Abstract
International Agency for Research on Cancer (IARC) classifies agents as to their potential carcinogenicity in humans. Monographs 1–123 categorize 120 agents as group 1 (carcinogenic to humans); 82 agents as group 2A (probably carcinogenic to humans); 311 agents as group 2B (possibly carcinogenic to humans); 499 agents as group 3 (not classifiable as to its carcinogenicity to humans); but only a single agent, caprolactam, a precursor for the manufacture of Nylon 6, fibers, and plastic as group 4 (probably not carcinogenic to humans). The evidence regarding carcinogenic potential to humans for a randomly selected sample of 100 group 3 agents was studied to determine whether the paucity of agents classified as group 4 represented the state of the scientific evidence or a reluctance on the part of IARC to classify a chemical into the lowest risk category. Of the 100 agents evaluated, a significant minority estimated at 24% did not possess structural elements of carcinogenicity, mutagenic activity in the Ames Salmonella assay or activity in a cell transformation assay. The results of this analysis suggest that the lack of agents classified as group 4 represents a reluctance on the part of IARC to place agents into the lowest category of risk. Failure to appropriately place agents into group 4 unnecessarily exhausts valuable staff resources as these low risk compounds require periodic updating of the evidentiary basis of their classification as a group 3 agent. It would be advantageous for both IARC and public health agencies if a clearer differentiation of group 3 and group 4 agents was employed.
Introduction
The International Agency for Research on Cancer (IARC) is under the auspices of the World Health Organization (WHO), which is a branch of the United Nations. 1 IARC classifies agents as group 1 (carcinogenic to humans; 120 agents), group 2A (probably carcinogenic to humans; 82 agents), group 2B (possibly carcinogenic to humans; 311 agents), group 3 (not classifiable as to its carcinogenicity to humans; 499 agents), and group 4 (probably not carcinogenic to humans; 1 agent). 2 Following the controversial classification of the widely used herbicide glyphosate (“Roundup” or glyphosphate) by the IARC Monographs Programme in March 2015 as “probably carcinogenic to humans (Group 2A),” 3 IARC’s classification protocols have come under scrutiny.
In this study, we examine the anomaly of the large number of agents classified as group 3 (not classifiable as to its carcinogenicity to humans), that is, 499 agents, as compared to only one agent classified as group 4 (probably not carcinogenic to humans), that is, caprolactam (Chemical Abstracts Reference Number (CASRN) 105-60-2). 4 Caprolactam’s IUPAC name is azepan-2-one, and its systematic IUPAC name is hexano-6-lactam. 4 Caprolactam is an organic colorless solid, highly soluble in water, with the formula (CH2)5C(O)NH. As a component of Nylon 6 filament, fiber and plastics, approximately five million tons of caprolactam is manufactured each year. 5 Using the classification rationale for caprolactam as a comparator, herein we evaluate the evidentiary basis underlying the placement of agents into either the IARC group 3 or group 4 classifications. In addition, agents currently classified as IARC group 3 that would more appropriately be classified as IARC group 4 are identified.
The relatively high false-positive rates of the major methods used to predict the potential carcinogenicity of chemicals necessarily introduces a significant measure of conservatism into the classification process. In addition, while somewhat smaller than the false-positive rates, the false-negative rates of the major predictive methods introduce additional subjectivity into the process of classifying agents into either IARC group 3 or group 4. Recently, our group analyzed the entire National Toxicology Program (NTP) 2-year rodent cancer study database. 6 –11 Of the 482 chemicals tested by NTP that resulted in the writing of a final report or were described in the two additional Report on Carcinogens (RoC) reports, 331 were negative in the Ames Salmonella mutagenicity test. Of the 331 Ames-negative chemicals, 204 chemicals induced tumors in either rats or mice (62%). If NTP’s contention that no battery of in vitro mutagenicity tests is better than just the Ames Salmonella mutagenicity assay alone in predicting tumorigenicity in rats and mice, 12 –14 then the high level of tumor induction by Ames-negative chemicals is consistent with a significant rate of nongenotoxic cytotoxicity-induced cellular proliferation leading to tumor formation. In addition to the large number of Ames-negative chemicals that nonetheless induce at least one rodent tumor, of the 331 chemicals that induced at least one tumor, 54 chemicals did not possess a structural alert of carcinogenicity resulting in a false-negative rate of 54 of 331 (16.3%). Similarly, OncoLogicTM (computer program developed by the US Environmental Protection Agency that mimics the judgment of human experts by following sets of knowledge-based rules derived from studies of how chemicals cause cancer in animals and humans) predicts that 50 chemicals predict either low or inactive carcinogenic potential for a false-negative rate of 50 of 331 (15.1%) chemicals. If marginal ratings are included, the false-negative rate predicted by OncoLogicTM (Oncologic) is 95 of 331 (28.7%). However, it should be noted that the relatively low false-negative rates predicted by structural alerts of carcinogenicity and Oncologic are depressed by the high false-positive rates of 40% for structural alerts and 43% for Oncologic.
