Abstract
In a subchronic (13-week) inhalation toxicity study with a terminal sacrifice (after 13 weeks inhalation) and several recovery period sacrifices (13, 26, 39, and 52 weeks), the effects of AEROSIL® 200 (pyrogenic synthetic amorphous silica (SAS)), AEROSIL® R 974 (surface-treated pyrogenic SAS), and SIPERNAT® 22 S (precipitated SAS) were tested in rats at multiple dose levels. The aforementioned materials are all SAS products. A comparative group of animals was exposed to quartz dust. This study attempts to reexamine the lung tissues originally evaluated in a study published by Reuzel et al. using the current standards. To reach a high level of credibility, the results of the reevaluation were subsequently examined by a pathology working group (PWG). In particular, the reevaluating pathologist and the PWG concluded that, even though quartz (crystalline silica) persisted, induced alterations in the lungs following 13 weeks of exposure to amorphous silicas were reversible following 52 weeks of recovery. A long-term adversity has not been established with SAS products. However, quartz dust damages lungs significantly by causing pulmonary fibrosis.
Keywords
Introduction
To characterize the toxicity of amorphous silica, including pyrogenic amorphous silica (AEROSIL® 200), surface-treated pyrogenic amorphous silica (AEROSIL® R 974), and precipitated amorphous silica (SIPERNAT® 22 S), a subchronic (13-week) inhalation toxicity study had been conducted in 1987. 1,2 Different test items were administered to rats over a period of 13 weeks, followed by recovery periods of 13, 26, and 52 weeks. The tested products were compared with effects caused by inhaled quartz (positive control). In the original study, rats were treated at multiple dose levels. The study was performed under good laboratory practice (GLP) conditions. The results were published in Reuzel et al. 1 There were however differences in the outcome of this study compared to the published data.
Synthetic amorphous silica (SAS) has been characterized by the United Nation’s Organization for Economic Co-operation and Development (OECD) for the hazardous properties of high production volume chemicals. In 2004, the “Screening Information Data Set” for SAS was released. Regarding human health, it was concluded that “The chemicals in this category are currently of low priority for further work.” It was summarized that SASs are rapidly eliminated from the lung tissue with no disproportionate disposition occurring in the mediastinal lymph nodes. Furthermore, intestinal absorption of SAS was considered to be insignificant in animals and humans. Beside inflammatory reactions, toxicity was limited. Moreover, …data collected from industrial hygiene surveillance over the last 50 years do not indicate any potential for skin sensitization. Given the inherent physico-chemical properties and ubiquitous nature of this class of compounds, there is no structural alert to indicate a sensitizing potential…
3
…Silica, amorphous, fumed (crystalline free) has a demonstrated lack of toxicity… …Silica, amorphous, fumed (crystalline free) is not classifiable, as to its carcinogenicity however, given its amorphous nature, it is not expected to pose a carcinogenic risk. Silicas are considered to be inert when ingested, and due to the high molecular weight it is unlikely to be absorbed through the skin. There should be no concerns for human health, whether the exposure is acute, subchronic, or chronic by any route…
4
The authors of the original study reported that …focal interstitial fibrosis, seen as amorphous eosinophilic, collagen-containing thickenings of the septa, was first observed 13 weeks after exposure in all exposed groups. During the subsequent post-exposure period, this condition disappeared completely in rats exposed to AEROSIL® R 974 or SIPERNAT® 22 S, but became more severe in rats exposed to 30 mg AEROSIL® 200 or quartz…
2
All test materials induced increases in lung weight, and pulmonary lesions such as accumulation of alveolar macrophages, inflammation, alveolar bronchiolization and fibrosis. In addition, rats exposed to AEROSIL® 200, AEROSIL® R 974, or quartz developed granulomatous lesions. Silicosis was observed only in quartz-exposed animals. At the end of the exposure period, AEROSIL® 200 and quartz had induced the most severe changes. Quartz dust was hardly cleared from the lungs and the changes in the lungs progressed during the posttreatment period, and eventually resulted in lesions resembling silicotic nodules.
Despite these findings other authors have reported that inhaled amorphous silica causes …limited and largely reversible cytotoxic and possibly fibrogenic effects… …cancer outcomes are inconclusive and for the most part negative. Epidemiologic investigations to date for any potential cancer risk are not informative because the effects of crystalline and amorphous silicas have not been separated…
5
The findings and conclusions described have been confirmed in a large number of subacute, subchronic, and chronic inhalation studies published in the literature using rats, guinea pigs, rabbits, and monkeys exposed to concentrations ranging from 0.5 mg SiO2/m3 to 150 mg SiO2/m3, performed with pyrogenic, precipitated, sol, and gel SAS, with and without surface modification. 6 –8 In exposed workers, there is no evidence for a fibrogenic effect of SAS to the human lung. 9
Hence, the question arises, as to whether fibrosis was induced after exposure to SAS and whether effects were fully reversible.
To answer these questions, a reevaluation was performed on available lung sections from the following groups (end of treatment after 13 weeks) of the original study: males only of Groups A–F, and both sexes of 13- and 52-week recovery animals (Groups A–G). Subsequently, a pathology working group (PWG) was formed to review the results of the reevaluation.
Material and methods
Originally, 10 specific pathogen free (SPF)-bred Wistar rats (Cpb: WU Wistar Random) per sex/group, at an age of 4 weeks at start of the acclimatization, were allocated per scheduled necropsy. The animals were treated with AEROSIL® 200 (pyrogenic SAS) at doses of 1.3, 5.9, or 31 mg/m3 (Groups B, C, and D, respectively), with AEROSIL® R 974 (surface-treated pyrogenic SAS) at 34.7 mg/m3 (Group E), and with SIPERNAT® 22 S (precipitated SAS) at 34.9 m/kg3 (Group F). Quartz (positive control) was administered as positive control at a concentration of 58.5 mg/m3 (Group G). A sham group (Group A) served as negative control and received air only. For the reevaluation, there were only slides from males available at the scheduled 13-week sacrifice, and sections from both sexes after 13-week recovery and after 52-week recovery. The study design schedule is presented in Table 1. The standard animal housing and care conditions, necropsy, organ preservation methods, and histotechnique procedures have been described in detail. 2
Study design, including treatment groups, test item, and dose and number of animal assigned per group.a
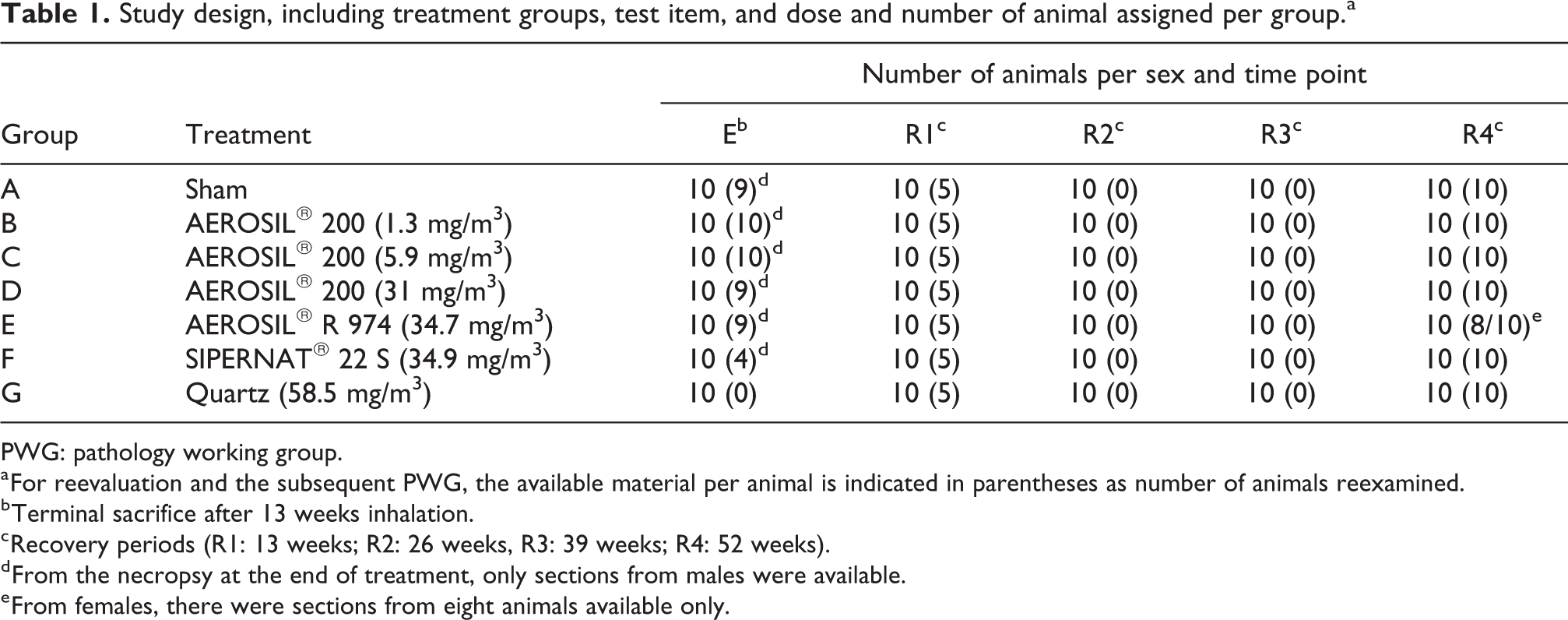
PWG: pathology working group.
a For reevaluation and the subsequent PWG, the available material per animal is indicated in parentheses as number of animals reexamined.
b Terminal sacrifice after 13 weeks inhalation.
c Recovery periods (R1: 13 weeks; R2: 26 weeks, R3: 39 weeks; R4: 52 weeks).
d From the necropsy at the end of treatment, only sections from males were available.
e From females, there were sections from eight animals available only.
The reevaluation was performed at AnaPath GmbH, Switzerland, by Dr Klaus Weber using the PathData system Version 6.2e2 (PDS Pathology Data Systems AG, Switzerland). For this reason, all previously hematoxylin and eosin (HE)–stained slides of the toxicity study (now more than 28 years old) were de-coverslipped and re-stained by HE owing to the fading of the original staining. The slides were coverslipped again and underwent histological evaluation. The evaluation on the re-stained sections of the lungs was performed according to current standards and under GLP conditions.
Diagnostic criteria and terminology used throughout the study were based upon recognized texts and current scientific literature, that is, according to International Nomenclature and Harmonization of Diagnostic Criteria (INHAND) nomenclatures. 10 Additional terminologies were applied to some of the induced findings. Details are provided in Table 2. All nonneoplastic findings were graded on a severity scale from 1 to 5 (minimal, slight, moderate, marked, and severe).
Diagnostic criteria used during reevaluation.

BALT: bronchiolar associated lymphoid tissue.
For group comparison, mean grades on lesions were calculated by the PathData System as sum of grades in groups divided by the number of affected individuals. A Fisher’s exact test was performed on all data. The data were evaluated at a significance level of p < 0.05 and p < 0.1. The trend test for nonneoplastic lesions (Armitage test) was applied comparing the findings after the 52-week recovery sacrifice, between controls and AEROSIL® 200 groups, controls and AEROSIL® R 974 groups, and controls and SIPERNAT® 22 S groups.
Following the reevaluation, it was decided to assemble a PWG. This PWG evaluation was performed as published elsewhere. 11,12 The PWG was chaired by Jerry F Hardisty, DVM, DACVP (EPL Inc., NC, USA) who organized and presented the material to the PWG participants. The PWG members included Chirukandath Gopinath, BVSc, PhD, FRC Path; Ernest E McConnell, DVM, DACVP, DABT; Günter Oberdörster, DVM, PhD, Professor Emeritus; Martin Rosenbruch, Dr med. vet., PhD Professor, FTA Pathol. (Bayer Pharma AG, Wuppertal, Germany); and Klaus Weber, DVM, MS Biol, DJSTP (AnaPath GmbH, Switzerland). Nils Krueger, Dr med. vet., FTA Pathol. (Evonik Resource Efficiency GmbH, Germany), was present as an observer.
The PWG focused on a specific question, that is, presence or absence of fibrosis in the lungs from animals exposed to SAS, as well as the reversibility of induced findings (increased alveolar macrophages/macrophage aggregates, interstitial inflammation, granulomatous inflammation, type II pneumocyte hyperplasia, alveolar/bronchiolar hyperplasia, foreign material). Since there were only a few reported differences between sexes, the PWG examined coded HE-stained slides only from male rats without knowledge of treatment group or previous diagnosis. There were up to nine sections per lung (animal) available for examination (see Peer Review), and of these just only one section, containing tissue from the left lung, was selected for the PWG review. During the review process of the PWG, additional sections were selected for discussion when necessary. The PWG examined slides containing sections of left lobe of the lung from the first five male rats in each exposure group at each sacrifice interval. Males were selected because there were only minor differences between both sexes, and the main question was targeted to persistent fibrosis. In addition, the left lobe of the lung of the remaining male control rats from the 13-week sacrifice and the 52-week recovery sacrifice was made available in an open manner to provide a basis for comparison. Each participant recorded his diagnoses and comments on worksheets that were prepared by the PWG chairperson. The PWG examined the lung slides only for possibly induced findings and did not consider other findings, which may have been present, since they were not related to the scope of the PWG.
