Abstract
The degree of correlation between tumors predicted by OncoLogic™ (Oncologic) and the actual formation of tumors as observed in the National Toxicology Program (NTP) 2-year rodent studies is lower for “justification reports” that incorporate historical data than for “data reports” that do not. The correlation between the ordinal ranking of the observed carcinogenicity of parent NTP chemicals and the predicted “level of carcinogenicity concern” from the justification reports obtained from Oncologic is poor (r = 0.56). Similarly, the correlation between the ordinal ranking of the carcinogenicity of metabolites from parent NTP chemicals and the predicted “level of carcinogenicity concern” from the justification report obtained from Oncologic is also poor (r = 0.43). In contrast, the correlation between the ordinal ranking of the observed carcinogenicity of parent NTP chemicals and the predicted level of carcinogenicity concern from the data reports obtained from Oncologic is comparatively better (r = 0.75). The correlation between the ordinal ranking of the carcinogenicity of metabolites from parent NTP chemicals and the predicted “level of carcinogenicity concern” from the data reports generated from Oncologic is also comparatively good (r = 0.68). The level of correlation between the ordinal tumorigenicity ranks of parent chemicals and between the ordinal tumorigenicity ranks of chemicals reported to induce liver tumors in the National Center for Toxicological Research liver cancer database was also investigated. There was a higher degree of correlation seen for Oncologic “data reports” as compared with Oncologic “justification reports.” Incorporation of additional information via “justification reports” weakens the predictive power of Oncologic.
Introduction
The National Toxicology Program (NTP) is a branch of the US Department of Health and Human Services. A major current emphasis of NTP is “The Toxicology in the 21st Century: The Role of the National Toxicology Program.”
1
NTP describes this program as follows: The Role of the National Toxicology Program is to support the evolution of toxicology from a predominantly observational science at the level of disease-specific models to a predominantly predictive science focused upon a broad inclusion of target-specific, mechanism-based, biological observations.
In a series of four earlier publications, we first analyzed the results from 60 2-year inhalation studies conducted by NTP and showed a high level of discordance in tumor formation between rats and mice. 2 Next, we analyzed the results from 212 2-year feed studies conducted in F334/N rats and B6C3F1 mice, and 31 2-year feed studies conducted in Osborne–Mendel rats and B6C3F1 mice. The feed studies showed a higher degree of concordance within either male or female rats or male and female mice, than between male rats and male mice, or female rats and female mice. 3 In the third study, 4 we analyzed the results from the following: 108 chemicals tested in 106 2-year studies conducted by exposing F334/N rats and B6C3F1 mice via gavage, 18 2-year gavage studies conducted in Osborne–Mendel rats and B6C3F1 mice on 19 different chemicals, 23 chemicals tested in 21 2-year studies conducted by exposing F334/N rats and B6C3F1 mice via drinking water, 18 chemicals tested in 18 2-year studies conducted by exposing F334/N rats and B6C3F1 mice via dermal application, and 11 chemicals tested in 11 2-year studies conducted by exposing F334/N rats and B6C3F1 mice via intraperitoneal injection. The neoplasticity of each chemical was analyzed for tumor incidence by species–sex category, tumor site concordance across species, and tumor site concordance across sex within species. When available, the Ames Salmonella mutagenicity assay results, and any results from a test for genotoxicity other than the Ames test, were correlated with the neoplasticity results. These three studies completed the summary and analysis of the various routes of exposure. 2 –4
The fourth study, and this the fifth study, considered the mechanistic aspects of the various factors potentially influencing tumor induction. In the fourth study, 5 each of the 478 chemicals was categorized from 1 to 48 by the level of “Clear” neoplastic evidence in male and female rats, and in male and female mice, and given an ordinal rank from 1 to 135 following additional considerations regarding tumor site concordance and tumor multiplicity. The resultant tumorigenicity category score and ordinal rank score were examined for associations with results in the Ames Salmonella mutagenicity assay, the presence or absence of structural alerts of carcinogenicity, and three Hansch QSAR parameters, that is, calculated base 10 logarithm of the octanol–water partition coefficient (ClogP), calculated molar refractivity, and McGowan molecular volume. As noted above, the NTP database consisted of a total of 478 organic chemicals, organic chemical mixtures, metals, inorganic chemicals, extracts, and natural products for which there were tumor and genotoxicity data available. Of these 478 chemicals, only 463 chemicals were used in this fifth report of the publication series, as 15 of the chemicals either could not be rated for carcinogenicity concern of the chemical by OncoLogic™ (Oncologic) or had no Chemical Abstracts Service Registry Number (CASRN) (e.g. a commercial product).
In this fifth study, the United States Environmental Protection Agency’s (USEPA) predictive program Oncologic 6 was used to compare the predicted carcinogenic potential (level of concern) for the same chemicals tested in the NTP studies with the actual experimental results. The qualitative Oncologic evaluations described by “degree of concern” were converted to “ordinal rankings of the Oncologic degree of concern” for each chemical in the following descending order: High or 1 > High-Moderate or 2 > Moderate or 3 > Low-Moderate or 4 > Marginal or 5 > Low or 6 > Inactive or 7. The relationship between the ordinal ranks predicted by Oncologic and the experimental ordinal ranks was examined via statistical analysis. In this fifth study, in addition to overall tumors, liver tumor data and median toxic dose (TD50) data on chemicals tested by NTP were obtained from the National Center for Toxicological Research liver cancer database (NCTRlcdb 7 ; https://www.fda.gov/ScienceResearch/BioinformaticsTools/ucm236173.htm). Of the 478 chemicals for which NTP prepared final reports, liver cancer and TD50 data on 128 chemicals were found in the NCTRlcdb. The NCTRlcdb data specifically indicate which rodent species and sex were induced with liver cancer. With this information, an ordinal scale was constructed to rank the concern associated with liver cancer as follows: rank 1 = male rats (MR), female rats (FR), male mice (MM), and female mice (FM) were all induced with liver cancer > rank 2 = any combination of three of the four MR, FR, MM, and FM was induced with liver cancer > rank 3 = MR and FM, or FR and MM, were induced with liver cancer > rank 4 = MR and FR, or MM and FM, were induced with liver cancer > rank 5 = MR or FR or MM or FM was induced with liver cancer > rank 6 = no species–sex of rodent was induced with liver cancer. The ordinal rankings of chemicals were used for the statistical analysis of liver cancer data. 5
Methods
Determination of ordinal rank number from 1 to 135
NTP classifies the level of evidence for neoplasia as Clear (Positive), Some, Equivocal, and Negative. Analysis of the entire NTP database demonstrated that only neoplastic evidence that rose to the level of “Clear” was sufficiently robust to facilitate meaningful statistical analysis. 2 –4 Each of the 478 chemicals for which final technical reports were available reported results for male rats, female rats, male mice, and female mice. In several cases, one of the four studies on a particular sex/species category was deemed as “Inadequate” due to technical problems with that arm of the study, while the other three arms reported valid results. This situation was amenable to statistical analysis with “Inadequate” ranked just higher than “Negative” due to the possibility that if that arm had been completed without technical difficulty, it might have shown a level of neoplasticity higher than “Negative.” The descending order of the categorical rank was as follows: Clear Evidence > Some Evidence > Equivocal Evidence > Inadequate Evidence > Negative Evidence. This ranking scheme resulted in the highest category of Clear (male rats), Clear (female rats), Clear (male mice), and Clear (female mice); and the lowest category of Negative (male rats), Negative (female rats), Negative (male mice), and Negative (female mice). Due to a sporadic presentation of species/sex categories ranked as “Inadequate,” the final number of categories is not set at 48 as the size of the NTP database grows, but rather that is the number of categories that result given the outcomes from the 478 current chemicals for which there are final technical reports.
Analysis of the entire NTP database across all routes of administration consistently showed that the highest hurdle of neoplastic evidence was tumor site concordance across species. 2 –4 This result created a boundary condition under which the ordinal rank could be further split within the neoplasticity category (from 1 to 48), but a chemical in a lower category could not be assigned a higher ordinal rank than that of any chemical in a higher category. The second highest hurdle of neoplastic evidence was tumor site concordance across sex within species. The final criterion influencing the ordinal rank was multiplicity of tumors that were not concordant with the organ site. These non-concordant tumors are referred to in the ranking scheme as “single tumors.” Online Supplementary Appendix shows the category ranking (1–48) and the ordinal ranking (1–135) for each of the 478 chemicals resulting from simultaneous consideration of the number of different tumors concordant with the tumor site across species, the number of different tumors concordant across sex within species, and the number of discordant tumors.
