Abstract
Nanomedicinal products (NMPs), due to their unique properties, are extensively investigated for their biomedical and pharmaceutical applications. Apart from being carriers of certain drugs, nanoparticles can also interact with both the innate and adaptive immune systems, thus eliciting immune responses. Following administration, their discrete physicochemical properties make each NMP act differently in the organism. Actually, the toxic effects of NMPs, in terms of specific end points, do not necessarily depend on the specific group or structural type of the particle. Furthermore, the nanoformulation may change the pharmacokinetic/toxicokinetic profile of the drug. Unveiling the structure–activity relationship of NMPs would help to clarify their immunomodulatory effects. Therefore, in addition to the current regulatory immunotoxicity testing strategies, development and regulatory approval of nano-sized pharmaceuticals still need to be discussed in order to identify potential gaps in the safety assessment.
Introduction
Nanomedicinal products (NMPs) can be designed to target specific cells and/or tissues, and in virtually all cases, NMPs can reach the immune system. It has been shown that eventually most nanomaterials (NMs) end up in organs of the mononuclear phagocytic system, notably the liver and spleen. 1 –3 Adverse immune effects, including allergy, hypersensitivity, and immunosuppression, have been reported after NMP administration. 3,4 Currently, no regulatory documents are specifically dedicated to the evaluation of the immunotoxicity of NMs or nanocarriers, 3 while the regulatory system for medicinal products is based on the provisions of Directive 2001/83/EC114. 5 Thus, all medicinal products, including NMPs, are evaluated on a case-by-case basis. 6 In Europe, the market authorization of medicines is granted by the European Commission after advice from the European Medicines Agency (EMA) and national regulatory authorities. 7 On the other hand, in contrast to the EMA guidance, the Food and Drug Administration (FDA) guideline considers information on immunosuppression in addition to immunotoxicology: immunogenicity, hypersensitivity, autoimmunity, and adverse immunostimulation. 3 The International Council for Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) S8 guidance on “Immunotoxicity Studies for Human Pharmaceuticals” was mainly based on the weight-of-evidence (WoE) review of the factors to evaluate the necessity for extra immunotoxicity tests following the initial evaluation by standard toxicity studies parameters. 7 WoE considers the class of the compound, the structural similarity to other compounds demonstrating immunosuppressive characteristics, pharmacological properties of the product, the disposition of the drug, and possible clinical findings that could lead to new nonclinical testing. 3 However, due to their unique structures, nanomedicines, nanocarriers, and NMPs have distinctive behaviors throughout the organism. The slowness and difficulty in establishing regulations are based on the novel properties of engineered NMs associated with their potential cytotoxic effects, the insufficiency of standardized methods to test those effects, and the overall lack of epidemiological evidence suggesting engineered NMs are toxic. 8 The toxic effects of NMPs, in terms of specific end points, are not associated with specific groups or structural types of NMPs. Second, the nanoformulation compared to the regular formulation may change the pharmacokinetic profile of the drug, and therefore its toxicokinetics. 9,10 Although some nanoparticles (NPs) exert no immunomodulatory activity, they can suppress inflammatory responses. However, recent available data on the immunomodulatory effects of NPs are not sufficient. Furthermore, it is fact that their immunological effects do not reflect their immunotoxicities. 11
Although various definitions of NMs and NMPs are used in regulations and scientific literature, NMs are defined as materials with lengths ranging from 1 nm to 100 nm in at least one dimension. This definition also includes particles in agglomerates or aggregates whenever the constituent particles are in the size range of 1 to 100 nm. 5,12 Because these materials are highly desirable for commercial and medical applications, the toxicity of NMs and ultrafine particles, in general, is a growing area of public health concern. 13,14 The development of new immunomodulatory agents, as such nanostructures may provide an opportunity, in a clinically significant fashion, to more effectively manipulate or deliver immunologically active components to target sites. DNA, RNA, peptides, proteins, and small molecules can all be used as cancer therapies when formulated in nanocarriers. The carriers of vaccines, such as viruses and NPs, have already been used in clinical studies for many years, but relatively little is known, imagined, or published about the associated toxicity. 15 –19 In this context, immunotoxicological studies of NMs have high priority and should be part of the risk assessment process that currently employed to protect human health and the environment. 13,14
