Abstract
Polycystic ovary syndrome (PCOS) is a heterogeneous endocrine and metabolic disorder, causes irregular menstrual cycles, dyslipidemia, excessive body weight, oxidative stress, hyperandrogenism, and infertility. This study focused on the impact of Cyperus esculentus (CES) on letrozole-induced PCOS in female Sprague-Dawley rats. Twenty (20) normal rats and twenty (20) PCOS rats (150–200 g, 8 weeks old) were randomly divided into four groups of ten (n = 10) rats each. Group A served as normal control group received 2 mL of normal saline, group B treated with 500 mg/kg body weight of CES, group C PCOS control group received 2 mL of normal saline, and group D PCOS rats post-treated with 500 mg/kg body weight of CES daily through gastric gavage for 30 days. Estrus cyclicity, body and ovaries weights, biochemical and histological parameters were measured. Observed irregular estrus cyclicity and multiple cysts in PCOS rats, increase glycemia, ovarian weight, triglycerides, total cholesterol, low-density lipoprotein cholesterol, very low-density lipoprotein cholesterol, malondialdehyde, luteinizing hormone (LH), testosterone, and decrease high-density lipoprotein cholesterol, estradiol, progesterone, catalase, reduced glutathione (GSH), and superoxide dismutase levels, compared with control. The intervention of CES ameliorated and restored the estrus cyclicity reproductive hormone, biochemical, and structural alterations. Moreover, CES significantly decreased cystic follicles, LH, and testosterone levels, but increased estradiol concentration. This plant may be beneficial in the management and treatment of PCOS-related reproductive and metabolic disorders.
Introduction
Polycystic ovarian syndrome (PCOS) is the most common cause of infertility in women of reproductive age. 1 According to the National Institutes of Health in the United States, it has a prevalence of 6 to 10% and 15% when the Rotterdam criteria are applied.2,3 Hyperandrogenism, anovulation, the presence of numerous ovarian cysts, abnormalities in the menstrual cycle, and fluctuating levels of gonadotropins are all symptoms of PCOS. 4 The etiology of PCOS is unclear, however, anomalies in androgen production and secretion are likely to be a contributing factor. 5 This condition is characterized by an excess of ovarian androgen production from numerous small follicles, anovulation, and insulin resistance in the reproductive life span of females. 6 Therefore, higher androgen level is the main marker of PCOS patients. 7 In women with PCOS, infertility could result from impaired ovarian folliculogenesis. Indeed, one of the main characteristics of PCOS is hyperandrogenism, which is believed to induce excessive production of gonadotropin-releasing hormone (GnRH) by hypothalamic neurons and luteinizing hormone (LH) by the pituitary gland. 8 Hyperandrogenism has also been linked to an increase in insulin secretion by pancreatic β-cells. 8 Hyperinsulinemia was discovered to reduce the hepatic synthesis of insulin-like growth factor (IGF) binding protein-1 and, as a result, enhance IGF-1 bioavailability. 9 Exposure to a single dose of letrozole in the adult rat can cause irregular cycles, lack of ovulation, and polycystic ovaries with a high number of atretic follicles and cysts. These ovarian morphological changes are comparable to those seen in women with PCOS. 10 The polycystic ovarian morphology in PCOS is presented by an excessive amount of growing follicles. 11 Oxidative stress (OS) is also linked to a higher risk of infertility in patients with PCOS. Many studies have found that patients with PCOS have abnormal levels of OS-related biochemical markers including malondialdehyde (MDA), glutathione peroxidase (GPx), catalase, and superoxide dismutase (SOD). 12 Furthermore, OS is linked to an excess of reactive oxygen species (ROS), which can lead to DNA damage and tumor suppressor gene mutations, resulting in uncontrolled ovarian cell proliferation, multiple cysts, and infertility. 13 Several drugs, including metformin and clomiphene citrate, have been used to treat PCOS, although they are frequently accompanied by significant adverse effects like headache, vomiting, stomach upset, sweating, blurred vision, and hypoglycemia.14,15 A new therapeutic approach with fewer side effects, easy availability, and broad-spectrum is required.
Cyperus esculentus (CES) is a member of the Cyperaceae family and is also known as chufa, yellow nutsedge, earth almond, and ground almond. It is a perennial crop widely grown in East Africa, Asia, the Arabian Peninsula, and parts of Europe, especially Spain. 16 CES appears to have more potential as a source of nutrition and industrial materials; it may be used to make beverages, milk or yogurt, flour, nougat, jam, beer, chocolate, a feed source, edible oil, and soaps, among other things. 17 Minerals such as potassium, phosphorus, magnesium, calcium, and siron are abundant in CES nuts. It’s also high in vitamins C and E, as well as a significant amount of vitamin B1. 18 Tiger nut helps the body adapt to stress by regulating the central nervous system. 19 Moreover, it defends the internal mechanisms and prevents constipation and diarrhea. 18 The researches done to identify the chemical composition of CES tubers indicated that the moisture content of tiger tubers was (8.50%). The carbohydrate content of these tubers was shown to be the leading component (45.73%), followed by the oil content (30.01%). Moreover, tiger nut tubers had 5.08% protein, 2.23% ash, and 14.80% crude fiber. The starch content of tiger nut tubers was (293.50 g/kg) followed by sucrose content (99.35 g/kg) and reducing sugar (27.61 g/kg).20,21 Tiger nut has been utilized as a healthy plant for over 4000 years due to its high amount of energy, minerals, and oleic acid. It has a significant amount of arginine, which releases the hormone that generates insulin, as well as carbohydrates with a sucrose and starch base. 22 Due to its high content of potassium, phosphorus, magnesium, calcium, and iron, tiger nut tubers are beneficial to bones, tissue healing, muscles, circulation, and body growth. 23 Tiger nut is also considered to have a protective impact against heart disease and cancer due to its high vitamin E concentration, which aids in the development and function of red blood cells. 24 Vitamin C in tiger nut is a good antioxidant, promotes iron absorption, and helps in maintaining vitamin E levels essential for the immunological system and tissue preservation. 21 Considering the above-mentioned pharmacological properties of CES may be a good candidate for the treatment of PCOS-related metabolic and reproductive disorders. Therefore, the present study investigated the impact of aqueous of CES on estrus cyclicity, blood glucose level, lipid profile, oxidative stress-related biochemical parameters, sex hormones, and ovarian histology in letrozole-induced PCOS in rats.
Materials and methods
Chemicals, drugs, reagents, and assay kits
Letrozole was obtained from FEGTOCHI Pharmacy, Akure, Nigeria, thiobarbituric acid and reduced glutathione were purchased from Sigma-Aldrich Corp. (St. Louis, MO USA). Assay kits for protein are products of Randox Laboratories Limited (Co Antrim, United Kingdom) while those of testosterone, follicle-stimulating hormone (FSH), luteinizing hormone (LH), estradiol, progesterone, and leptin were obtained from Monobind Inc. California, USA. All other reagents used were of analytical grades.
Plant collection and identification
Cyperus esculentus were purchased from Shasha market, Akure and taken to the Centre for Research and Development (CERAD), Federal University of Technology, Akure (FUTA), Ondo State, Nigeria for proper identification and authentication. The samples of the CES nut were identified and authenticated by Mr. Omomoh Bernard and a sample of the plant voucher FUTA/0196 deposited for reference purpose.
Extraction of plant material
The fresh CES nut was thoroughly washed in sterile water and air-dried under shade for 6 weeks to a constant weight in the laboratory. The air-dried nuts were weighed using CAMRY (EK5055, Indian) electronic weighing balance and were milled with an automatic electrical blender (model FS-323, China) to powdered form. Seven hundred grams of the milled plant sample was later soaked in 1500 mL of phosphate-buffered saline (PBS) for 2 h at room temperature and was later filtered through cheesecloth and then through Whatman No 1 filter paper, the filtrate was then bottled in clean screw-cap bottles and stored in a refrigerator until use [Figure 1].
25
Fresh Cyperus esculentus tubers Dried Cyperus esculentus tubers (Maduka and Ire, 2018).
Phytochemical screening
Qualitative phytochemical analysis of the extract of CES nuts was done in accordance with Soni and Sosa. 26 While modifications on the report by Grindberg and Williams, 27 on high performance liquid chromatography was adopted to quantify the vitamins.
Experimental animals
A total number of forty female Sprague-Dawley (SD) rats (150–200 g, 8 weeks old) were used for this study. The animals were obtained from the laboratory animal’s House, Faculty of Basic Medical Sciences, Ladoke Akintola University of Technology, Ogbomoso, Nigeria. All rats were kept in a pathogen-free environment conserved in stainless steel cages and the temperature was adjusted at 21–24°C. Relative humidity was nearly 60% with a 12-h light-dark cycle. All rats had standard rodent chow and filtered water during the experimental period ad libitum. The rats were left to adapt for 14 days before use in the study herein. All experimental procedures followed the recommendations provided in accordance with the Basic & Clinical Pharmacology & Toxicology policy for experimental studies. 28
Induction of polycystic ovary syndrome
Twenty (20) Healthy adult female Sprague-Dawley rats were treated with 1 mg/kg of letrozole daily through gastric gavage for 21 days to induce PCOS. 29
Experimental design
Twenty (20) normal female rats and twenty (20) PCOS rats were randomly divided into four groups of ten (n = 10) rats each. Group A served as normal control group received 2 mL of normal saline, group B treated with 500 mg/kg body weight of CES only, group C PCOS (1 mg/kg of letrozole only) group and group D PCOS (letrozole-induced) rats post-treated with 500 mg/kg body weight of CES daily through gastric gavage the experiment lasted for 40 days.
