Abstract
Although sodium iodate (NaIO3)-induced retinal injury model has been widely used in rodents, its application in large animal species has encountered variation in retinal toxicity. NaIO3 induced retinal degeneration and functional changes in sheep, but not in swine. In monkeys, administration of NaIO3 via a carotid artery affected only the cell function of ipsilateral retinal pigment epithelium. The aim of the present study was to identify the dosage and route of NaIO3 administration resulting in morphologic and functional retinal changes in cynomolgus monkeys. Separate groups of animals received NaIO3 intravenously in three different dosing paradigms. Vehicle control animals received phosphate-buffered saline. At selected time points following dosing, flash electroretinograms (ERGs) were recorded followed by necropsy. The eyes were examined microscopically post-necropsy and the levels of circulating microRNA-183 cluster were evaluated in the blood samples collected on days 1, 4, and 5 postdose. A statistically significant reduction in both scotopic a-wave and scotopic and photopic b-wave signals (p < 0.05) were observed between the ERG signals acquired from NaIO3-treated and vehicle control animals, coupled with time-dependent elevations in plasma miR-183 cluster. Mild to moderate retinal degeneration was observed in the outer layer of the retina, which correlated well with the functional and clinical observations. There were no statistically significant differences in scotopic oscillatory potentials. These findings suggest that intravenous injection of sublethal NaIO3 markedly damaged the cone and rod photoreceptors both functionally and morphologically, and plasma miR-183 reflected the retinal toxicity in those animals with moderate retinal damage.
Keywords
Introduction
Adverse drug-induced ophthalmic effects are not uncommon in clinic, 1,2 particularly in patients with psychotropic agents, 3 and have been reported frequently as adverse events in humans. 4 In drug development, adverse ophthalmic events also have significant impact on advancement of new pharmaceutical candidates, with ∼6.8% of drugs removed from clinical trials because of visual toxicity concerns. 5,6 Animal models of retinal injury are an essential part of drug development, as they serve as a positive control for proof of principle and testing of efficacy and safety for drug candidates. Retinal degeneration models induced by pharmaceuticals, such as sodium iodate (NaIO3), N-methyl-N-nitrosourea, 7 and adenosine triphosphate, 8 are widely used in drug discovery. Among them, the murine NaIO3 model is the most widely used because of reproducible patchy retinal degeneration 9 –12 and is also used in retinal mechanistic studies in a wide variety of wild-type 13 and genetic knockout mouse strains. More recently, this model has gained a resurgence of interest because of its ability to utilize high-resolution in vivo imaging to study structural changes in the retina. New techniques such as optical coherence tomography and confocal scanning laser ophthalmoscopy allow longitudinal evaluation of retinal structure, 14,15 thus making this model amendable to precisely evaluate efficacy and toxicity of the novel therapeutics, including stem cell-derived cells. 16 Potential limitations with regard to the use of murine models in testing the efficacy and safety of new drug candidates for use in humans include the rodent eye anatomy and physiology. The nonhuman primate eye most closely resembles the human eye with regard to the presence of a macula. 17 In addition, dose volumes intended for use in humans cannot be tested in the rodent eye.
Application of NaIO3 in large laboratory animal species to induce retinal degeneration has, however, encountered species-dependent variation in toxicity. For example, single intravenous dosages of NaIO3 at 15–30 mg/kg resulted in moderate to severe retinal pigment epithelium cell (RPE) damage with subsequent photoreceptor loss in rodents, 18 while dosages of up to 90 mg/kg did not affect visual function in swine. 19 In monkeys, administration of 1 mg/kg NaIO3 via a carotid artery affected RPE function but did not affect the contralateral eye. 20 Thus, administration of NaIO3 at higher doses (mg/kg) may more reproducibly induce retinal injury in nonhuman primates.
In the present study, we evaluated three different dosing paradigms of NaIO3: (1) a single 30 mg/kg dose on day 1, (2) two 30 mg/kg doses administered on days 1 and 2, and (3) a single 25 mg/kg dose on day 1 and 15 mg/kg dose on day 2 in conjunction with current electrophysiologic and plasma biomarker assay techniques to develop a consistent model of acute retinal toxicity capable of elucidating both morphologic and functional changes in cynomolgus monkeys.
Full-field electroretinogram (ERG) has been used to assess retinal function in human and nonhuman primates. 21 The standard flash ERG trace consists of several distinct light-driven components, with the a-wave, b-wave, and oscillatory potentials (OPs) constituting the major ones. By testing at different background light conditions and dark or light adaptation, ERG reveals the activities of rod- and cone photoreceptor-driven responses as well as the responses originated from the inner retinal structure. It is not only a gold standard end point for retinal function but also a translatable biomarker from preclinical to clinical phase in drug development, 22 since the origins and similarities between laboratory animals and humans of the various waveforms of ERG have been well-defined without doubt in retinas of both animals and humans over the past 40 years.
Visual disturbance was reported as a frequent adverse event in patients administered the oral small-molecule tyrosine kinase inhibitor, crizotinib. 4,23 The use of the ERG in rodent models detected visual disturbances that were not detectable in standard nonclinical toxicological evaluation. 24 The ERG was validated as an efficient electrophysiological functional assay with many known retinal toxicants/drugs. 6 In drug discovery industry, ERG technology has been widely used as an efficient tool to predict retinal toxicity to reduce attrition at later stages. Although ERG is commonly recognized as translatable to clinic, for statistical analysis, to obtain the exact number of the translation rate would need more preclinical and clinical data.
In the past few years, microRNA (miRNA, miR) has been reported to be involved in the development and functions of the retina and the eye. 25 miR-182, miR-183, and miR-96 are members of a conserved sensory organ–enriched cluster and miR-183/96/182 or the miR-183 cluster are reported to express in rods, cones, and bipolar cells in mice 26 and human. 25 Changes in miRNA levels in the vitreous humor have been reported to be correlated with various ocular diseases. 27 Our previous study demonstrated that circulating miR-183 cluster are emerging biomarkers for retinal damage in rat. 28 To date, there have not been any reports of miRNA profiling in nonhuman primate models. Such blood-borne miRNAs may be excellent and less invasive biomarkers for monitoring retinal health during drug discovery and development.