Adverse effects of NPs on human health depend on individual mechanical and physicochemical properties, as well as exposure, and NP chemistry, particle size, shape, agglomeration state, chemical composition, surface area, surface chemistry, surface charge, porosity, and electromagnetic properties. NMs can stimulate and/or suppress the immune response, and the compatibility of an NM with the immune system is largely determined by the properties mentioned above. 20 –22
The interaction of immune system components with NPs
NPs interact with both innate and adaptive immune cells, and can stimulate and/or suppress the immune response. 23 Their compatibility with the immune system is largely determined by their surface chemistry. 24 Actually, inflammation is a defense mechanism of the body that involves several immune regulatory molecules, following the infiltration of phagocytic cells. Several studies with single- and multi-walled carbon nanotubes and fullerene derivatives have shown that NPs may induce the release of inflammatory mediators from various cell types, including alveolar and bronchial epithelial cells, epidermal keratinocytes, and cultured monocyte–macrophage cells. Collectively, NMs provoke considerable inflammation due to their physicochemical properties. Further the length- and size-related structure–activity relationship of NPs is important in stimulating host inflammatory responses. 25 –28 NP uptake by immune cells may occur both in the bloodstream by monocytes, platelets, leukocytes, and dendritic cells (DCs) or in tissues by resident phagocytes such as Kupffer cells in the liver, DCs in lymph nodes, macrophages, and B cells in the spleen. NP uptake by immune cells can be facilitated by the adsorption of plasma proteins to the surface of the NP. 29 How NPs potentially influence the immune system can be examined by NP-mediated hemolysis, NP-induced platelet aggregation, and the ability of the NP to activate the complement system. 29 In general, larger particles are taken up by cells more efficiently than smaller particles of the same composition and surface properties. Additionally, the distance between two neighboring NPs strongly affects the capacity of adsorption and phagocytic uptake. 30 Particles bearing cationic or anionic surface charges have been shown to be more attractive to phagocytes than neutral particles of the same size. A hydrophilic surface with a low negative charge reduces protein adsorption and uptake of NPs. 31
However, naked NMs dynamically interact with encountered biomolecules and form the so-called “protein bio-corona.” 32 Actually, in biological fluids, proteins compete for the NP “surface,” leading to a protein corona that largely defines the biological identity of the particle. 33 As for NPs targeting, the chemical and physical characteristics of the NP and the cellular phenotypes play a relevant role in the potential toxic or nontoxic effects exerted by the protein corona. Even in the case of NPs with the same chemical and physical properties, relative quantities of the adsorbed proteins can change their toxicological profile. In some cases, the interaction between NPs and the protein corona can reduce systemic toxicity by increasing the stability of particles and by inhibiting the generation of reactive oxygen species (ROS). 34,35 According to one report, the formation of a protein corona can increase internalization specificity in macrophages and endothelial cells, causing an increase in the risk of cytoplasmic and systemic toxicity. According to others, the elements of the innate immune system can exert a completely different action toward the presence of a protein corona and can lead to activation of an immune response. 34
Although interactions of NMs with the immune system can involve both innate and adaptive immune responses, macrophage-based reactions are at large in charge of NMs such as recognition, uptake, processing, and clearance. 23 Protein aggregates, as NPs, mimicking danger signals lead to a first-line effect on DCs having alterations on the adaptive immune system response. 36 In this respect, uptake kinetics, cell surface marker expression, soluble protein antigen uptake, and degradation, as well as T-cell stimulating capacity of NPs, determine the modulation of host immune response. 37 While the cellular components of the innate immune system form the interface between NPs and host, the noncellular components of the innate system strive to prevent the entry of NPs through biological barriers. Unlike cells in the innate system, which use a fixed repertoire of inherited receptors, via recombination, T and B cells generate antigen receptors that may recognize practically any antigen. Thereby, elements of the adaptive response also support the function of the innate immune system. In this manner, DCs form the main interface between the innate and adaptive immune systems. 36,38,39 Therefore, the interaction between the components of the immune system and the NPs has been discussed taking into account the successive stimuli that occur in innate and adaptive immunological systems.
