Abstract
This in vitro skin study determined absorption, diffusion, and binding rates of four [14C]-labeled nanoparticles (NPs): 12 nm Fe3O4, 32 nm Fe3O4@SiO2, 33 nm SiO2, and 78 nm SiO2 in each layer of human cadaver skin. In vitro microdialysis device and flow-through skin diffusion system were used to measure the binding affinity to the stratum corneum (SC) and permeability into/through skin layer of the four NPs with different physical–chemical properties, respectively, in short (30 min) and/or long (24 hours) exposures. Results show that NP size is an important factor affecting NP percutaneous absorption. The 12 nm Fe3O4 NPs reached the SC and viable epidermis; 32 nm Fe3O4@SiO2 core/shell NPs only reached SC. However, 33 nm and 78 nm silica NPs did not permeate SC. Similar patterns were observed for NP binding affinity to SC and dermatopharmacokinetic analysis using the tape stripping method. The binding affinity determination may be a useful method to efficiently screen skin penetration of NPs.
Keywords
Introduction
The use of nanoparticles (NPs) has recently increased dramatically in diverse areas including industry, agriculture, medicine, and cosmetics. 1 –5 Thus, their potential hazardous effects on human health are a matter of increasing interest and concern. The European Commission on Public Health 6 cautioned that NPs might have the ability to “stick” to molecular surfaces and cross cell membranes into a living system. NPs may interact with living systems, depending on the dimensions of both NPs and biomolecules such as proteins, if they are of similar size. 7 In a living system, if NPs dissolve easily they can spread through the entire body and exert chemical effects. 8 If NPs are not soluble, they may accumulate in biological systems and persist. 9
Skin has a surface area of approximately 1.8 m2 that acts as the primary physical barrier between our body and exogenous environmental factors. 10,11 Dermal exposure to NPs can result from medical, cosmetic, industrial, and agricultural products as well as environmental contamination. The permeability of NPs into and through the skin and their potential toxicity to human health has been recently investigated. 12 –15
Skin irritation potential and cytotoxic effects of various sizes of nanosilica particles (7 and 10–20 nm) were observed in a cultured human keratinocytes (CHK) model, but no similar observation was found when using a human skin equivalent model (HSEM) with nanosilica concentration at 500 g/ml. 16 Skin permeability and toxicological property of well-dispersed amorphous silica particles for sizes ranging from 70 nm to 1000 nm was investigated using an in vitro artificial skin model for the penetration assay and in vivo mice model for penetration and toxicity evaluation. 17 The result suggested that decreasing particle size (down to 70 nm) increases NPs skin permeability. Most current studies on the distribution and toxicology of NPs on systems or organs emphasize the liver, kidney, lungs, and gastrointestinal (GI) tract, and skin penetration studies are largely qualitative and sometimes contradictory. 18
Medical research of iron oxide (Fe3O4) and silica oxide (SiO2) NPs recently has been focused on quantitative pharmacokinetic studies of systemic circulation and organs such as liver, kidney, lungs, and GI tract, but minimal quantitative skin absorption and penetration study has been reported to date. 2 –5,14 Hence, research in skin absorption and penetration of NPs with different physicochemical properties needs to accelerate and gain advanced knowledge.
Here, we studied four metal oxide NPs of varying sizes (12 nm Fe3O4, 32 nm Fe3O4@SiO2, and 33 nm/78 nm SiO2) with carbon-labeling and prepared as aqueous topical dosing formulations to determine in vitro NP binding affinity to the human stratum corneum (SC), permeation capacity into and through human skin layer, and dermatopharmacokinetic analysis These radiolabeled NP formulations allowed for easy topical application, stability, and accurate measurement. They also possess similarities as well as different physicochemical properties which should provide benefits for skin binding and penetration study.
Materials and methods
NP synthesis and characterization
Four [14C]-radiolabeled NP topical formulations, 12 nm Fe3O4 (10 mg/ml water, 24,051 disintegrations per minute (DPM)/mg particles), 32 nm Fe3O4@SiO2 (10 mg/ml water, 1224 DPM/mg particles), 33 nm SiO2 (10 mg/ml water, 557 DPM/mg particles), and 78 nm SiO2 (10 mg/ml water, 372 DPM/mg particles) were synthesized, characterized by Oak Ridge National Laboratory (Oak Ridge, Tennessee, USA), and gifted from Lawrence Livermore National Laboratory (Livermore, California, USA). These NP formulations were individually administered in SC binding, skin penetration, and dermatopharmacokinetic studies without further dilution.
These synthesized NPs were modified with chemically grafted carboxylate groups (–COO–) on surfaces through a previously reported method,
19
and thus the NPs are hydrophilic and stable in aqueous phase. Particle sizes, polydispersity, and zeta potential (
Human skin preparations
The in vitro skin binding and penetration study using human (cadaver and surgical) skin was approved by the UCSF Human Research Committee. Skin samples were freshly dermatomed from six human adult cadavers (age 47–78 years, average 63 ± 12 years, three males and three females) within 24 h after death at the UCSF Anatomy Laboratory. Skin collected from abdominal and thigh regions is normally recommended for in vitro skin exposure assessment and modelling studies since it provides realistic dermal absorption values. 21 Dermatomed skin containing SC, “viable” epidermis, and partial dermis in approximately 500-µm thickness was stored at 0–4°C for approximately 2 weeks before being used or further separated to obtain SC membrane.
To ensure the integrity of each skin section, its physical condition was first examined using a 5–30× binocular stereo microscope (American Optical Corp., Buffalo, New York, USA) to eliminate any surface damage or potential holes. The thickness of the skin was then measured with an electronic digital caliper (Fisher Scientific, Fair Lawn, New Jersey, USA). Trans-epidermal water loss (TEWL) from SC was assessed before application of the test formulations. Following a brief (1–2 h) equilibrium of skin and phosphate-buffered saline, 0.1 M and pH 7.0, each skin sample was measured with a VapoMeter (Delfin Technologies Ltd, Kuopio, Finland). A skin specimen in which the TEWL value was less than 15 g/h m2 was considered acceptable. 22
SC membrane, used for in vitro SC binding study, was separated from the above dermatomed skin sample using modified methods of Kassis and Søndergaard 23 and Surber. 24 Briefly, the dermatomed skin was submerged in phosphate-buffered saline (0.05 M, pH 7.2) at 60°C for 1 min. SC-epidermis layer was then peeled from the dermis with dissection forceps and placed dermal side down on a filter paper soaked with 0.5% trypsin solution (w/w, pH 8.2) at 36°C for 15 h. After incubation, SC-epidermis layer was smoothed out on a flat surface and mushy epidermis removed by firmly rubbing with a moistened cotton-tipped applicator. The SC sheet was rinsed with water three times and then lifted out on a steel wire mesh to dry at 37°C in an evaporative incubator (Fisher Scientific) overnight. Dry SC was stored in a vacuumed desiccator (Fisher Scientific) at room temperature until use.
