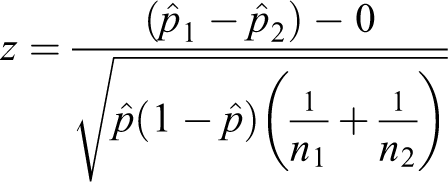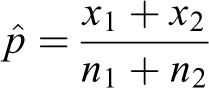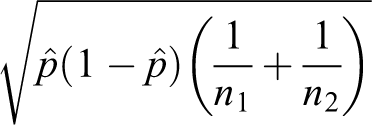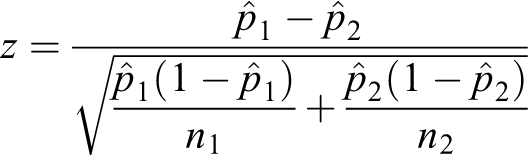Abstract
This study presents an analysis of species, sex concordance in tumorigenicity among National Toxicology Program (NTP) chemicals tested for carcinogenicity by feed. It includes mutagenicity and other genetic toxicology test results, as well as Log P values. A total of 213 chemicals were tested in 212 2-year studies by exposing F334/N rats and B6C3F1 mice in their feed. Twenty-two of the 213 chemicals tested were clearly neoplastic in male and female rats and mice. Thirteen of these 22 chemicals were positive in at least one Ames test representing a prediction accuracy of 59.1%. Seventy-six of 213 chemicals tested were not neoplastic in either rodent species. Of the 22 ubiquitously neoplastic chemicals, 14 were concordant for developing tumors at the same anatomical site both across species and across sex within species. Of the 22 ubiquitously neoplastic chemicals, none were concordant for developing tumors at the same anatomical site across species, but not across sex within species. The neoplastic response to chemicals administered in feed displays more concordance within either male or female rats, or male and female mice, than between male rats and male mice, or female rats and female mice. In the NTP feed studies, negative Ames test results are more predictive of neoplastic responses than positive Ames test results. Log P values for the chemicals that produced tumors in male and female rats and mice were compared to chemicals that did not produce tumors. A statistical comparison between these Log P values showed no relationship between Log P values and neoplasticity.
Introduction
The National Toxicology Program (NTP) is a branch of the US Department of Health and Human Services. A major current emphasis of NTP is “The Toxicology in the 21st Century: The Role of the National Toxicology Program.”
1
NTP describes this program as follows: The Role of the National Toxicology Program is to support the evolution of toxicology from a predominantly observational science at the level of disease-specific models to a predominantly predictive science focused upon a broad inclusion of target-specific, mechanism-based, biological observations.
In the current study, we analyze the results from 213 chemicals tested in 212 2-year studies conducted by exposing F334/N rats and B6C3F1 mice in their feed. Additionally, 31 2-year feed studies have been conducted in Osborne-Mendel rats and B6C3F1 mice on 34 different chemicals. The results from these 31 studies are analyzed and discussed separately. The neoplasticity of each chemical was analyzed for tumor incidence by species–sex category, tumor site concordance across species, and tumor site concordance across sex within species. When available, the Ames Salmonella mutagenicity assay results, and any results from a test for genotoxicity other than the Ames test, were correlated with the neoplasticity results.
NTP considers results from the Ames assay test to be very important in its deliberations as illustrated by the following statement from a recent Report on Carcinogens.
3
DNA reactivity combined with Salmonella mutagenicity is highly correlated with induction of carcinogenicity in multiple species/sexes of rodents and at multiple tissue sites.
4
A positive response in the Salmonella test was shown to be the most predictive in vitro indicator for rodent carcinogenicity (89% of the Salmonella mutagens are rodent carcinogens).
5,6
Additionally, no battery of tests that included the Salmonella test improved the predictivity of the Salmonella test alone…
Statistical methods
The following tests were applied to assess the statistical significance of the differences in proportions. 8
Pooled test
The null hypothesis is as follows:
The formula for the pooled test statistic comparing two proportions is as follows:
where
The standard error is as follows:
Unpooled test
The null hypothesis is as follows:
Significance tested at p < 0.05.
Results for 212 feed studies in F344/N rats and B6C3F1 mice (213 chemicals tested)
Clear evidence of neoplasia in male rats, female rats, male mice, and female mice
Twenty-two of 213 chemicals tested were clearly neoplastic in male and female rats, and in male and female mice (Table 1). These 22 chemicals are the following: nitrilotriacetic acid (NTA; CASRN 139-13-9) and nitriloacetic acid, trisodium salt (Na3-NTA-H2O; CASRN 18662-53-8), 2-methyl-1-nitroanthraquinone (CASRN 129-15-7), 4,4′-thiodianiline (CASRN 139-65-1), 4-chloro-o-phenylenediamine (CASRN 95-83-0), tris (2,3-dibromopropyl) phosphate (TBP; CASRN 126-72-7), 2,4-diaminoanisole sulfate (CASRN 615-05-4), o-anisidine hydrochloride (CASRN 134-29-2), 3-amino-9-ethylcarbazole hydrochloride (CASRN 132-32-1), cupferron (CASRN 135-20-6), p-cresidine (CASRN 120-71-8), o-toluidine hydrochloride (CASRN 636-21-5), Michler’s ketone (CASRN 90-94-8), 4,4′-oxydianiline (CASRN 101-80-4), di(2-ethylhexyl)phthalate (CASRN 117-81-7), C.I. Basic Red 9 monohydrochloride (pararosaniline; CASRN 569-61-9), 1-amino-2,4-dibromoanthraquinone (CASRN 81-49-2), 1,2,3-trichloropropane (CASRN 96-18-4), ethylene thiourea (ETU; CASRN 96-45-7), polybrominated biphenyls (CASRN 67774-32-7), 2,2-bis(bromomethyl)-1,3-propanediol (FR-1138® [Dow Chemical Company]; CASRN 3296-90-0), o-nitrotoluene (CASRN 88-72-2), and acrylamide (CASRN 79-06-1; Table 1).
