Abstract
Introduction:
Cerebral venous thrombosis (CVT) is a less common stroke subtype. While long term outcome factors have been extensively studied, short term deterioration remains poorly understood.
Patients and methods:
We conducted a 10-years retrospective analysis at a high-volume tertiary center, including consecutive patients diagnosed with CVT. The primary outcome was early deterioration (ED), defined as decrease in Glasgow Coma Scale, de novo or worsening of focal deficit, death from neurological cause, new or enlarged parenchymal lesions or subarachnoid hemorrhage during hospitalization. Multivariable logistic regression analysis was performed to identify factors associated with ED.
Results:
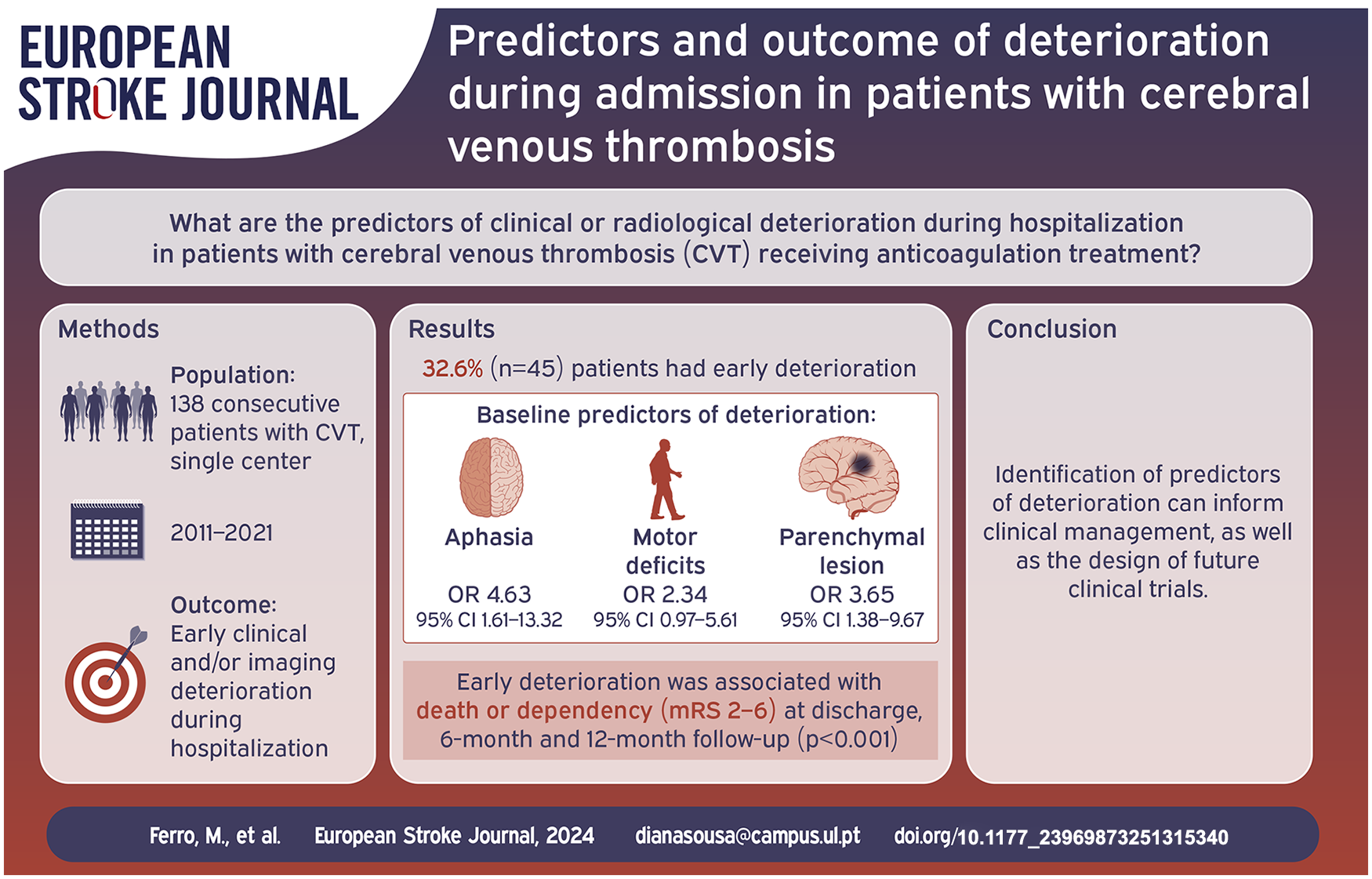
We included 138 patients (81.2% female, median age 42.0 years (IQR 29.3–49.0)). Forty-five (32.6%) patients had ED, with 33 (23.9%) showing clinical deterioration and 35 of 104 (33.7%) imaging worsening. Variables selected from the multivariate model for association with ED were aphasia (OR 4.63, 95% CI 1.61–13.32), motor deficits (OR 2.34, 95% CI 0.97–5.61), and parenchymal lesion (OR 3.65, 95% CI 1.38–9.67). Twenty-seven patients underwent endovascular treatment after deterioration. Patients in the ED group had worse functional outcome at discharge, 6 and 12 months (p < 0.001).
Discussion:
One third of patients in this cohort experienced ED. Patients with aphasia, motor deficit, or parenchymal brain lesion at baseline were at higher risk. These patients performed worse at long term follow-up.
Conclusion:
We identified predictors of ED in patients with CVT. These patients should be carefully monitored. These findings may inform the design of future clinical trials aimed at evaluating additional therapeutic interventions in the acute phase.
Introduction
Cerebral venous thrombosis (CVT) is caused by complete or partial occlusion of the major cerebral venous sinuses or the feeding cortical veins. 1 The incidence of CVT is estimated to be 1.3–1.6 per 100.000, mainly in adults between 20 and 50 years old, and threefold more common in women than in men. 2