Evaluation of carcinogenic potential by Oncologic
Oncologic is an expert system that predicts the potential carcinogenicity of chemicals by applying the rules of structure activity relationship (SAR) analysis and expert judgment that incorporates what is known about metabolism, mechanisms of action, and human epidemiological studies. There are two types of reports produced. One report is called a justification report. This report contains a summary of the predicted concern level for the compound and the line of reasoning used by the expert system to arrive at the concern level. The other report is a data report. The data report restates the information the user enters into Oncologic and uses for its concern level evaluation. Both reports use the rules of SAR analysis and expert judgment to predict the concern level.
The justification report consists of two parts: a summary of the evaluation and a line of reasoning (justification) of how the final conclusions were derived. Within the summary section, a final level of concern is stated along with any other messages that merit special attention. The concern levels used by the Oncologic system are semantic terms used by the EPA for ranking the hazard levels of chemicals. The specific terms in order from the highest concern to the lowest concern are High (highly likely to be a potent carcinogen), High-Moderate (highly likely to be a moderately active carcinogen), Moderate (likely to be a moderately active carcinogen), Low-Moderate (likely to be weakly carcinogenic), Marginal (likely to have equivocal carcinogenic activity), Low (unlikely to be carcinogenic), and Inactive (not carcinogenic).
The data report contains the information that was entered about the compound. Essentially, it becomes a reference report of the data entered upon which the Oncologic system based its evaluation. It should be noted that not all the data that are available from the sources above were incorporated into Oncologic. For example, only about 45% or less of the studies performed by NTP are in the database. Oncologic is free from the USEPA at http://www.epa.gov/oppt/sf/pubs/oncologic.htm. 6
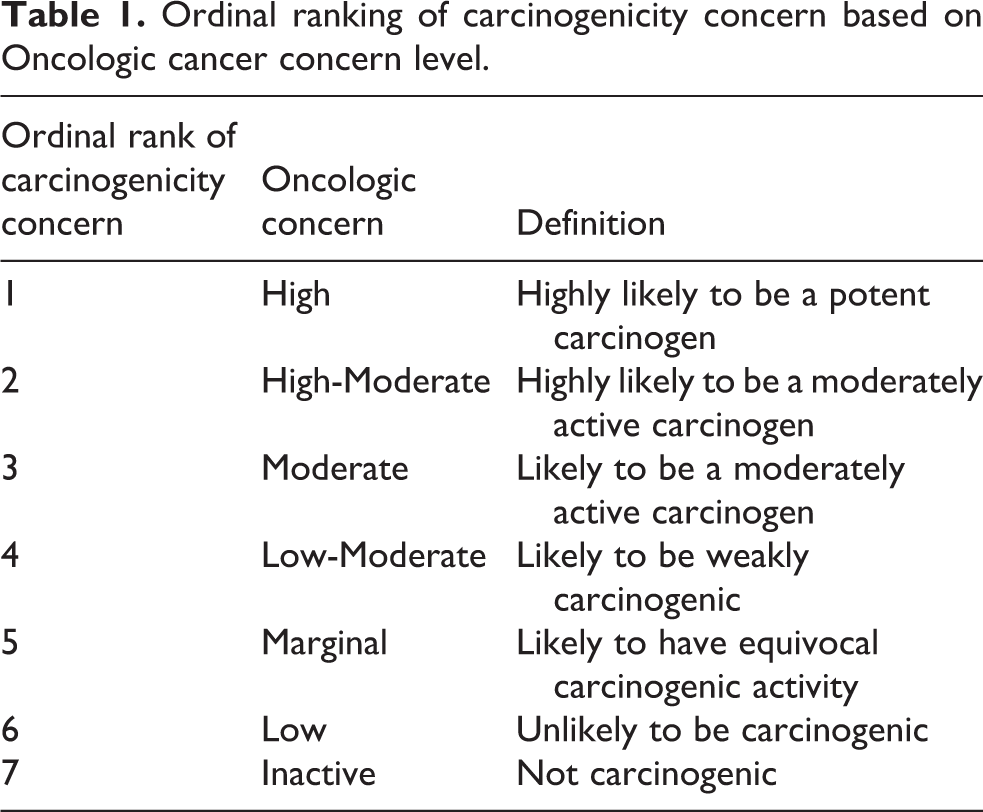
Oncologic rates the level of carcinogenicity concern expressed as a verbal qualitative description (High to Inactive). With this information, an ordinal scale was constructed to rank the concern associated with the induction of cancer. High = Ordinal Rank 1, High-moderate = Ordinal Rank 2, Moderate = Ordinal Rank 3, Low-moderate = Ordinal Rank 4, Marginal = Ordinal Rank 5, Low = Ordinal Rank 6, and Inactive = Ordinal Rank 7. The ordinal rankings of chemicals were used for the statistical analysis of Oncologic cancer concern data (Table 1).
Ordinal ranking of carcinogenicity concern based on Oncologic cancer concern level.
Prediction of carcinogenicity of metabolites
An estimate for the carcinogenicity of metabolites was assigned as follows. For direct-acting chemicals (electrophiles), the carcinogenicity concern level of the parent chemical was equal to the metabolite. Therefore, the two ratings are the same. For chemicals like cupferron where a metabolite is formed and acts as an electrophile, the predicted carcinogenicity of the metabolite is one step higher (e.g. High-Moderate goes to High). If the prediction from Oncologic is already High, then the ranking of the metabolite remains High. The reasoning is as follows. The parent compounds are often less carcinogenic than their metabolite, except in direct-acting chemicals where they are identical. 8,9 The parents are very often procarcinogens. 8 For example, benzo[a]pyrene (B[a]P) is only mildly carcinogenic by itself. However, its metabolite benzo[a]pyrene-7,8-oxide is highly carcinogenic. 8,9
The NCTRlcdb
The NCTRlcdb contains 999 chemicals and was developed based on liver-specific carcinogenicity. The assumption underlying the construction of the database was that organ-specific carcinogenicity is a more definitive end point for building a biologically predictive model. In the NCTRlcdb, each chemical is assigned one of the following toxicity classifications based on the studies of male and female rats and mice: liver carcinogen (273), other carcinogens (293), noncarcinogen (304), and others (129). Additionally, the NCTRlcdb provides information on CASRNs, chemical formula, ClogP, TD50, source of information (literature or National Cancer Institute [NCI]), and carcinogenicity index. 7
For this part of the analysis, only the chemicals studied in the 2-year NTP studies and that also induced liver cancer were considered. Of the 478 chemicals for which NTP prepared reports, liver cancer data on 128 chemicals were found in the NCTRlcdb. The NCTRlcdb also contained TD50 information on 128 chemicals. 7 The NCTRlcdb provides data on liver cancer and specifically indicates which species and sex were induced with liver cancer. Using this information, an ordinal scale was constructed to rank order the degree of concern associated with liver cancer (Table 2). Ordinal Rank 1 represented the induction of liver cancer in all four species/sex categories, that is, male rat (MR), female rat (FR), male mouse (MM), and female mouse (FM); Ordinal Rank 2 is any combination of three of the four categories above; Ordinal Rank 3 is either MR and FM or FR and MM; Ordinal Rank 4 is any combination of MR and FR or FM and MM; Ordinal Rank 5 consists of the individual categories of MR, FR, MM, or FM; and Ordinal Rank 6 consists of “None” (Table 2). The ordinal rankings of chemicals were used for the statistical analysis of liver cancer data for each chemical. Of the 128 chemicals that were evaluated, only 36 were part of the Oncologic database where justification reports were available. The remaining 92 NTP-tested chemicals had data reports generated.
Ordinal ranking of carcinogenicity concern based on the NCTRlcdb data on species–gender induced with liver cancer.