Materials and methods
Animals
A total of 13 adult cynomolgus monkeys (Macaca fascicularis, weighing 5.5–8.5 kg, 4–5 years old; Charles River, MA, USA) were used in this study. Animals were housed in a temperature- and humidity-controlled environment with a 12-/12-h light/dark cycle. Animals had free access to food and water. All in vivo procedures were approved by the institutional animal care and use committees at Pfizer (Groton, CT, USA and Pearl River, NY, USA) and conformed to the statement of the Association for Research in Vision and Ophthalmology for the Use of Animals in Ophthalmic and Vision Research. All efforts were made to minimize the animal’s pain and distress and to minimize the number of animals used. Five monkeys (animals 0-1, 0-2, 0-3, 0-4, and 50-126) received no drug or vehicle injection. Two monkeys (110-148 and AT195A) received phosphate-buffered saline (PBS) vehicle injection and the other six received NaIO3 injections (Table 1).
NaIO3 dosage regimen and clinical course.

NaIO3: sodium iodate;–: no findings; HPD: hours postdose.
Severity grades: 1 = minimal, 2 = mild, 3 = moderate, 4 = marked, and 5 = severe.
aAnimals 50-126 did not receive vehicle injection.
bHours post the first dose.
cAnimals 2-2 was euthanized at 48 HPD.
Chemicals and administration
NaIO3 (Sigma-Aldrich, St. Louis, MO, USA) was dissolved in PBS (10–20 mg/ml), filtered through a 0.22 µm filter and dosed as either a one-time 30 mg/kg (n = 2) or two doses on consecutive 2 days (n = 2). In the other two animals, NaIO3 was injected at 25 mg/kg for first dose and at 15 mg/kg for the second dose on the following day (Table 1). Doses were administered at the volume of 1.5 ml/kg via the saphenous vein as a slow bolus infusion over a period of 30–60 s. PBS was administered to two animals in the same manner.
Blood sample collection, RNA extraction, and miRNA measurement
Blood samples were obtained at predose, 48 and 72 h time points following the first dose of NaIO3 (day 1). A terminal blood sample was also collected at euthanasia. A gross necropsy and tissue collection were performed on day 5. Total RNAs from 100 µl of plasma were purified by Qiagen’s miRNAeasy kit (Valencia, CA, USA) according to the manufacturer’s protocol and a total 30 µl of the purified RNAs was obtained. Ten microliter of the fixing amount of the purified miRNA was subjected to real-time polymerase chain reaction (RT-qPCR). Quantification of circulating plasma miRNAs in blood was performed in duplicate in final PCRs. All reactions were run on a ViiA™ 7 Real Time PCR System (ThermoFisher Scientific, Waltham, MA, USA) using the following conditions: 95°C for 10 min, followed by 40 cycles at 95°C for 15 s and 60°C for 50 s. Data are presented as fold change (or fold increase) relative to predose or vehicle control ± Standard error of the mean (SEM), in which relatively stable miRNAs in plasma such as miR-181, miR-92, and miR-192 were selected as reference miRNAs for normalization purpose and in addition, exogenous spike in control Cel miR-39 was measured to improve the quantification accuracy in RT-qPCR. 28
Clinical chemistry and biomarkers
Terminal blood samples were collected from the animals in cohort 3 for measuring the following chemistry analytes: alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, gamma-glutamyl transferase, glutamate dehydrogenase, creatine kinase, total bilirubin, cholesterol, triglyceride, glucose, total protein, albumin, globulin, albumin/globulin ratio, blood urea nitrogen, creatinine, phosphorus, calcium (Ca), sodium (Na), potassium (K), and chloride (Cl), hemolysis index (HEM IND), icterus index (ICT IND), and lipemia index (LIP IND). All measures were conducted according to Siemen Advia 1200 protocols, including calibrations of all the analytes along with pre- and postrun controls (Siemens Advia 1200; all reagents and controls purchased from Siemens Healthcare Diagnostics, Malvern, PA, USA).
Clinical observations
Cage-side clinical observation was conducted twice daily after NaIO3 dosing until the animals were euthanized.
Flash ERG recording
Standard full-field ERGs were recorded simultaneously from both eyes at 96 h post-NaIO3 or vehicle injection. The monkeys were anesthetized by intramuscular injection of 10 mg/kg ketamine hydrochloride (Pfizer, Lake Forest, IL, USA) and 0.01 mg/kg glycopyrrolate. Induction and maintenance (via endotracheal intubation) of anesthesia were achieved with ∼2% isoflurane in oxygen (0.80–1.0 L/min). A Bair Hugger warm air blanket (Arizant, Eden Prairie, MN) provided thermal support during anesthesia. Heart rate, respiratory rate, blood oxygen saturation, and anesthetic depth were monitored at 10-min intervals throughout the procedure.
ERG procedures used in this study were similar to our previously reported method. 24 In brief, the animals were dark adapted for 40 min prior to scotopic response testing. 29 Mydriacyl (1% tropicamide; Bausch & Lomb Inc., Tampa, FL, USA) was applied to the eyes to induce pupil dilation. The cornea was anesthetized with topical proparacaine hydrochloride eye drops (Bausch & Lomb Inc.). ERG lens electrodes (Fabrinal SA, Switzerland) were placed onto the surface of the eyes with 0.3% hypromellose (Hub Pharmaceutical, LLC, Rancho Cucamonga, CA, USA) and saline as coupling agents bilaterally. A platinum needle reference electrode (Natus Neurology, Middleton, WI, USA) was inserted subcutaneously in the forehead and a ground electrode clip was placed on an ear.
A UTAS Visual Electrodiagnostics System with a BigShot Ganzfeld (LKC Technologies, Inc., Gaithersburg, MD, USA) stimulator was used to evoke and acquire the ERG signals (high-pass filtered at 0.3 Hz and low-pass filtered at 500 Hz). After 40 min of dark adaptation, ERGs were recorded to test rod responses (0.007 cd s/m2), maximum rod and cone responses (2.5 cd s/m2) and scotopic OPs (6.43 cd s/m2). Then following 10 min of light adaptation, photopic flicker responses (2.5 cd s/m2, 30 Hz) and cone responses (2.5 cd s/m2) were acquired. 29 All photopic responses were obtained with the background Ganzfeld illumination of 30 cd s/m2.