Vagina smear
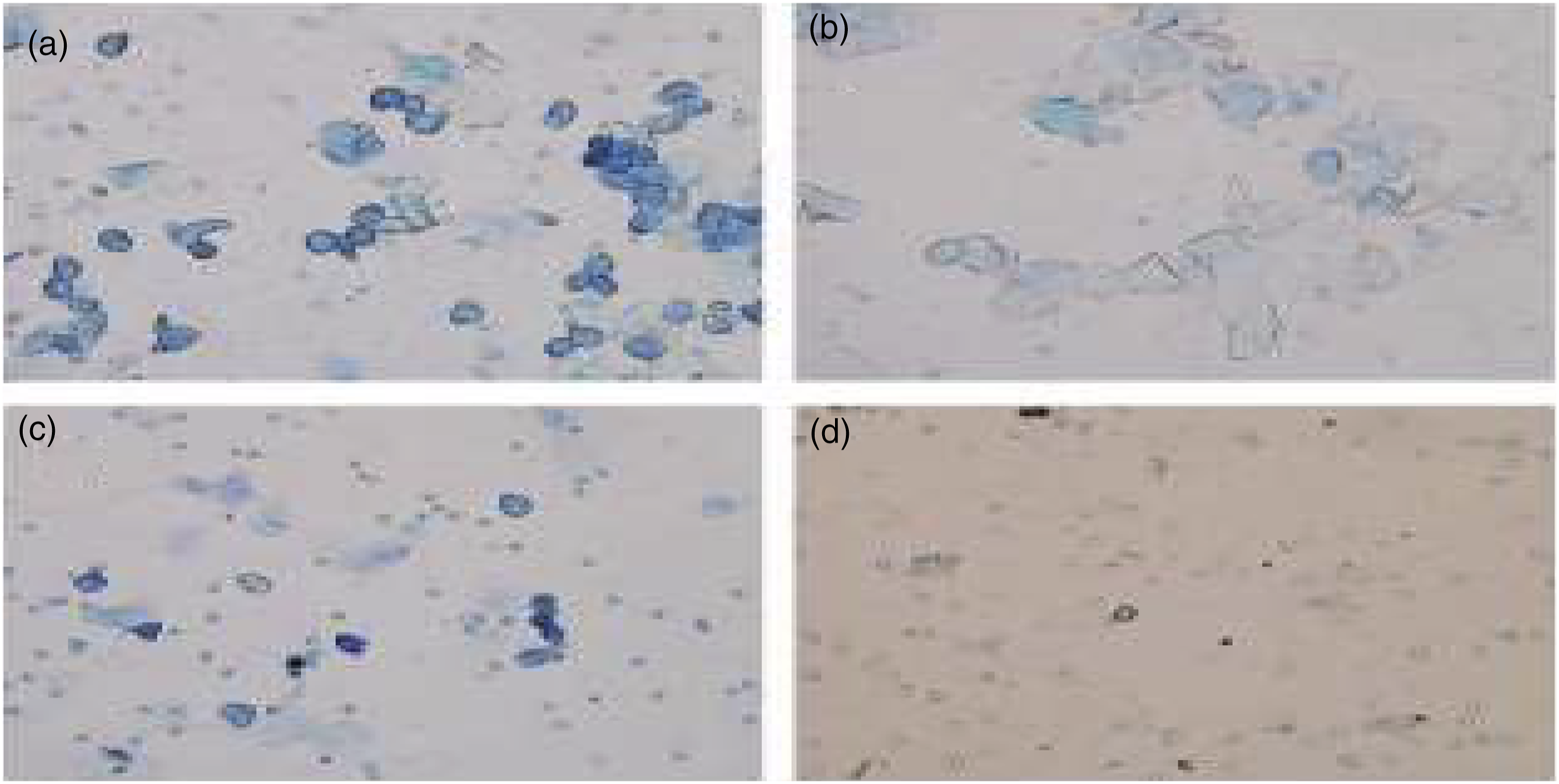
The PCOS condition was confirmed by the preparation of a vaginal smear to observe reproductive cycle irregularities. Vaginal secretion was collected daily with a plastic pipette by inserting the tip in the vagina of female rats, filled with 15 μL of normal saline, one drop of collected vaginal fluid was placed on glass slides. Vaginal fluid was fixed by placing the slides on a slide warming table and stained with methylene blue staining solution. After staining, slides were washed to remove the excess stain, dried, and observed under a light microscope to assess the changes in the estrus cycle. Three types of cell were recognized: round and nucleated cells were epithelial cells; irregular cells without a nucleus were the cornified cells and the small round cells were the leucocytes, their mutual proportion was used for the determination of the different phases of the estrus cycle [Figure 2].
30
Figures and cell types of vaginal smears. (a): Preoestrus, loaded mainly with epithelial cell; (b): Estrus, loaded mainly with keratinocytes; (c): Metoestrus, displaying epithelial cell, keratinocytes, and leukocytes; (d): Diestrus, displaying full of leukocytes.
31
Sample collection
At the end of the experiment, the rats fasted overnight, weighed and blood samples were collected from the retro-orbital venous sinus,32,33 into a sterile plain sample bottle, left at room temperature for 30 min to enable clotting, and then centrifuged at 3500 rpm for 20 min using a benchtop centrifuge (Midfield Equipment & Scientific limited, England) to separate serum, and the fresh serum was used immediately for the analysis of sex hormones. After blood samples collection, the animals from all groups were euthanized in an airtight container containing 4% fluothane in oxygen followed by cervical dislocation. The ovaries of all rats were immediately dissected out and weighed. The right ovary of each rat was homogenized in a glass homogenizer in cold phosphate-buffered saline (1:4) (PBS, 0.01 mol/L, pH 7). The prepared homogenate was centrifuged for 5 min at 5000×g; then filtered and used for measurement of oxidative stress markers. The left ovaries were fixed in Bouin’s for histological assessment.
Determination of sex hormone
Serum testosterone, LH, FSH, progesterone, estradiol, leptin, and Anti-mullerian hormone (AMH) of the rats were quantitatively measured by ELISA using specific kits (Monobind Inc. California, USA). The experiment was performed as per the manufacturer’s instructions.
Biochemical analysis
Determination of ovarian malondialdehyde (MDA)
Lipid peroxidation level in form of malondialdehyde in the ovary was measured using thiobarbituric acid reactive substances (TBARS) with some modifications as previously described. 34 The ovary was homogenized in ice-cold 0.15 M KCl (10%) and the concentration of TBARS was expressed as nmol of MDA per mg protein using 1,1,3,3-tetra methoxy propane as standard. The absorbance was read at 532 nm.
Determination of ovarian reduced glutathione (GSH)
The reduced form of glutathione was determined using Ellman’s reagent 5-5-dithiol-bis (2-nitrobenzoic acid) (DTNB) as a coloring reagent. 35 The absorbance was read at 412 nm by a spectrophotometer. GSH concentration was calculated from a standard curve.
Determination of ovarian superoxide dismutase (SOD)
Ovarian superoxide dismutase was assayed by the method of, 36 which involves the inhibition of photochemical reduction of nitro blue tetrazolium (NBT) at pH 8.0. A single unit of enzyme is defined as the quantity of superoxide dismutase required to produce 50% inhibition of photochemical reduction of NBT. The absorbance was read at 580 nm against a blank using a UV – Vis spectrophotometer. The activity was expressed as U/mg protein.
Determination of ovarian catalase (CAT)
Catalase activity was estimated in ovary homogenate by the method reported by. 37 The specific activity of catalase has been expressed as mmoles of H2O2 consumed/min/mg protein. The difference in absorbance at 240 nm per unit time is a measure of catalase activity.
Determination of ovarian COX activity
COX activity assay kit (Cayman, Ann Arbor, MI, USA) for measuring the activity of COX in rat’s ovaries were used. The harvested ovarian tissues were washed thoroughly with ice-cold Tris buffer, pH 7.4, containing 0.16 mg/mL of heparin, to remove any red blood cells and clots, and then stored at −800C. For each rat, a sample of ovarian tissue was homogenized in 5 mL of cold buffer (0.1 M Tris-HCl, pH 7.8, containing 1 mM EDTA) per gram of tissue and centrifuged at 10 000xg for 15 min at 40C. Removed supernatant was kept on ice. The peroxidase activity of COX was measured by the COX activity assay kit. This was assayed colorimetrically by monitoring the appearance of oxidized N, N, N, N′-tetramethyl-phenylenediamine at 590 nm. COX-2 activity was measured by using the COX-1-specific inhibitor. 38 Results for COX activities were given as units per milligram of protein. The activity of COX was expressed as nmol/min/mg protein (U/mg protein).
Determination of glucose and lipid profile
Glucose was determined by the glucose oxidase method using a commercially available kit (Fine test, Infopia Diagnostics). The lipid profile, including plasmatic total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), very low-density lipoprotein cholesterol (VHDL-C), high-density lipoprotein cholesterol (HDL-C), and triglycerides (TG), was evaluated using standard colorimetric kits (CORMAY, Łomianki, POLAND) as described previously.39,40 The LDL-C and very low-density lipoprotein cholesterol (VLDL-C) levels were calculated based on Friedewald’s equation: LDL = TC – TG/5 – HDL; VLDL = TG/5. 37
Determination of ovarian histology, follicular count, and histopathology
The left ovary was harvested and fixed in Bouin’s fluid for 24 h, moved to 70% alcohol for dehydration. Then they passed through 90% and absolute alcohol and xylene for different durations before being transferred into molten paraffin wax for 1 h each in an oven at 65°C for infiltration. The tissues were embedded, and serial sections on a rotary microtome set at 5 μm were performed. The tissues were picked up with albumenized slides and allowed to dry on hot plates for 2 min. The slides were dewaxed with xylene and passed through descending concentrations of alcohol, 100% alcohol, and 50% alcohol (in that order), and then in water for 5 min. The slides were then stained with hematoxylin and eosin (H&E), mounted in DPX, and observed under a light microscope (Olympus, Japan), photomicrographs were taken at a magnification of ×100, x400. Follicles were defined primarily if had a single layer of cuboidal granulosa cells; preantral if had one or two small spaces filled by follicular fluid; antral if had a single large antral space; preovulatory if oocyte was surrounded by a rim of cumulus cells 41 ; atretic if follicles were deformed or oocyte was absent or pyknotic nuclei presented in granulosa cells, and corpora lutea. All types of follicles were counted in each ovary. Then all follicles were classified as healthy (primary, preantral, antral, and preovulatory) or atretic. 42 All types of follicles, cysts, and corpus luteal in each ovary were counted by two persons within 9 representative sections at least 25 μm apart.