Compounds that produced clear evidence of neoplasia in male rats, female rats, male mice, and female mice

Thirteen of these 22 chemicals were positive in the Ames test (13/22 observed vs 22/22 expected, p pooled = 0.0004; p unpooled = 0.0000). These 13 chemicals are the following: 2-methyl-1-nitroanthraquinone (CASRN 129-15-7), 4,4′-thiodianiline (CASRN 139-65-1), 4-chloro-o-phenylenediamine (CASRN 95-83-0), TBP (CASRN 126-72-7), 2,4-diaminoanisole sulfate (CASRN 615-05-4), 3-amino-9-ethylcarbazole hydrochloride (CASRN 132-32-1), cupferron (CASRN 135-20-6), p-cresidine (CASRN 120-71-8), 4,4′-oxydianiline (CASRN 101-80-4), C.I. Basic Red 9 monohydrochloride (pararosaniline; CASRN 569-61-9), 1-amino-2,4-dibromoanthraquinone (CASRN 81-49-2), 1,2,3-trichloropropane (CASRN 96-18-4), and 2,2-bis(bromomethyl)-1,3-propanediol (FR-1138; CASRN 3296-90-0; Table 1).
The Ames test was only able to predict an across-the-board neoplastic response in 59.1% of this set of 22 chemicals. Eighteen of the 22 ubiquitously neoplastic chemicals were positive in at least one other test of genetic toxicity (18/22 observed vs 22/22 expected, p pooled = 0.0180; p unpooled = 0.0135). Overall, a single positive result in the Ames test was less common than a single positive test in a different test for genetic toxicity (13/22 Ames positive vs 18/22 other genetic test positive, p pooled = 0.0493; p unpooled = 0.0440).
Of the 22 ubiquitously neoplastic chemicals, 14 were concordant for developing tumors at the same anatomical site in both rats and mice, and in both males and females (14/22 observed vs 22/22 expected, p pooled = 0.0001; p unpooled = 0.0000). These 14 chemicals are the following: NTA (CASRN 139-13-9) and nitriloacetic acid, trisodium salt (Na3-NTA-H2O; CASRN 18662-53-8), 4,4′-thiodianiline (CASRN 139-65-1), TBP (CASRN 126-72-7), 2,4-diaminoanisole sulfate (CASRN 615-05-4), o-anisidine hydrochloride (CASRN 134-29-2), 3-amino-9-ethylcarbazole hydrochloride (CASRN 132-32-1), cupferron (CASRN 135-20-6), p-cresidine (CASRN 120-71-8), 4,4′-oxydianiline (CASRN 101-80-4), di(2-ethylhexyl)phthalate (CASRN 117-81-7), 1-amino-2,4-dibromoanthraquinone (CASRN 81-49-2), 1,2,3-trichloropropane (CASRN 96-18-4), ETU (CASRN 96-45-7), and polybrominated biphenyls (CASRN 67774-32-7).
Of these same 22 chemicals, one (o-toluidine hydrochloride (CASRN 636-21-5)) was completely discordant for developing tumors at the same anatomical site in both rats and mice, and in both males and females for both rats and mice (1/22 observed vs 0/22 expected, p pooled = 0.1559; p unpooled = 0.1530). Of the 22 ubiquitously neoplastic chemicals, none (0/22) were concordant for developing tumors at the same anatomical site in just male rats and male mice, or in just female rats and female mice, but not in both male and female rats, or in male and female mice. Of the 22 ubiquitously neoplastic chemicals, 7 were discordant for developing tumors at the same anatomical site in rats and mice but were concordant for developing tumors at the same anatomical site across sex within species (7/22 observed vs 0/22 expected, p pooled = 0.0020; p unpooled = 0.0007). These seven chemicals are the following: 2-methyl-1-nitroanthraquinone (CASRN 129-15-7), 4-chloro-o-phenylenediamine (CASRN 95-83-0), Michler’s ketone (CASRN 90-94-8), C.I. Basic Red 9 monohydrochloride (pararosaniline; CASRN 569-61-9), 2,2-bis(bromomethyl)-1,3-propanediol (FR-1138; CASRN 3296-90-0), o-nitrotoluene (CASRN 88-72-2), and acrylamide (CASRN 79-06-1; Online Appendix A).
No evidence of neoplasia in male rats, female rats, male mice, female mice
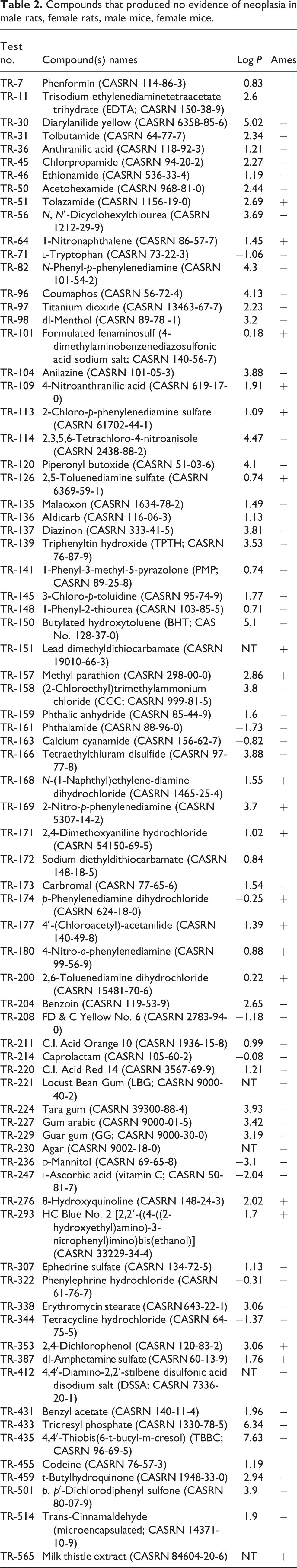