In vitro SC binding study
SC was cut into a 1 cm2 disc and embedded in 1 mL of dosing solution in a 10-mL glass vial in a shaking water bath (Fisher Scientific Isotemp Shaking Water Bath) at 37°C. Binding rates were determined at different incubation times (0.5 h, 2 h, 4 h, and every 4 h until reaching equilibrium). When the incubation ended, the SC samples were removed and rinsed thrice with 1 mL PBS(10 mM, pH 7.4), and all samples were individually measured for radioactivity.
In vitro skin penetration study
Human dermatomed skin samples were placed on a continuous flow-through diffusion cell system (PermeGear, Inc., Hellertown, Pennsylvania, USA). The donor chamber (above the skin, about 1 cm2 surface area) remained open to air. The receiving chamber (below the skin, 3 mL in volume) was filled to capacity with receptor fluid, PBS (10 mM, pH 7.4), and stirred magnetically at approximately 600 r/min. PBS was pumped through the diffusion cell at a rate of 4 mL/h using a Pump Pro MPL (Watson-Marlow, Inc., Wilmington, Massachusetts, USA). The cell was maintained at approximately 32°C using a LAUDA heating circulator (LAUDA, Lauda-Königshofen, Germany).
A single dose of 20 µL of each [14C]-NP dosing solution was applied to the skin surface (1 cm2) and left for 30 min or 24 h. After defined exposure times, skin surface dose residue was immediately washed off with a small piece of cotton pad, wetted once with 5% soap (soft soap, Colgate-Palmolive), twice wetted with water, and finally dried with a cotton pad. All washed samples per skin surface were placed into a glass scintillation vial to quantify radioactivity. Receptor fluid samples were individually collected into glass tubes at defined time points after dose using a Retriever Fraction Collector (Teledyne ISCO, Inc., Lincoln, Nebraska, USA) at 30 and 60 min, 2 h, and then every 2 h until the end of the experiment at 24 h.
After a 24-h experiment finished, the skin was removed from the glass cell and 10 consecutive D-Squame® tape strips (CuDerm Corp., Dallas, Texas, USA) were performed to remove SC adopted methods of Rougier et al. 25 and USFDA. 26 The first two stripped samples were counted separately as dose residues. The rest of the tape stripped samples (numbers 3 to 10) were pooled together in a glass scintillation vial. The remaining skin was separated into two parts, epidermis and dermis, using short-period heat contact (60°C for 2 min) as described by Kassis and Søndergaard. 23 All skin samples were digested by adding 2–5 mL of Soluene-350™ (PerkinElmer Life and Analytical Sciences, Boston, Massachusetts, USA) to be liquidized. An aliquot (1 mL) of the receptor fluid samples was collected from each time point for radioactive assay.
In vitro dermatopharmacokinetic study
For dermatopharmacokinetic study, the experiment procedure was similar to that of in vitro skin penetration study. The main differences were: (1) a single dose was applied to the skin surface (1 cm2) for 24 h of dermal exposure, (2) skin surface dose residue was washed off at 24 h post dose, and (3) after skin washes, the SC surface was tape stripped by 20 consecutive D-Squame® tape strips modified from methods of Weigmann et al. 27 and Leal et al. 28 The first two stripped samples were counted separately as dose residues. For the rest of the stripped samples (numbers 3 to 20), each was placed individually into a glass scintillation vial to examine NPs distribution in SC.
Radioactivity measurements
Samples were mixed with the Ultima Gold™ scintillation cocktail (PerkinElmer Life and Analytical Sciences, Boston, Massachusetts, USA) to measure radioactivity with a PerkinElmer Tri-Carb 2900 TR liquid scintillation spectrometer (Perkin Elmer Life and Analytical Sciences, Downer Grove, Illinois, USA).
Radioactivity of each sample was adjusted by subtracting background activity. The percentage of applied dose was then calculated by dividing by standard activity of each NP sample and multiplying by 100.
Data calculation and analysis
Dose calculations
Immediately before and after a single-dose application, a standard dose (20 µL for skin penetration or PK studies, or 1 mL for binding study) was weighed, and radioactivity was measured for each formulation.
Weight-normalized standard dose was expressed as radioactivity versus net dose weight (DPM/g standard dose). Based on the ratio of radioactivity and dose weight of the standard dose, radioactivity (DPM) of each individual sample was then calculated from its dose weight and normalized standard dose using the calculation formula below.

Radioactivity recovery of each sample as percent dose applied (%D) was calculated using the following formula:

Data analysis
Individual and mean (±SD) amounts of test NP equivalent in the SC, epidermis, dermis, receptor fluid, and wash samples are presented as percent administered dose at each time point. Binding rates were determined after reaching equilibrium, approximately in 20 h post.
Dermatopharmacokinetic analysis was performed with SigmaPlot (v13, Systat Software, Inc., San Jose, California, USA) for logarithm curve fitting.