Given that the carcinogen classification is an inexact process, our intent is not to provide a definitive determination as to which agents should be classified as IARC group 3 or group 4. Rather, the purpose of this article is to demonstrate that a reasonable examination of the evidence within relatively wide boundaries of interpretation would result in at least some of the agents currently placed by IARC into group 3 being more logically categorized into group 4.
Methods
Randomized selection of a subset of 100 group 3 agents for evaluation
Table 1 presents the current group classification, cancer classification, and the number of agents in each group. An examination of the evidence regarding potential carcinogenicity of each of the 499 agents classified as IARC group 3 was beyond the logistical capabilities of this analysis. As a surrogate for the entire database of 499 group 3 agents, a randomized subset of 100 group 3 agents was selected for study. First, the CASRN for each of the 499 group 3 agents was written on an index card. The index cards (499) were thoroughly mixed. The first 100 index cards were pulled via blind draw from the intermixed 499 cards. This random subset of 100 IARC group 3 agents constituted the agents evaluated in this analysis.
Agents classified by the IARC monographs, volumes 1–123. 2
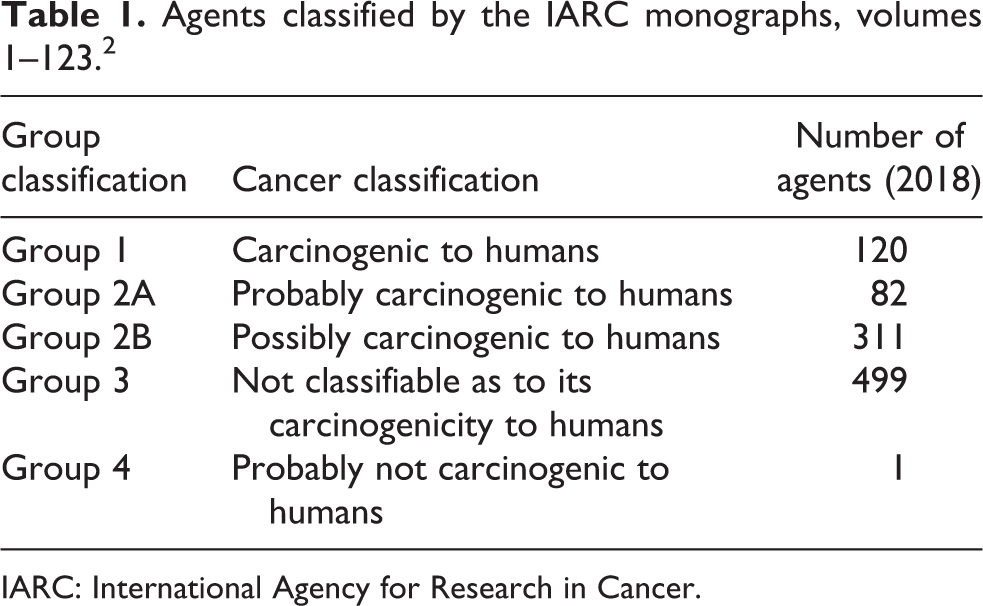
IARC: International Agency for Research in Cancer.
Construction of Online Table 2A to 2C (master data tables)
Online Table 2A has 7 columns. Column one lists the chemical name of the IARC group 3 or 4 agents studied. Column two provides the corresponding CASRN. Columns three through five contain the IARC group; IARC volume; and year, respectively. The information for all 101 agents (100 group 3 agents and 1 group 4 agent) in columns 1–5 were excerpted from the IARC list of classifications, volumes 1–123, found at https://monographs.iarc.fr/list-of-classifications-volumes/ and downloaded in Excel format. 2
Column six titled “Explanation of human tests for carcinogenicity” and column seven titled “Explanation of animal tests for carcinogenicity” were derived from the summary sections of the IARC volume and year listed for each agent. Each cited IARC volume was examined and the information was excerpted, condensed, and incorporated into columns six and seven. Table 2A can be found in the Supplemental Material.