Interaction of NPs with complement system
Macrophages use phagocytosis to internalize NP–protein corona complexes, while naked NPs can be taken up through clathrin- or dynamin-dependent endocytosis. 40 The amounts of internalization of NPs in monocytes and macrophage-like cells differ significantly depending on the presence or absence of a protein corona. NP uptake by THP-1 monocytes can be strongly inhibited by the presence of serum albumin. On the contrary, in the case of differentiated THP-1 type macrophage-like cells, the presence of albumin on the NP surface induces an increase in the internalization rate. This process is accompanied by the secretion of inflammatory cytokines as proof of the activated phagocytosis. 40,41
While complement system can be activated through three pathways; classical, lectin, or alternative, NP–protein corona complexes seem to initiate the classical pathway. 42 The classical pathway is activated when the complement protein C1 binds to immunoglobulin G (IgG) molecules adsorbed on the NP surface. The alternative pathway is independent of C1 activation and is triggered through the spontaneous hydrolysis of C3 to C3b. The alternative pathway activation provides the C3b deposition that adds 25% more mass to the NP surface 43 (Figure 1). Furthermore, the classical pathway of the complement system is initiated when the head group of C1q binds to IgG molecules present on the surface. C1q binding promotes a damaging effect of excessive complement activation. 44 Classical complement activation is markedly reduced on the nanostructured hydrophilic surface compared with smoother ones. The ability of human IgG to activate the complement system is also significantly reduced by the surface of the nanostructure. In this manner, the physicochemical surface properties of NPs could mediate protein corona formation, and thus control the activation of complement pathways. 