Statistical difference of each NP between short (30-min) surface exposure and long exposure (24-h) was tested by Student’s
Results
Table 1 provides the physical characteristics of the four synthesized NP samples. The NPs used in this research are surface functionalized by carboxylate groups, thus they have hydrophilic surfaces and only effects from NPs of a hydrophilic nature were examined, as noted in the text description. Particle sizes, polydispersity, and
Size, zeta potential, and specific radioactivity of synthesized 14C-labeled nanoparticles.a
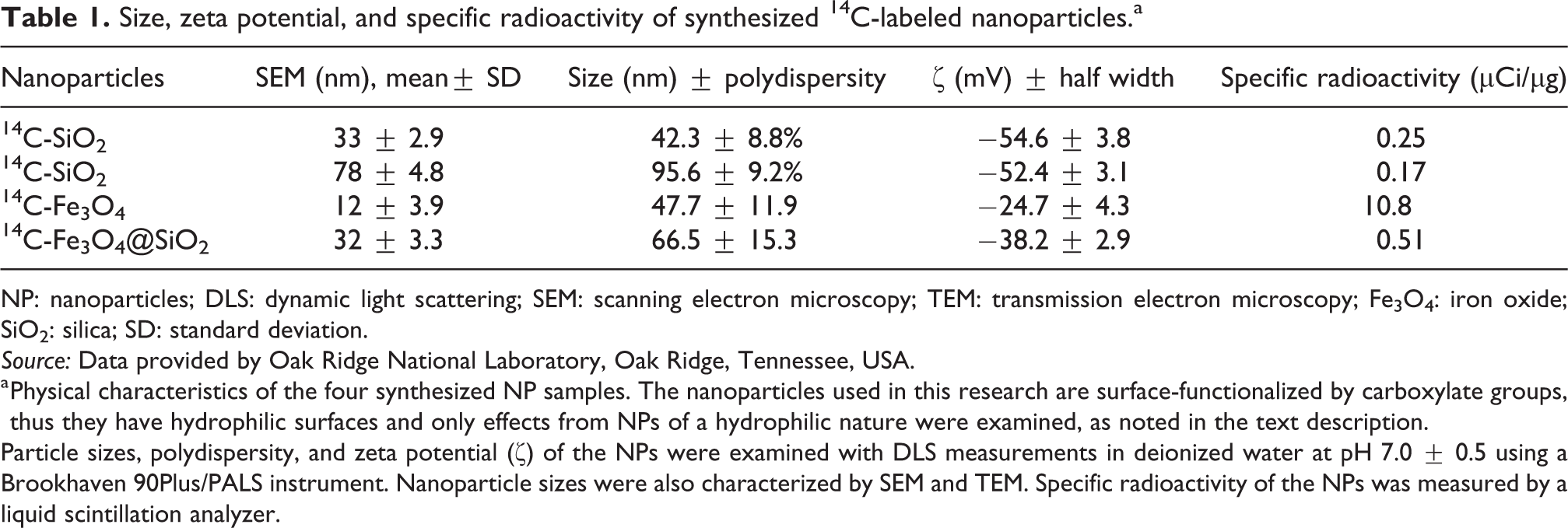
NP: nanoparticles; DLS: dynamic light scattering; SEM: scanning electron microscopy; TEM: transmission electron microscopy; Fe3O4: iron oxide; SiO2: silica; SD: standard deviation.
a Physical characteristics of the four synthesized NP samples. The nanoparticles used in this research are surface-functionalized by carboxylate groups, thus they have hydrophilic surfaces and only effects from NPs of a hydrophilic nature were examined, as noted in the text description.
Particle sizes, polydispersity, and zeta potential (
Table 2 summarizes the binding affinity of the four NP samples to SC after reaching equilibrium (20–24 h post incubation). Binding rates of SC to the NPs quickly reached 90% or more of its capacity 30 min post dosing/incubation and then slowly increased to equilibrium at approximately 20 h. We thus used the binding rate at 24 h as SC maximum binding capacity for these four NPs. [14C]-Fe3O4@SiO2 NP (32 nm) has the highest binding rate, followed by [14C]-Fe3O4 NP (12 nm) and [14C]-SiO2 (78 nm). [14C]-SiO2 (33 nm) has the lowest binding rate.
SC binding affinity (as percent of applied dose) of four NPs after reaching equilibrium.a

Fe3O4: iron oxide; NP: nanoparticle; SC: stratum corneum; SiO2: silica.
a Binding rates of the SC sheet to four NPs were determined at different incubation times (0.5 h, 2 h, 4 h, and every 4 h until reaching equilibrium). The rates quickly reached 90% of its capacity in 30 min post dosing/incubation, and then slowly increased to the maximum value around 20–24 h. The SC sheet was then washed three times, followed by counting radioactivity of each fraction. The results indicated the SC binding affinity for each type of NP. Each number represents the mean ± SD of six samples of percent applied dose (% D).
Table 3 explores the effect of a short dermal exposure (30 min) on penetration and distribution of four NPs in skin layers—SC, epidermis, and dermis, as well as receptor fluid. [14C]-Fe3O4 NPs (12 nm) permeated into SC (1.0% ±0.4) and epidermis (1.1% ± 0.6) but were not found in dermis and receptor fluid (RF). [14C]-Fe3O4@SiO2 NPs (32 nm) were only detected in SC (3.2% ±2.7), but not found in epidermis, dermis, and RF. No evidence of skin absorption/penetration was observed for the other two NPs, [14C]-SiO2 (33 nm) and [14C]-SiO2 (78 nm).