The PWG chairperson and the observer were nonvoting members during the PWG review. Following the initial introductions, the diagnostic criteria and nomenclature proposed by the INHAND project (International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice) for Proliferative and Nonproliferative Lesions of the Rat and Mouse Respiratory Tract 10 were used as a guideline by the PWG while examining lung slides. The PWG was performed according to the procedures recommended in the EPA Pesticide Regulation (PR) Notice 94-5: “Requests for Reconsiderations of Carcinogenicity Peer Review Decisions Based on Changes in Pathology Diagnoses” 13 and in compliance with GLP requirements. The previously performed reexamination at AnaPath, Switzerland, was used as the basis for the PWG report.
Results
During the reevaluation of the original sections, there were a number of degenerative and inflammatory lesions recorded in all test item treated as well as in the control groups. The treatment of animals by daily inhalation of AEROSIL® 200 at a concentration of 1.3–31 mg/m3, AEROSIL® R 974 (34.7 mg/m3), and SIPERNAT® 22 S (34.9 mg/m3) did not cause any irreversible changes in the lungs as seen over the complete recovery period.
Fibrosis/fibrogenesis
Fibrosis was not observed after the 13-week main test period, and after the 13-week recovery period. At the latter sacrifice time point, in two animals of the high-dose AEROSIL® 200 group, a minimal fibrogenesis (not statistically significant in the Fisher’s test) was noted. After the 52-week recovery period, a minimal focal fibrosis was detected only in one animal from the control group, one animal from each of the low- and mid-dose AEROSIL® 200–treated animals, and in three animals of the high-dose AEROSIL® 200–treated animals. No fibrosis was present in AEROSIL® R 974 and SIPERNAT® 22 S–treated groups. Hence, it appears that aside from some reactive changes including the presence of alveolar macrophages and macrophage aggregations in high-dose AEROSIL® 200 animals, there was no significant difference compared to the control animals.
In contrast, a high level of multifocal fibrosis was recorded in quartz-treated animals after the 52-week recovery period. In addition, the pleura was affected showing a multifocal pleural mesothelial hyperplasia in a high portion of the animals.
Thirteen-week terminal sacrifice
At the 13-week sacrifice (Table 3), the lesions did not differ in sections evaluated from males only, between all test item–treated groups. In control animals, there was interstitial inflammation in controls which was lower in incidence than in AEROSIL® 200-treated animals (Figure 1).
Incidence and mean severity of main findings in lungs, 13-week sacrifice, males.a
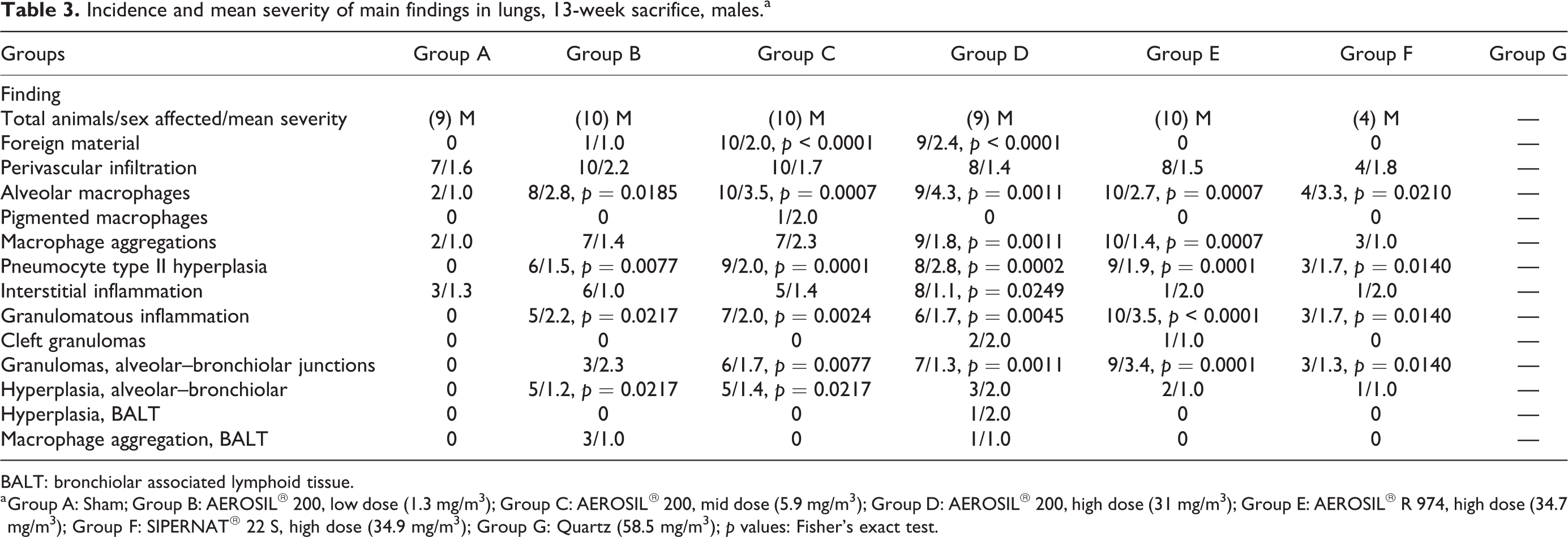
BALT: bronchiolar associated lymphoid tissue.
a Group A: Sham; Group B: AEROSIL® 200, low dose (1.3 mg/m3); Group C: AEROSIL® 200, mid dose (5.9 mg/m3); Group D: AEROSIL® 200, high dose (31 mg/m3); Group E: AEROSIL® R 974, high dose (34.7 mg/m3); Group F: SIPERNAT® 22 S, high dose (34.9 mg/m3); Group G: Quartz (58.5 mg/m3); p values: Fisher’s exact test.
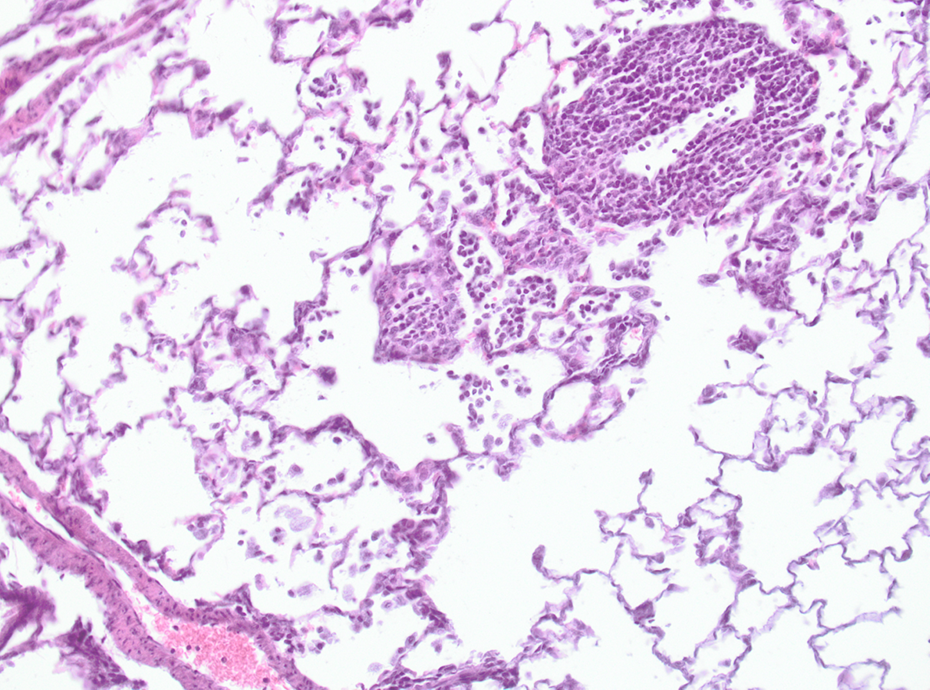
Control rat after 13 weeks of treatment. Slight perivascular and interstitial inflammation; HE, ×100. HE: hematoxylin and eosin.
Induced lesions consisted mainly of the presence of foreign material appearing in a dose-dependent manner in AEROSIL® 200-treated groups; increasing severity of alveolar macrophages; macrophage aggregations (low severity in the SIPERNAT® 22 S-treated group); reactive pneumocyte macrophage type II hyperplasia; and granulomatous inflammation and the presence of alveolar/bronchiolar junctional granulomas (highest incidence and severity in the AEROSIL® R 974-treated group), and cleft granulomas.
The incidence and severity between the AEROSIL® 200 high-dose and AEROSIL® R 974 and SIPERNAT® 22 S–treated groups was similar, except for cleft granulomas and a lower incidence of junctional granulomas (lowest incidence in SIPERNAT® 22 S-treated animals, highest incidence in AEROSIL® R 974-treated animals). Alveolar/bronchiolar epithelial hyperplasia was not dose-related, but appeared in all test item–treated groups. Furthermore, there were isolated cases of hyperplasia and of reactive macrophages in the bronchiolar associated lymphoid tissue (BALT) in AEROSIL® 200-treated animals only (Figures 2 to 7).
Low-dose AEROSIL® 200–treated animal after 13 weeks of treatment. Alveolar macrophages, perivascular and interstitial inflammation; HE, ×40. HE: hematoxylin and eosin.
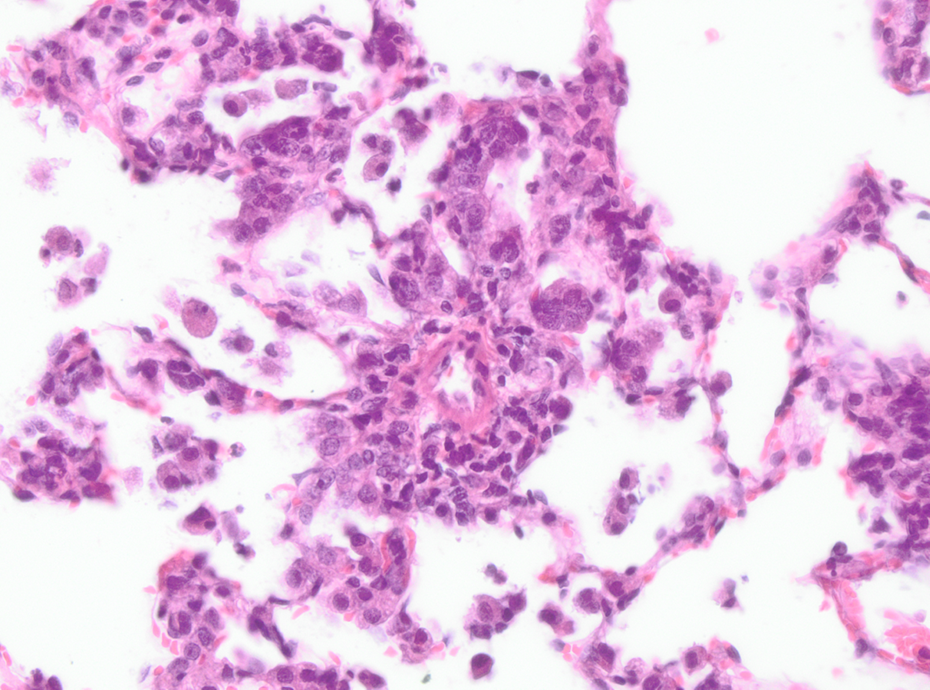
Low-dose AEROSIL® 200–treated animal after 13 weeks of treatment. Alveolar macrophages, macrophage aggregation, perivascular and interstitial inflammation; HE, ×200. HE: hematoxylin and eosin.
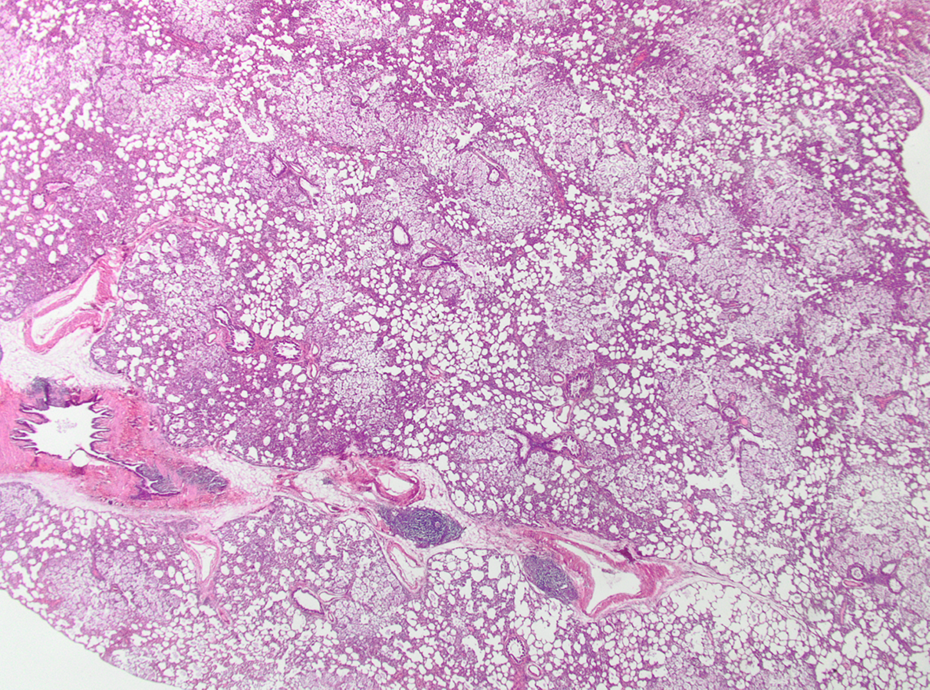
Mid-dose AEROSIL® 200–treated animal after 13 weeks of treatment. Intra-alveolar foreign material; HE, ×12.5. HE: hematoxylin and eosin.