45 In this respect, the conformational state of the glycopolymer chain on the NP surface acts as a “molecular switch” for complement activation and amplification. The protein corona on the NP surface simultaneously affects this process. Thus, the elevated levels of two pro-complement proteins, factors B and C3, present on the NP surface grafted with glycopolymer chains at high grafting density compared to low grafting density surface, modulate complement activation, and the immune response. 46,47
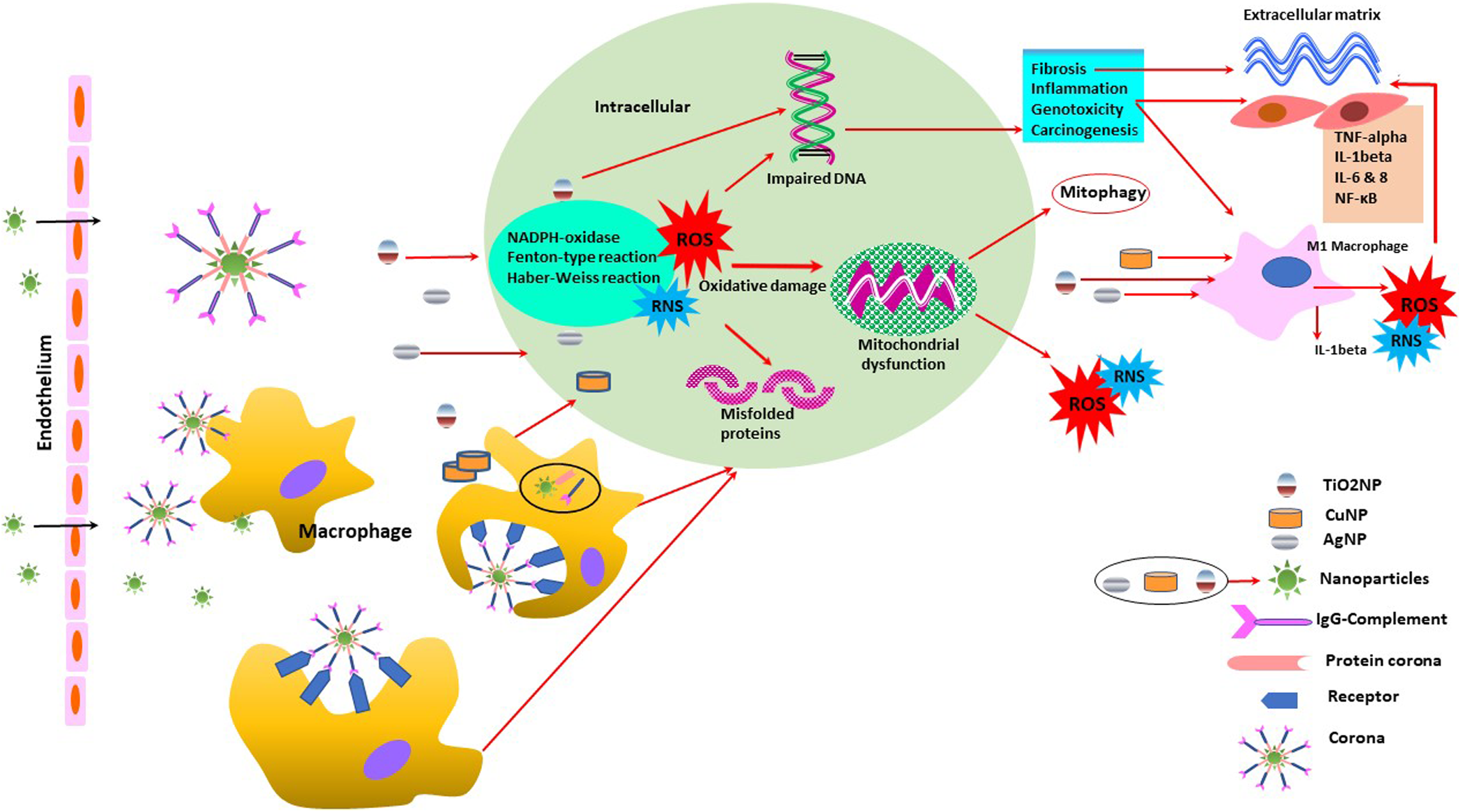
Proteins compete for the NP “surface,” leading to a protein “corona” that largely defines the biological identity of the NPs. Macrophages internalize the agglomerates of NPs via actin-dependent endocytosis. Biodegradable NPs are digested and, thus, facilitate elimination, but nondegradable NPs can be stored in cells for long periods of time. ROS-mediated oxidative stress is considered a key effect for the toxicity of NPs. Excess ROS and RNS production is a key to the mitochondrial damage. NP-induced oxidative stress in mitochondria can lead to the activation of mitochondrial apoptotic pathways that are the major mechanisms of cell death caused by NPs. The autophagic activity is a key mechanism for cellular clearance of aggregated proteins leading to cell survival. Inside the nucleus, NPs can affect DNA, thereby cause DNA breakage or alter the gene expression. ROS generation also increases cell death and enhances DNA damage and histone phosphorylation. ROS and RNS act as second messengers to activate the pro-inflammatory signaling cascades. Activation of the pro-inflammatory signaling cascade results in a polarization of macrophages from M2 to M1 type. NP: nanoparticle; ROS: reactive oxygen species; RNS: reactive nitrogen species; TNF-α: tumor necrosis factor-α; IL-1β: interleukin-1β; NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells; NADPH: nicotinamide adenine dinucleotide phosphate; TiO2: titanium dioxide; Ag: silver; IgG: immunoglobulin G; Cu: copper.