Radioactivity as percentage of applied dose of four NP samples following a 30-min dermal exposure in vitro.a
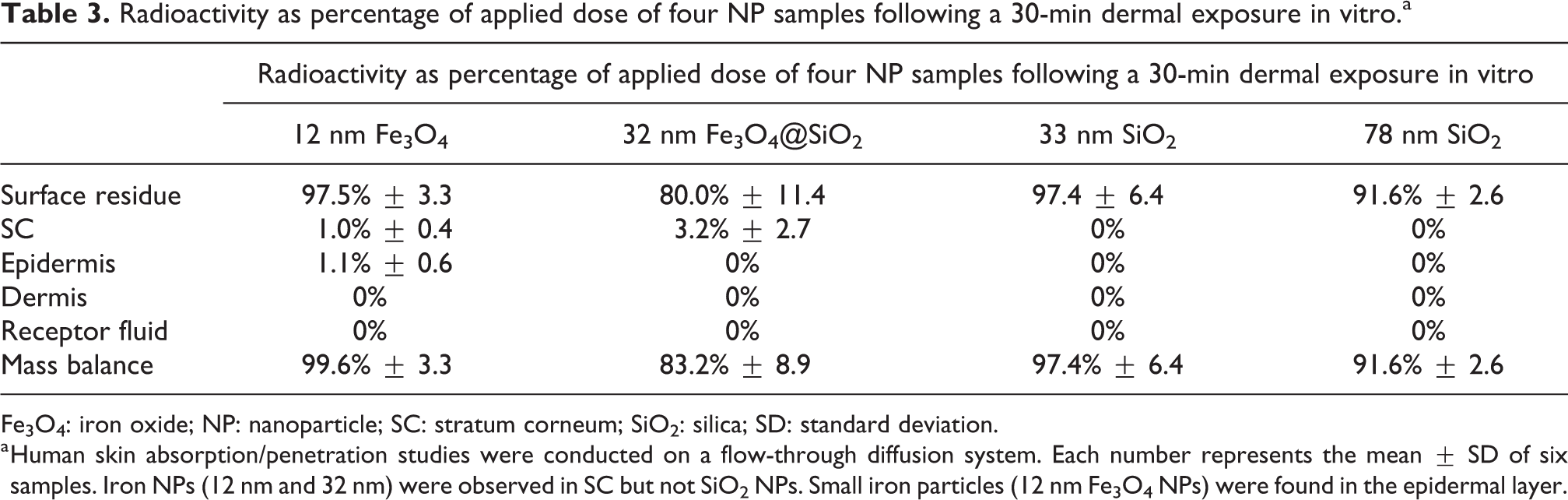
Fe3O4: iron oxide; NP: nanoparticle; SC: stratum corneum; SiO2: silica; SD: standard deviation.
a Human skin absorption/penetration studies were conducted on a flow-through diffusion system. Each number represents the mean ± SD of six samples. Iron NPs (12 nm and 32 nm) were observed in SC but not SiO2 NPs. Small iron particles (12 nm Fe3O4 NPs) were found in the epidermal layer.
Table 4 further demonstrates that after extending dermal exposure time to 24 h, all NPs permeated SC but only [14C]-Fe3O4 NPs (12 nm) were in both SC (0.4% ± 0.1) and epidermis (0.1% ± 0.1). No NPs were found in the dermis and receptor fluid collected at 24 h.
Radioactivity as percentage of applied dose of four NP samples following a 24-h dermal exposure in vitro.a
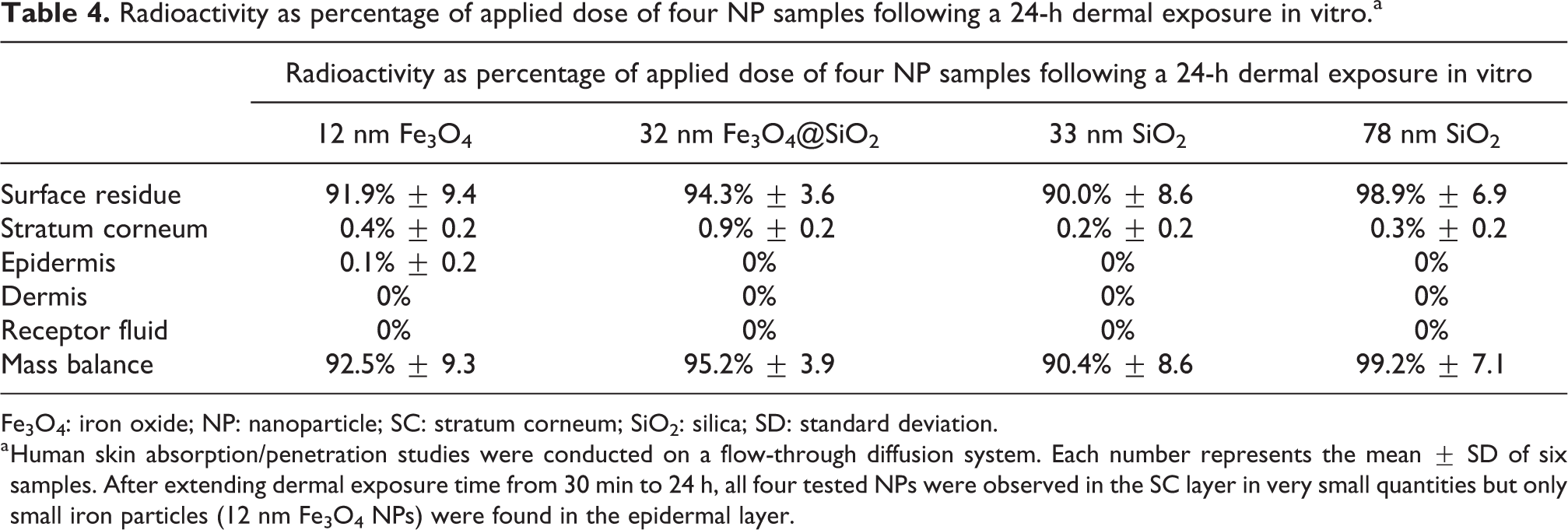
Fe3O4: iron oxide; NP: nanoparticle; SC: stratum corneum; SiO2: silica; SD: standard deviation.
a Human skin absorption/penetration studies were conducted on a flow-through diffusion system. Each number represents the mean ± SD of six samples. After extending dermal exposure time from 30 min to 24 h, all four tested NPs were observed in the SC layer in very small quantities but only small iron particles (12 nm Fe3O4 NPs) were found in the epidermal layer.
SC penetration kinetics of the NPs was studied using the in vitro SC tape stripping method. Twenty-four hours post topical application, tested skin was tape stripped 20 times to remove approximately 50% of SC by thickness, and the results represent the distribution of NPs in the SC layer. [14C]-Fe3O4 NPs (12 nm) were observed as deep as 50% of SC layer in depth (Figure 1), while 32 nm Fe3O4@SiO2 NPs were mostly found in the first 10 strips (Figure 2). The 33 nm and 78 nm SiO2 NPs, however, were only found in the first four tape strips. In general, test substance retained in the top few layers of SC (i.e. contained in the first few tape strips) may be removed by desquamation and therefore may not be absorbed systemically. 29,30 We therefore concluded that no actual SC absorption/penetration was present for two SiO2 NPs (Figures 3 and 4).