Long term poor outcome factors are widely studied and classically included age, coma, thrombosis of the deep venous system, concomitant malignancy, mental status disturbance, male sex, and intracranial hemorrhage.3–6 Some more recent cohort studies have also identified focal neurological deficits and parenchymal brain lesions as predictors of worse outcomes.4,7,8
In contrast, early deterioration during admission remains poorly understood, despite estimates suggesting that neurological worsening occurs in one-quarter to one-third of patients during the acute phase.3,9 Moreover, endovascular treatment is currently mostly used as a rescue treatment for patients who are experiencing clinical or imaging deterioration despite standard therapy, or that have contraindications to anticoagulation.10,11
Therefore, we aimed to explore predictors of clinical or radiological deterioration during admission in patients with CVT under treatment with anticoagulation. This may contribute to a timelier identification of this high-risk group and eventually consideration for patient selection in future prospective trials of adjunctive therapies for this population.
Patients and methods
Study design and participants
This retrospective analysis included consecutive adult patients with CVT admitted to the stroke unit of a high-volume tertiary centre, which serves as a referral hub for endovascular treatment (EVT), between January 2011 and August 2021. Patients were identified through a prospective registry maintained at the centre, which provided a basis for data collection. Demographic, clinical, and radiological data was thoroughly reviewed through detailed examination of medical records. CVT diagnosis was confirmed using Computed Tomography (CT) venography, Magnetic Resonance (MR), or catheter-based angiography. For patients undergoing EVT, follow-up imaging with CT or MRI was performed 24 hours post-procedure to screen for potential complications. Additional imaging was conducted in cases of neurological deterioration or as clinically indicated by the treating physician, often including a scan before discharge.