Seventy-six of 213 chemicals tested were not neoplastic in male rats, female rats, male mice, or female mice (Table 2). Twenty of the 76 ubiquitously non-neoplastic chemicals were positive in at least one reported Ames test (20/76 observed vs 0/76 expected, p pooled = 0.0001; p unpooled = 0.0001). These 20 chemicals are the following: tolazamide (CASRN 1156-19-0), 1-nitronaphthalene (CASRN 86-57-7), formulated fenaminosulf (4-dimethylaminobenzenediazosulfonic acid sodium salt; CASRN 140-56-7), 4-nitroanthranilic acid (CASRN 619-17-0), 2-chloro-p-phenylenediamine sulfate (CASRN 61702-44-1), 2,5-toluene diamine sulfate (CASRN 6369-59-1), lead dimethyldithiocarbamate (CASRN 19010-66-3), methyl parathion (CASRN 298-00-0), N-(1-naphthyl)ethylene-diamine dihydrochloride (CASRN 1465-25-4), 2-nitro-p-phenylenediamine (CASRN 5307-14-2), 2,4-dimethoxyaniline hydrochloride (CASRN 54150-69-5), p-phenylenediamine dihydrochloride (CASRN 624-18-0), 4′-(chloroacetyl)-acetanilide (CASRN 140-49-8), 4-nitro-o-phenylenediamine (CASRN 99-56-9), 2,6-toluenediamine dihydrochloride (CASRN 15481-70-6), 8-hydroxyquinoline (CASRN 148-24-3), HC Blue No. 2 [2,2′-((4-((2-hydroxyethyl)amino)-3-nitrophenyl)imino)bis(ethanol)] (CASRN 33229-34-4), 2,4-dichlorophenol (CASRN 120-83-2), dl-amphetamine sulfate (CASRN 60-13-9), and milk thistle extract (CASRN 84604-20-6). This result could be considered as a false-positive rate of 26.3%. Six of the 76 ubiquitously non-neoplastic chemicals were not tested in another genetic toxicology test other than the Ames test. Of the 70 non-neoplastic chemicals tested in another genetic toxicology test, 39 were positive in at least one non-Ames genetic toxicology test (39/70 observed vs 0/70 expected, p pooled = 0.0000; p unpooled = 0.0000). This represents a false-positive rate of 55.7%. The false-positive rate for tests other than Ames is higher than for the Ames tests (20/76 Ames positive vs 39/70 other genetic toxicology test positive, p pooled = 0.0001; p unpooled = 0.0001).
Compounds that produced no evidence of neoplasia in male rats, female rats, male mice, female mice.
Three of four categories of species/sex were clear
Eighteen of 213 chemicals tested were neoplastic in ¾ categories of rodent species and sex (Table 3). Eleven of these 18 chemicals were positive in the Ames test (11/18 observed vs 18/18 expected, p pooled = 0.0016; p unpooled = 0.0004). These eleven chemicals are as follows: hydrazobenzene (CASRN 122-66-7), 3-(chloromethyl) pyridine hydrochloride (CASRN 6959-48-4), phenazopyridine hydrochloride (CASRN 136-40-3), 1-amino-2-methylanthraquinone (CASRN 82-28-0), 5-nitroacenaphthene (CASRN 602-87-9), 5-nitro-o-anisidine (CASRN 99-59-2), 1,5-naphthalenediamine (CASRN 2243-62-1), 2,4,5-trimethylaniline (CASRN 137-17-7), 2,4-diaminotoluene (CASRN 95-80-7), 4,4′-methylenebis-(N, N-dimethyl)benzeneamine (CASRN 101-61-1), and o-nitroanisole (CASRN 91-23-6).
Compounds that produced three of four categories of species/sex were clear.
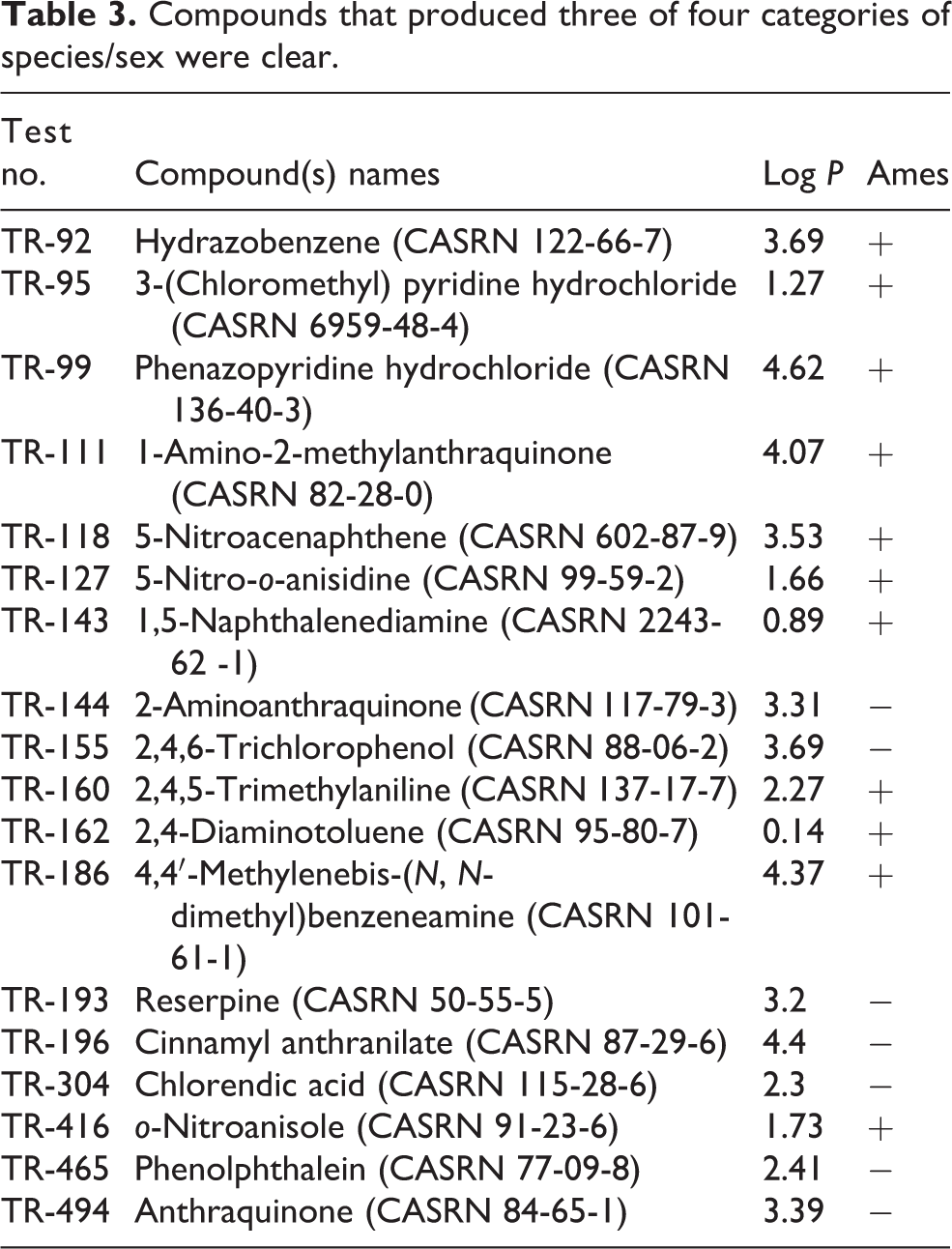
Fifteen of these 18 chemicals were positive in another genetic toxicology test (15/18 observed vs 18/18 expected, p pooled = 0.0352; p unpooled = 0.0289). Overall, a single positive result in the Ames test was less common than a single positive test in a different test for genetic toxicity (11/18 Ames positive vs 15/18 other genetic toxicology test positive, p pooled = 0.0683; p unpooled = 0.0622). Seven of these 18 chemicals were negative in the Ames test (7/18 observed vs 0/18 expected, p pooled = 0.0000; p unpooled = 0.0000). These seven chemicals include 2-aminoanthraquinone (CASRN 117-79-3), 2,4,6-trichlorophenol (CASRN 88-06-2), reserpine (CASRN 50-55-5), cinnamyl anthranilate (CASRN 87-29-6), chlorendic acid (CASRN 115-28-6), phenolphthalein (CASRN 77-09-8), and anthraquinone (CASRN 84-65-1).