ERG waveforms were analyzed with LKC Technologies software using the International Society for Clinical Electrophysiology of Vision guidelines. The a-wave amplitude was measured from baseline to trough, and implicit time was measured between stimulus and a-wave trough. The b-wave amplitude was measured from a-wave trough to b-wave peak, and b-wave implicit time was measured from stimulus to b-wave peak. The OPs were isolated off-line from the underlying b-wave using the automatic electronic filtering provided in the LKC software. The OP amplitudes were determined by summing the amplitudes of five consecutive wavelets. 30 The flicker response was the mean amplitude of two consecutive b-waves at 30 Hz. The following parameters were analyzed statistically: ERG a-wave amplitudes and implicit times (scotopic), ERG b-wave (scotopic and photopic) amplitudes and implicit times, and OP amplitudes. 31 The ERG data obtained from one vehicle-dosed monkey was excluded as a statistical outlier due to atypically low amplitudes in all the parameters tested.
Histopathological evaluation
At study termination, animals were euthanized with pentobarbital (100 mg/kg) administered intravenously. Eyes were harvested immediately after euthanasia. Both eyes were perfused with 5–10 ml of a 3% glutaraldehyde fixative solution via needle and syringe puncturing the sclera opposite a small incision made on the equator of each globe. The fixative was slowly perfused through the vitreal chamber to exit the perforation. Eyes were placed in 3% glutaraldehyde fixative for a 2-week period, followed by a 1-week period of 10% buffered formalin fixation prior to processing on slides for microscopic evaluation. For each eye, a horizontal section just below the optic nerve and at least five step sections were taken at 100 µm intervals and were examined starting below the optic nerve and toward the optic disk. All sections were stained with hematoxylin and eosin.
Statistical analysis
The data are reported as the means ± standard deviation (SD) or SEM. Differences were tested for significance using unpaired Student’s t-test (Prism; GraphPad Software, La Jolla, CA). A p value <0.05 was considered statistically significant.
Results
Clinical observation
The clinical courses of six NaIO3-treated monkeys is shown in Table 1. One animal (2-2) administered with repeated doses of NaIO3 at 30 mg/kg developed adverse effects, initiating 24 h postdose (HPD). Clinical signs of syncope, hypoactivity, and hypothermia resulted in a poor clinical prognosis and terminal end point at 48 HPD prior to ERG testing. Animals administered both single and repeat doses of NaIO3 at 30 mg/kg demonstrated clinical signs of decreased visual acuity, as demonstrated by difficulty focusing on external stimuli and grasping food. No significant adverse effects were noted in the two animals administered with the repeat, decreasing dosages of NaIO3 at 25 mg/kg on day 1 and 15 mg/kg on day 2. Transient postdose emesis was observed in five out of six animals administered NaIO3.
NaIO3 administration decreases ERG signals originated from the outer retina but not the inner retina
Typical waveforms of the full-field ERGs recorded from animals 4 days after PBS dosing are shown in Figure 1 (left panels of F-J). Most of the ERG parameters we measured from the four control and two PBS-dosed monkeys are similar to that reported by Bee 29 (Table 2). Shapes and implicit times of the waveforms are highly comparable. Higher amplitudes in the literature could be explained by other experiment conditions.
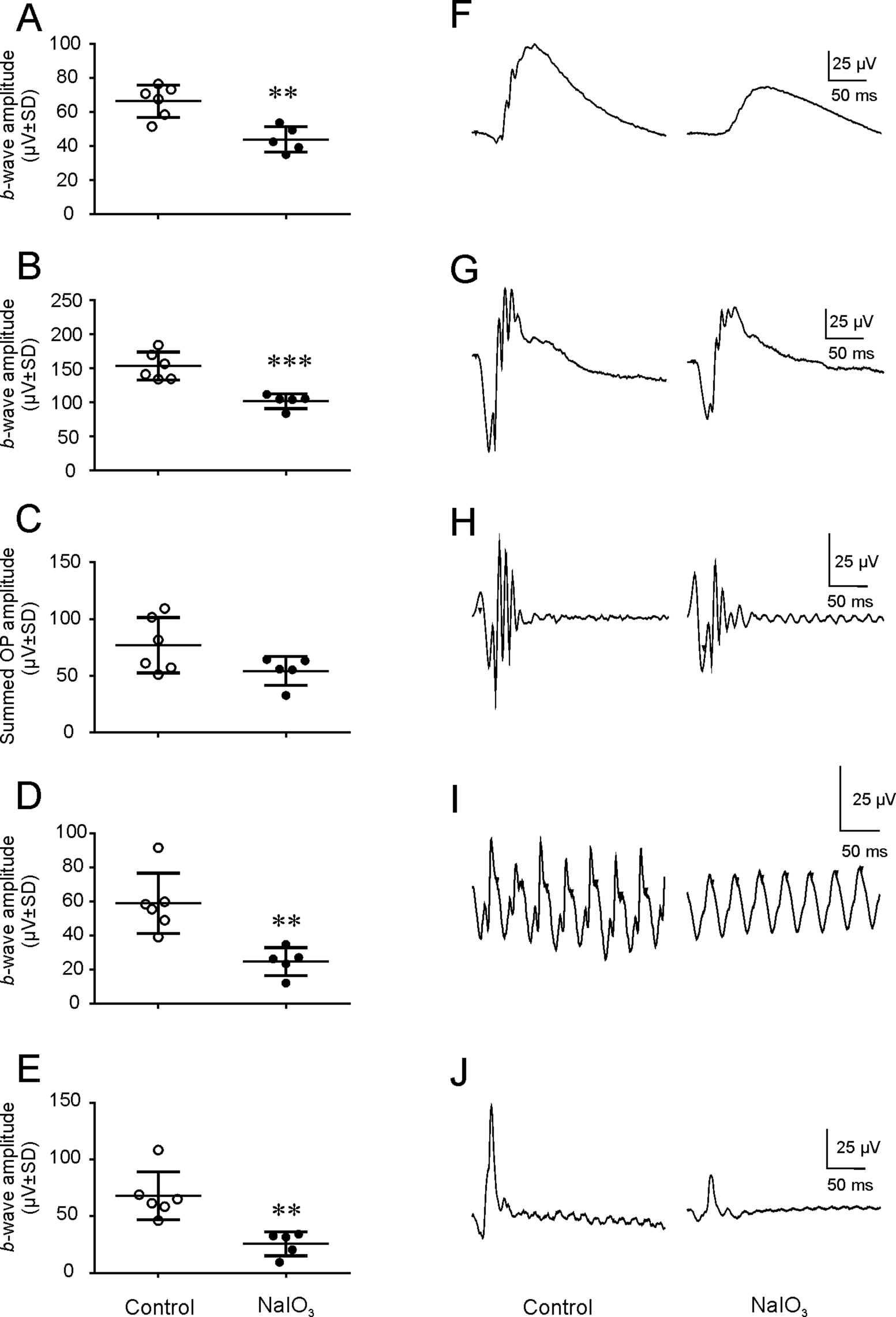
NaIO3 administration induces reductions in the dark- and light-adapted ERG signals in cynomolgus monkeys on Day 5 post_injection. b-Wave amplitudes in the rod_mediated scotopic ERG signals (A and F) and rod and cone mediated scotopic maximal responses (B and G) in the NaIO3-dosed animals were reduced significantly (**P < 0.01 and ***P < 0.001, respectively) compared with those in the PBS-control animals. b_Waves of the cone-mediated photopic flicker and cone responses were also decreased in the NaIO3-dosed animals (both **P < 0.01, D, I, E and J). Note that mean OP amplitude was not significantly affected (P > 0.05, C and H) by NaIO3 injection. ^ in A-E represent values from control or PBS injected animals. • in A-E represent the values from all animals dosed with NaIO3 in all three cohorts. Traces in F-J represent averages of 3-10 responses.