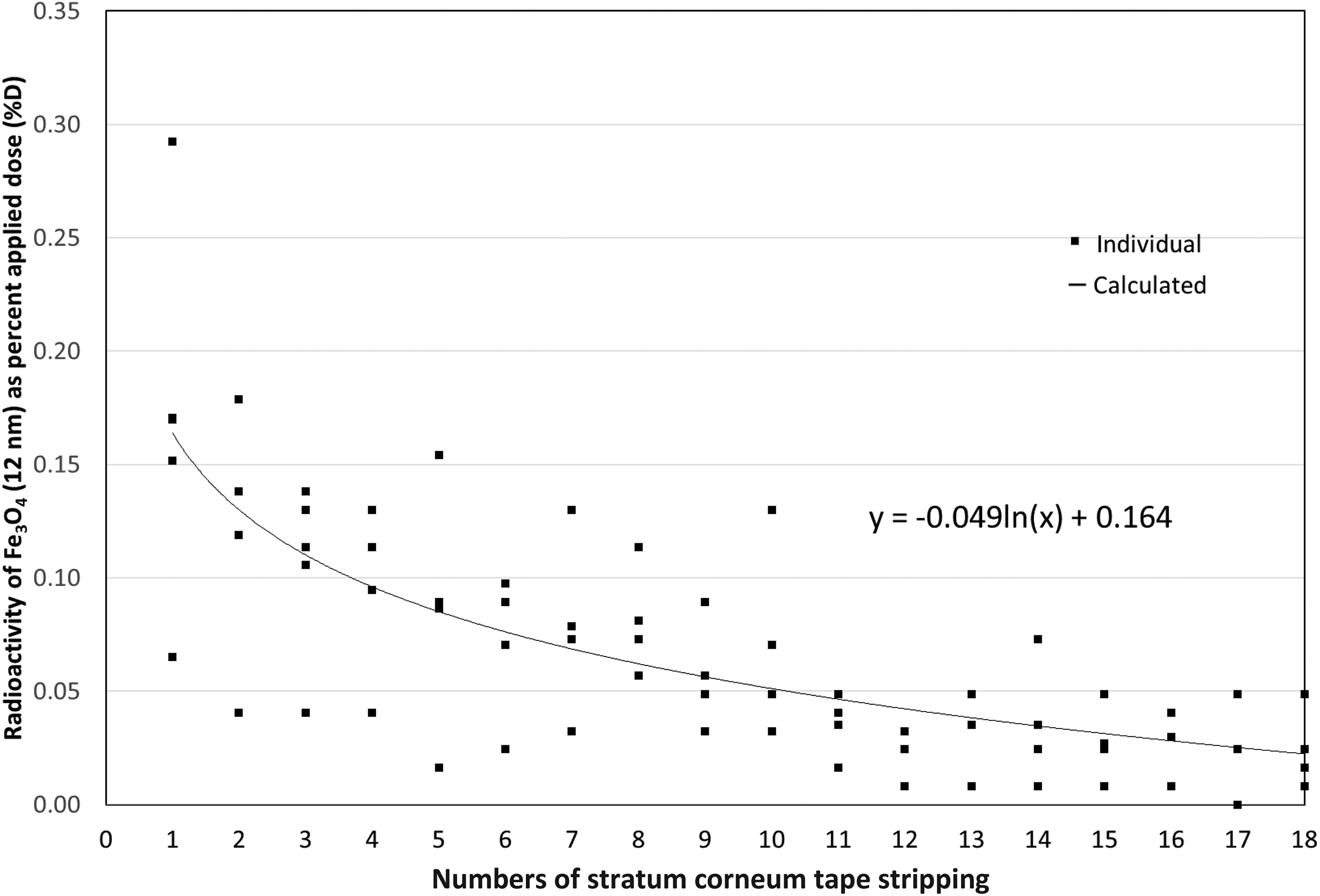
Dermatopharmacokinetic analysis of Fe3O4 (12 nm) nanoparticles. Human skin was exposed to testing NPs for 24 h and then SC samples were collected via 20 consecutive D-Squame® tape strips. The first two stripped samples were considered as unabsorbed dose residues. Numbers 3 to 20 stripping samples (total 18) were measured individually to examine NPs distribution in SC. Small size (12 nm) Fe3O4 NPs easily penetrate into the SC layer. Solid squire symbol: individual observed, line: calculated. Each number represents the mean ± SD of five samples. Fe3O4: iron oxide; NP: nanoparticle; SC: stratum corneum; SiO2: silica; SD: standard deviation.