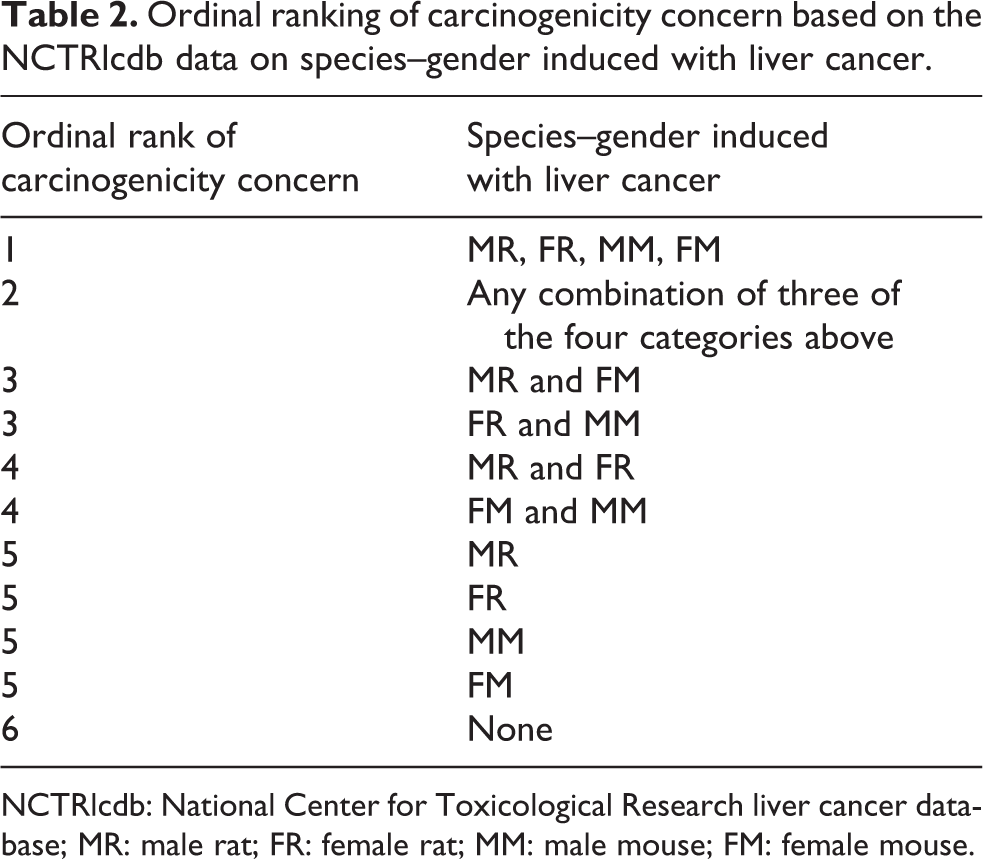
NCTRlcdb: National Center for Toxicological Research liver cancer database; MR: male rat; FR: female rat; MM: male mouse; FM: female mouse.
Statistical methods
Two statistical methods were employed: correlation matrices and analysis of variance (ANOVA). A correlation matrix is a table showing correlation coefficients between sets of variables. Each random variable (Xi ) in the table is correlated with each of the other values in the table (Xj ). This illustrates which pairs have the highest correlation. The diagonal of the symmetric matrix table is always a set of ones, because the correlation between a variable and itself is always 1. The correlation coefficient indicates how strongly two variables are related to each other. The statistic used to produce the correlation coefficient is Pearson’s r, the Pearson correlation coefficient. Pearson’s r is a measure of the linear correlation between two variables X and Y. It has a value between +1 and −1, where 1 is total positive linear correlation, 0 is no linear correlation, and −1 is total negative linear correlation. 10
ANOVA is a particular form of statistical hypothesis testing used to analyze experimental data. A test result (calculated from the null hypothesis and the sample) is called statistically significant if it is deemed unlikely to have occurred by chance, assuming the truth of the null hypothesis. A statistically significant result, when a probability (p value) is less than a threshold (significance level), justifies the rejection of the null hypothesis, but only if the a priori probability of the null hypothesis is not high. 11
Results
Results from carcinogenicity predictions by Oncologic compared with actual tumors from NTP final reports
NTP tested 478 chemicals and 463 possessed sufficient data to facilitate an ordinal ranking of carcinogenicity from 1 to 135. 4,5 The 15 remaining chemicals could not be rated for several reasons. Of these 463 NTP-tested chemicals, 204 were included in the Oncologic database. Of the 463 chemicals tested by NTP for which there were sufficient data to assign an ordinal ranking (1–135) of carcinogenicity, 4,5 there were 259 NTP-tested chemicals that were not part of the formalized Oncologic database but for which an Oncologic “level of carcinogenic concern” could be determined. By manually entering the toxicological data requested by the Oncologic program, a “level of carcinogenic concern” was developed by Oncologic and a “data report” was generated. The toxicological data entered into the Oncologic data report for 259 NTP-tested chemicals were obtained from the NTP databases of final reports. No other sources of data were used.
A table of data (Online Supplementary Appendix 1) was produced that contained the name and CASRN of the NTP-tested chemical; the ordinal ranking of the observed carcinogenicity of parent NTP chemicals; the predicted carcinogenicity of the parent compound via Oncologic; a discussion on the predicted metabolites/mode of carcinogenic action/results from NTP testing; the predicted carcinogenicity of metabolites, liver tumors produced by NTP-tested chemicals (sex–species data from the NCTRlcdb); other tumors produced by NTP-tested chemicals (sex–species data from the NCTRlcdb); and TD50 information from the NCTRlcdb.
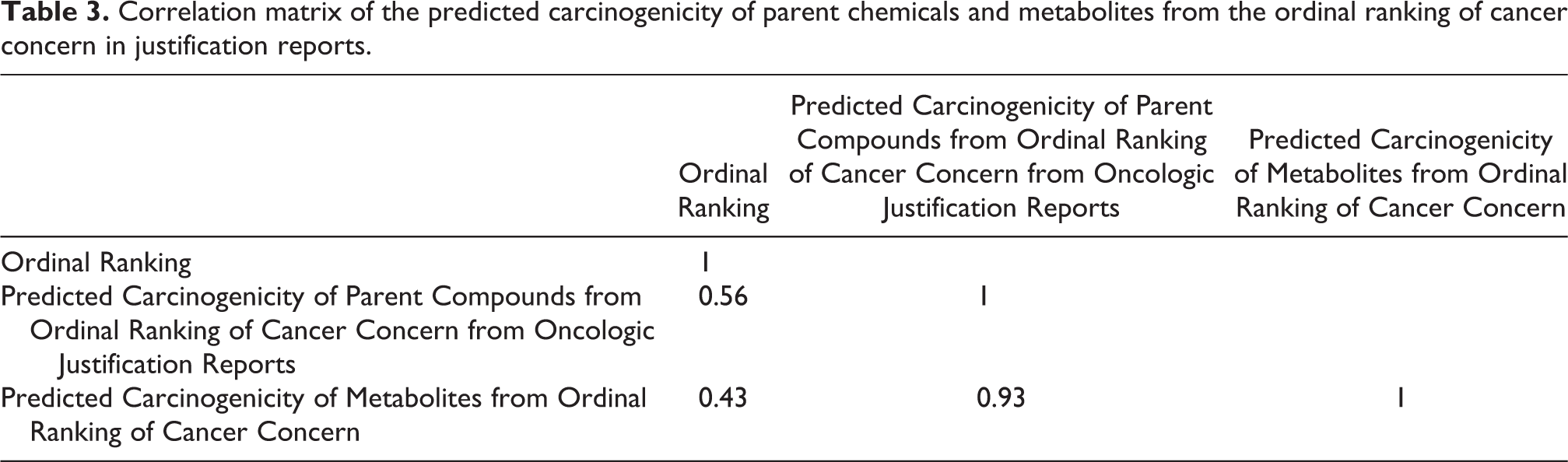
A correlation matrix on the raw data from 204 NTP-tested chemicals for which there were justification reports was created (Table 3). The correlation between the ordinal ranking (1–135) of the observed carcinogenicity of parent NTP chemicals 5 and the predicted “level of carcinogenicity concern” from the justification reports obtained from Oncologic is poor (r = 0.56). Similarly, the correlation between the ordinal ranking of the carcinogenicity of metabolites from parent NTP chemicals and the predicted “level of carcinogenicity concern” from the justification report obtained from Oncologic is also poor (r = 0.43). As expected, there was a high correlation between the predicted carcinogenicity of parent chemicals from Oncologic justification reports and the predicted carcinogenicity of metabolites (r = 0.93).
Correlation matrix of the predicted carcinogenicity of parent chemicals and metabolites from the ordinal ranking of cancer concern in justification reports.
A correlation matrix on the raw data from 259 NTP-tested chemicals for which final reports were generated is shown in Table 4. The correlation between the ordinal ranking (1–135) of the observed carcinogenicity of parent NTP chemicals 5 and the predicted level of carcinogenicity concern from the data reports obtained from Oncologic is comparatively good (r = 0.75). The correlation between the ordinal ranking of the carcinogenicity of metabolites from parent NTP chemicals and the predicted “level of carcinogenicity concern” from the data reports generated from Oncologic is also comparatively good (r = 0.68). As expected, there was a high correlation between the predicted carcinogenicity of parent chemicals from Oncologic data reports and the predicted carcinogenicity of metabolites (r = 0.96).
Correlation matrix of the predicted carcinogenicity of parent chemicals and metabolites from the ordinal ranking of cancer concern in data reports.