Baseline ERG parameters (n = 6).

ERG: electroretinogram; PBS: phosphate-buffered saline.
Values are means obtained from control (n = 4) and PBS-dosed (n = 2) animals. The values within the parentheses are from literature reported by Bee. 29
Intravenous administration of NaIO3 at all regimens (Table 1) demonstrated statistically significant decreases in both the a-wave (Figure 2) and b-wave components (Figure 1B and G) of the rod and cone photoreceptor–mediated scotopic maximum ERG responses, b-wave (Figure 1A and F) of the rod receptor–mediated scotopic responses, and b-wave (Figure 1D and I) of the photopic flicker and cone responses (Figure 1E and J). However, NaIO3 administration did not cause statistically significant decrease in the amplitude of the OPs (p > 0.05), compared with those obtained from the control and PBS-injected animals (pooled; Figure 1C and H). The implicit times of either a-wave or b-wave were not affected by NaIO3 (data not shown).
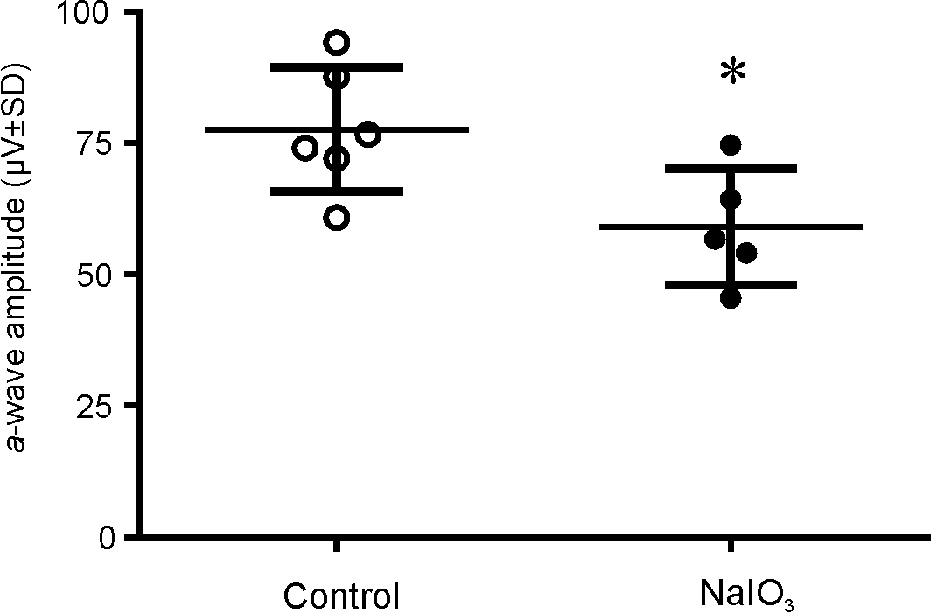
NaIO3 administration causes reductions in photoreceptor-driven ERG signals in the cynomolgus monkeys on Day 5 post_injection. NaIO3 reduces dark-adapted ERG a_wave on 4 days post-injection. a_Wave amplitudes in rod and cone mediated scotopic maximal responses were reduced significantly (*P < 0.05). ^ represent values from control or PBS injected animals. • represent the values from all animals dosed with NaIO3 in all three cohorts.
Circulating miRNAs in plasma of monkeys dosed with NaIO3
Cynomolgus monkeys (n = 6) administered NaIO3 were divided between three separate cohort studies. Each cohort study consisted of three monkeys: one monkey as PBS vehicle control and other two monkeys administered with NaIO3. The three cohort studies were carried out with different NaIO3 dosing regimens (Table 1).
Standard curves were produced with the known oligoribonucleotides as standard template in the RT-qPCR, so the copy number of the unknown miRNA samples of interest were derived from the standard curves. Although miR-183, miR-182, and miR-96 (miR-183 cluster) were measured in all the plasma samples collected, miR-96 was not consistently quantified due to miR-96 had the quality control issues such as undetectable or borderline detection (Ct values 35–38) in some plasma samples, and no specific amplification curves in melting curve analysis post-PCR amplification. Therefore, data regarding miR-96 quantification was excluded from this study. Levels of miR-182 and miR-183 in plasma were increased in four out of six monkeys administered different doses of NaIO3 when compared with vehicle control or baseline (Figure 3). In the third cohort study, miR-183 level in plasma was significantly increased (p < 0.05; Figure 3C). Reference miR-181, miR-192, and miR-92 were measured in cohort 3 animals and no elevation was observed with NaIO3 administration compared with PBS-dosed animal (Figure 4). MiR-124 was also quantified in all the plasma samples by RT-qPCR. However, there were no notable miR-124 alterations in plasma samples pre- or postdose of NaIO3 administration (data not shown).