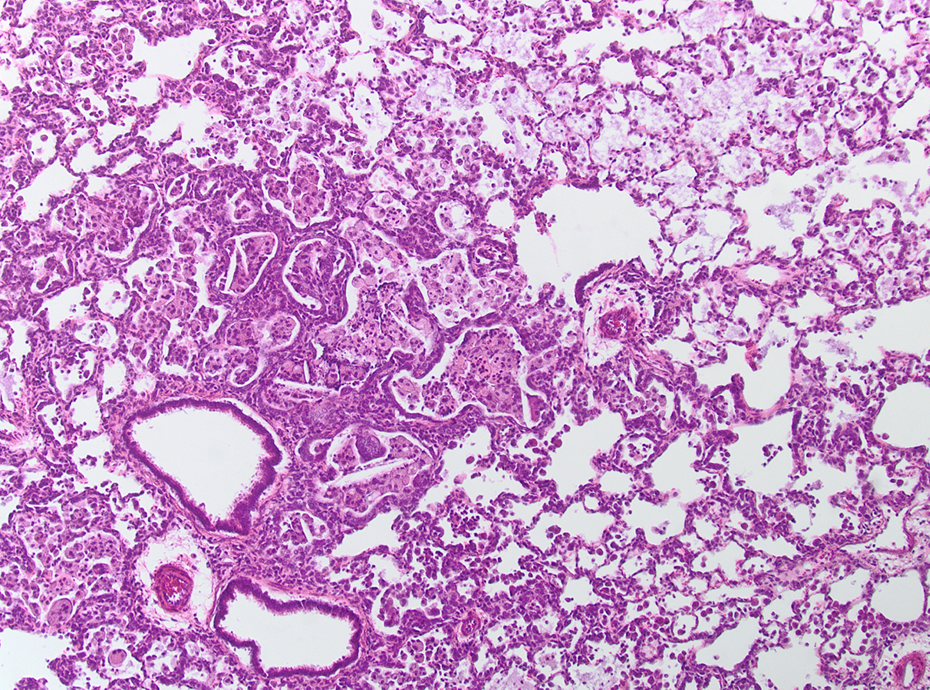
High-dose AEROSIL® 200–treated animal after 13 weeks of treatment. Intra-alveolar foreign material, alveolar macrophages, alveolar–bronchiolar hyperplasia, and interstitial inflammation; HE, ×100. HE: hematoxylin and eosin.
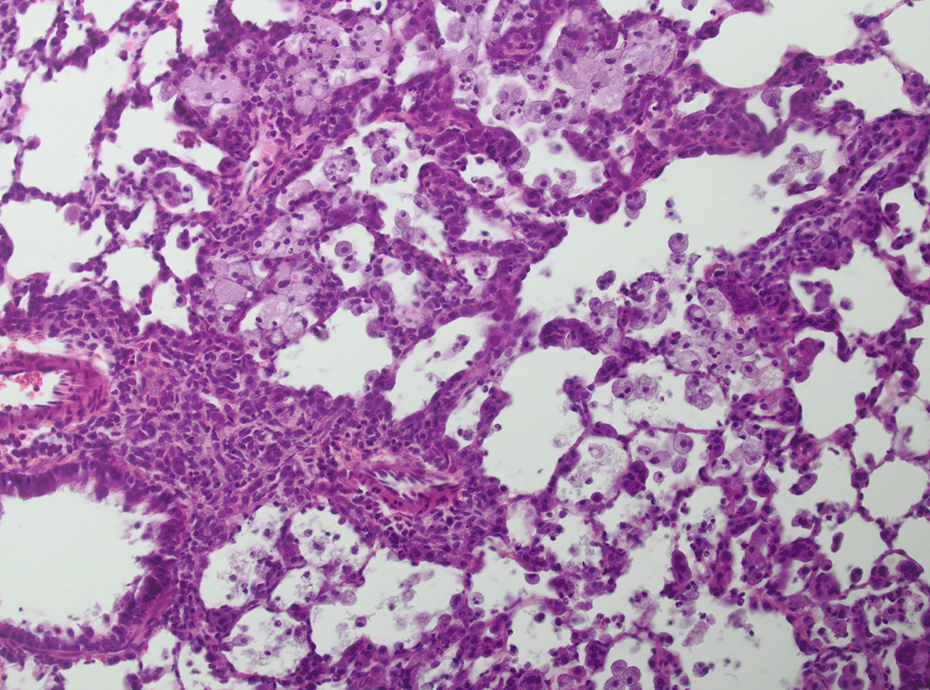
AEROSIL® R 974-treated animal after 13 weeks of treatment. Alveolar macrophages, type II pneumocyte hyperplasia, and granulomatous inflammation; HE, ×200. HE: hematoxylin and eosin.
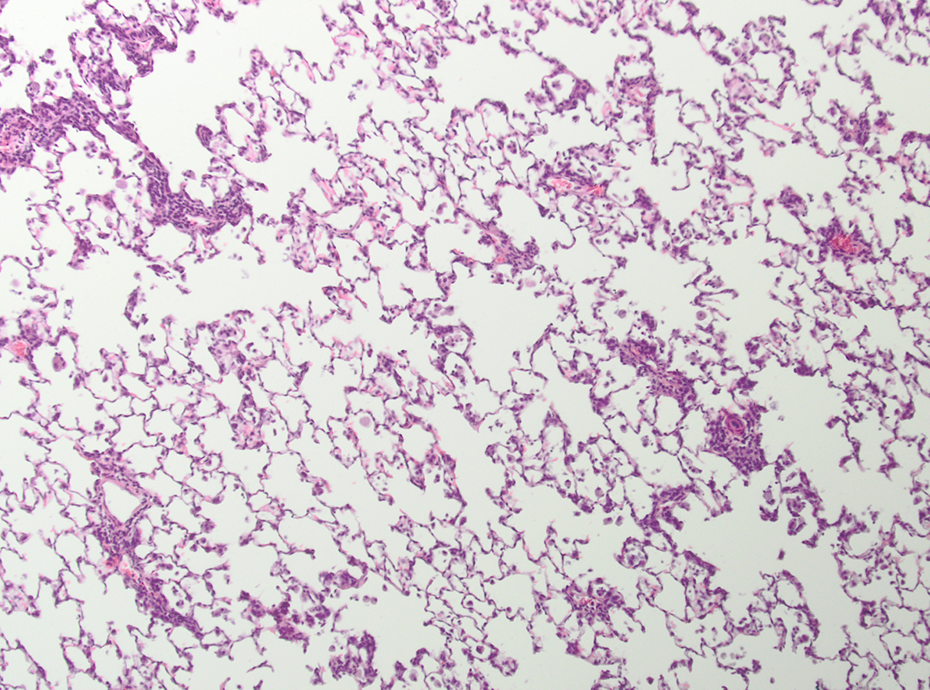
SIPERNAT® 22 S-treated animals after 13 weeks of treatment. Perivascular inflammation, alveolar macrophages, and granulomatous inflammation; HE, ×100. HE: hematoxylin and eosin.
Thirteen-week recovery sacrifice
In the 13-week recovery animals (Tables 4 and 5), all findings noted after 13 weeks of treatment were still present although mostly appearing at lower severities. From this sacrifice onward, there were also sections from quartz-treated animals available.
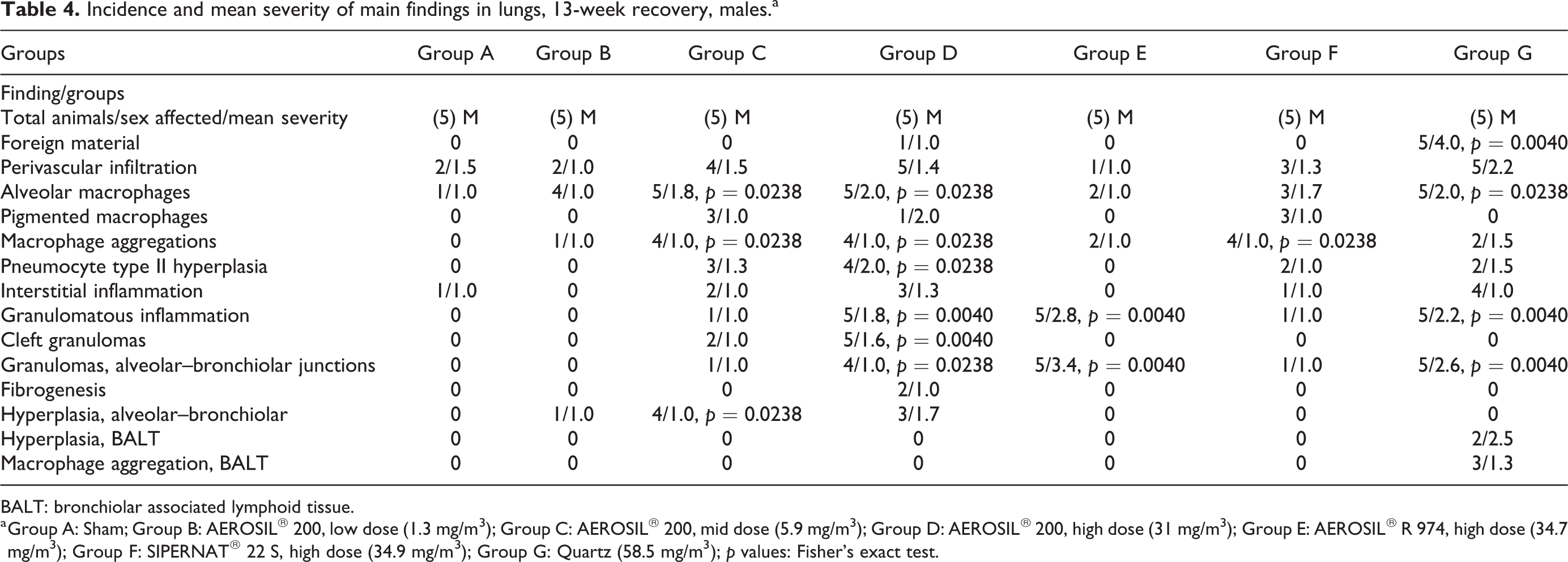
Incidence and mean severity of main findings in lungs, 13-week recovery, males.a
BALT: bronchiolar associated lymphoid tissue.
a Group A: Sham; Group B: AEROSIL® 200, low dose (1.3 mg/m3); Group C: AEROSIL® 200, mid dose (5.9 mg/m3); Group D: AEROSIL® 200, high dose (31 mg/m3); Group E: AEROSIL® R 974, high dose (34.7 mg/m3); Group F: SIPERNAT® 22 S, high dose (34.9 mg/m3); Group G: Quartz (58.5 mg/m3); p values: Fisher’s exact test.
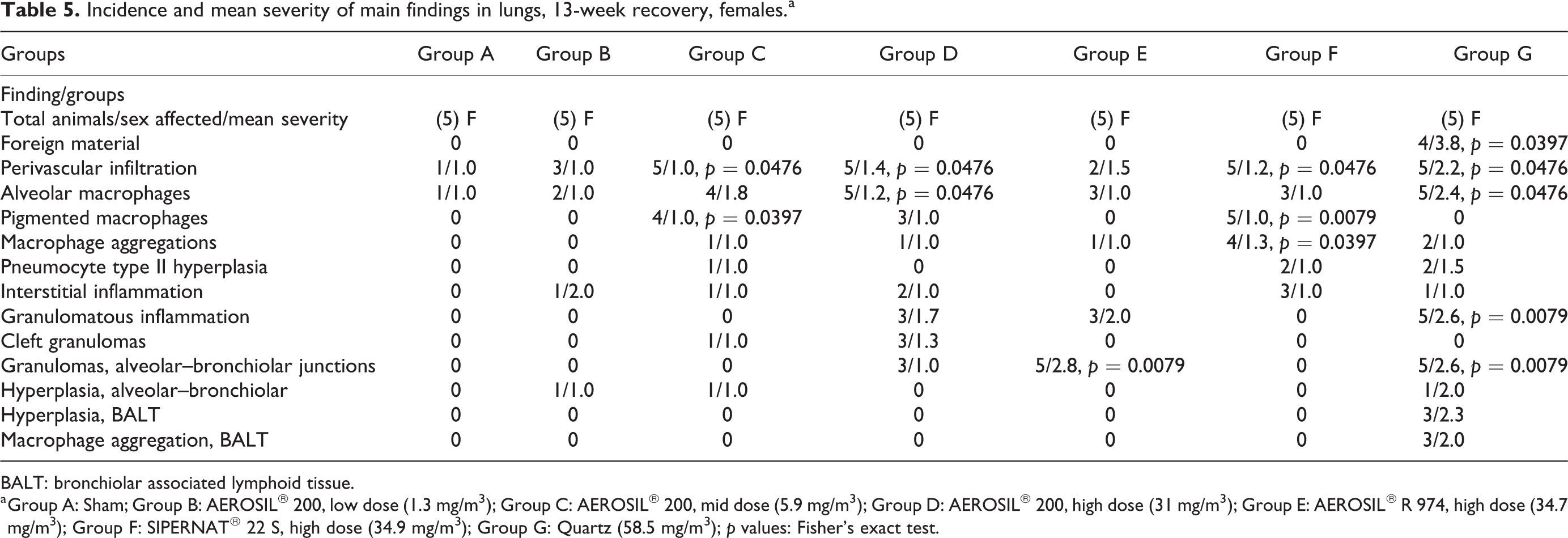
Incidence and mean severity of main findings in lungs, 13-week recovery, females.a
BALT: bronchiolar associated lymphoid tissue.
a Group A: Sham; Group B: AEROSIL® 200, low dose (1.3 mg/m3); Group C: AEROSIL® 200, mid dose (5.9 mg/m3); Group D: AEROSIL® 200, high dose (31 mg/m3); Group E: AEROSIL® R 974, high dose (34.7 mg/m3); Group F: SIPERNAT® 22 S, high dose (34.9 mg/m3); Group G: Quartz (58.5 mg/m3); p values: Fisher’s exact test.