Data
Collected variables included the following: 1) Demographic information: sex, ethnicity, age at CVT, previous modified Rankin Score (mRS) and risk factors for CVT; 2) CVT diagnosis-related data: presenting signs and symptoms at baseline, diagnostic modality, thrombus load, parenchymal lesions, subarachnoid hemorrhage, and International Study on Cerebral Vein and Dural Sinus Thrombosis Risk Score (ISCVT-RS) calculated as described by Ferro et al. 12 ; 4) Treatment: anticoagulation therapy and endovascular treatment; 5) Clinical evolution: intensive care unit (ICU) admission, decompressive craniectomy, neurological complications (de novo impairment of consciousness or decline of level of consciousness, de novo headache or worsening of headache intensity, seizures, new focal neurological deficit or worsening of baseline focal neurological deficit, namely aphasia, neglect, motor, sensory or visual field deficit, ataxia); 6) Imaging evolution: new parenchymal lesions or subarachnoid hemorrhage in follow-up imaging during hospital admission, worsening of parenchymal lesion defined as increase, or de novo, non-hemorrhagic lesion or hematoma volume from baseline; 7) Outcome at discharge, 6 and 12 months after diagnosis: mRS score; focal neurological symptoms, seizures, headache or self-reported cognitive complaints (considered if patients mentioned memory difficulties, attention deficit, linguistic difficulties or other disturbance of cognitive domains, interfering with daily activities or work); Recurrent CVT; 8) Death during follow up.
Symptoms at baseline and during admission were based on a thorough review of medical records, including emergency department admission notes, and discharge summaries. Onset was considered based on the last day well, prior to the onset of the first symptom thought to be related to CVT. 13
Thrombus load was evaluated using a scoring system14–16 that assigns a score of 1 to each blocked segment of the cerebral venous system. The system categorizes the cerebral venous system into anatomical divisions, namely: superior sagittal sinus, right transverse sinus, left transverse sinus, right sigmoid sinus, left sigmoid sinus, torcula, straight sinus, deep venous system, cortical veins, right jugular vein, left jugular vein, cavernous sinuses, cerebellar veins. For the deep venous system, cortical veins, and cerebellar veins, a single point is given for each group, regardless of the number of occluded veins within that group. Specifically, these groups receive 0 points if none of the veins are fully blocked or 1 point if at least one vein is fully blocked. Additionally, the ISCVT Risk Score was calculated for each patient, and assessed as a potentially relevant variable in the analysis.
This study was performed in accordance with the ethical standards of the Declaration of Helsinki and was approved by the local institutional Ethics Committee. As the study was retrospectively designed and deidentified data were used, the requirement to obtain informed consent was waived by the board.
Outcomes
The primary outcome measure was early deterioration during hospitalization. Early deterioration was defined as a decrease in the Glasgow Coma Scale category (grouped as GCS 15; 13–14; 9–13; <9), de novo or worsening of focal deficit, death from a neurological cause, new parenchymal lesions, new subarachnoid haemorrhage or an increase of non-hemorrhagic lesion or hematoma volume from baseline, during hospitalization. The stratification of GCS in categories commonly used in CVT research was selected to standardize the classification of changes in consciousness level. Neurological worsening and new or enlarged brain lesions were classified by consensus between two investigators (MF and MS for clinical findings, with discrepancies resolved by a senior neurologist, DAS; SB and MB for imaging findings, with discrepancies resolved by a senior neuroradiologist, IF).
Statistical analysis
Categorical variables are presented as absolute frequencies and percentages. For continuous variables, those that are normally distributed are presented as means and standard deviations (SD), while those not normally distributed are presented using medians and interquartile ranges (IQR). Participants were divided into two groups for each observed outcome. For comparing group differences, t-tests were used for normally distributed variables and Mann–Whitney U tests for non-normally distributed variables. For categorical data, Pearson’s chi-squared tests were employed, and Fisher’s exact test was used when expected frequencies were less than five.