Histopathological assessment was carried out by a pathologist, who was blinded to all experimental data. Ovarian degeneration criteria were graded between 0-3. While the normal histological structure appearance was scored as grade 0, the degenerative changes were scored as mild (grade 1), moderate (grade 2), and severe (grade 3). While histochemical scoring was performed, scoring was done by evaluating one central and five peripheral areas.
Fertility of female rats
Fertility was estimated by modification method that was previously described by. 43 The adult female rats were treated with letrozole, CES nut, and control female counterparts. Each female rat was placed in an individual cage with two untreated male rats of the same strain. They were left together for 10 days during which two oestrone cycles had elapsed in female rats. And after 10 days the untreated male rats are removed, pregnant females were sacrificed under diethyl ether anesthesia and the number of pregnant rats, implantation sites, and the number of fetuses was recorded.
Statistical analysis
The results were reported as mean ± standard error of the mean (S.E.M). An instant-3 computer program (v2.04, GraphPad Software Inc., San Diego, CA, USA) was used to analyze the numbers and to evaluate the significant differences; the comparison of means between every two experimental groups was made using one-way ANOVA with Newman-Keuls Multiple Comparison significant difference test. The differences were considered significant if p < 0.05.
Results
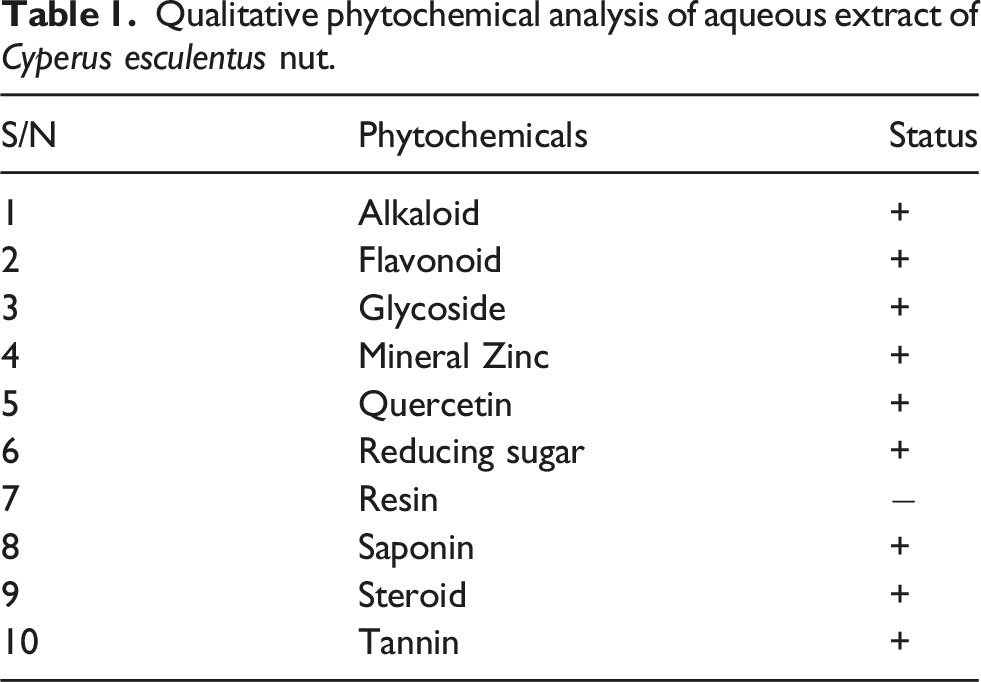
Phytochemical screening
Qualitative phytochemical analysis of aqueous extract of Cyperus esculentus nut.
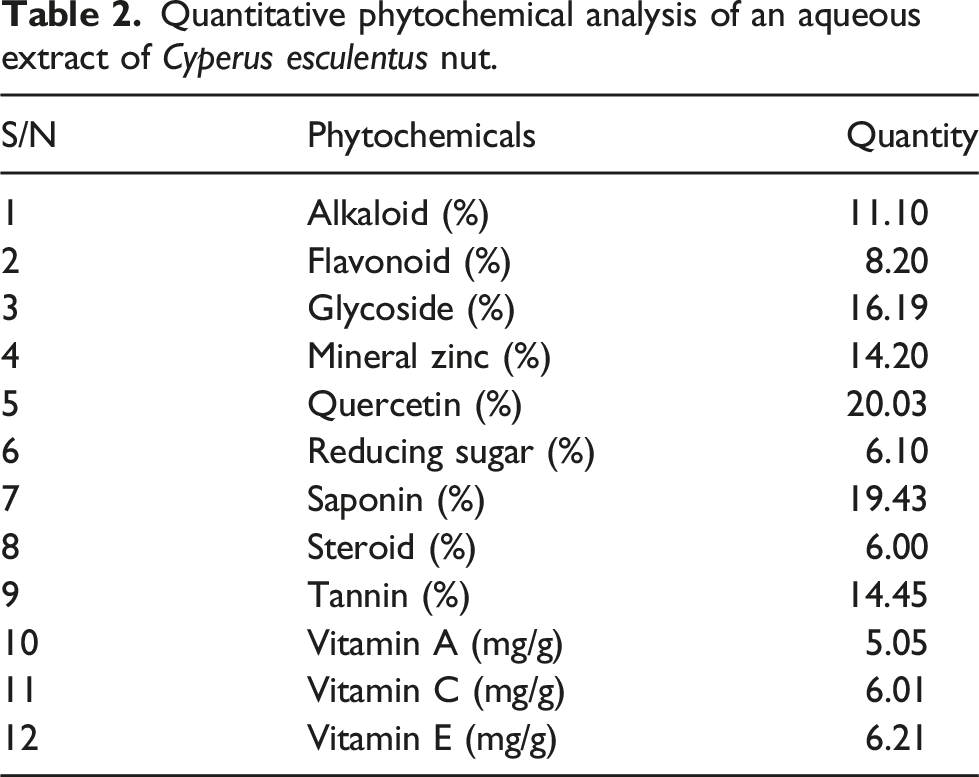
Quantitative phytochemical analysis of an aqueous extract of Cyperus esculentus nut.
Oestrus cycle’s
Ameliorating potential of Cyperus esculentus tubers (Tiger Nut) extract on oestrus cycles in letrozole-induced PCOS in Sprague-Dawley rats.
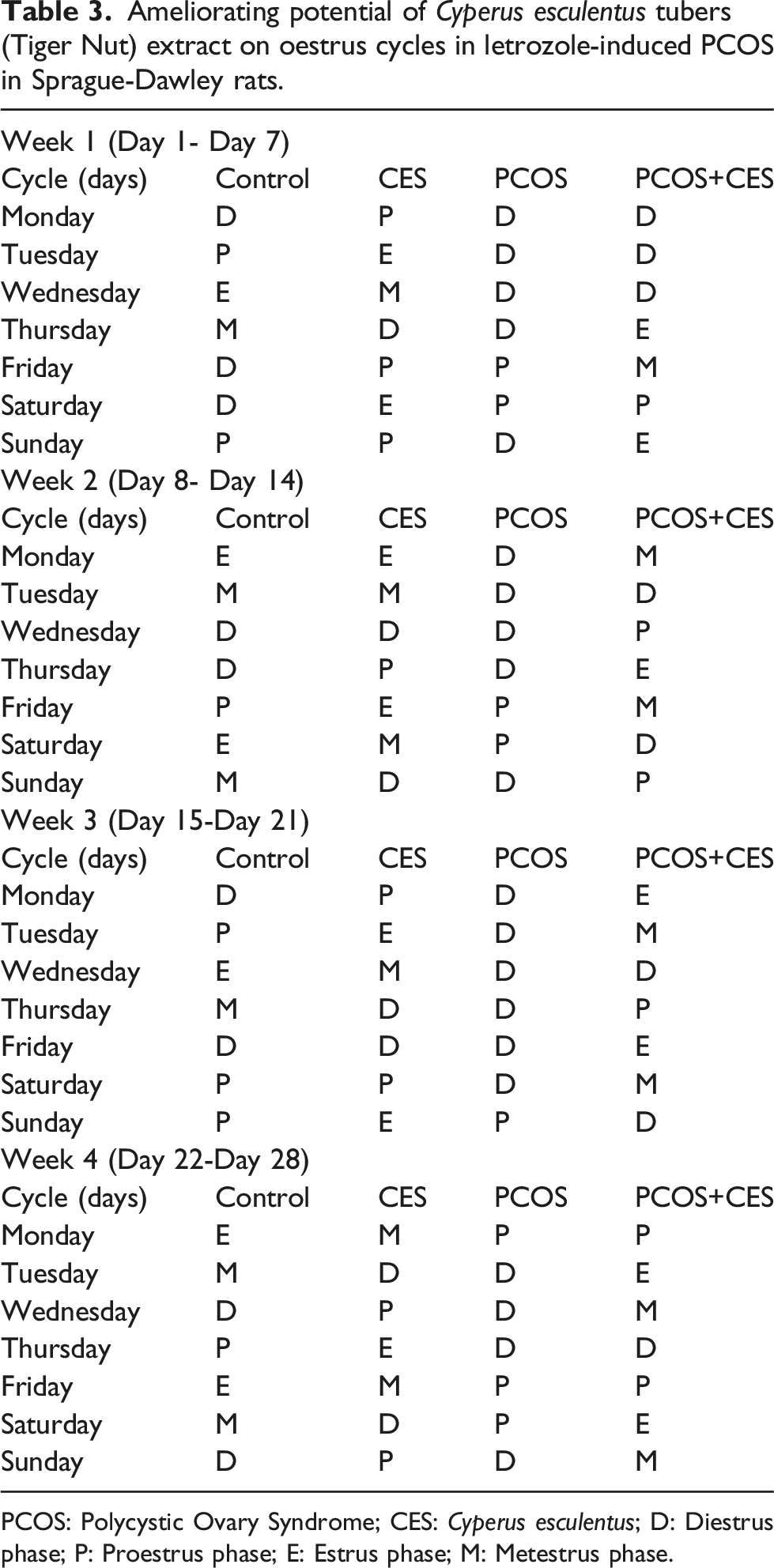
PCOS: Polycystic Ovary Syndrome; CES: Cyperus esculentus; D: Diestrus phase; P: Proestrus phase; E: Estrus phase; M: Metestrus phase.