Online Table 2B has eight columns. The first column lists the chemical name of the IARC group 3 or 4 agents studied. The second column denotes whether an “Agent is clearly Ames positive (Y/N),” the third column denotes whether an “Agent was positive in some other genotoxicity assay (Y/N),” and the fourth column delineates whether the “Group 3 or 4 agent is clearly non-genotoxic (Y/N).” The information in these columns (second through fourth) was obtained from the European Union Reference Laboratory, European Centre for the Validation of Alternative Methods (EURL ECVAM) Genotoxicity and Carcinogenicity consolidated database at https://cidportal.jrc.ec.europa.eu/ftp/jrc-opendata/EURL-ECVAM/datasets/genotox/ECVAM_Ames_positives_DB.xls. 15 The information for the fifth (Structural Alerts determination), sixth (Pro-inflammatory designation), seventh (Cell Transformation Assays (CTAs)), and eighth (in vivo Micronucleus (MN) assay) columns was obtained from the chemical listings of the European Molecular Biology Laboratory (ChEMBL or ChEMBLdb) at https://www.ebi.ac.uk/chembl/ 16 ; multiple searches of the internet using the terms “chemical agent” and “pro-inflammatory”; the Environment Directorate, Organisation for Economic Co-operation and Development at http://www.oecd.org/officialdocuments/publicdisplaydocumentpdf/?doclanguage=en&cote=env/jm/mono(2007)18, 17 and the European Union Reference Laboratory, European Centre for the Validation of Alternative Methods (EURL ECVAM) Genotoxicity and Carcinogenicity consolidated database at https://cidportal.jrc.ec.europa.eu/ftp/jrc-opendata/EURL-ECVAM/datasets/genotox/ECVAM_Ames_positives_DB.xls, 15 respectively. Additional information was also obtained via Internet searches using the terms “chemical agent” and “cell transformation assay”. Additional data on Ames results and information on “other genotoxic assays” was obtained from Li et al. (2018) 18 and the Carcinogenic Potential Database at https://toxnet.nlm.nih.gov/cpdb/chemicalsummary.html. 19 Table 2B can be found in the Supplemental Material. Online Table 2C has four columns. The first column lists the chemical name of the IARC group 3 or 4 agents studied. Column two is a “Carcinogenicity Designation” (Table 2). Data to assess the carcinogenicity designation were obtained from Table 2B.
Categorization of carcinogenicity for 100 IARC group 3 agents based on Ames, Structural Alerts, and CTAs data.

IARC: International Agency for Research in Cancer; Sal: Ames test; SA: structural alerts; CTA: cell transformation assay; NGC: non-genotoxic carcinogen; NC: non-carcinogen; U: unknown; GC: genotoxic carcinogen; NT: not tested.
Table 2 lists the categories of carcinogenic potential shown in Online Table 2C. The highest category is genotoxic carcinogen (GC). Consideration as a GC is based on a positive result in the Ames test (Sal), 20 possession of structural alerts (SA), 9 and a positive result in one or more CTAs. 17 If an agent is positive for both the Ames test and structural alerts, but lacks cell transformation data, it is also considered to be a potential GC. 9 An agent was considered a non-genotoxic carcinogen (NGC) if one or both results for Ames test and/or structural alerts were negative (or absent), but the CTA was positive. 21 –23 Benigni et al. 21 have shown a high level of concordance in prediction of animal carcinogenicity from chemicals having negative Ames tests and/or structural alerts but positive CTA results. A non-carcinogen (NC) was designated if the Ames test and structural alerts were negative 24 and the CTAs were negative or not tested. For all other combinations of results for Ames, structural alerts and CTAs the carcinogenicity classification was unknown (U).
Our previous analysis of the NTP database on 2-year rodent cancer studies 9 confirmed the positive correlation between mutagenicity as measured in the Ames Salmonella bacterial assay 20,25,26 and the presence of structural alerts of carcinogenicity 21,27 –29 and development of rodent tumors. In addition to the Ames test and structural alerts, Berwald and Sachs, 30 Creton et al., 31 and the OECD 17 have suggested that the results from CTAs provide additional power toward predicting whether a chemical agent will induce rodent tumors following chronic administration. Several in vitro CTAs have been developed and measure induction of phenotypic alterations characteristic of tumor cells, and cells transformed in vitro have been shown to induce tumors when injected into immunosuppressed experimental animals. 30,32 CTAs mimic some key stages of in vivo multistep carcinogenesis and have been shown to have good concordance with rodent bioassay results, detecting both genotoxic and NGCs. 17,31 Based on the established predictive power of the Ames test (Sal), structural alerts of carcinogenicity (SA), and the CTA, a categorization scheme for carcinogenic potential was developed. Column 14 provides the carcinogenic categorization for each agent.
The third column in Online Table 2C denotes whether information from one or more 2-year tumor study(s) was available on the group 3 agents or group 4 agent. This information was obtained by reviewing the monograph(s) and supplements list for each agent in Online Table 2A. The fourth column of Online Table 2C shows the Group Rating Designation. The criteria and number of group 3 agents that met each criterion are listed in Table 3. Online Table 2C and the references to Online Table 2C can be found in the Supplemental Material.
Criteria and number of the 100 group 3 agents that met the criteria for group 4.

IARC: International Agency for Research in Cancer; Sal: Ames test; SA: structural alerts; CTA: cell transformation assay.
Results
There are 100 group 3 IARC agents and 1 group 4 agent listed in Table 2A. Table 3 has seven columns of data that come directly from the IARC site at https://monographs.iarc.fr/list-of-classifications-volumes/ and were downloaded in Excel format. 2 The seven columns of data provide the IARC group 3 or 4 agent name, the CASRN, the IARC group that the agents belong to, the IARC volumes and supplements where the agent is reviewed, the year that the agent was adopted into its group, an explanation of human tests for carcinogenicity, and an explanation of animal tests for carcinogenicity, respectively. The information in the first four columns is self-explanatory. Column five lists the year each agent was adopted into its group and is anomalous in that a large percentage (46%) of the agents were adopted in 1987 (Figure 1).