Table 5 shows the predicted Oncologic cancer concern ordinal ranks (1–7) for the 204 NTP-tested parent chemicals contained in the Oncologic database (justification reports) regressed against the observed average ordinal ranking (1–135) of 204 NTP-tested chemicals. The data sets are linearly related and highly correlated (Figure 1). The equation that predicts the ordinal ranking of carcinogenicity for NTP-tested chemicals is as follows
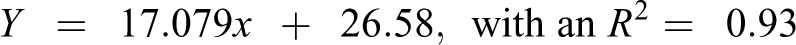
Regressions of Oncologic cancer concern ratings (in justification reports) versus average ordinal ranking from NTP chemicals and metabolites.
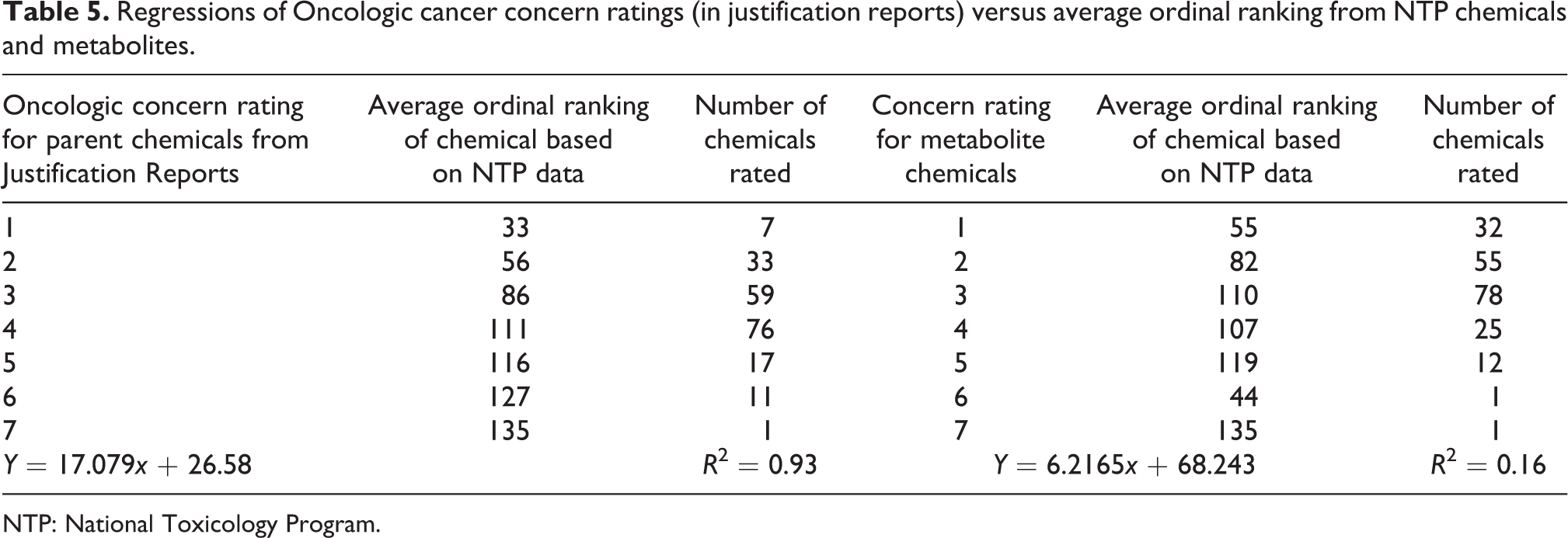
NTP: National Toxicology Program.
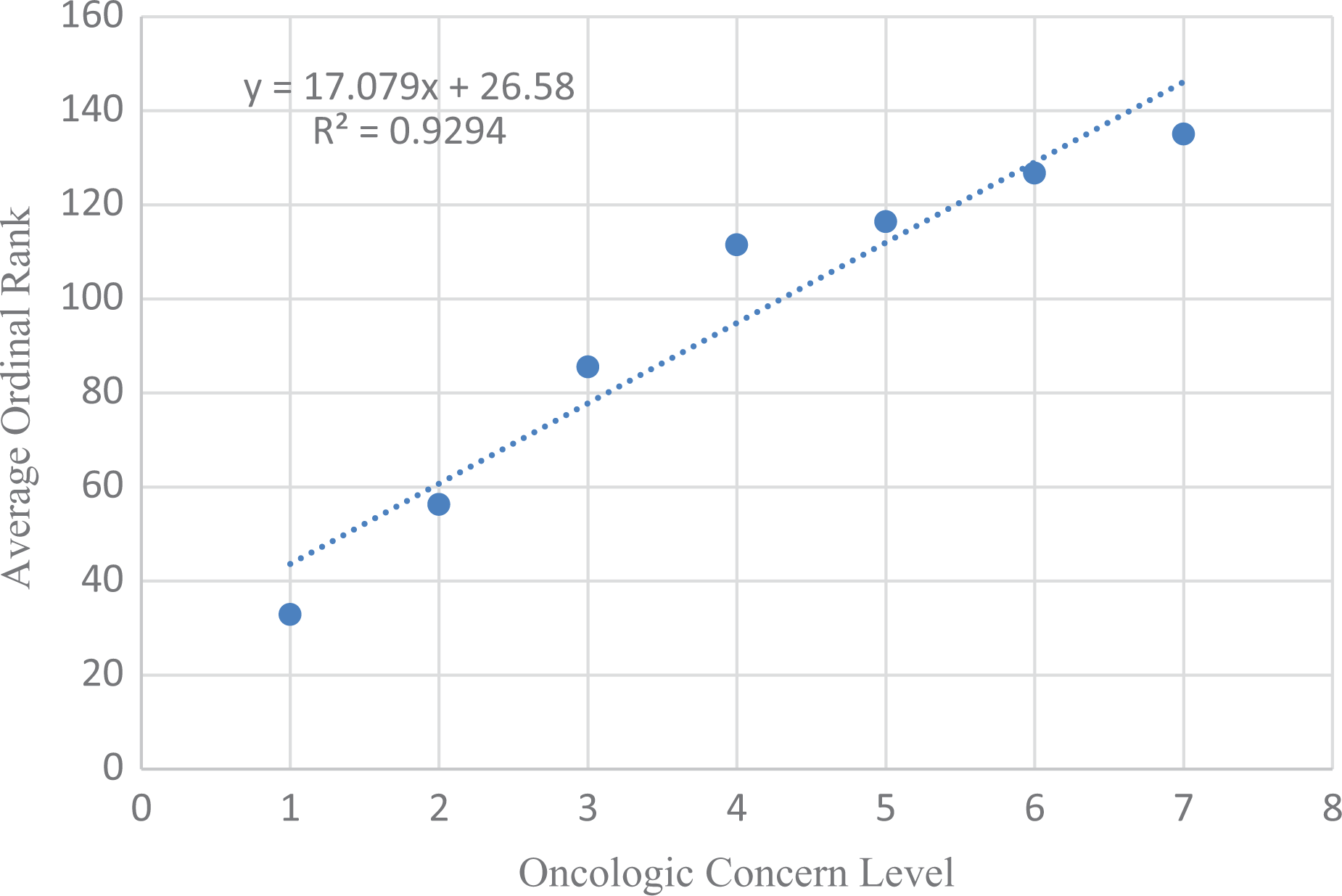
Oncologic concern level for parent chemicals from justification reports versus NTP average ordinal ranking. NTP: National Toxicology Program.
Table 5 also shows the predicted Oncologic cancer concern ordinal ranks (1–7) for the metabolites of 204 NTP-tested chemicals regressed against the observed average ordinal ranking (1–135) of 204 NTP-tested chemicals. The data sets were poorly correlated. The equation that predicts the ordinal ranking of carcinogenicity for the metabolites of 204 NTP-tested chemicals is as follows
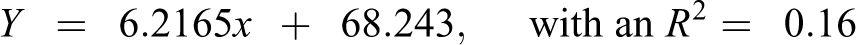
Table 6 shows the predicted Oncologic cancer concern ordinal ranks (1–7) for the 259 NTP-tested parent chemicals that are not part of the Oncologic database but where data reports were generated regressed against the observed average ordinal ranking (1–135) of 259 NTP-tested chemicals. The data sets were linearly related and the correlation was high (Figure 2). The equation that predicts the ordinal ranking of carcinogenicity for NTP-tested chemicals is as follows
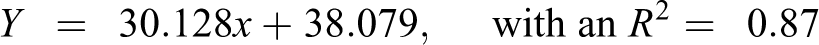
Regressions of Oncologic concern ratings (in data reports) versus average ordinal ranking from NTP chemicals and metabolites.