Three of these 18 chemicals were negative in another genetic toxicology test (3/18 observed vs 0/18 expected, p pooled = 0.0352; p unpooled = 0.0289). Overall, a single negative result in the Ames test was more common than a single negative test in a different test for genetic toxicity (7/18 Ames negative vs 3/18 other genetic toxicology test negative, p pooled = 0.0683; p unpooled = 0.0622).
Two chemicals that were neoplastic in ¾ categories of rodent species and sex were concordant for tumor development both across species, and across sex within species. Hydrazobenzene in the feed induced liver tumors in both rats and mice, and also in male and female rats. 2,4,5-Trimethylaniline in the feed induced hepatocellular carcinomas in both rats and mice, and similar neoplastic nodules in male and female rats. Two chemicals that were neoplastic in ¾ categories of rodent species and sex were discordant for tumor development both across species, and across sex within species, that is, 5-nitro-o-anisidine and reserpine (Online Appendix A).
Fourteen of 18 chemicals that were neoplastic in ¾ categories of rodent species and sex were discordant for tumor development across species, but concordant for tumor development across sex within species. Eight of 14 chemicals were concordant across sex within species for mice. Of these eight chemicals, reportedly concordant between male and female mice, five induced liver tumors. Six of 14 chemicals were concordant across sex within species for rats.
Overall, within-sex concordance for mice was somewhat more common than within-sex concordance for rats (8/14 vs 6/14, p pooled = 0.2248; p unpooled = 0.2225), although the difference was not statistically significant. Three of the six within-sex concordant chemicals in rats induced liver tumors, that is, 1-amino-2-methylanthraquinone, 2,4-diaminotoluene, and chlorendic acid. Induction of liver tumors was somewhat less common in rats than in mice (3/6 liver tumors rats vs 5/8 liver tumors mice, p pooled = 0.3200; p unpooled = 0.3195; Tables 4 and 5), although the difference did not approach statistical significance.
Comparisons of the proportions of Ames test positives and OAT test positives for different degrees of neoplastic evidence.
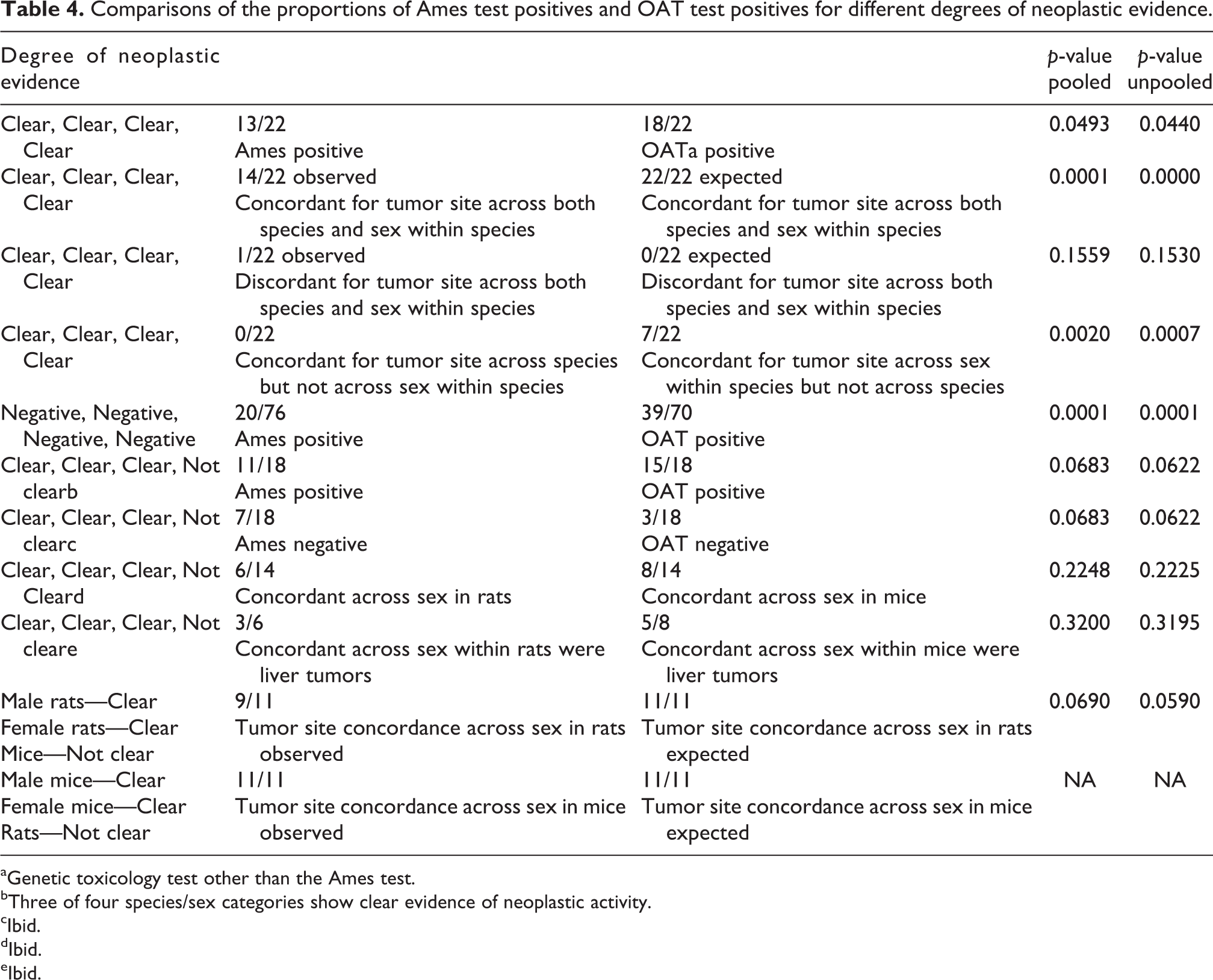
aGenetic toxicology test other than the Ames test.