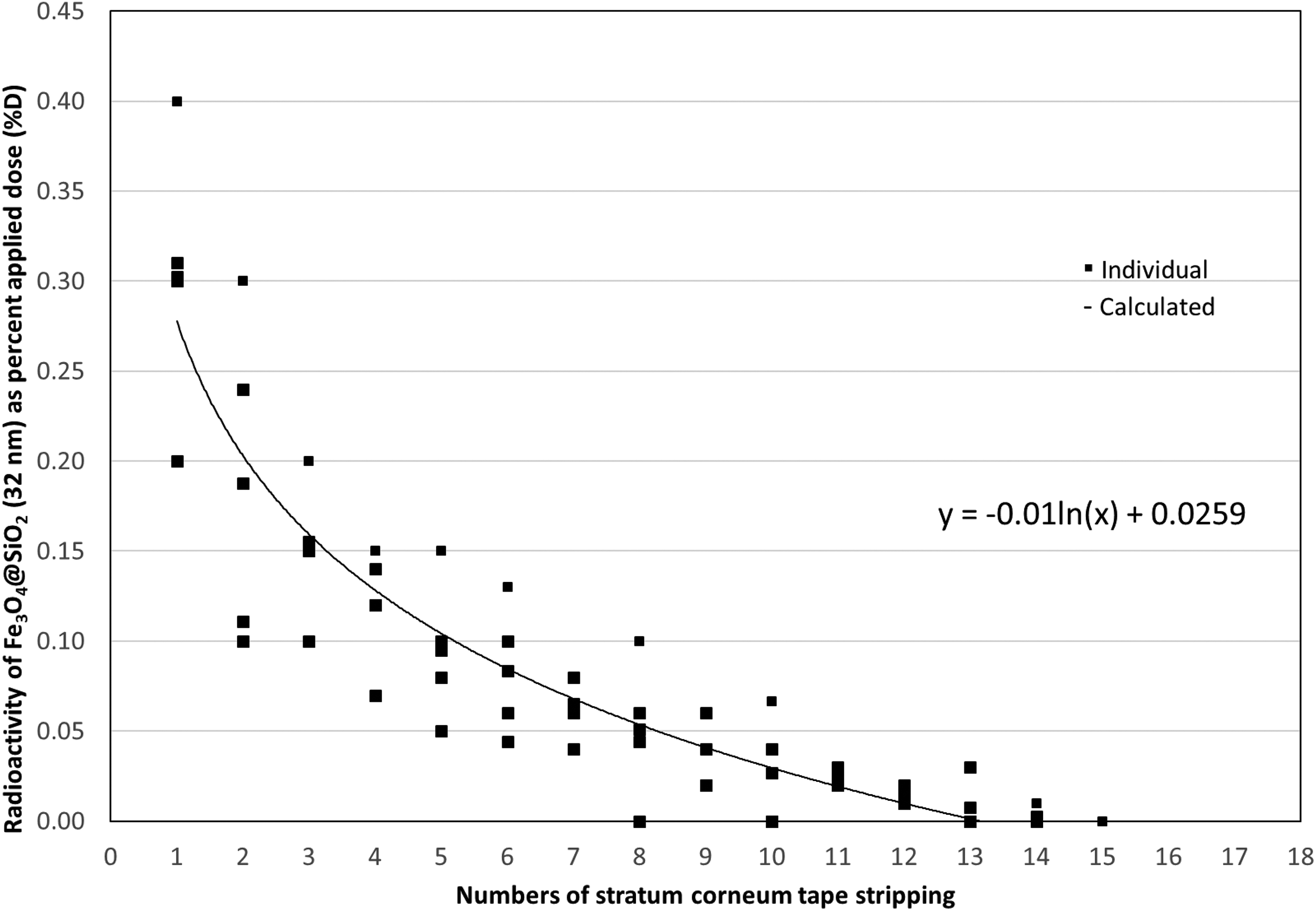
Dermatopharmacokinetic analysis of Fe3O4 @SiO2 (32 nm) NPs. Human skin was exposed to testing NPs for 24 h and then SC samples were collected via 20 consecutive D-Squame® tape strips. The first two stripped samples were considered as unabsorbed dose residues. Numbers 3 to 20 stripping samples (total 18) were measured individually to examine NPs distribution in SC. Enlarging particle size to 32 nm, Fe3O4 @SiO2 NPs reduced the amount to penetrate into the SC layer. Solid squire symbol: individual observed, line: calculated. Each number represents the mean ± SD of five samples. Fe3O4: iron oxide; NP: nanoparticle; SC: stratum corneum; SiO2: silica; SD: standard deviation.
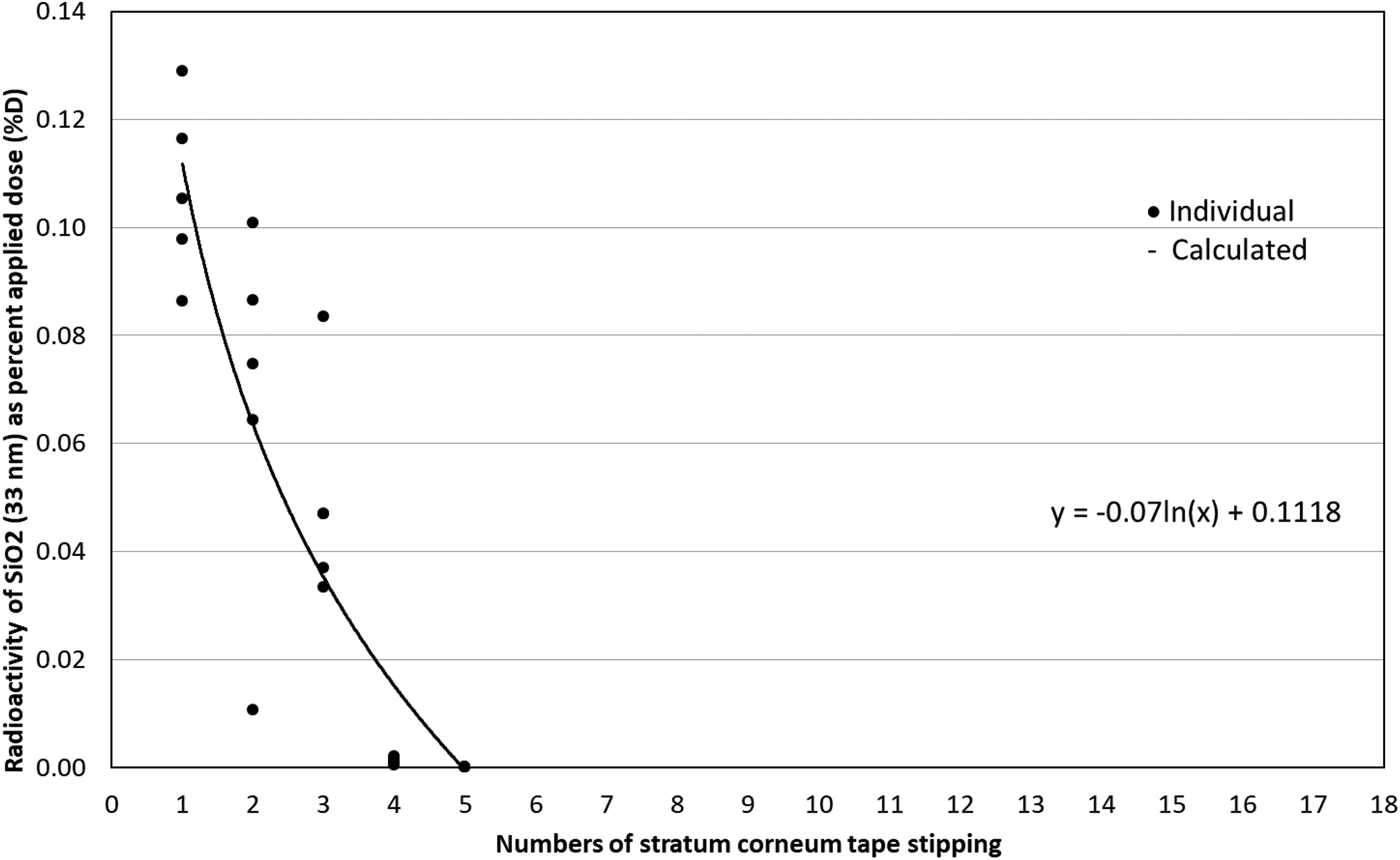
Dermatopharmacokinetic analysis of SiO2 (33 nm) NPs. Human skin was exposed to testing NPs for 24 h and then SC samples were collected via 20 consecutive D-Squame® tape strips. The first two stripped samples were considered as unabsorbed dose residues. Numbers 3 to 20 stripping samples (total 18) were measured individually to examine NPs distribution in SC. Particle size (33 nm) SiO2 NPs are mostly retained in the superficial layers of the SC (numbers 3–6 of tape strips). Solid circle symbol: individual observed, line: calculated. Each number represents the mean ± SD of five samples. NP: nanoparticle; SC: stratum corneum; SiO2: silica; SD: standard deviation.