Adaptive immunity can be directed against the NP itself, even subsequent to the formation of a protein corona. The protein corona, when interacting with NPs, can stimulate the adaptive immune process if the complex created represents a danger signal, which in turn activates the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling pathway. 34,48 Even slight changes in the dimensions of the NPs can evidently influence the binding of protein with the NP’s surface and within the protein corona. In addition, variations in serum protein adsorption correlate with differences in the mechanism and efficiency of NP uptake by macrophages. 49,50 Corona proteins present in the blood serum quickly bind to conventional non-stealth NPs, allowing macrophages of the mononuclear phagocytic system to easily recognize and eliminate these drug delivery devices before they can perform their designed therapeutic function. However, copolymer coating on the surface of NPs results in an increase in the blood circulation half-life of the particles. 51
After the NMs are taken up by the immune cells, their fate depends on their biodegradability. Biodegradable NMs are digested and, thus, facilitate elimination, but nondegradable NMs can be stored in the cells for long periods of time. 29 Increasing the circulation time of drug nanocarriers and enhancing the biodegradation process of NMs cause inflammatory responses and toxicity after inadvertent exposures. 52,53
The interaction of the multi-/single-walled carbon nanotubes and C60 fullerene with the Toll-like receptors (TLRs) triggers the innate immune response in the body. Secretion of inflammatory protein mediators such as interleukins and chemokines results from the ligand molecule-dependent inhibition or activation of TLR-induced signal transduction. 54 Furthermore, activation of the complement cascade on exposure to liposomes and other lipid-based NPs may lead to hypersensitivity reactions and even anaphylaxis. Therefore, NPs, which are administered systemically, should be tested for the tendency to activate the complement system. 29,55 Thus, following introduction of the NPs into the bloodstream, their lipid surface is immediately modified by the adsorption of a “protein corona”. Long-standing interaction of corona with the receptor-binding sites of target cells activates the cell’s uptake machinery, and triggers liposome endocytosis. 56
As noted above, nevertheless, there may be undesirable interactions between NPs and the immune system, such as immunostimulation or immunosuppression, which may promote inflammatory or autoimmune disorders.
Anti-inflammatory versus pro-inflammatory response induced via NPs—“safe-by-design”
The exact mechanism of how these complement proteins mediate nanotoxicity has not yet been elucidated. 57 Immunosuppression is often mediated by toxicity of a substance to T cells. Although the accumulation of metal NPs is higher in the thymus than in the spleen, NPs affect CD4+ and CD8+ lymphocytes in the spleen but not in the thymus. 58 On the other hand, induction of immune tolerance by NPs can be considered a form of desirable immunosuppression. 57 NPs elicit a nonspecific inflammatory process with engagement of neutrophils, macrophages, and other effector cells; however, it is important to distinguish between acute and chronic inflammation to identify the potential hazards of NPs for human health. In this process, complement activation represents a dynamic corona formation, which has important implications in clearance and safety of NPs. 59
The cyto-genotoxicity of zinc oxide NPs induces a significant decrease in mitochondrial membrane potential that results in ROS generation and apoptosis. 60 Noble metal NPs modulate ROS and reactive nitrogen species (RNS) production, suppressing the antioxidant system of tumor-associated macrophages. Moreover, ROS and RNS act as second messengers to activate the pro-inflammatory signaling cascades. Thus, while there is a down regulation of tumor necrosis factor-α (TNF-α) and interleukin-10 (IL-10), the pro-inflammatory cytokine interleukin-12 (IL-12) is upregulated. Activation of the pro-inflammatory signaling cascade results in a polarization of macrophages from M2 (anti-inflammatory) to M1 (pro-inflammatory) type. 61 At lower doses of metal NPs, the Th1 or M1 response is counteracted by Th2 or M2 response, resulting in insignificant oxidative damage. However, with increasing dose of NPs, the M1 response increases over the M2 response resulting in significant tissue damage. 