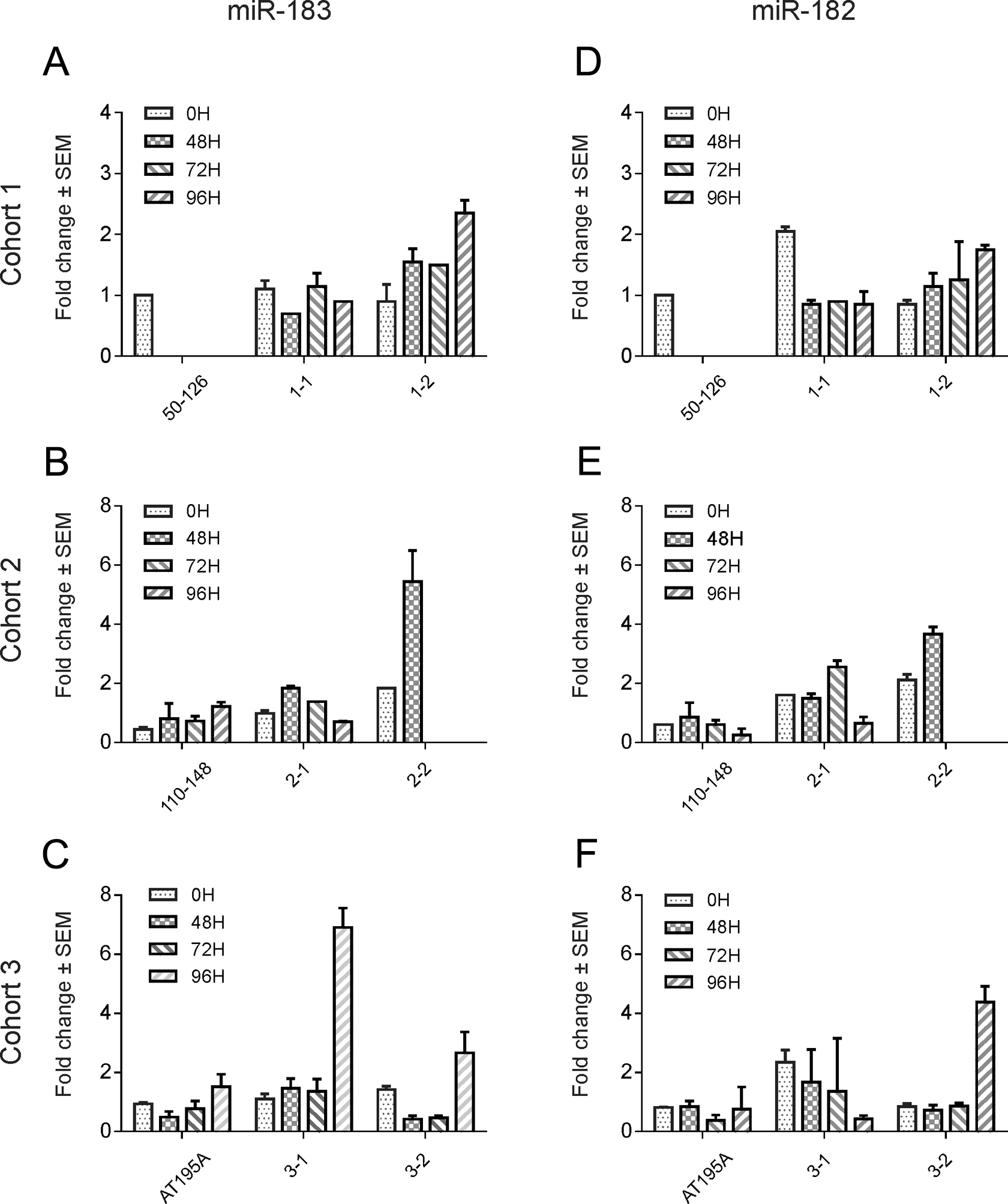
Changes in miR-183 and 182 levels in plasma of cynomolgus monkeys administered intravenous NaIO3 in comparison with control or vehicle controlled animals. Cohort 1: miR-183 level was higher (2.2× baseline) at 96 h post dose in animal 1-2 administered a single intravenous dose of at 30 mg/kg but not in the other NaIO3-dosed animal 1-1 (A). No dramatic changed was observed in miR-182 levels (D). Cohort 2: miR-183 level was higher (5.5× baseline at 48 h in animal 2-2 administered NaIO3 on Day 1 and 2 at 30 mg/kg (B). Note that miR-182 was also higher (3.7× baseline) in this animal (E). Cohort 3: miR-183 was higher (2.6× baseline, C) and miR-182 was higher (4.4× baseline, F) in animal 3-2 administered NaIO3 at 25 mg/kg and 15 mg/kg on Day 1 and 2, respectively. Only miR-183 was higher (6.9× baseline) in animal 3-1.