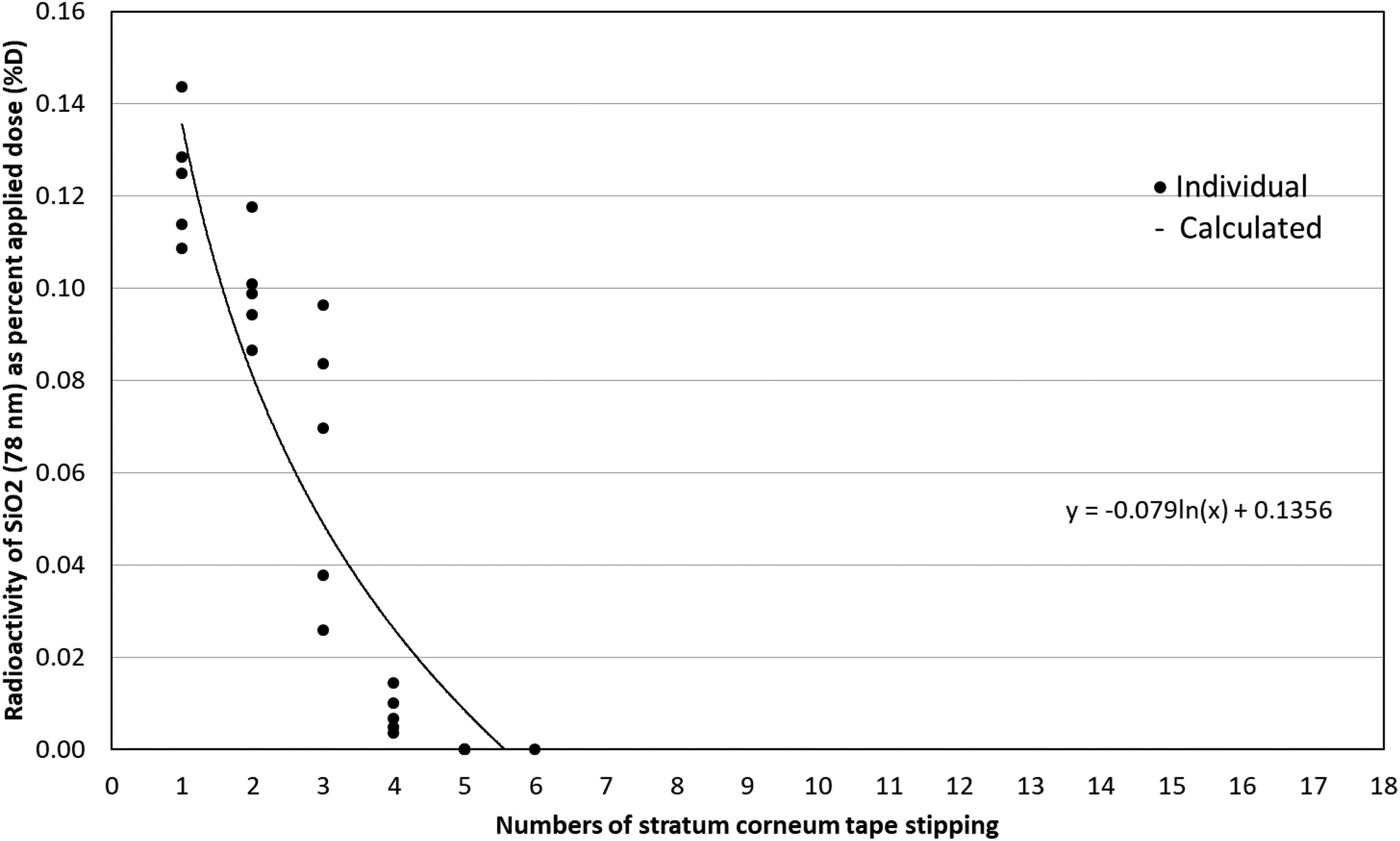
Dermatopharmacokinetic analysis of SiO2 (78 nm) NPs. Human skin was exposed to testing NPs for 24 h and then SC samples were collected via 20 consecutive D-Squame® tape strips. The first two stripped samples were considered as unabsorbed dose residues. Numbers 3 to 20 stripping samples (total 18) were measured individually to examine NPs distribution in SC. Particle size (78 nm) SiO2 NPs are mostly retained in the superficial layers of the SC (numbers 3–6 of tape strips). Solid circle symbol: individual observed, line: calculated. Each number represents the mean ± SD of 5 samples. NP: nanoparticle; SC: stratum corneum; SiO2: silica; SD: standard deviation.
Discussion
This study demonstrated that 12 nm Fe3O4 NPs penetrated into SC and through to viable epidermis (Table 3) 30 min post dermal exposure, while other larger sizes of NPs (32 nm Fe3O4@SiO2 or 33 nm and 78 nm SiO2) needed much longer exposure times (24 h) to partition into SC (Table 4) but not in the epidermis. The NPs’ size differences contributed to these variations in their permeability. Similar observations were reported by Baroli et al. 31 and Rouse et al. 32 that only when NP size is small enough, such as when iron oxide size is 10 nm or less, is it possible to diffuse through the SC to the epidermis. 31 Thus, NP size is an important factor in determination of SC permeability.