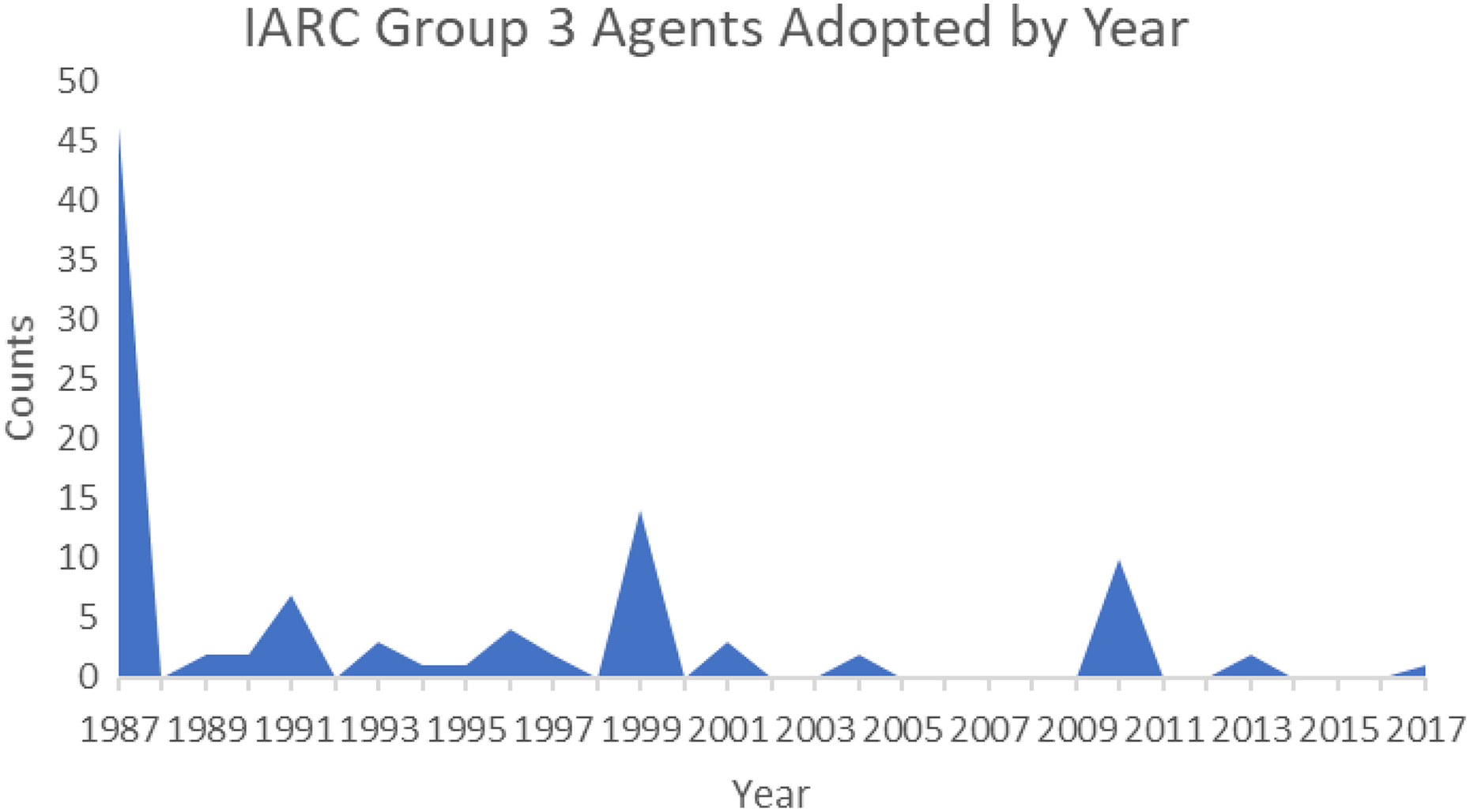
Adoption year of 100 group 3 agents.
In September 1986, an IARC Working group prepared the first version of the categorical grouping classification currently used by IARC (i.e. groups 1, 2A, 2B, 3, and 4). Prior to 1987 (and still in use today), IARC used only an evidence-based classification. The evidence for carcinogenicity from studies in humans (and animals) was assessed by the Working Group to fall into one of five groups:
Sufficient evidence of carcinogenicity indicates that there is a causal relationship between exposure and human cancer.
Limited evidence of carcinogenicity indicates that a causal interpretation is credible, but that alternative explanations, such as chance, bias or confounding, could not adequately be excluded.
Inadequate evidence of carcinogenicity, which applies to both positive and negative evidence, indicates that one of two conditions prevailed: (a) there are few pertinent data or (b) the available studies, while showing evidence of association, do not exclude chance, bias, or confounding.