62 In addition to direct prooxidant effects of NPs, they may activate the redox-machinery of the cell especially in the lungs where inflammatory cells—neutrophils and macrophages—can act as potent generators of ROS. 53 Resting NLRP3 localizes to endoplasmic reticulum structures, whereas on inflammasome activation both NLRP3 and its adaptor ASC redistribute to the perinuclear space where they co-localize with the endoplasmic reticulum and mitochondria. Mitochondria are the major source of ROS in the cell. ROS-producing mitochondria can trigger inflammasome activation in phagocytic cells, 63 thus providing a link between mitochondria and inflammation, with ROS acting as the signal. Active cellular internalization of NPs or fibers through phagocytic pathways may, at least in professional phagocytic (professional antigen presenting) cells, result in activation of the nicotinamide adenine dinucleotide phosphate (NADPH) oxidase with resultant massive generation of ROS. 64 Moreover, cationic NPs interact with the cell membrane, triggering membrane disruption and Ca2+ influx. The elevation of intracellular Ca2+ induces degranulation and oxidative stress. 65 Fundamentally, two enzymatic systems, NADPH oxidase and nitric oxide (NO) synthase (NOS), produce superoxide radicals and RNS, respectively. The NADPH oxidase produces more superoxide ions, hence induces higher oxidative stress. 66 Both of these are not reactive enough to oxidatively biodegrade NPs. However, superoxide radicals and RNS can effectively react to yield peroxynitrite, whose oxidizing potency is sufficient to cause biodegradation. The majority of NO produced by activated macrophages are converted to peroxynitrite. 67,68 Indeed, biodegradation reactions taking place in inflammatory cells are substantial contributors to the overall elimination of NPs from the tissues. The “safe-by-design” principle is a prerequisite in the development of new NMs. In this context, for the optimizing lifetime and clearance of NPs from the body, the “safe-by-biodegradation” concept is very important. 68
Furthermore, pro-inflammatory effects of NPs increase the expression of interleukin-1β (IL-1β), macrophage inflammatory protein-1α (MIP-1α), monocyte chemoattractant protein-1 (MCP-1), keratinocyte chemoattractant, thymus and activation-regulated chemokine (TARC), granulocyte-macrophage colony-stimulating factor (GM-CSF), and the activation of the stress-activated mitogen-activated protein kinases (MAPKs) p38 and c-Jun N-terminal kinases (JNKs). All considered, the available data suggest that oxidative stress is at least one of the mechanisms by which engineered NPs exert their potential toxicity. 69
Immunological dysfunction caused by titanium dioxide (TiO2) NPs is involved in the increased expression of NF-κB, TNF-α, and IL-1β, and as an inhibitor of kappa B (IκB) expression. TiO2 NPs significantly decrease Ca2+-ATPase and Ca2+/Mg2+-ATPase activity and enhance intracellular Ca2+ levels, and upregulate the expression of p-PKC and p-p38 MAPK in a dose-dependent manner. 70 The metal oxide NPs, in general, dose dependently activate NF-κB, a pathway that is mainly related with inflammation and the immune response. Furthermore, the release of inflammatory cytokines is related to the activation of NF-κB and JNKs. 71 Metal oxide NPs induce mRNA and protein expression of NF-κB and act as an inhibitor of κB kinase-α (IKK-α) and the nuclear factor-inducing kinase (NIK). Inflammatory response and nitrative stress enhanced by NPs are related to the NF-κB activation and Th1/Th2 imbalance. 72 In the event of oxidative stress, the IκB undergoes degradation, thus freeing NF-κB which then translocates into the nucleus to regulate the transcription of its target genes. 73
Inflammation induced oxidative stress via NPs and cell death
Cerium oxide NPs exhibit their toxicity through oxidative stress, which is provided by the p38-Nrf-2 signaling pathway-mediated induction of heme oxygenase-1 (HO-1). 74 In the same way, TiO2 NPs cause significant increases in splenic ROS accumulation. Subsequent lipid peroxidation and HO-1 expression occur via the p38-Nrf-2 signaling pathway. 75 Silver NPs (AgNPs) exposure of different sizes (4.7 and 42 nm) cause ROS production, glutathione depletion, and a slight inhibition of superoxide dismutase. 76
Exposure to silica NPs (SiNPs) activates the MAPKs signaling pathway, as shown by the increased expression of phosphorylated JNK and p38 MAPK, but inactivates the extracellular signal-regulated kinases (ERKs). It has been reported that NPs induced ROS production and oxidative stress through the MAPK pathway that regulates apoptosis.