Size-dependent NP penetration is not only observed in normal integrity skin but also in disrupted SC. Effect of partial SC disruption on silica oxide NPs (40 nm size or larger) penetration was tested after cyanacrylate skin surface stripping (CSSS) pretreatment, 33 or CSSS pretreatment plus positive or negative surface charging as skin penetration promotion. 7 Results indicated that even when SC barrier structure was partially disrupted, only 40 nm SiO2 NPs were found in epidermal Langerhans cells (LC), and no larger sizes of silica NPs penetrated into the epidermis. 7,33 The results further emphasized the importance of NP size in determination of skin (deep) penetration even when the skin barrier has been disrupted.
NP size, however, may not be the only factor in determining SC permeability. Two structurally different NPs Fe3O4@SiO2 and SiO2 had similar sizes 32 nm and 33 nm, respectively, but showed different SC penetration rates. When they were topically dosed on the skin in vitro for 24 h, total SC penetration (% dose) of 32 nm Fe3O4@SiO2 was four times that of 33 nm SiO2 (Table 4). This result demonstrated that NPs’ structurally related physicochemical property variation could result in different skin permeability.
NPs’ structural variations also change their SC binding rates. The main barrier function of the skin is dominated by protein (keratin, 70–80%) and lipids. 34 Chemicals’ binding affinities to SC protein/lipids are associated with their skin permeability. 35 As presented in Table 2 after 24 h of incubation, SC binding rate of 32 nm Fe3O4@SiO2 was also four times of that of 33 nm SiO2. The results suggest that Fe3O4NPs have higher SC affinity than that of SiO2 NPs. Thus, determination of the SC binding affinity of NPs may be a useful method to estimate NP diffusion in SC.
SC has heterogeneous structure composed of 15–20 layers of flattened corneocytes embedded in lipid bilayers. 36 SC tape stripping method measures SC mass, absorption, reservoir, and pharmacokinetics of testing chemicals in a defined exposure period. 37,38
Tables 3 and 4 provided information of radioactivity recovery of total SC absorbance for each tested NP (% dose) after pooling together all stripped samples to count. The results demonstrated that NP size is an important factor in determination of SC permeability and extension of exposure did not significantly alter the permeability of these NPs (
Figures 1 to 4 emphasized NP distribution in the SC presented by individual tape-stripped sample after a 24-h exposure. These continuously collected SC stripped samples gave the concentration profile of a NP in SC in relation to the relative SC depth.
Small size Fe3O4 NPs (12 nm) reached approximately 40#60% of the whole SC thickness, while larger 32 nm Fe3O4@SiO2 was approximately 20% deep (Figures 1 and 2). This further supports the importance of NP size in skin penetration. However, compared to 32 nm Fe3O4@SiO2, 33 nm SiO2 only reached the superficial layers of the SC (Figure 3) and showed no difference compared to 78 nm SiO2 (Figure 4). This may be explained by NPs’ SC binding affinity.
Based on structural and functional differences, SC can be further divided into three layers. The superficial layer (0–30% SC depth) of SC has highly folded keratin filaments, which are loosely bound to outer corneocytes to allow chemicals to pass quickly during influx and efflux, and retaining. 39,40 Thus, the initial tape strips removed higher amounts of SC protein and retained testing chemicals. The intermediate (30–70%) and the bottom (80–100%) layers exhibited distinct barrier properties to chemical permeation. The major difference is that the former has unfolded keratin filaments and the most water binding, while in the latter the water binding sites are already occupied with water and cannot swell substantially. 39,40 Thus, the quantity of SC removed by tape stripping is not linearly proportional to the number of tape strips removed and the values progressively decreased as the tape number increased. As illustrated by Figures 1 to 4, whether or not these NPs penetrated into the low part of the SC, they were present in high amount in the superficial layer of the SC and then quickly or slowly declined.
Knowledge of the concentration profile of a NP or chemical in SC in relation to the relative SC depth is important to predict its permeation status, whether it will desquamate, retain, or further diffuse to systemic. The superficial layer of SC is involved in the process of desquamation, therefore the amount of chemical found in this layer is usually not considered as skin absorbed. Chemicals retained in the intermediate and bottom layers, however, might further penetrate into the deep tissues or systemic. 41
The relation between the number of tape strips and SC depth (relative thickness of the horny layer) can be used to estimate chemical penetration and distribution in SC, in which the total absorbance corresponds to cumulated values of pseudo-absorption of all removed tape strips. Based on this relation, the relative amount of SC stripped with each tape can be calculated in terms of the percentage of SC thickness. 42,43 For example the range of relative SC removed (%) by 20 tape strips can be from 40% to 60% depending upon variations of the test chemical and vehicle used and the skin sample’s regional and physical variations. 37,44
In conclusion, this study confirmed that NP size is an important factor affecting percutaneous absorption of NPs. Only when Fe3O4 NP size was 12 nm could it reach the epidermis while other larger sizes of NPs (Fe3O4@SiO2 NP and silica NPs) were only retained in SC. Even with the exposure extended from 30 min to 24 h, penetration was not enhanced. Binding rates of these NPs to SC were similar to the amounts of the NPs retained in SC after a 24-h dermal exposure. Fe3O4 NPs had relatively higher binding rates to SC compared to those of SiO2 NPs. The relationship of these NPs to skin was further demonstrated by in vitro dermatopharmacokinetic analysis using SC tape stripping method, which gave a clear illustration of NPs. The binding affinity determination may be a useful method to quickly screen skin penetration of NPs.
At this point, we await confirmation of these results with in vivo studies—evaluating not only the skin penetration effect of different NPs with various particle sizes but also related permeation kinetics as well.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial supports for the research, authorship, and/or publication of this article: This work was supported by the Defense Threat Reduction Agency (DTRA) Grant: HDTRA1-14-0005 UCSF (BRBAA11-PerC-9-2-0054-Base). This manuscript was also supported by University of California San Francisco (UCSF) library's “Open Access Publishing Fund.”