No evidence of carcinogenicity applies when several adequate studies are available which do not show evidence of carcinogenicity https://monographs.iarc.fr/preamble-to-the-iarc-monographs-amended-january-2006/.
Evidence suggesting lack of carcinogenicity.
Each classification has a very specific meaning for making definitive evaluations. 33
In 1987, Supplement 7 was published by IARC,
34
and all compounds were further classified by groups as follows: Group 1—The agent is carcinogenic to humans. This category is used only when there is sufficient evidence of carcinogenicity in humans. Group 2A—The agent is probably carcinogenic to humans. This category is used when there is limited evidence of carcinogenicity in humans and sufficient evidence of carcinogenicity in experimental animals. Exceptionally, an agent may be classified into this category solely on the basis of limited evidence of carcinogenicity in humans or of sufficient evidence of carcinogenicity in experimental animals strengthened by supporting evidence from other relevant data. Group 2B—The agent is possibly carcinogenic to humans. This category is generally used for agents for which there are limited evidence in humans in the absence of sufficient evidence in experimental animals. It may also be used when there is inadequate evidence of carcinogenicity in humans or when human data are nonexistent, but there is sufficient evidence of carcinogenicity in experimental animals. In some instances, an agent for which there is inadequate evidence or no data in humans but limited evidence of carcinogenicity in experimental animals together with supporting evidence from other relevant data may be placed in this group. Group 3—The agent is not classifiable as to its carcinogenicity to humans. Agents are placed in this category when they do not fall into any other group. Group 4—The agent is probably not carcinogenic to humans. This category is used for agents for which there is evidence suggesting lack of carcinogenicity in humans together with evidence suggesting lack of carcinogenicity in experimental animals. In some circumstances, agents for which there is inadequate evidence of, or no data on, carcinogenicity in humans but evidence suggesting lack of carcinogenicity in experimental animals, consistently and strongly supported by a broad range of other relevant data, may be classified in this group.
34
Table 4 lists the number of agents in 2018 and 1987 by group classification. In Figure 1, 100 of the current 499 group 3 agents were randomly selected. Of these 100 group 3 agents, 46 were adopted in 1987. The large number of adoptions in 1987 seemed unusual. Upon investigation of this anomaly, it was found that 1987 was the first year that the group classification system was used and the backlog of chemicals at IARC was distributed into the five groups.
The number of IARC agents in 2018 and 1987 by group classification.
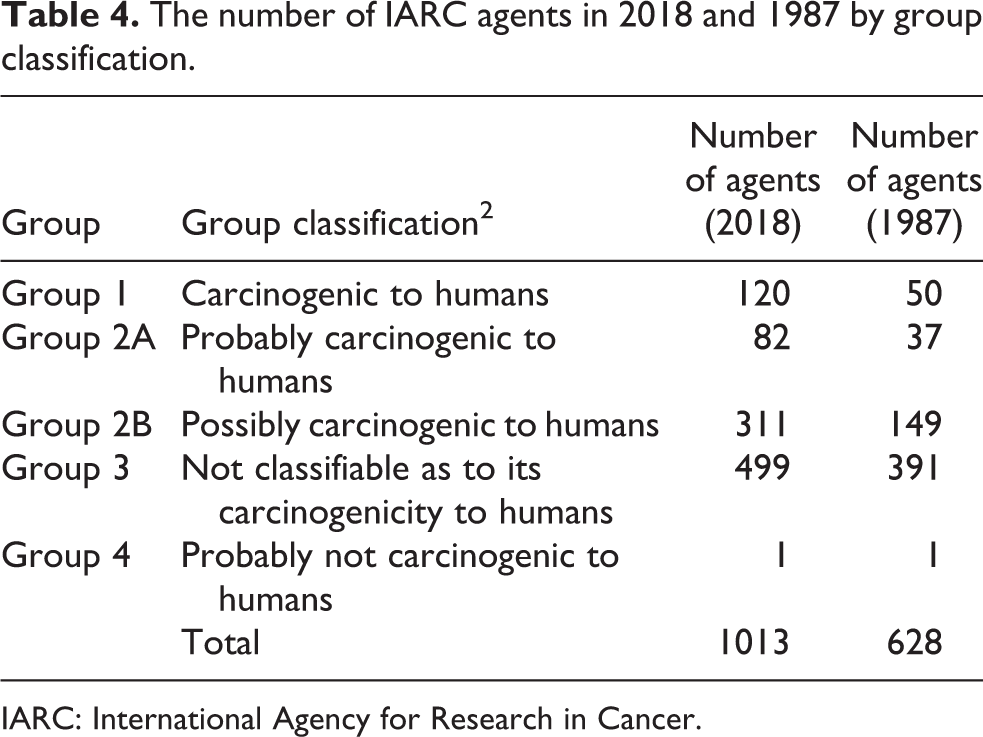
IARC: International Agency for Research in Cancer.