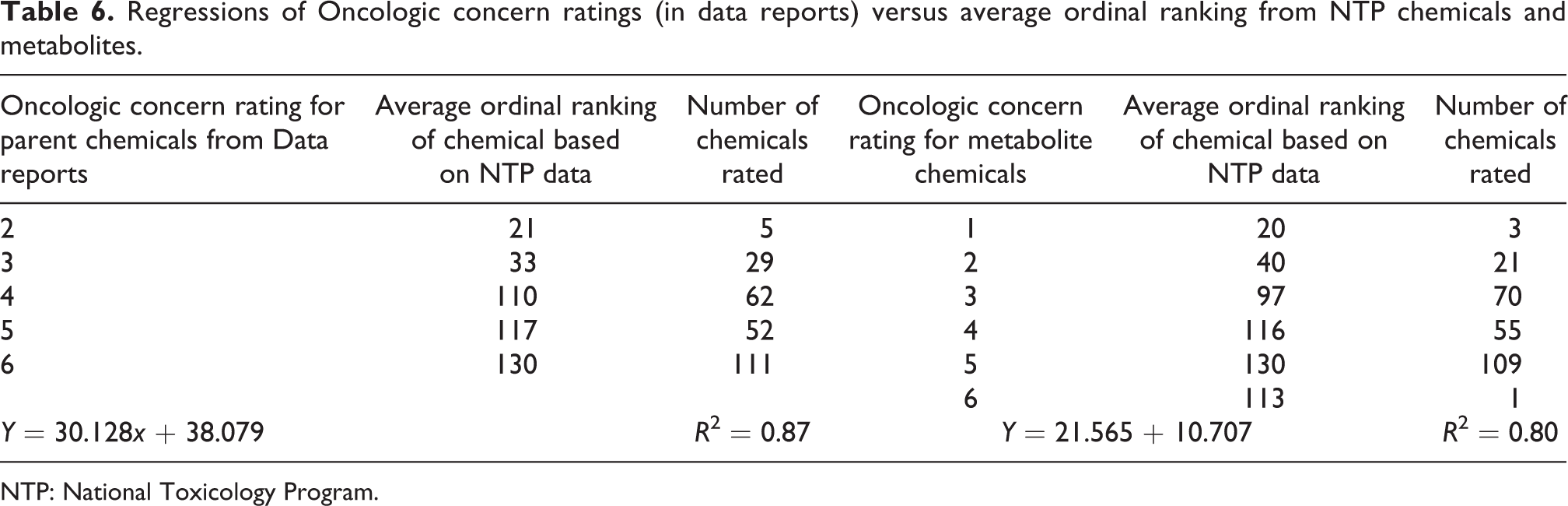
NTP: National Toxicology Program.
Oncologic concern level for parent chemicals from data reports versus NTP average ordinal ranking. NTP: National Toxicology Program.
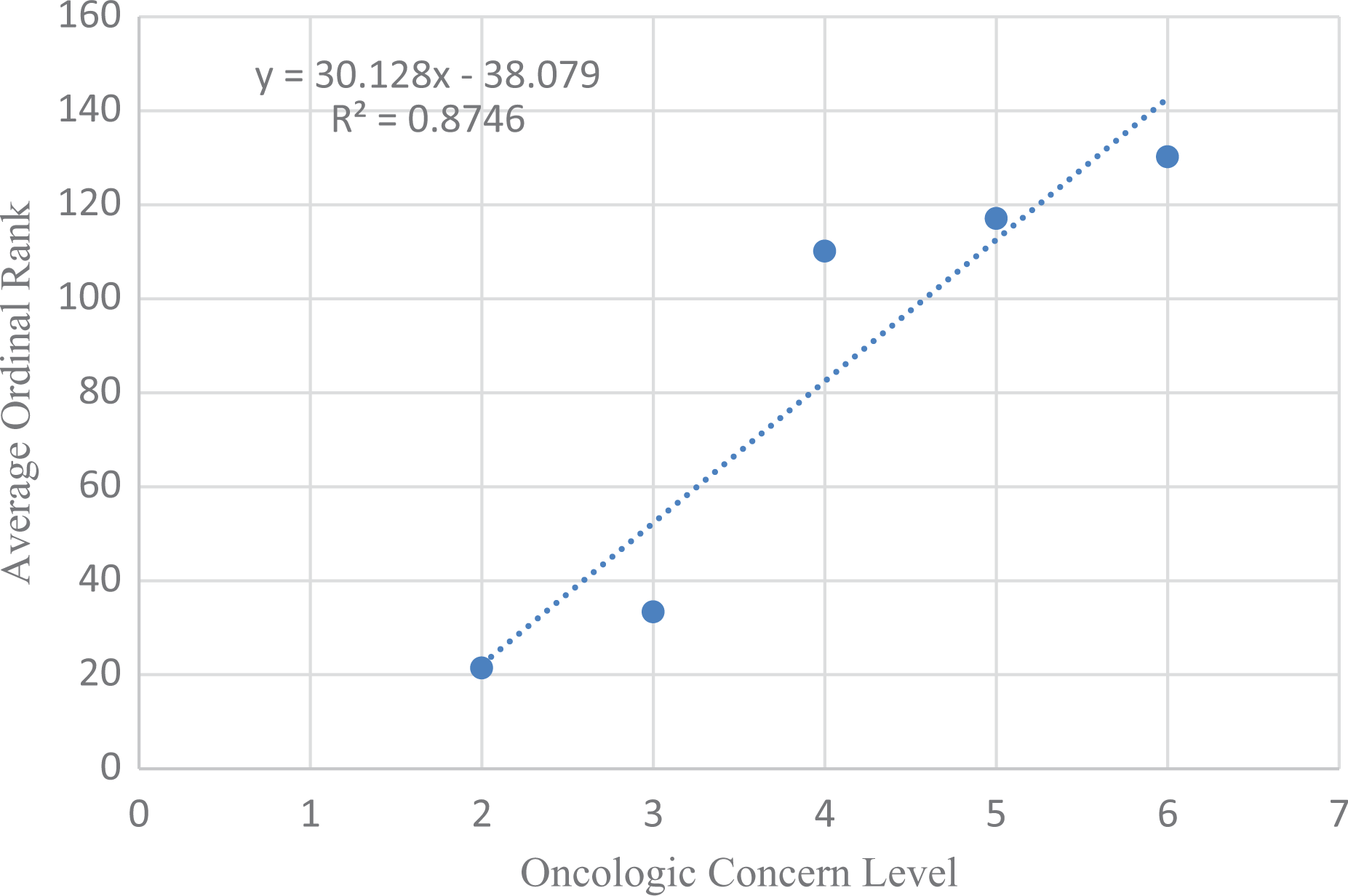
Table 6 also shows the predicted Oncologic cancer concern ratings for the metabolites of 259 NTP-tested chemicals regressed against the observed average ordinal ranking (1–135) of 259 NTP-tested chemicals. The data sets were linearly related and the correlation was very good (Figure 3). The equation that predicts the ordinal ranking of carcinogenicity for NTP-tested chemicals is as follows
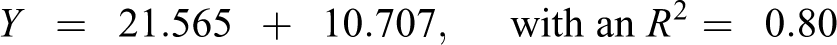
Oncologic cancer concern ratings for the metabolites from data reports versus NTP average ordinal ranking. NTP: National Toxicology Program.
Correlations between NTP database tumors and NCTRlcdb tumors
Of the 478 chemicals for which NTP generated final reports, liver cancer data on 128 chemicals were found in the NCTRlcdb. The NCTRlcdb also contained TD50 information on these 128 chemicals. Of the 128 chemicals that were NTP-tested chemicals and were also found in the NCTRlcdb database, 92 were chemicals that had Oncologic justification reports. The remaining 36 NTP-tested chemicals had cancer concerns generated via Oncologic data reports.
A correlation matrix (Table 7) was produced to determine significant correlations among the Ordinal Ranking on NTP-tested chemicals (1–135), Ordinal Ranking of Predicted Carcinogenicity of Parent Compound via Oncologic (Justification Reports), Ordinal Ranking of Predicted Carcinogenicity of Metabolites, Ordinal Ranking of Liver Tumors by Species, and Ordinal Ranking of Tumors Other than Liver by Species from the 92 chemicals that had Oncologic justification reports. There is only one significant correlation, that is, as expected there was a high correlation between the ordinal ranking of the predicted carcinogenicity of parent chemicals from Oncologic justification reports and the ordinal ranking of the predicted carcinogenicity of metabolites (r = 0.90).
Correlation matrix of the predicted carcinogenicity of 92 parent chemicals, metabolites, liver tumors, and other tumors in the NCTRlcdb database based on the ordinal ranking of cancer concern in justification reports.