77,78
Glutathione
Macrophages can be affected by NPs in various ways. Macrophage activation occurs following phagocytosis of a number of pathogenic particles leading to the release of cytokines. Exposure of primary macrophages to zinc oxide NPs (2.5 μg/ml) through the caveolae receptor pathway causes a significant enhancement of Ras, PI3K, and enhanced phosphorylation and subsequent activation of its downstream signaling pathways via ERK1/2, p38, and JNK MAPKs. Zinc oxide NPs induce the generation of RNS and the overexpression of cyclooxygenase-2 (COX-2), inducible NOS (iNOS), pro-inflammatory cytokines (IL-6, IFN-γ, TNF-α, IL-17, and regulatory cytokine IL-10), and MAPKs. 85 SiNPs disturb the NO/NOS system, induce inflammatory response, activate autophagy, and eventually lead to endothelial dysfunction via the PI3K/Akt/mTOR pathway. 86 In brief, oxidative stress can result in the release of pro-inflammatory mediators through the NF-κB, MAPK, and PI3K pathways. This suggests that NP-induced oxidative stress is linked to inflammation.
The lysosomal compartment is the most common intracellular site of NP sequestration and degradation. While the autophagy and endo-lysosomal pathways have the potential to influence the disposition of NMs, biopersistent NMs can, in turn, negatively impact these pathways. 87 Autophagy may be a cellular defense mechanism against oxidative stress toxicity. 88 Thereby, ROS-activated autophagy is a feedback mechanism to limit ROS-mediated cell activation by removing oxidatively damaged molecules and cell structures. 89 The autophagic activity is a key mechanism for cellular clearance of aggregated proteins leading to cell survival following exposure to NPs under 20 nm. 90 However, prolonged and uncontrolled autophagy can result in harmful cellular dysfunction and cell death. 83 NPs accumulation in lysosomes is one of the mechanisms of cytotoxicity, and its overload will lead to delayed clearance of particles. Thus, particle persistence can negatively impact lysosomal functions. 91
Conclusion
Immunotoxicity is a new and emerging field. There is a staggering heterogeneous group of molecular entities such as dendrimers, fullerenes, quantum dots, nanotubes, nanospheres, nanohorns, nanoshells, and liposomes, with highly variable physicochemical characteristics including size, shape, surface area, and reactivity, which in turn certainly interact with immune cells, like macrophages, monocytes, DCs, and lymphocyte, and trigger nonspecific inflammatory responses via generation of active oxygen species (oxidative burst) and release of pro-inflammatory cytokines. 19 Thus, interactions of NMs with the immune system can potentially lead to immunosuppression, hypersensitivity, immunogenicity, and autoimmunity, involving both innate and adaptive immune responses. Inherent physical and chemical NP characteristics more than likely influence their immunotoxicity. In this context, the successful adaptation of nanoformulations to clinical practice involves a detailed assessment of their safety profiles and includes evaluation of potential immunotoxicity. There is no comprehensive data available on the immunological effects of NPs and nanomedicines, and no clear understanding of the mechanisms involved to account for widely differing immunological effects. A better understanding of the structure–activity relationship between the physicochemical properties of a NP and their potential untoward effects on the immune system will help guide us toward safer drug delivery systems.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