bThree of four species/sex categories show clear evidence of neoplastic activity.
cIbid.
dIbid.
eIbid.
False positive and false negative rates for the Ames test and genetic toxicology assays OAT.
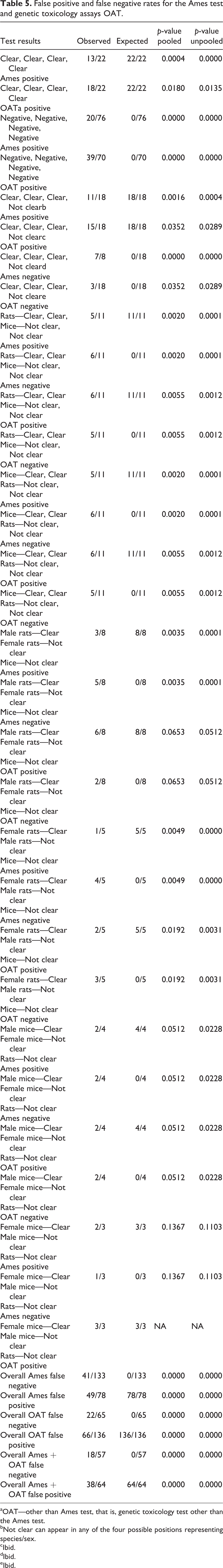
aOAT—other than Ames test, that is, genetic toxicology test other than the Ames test.
bNot clear can appear in any of the four possible positions representing species/sex.
cIbid.
dIbid.
eIbid.
Clear evidence of neoplasms in male and female rats; clear evidence of neoplasms lacking in male and female mice
Eleven of 213 chemicals tested were neoplastic in both male and female rats, but were not neoplastic in either male or female mice, including the following: 2,4-dinitrotoluene (CASRN 121-14-2), 4-amino-2-nitrophenol (CASRN 119-34-6), 3,3′-dimethoxybenzidine-4,4′-diisocyanate (CASRN 91-93-0), azobenzene (CASRN 103-33-3), N-nitrosodiphenylamine (CASRN 86-30-6), aniline hydrochloride (CASRN 142-04-1), C.I. Solvent Yellow 14 (CASRN 842-07-9), oxytetracycline hydrochloride (CASRN 2058-46-0), diphenhydramine hydrochloride (CASRN 147-24-0), rhodamine 6G (C.I. Basic Red 1; CASRN 989-38-8), and pentaerythritol tetranitrate (PETN; CASRN 78-11-5). Five of these 11 chemicals were positive in the Ames test (5/11 observed vs 11/11 expected, p pooled = 0.0020; p unpooled = 0.0001), including 2,4-dinitrotoluene (CASRN 121-14-2), 4-amino-2-nitrophenol (CASRN 119-34-6), 3,3′-dimethoxybenzidine-4,4′-diisocyanate (CASRN 91-93-0), azobenzene (CASRN 103-33-3), and N-nitrosodiphenylamine (CASRN 86-30-6). Six of these 11 chemicals were negative in the Ames test (6/11 observed vs 0/11 expected, p pooled = 0.0020; p unpooled = 0.0001). Six of these 11 chemicals were positive in a non-Ames genetic toxicology test (6/11 observed vs 11/11 expected, p pooled = 0.0055; p unpooled = 0.0012). Five of these 11 chemicals were negative in an “other than Ames” genetic toxicology test (5/11 observed vs 0/11 expected, p pooled = 0.0055; p unpooled = 0.0012). For the 11 chemicals that were neoplastic in both sexes of rats and not neoplastic in either sex of mice, 9 chemicals induced tumors at the same anatomical site for both male and female rats (9/11 tumor site concordance in rats only/no mice vs 11/11 tumor site concordance in mice only/no rats, p pooled = 0.0690; p unpooled = 0.0590), including 2,4-dinitrotoluene (CASRN 121-14-2), 4-amino-2-nitrophenol (CASRN 119-34-6), 3,3′-dimethoxybenzidine-4,4′-diisocyanate (CASRN 91-93-0), azobenzene (CASRN 103-33-3), N-nitrosodiphenylamine (CASRN 86-30-6), aniline hydrochloride (CASRN 142-04-1), C.I. Solvent Yellow 14 (CASRN 842-07-9), oxytetracycline hydrochloride (CASRN 2058-46-0), and PETN (CASRN 78-11-5; Online Appendix A).