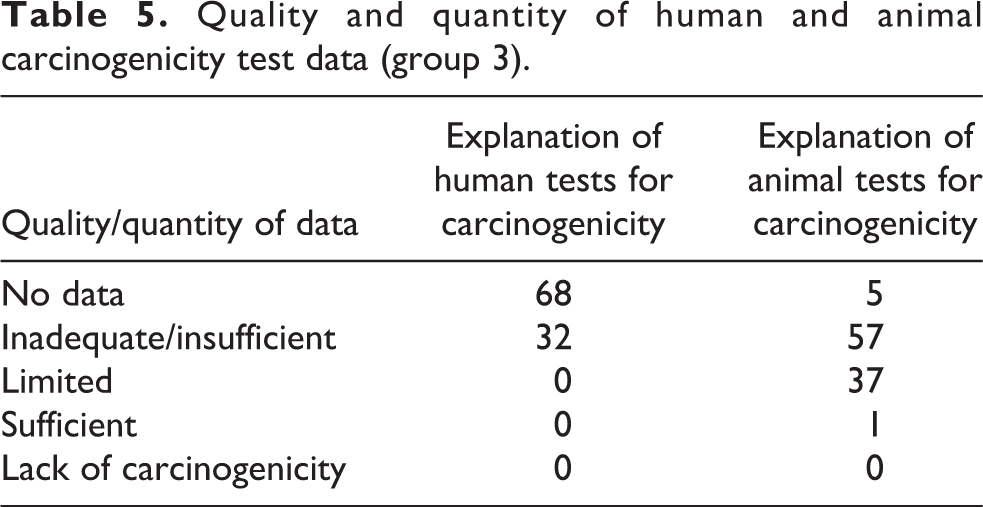
The preamble to the IARC Monographs describes the objective and scope of the program, the scientific principles and procedures used in developing a monograph, the types of evidence considered and the scientific criteria that guide the evaluations. In Part B (Scientific Review and evaluation), Section 6 (Evaluation and rationale) definitions and particular phrases are provided to classify the evidence for human and animal carcinogenicity. Table 5 shows that 68% of the group 3 agents have no human carcinogenicity data and the 32% of the remaining agents have inadequate human carcinogenicity test data. In terms of animal carcinogenicity data, 5% of the group 3 agents have no data, 57% with inadequate/insufficient data, 37% with limited data, and only 1% (a single compound) with sufficient animal carcinogenicity data. For caprolactam (the only group 4 agent), there are no human carcinogenicity data nor evidence suggesting a lack of carcinogenicity in experimental animals.
Quality and quantity of human and animal carcinogenicity test data (group 3).
Online Table 2B lists genotoxicity test results for the 101 agents classified by IARC as group 3 or 4. Table 6 is a summary of the Online Table 2B data. Thirty-seven percent of the 100 group 3 agents had positive Ames test results. Fifty-one percent had negative Ames test results and 12% displayed either equivocal results, or were not tested, or the information was not available. Fifty-six percent of the 100 group 3 agents showed positive test results for some other genotoxic assay (e.g. sister chromatid exchange, chromosomal aberrations, etc.). Twenty percent had negative test results for some other genotoxic assay and 24% had either equivocal results, or were not tested, or the information was not available. Only 15% of the 100 group 3 agents were clearly non-genotoxic. Eighty-four percent of the 100 group 3 agents possessed one or more structural alerts. Fifteen group 3 agents had no structural alerts. Of the 100 group 3 agents, 26% were considered to be pro-inflammatory. Twenty percent of the 100 group 3 agents were not pro-inflammatory, and 54% had either equivocal results, or were not tested, or the information was not available. Nineteen percent of the 100 group 3 agents were positive for one or more CTAs. Twenty-six percent were negative in CTAs. Fifty-five percent had either equivocal results, or were not tested, or the information was not available. Eleven percent of the 100 group 3 agents exhibited a positive result in in vivo micronucleus assays, while 50% gave negative results, 39% of the 100 group 3 agents had either equivocal results, or were not tested, or the information was not available. Online Table 2C lists carcinogenicity designation and group 4 rating designations for the 100 group 3 agents classified by IARC as group 3 or 4 (See Tables 2 and 3). Table 7 is a summary of the Online Table 2C data.
Genotoxicity test results for 100 group 3 agents.