For induced findings, there were no distinctive differences between both sexes. Foreign material in the alveoli was almost not visible any longer except for quartz-treated animals. Perivascular inflammation appeared at similar incidences and severities in all groups, besides the quartz group, where the incidence and severity was recorded at higher levels. Alveolar macrophages increased in incidence and severity, and macrophage aggregations were recorded in all test item–treated groups. Pneumocyte type II hyperplasia was present in the mid- and high-dose AEROSIL® 200 and the SIPERNAT® 22 S groups, and also in the quartz group. Interstitial inflammation was distributed randomly throughout the groups at higher incidences than in controls. Granulomatous inflammation was recorded at low severity degrees in test item–treated groups only. There were single cases of cleft granulomas in animals of the mid- and high-dose AEROSIL® 200 groups. Granulomas at the bronchiolar/alveolar junction were present in the test item–treated groups, except for the low-dose AEROSIL® 200 group. The latter finding was of minimal severity apart from the AEROSIL® R 974 and quartz–treated animals where it was recorded as moderate or severe (Figures 7 to 13).
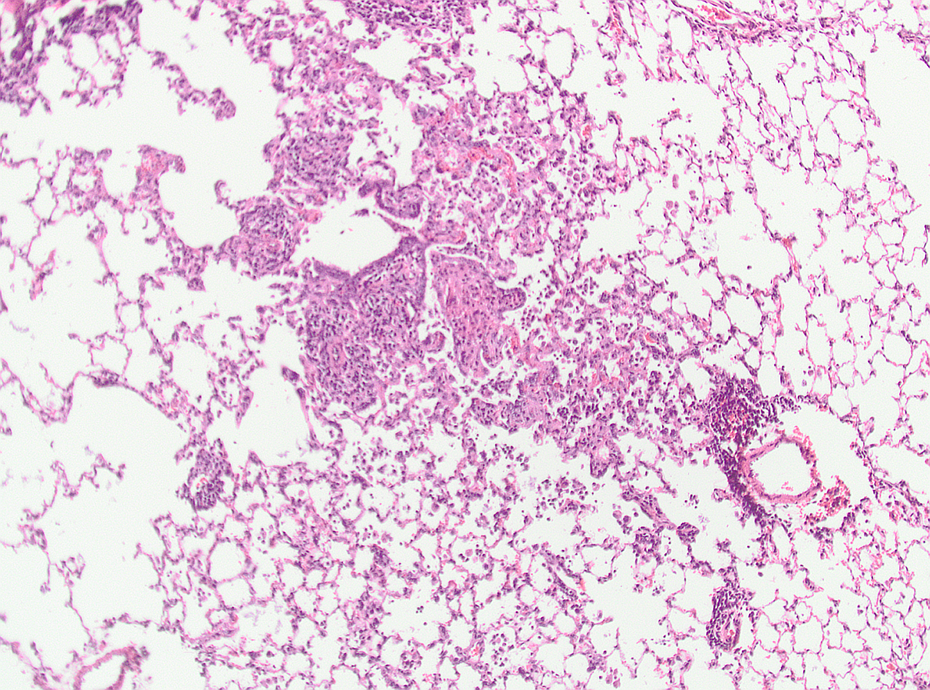
Mid-dose AEROSIL® 200–treated animal after 13 weeks recovery. Alveolar macrophages and interstitial inflammation; HE, ×100. HE: hematoxylin and eosin.
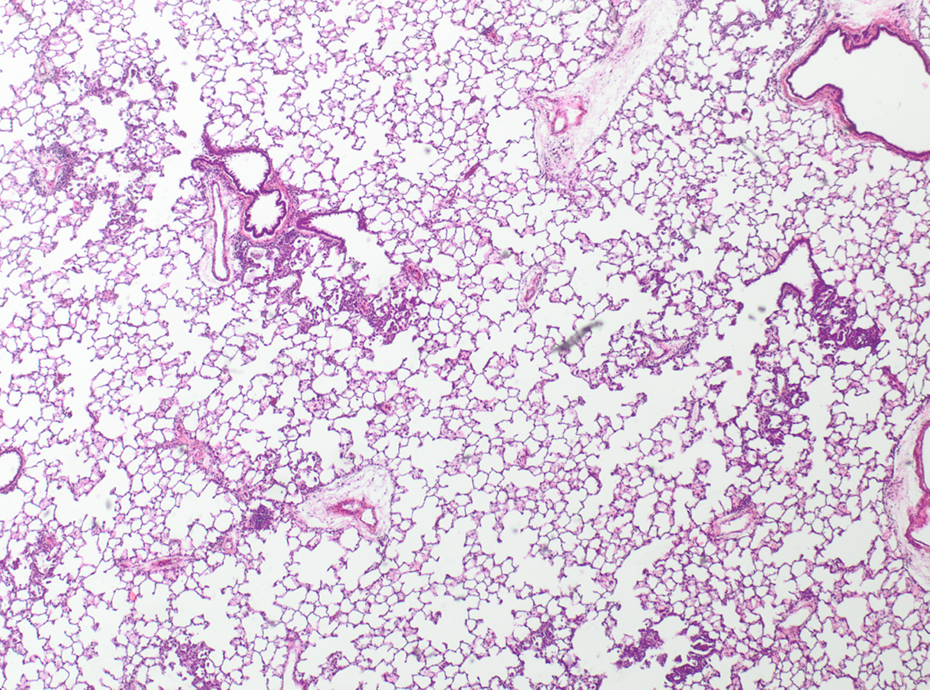
High-dose AEROSIL® 200–treated animal after 13 weeks recovery. Type II pneumocyte proliferation and alveolar macrophages; HE, ×40. HE: hematoxylin and eosin.
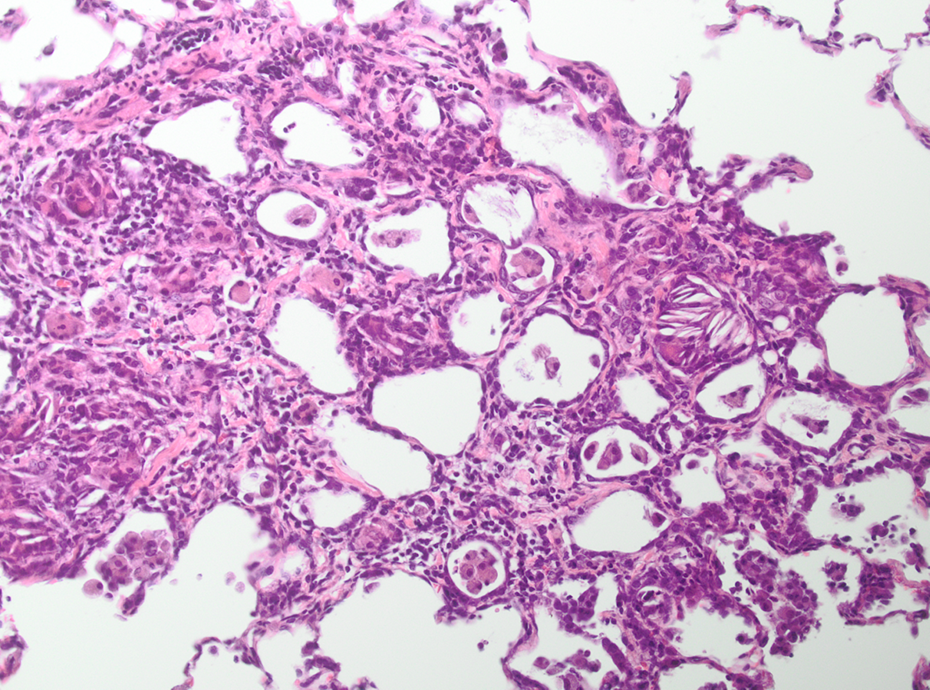
High-dose AEROSIL® 200–treated animal after 13 weeks recovery. Granulomatous inflammation and fibrogenesis; HE, ×200. HE: hematoxylin and eosin.
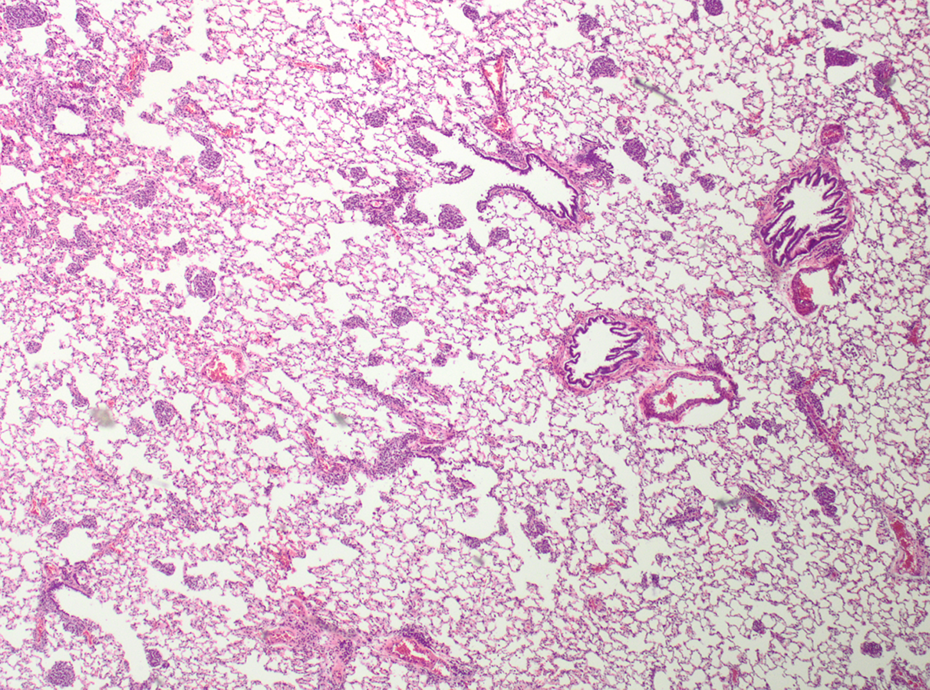
AEROSIL® R 974-treated animal after 13 weeks recovery. Granulomatous inflammation; HE, ×40. HE: hematoxylin and eosin.
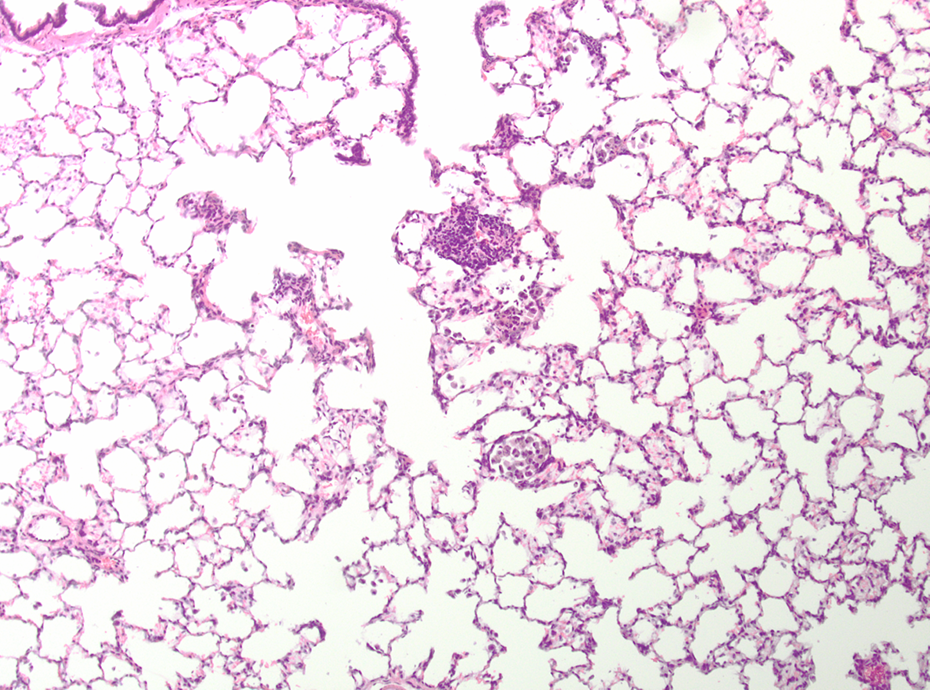
SIPERNAT® 22 S-treated animal after 13 weeks recovery. Minor inflammatory lesions; HE, ×100. HE: hematoxylin and eosin.