Clear evidence of neoplasms in male and female mice; clear evidence of neoplasms lacking in male and female rats
Eleven of 213 chemicals tested were neoplastic in both male and female mice, but were not neoplastic in either male or female rats, including 5-nitro-o-toluidine (CASRN 99-55-8), 6-nitrobenzimidazole (CASRN 94-52-0), 4-chloro-o-toluidine hydrochloride (CASRN 3165-93-3), nitrofen (CASRN 1836-75-5), 5-chloro-o-toluidine (CASRN 95-79-4), di(2-ethylhexyl)adipate (DEHP; CASRN 103-23-1), 2,6-dichloro-p-phenylenediamine (CASRN 609-20-1), 2-biphenylamine hydrochloride (CASRN 2185-92-4), zearalenone (CASRN 17924-92-4), manganese (II) sulfate monohydrate (CASRN 10034-96-5), and methylphenidate hydrochloride (CASRN 298-59-9). Five of these 11 chemicals were positive in the Ames test (5/11 observed vs 11/11 expected, p pooled = 0.0020; p unpooled = 0.0001). Six of these 11 chemicals were negative in the Ames test (6/11 observed vs 0/11 expected, p pooled = 0.0020; p unpooled = 0.0001). Six of these 11 chemicals were positive in non-Ames genetic toxicology tests (6/11 observed vs 11/11 expected, p pooled = 0.0055; p unpooled = 0.0012). Five of these 11 were negative in non-Ames genetic toxicology tests (5/11 observed vs 0/11 expected, p pooled = 0.0055; p unpooled = 0.0012). For the 11 chemicals that were neoplastic in both sexes of mice and not neoplastic in either sex of rats, all chemicals induced tumors at the same anatomical site for both male and female mice (11/11 observed vs 11/11 expected, p pooled = NA; p unpooled = NA; Online Appendix A).
Clear evidence of neoplasms in male rats only
Eight of 213 chemicals tested were neoplastic in male rats only including dapsone (CASRN 80-08-0), 2-amino-5-nitrothiazole (CASRN 121-66-40, 11-aminoundecanoic acid (CASRN 2432-99-7), D & C Red No. 9 (CASRN 5160-02-1), ziram (CASRN 137-30-4), melamine (CASRN 108-78-1), monuron (CASRN 150-68-5), and HC Yellow 4 (CASRN 59820-43-8). Three of these eight chemicals were positive in the Ames test (3/8 observed vs 8/8 expected, p pooled = 0.0035; p unpooled = 0.0001) including 2-amino-5-nitrothiazole (CASRN 121-66-40), ziram (CASRN 137-30-4), and HC Yellow 4 (CASRN 59820-43-8). Five of these eight chemicals were negative in the Ames test (5/8 observed vs 0/8 expected, p pooled = 0.0035; p unpooled = 0.0001). Six of these eight chemicals tested were positive in an “other than Ames” genetic toxicology test (6/8 observed vs 8/8 expected, p pooled = 0.0653; p unpooled = 0.0512). Two of these eight chemicals tested were negative in an “other than Ames” genetic toxicology test (2/8 observed vs 0/8 expected, p pooled = 0.0653; p unpooled = 0.0512; Online Appendix A).
Clear evidence of neoplasms in female rats only
Five of 213 chemicals tested were neoplastic in female rats only including daminozide (CASRN 1596-84-5), 1,1,3-trimethyl-2-thiourea (CASRN 2489-77-2), p-quinone dioxime (CASRN 105-11-3), acetaminophen (CASRN 103-90-2), and p-nitrobenzoic acid (CASRN 62-23-7). Two of these five chemicals (p-quinone dioxime and p-nitrobenzoic acid) were positive in the Ames test (2/5 observed vs 5/5 expected p pooled = 0.0192; p unpooled = 0.0031) including p-quinone dioxime (CASRN 105-11-3) and p-nitrobenzoic acid (CASRN 62-23-7). Three of these five chemicals were negative in the Ames test (3/5 observed vs 0/5 expected, p pooled = 0.0192; p unpooled = 0.0031). Two of five chemicals were positive in an “other than Ames” genetic toxicology test (2/5 observed vs 5/5 expected, p pooled = 0.0192; p unpooled = 0.0031). Three of five chemicals were negative in an “other than Ames” genetic toxicology test (3/5 observed vs 0/5 expected, p pooled = 0.0192; p unpooled = 0.0031; Online Appendix A).
Clear evidence of neoplasms in male mice only
Four of 213 chemicals tested were neoplastic in male mice only including 3-amino-4-ethoxyacetanilide (CASRN 17026-81-2), hydrochlorothiazide (CASRN 58-93-5), fenthion (CASRN 55-38-9), and C.I. Vat Yellow 4 (CASRN 128-66-5). Two of these four chemicals, that is, 3-amino-4-ethoxyacetanilide (CASRN 17026-81-2) and hydrochlorothiazide (CASRN 58-93-5), were positive in the Ames test (2/4 observed vs 4/4 expected, p pooled = 0.0512; p unpooled = 0.0228). Two of the four chemicals were negative in the Ames test (2/4 observed vs 0/4 expected, p pooled = 0.0512; p unpooled = 0.0228). Two of the four chemicals were positive in an “other than Ames” genetic toxicology test (2/4 observed vs 4/4 expected, p pooled = 0.0512; p unpooled = 0.0228). Two of the four chemicals were negative in an “other than Ames” genetic toxicology test (2/4 observed vs 0/4 expected, p pooled = 0.0512; p unpooled = 0.0228; Online Appendix A).
Clear evidence of neoplasms in female mice only
Three of 213 chemicals tested were neoplastic in female mice only including nitrofurantoin (CASRN 67-20-9), trimethylphosphate (CASRN 512-56-1), and citral (microencapsulated; CASRN 5392-40-5). Two of these three chemicals were positive in the Ames test (2/3 observed vs 3/3 expected, p pooled = 0.1367; p unpooled = 0.1103), that is, nitrofurantoin (CASRN 67-20-9) and trimethylphosphate (CASRN 512-56-1). One of the three chemicals was negative in the Ames test (1/3 observed vs 0/3 expected, p pooled = 0.1367; p unpooled = 0.1103). Three of three chemicals were positive in an “other than Ames” genetic toxicology test (3/3 observed vs 3/3 expected, p pooled = NA; p unpooled = NA; Online Appendix A).