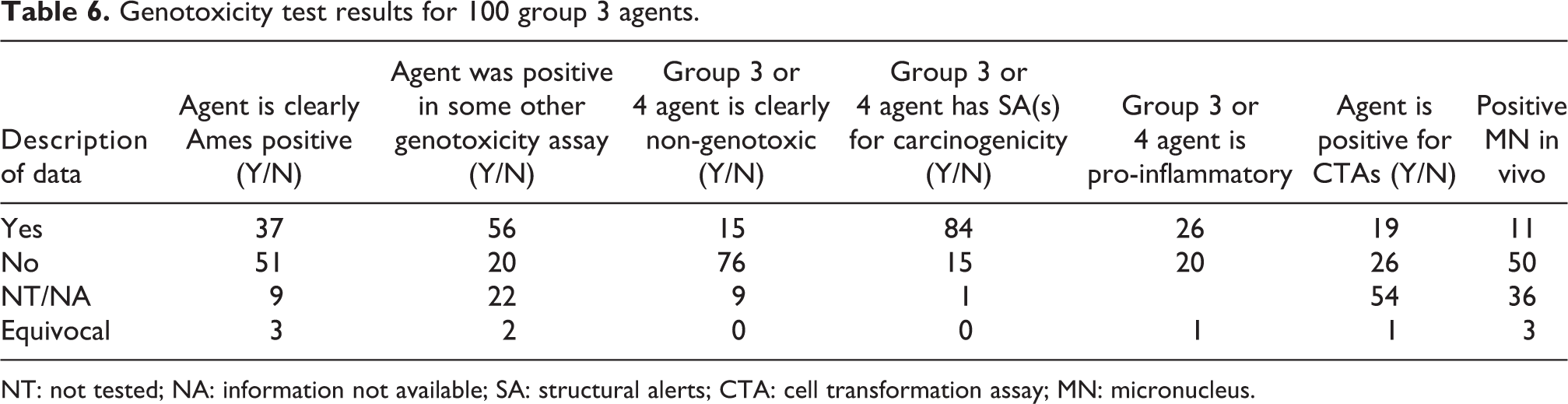
NT: not tested; NA: information not available; SA: structural alerts; CTA: cell transformation assay; MN: micronucleus.
Carcinogenicity designation and number of group 3 agents meeting the criteria of Table 3.

GC: genotoxic carcinogen; NGC = non-genotoxic carcinogen; NC: non-carcinogen; U: unknown.
Discussion
Twenty-four percent of the 100 randomly selected 499 group 3 agents meet the criteria set in Table 3 to be considered as NCs. When applied to all of the relevant agents, this 24% represents about 120 of the 499 group 3 agents. These 24 agents from the 100 randomly selected 499 group 3 agents currently classified as group 3 could reasonably be reclassified as group 4 (probably not carcinogenic to humans). Ten percent of the 100 randomly selected 499 group 3 agents are classified as NGCs. When applied to all of the relevant agents, this 10% represents about 50 agents of the 499 group 3 agents. It is difficult to determine where these 50 agents could best be reclassified as previous investigators have found similar anomalies with non-genotoxic agents being classified from group 1 to group 3. 35,36 Twenty-six percent (26 agents) of the 100 randomly selected 499 group 3 agents are classified as GCs. When applied to all of the relevant agents, this 26% represents about 130 agents of the 499 group 3 agents. These 26 genotoxic agents from the 100 randomly selected 499 group 3 agents currently classified as group 3 could conceivably be reclassified across a wide spectrum of categories depending on the particular exposure scenarios, and presence or absence of clinical case reports or epidemiological data. Forty percent of the 100 randomly selected 499 group 3 agents have an unknown classification due to a lack of data. When applied to all of the relevant agents, this 40% represents about 200 agents of the 499 group 3 agents. While caprolactam meets the criteria in Table 3, each of the other 24 group 3 agents examined also met the same criteria. When applied to the entire database, these 24 group 3 agents would extrapolate to about 120 of the 499 group 3 agents being reclassified as group 4 agents.
IARC uses very specific wording for describing definitive evaluations.
33
However, IARC says that it does not measure risk, that is, the “likelihood that cancer will occur as a result of exposure to the agent” but rather classifies agents based on “the weight of the evidence as to whether an agent is capable of causing cancer (technically called ‘hazard’).”
37
IARC states that the classification (group 1–group 4) indicates the strength of the evidence that a substance or agent causes cancer. The cancer risk associated with substances or agents assigned the same classification may be very different, depending on factors such as the type and extent of exposure and the strength of the effect of the agent.
37,38
IARC selects agents for review based on two criteria: evidence of human exposure and some evidence or suspicion of carcinogenicity. Ad hoc advisory groups generally meet every 5 years to make recommendations as to which agents should be reviewed or reevaluated in the Monographs series. IARC may schedule other current agents for review as it becomes aware of new scientific information or as national health agencies identify an urgent public health need related to cancer. There is no strict rule as to when an agent is reevaluated.