All values are reported with a 95% confidence interval. To answer the main objective, independent variables with a significance level <0.05 in the bivariate analysis were included in a backward stepwise multiple logistic regression model (Wald) to identify predictors for each outcome, with a model significance level set at 0.10. The location of sinus thrombosis was not included as a predictive variable due to the frequent occurrence of multiple thrombosis locations, which complicates the attribution of effects to specific sites within the logistic regression framework. We also excluded midline shift and signs of impending brain herniation at baseline as predictive variables in our model due to the known strong probability of early deterioration and need for prompt surgical intervention. A sensitivity analysis was performed, excluding patients with midline shift at baseline, to further explore its potential impact. To prevent collinearity in the model, variables that could overlap significantly were carefully considered and, where appropriate, excluded. Additionally, variables with fewer than five cases were also excluded to ensure stability of the model estimates. Goodness-of-fit of the logistic regression was evaluated using the Hosmer-Lemeshow test to assess model calibration. We used complete case analysis. No imputation techniques were applied. The statistical analysis was carried out using SPSS Statistics version 29.0.
Results
Study population
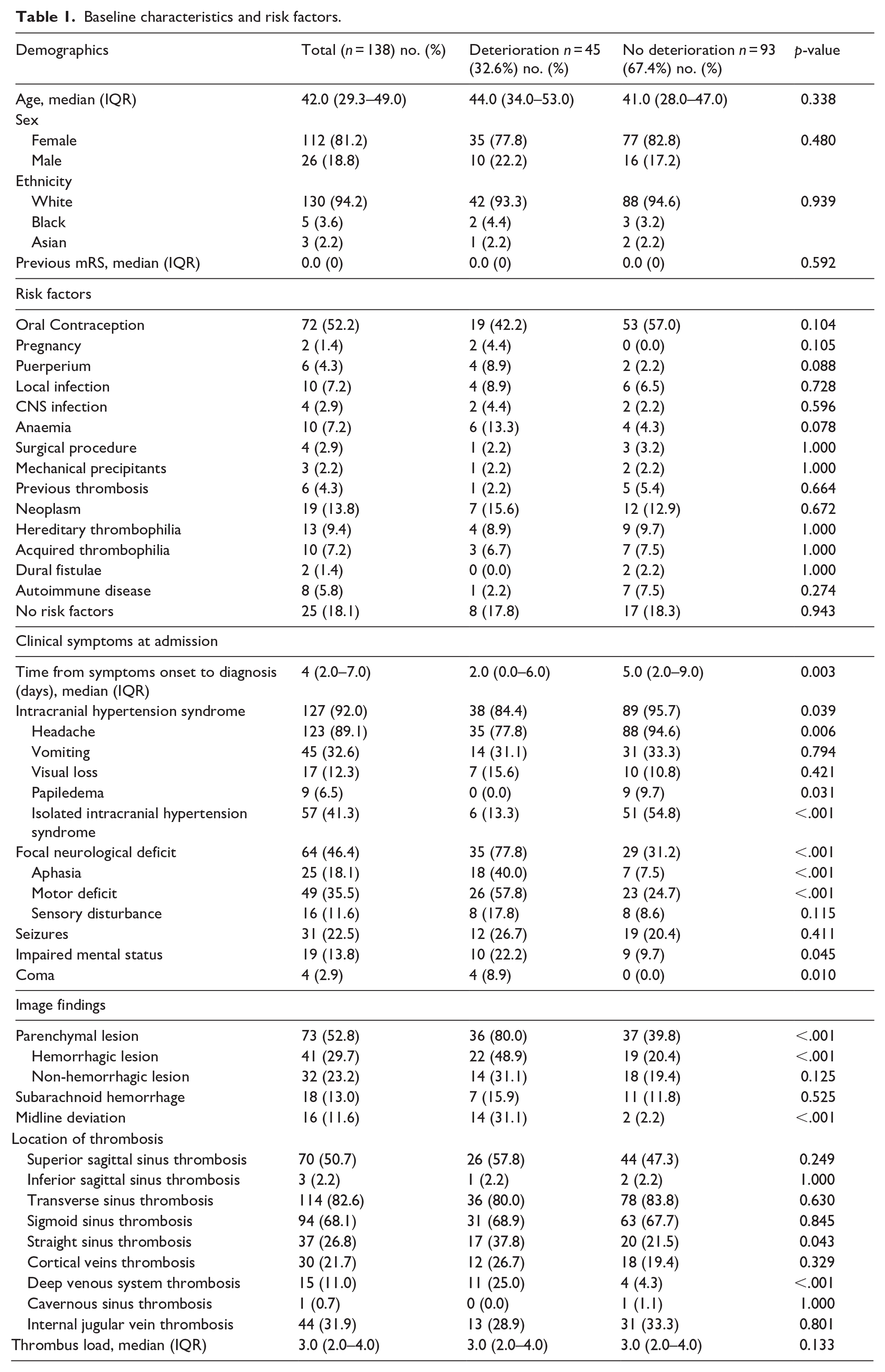
From the 139 individuals admitted with diagnosis of CVT during the study period, we included 138 patients. Among these, 85 patients (61.6%) were transferred from peripheral hospitals lacking stroke unit care. One patient was excluded, as the admission was for endovascular treatment at our center, and a prompt transfer back to the referring tertiary hospital precluded sufficient data collection. The median age of the patients was 42.0 (IQR 29.3–49.0) and 112 were female (81.2%). The most common risk factor for CVT was oral contraception (n = 72; 52.2%), followed by malignancy (n = 19; 13.8%) and hereditary thrombophilia (n = 13; 9.4%) (Table 1).
Baseline characteristics and risk factors.
Baseline clinical and neuroimaging features