Bodyweight, body length, ovarian weight, and lee index
Ameliorating potential of Cyperus esculentus tubers (Tiger Nut) extract on body and ovary weight in letrozole-induced PCOS in Sprague-Dawley rats.
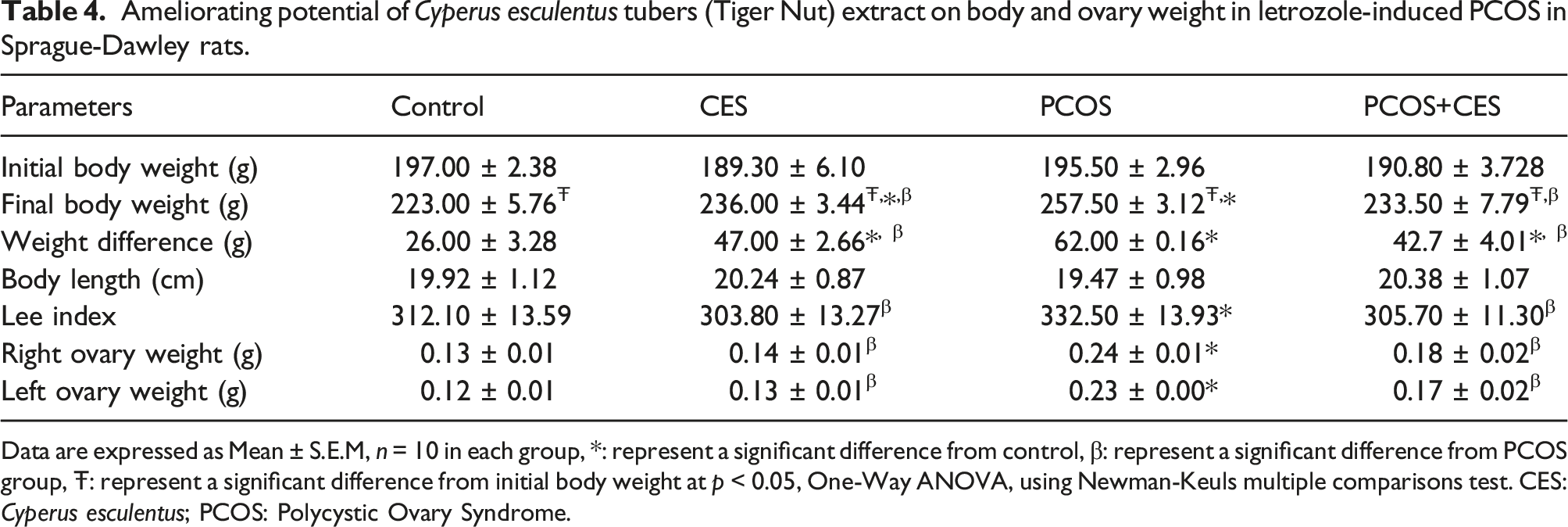
Data are expressed as Mean ± S.E.M, n = 10 in each group, *: represent a significant difference from control, β: represent a significant difference from PCOS group, Ŧ: represent a significant difference from initial body weight at p < 0.05, One-Way ANOVA, using Newman-Keuls multiple comparisons test. CES: Cyperus esculentus; PCOS: Polycystic Ovary Syndrome.
Hormone concentration
Ameliorating potential of C. esculentus tubers (Tiger Nut) extract on hormone parameters in letrozole-induced PCOS in Sprague-Dawley rat.
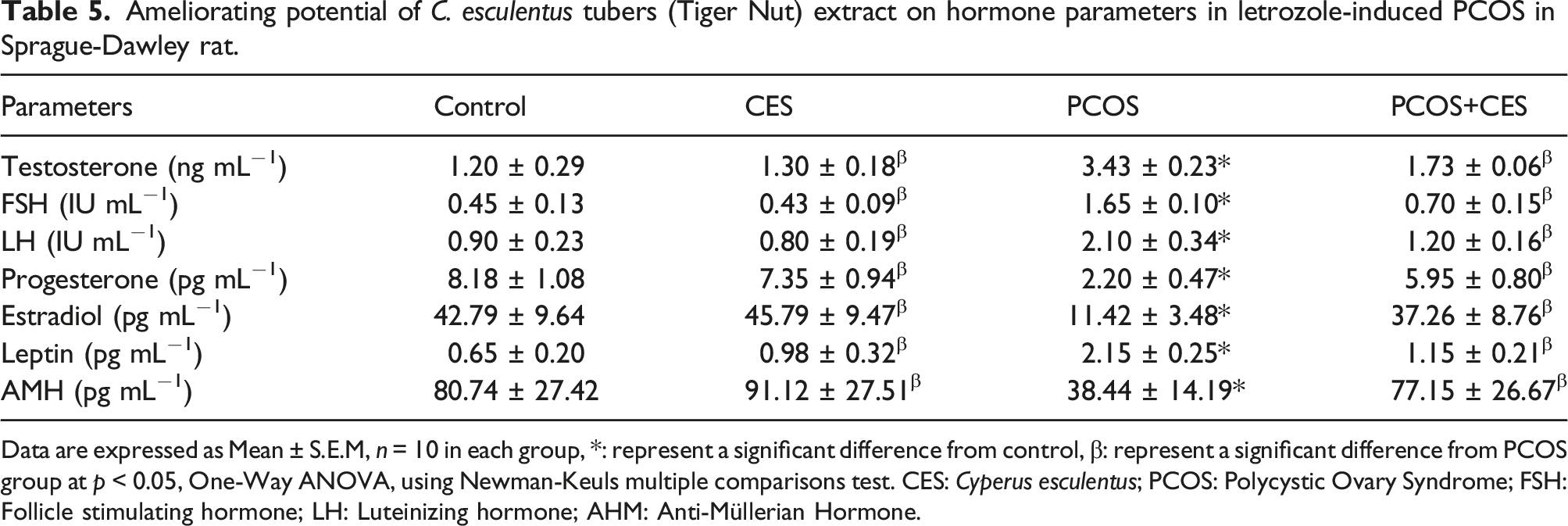
Data are expressed as Mean ± S.E.M, n = 10 in each group, *: represent a significant difference from control, β: represent a significant difference from PCOS group at p < 0.05, One-Way ANOVA, using Newman-Keuls multiple comparisons test. CES: Cyperus esculentus; PCOS: Polycystic Ovary Syndrome; FSH: Follicle stimulating hormone; LH: Luteinizing hormone; AHM: Anti-Müllerian Hormone.
Follicular count
Ameliorating potential of Cyperus esculentus tubers (Tiger Nut) extract on ovary histopathology score parameters in letrozole-induced PCOS in Sprague-Dawley rat.
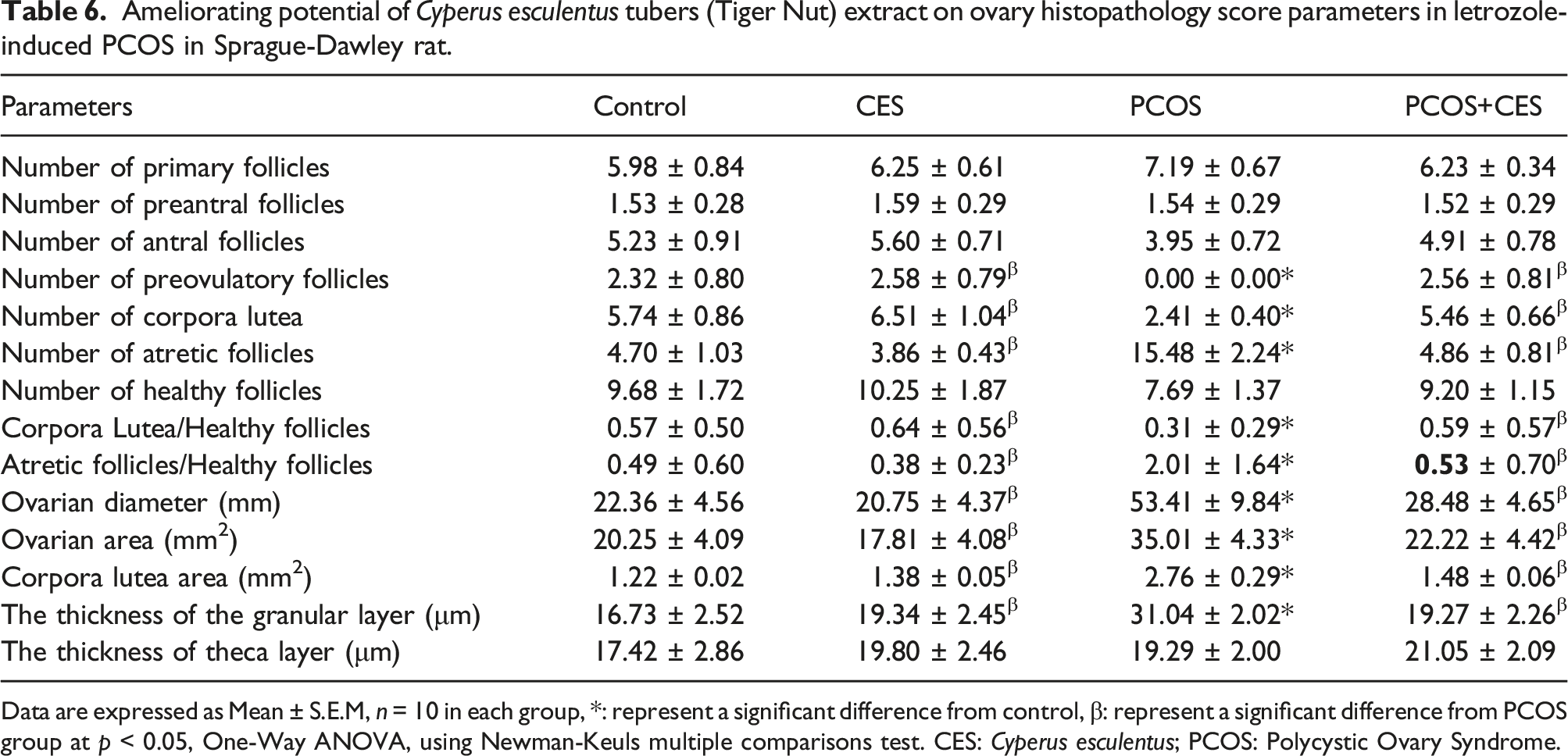
Data are expressed as Mean ± S.E.M, n = 10 in each group, *: represent a significant difference from control, β: represent a significant difference from PCOS group at p < 0.05, One-Way ANOVA, using Newman-Keuls multiple comparisons test. CES: Cyperus esculentus; PCOS: Polycystic Ovary Syndrome.