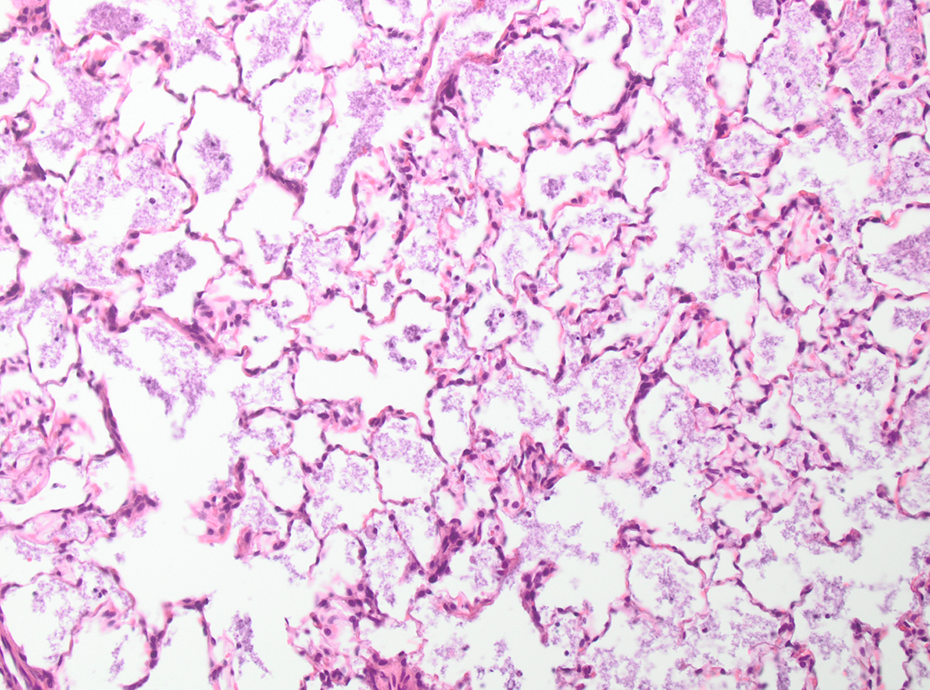
Quartz-treated animal after 13 weeks recovery. Intra-alveolar foreign material; HE, ×200. HE: hematoxylin and eosin.
Fibrogenesis, accompanying inflammatory processes, was diagnosed in two males of the high-dose AEROSIL® 200 group (Figure 10). In addition, a finding not observed after the 13-week terminal sacrifice was noted, that is, a brownish pigment in alveolar macrophages in the alveoli of some animals from both sexes accumulated in the mid- and high-dose AEROSIL® 200 and SIPERNAT® 22 S–treated groups.
In quartz-treated animals, findings present at a higher severity compared to other groups consisted of foreign material (Figure 13), perivascular inflammation, alveolar macrophages (females only), granulomas at the bronchial alveolar junction, and granulomatous inflammation. Moreover, there was a clear affection of the BALT, that is, hyperplasia and accumulation of reactive macrophages.
Fifty-two-week recovery sacrifice
After the planned 52-week recovery (Tables 6 and 7), in controls, the number of inflammatory and reactive lesions increased compared to previous sacrifices probably owing to age-related changes (Figures 14 and 15).
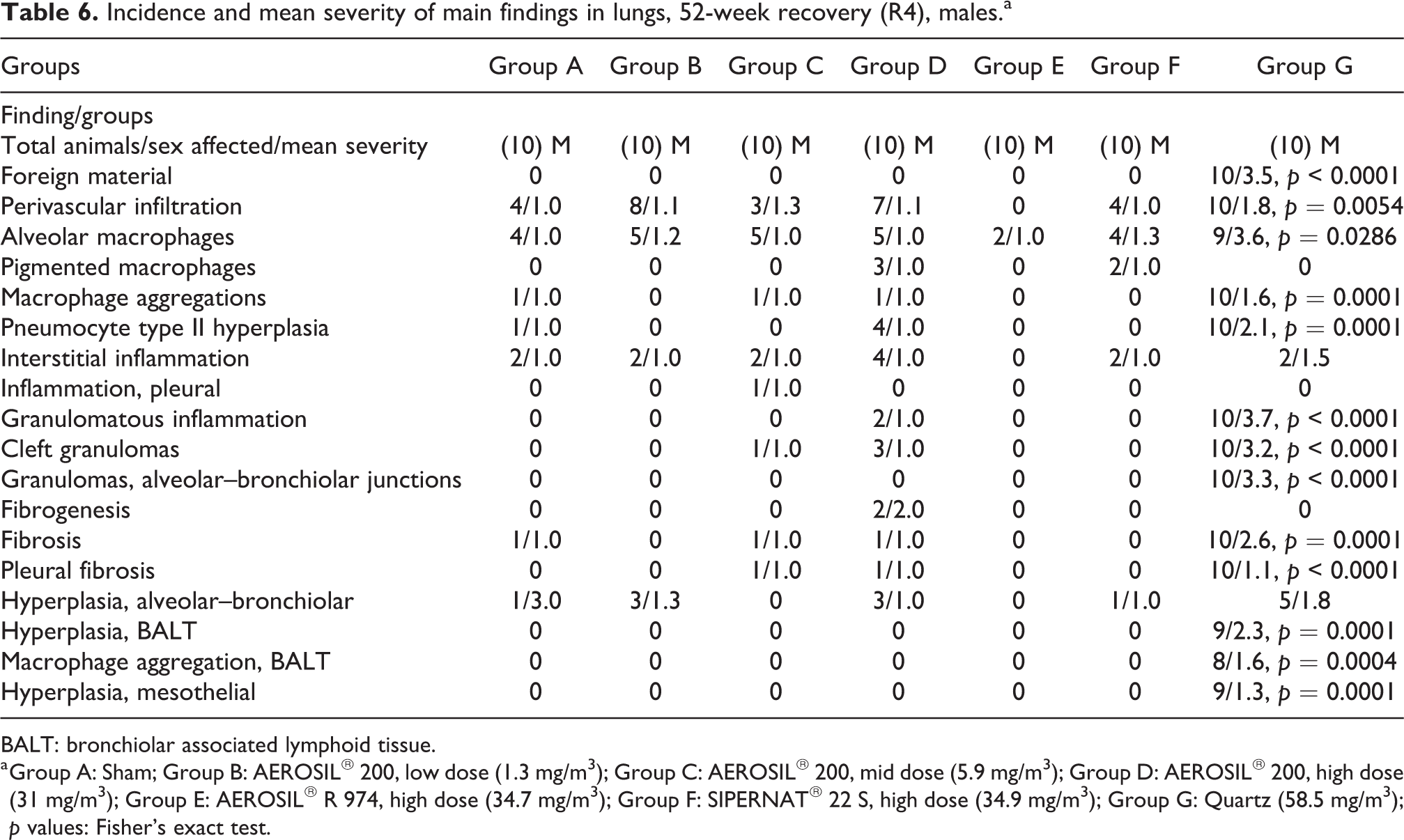
Incidence and mean severity of main findings in lungs, 52-week recovery (R4), males.a
BALT: bronchiolar associated lymphoid tissue.
a Group A: Sham; Group B: AEROSIL® 200, low dose (1.3 mg/m3); Group C: AEROSIL® 200, mid dose (5.9 mg/m3); Group D: AEROSIL® 200, high dose (31 mg/m3); Group E: AEROSIL® R 974, high dose (34.7 mg/m3); Group F: SIPERNAT® 22 S, high dose (34.9 mg/m3); Group G: Quartz (58.5 mg/m3); p values: Fisher’s exact test.
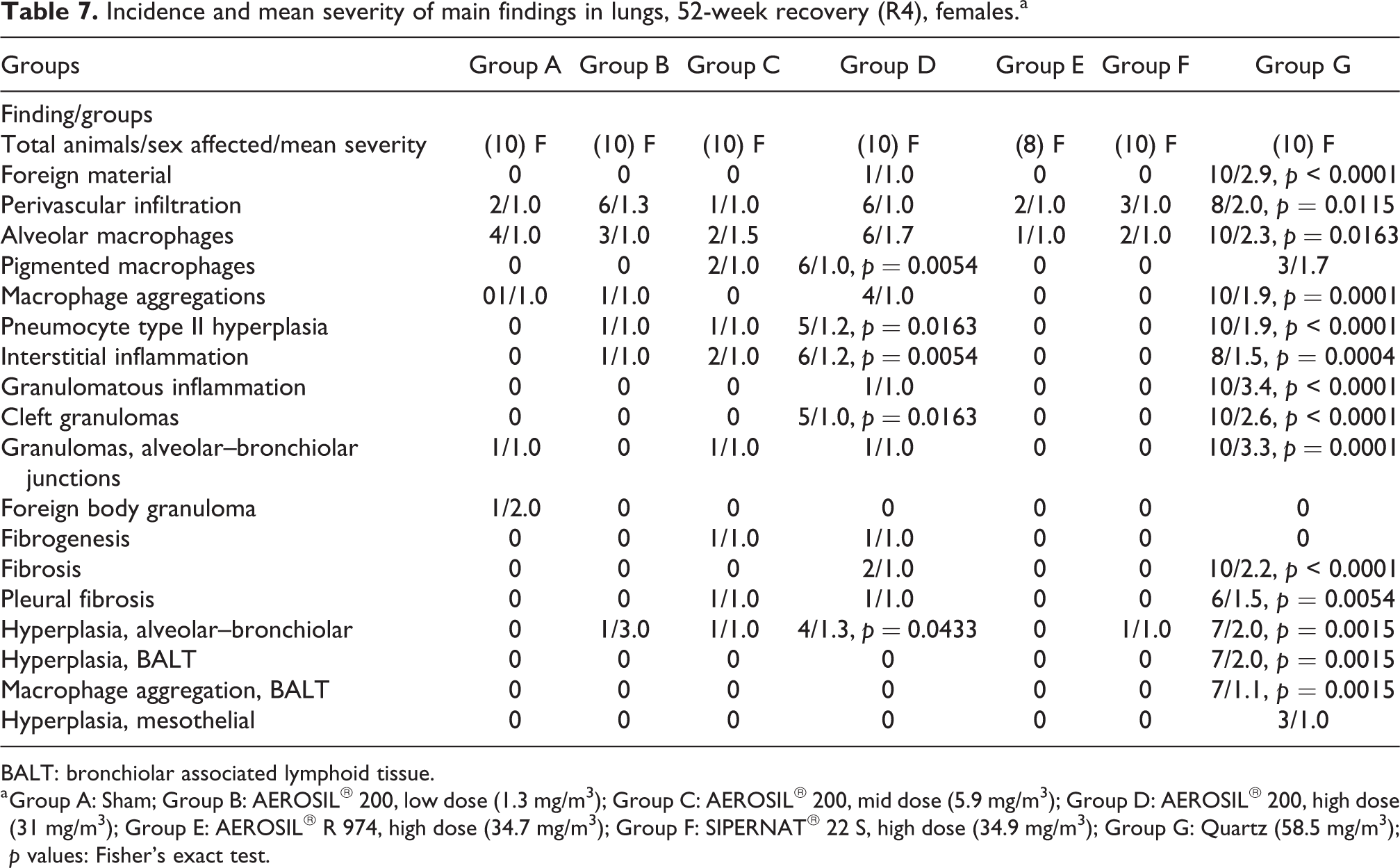
Incidence and mean severity of main findings in lungs, 52-week recovery (R4), females.a
BALT: bronchiolar associated lymphoid tissue.
a Group A: Sham; Group B: AEROSIL® 200, low dose (1.3 mg/m3); Group C: AEROSIL® 200, mid dose (5.9 mg/m3); Group D: AEROSIL® 200, high dose (31 mg/m3); Group E: AEROSIL® R 974, high dose (34.7 mg/m3); Group F: SIPERNAT® 22 S, high dose (34.9 mg/m3); Group G: Quartz (58.5 mg/m3); p values: Fisher’s exact test.