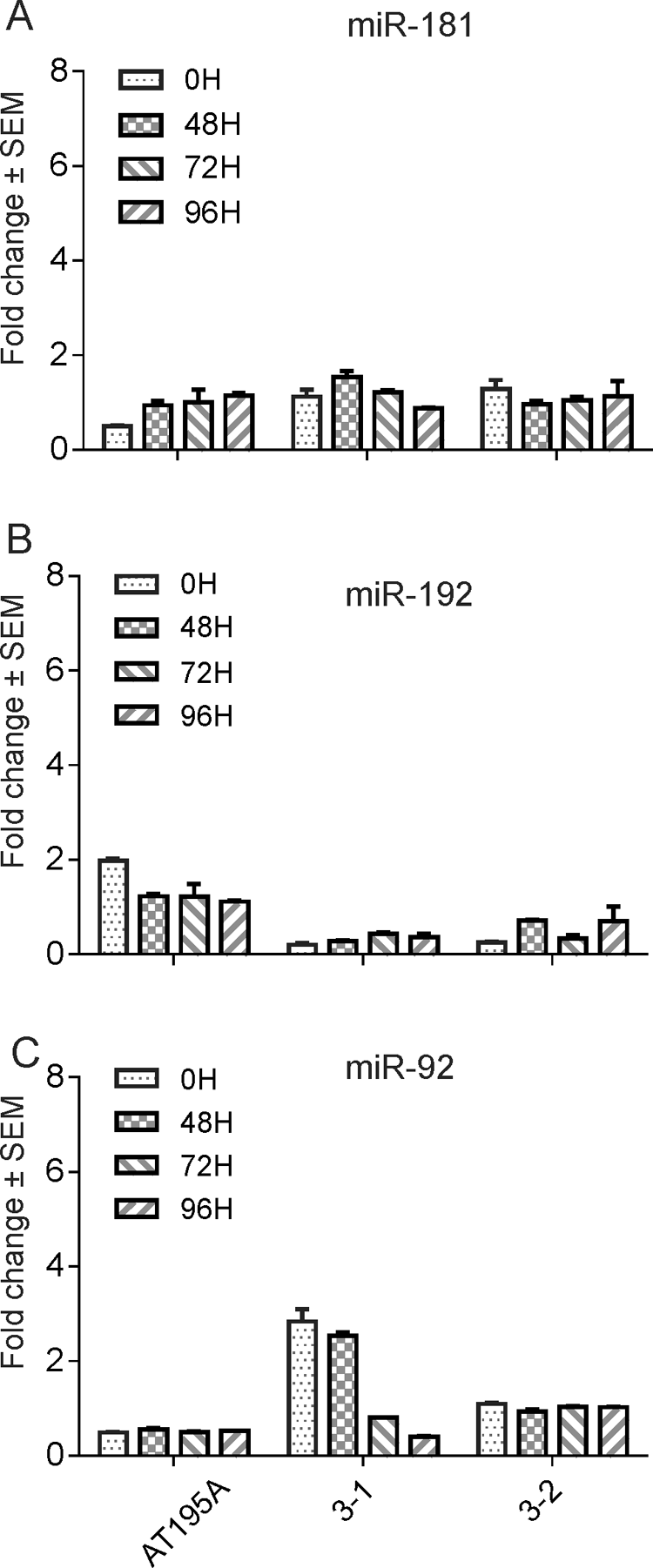
NaIO3 administration does not cause changes in the levels of circulating reference miRNAs in the monkeys. MiR-181 (A), miR-192 (B) and miR-92 (C) were not elevated in the animals dosed with NaIO3 (animals 3-1 and 3-2) compared with PBS_dosed animal (AT195A) in cohort 3. Note that miR-92 was higher in two time points of 0 and 48 HPD in animal 3-1 most likely due to notable hemolysis of the blood samples collected (C).
NaIO3-induced retinal degeneration
Retinal degeneration was mild to moderate in cohort 1 cynomolgus monkeys administered a single intravenous dose of NaIO3 at 30 mg/kg and absent or mild in cohort 2 cynomolgus monkeys administered daily intravenous dose of NaIO3 at 30 mg/kg on days 1 and 2. In cohort 3, retinal degeneration was absent or mild in cynomolgus monkeys administered two intravenous doses of NaIO3 at 25 and 15 mg/kg on days 1 and 2, respectively. The retinal degeneration, when present, primarily affected the central retina at the fovea centralis, macula lutea, and an area adjacent to the macula lutea (Figure 5). Retinal degeneration was characterized by the presence of enlarged activated pigmented and/or detached RPE cells within retinal outer segment space of the photoreceptor layer, vacuolation of the photoreceptor outer segment, and displacement of nuclei from the outer nuclear layer into the photoreceptor layer. Interestingly, the peripheral retina was not affected by NaIO3 treatment at any dose-level tested. Cynomolgus monkeys administered a single intravenous dose of NaIO3 at 30 mg/kg or two intravenous doses of NaIO3 at 30 mg/kg on days 1 and 2, and two intravenous doses 25 mg/kg (day 1) and 15 mg/kg (day 2) induced retinal degeneration that was limited to the RPE, inner segment, and outer segment of the photoreceptor layers (Table 3).
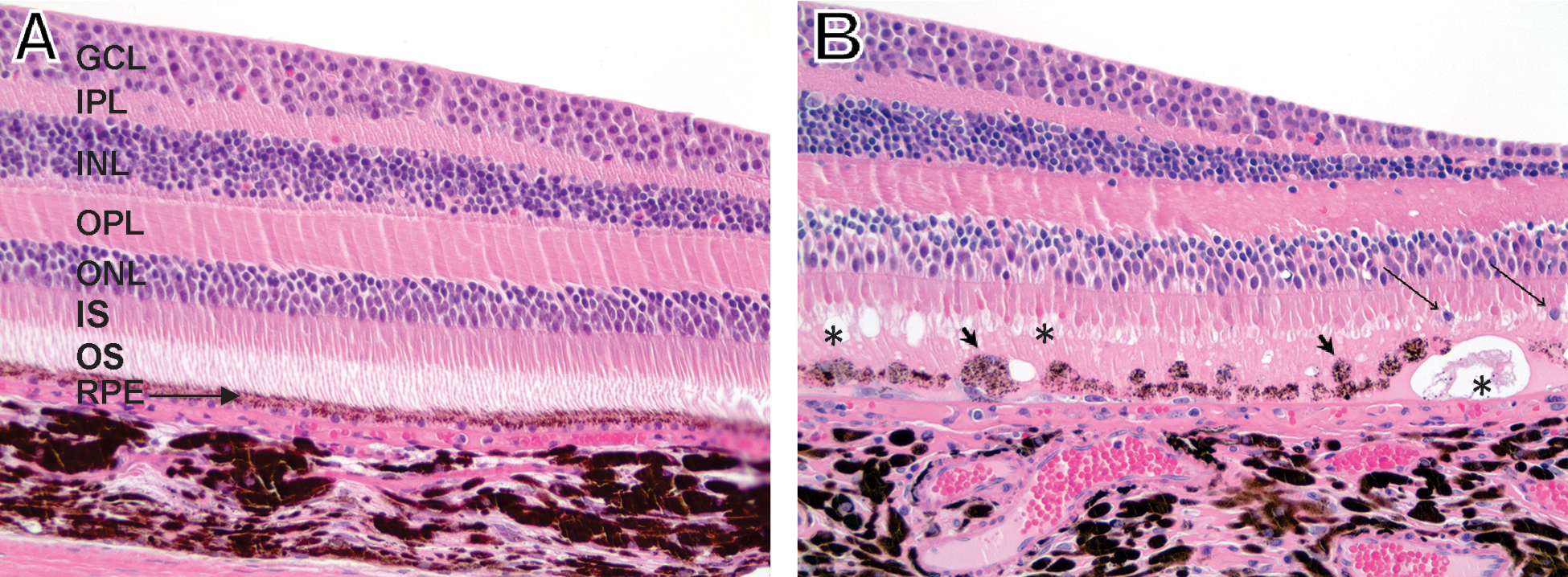
Photomicrographs of cynomolgus monkey retinas. (A) Central retina at the macula lutea (on the right) from a control cynomolgus monkey AT195A on Day 5 after two intravenous doses of PBS administered on Day 1 and Day 2 of study. Layers of the normal retina are labeled from the top or inner layer of the retina to the outer layer of the retina on the bottom. Ganglion cell layer, GCL, inner plexiform layer, IPL, inner nuclear layer, INL, outer plexiform layer, OPL, outer nuclear layer, ONL, inner segment of the photoreceptor layer, IS, outer segment of the photoreceptor layer, OS and retinal pigment epithelium, RPE. Hematoxylin and eosin stain. 