The PCOS-induced rats’ exhibit significant (p < 0.05) reduction in ovarian diameter (OD), ovarian area (OA), corpora lutea area (CLA), and thickness of the granular layer (TGL) as compared to animals in the control group. However, there was a significant (p < 0.05) increase in OD, OA, CLA, and TGL in the group treated with CES only and PCOS+CES treated group compared to PCOS untreated group. Also, no observed significant difference in OD, OA, CLA, and TGL among CES, PCOS and control group (p > 0.05). In addition, no significant (p > 0.05) difference in thickness of theca layer between the control and other experiment groups [Table 6].
Ovulation rate, ova shed, fertility index, and gestation rate
Ameliorating potential of Cyperus esculentus tubers (Tiger Nut) extract on ovulation rate, ova shed, fertility index, and gestation rate in letrozole-induced PCOS in Sprague-Dawley rat.

Data are expressed as Mean ± S.E.M, n = 10 in each group, *: represent a significant difference from control, β: represent a significant difference from PCOS group at p < 0.05, One-Way ANOVA, using Newman-Keuls multiple comparisons test. CES: Cyperus esculentus; PCOS: Polycystic Ovary Syndrome.
Ovarian oxidative stress and antioxidant concentration
Ovarian MDA concentration of Polycystic Ovary Syndrome rats shows a significant (p < 0.05) increase with a concurrent decrease in ovarian GSH concentration compared with the control group, indicating the oxidative action of letrozole on ovarian tissues. However, CES post-treatment with down-regulate MDA concentration and up-regulate GSH concentration, indicating the antioxidant capacity of CES [Figure 3b, 3d]. The concentration of ovarian TP, SOD, CAT, and GSH were significantly (p < 0.05) lowered in PCOS rats as compared with the control rats [Figure 3a, 3c-e). However, the intervention of CES insignificantly (p > 0.05) increase the concentration of ovarian TP, SOD, CAT, and GSH in PCOS+CES treated group compared with the control group but significantly (p < 0.05) higher in comparison with PCOS rats. Administration of CES extracts alone significantly (p < 0.05) reduced ovarian MDA compared to the PCOS group and no significant (p > 0.05) difference when compared with the control and PCOS+CES rats. Also, CES-only treatment revealed a significant (p < 0.05) increase in ovarian TP, SOD, CAT, and GSH in relation with PCOS rats and insignificantly increase in comparison with control and PCOS+CES rats [Figure 3a, 3c-e]. Ameliorating potential of Cyperus esculentus tubers (Tiger Nut) extract on Total protein (TP), malondialdehyde (MDA), superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH), activities in letrozole-induced PCOS in Sprague-Dawley rats. Data are expressed as the mean ± SEM (n = 10), *: Significant change from the control group at p < 0 05; β: significant change from the PCOS group at p < 0 05, One-Way ANOVA, using Newman-Keuls multiple comparisons test. CES: Cyperus esculentus; PCOS: Polycystic Ovary Syndrome.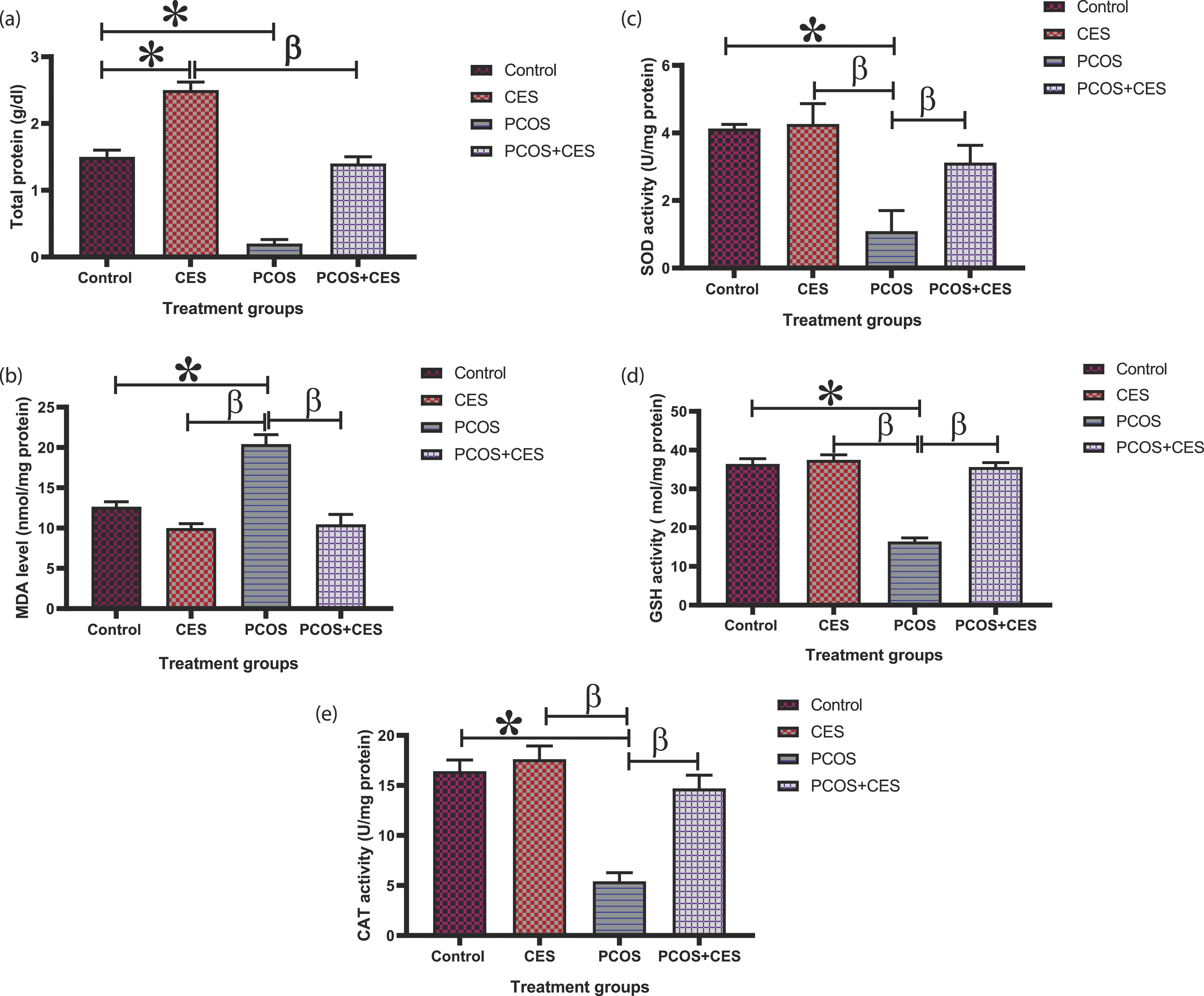
Glucose analysis and lipid profile