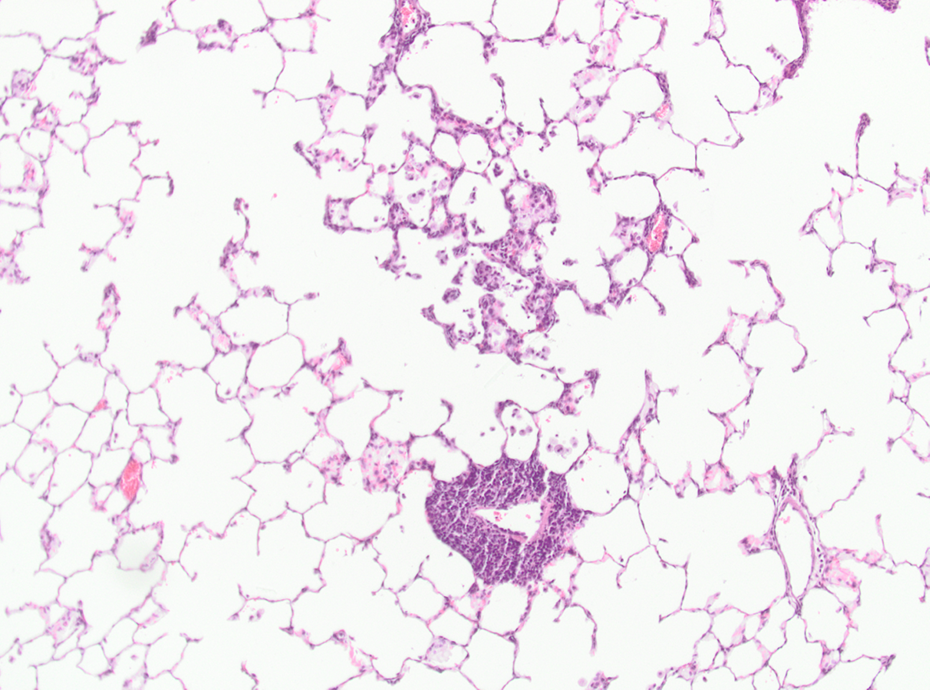
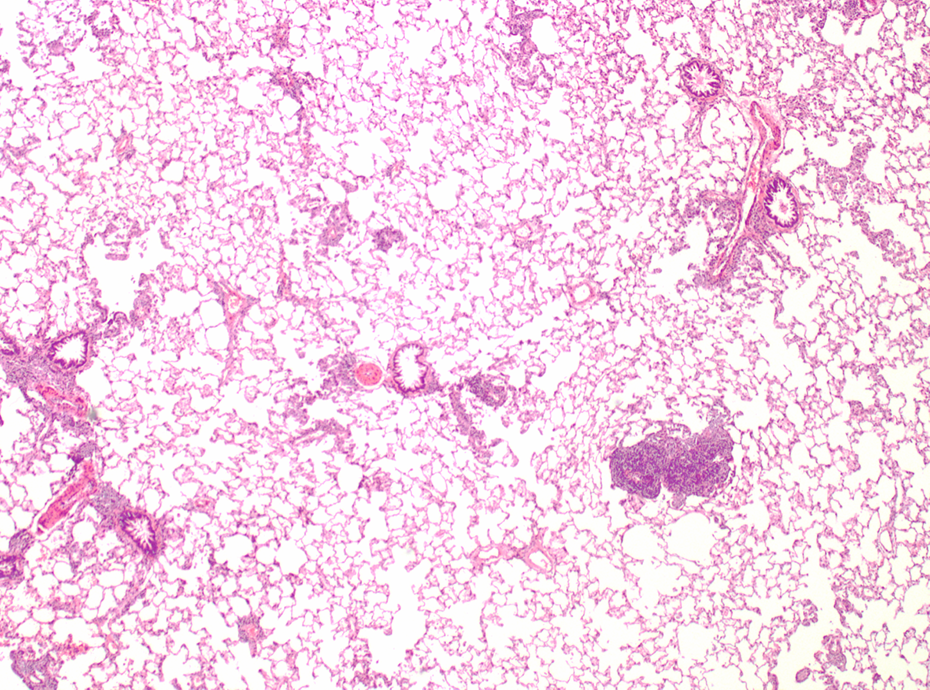
Control animal after 52 weeks recovery. Perivascular and interstitial inflammation; HE, ×100. HE: hematoxylin and eosin.
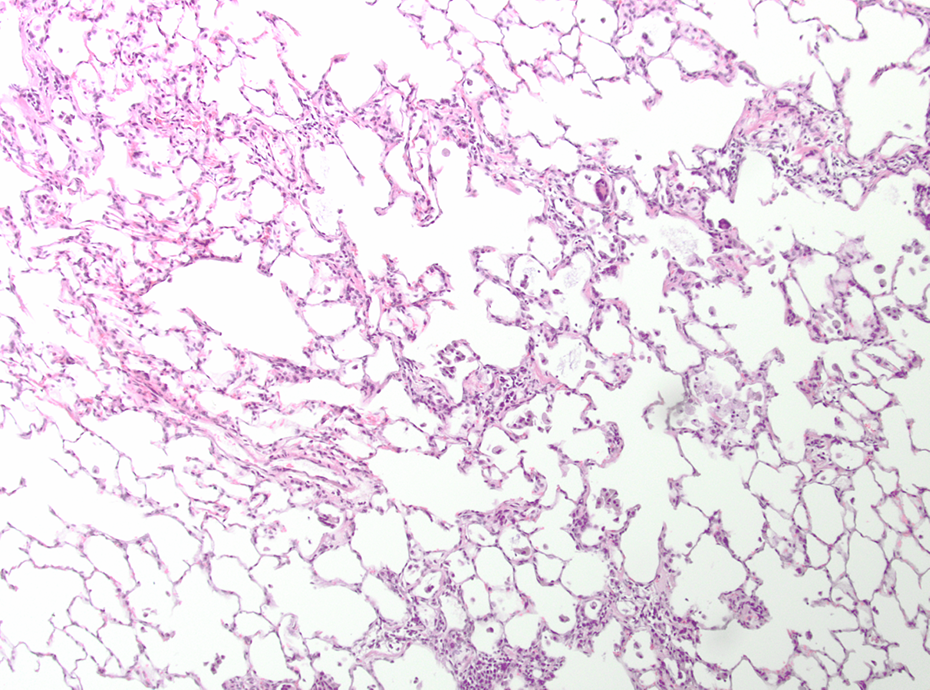
Control animal after 52 weeks recovery. Minimal focal fibrosis; HE, ×400. HE: hematoxylin and eosin.
In test item–treated groups, all findings recorded in AEROSIL® 200, AEROSIL® R 974, and SIPERNAT® 22 S–treated males were similar to control animals (Figure 16), including one case of focal alveolar/bronchiolar hyperplasia (Figures 17 and 18). In females of the high-dose AEROSIL® 200 group, there was still a higher incidence of reactive changes. However, these findings were similar to control lesions that may represent age-related changes (Figure 19). A focal minimal pleural fibrosis was recorded in one animal per sex of the mid- and high-dose AEROSIL® 200 (Figure 20).
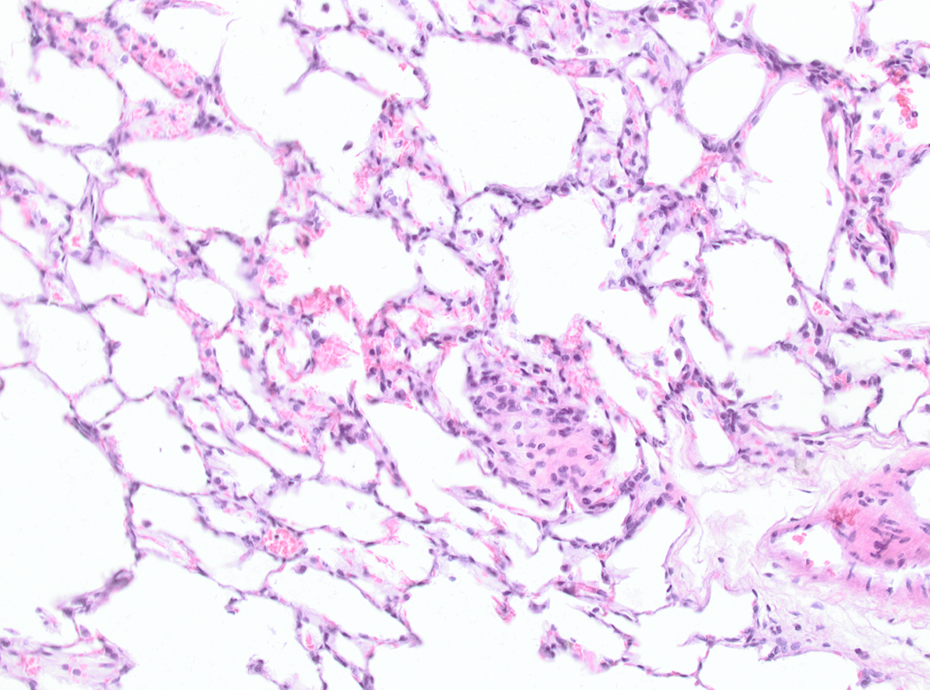
High-dose AEROSIL® 200–treated animal after 52 weeks recovery. Macrophages aggregation and interstitial inflammation; HE, ×200. HE: hematoxylin and eosin.
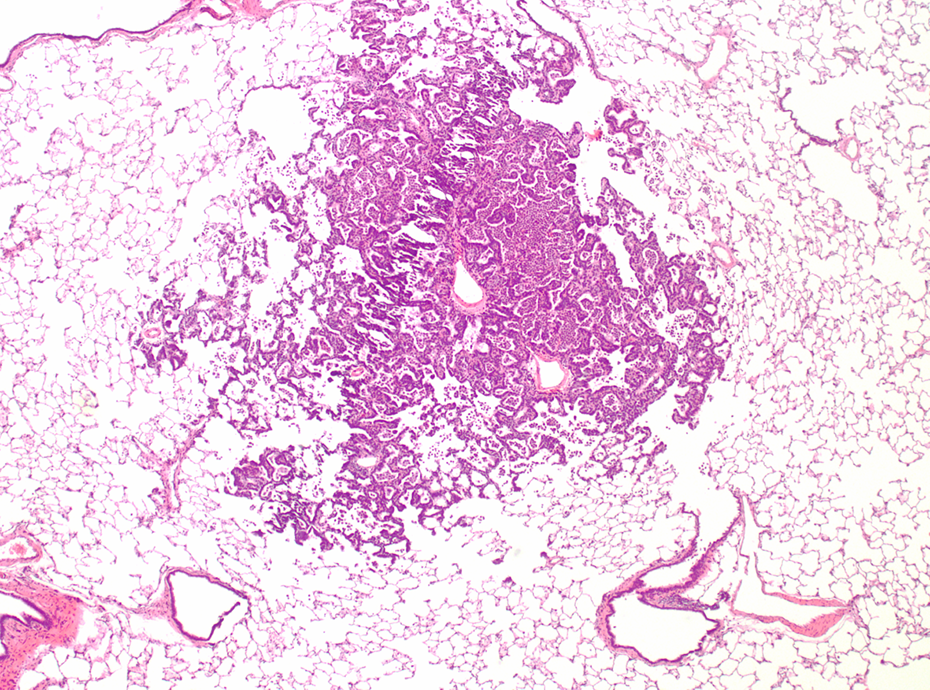
Control animal after 52 weeks recovery. Focal moderate alveolar–bronchial hyperplasia; HE, ×40. HE: hematoxylin and eosin.
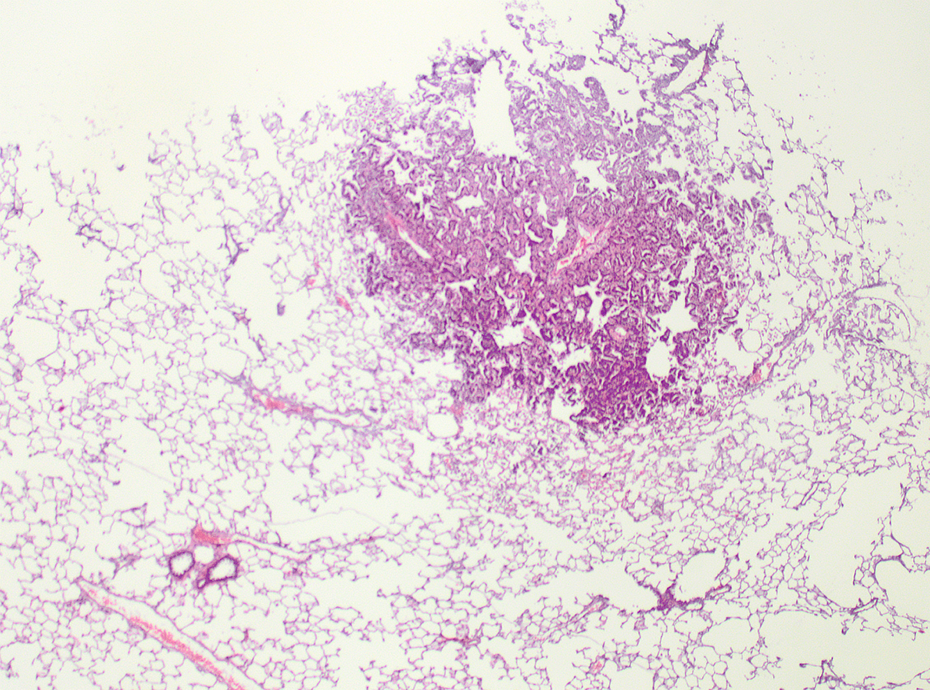
Low-dose AEROSIL® 200–treated animal after 52 weeks recovery. Focal moderate alveolar–bronchial hyperplasia; HE, ×40. HE: hematoxylin and eosin.