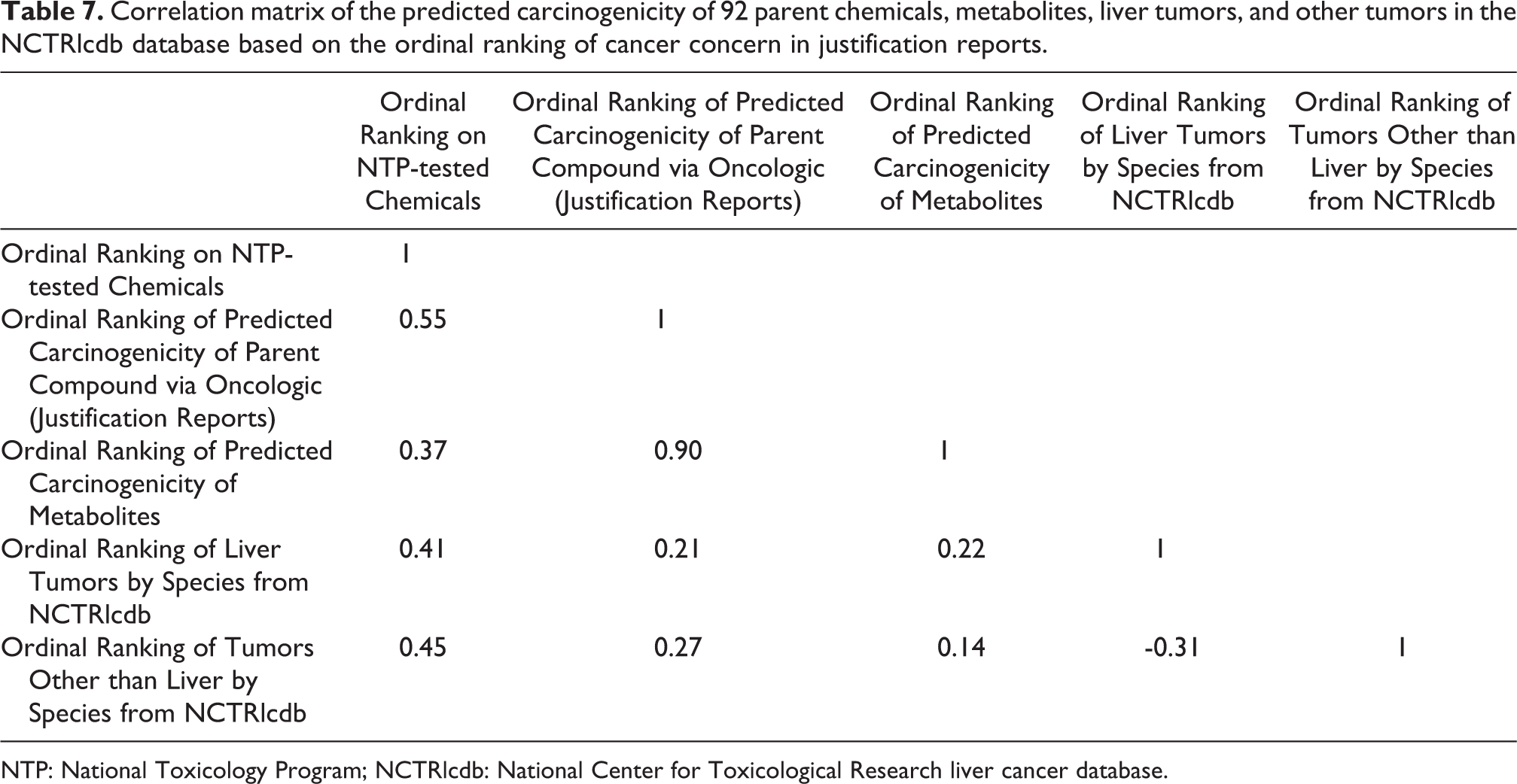
NTP: National Toxicology Program; NCTRlcdb: National Center for Toxicological Research liver cancer database.
Table 8 shows the p value results of ANOVA for the relationships of interest (Ordinal Ranking of Carcinogenicity, Ordinal Ranking of Predicted Carcinogenicity of Parent Compound via Oncologic (Justification Reports), Ordinal Ranking of Predicted Carcinogenicity of Metabolites, Ordinal Ranking of Liver Tumors by Sex–Species, Ordinal Ranking of Tumors Other than Liver by Sex–Species, and TD50) for the 92 NTP-tested chemicals that had cancer concerns generated via Oncologic justification reports. None of the relationships are significant. The ANOVA results between the median toxic doses at which toxicity occurs in 50% of the rodents (TD50) are relatively insignificant for Ordinal Ranking of Predicted Carcinogenicity of Parent Compound via Oncologic (Justification Reports; p = 0.34), Ordinal Ranking of Predicted Carcinogenicity of Metabolites (p = 0.16), Ordinal Ranking of Liver Tumors by Sex–Species (p = 0.19), and Ordinal Ranking of Tumors Other than Liver by Sex–Species (p = 0.57).
Table of the p value results of ANOVA for relationships of interest for the 92 NTP-tested chemicals that had cancer concerns generated from justification reports based on ordinal rankings.
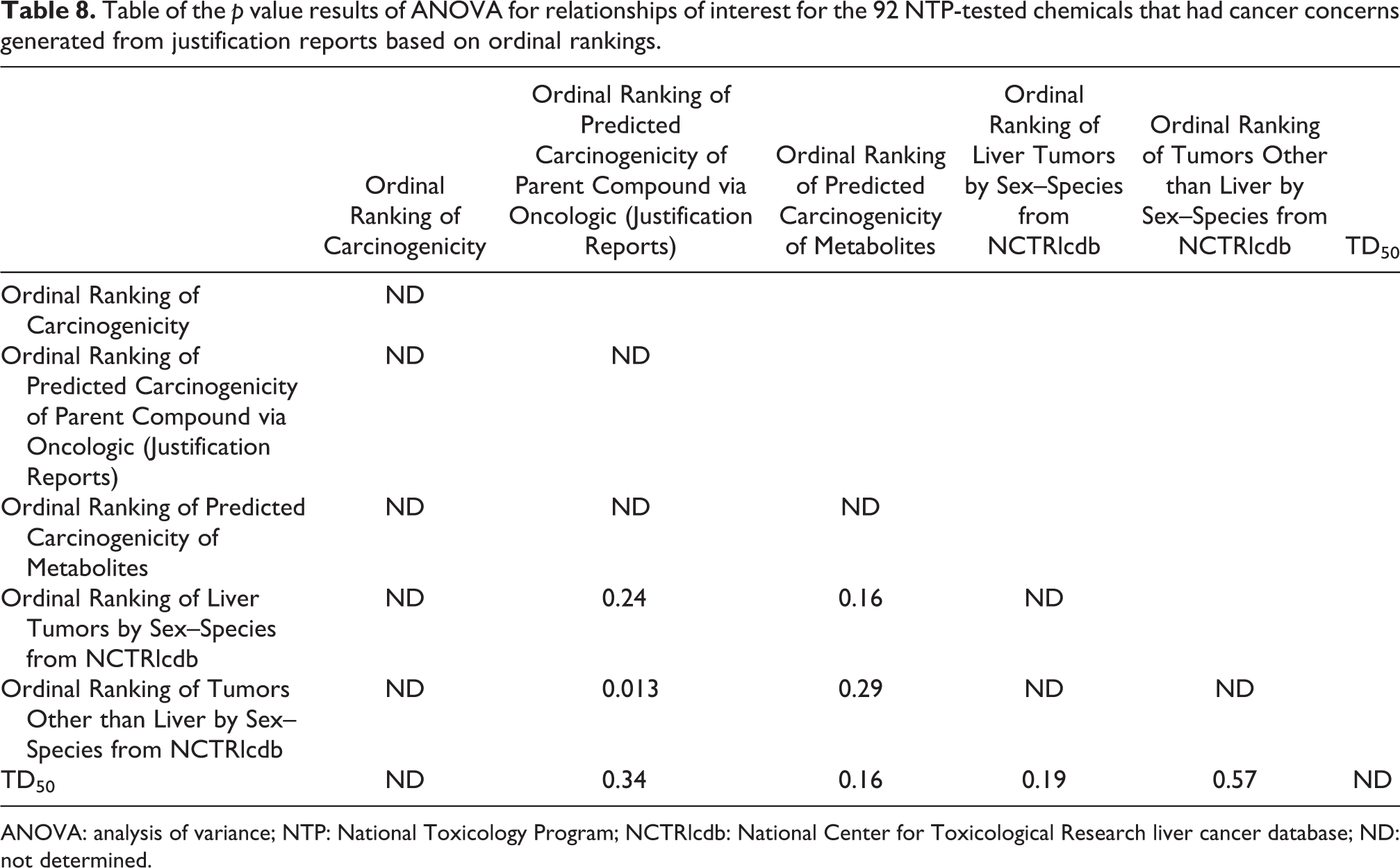
ANOVA: analysis of variance; NTP: National Toxicology Program; NCTRlcdb: National Center for Toxicological Research liver cancer database; ND: not determined.