There was an observed significant (p < 0.05) increase in glucose, cholesterol, and triglyceride levels in PCOS-induced rats compared to the control group. However, treatment with CES and administration of CES only significant (p < 0.05) decrease the level of glucose, cholesterol, and triglyceride as compared to PCOS-induced rats [Figure 4(a)–(c)]. Also, PCOS rats present a significant (p < 0.05) increase in LDL-C and VLDL-C in the PCOS group compared to the control group, whereas HDL-C levels were considerably decreased in PCOS rats compared to control. Treatment with CES and administration of CES only reduced LDL-C and VLDL-C and increase HDL-C in comparison to the PCOS group [Figure 4(d)–(f)]. Similarly, there was a profound increase (p < 0.05) in ratios of LDLC/HDL-C, TC/HDL-C, and TG/HDL-C in PCOS compared to control. Whereas, treatment with CES reduced the ratios of LDLC/HDL-C, TC/HDL-C, and TG/HDL-C significantly higher (p < 0.05) when compared to that of PCOS animals [Figure 4(g)–(i)]. (a-i): Ameliorating potential of Cyperus esculentus tubers (Tiger Nut) extract on (a) Glucose, (b) Cholesterol, (c) Triglycerides, (d) HDL-C, (e) LDL-C, (f) VLDL-C, (g) LDL-C/HDL-C (h) TC/HDL-C and (i) TG/HDL-C in letrozole-induced PCOS in Sprague-Dawley rats. Data are expressed as the mean ± SEM (n = 10), *: Significant change from the control group at p < 0 05; β: significant change from the PCOS group at p < 0 05, One-Way ANOVA, using Newman-Keuls multiple comparisons test. CES: Cyperus esculentus; PCOS: Polycystic Ovary Syndrome.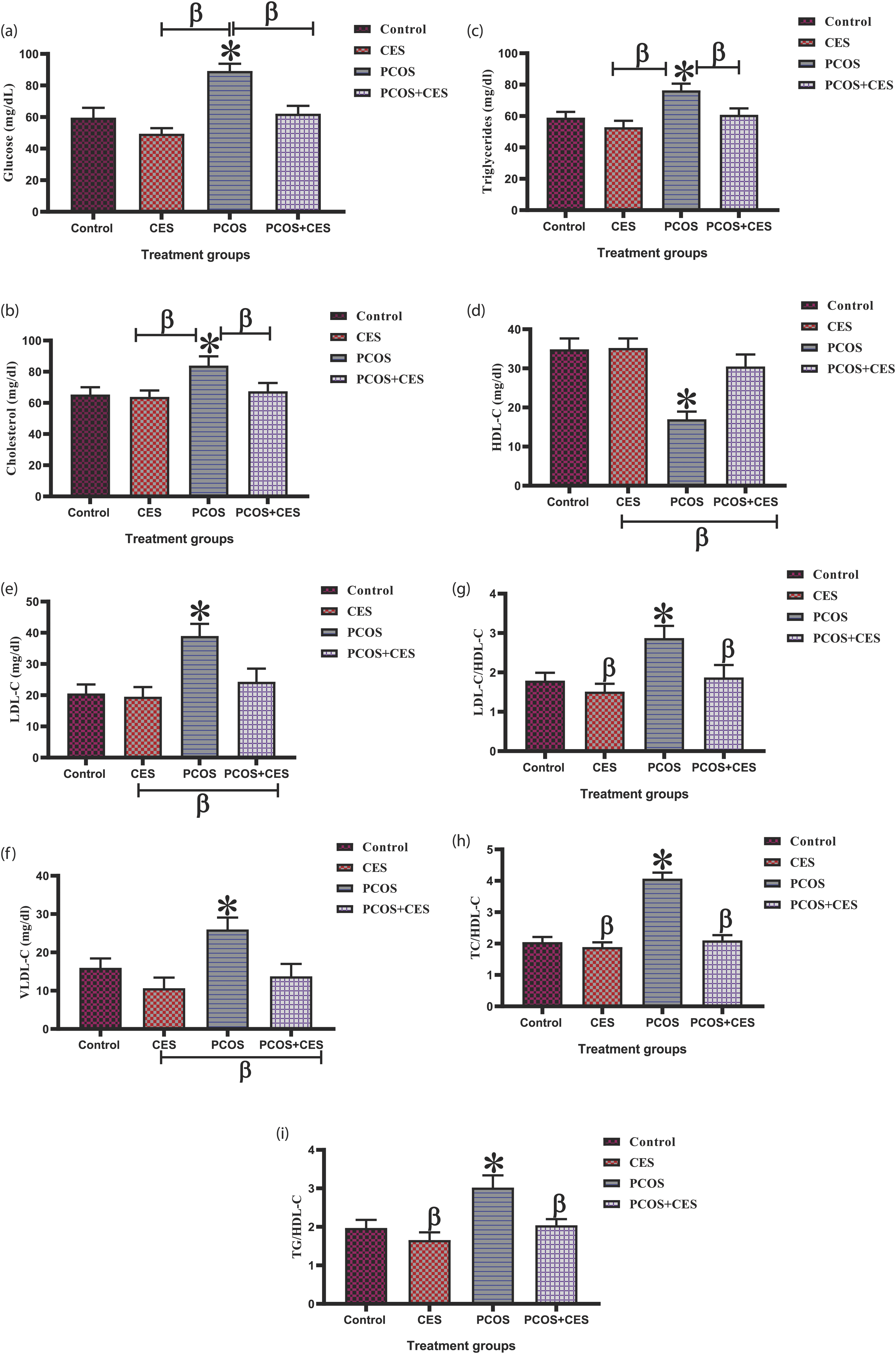
Ovarian COX-1 and COX-2 concentration
The concentration of ovarian COX-1 significantly (p < 0.05) decrease in PCOS animals compared to control. Administration of CES and post-treatment with CES significantly (p < 0.05) increase the activity of concentration of ovarian COX-1 as compared with PCOS-induced animals. The CES treatment group showed an insignificant higher value in COX-1 than PCOS+CES treated SD rats (p > 0.05) [Figure 5(a)]. (a,b): Ameliorating potential of Cyperus esculentus tubers (Tiger Nut) extract on ovarian cyclooxygenase (a). (COX)-1 and (b). COX-2 concentration in Letrozole-induced PCOS in Sprague-Dawley rats. Data are expressed as the mean ± SEM (n = 10), *: Significant change from the control group at p < 0 05; β: significant change from the PCOS group at p < 0 05, One-Way ANOVA, using Newman-Keuls multiple comparisons test. CES: Cyperus esculentus; PCOS: Polycystic Ovary Syndrome.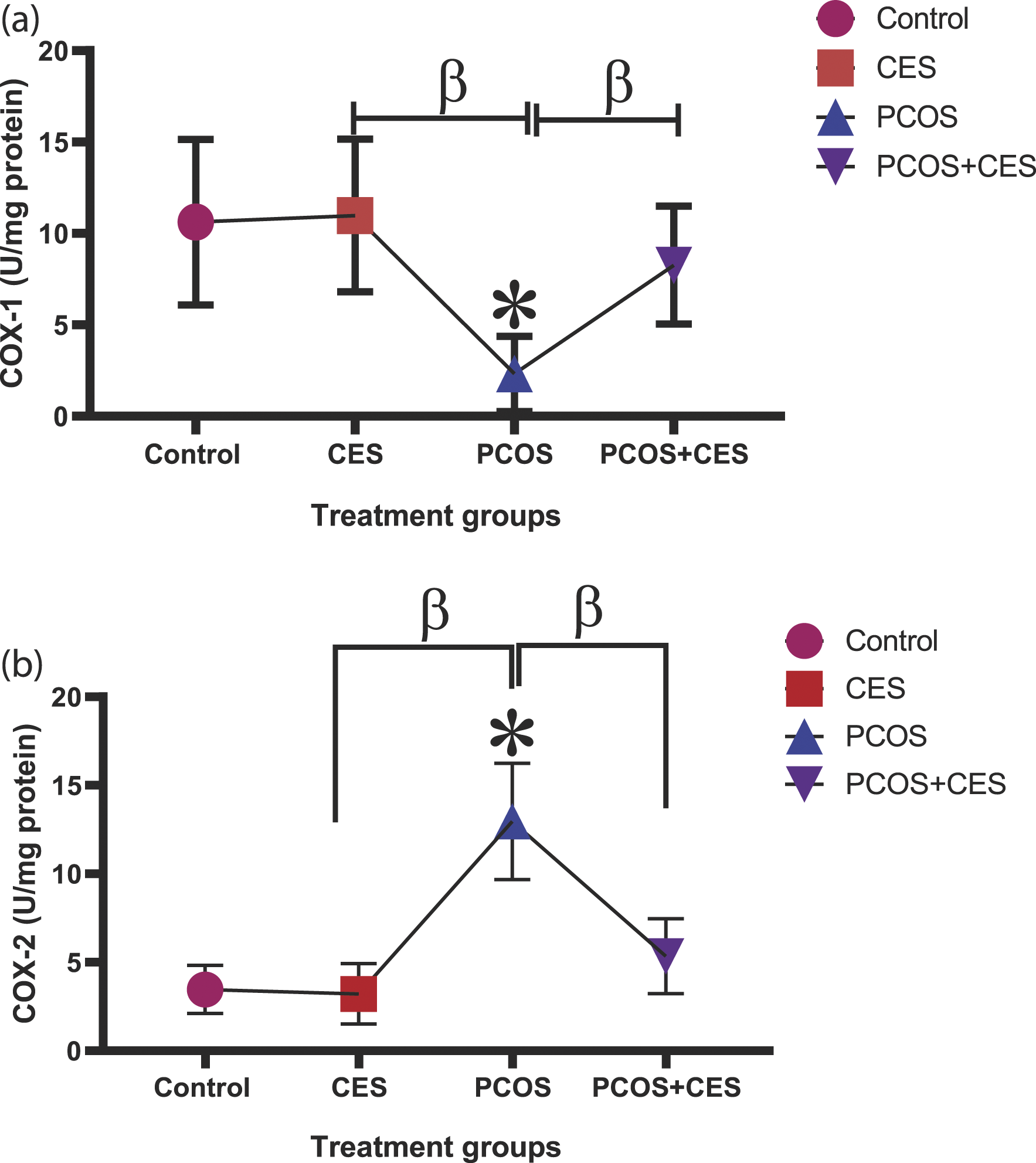
There was an observed significant (p < 0.05) increase in the activity of COX-2 in the ovary of PCOS-induced rats when compared to that of rats in the control group. However, treatment of PCOS rats with CES revealed a significant (p < 0.05) decline in the activity of ovarian COX-2 compared to PCOS-induced rats. Also, administration of CES significantly (p < 0.05) decrease ovarian COX-2 compared to PCOS untreated rats. Ovarian COX-2 shows no significant (p > 0.05) difference in the group treated with CES only and PCOS+CES treated group [Figure 5(b)].