Clear evidence of neoplasms in male rats and male mice; clear evidence of neoplasms lacking in female rats and female mice
Only one of 213 chemicals, that is, p-nitrosodiphenylamine (CASRN 156-10-5), induced neoplasms in both male rats and mice, but not in female rats or female mice. This chemical was both positive in the Ames test and in an “other than Ames” genetic toxicology test. This chemical caused liver neoplasms in both male rats and mice (Online Appendix A).
Clear evidence of neoplasms in female rats and female mice; clear evidence of neoplasms lacking in male rats and male rats
Only one of 213 chemicals induced neoplasms in both female rats and mice, but not in male rats or male mice. This chemical, nitrofurazone (CASRN 59-87-0), was positive in both the Ames test and in an “other than Ames” genetic toxicology test. Nitrofurazone was discordant for tumor type between the female rats and female mice (Online Appendix A).
Summary of Ames salmonella mutagenicity test data
Of the 213 chemicals tested in 2-year feed studies in F334/N rats and B6C3F1 mice, 133 were negative in the Ames test. Forty-one of the 133 Ames negative chemicals (41/133) showed clear neoplastic activity in at least one species/sex category. For F344/N rats and B6C3F1 mice, the false-negative rate for the Ames test was 30.83% (41/133 observed vs 0/133 expected, p pooled = 0.0000; p unpooled = 0.0000; Online Appendix A).
Of the 213 chemicals, 78 had at least one positive Ames test result. Of those 78 Ames positive chemicals, 49 (49/78) showed clear neoplastic activity in at least one species/sex category (49/78 observed vs 78/78 expected, p pooled = 0.0000; p unpooled = 0.0000). Twenty-nine chemicals that were Ames positive showed no clear neoplastic activity in at least one species/sex category. For F344/N rats and B6C3F1 mice, the false-positive rate for the Ames test was 29/78 or 37.18% (Online Appendix A).
Summary of “other genetic toxicology test” data
Of the 213 chemicals, 136 were positive in at least one genetic toxicology test other than the Ames test. Sixty-six of 136 chemicals (48.53%) showed clear neoplastic activity in at least one species/sex category (66/136 observed vs 136/136 expected, p pooled = 0.0000; p unpooled = 0.0000). The false-positive rate was 51.47%. Of the 213 chemicals, 65 were negative in genetic toxicology tests other than Ames. Twenty-two of 65 chemicals showed clear neoplastic activity in at least one species/sex category. The false-negative rate was 33.85% (22/65 observed vs 0/65 expected, p pooled = 0.0000; p unpooled = 0.0000).
Correlation of Ames with other genetic toxicity tests
For 57 chemicals, both the Ames test and genetic toxicology assays other than Ames were negative. Of these 57 ubiquitously negative genetic toxicity test results, 18 showed clear neoplastic activity in at least one species/sex category. The false-negative rate for the Ames test combined with another genetic toxicology test was 18/57 (31.58%; 18/57 observed vs 0/57 expected, p pooled = 0.0000; p unpooled = 0.0000).
For 64 chemicals, both at least one Ames test and one other genetic toxicology assay other than Ames were positive. Of these 64 ubiquitously positive genetic toxicity test results, 38 showed clear neoplastic activity in at least one species/sex category (38/64 observed vs 64/64 expected, p pooled = 0.0000; p unpooled = 0.0000). The false-positive rate for the combined Ames test and other genetic toxicology test data was 26/64 or 40.63%.
Using the criterion that a single positive result in either the Ames or other genetic toxicology assay renders that chemical positive, the Ames test result was concordant with the “other genetic toxicology” test result for 135 chemicals (positive or negative concordance) and discordant with the “other genetic toxicology” test result for 73 chemicals.
Lack of correlation for Log P values and neoplasticity
Table 1 lists the Log P values for the chemicals that are produce tumors in male rats, female rats, male mice, and female mice. Table 2 lists the Log P values for the chemicals that do not produce tumors in male rats, female rats, male mice, or female mice. A statistical comparison between the Log P values in Tables 1 and 2 showed no difference, that is, no relationship between Log P values and neoplasticity. The mean Log P value for Table 1 was 1.92 and 1.80 for Table 2. The variance for the Log P values in Table 1 was 6.03 and 4.66 for Table 2. Considering 24 observations from Table 1, and 73 observations from Table 2, and assuming a hypothesized mean difference = 0, the following statistical values were calculated: df 35; t Stat 0.21; p(T ≤ t) one-tail 0.42; t critical one-tail 1.69; p(T ≤ t) two-tail 0.84; and t critical two-tail 2.03. In addition, a plot of the Log P values from Tables 1, 2, and 3 shows no difference in the distributions (data not shown).
Results for 31 NTP feed studies in Osborne-Mendel rats and B6C3F1 mice (34 chemicals tested)
NTP conducted 31 2-year feeding studies on 34 different chemicals using the Osborne-Mendel rat strain and B6C3F1 mice (Online Appendix B). Two of 34 chemicals induced tumors in male rats, female rats, male mice, and female mice, that is, sulfallate (CASRN 95-06-7) and technical grade chlordecone (Kepone; CASRN 104-50-0). Eleven of 34 chemicals did not induce tumors in either male and female rats, or male and female mice: mexacarbate (CASRN 315-18-4); dichlorodiphenyltrichloroethane (DDT) (CASRN 50-29-3), dioxathion (CASRN 78-34-2), dibenzo-p-dioxin (UDD; CASRN 262-12-4), pentachloronitrobenzene (PCNB; CASRN 82-68-8), methoxychlor (CASRN 72-43-5), malathion (CASRN 121-75-5), photodieldrin (CASRN 13366-73-9), lindane (CASRN 58-89-9), endrin (CASRN 72-20-8), and dichlorvos (CASRN 62-73-7). Ten of these 11 ubiquitously non-neoplastic chemicals were tested in the Ames assay. Of the 10 Ames tested chemicals, only one tested positive, representing a false-positive rate of only 10%.