At admission, headache was the most common symptom (n = 123; 89.1%), followed by motor deficit (n = 49; 35.5%), vomiting (n = 45;32.6%) and seizures (n = 31; 22.5%). Symptoms were also grouped into the following categories: 127 (92.0%) had symptoms of intracranial hypertension (headache, vomiting, reduced visual acuity, papilledema or cranial nerve palsy), 64 (46.4%) had focal neurological deficits and 43 (31.2%) had disturbance of mental status or consciousness (Table 1). Median time from beginning of the symptoms to diagnosis was 4 (IQR 2–7) days.
CVT diagnosis was confirmed using CT venography in 73 patients (52.9%), MR in 63 patients (45.7%), and catheter-based angiography in two patients (1.4%). At baseline imaging, most patients had parenchymal lesions (n = 73; 52.8%) mainly in the left hemisphere (n = 35) or bilateral (n = 23). Forty-one (29.7%) patients had hemorrhagic parenchymal lesions, 32 (23.2%) had non-hemorrhagic lesions and 18 patients (13.0%) had subarachnoid hemorrhage. Sixteen patients (11.6%) had midline shift (median, IQR; 4.0, 3.0–5.0 mm).
The distribution of thrombosis location was as follows: transverse sinus was the most commonly involved, affecting 114 patients (82.6%), followed by the sigmoid sinus in 94 (68.1%), superior sagittal sinus in 70 (50.7%), internal jugular vein in 44 (31.9%), straight sinus in 37 (26.8%), cortical veins in 30 (21.7%) and deep venous system in 15 (11.0%). Median thrombus load was 3.0 (IQR 2.0–4.0).
Treatment
All patients but one received treatment with anticoagulation (137; 99.3%). One patient died shortly after admission before therapy initiation. Forty patients (29.0%) received endovascular treatment, either local thrombolysis (n = 35; 89.7%) and/or mechanical thrombectomy (aspiration n = 26; 66.7%, stent-retriever n = 1; 2.6%, balloon n = 29; 74.6%) (Table S1).
Deterioration during hospitalization
The median duration of hospitalization was 6.0 (IQR 3–13) days. During hospitalization, 45 (32.6%) patients had early deterioration. Thirty-three patients (23.9%) had clinical deterioration and 35 patients (33.7%) of the 104 who performed a second exam had imaging worsening (Supplemental Table S2). In patients with imaging deterioration, follow-up imaging was performed at a median of 1.5 days (IQR 0.75–3.0) after diagnosis. Most patients (N = 26; 57.8%) met at least two criteria for deterioration. Moreover, seven (5.1%) patients had new-onset seizures, 21 (15.2%) patients were admitted to the ICU and 9 (6.5%) were submitted to craniectomy.