Histomorphological findings
The histological examination of the control ovary showed the normal structure of surface epithelium and well-developed stroma with cortical region. Primary and secondary follicles showed a compact appearance. Preantral and antral follicles were also apparent with a fluid-filled cavity antrum present among the granulosa cells [Figure 6(a) and (b) (A1, A2)]. The Ovarian section of CES alone treated rats shows many healthy ovarian follicles in various stages of development. The theca and granulosa cells around the developing oocytes are well arranged. [Figure 6(a) and (b) (B1, B2)]. Ovarian section of PCOS rats revealed distorted histological architecture with a population of immature cysts with marked atresia, highly damaged primordial and primary follicles surrounded by atrophied follicular cells with withering granulosa cells, the population of an atretic follicle in the cortex of ovary and cells debris in the antrum of the follicle [Figure 6(a) and (b)(C1, C2)]. Ovarian section of PCOS+CES treated rats revealed regenerated histo architecture and normal structure of surface epithelium with a population of mature follicles and regeneration of damaged primordial and primary follicle, few atretic follicles in the cortex of ovary disappearance of cells debris in the antrum of the follicle [Figure 6(a) and (b)(D1, D2)]. (a): Control (A1): Ovarian section of a control rat showing follicles at various stages with normal follicle and corpus luteum. B1(CES): Ovarian section showing normal follicles with abundant corpus luteum, C1(PCOS): Ovarian section of PCOS rat showing cystic, atretic, and antral follicles and decreased corpus luteum. D1 (PCOS+CES) ovarian section showing decrease numbers of atretic and cystic follicles with an increase in normal follicles and corpus luteum. CF: Cystic follicle. H&E, Mag x100. CF: Cystic Follicle, GL: Granulosa layer, O: Oocyte, NF: Normal follicle, CL: Corpus luteum. (b): Control (A1): Ovarian section of a control rat showing follicles at various stages with normal follicle and corpus luteum. B1(CES): Ovarian section showing normal follicles with abundant corpus luteum, C1(PCOS): Ovarian section of PCOS rat showing cystic, atretic, and antral follicles and decreased corpus luteum. D1 (PCOS+CES) ovarian section showing decrease numbers of atretic and cystic follicles with an increase in normal follicles and corpus luteum. CF: Cystic follicle. H&E, Mag x400. CF: Cystic Follicle, GL: Granulosa layer, O: Oocyte, NF: Normal follicle, CL: Corpus luteum.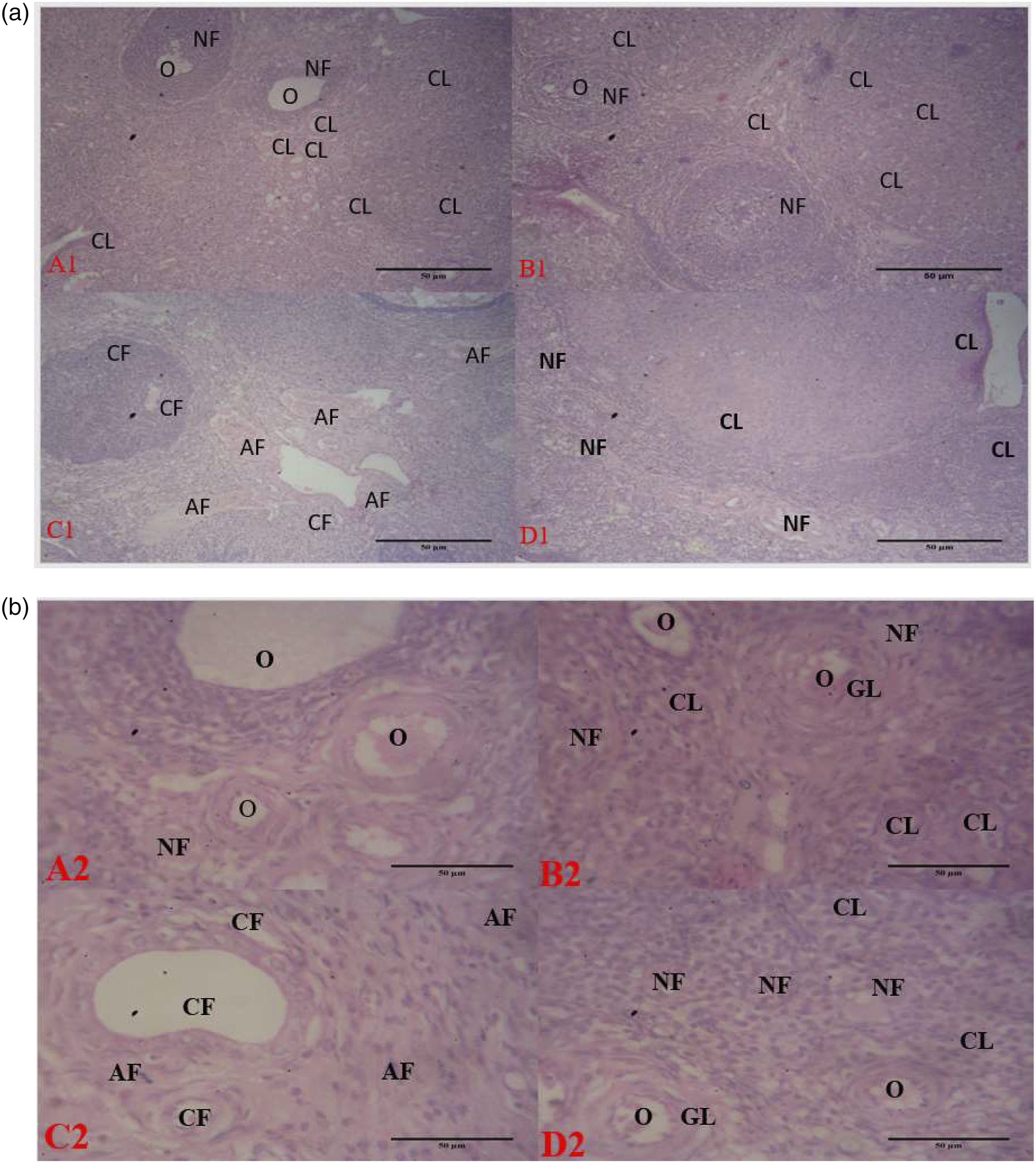
The histopathological score of ovarian tissues
There was an observed significant (p < 0.05) increase in cell degeneration, vascular congestion, interstitial edema, and vascular hemorrhage in PCOS induced animals, CES and PCOS+CES treated animals in comparison with that of the SD rats in the control group but, CES and PCOS+CES treated groups present significant lower value than the PCOS induced animals. Conversely, the intervention of CES (PCOS+CES) significantly (p < 0.05) reduced cell degeneration, vascular congestion, interstitial edema, and vascular hemorrhage as compared to PCOS untreated animals. In addition, no significant (p > 0.05) difference in ovarian tissues tissue score between the CES and PCOS+CES treated SD rats. [Figure 7]. (a-d) Ameliorating potential of Cyperus esculentus tubers (Tiger Nut) extract on ovarian tissues score in letrozole-induced PCOS in Sprague-Dawley rats. Data are expressed as the mean ± SEM (n = 10), *: Significant change from the control group at p < 0 05; β: significant change from the PCOS group at p < 0.05, One-Way ANOVA, using Newman-Keuls multiple comparisons test. CES: Cyperus esculentus, PCOS: Polycystic Ovary Syndrome.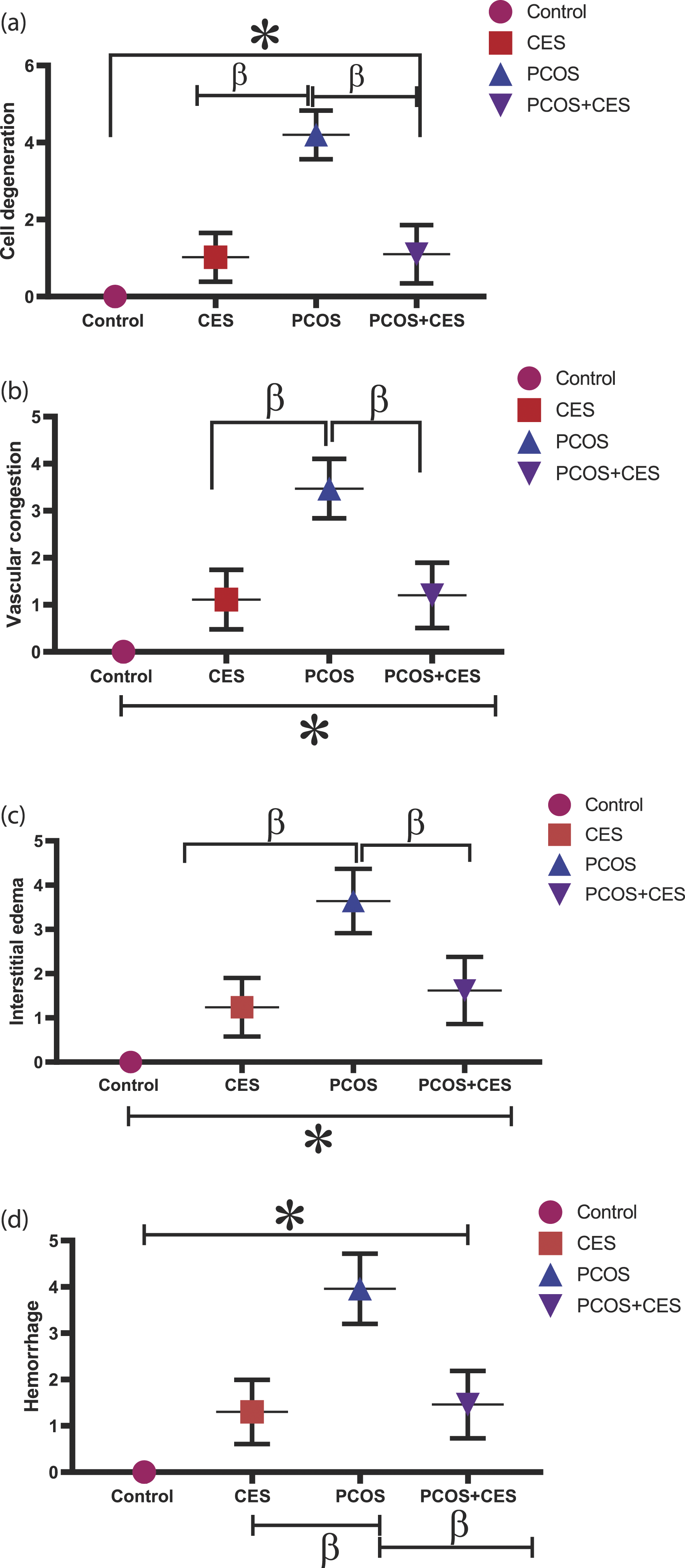
Discussion
Polycystic ovary syndrome (PCOS) is a common complex metabolic and endocrinal disorder that afflicts women of childbearing age. 44 This disorder is characterized by irregular menstrual cycles, weight gain, subfertility, infertility, irregular ovulation, abdominal obesity, and hyperandrogenism.45,46 The estrus cycle is related to alterations in the circulating sex hormone, which controls ovarian function, including follicular maturation and hormonal imbalance. 47
In this study, there was an observed increase in body and ovarian weight in PCOS rats and a prolonged estrus cycle. Increased body and ovarian weight support the report of.48,49,51 The increase in body and ovarian weight is attributed to abdominal fat deposits and thickening of ovarian capsules, and hyperplasia of theca internal cells in the ovary. Also, increased body and ovarian weight may be connected to letrozole’s anabolic characteristics, which have been linked to fat accumulation and the formation of multiple cysts in the ovary, respectively. 52 In addition, rats with letrozole-induced PCOS had higher body weights, which were comparable to those seen in women with PCOS. 49 Furthermore, rats with PCOS had an extended diestrus phase and did not have a regular estrus cycle. 53 which corroborate our findings in this study, since the reduction in the frequency of estrous cycles in PCOS rats resulting in anovulation which occurs at an early stage of pro-estrous to the end of oestrus, 54 leading to female infertility. 55 Treatment of PCOS rats with CES aqueous extract significantly decreases rats’ body and ovarian weight relative to control and regulate the prolonged diestrus phase of the estrus cycle. This CES action could be linked to its mineral and vitamin constituents. Zinc promotes growth, sexual maturation, and reproduction, vitamins C and E increase the development of an egg in the ovary thereby, leading to better ovulation. 56
In the current study, there was observed increase in serum testosterone, FSH, and LH concentration and decrease in progesterone, estradiol, and leptin concentration in PCOS rats when compared with the control, which confirmed the observation of, 57 that increased plasma levels of androgen and low level of progesterone and estradiol were the most consistent hormonal feature of rats with PCOS however, post-intervention of CES extract regulate the hormonal imbalance occasion by PCOS, we can deduce from our observation that dairy supplementation of CES can regulate hormone level since the administration of CES alone maintain the hormone concentration and downregulate the concentration of FSH and LH and elevate the level of progesterone, estradiol, and leptin. Over secretion of testosterone could be linked to the pathogenesis of PCOS and a decrease in high levels of testosterone in PCOS rats by CES extract treatment may have beneficial effects on PCOS disorders. Consumption of CES aqueous extract may be a natural potent therapeutic diary supplement for the treatment of hormonal imbalance related to PCOS. Since hormonal drugs and synthetic substances are the most effective remedies for the treatment of female reproductive dysfunction. Hormonal drugs and chemicals occasion adverse effects, such as uterine bleeding and hyperplasia.44,58 The impact of CES on serum levels of hormones and ovarian dynamic would have contributed to the resumption of the estrous cycle, the hypertrophy of uterine epithelial cells, and finally the restoration of PCOS rat fertility. Indeed, as compared to PCOS-induced rats, the fertility index and gestation rate increased in CES-treated rats, with more than 82% of female rats pregnant and 100% of gestational female rats having viable and healthy fetuses at delivery.