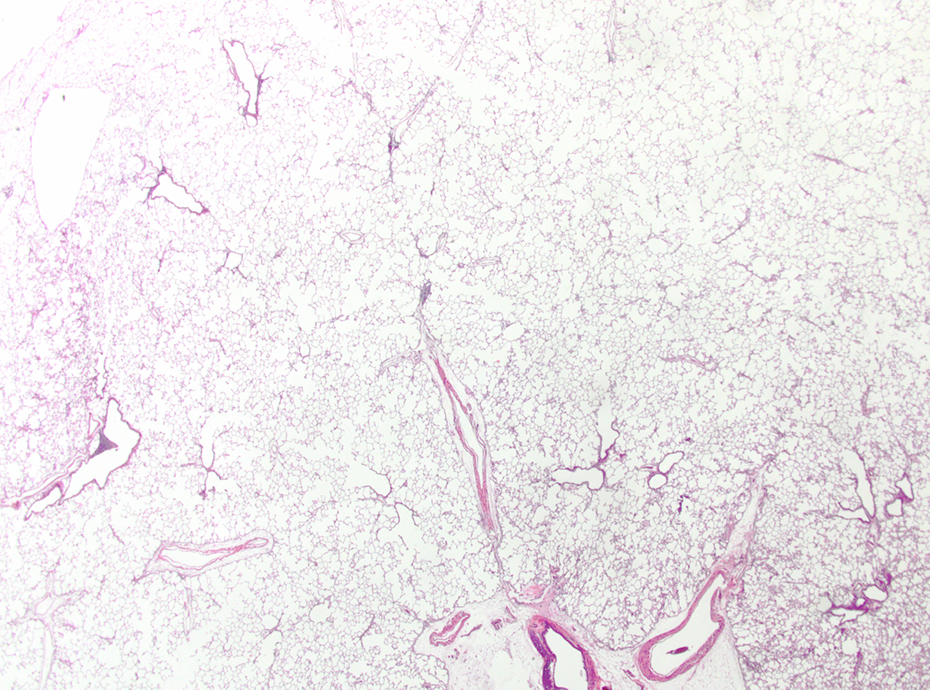
Mid-dose AEROSIL® 200–treated animal after 52 weeks recovery. No findings present; HE, ×40. HE: hematoxylin and eosin.
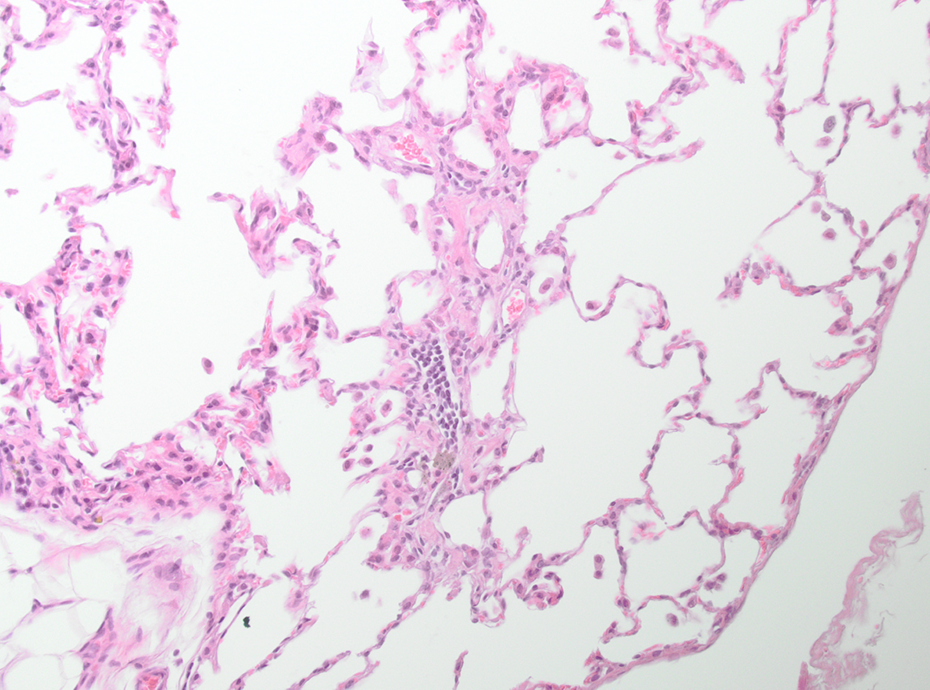
Mid-dose AEROSIL® 200–treated animal. Minimal focal interstitial fibrosis; HE, ×200. HE: hematoxylin and eosin.
In contrast, the quartz-treated animals showed severe intra-alveolar foreign material, associated with a high incidence and severity of reactive alveolar macrophages, macrophage aggregations, the formation of alveolar/bronchiolar junctional granulomas and especially cleft granulomas, as well as granulomatous inflammation, pneumocyte type II hyperplasia, multifocal fibrosis, and macrophage aggregates affecting the BALT. In addition, a high number of males, but less of females was affected by mesothelial hyperplasia (Figures 21 to 24). Additionally, there was one cystic keratinizing epithelioma in the quartz-treated animals after 52 weeks of recovery, an expansile nodule with central keratinization and necrosis located within the pulmonary parenchyma. It was characterized by a thick, irregular, and more complex cyst wall that lacked orderly maturation. Epithelial cells showed an increased number of mitotic figures.
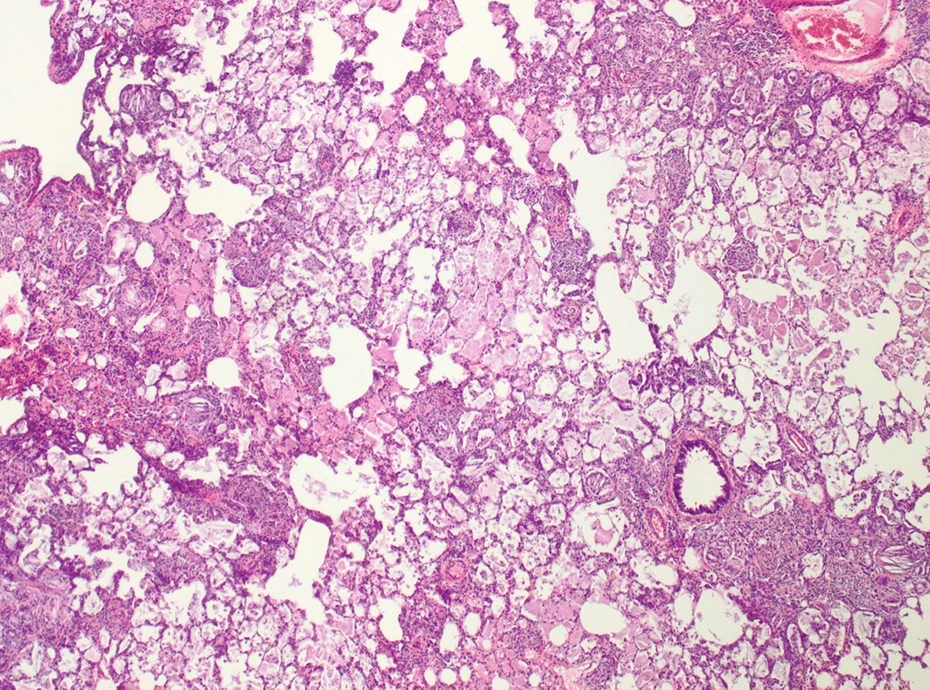
Quartz-treated animal. Inflammatory changes; HE, ×40. HE: hematoxylin and eosin.
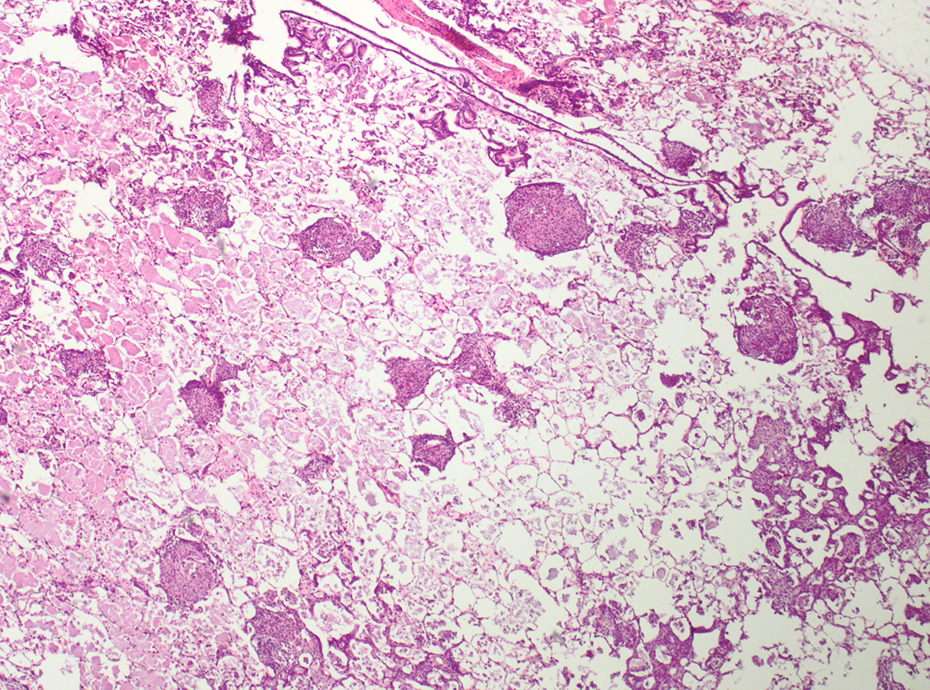
Quartz-treated animal. Granulomatous inflammation; HE, ×40. HE: hematoxylin and eosin.
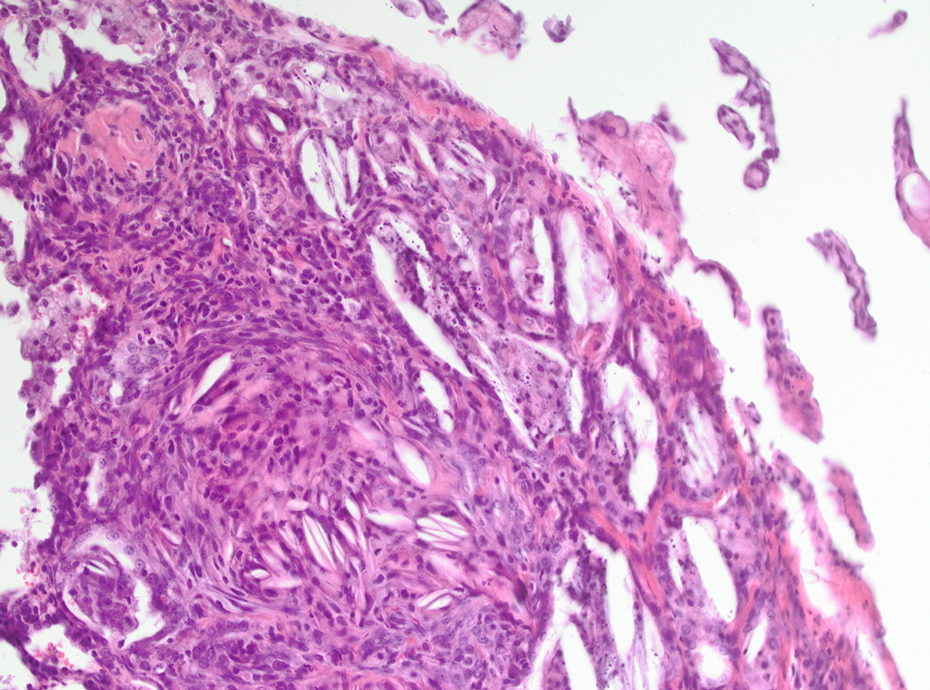
Quartz-treated animal. Granulomatous inflammation, cleft granuloma, fibrosis, and mesothelial hyperplasia; HE, ×200. HE: hematoxylin and eosin.