Of note, 27 patients underwent EVT after early deterioration, which accounts for 67.5% of the total number of patients selected for EVT in this cohort (n = 40). Baseline ISCVT-RS did not correlate significantly to early deterioration (p = 0.06). Seizures during admission were not significantly more prevalent in the early deterioration group (p = 0.411). Time from symptom onset to diagnosis was shorter in patients that deteriorated - 2.0 (IQR 0.0–6.0) versus 5.0 (IQR 2.0–9.0) days (p = 0.003).
From five variables initially identified through their significant bivariate associations, namely time from symptom onset to admission (p = 0.003), aphasia (p < 0.01), motor deficit (p < 0.01), parenchymal lesion (p < 0.01), and mental status disturbance (p = 0.045), three were selected from the multivariate logistic regression model: aphasia (OR 4.63, 95%CI 1.61–13.32), motor deficits (OR 2.34, 95%CI 0.97–5.61) and parenchymal lesion (OR 3.65, 95%CI 1.38–9.67) at baseline (Table 2). Model calibration was assessed using the Hosmer-Lemeshow test, which indicated an acceptable fit (p = 0.095). On the other hand, patients who presented with isolated intracranial hypertension syndrome (n = 57) where significantly less likely to deteriorate during hospitalization, although deterioration still occurred in approximately 1 in 10 patients (n = 6) with this presentation. Patients with both parenchymal brain lesions and aphasia or motor deficits had the highest risk of early deterioration. Specifically, 73.9% of patients with aphasia and parenchymal lesions experienced early deterioration, compared to only 29.2% of those with parenchymal lesions but without these clinical signs (Supplemental Table S3).
Multivariate analysis of variables predicting early deterioration.

In the early deterioration group, 14 patients had midline shift at baseline, compared to 2 in the group without deterioration (p < 0.001). The sensitivity analysis excluding patients with midline shift at baseline found that mental status disorder (p = 0.038), aphasia (p = 0.022), motor deficit (p = 0.004), and parenchymal lesion (p < 0.001) were significantly associated with early deterioration, as in the original cohort. However, in the multivariate logistic regression model, only parenchymal lesion was retained as a significant independent predictor of early deterioration (OR 4.74, 95% CI 1.91–11.77).
Long term outcomes
In the overall cohort, the Modified Rankin Scale at discharge was 0 or 1 in 96 patients (69.6%). Five patients died during hospitalization: four due to brain herniation and one due to myocardial infarction. Of the 133 patients who survived, 62 (46.6%) were transferred to other hospitals for continuation of care or rehabilitation. Of these, 55 (88.7%) had been referred from non-stroke centre emergency departments at peripheral hospitals. Most patients (n = 72; 54,1%) were discharged home. Follow-up at 6 and 12 months was available for 128 patients, as 5 patients died and 5 patients were lost to follow-up. Four of the five patients lacking follow-up in the early deterioration group died from neurological causes during admission. The majority of patients (n = 124; 96.9%) were receiving anticoagulation therapy at 6 months, and 87 (67.9%) continued treatment at 12 months.
Deterioration during hospitalization was associated with death or dependency (mRS 2–6) at discharge (p < 0.001), 6-month (p < 0.001), and 12-month (p < 0.001) follow-up, as well as with cognitive complaints at the 12-month follow-up (p = 0.007) (Table 3).
Clinical outcomes and events during the first 12 months of follow-up.