200X magnification. (B) Central retina at macula lutea and fovea centralis (on the right) from a cynomolgus monkey 3-1 on Day 5 after two intravenous doses of NaIO3 administered on Day 1 (25 mg/kg) and on Day 2 (15 mg/kg). Retinal degeneration was characterized by vacuolation of the OS of the photoreceptor layer (asterisks), enlargement of the activated RPE (short arrows) and displacement of nuclei from the ONL into the OS of the photoreceptor layer (long arrows). Hematoxylin and eosin stain, 200X.
Retinal histopathological finding and miR biomarker changes in NaIO3-dosed animals.
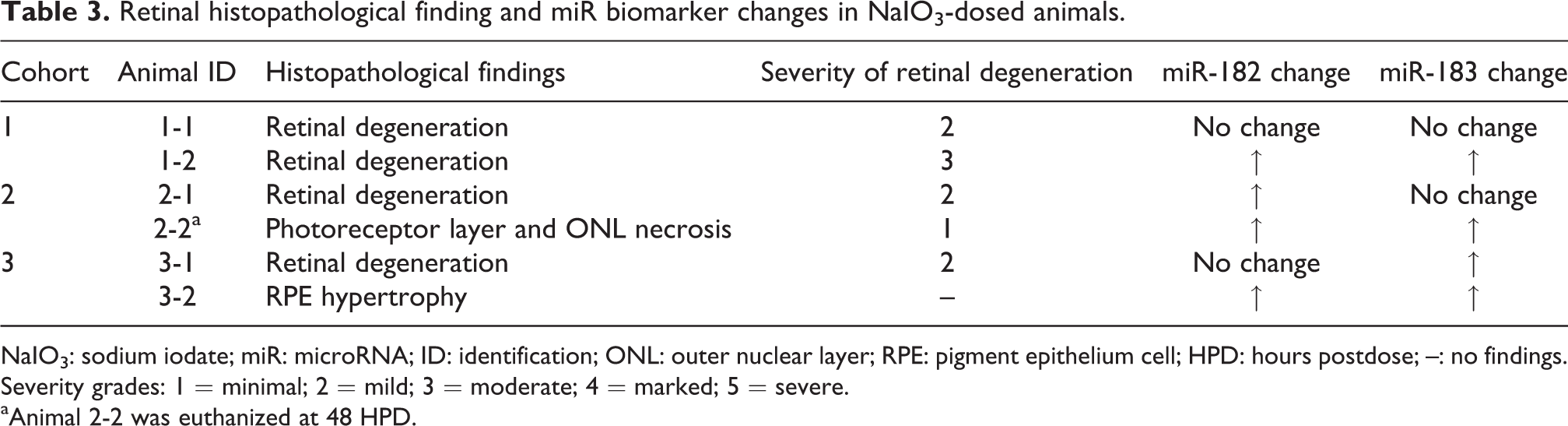
NaIO3: sodium iodate; miR: microRNA; ID: identification; ONL: outer nuclear layer; RPE: pigment epithelium cell; HPD: hours postdose; –: no findings.
Severity grades: 1 = minimal; 2 = mild; 3 = moderate; 4 = marked; 5 = severe.
aAnimal 2-2 was euthanized at 48 HPD.
NaIO3 does not affect clinical chemistry or biomarkers of vital organs
Compared with the monkey dosed with PBS, the hepatic, renal, and cardiovascular injury biomarkers were not changed in the two monkeys dosed with NaIO3 in cohort 3 study (data not shown).
Discussion
To our knowledge, NaIO3 has been tested in primates in only a few previous studies. 20 Our current study provided evidence that at sublethal concentrations, NaIO3 causes central degeneration of the RPE and secondarily inner and outer segments of the photoreceptors without significant damage to the inner retinal layers in cynomolgus monkey. We also found both the cone and rod photoreceptor functions were impaired by NaIO3 after 4 days.
It has been well-documented that NaIO3 intravenous administration in rodent and rabbit causes selective necrosis of the RPE, 10 with subsequent photoreceptor degeneration. 10,32 In the current study, we observed similar pathology in the nonhuman primate retina, particularly in the central retina close to the fovea, 4 days following NaIO3 administration.