In this study, histopathological observation of the ovarian tissue of PCOS rats revealed a lack of ovulation as evidence of insignificant differences in preantral and antral follicles between the control and experimental group this confirmed the report of,59,60 that single dose of exogenous estradiol valerate injection inhibits ovulation and preantral and antral follicles show no significances significant between the control and estradiol valerate treated animals. Follicle count of the ovarian tissue of PCOS rats revealed a significant decrease in the number of preovulatory follicles and corpora lutea as compared to control this could be a result of a decrease in serum progesterone, estradiol and leptin concentration leading to a lack of hormone for developing follicles to be ovulated at the beginning of folliculogenesis thereby, usher in anovulation or reduced ovulatory rate. It is known that polycystic ovary is a disorder of follicle development, ovarian follicles are arrest at immature stages this may be linked to a lack of positive estrogen feedback to the hypothalamus and pituitary axis. Therefore, the number of atretic follicles significantly increase in PCOS, and the ratio of atretic to healthy follicles also, increase as compared with the control rats. This is a result of the inflow of androgen into the granulosa layer of preantral follicles and binds to the cell receptor and cell death. A higher concentration of androgen may occasion deterioration of follicles by increasing the number of pycnotic granulosa cells and degenerating oocytes.50,61 In addition, edema, cell degeneration, hemorrhage, and vascular congestion in the PCOS group’s ovarian tissues suggest prooxidant and proinflammatory ovarian tissue lesions. In this sense, there is a statistically significant injury in the PCOS group compared to the control and PCOS+CES in cell degeneration, vascular congestion, and interstitial edema, and hemorrhage results obtained in these histopathological examinations. However, treatment of PCOS rats with aqueous extract of CES induced regeneration of the ovarian tissue and disappearance of polycystic follicles resulting in the formation of corpora lutea suggesting the restoration of ovulation and development of follicular cells at a different stage. This pro ovulation potential of CES extract could be related to its bioactive phytochemical component which plays an important role directly on the cellular level resulting in upregulation of progesterone, estradiol, leptin, and antioxidative enzymes and downregulation of lipid peroxidation thereby, leading to ovulation. The COX-1 enzyme is responsible for the synthesis of cytoprotective prostaglandins whereas COX-2 is responsible for the synthesis of pro-inflammatory prostaglandins. 62 Consequently, while COX-1 enzyme inhibition leads to tissue injury, COX-2 inhibition protects the tissue from inflammatory injury. 63 In this study, there was an increase in ovarian COX-1 and decreased COX-2 levels in PCOS-induced rats. However, the COX-1 level was low and the COX-2 level was high in the ovarian PCOS+CES. These suggest the anti-inflammatory effect of CES, the COX-1 and COX-2 levels observed suggest that CES has anti-inflammatory effects, may be protective in the PCOS ovarian tissue.
In the current study, we observed a significant (p < 0.05) decrease in total protein in PCOS rats as compared with the control and treated rats. Treatment with CES extract increases the total protein relative to control, PCOS decreases insulin production and insulin is an anabolic hormone involved in protein synthesis. We can, therefore, deduce from our observation that CES supplementation is capable of regulating insulin secretion thereby, increases the synthesis of protein. The endogenous antioxidant enzymes SOD and CAT are the first line of defense against oxidative stress. The SOD catalyzes the dismutation of superoxide anion radical to H2O2 whereas CAT causes final detoxification of H2O2 to H2O and O2. 64 The activity of these two antioxidant enzymes was significantly reduced in ovarian tissues of PCOS rats while post-treatment with CES extract showed significantly (p < 0.05) increased activity of these enzymes, and this can be attributed to the ability of CES to upregulate and improves the activity of these enzymes. The depletion of these two enzymes in PCOS rats can be associated with high lipid peroxidative products (MDA) and a high amount of H2O2 generated in ovarian tissues. GSH is known to function as a free radical scavenger in the repair of radical-induced cellular damage and also serves as a co-factor for GPx and GST. 65 The decrease in ovarian anti-oxidant enzymes such as GSH concentration in PCOS rats observed in this study could be linked to the production of free radicals resulting in derangement of anti-oxidant levels. A deranged level of anti-oxidant could affect the immune system leading to free radical overload. The intervention of CES extracts significantly increases the concentration of ovarian GSH this is a result of the antioxidant characteristics of bioactive constituents of CES radical load in the ovary of PCOS rats. Thus, CES might serve as a potential therapeutic natural diary supplement for treatment of female infertility, ovarian dysfunction occasion by PCOS. Lipid peroxidation is described as an oxidative deprivation of lipids, initiating a free radical chain reaction of polyunsaturated fatty acids of the fatty acid membrane. 66
Hyperglycemia is an important indicator of PCOS. There was significant (p < 0.05) increase in blood glucose levels in PCOS-induced rats as compared to control. Low glucose tolerance and insulin resistance occasion by letrozole could be linked to elevated androgen concentrations. 48 Treatment with CES significantly (p < 0.05) decreased the glucose levels we could, therefore, deduce that CES reduces glucose resistance by controlling glucose homeostasis, improving insulin secretion, and potentiating the insulin-mediated uptake of glucose.30,48 Dyslipidemia was seen in the diseased control group, with lower HDL-C and higher levels of plasma triglycerides, TBARS, LDL-C, and total cholesterol.67,68 Dyslipidemia is a root cause of coronary artery disease in PCOS patients. 69 PCOS has been linked to dyslipidemia. When compared to the control group, there was a significant reduction in HDL cholesterol but an increase in triglycerides, LDL cholesterol, total cholesterol, and VLDL cholesterol in the untreated PCOS group. However, treatment of PCOS-induced rats with CES significantly improved the parameters these could be attributed to the strong antioxidant capacity of CES. CES protects cellular injury directly by scavenging free radicals and prevents atherosclerosis by inhibiting LDL from oxidation, our data has also verified it. 70 The present study is the first to show that the consumption of CES could serve as a potential diary supplement for the treatment of PCOS, to the best of our knowledge.
Conclusion
Polycystic ovary syndrome causes female infertility through increased oxidative stress in the ovarian tissue, increase testosterone, FSH, and LH, decrease progesterone, estradiol, and leptin. Based on present observation we, therefore, deduce that CES mitigates the release of GnRH, LH, and FSH, amplifying the aromatization of androgens into estrogens, and stimulating estrogens production by adipocytes, responsible for the restoration of estrus cyclicity and ovulation induction. Additionally, the importance of CES could be mediated through its antihyperglycemic and antioxidant capacity.
Footnotes
Acknowledgements
We thank the Department of Anatomy, Federal University of Technology, Akure, Nigeria for providing enabling environment for the research. The contribution of Mr. Boboye of our histology laboratory in histological processing during the different phases of the study is well acknowledged.
Authors’ contributions
Participate in the conception and initial design: SAA,OAA,SOJ. Participate in administration and animals’ management: SAA,AOO. Participate in the analysis and interpretation of results: SAA,OAA,SOJ. Participate in the drafting of the manuscript: SAA,OAA. Histological analysis: SAA,OAA,AOO. Statistical analysis and revising manuscript for intellectual content: SAA,OAA,SOJ,AOO. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