The unique case of group 4 agent caprolactam is instructive. Over a 20-year period caprolactam was reviewed (initially) and then reevaluated an additional three times. IARC published three volumes and one supplement citing caprolactam. 43 –46 The first evaluation of caprolactam was in 1979. 47 At that point, no human or animal studies had been completed on caprolactam for the Working Group to evaluate. However, there was a footnote that described a future planned oral carcinogenicity study in rats and mice. 48 The only carcinogenicity study on caprolactam was conducted in 1982 by the NTP. 49 The 1982 study was deemed to be adequate by the NTP. The results reported by the NTP were as follows: “Under the conditions of this bioassay, caprolactam was not carcinogenic for F344 rats or B6C3F1 mice.” In 1986, in IARC vol. 39, caprolactam was reevaluated. 44 Conclusions from the Working Group in 1986 were, “There is no evidence for the carcinogenicity of caprolactam to experimental animals. No data on humans are available. In 1987, IARC supplement 7 was published and contained the following excerpt: “Evidence for carcinogenicity to animals (evidence suggesting lack of carcinogenicity). 45 Caprolactam was tested adequately by oral administration in the diet of mice and rats. There was no increase in tumour incidence over that in controls.” In 1987, no new data were available to the Working Group. In 1999, IARC vol. 71 was published and contained the following information on the same animal carcinogenicity study conducted and published in 1982: No epidemiological data relevant to the carcinogenicity of caprolactam were available. There is evidence suggesting a lack of carcinogenicity of caprolactam in experimental animals. 46 Based on the data in Online Table 2, the 100 group 3 agents were on-average reviewed and then reevaluated once every 13 years (range was 26 years). None of the group 3 agents were evaluated more than three times. The only group 4 agent was reviewed initially and then reevaluated three additional times over a 20-year period.
Conclusions
When a chemical is shipped from a manufacturer in the United States to a downstream user, the shipment must contain a Safety Data Sheet (SDS). 50 While not a legally mandated section, the SDS for most chemicals and especially for toxic chemicals, contains “Section 14: Transport Information.” One of the elements of an SDS Section 14 is the “packing group number,” assigned based on the degree of hazard. 50 Corrosivity is a very common contributor to selection of packing group numbers of concern, that is, Packing group I: high danger or Packing group II: medium danger, in contrast with Packing group III: low danger. 51 For well-studied chemicals, the degree of corrosivity has already been established and assignment of the chemical to one of the three possible packing groups has already been completed. However, for experimental chemicals possessing only knowledge of chemical structure, behavior of other chemicals in the same structural class, or having limited toxicology data, a staff toxicologist is usually called upon to assist the shipping department in selecting the appropriate packing group. The question readily arises, “Why not classify any chemical not possessing a formal test of corrosivity as a ‘Packing Group I’ chemical thereby maximizing worker safety in both the shipping chain and at the downstream user?” The reason this cannot be done is that arbitrary assignment to Packing group I necessitates use of expensive shipping containers, 52 limits the potential modes of transportation allowed to ship the chemical, 53 affects the types and places for warehousing the chemical, 54 and activates regulations promulgated by local emergency responders based on optimizing safety for hazmat personnel and firefighters. 55 In the event of a fire or flood at a chemical storage facility, inappropriate over-classification of stored chemicals as Packing group I could waste valuable time and misallocate hazmat and firefighter efforts toward ameliorating non-existent risks. For well-studied chemicals, individuals must prepare the package in the same manner in which it was tested and certified. 56
The simple case of the burden placed on a toxicologist to make a data-based decision, in the absence of a definitive data set, for the hazard classification and shipping of a small quantity of a research chemical demonstrates a more generalizable principle extensively studied by economists, scientists, and business experts for many decades, that is, the tendency toward risk aversion among decision makers within an organization tasked with the protection of human health. Luminaries who have analyzed and described risk aversion in decision-making processes include the Noble Laureate scientist George Hitchings and Nobel Laureate economist Milton Friedman regarding the US Food and Drug Administration and Nobel Laureate economist Friedrich Hayek on governmental bureaucracy in general. 57 –61 Agents that have achieved the status of becoming candidates for IARC classification have usually filled an important market niche by outcompeting other agents on their potential usefulness, price, safety, ease of manufacture, handling, shipping, or logistics of disposal. (One of the factors in the decision to prioritize a chemical for classification is the potential for exposure of workers, the general population, and the environment. Chemicals, especially high-volume chemicals in general commerce, are more likely to be associated with the above noted exposures.) Replacing such an important chemical due to overly conservative not scientifically based carcinogen classification with another chemical could be disadvantageous economically and also from a human and environmental health perspective.
It would be possible to perform margin of error calculations to evaluate the validity of IARC classifications of chemicals. 62,63 Additionally, econometric studies could be performed to determine the “costs” of IARC category 3 versus 4 miss-classification. IARC has performed similar studies in the past. 64
As noted previously, IARC has classified 499 agents as group 3 (not classifiable as to its carcinogenicity to humans) and only a single agent as group 4 (probably not carcinogenic to humans). The results of this analysis demonstrating that a significant minority of agents classified as group 3 do not possess evidence of carcinogenic potential suggests that the absence of agents classified as group 4 is due at least in part to a reluctance on the part of IARC committee members to designate agents as “Probably not carcinogenic to humans.”
Supplemental material
Supplemental Material, IARC_Supplemental_Materials - An approximated one-quarter of IARC Group 3 (unclassifiable) chemicals fit more appropriately into IARC Group 4 (probably not carcinogenic)
Supplemental Material, IARC_Supplemental_Materials for An approximated one-quarter of IARC Group 3 (unclassifiable) chemicals fit more appropriately into IARC Group 4 (probably not carcinogenic) by Carr J Smith and Thomas A Perfetti in Toxicology Research and Application
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