A correlation matrix was produced to determine if there were any significant correlations among the data sources from the 36 chemicals for which we generated Oncologic data reports (Table 9). Several significant correlations exist. As expected, there was a high correlation between the ordinal ranking of the predicted carcinogenicity of parent chemicals from Oncologic data reports and the ordinal ranking of the predicted carcinogenicity of metabolites (r = 0.92). There were also several high correlations between Ordinal Ranking of Carcinogenicity (1–135) and Ordinal Ranking of Predicted Carcinogenicity of Parent Compound via Oncologic Data Report (1–7; r = 0.77), Ordinal Ranking of Carcinogenicity (1–135) and Ordinal Ranking of Predicted Carcinogenicity of Metabolites (r = 0.76), and Ordinal Ranking of Carcinogenicity and Ordinal Ranking of Liver Tumors by Sex–Species (r = 0.79).
Correlation matrix of the predicted carcinogenicity of 36 parent chemicals, metabolites, liver tumors, and other tumors in the NCTRlcdb database based on the ordinal ranking of cancer concern in data reports.
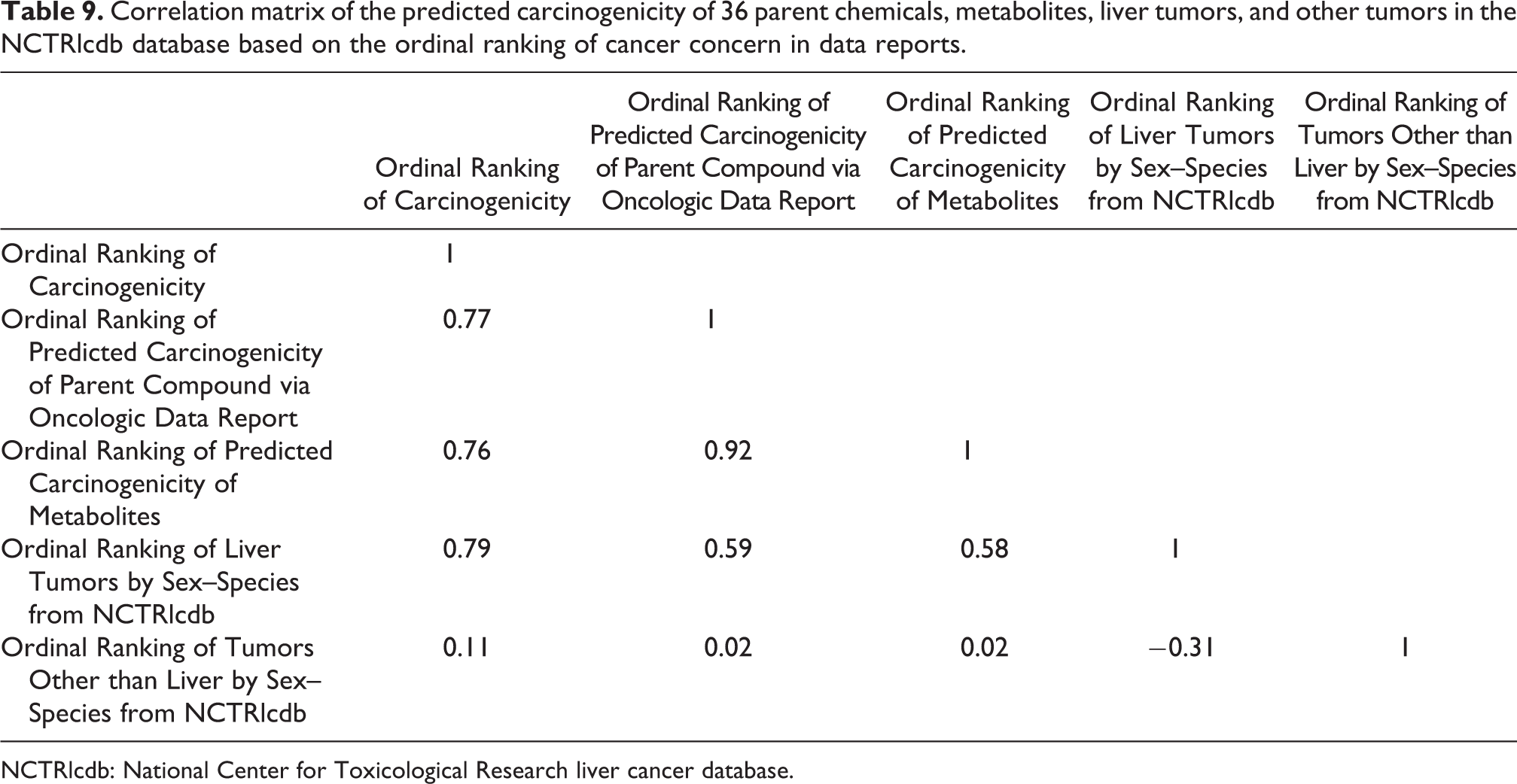
NCTRlcdb: National Center for Toxicological Research liver cancer database.
Table 10 shows the p value results of ANOVA for the relationships of interest for the 36 NTP-tested chemicals that had cancer concerns generated via data reports. Table 10 shows the p values for the various relationships. Several relationships are statistically significant. The ordinal ranking of the predicted carcinogenicity of parent compound via Oncologic and the ordinal ranking of metabolites versus ordinal ranking of liver tumors by sex–species are both significant (with p values of 3.12E−05 and 0.000525, respectively). The ordinal ranking of the predicted carcinogenicity of metabolites versus TD50 was significant (with p values of 0.005062).
Table of the p value results of ANOVA for relationships of interest for the 36 NTP-tested chemicals that had cancer concerns generated from data reports based on ordinal rankings.
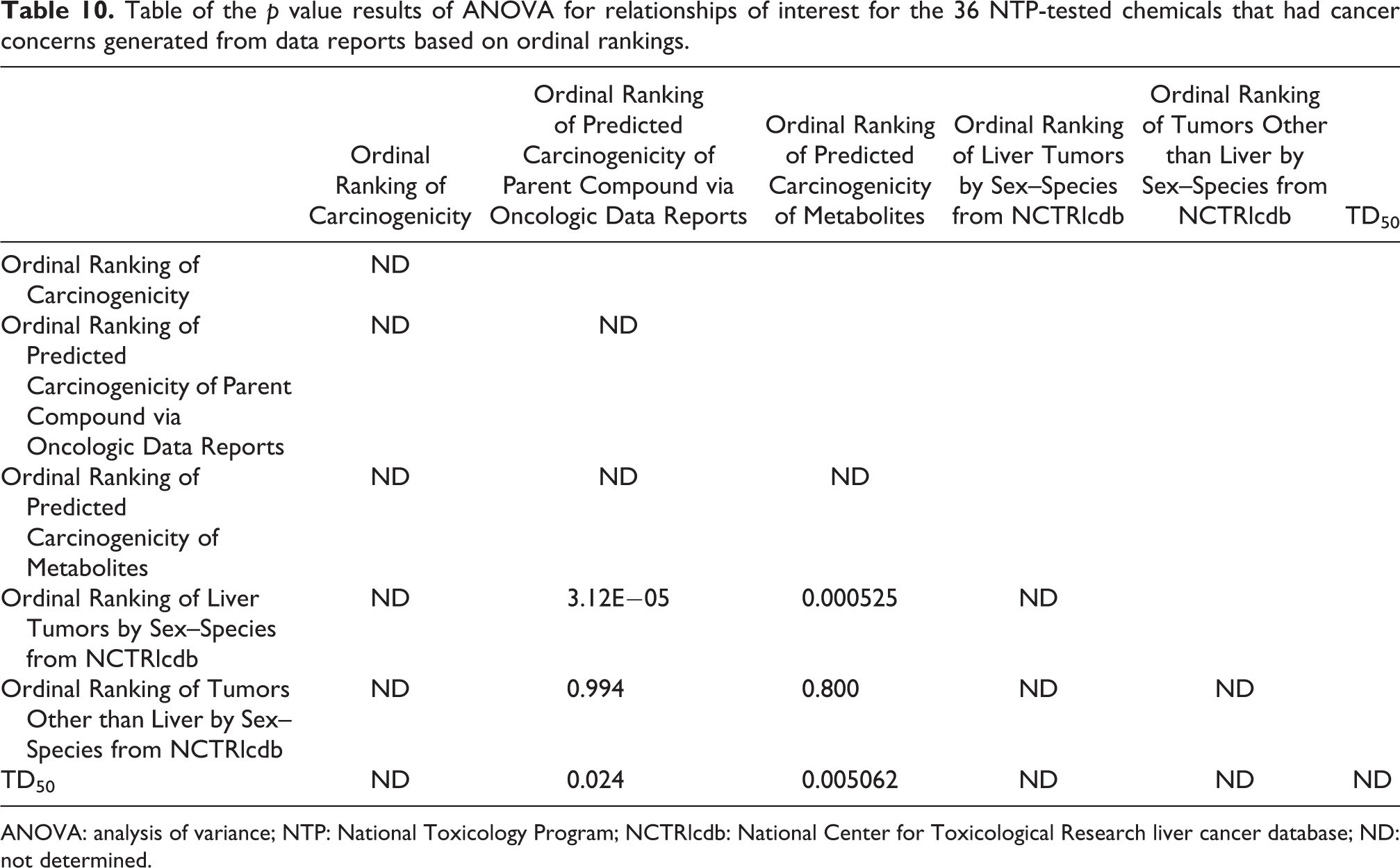
ANOVA: analysis of variance; NTP: National Toxicology Program; NCTRlcdb: National Center for Toxicological Research liver cancer database; ND: not determined.
