus, intensification in the reduction of GPx and GST activities observed in rats co-exposed to DCM and EtOH connotes GSH overutilization and inhibition of detoxification process mediated by these GSH-dependent enzymes in the treated rats. The present investigation demonstrated that co-exposure to DCM and EtOH markedly exacerbated hepatic and renal oxidative damage evidenced by the increase in the MDA level in the investigated organs.
The marked increase in the LPO and the decrease in the cellular redox status in rats co-treated with DCM and EtOH were accompanied by histopathological damage in the liver and kidney of the treated rats. Light microscopic evaluation revealed that rats co-exposed to DCM and EtOH caused discernible histological degeneration characterized by multifocal area of marked inflammation and tubular necrosis in the kidney, 43 whereas disseminated congestion, focal area of portal fibrosis, and periportal infiltration by inflammatory cells were seen in the liver.
Conclusion for future biology
Taken together, the data from the present study highlight that co-exposure to DCM and EtOH elicited more harmful effects on the liver and kidney than the individual chemical exposure that is attributable to the intensified oxidative stress in the treated rats.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