To detect the potential functional change in the retina, especially the different retinal layers, we performed full-field flash ERG measurement prior to the necropsy. In primates, flash ERG a-wave is generated in the photoreceptor layer 33 and the b-wave originated mainly from the ON-bipolar cells 34 and the inner plexiform layer and the glia cells and depends on the a-wave. As expected, we observed significant reduction in scotopic b-wave (∼34%; Figure 1). The decrease could arise primarily from the defect in the rod bipolar cells, the reduced input from the rod photoreceptors, or the synaptic connections between the two. Further analysis indicates that the amplitudes of scotopic a-wave were also decreased (Figure 2), so the diminished scotopic b-wave most likely was due to the decreased input to the bipolar cells attributed to the degeneration of the photoreceptors. This result indicated that NaIO3 mainly damaged the functions of photoreceptors. Furthermore, the cone photoreceptor responses were impacted more (∼62%) than the rod photoreceptor responses (∼34%). Indeed, in most of the monkeys we tested, our ERG data were consistent with the histopathologic findings of mild to moderate central photoreceptor degeneration and RPE damages (Figure 5B). The OP, however, was not statistically significantly affected by NaIO3 administration. Lack of an OP response indicated that the inner retinal function (from the inner nuclear layer to ganglion cell layer) was not impacted by this retinal toxicant, since the OP response reflected inner retinal activities in monkeys 35 and humans. 36 In this study, we did not further explore the functional changes in the retinal ganglion cells with other special ERG waveforms, such as PhNR 37 or pattern ERG.
In this study, a single dose of 30 mg/kg NaIO3 in two monkeys resulted in limited clinical signs and ERG changes in 1 of 2 animals. Therefore, in the subsequent cohort (n = 2), the 30 mg/kg NaIO3 dose was escalated in a 2-day repeat-dose study. The 2-day repeat-dose study with 30 mg/kg NaIO3 in monkeys was too toxic and resulted in severe adverse clinical signs and early euthanasia of one animal. As a result, the 30 mg/kg dose of NaIO3 was scaled down to 25 and 15 mg/kg in the next 2-day repeat-dose study. In this subsequent study, two animals administered 25 and 15 mg/kg as single, decreasing dosages tolerated the treatments with minimal adverse clinical signs and significant reductions in ERG signals. Furthermore, no changes were observed in the hepatic, heart, and renal injury biomarkers in the circulating blood with the NaIO3 dose reductions. Hence, 25 and 15 mg/kg doses produced retinal toxicity in a 2-day repeat-dose study design in cynomolgus monkeys and were considered the optimal doses because of the selective ocular toxicity. This repeat, decreasing dose paradigm resulted in comparable effects observed in sheep at a single intravenous dose of 30 mg/kg NaIO3, which caused pronounced reductions in a- and b-waves of the ERG recorded 3 days following the injection. 38 Thus, repeated doses at lower level seems better than single dose at higher level, presumably because the first dose breaks down the RPE barrier initially and facilitates the subsequent NaIO3 distribution at lower dose. 39
The major pathology finding in cynomolgus monkeys occurred in the RPE and photoreceptors but not in the inner layers of the retina. This is in agreement with the literature. For example, in light microscopy, Noell 32 observed very little or no inner segment involvement in the rabbit NaIO3 model. Another notable finding of our study is that the major retinal degeneration was observed around the fovea area, but not in the peripheral retina, which had been shown severely impacted in rodent as reported in a NaIO3 rat study. 40 The distribution of the retinal degeneration observed in the nonhuman primate may more accurately reflect the retinal toxicity in humans because of the presence of the macula. The sensitivity of the macula to retinal toxicity may be attributed to abundant distribution of vessels around the fovea and packed cone photoreceptors in this area, more light stimulation in this region, or different mitochondria in the more bioenergetical active cone 41 versus rod photoreceptors. 42 We hypothesized that systemically administered NaIO3 in the blood stream damaged the RPE via choroid at the outer blood retinal barrier and/or via the retinal vessels at the inner blood retinal barrier in the macula lutea.
Due to the limited number of cynomolgus monkey available, we could not monitor the retinal changes with ERG or pathology at multiple time points as with miRNA biomarker measurements, thus, we can’t provide further evidence of the temporal function–structure relationship in the retinal degeneration. The functional impairment may well precede the morphologic changes on 4 days after drug treatment. Previously, Nissan and coworkers examined the changes in ultrastructure and function of sheep retina and observed the reduction in a- and b-amplitudes prior to ultrastructural changes in the neuroretina. 38 In future studies, multiple time point monitoring would be interesting.
In this study, we monitored the development and progression of the retinal damage by measuring the potential retinal injury biomarker miR-183 cluster in the circulating blood. The miR-183 cluster was increased in some animals as early as 48 HPD and appeared to be correlated to the severity of the retinal degeneration after NaIO3 treatment (Table 3). Although the exact reasons and underlying mechanisms of increased plasma miRNA-183 cluster levels in monkeys following NaIO3 administration remains to be explored, however, previous studies in adult mice revealed that 19 miRNAs are specifically expressed in the retina. 26,43 Among these, a polycistronic, sensory organ-specific paralogous miRNA cluster that includes miR-96, miR-182, and miR-183 were found to be conservation of synteny to human chromosome. In situ hybridization showed that members of this cluster are expressed in photoreceptors, bipolar, and amacrine cells 26 but not in the RPE. 44 Therefore, these miRs have been speculated to play important role in normal function of the retina. In spite of the absence of report of expression in primates, the expression in primates may be likewise, because miR-183 family have been found to be highly conservative across vertebrates. 25 Our hypothesis stated that the increased miR-183 cluster in the circulating blood was due to the leakage of RPE and Bruch’s membrane by NaIO3 toxicity. There is a possibility that cells of the neurosensory retina (ganglion cell, amacrine, or horizontal cells) 45 and photoreceptors 46 may also be directly involved in NaIO3-mediated toxicity. To date, this is the first report describing the plasma miRNA profile in a nonhuman primate in response to toxic doses of intravenously administered NaIO3, therefore the translatability of the nonhuman primate data to the human as well as the neuronal/neurotrophic factor pathways associated with the neuronal retina injury remains to be explored in future research.
We concluded that the sublethal doses of 25 and 15 mg/kg NaIO3 in cynomolgus monkeys in 2-day repeat-dose study design markedly damaged the photoreceptors both functionally and morphologically. Therefore, this nonhuman primate model and the intravenous doses of NaIO3 used in this study could be used in future toxicity studies to serve as a positive control for retinal injury or in efficacy studies for pharmaceutical intervention.
Footnotes
Acknowledgments
The authors would like to thank Steven Kreuser and Amy Martunas for anesthesia support and Carlos Esparza for his excellent expertise and support for tissue processing and staining. We also thank Chris Somps for helpful comments on the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