Six of 34 chemicals induced neoplasms in male mice and female mice, but not in male rats and female rats: p, p′-DDE CASRN 72-55-9), chlorobenzilate (CASRN 510-15-6), toxaphene (CASRN 8001-35-2), captan (CASRN 133-06-2), heptachlor (CASRN 76-44-8), and chlordane (CASRN 57-74-9). Only one chemical, chlorothalonil (CASRN 1897-45-6), induced tumors in male rats and female rats, but not in male mice or female mice. Chlorothalonil induced kidney tumors in both male rats and female rats. Dicofol (CASRN 115-32-2) was the only chemical that induced tumors in male rats, but not in female rats, male mice, or female mice. Two chemicals induced tumors in female mice only, that is, trifluralin (CASRN 1582-09-8) and chloramben (CASRN 133-90-4). Aldrin (CASRN 309-00-2) induced tumors in male mice only.
As was observed previously in the F344/N rats and B6C3F1 mice feeding studies, concordance within male and female rats, or male and female mice, was stronger than concordance across rodent species with nine chemicals displaying discordance across species, but concordance across sex within species: p, p′-DDE, chlorobenzilate, chlorothalonil, toxaphene, tetrachlorvinphos, nitrofen, captan, heptachlor, and chlordane. In contrast, there was only one case of concordance across species but not across sex within species, that is, sulfallate induced mammary tumors in both female rats and female mice. In addition, chlordecone was the only case of concordance both across species and across sex within species. Overall, 14 of the 34 chemicals induced a neoplasm in at least a single species/sex category. Ten of these 14 neoplastic chemicals were negative in the Ames test representing an extraordinarily high false-negative rate of 71.4%.
Discussion
Since the inception of its testing program in 1977, the NTP has conducted 594 2-year studies in rats and mice via different routes of administration including inhalation, oral, dermal, and intraperitoneal. Previously, we were able to evaluate 58 of the 60 2-year inhalation studies in the NTP database and showed a high level of discordance in both overall tumor incidence and tumor site distribution between rats and mice. 2 In the current study, 213 chemicals tested by conducting 2-year feeding studies in F344/N rats and B6C3F1 mice, and 34 chemicals tested in 2-year feeding studies in Osborne-Mendel rats and B6C3F1 mice were analyzed.
Of the 213 chemicals administered in the feed, 23 showed clear evidence of neoplasia in male and female F344/N rats, and in male and female B6C3F1 mice. Twenty-one of these 23 ubiquitously neoplastic chemicals possessed structural alerts suggestive of carcinogenic potential. 9 The 21 ubiquitously neoplastic chemicals containing structural alerts were as follows: 2-methyl-1-nitroanthraquinone, 4,4′-thiodianiline, 4-chloro-o-phenylenediamine, TBP, 2,4-diaminoanisole sulfate, o-anisidine hydrochloride, 3-amino-9-ethylcarbazole hydrochloride, cupferron, p-cresidine, o-toluidine hydrochloride, Michler’s ketone, reserpine, 4,4′-oxydianiline, C.I. Basic Red 9 monohydrochloride (pararosaniline), 1-amino-2,4-dibromoanthraquinone, 1,2,3-trichloropropane, ETU, polybrominated biphenyls, 2,2-bis(bromomethyl)-1,3-propanediol (FR-1138), o-nitrotoluene, and acrylamide. The two ubiquitously neoplastic chemicals that did not possess a structural alert were NTA and nitriloacetic acid, and trisodium salt (Na3-NTA-H2O) and di(2-ethylhexyl)phthalate. The two outliers not possessing a structural alert for carcinogenicity represent the extremes of the water/lipid solubility scale. NTA has an extremely hydrophilic Log P value of −2.04, and Na3-NTA-H20 with a Log P = −1.6, and di(2-ethylhexyl)phthalate is extremely hydrophobic with a calculated Log P = 7.45. Whether these extreme Log P values adversely affect the predictability of the structural alerts is unknown. Eighteen of the 213 chemicals administered in the feed displayed clear evidence of neoplasia in three of the four categories of male rats, female rats, male mice, and female mice for F344/N rats and B6C3F1 mice. Each of these 18 chemicals either possessed a structural alert or was a phenolic tumor promoter. Therefore, these results were consistent with the structural predictions.
Seventy-seven of the 213 chemicals tested in feed were negative in male and female F344/N rats, and in male and female B6C3F1 mice. Eighteen of these 77 chemicals ubiquitously negative for neoplasia nonetheless contained a structural alert representing a false-negative rate of 23% (18/77). An additional seven of these 77 chemicals could be categorized as phenolic tumor promoters. In contrast with the high degree of association between possession of a structural alert and development of a tumor for the ubiquitously neoplastic chemicals and the chemicals neoplastic in ¾ species/sex categories, only 52/77 (67.5%) of the completely non-neoplastic chemicals did not possess structural characteristics frequently associated with neoplasia. The remaining 52 ubiquitously non-neoplastic chemicals did not contain a structural alert and were classifiable as primary and secondary amines, amides, acetates, phosphates, sulfates, sulfones, lactams, anhydrides, amino acids, gums, natural products, sugars, acids, thiocarbamates, thioureas, and azo dyes. 10
Of the 22 ubiquitously neoplastic chemicals, 14 were concordant for developing tumors at the same anatomical site both across species, and across sex within species. Of the 22 ubiquitously neoplastic chemicals, none (0/22) was concordant for developing tumors at the same anatomical site across species, but not across sex within species. Of the 22 ubiquitously neoplastic chemicals, 7 were discordant for developing tumors at the same anatomical site across species but were concordant for developing tumors at the same anatomical site across sex within species. The neoplastic response to chemicals administered in feed displays a higher degree of concordance within either male or female rats, or male and female mice, than between male rats and male mice, or female rats and female mice. This same pattern was also observed in the 2-year feeding studies employing the Osborne-Mendel rat strain and B6C3F1 mice. The tendency toward concordance in males and females within a rodent species should be considered when weighing the strength of neoplastic evidence via the feed route of exposure.
Over the years, there have been several studies that have examined or reexamined the carcinogenicity of chemicals, groups of chemicals, and exposure circumstances in animals (and in certain circumstances humans) by the International Agency for Research on Cancer (IARC) and the NTP. 11– 13 The results of this study compare favorably to past studies and support the previous results that there is a low level of concordance in chemicals tested causing cancer in each of the four sex/species groups used for testing (female rats, male rats, female mice, and male mice; 14% for all chemicals tested by NTP vs. 11% (23/213) in the NTP feed studies).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.