Chemicals predicted by Oncologic as carcinogenic or potentially carcinogenic that did not induce tumors
Of the 478 chemicals tested by way of inhalation, feed, gavage, drinking water, dermal administration, or intraperitoneal injection, 134 were ubiquitously negative in male and female rats, and in male and female mice. Fifty-eight of these 134 chemicals ubiquitously negative for neoplasia nonetheless had a concern rating from Oncologic of Moderate (five chemicals), Low-Moderate (31 chemicals), and Marginal (22 chemicals) (indicating at least an equivocal carcinogenic activity) representing a false positive rate of 43% (58/134).
There are 330 chemicals that induce at least one tumor in either rats or mice. The concern ratings from Oncologic are as follows: High (7), Moderate-High (38), Moderate (83), Low-Moderate (107), Marginal (45) (indicating at least an equivocal carcinogenic activity), Low (3) (indicating unlikely to be carcinogenic), and Inactive (47). Therefore, at least 50 (Low + Inactive) of 330 chemicals represent a false negative rate of 50/330 (15.2%). If marginal ratings are included, the false negative rate is 95/330 (28.8%).
Discussion
In the first four papers of this series, 2 –5 statistical analysis of the entire 594 NTP 2-year rodent cancer study database was conducted. This analysis demonstrated that reliance on historical Ames data, that is, examining every Ames test ever conducted on the chemical of interest and classifying a single positive Ames test result as indicative of genotoxicity, leads to spurious results. The biological implausibility of this methodology is demonstrated by poor correlations between positive Ames test results and the induction of rodent tumors, and concomitantly, poor correlations between negative Ames test and the lack of induction of rodent tumors.
Similarly, in this fifth study of the series, the degree of correlation between tumors predicted by the Oncologic program and the actual formation of tumors as observed in the NTP 2-year rodent studies is lower for “justification reports” that incorporate historical data than for “data reports” that do not. The correlation between the ordinal ranking (1–135) of the observed carcinogenicity of parent NTP chemicals 5 and the predicted “level of carcinogenicity concern” from the justification reports obtained from Oncologic is poor (r = 0.56). Similarly, the correlation between the ordinal ranking of the carcinogenicity of metabolites from parent NTP chemicals and the predicted “level of carcinogenicity concern” from the justification report obtained from Oncologic is also poor (r = 0.43). In contrast, the correlation between the ordinal ranking (1–135) of the observed carcinogenicity of parent NTP chemicals 4,5 and the predicted level of carcinogenicity concern from the data reports obtained from Oncologic is comparatively good (r = 0.75). The correlation between the ordinal ranking of the carcinogenicity of metabolites from parent NTP chemicals and the predicted “level of carcinogenicity concern” from the data reports generated from Oncologic is also comparatively good (r = 0.68).
Tables 7 and 9 show the level of correlation between the ordinal tumorigenicity ranks of parent chemicals and metabolites in the NTP database and between the ordinal tumorigenicity ranks of chemicals reported to induce liver tumors in the NCTRlcdb. The higher degree of correlation seen for Oncologic “data reports” as compared with Oncologic “justification reports” is seen by comparing the comparable correlation coefficients in Tables 7 and 9 for the relationship between Ordinal Ranking of Liver Tumors by Sex–Species and Ordinal Ranking of Predicted Carcinogenicity of Parent Compound via Oncologic (Justification Reports) (r = 0.21) and between Ordinal Ranking of Liver Tumors by Sex–Species and Ordinal Ranking of Predicted Carcinogenicity of Parent Compound via Oncologic (Data Reports) (r = 0.59). In Table 8, the ANOVA results between the median toxic doses at which toxicity occurs in 50% of the rodents (TD50) are relatively low for Ordinal Ranking of Predicted Carcinogenicity of Parent Compound via Oncologic (Justification Reports) (r = 0.34), Ordinal Ranking of Predicted Carcinogenicity of Metabolites (r = 0.16), Ordinal Ranking of Liver Tumors by Sex–Species (r = 0.19), and somewhat higher for Ordinal Ranking of Tumors Other than Liver by Sex–Species (r = 0.57).
In contrast with the relatively weak relationships between the predictions made in Oncologic justification reports and TD50, Table 10 shows the p value results of ANOVA for the relationships of interest for the 36 NTP-tested chemicals that had cancer concerns generated via data reports. TD50 and Ordinal Ranking of Predicted Carcinogenicity of Parent Compound via Oncologic Data Reports were related to one another at a p value = 0.024. Similarly, TD50 and Ordinal Ranking of Predicted Carcinogenicity of Metabolites were related to one another at a p value = 0.005062.
Another interesting result is illustrated in Tables 5 and 6 and Figures 1 and 2. In the fourth paper of this series, we published an ordinal ranking scheme described herein at the beginning of the “Methods” section under “Determination of ordinal rank number from 1 to 135.” The extremely high degree of linear correlation between ordinal ranks assigned using this scheme and the levels of cancer concern as predicted by Oncologic provides external validation for the ordinal ranking methodology.
The results in this fifth study support the contention of Ames and Gold 12 that the NTP 2-year rodent studies overreport the carcinogenic potential that would be expected at lower, noncytotoxic doses not leading to reparative cell proliferation with the attendant amplification of background mutation rates. First, when compared against the actual experimental results in the NTP database, Oncologic predicts that tumors should not form 15.2% of the time when they in fact do, and if marginal ratings are included, 28.8%. This result is consistent with the thesis of Ames and Gold 12 that tumors can form from the amplification of the background mutation rate due to the cytotoxicity-induced acceleration of cell proliferation rates in the absence of exposure to genotoxins. It should also be noted that the high false positive rate (43%) associated with Oncologic crowds out the false negative rates in a reciprocal fashion.
Second, the Ames and Gold 12 hypothesis predicts a continuum of tumorigenic effects based on the dose, that is, genotoxic chemicals causing tumors at comparatively low doses and cytotoxic chemicals inducing tumors at comparatively higher doses. In addition, Kiraly et al. 13 have shown that increased cell proliferation rates interact synergistically with mutation, thereby implying that in many cases rodent tumors form with nonlinear contributions to carcinogenesis from both mechanisms. In Tables 8 and 10, the poor correlation between TD50 values and Oncologic predictions of the level of concern, or of liver tumor formation, is consistent with tumor formation occurring via a mixture of mechanistic effects.
Conclusions
Predictions made by Oncologic “data reports” for the level of carcinogenic concern for both the parent compounds and the predicted metabolites correlate well with the actual tumor formation seen in NTP 2-year rodent bioassays. Incorporation of additional information via “justification reports” weakens, rather than strengthens, the predictive power of Oncologic.
Supplemental material
NTPPaper5FinalSupplementalMaterial - Comparison of carcinogenicity predictions by the Oncologic expert system with NTP 2-year rodent study tumorigenicity results
NTPPaper5FinalSupplementalMaterial for Comparison of carcinogenicity predictions by the Oncologic expert system with NTP 2-year rodent study tumorigenicity results by Carr J Smith, and Thomas A Perfetti in Toxicology Research and Application
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