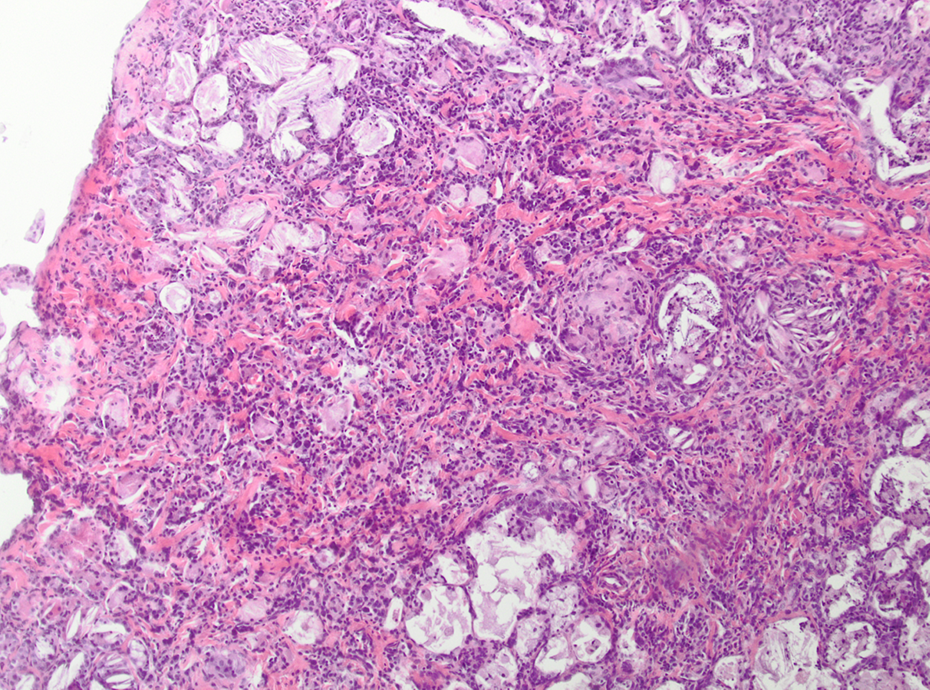
Quartz-treated animal. Granulomatous inflammation, fibrosis, and mesothelial hyperplasia; HE, ×200. HE: hematoxylin and eosin.
Discussion
During a reevaluation performed of the available study material, there were a number of degenerative and inflammatory lesions recorded in all test item–treated and the control groups. This was in agreement with published data and consistent with evidence from a limited number of in vivo studies that there is a largely reversible lung inflammation, granuloma formation, and focal emphysema. Despite these findings, no progression toward lung fibrosis was reported. 14
The reevaluation process followed the INHAND terminology wherever appropriate.
10
Some terms had to be defined to describe induced unique findings. Moreover, a differentiation was made between reversible fibrogenesis and irreversible fibrosis. …The terms fibrogenesis or fibroblastic response may be used to distinguish potentially reversible fibroblast proliferation with minimal cross-linking from irreversible pulmonary fibrosis with extensive cross-linking…
10
At the 13-week sacrifice (K0), the lesions did not differ significantly between test item–treated groups. During reevaluation and PWG, the findings of the original study report were confirmed using an adapted terminology. In sections evaluated from males of all test item–treated groups, the changes consisted mainly of deposited test material (termed “foreign material”) associated with inflammatory lesions (increasing severity of alveolar macrophages, macrophage aggregations, granulomatous inflammation, granulomas) and with reactive lesions (reactive pneumocyte type II proliferation), alveolar/bronchiolar hyperplasia, and some cases of BALT hyperplasia and with reactive macrophages in the BALT (in AEROSIL® 200-treated animals only).
In the original study report, in the 13-week recovery (R1) animals, there was the description of fibrosis, defined as “…an amorphous eosinophilic thickening of the septa, which appeared to be positive for collagen (Weigert van Gieson) stain…”
2
Weigert van Gieson stain is combined trichrome stain for collagen and elastic fibers. Rechecking the original sections, no fibrosis could be detected by the present material. Furthermore, no special stains could be applied to the old study material. Fibrosis was diagnosed in a high number of animals from treated groups. It was recorded in: AEROSIL® 200 (1.3 mg/m3): one male; AEROSIL® 200 (5.9 mg/m3): two males, one female; AEROSIL® 200 (31 mg/m3): five males, four females; AEROSIL® R 974 (34.7 mg/m3): three males; SIPERNAT® 22 S (34.9 mg/m3): one male; and Quartz (58.5 mg/m3): two males.
In addition, the diagnosis “granulomatous fibrosis” was made in four males and five females from the positive control (quartz (58.5 mg/m3).
On the contrary, during the reevaluation and the PWG, fibrogenesis was diagnosed in two males of the high-dose AEROSIL® 200 group that was associated with inflammatory processes. Furthermore, pigmented alveolar macrophages accumulated in the alveoli in the mid- and high-dose AEROSIL® 200 groups and SIPERNAT® 22 S-treated group. These were considered to represent remnants of test material appearing as pigment. In addition to the findings noted after 13 weeks of treatment, there were “cleft granulomas” (focal granuloma formation with clefts) in single animals of the mid- and high-dose AEROSIL® 200.
Other findings described in the original report were diagnosed similarly except for the presence of pigmented macrophages and BALT-related lesions.
Regarding the 52-week recovery (R4), there were differences between the original report and the reevaluation/PWG. In the original report, interstitial fibrosis was diagnosed in: AEROSIL® 200 (1.3 mg/m3): 1 male; AEROSIL® 200 (5.9 mg/m3): 2 males, 1 female; AEROSIL® 200 (31 mg/m3): 10 males, 10 females; and Quartz (58.5 mg/m3): 10 males, 10 females.
In addition, “intragranulomatous fibrosis” was noted in 10 males and 8 females treated with Quartz (58.5 mg/m3).
During reevaluation and the subsequent PWG, fibrogenesis was diagnosed in: AEROSIL® 200 (5.9 mg/m3): 1 female, and AEROSIL® 200 (31 mg/m3): 2 males, 1 female;
and fibrosis was diagnosed in: Sham: 1 male (control, minimal), AEROSIL® 200 (5.9 mg/m3): 1 male (minimal), AEROSIL® 200 (31 mg/m3): 1 male, 2 females (minimal), and Quartz (58.5 mg/m3): 10 males, 10 females (moderate to severe).
In the original report, a “focal increased septal cellularity” was recorded. This finding was defined as “…increase in the number of cells within the alveolar walls. The type of these cells could not be established unequivocally…” Additionally, (focal) alveolar bronchiolization was defined as …cuboidal cells along the alveolar spaces instead of the normal flat cells. Discrimination between this lesion and accumulation of alveolar macrophages was not always easy…
2
In general, all findings recorded in AEROSIL® 200, AEROSIL® R 974, and SIPERNAT® 22 S–treated group males were similar to control group lesions, except in males of the high-dose AEROSIL® 200 group. In the latter animals, there was still a higher incidence of reactive changes including alveolar macrophages, macrophage aggregations, and cleft granulomas. However, the lungs generally appeared similar to control lungs with age-related changes. Therefore, induced lesions were considered to be equally unlikely in females, even though sections of females were not evaluated by the PWG since the sections were missing for interim necropsies.
Summing up, during reevaluation and PWG, fibrosis (as an observable increase in the amount or abnormal location of collagen in lung parenchyma with an occurrence in alveolar septa, interstitium, and pleura, resulting in disruption of the normal lung architecture) was not seen at all after the 13-week exposure period, and after the 13-week recovery period. At the latter sacrifice time point, in two animals of the high-dose AEROSIL® 200 group, there was a minimal fibrogenesis (characterized by single fibers within the inflammatory foci and associated with an increase in septal or interstitial thickness resulting from edema or inflammation without substantial fiber cross-linking).
In general, asbestos and crystalline silica (e.g. quartz and cristobalite) produce fibrosis in a dose-dependent manner that progresses with time, whereas amorphous forms of silica, granite, coal, and silicates such as kaolin, bentonite, talc, and mica do not induce such an aggressive fibrogenic response in the lung. 15 Nonetheless, the exposure to other minerals, for example, feldspar and quartz, as mining contaminants must be considered as a contributing factor toward aggressive lesions. 15,16
The development of a fibrotic lesion is a consequence of the inflammatory process that is associated with the proliferation of epithelial and connective tissue cells that are associated with the release of cytokines and mediators. Fibrogenesis is part of every inflammatory process, since fibroblasts secrete collagen fibers, that will be resolved afterward. Hence, this finding is not considered as being very relevant for the study. Fibrogenesis needs to be differentiated from true fibrosis. Secreted components of the extracellular matrix, namely fibrin, fibrinogen and fibronectin stabilizes, cross-link and interact with collagen, and are thereby forming fibrosis. Fibrogenesis is reversible as opposed to fibrosis, which is clearly not reversible.
After the 52-week recovery period, a minimal focal fibrosis was noted only in one each of control, and low- and mid-dose AEROSIL® 200–treated animals, and in three high-dose AEROSIL® 200–treated animals. This fibrosis was very minimal in severity and only appeared focally. Historical control data show that focal fibrosis is not an uncommon change in aged animals. 17 Therefore, the presence of these lesions is in accordance with spontaneous background findings that may be recorded in control animals. Furthermore, in quartz-treated animals (positive control), there was a significant increase of relative and absolute lung weight during recovery which was not observed in amorphous silica–exposed animals. 1,2
It could be argued, however, that the drastic differences in histopathological findings between crystalline silica (quartz) and amorphous silica–exposed rats in this study might be due to the much higher exposure concentration of the crystalline (59 mg/m3) versus the amorphous (31–35 mg/m3) aerosols inhaled during the subchronic exposures. Assuming similar aerosol size distribution for both the crystalline and amorphous silicas, the deposited lung doses predictably were also much higher in quartz-exposed rats compared to the rats of the amorphous silica groups. It could, thus, be claimed that the greater inflammatory response observed in the quartz animals was to be expected.
Nonetheless, a subchronic rat inhalation study was designed to achieve the same retained lung burden of crystalline and amorphous SiO2 at the end of a 13-week inhalation exposure. 18 To achieve this, the amorphous SiO2 (AEROSIL® 200) concentration had to be increased to an extremely high unrealistic level of 50 mg/m3, whereas the crystalline silica (cristobalite) had to be kept at 3 mg/m3. Although the pulmonary inflammatory response was judged to be twice as high in the AEROSIL® 200 group compared to the cristobalite group at the end of exposure, the inflammatory indicators decreased rapidly in the postexposure observation period in this group, whereas the particular response remained increased in cristobalite-exposed rats. This was consistent with a fast decrease of amorphous SiO2 lung burden in the postexposure recovery phase vis-à-vis a persistent pulmonary retention of crystalline SiO2. 18 At 8 months of recovery, inflammatory markers were still elevated in cristobalite-exposed rats, but were not significantly different in the AEROSIL® 200-exposed animals compared to controls. Genotoxic effects (HPRT mutations in alveolar epithelial cells) occurred only in cristobalite but not AEROSIL® 200-exposed rats, despite the high inflammatory response at the end of the subchronic exposure. Targeted cell death (apoptosis or necrosis) was observed to a high degree in AEROSIL® 200-exposed rats and only at lower incidence in cristobalite-exposed rats.
Thus Johnston et al. suggested that in addition to an inflammatory response, particle biopersistence, solubility, and direct or indirect epithelial cell cytotoxicity may be the key factors for the induction of either mutagenic events or targeted cell death. 18
Similarly, Arts et al. could not detect irreversible changes after treatment of rats by inhalation with three different types of SAS at different doses after a 1- or 3-month observation period following a 5-day inhalation period. In contrast, the treatment by quartz caused the development of silicosis. 19
The results are in agreement with data published by Richards et al., 15 who reported that in rats at comparable doses of quartz produced fibrosis, however, amorphous silica caused a transient fibrogenesis, and titanium dioxide was without effect.
Furthermore, epidemiological studies do not reveal data supporting the hypothesis that SASs have any relevant potential to induce fibrosis in workers with high occupational exposures. 9,20,21 Moreover, there is no study that provides evidence for carcinogenic potential of amorphous silica to human. 9
Other findings recorded by the PWG in the lungs from animals after 52 weeks recovery were distributed randomly throughout the groups, except for some reactive changes in high-dose AEROSIL® 200 animals (macrophage aggregation, pneumocyte type II hyperplasia, pigmented macrophages, cleft granuloma, interstitial inflammation, and alveolar–bronchiolar hyperplasia). Such responses are not unusual in the lungs of aged animals. They were regarded as being within the range of spontaneous background lesions. 17,22,23
The PWG confirmed the absence of an induced fibrosis in this study. Some minor differences in inflammatory parameters were established (namely increased incidences of alveolar macrophages after 52 weeks), which was likely due to the fact that not all sections per lung have been reevaluated by the PWG. However, the incidence and severity was similar between study controls/historical controls and treated animals for this time point considering all sections.
In conclusion, after 52 weeks of recovery, a number of minor inflammatory lesions were recorded in control and amorphous silica–treated animals. Based on these findings, no differences could be established between control group and the treated animals. Fibrogenesis and/or minimal focal fibrosis were diagnosed in single animals in all groups. Similar level of these findings were equally noted in untreated animals. In contrast, slight to moderate fibrosis was diagnosed in all quartz-treated animals.
Considering the findings in the lungs, the various types of amorphous silica did not result in any significant differences. Most importantly, since the findings were fully reversible a long-term adversity is beyond a further viable consideration.
Footnotes
Authors’ note
The coauthors are listed in alphabetic order.
Authors’ contribution
KW performed the reevaluation and was member of the PWG. CG, JFH (chairman), EEM, and GO were members of the PWG. MB, AB, and NK supported text writing and design.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Evonik Resource Efficiency GmbH and Wacker Chemie AG produce SAS. The reevaluation and the performance of the PWG have been funded by Evonik Resource Efficiency GmbH.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The reevaluation and the performance of the PWG have been funded by Evonik Resource Efficiency GmbH.