Discussion
One third of patients in this cohort experienced early deterioration which is slightly higher than the rates described by ISCVT and Bushnaq et al, at 23% and 25.85% respectively.17,18 However, our findings align with those of Yii et al., who also reported in-hospital deterioration in 34% of the patients. 9 This higher incidence of early deterioration may reflect a selection bias, as almost two thirds of our patients were referrals from other hospitals. Some of these referrals included critically ill or at-risk patients selected for endovascular treatment, likely leading to an increased proportion of patients presenting with more severe phenotypes. Additionally, our definition of deterioration included not only clinical but also imaging findings, specifically the presence of new or worsening intracranial lesions at the follow-up imaging. This approach is based on the assumption that even asymptomatic worsening on imaging indicates progression of the underlying pathological process and, therefore, should be valued in this context as it represents potential exacerbation of brain damage. This is also supported by a previous analysis from the ISCVT cohort, where new parenchymal lesions were noted in one-third of deteriorating patients during hospitalization. 3
We found that focal deficits, particularly aphasia or motor deficits, and evidence of parenchymal lesion in the baseline imaging assessment were independent predictors of early deterioration. Aphasia, a common focal deficit in CVT patients, was observed in 19% of cases in the ISCVT cohort. 3 It results from lesions in the dominant hemisphere, which is more likely to be symptomatic. 19 Also, worsening in language deficits is particularly noticeable and, in this cohort, left hemisphere lesions were more prevalent.
Furthermore, aphasia is frequently linked to cortical vein thrombosis, which may be more prone to deterioration, as it corresponds to terminal circulation in critical brain areas responsible for language and other cognitive functions. Prehospital focal motor deficit has similarly been associated with higher risk of early deterioration in patients with acute stroke. 13 However, aphasia and motor deficits may also not reflect stronger biological associations with early deterioration than other deficits but rather the challenge of assessing changes in subtler symptoms, including cognitive impairments.
The association between baseline parenchymal brain lesions and ED aligns with the findings of Yii et al. 9 In the sensitivity analysis that excluded patients with midline shift at baseline, who might have a higher likelihood of deterioration, parenchymal lesions remained an independent predictor of early deterioration in multivariate analysis. This reinforces the robustness of this association, although the smaller sample size may also have limited the detection of associations with other possible predictors.
Moreover, the combination of aphasia or motor deficits, and the presence of parenchymal brain lesions on imaging was associated with the highest proportion of patients experiencing early neurological deterioration, exceeding the rates observed in patients with only one predictor, such as parenchymal brain lesions alone. Among patients with both aphasia and parenchymal lesions, 73.9% experienced early deterioration, compared to 29.2% in those with parenchymal lesions but no clinical signs. On the other hand, we could confirm that patients with isolated intracranial hypertension rarely deteriorate.
Identifying at-risk patients for early deterioration revealed a possible impact on outcomes at discharge, 6 and 12 months, as these patients performed worse. This also aligns with the findings reported by the VENOPORT study, in which worsening after admission was identified as a predictor of poorer long-term outcomes. 20
The TO-ACT trial 21 found no improvement in functional outcome in patients receiving endovascular treatment over best medical management alone. The inclusion criteria of this study focused on patients with known poor prognosis criteria (at least one), namely mental status disorder, coma, ICH, or thrombosis of the deep cerebral venous system. We hypothesize that the distinction between early deterioration and overall poor outcome is relevant, because the identification of patients at risk for deterioration may offer an opportunity to mitigate progression and potentially salvage brain tissue, whereas indicators of worse prognosis often may also reflect stages of irreversible brain damage. However, and due to the current lack of evidence supporting endovascular treatment in these patients, most patients selected for endovascular treatment in our cohort, as in most others, had already experienced neurological or imaging deterioration during hospital admission before receiving the intervention. 22 Future studies could explore whether earlier endovascular intervention, applied before deterioration sets in, could prevent further complications in high-risk patients.
When considering other more common types of stroke, early neurological deterioration has been extensively assessed in patients with acute ischemic stroke23,24 and in intracerebral haemorrhage. 25 As in our study, early deterioration in these conditions was usually predicted by a combination of clinical (e.g. higher NIHSS score/focal symptoms and lower GCS) and imaging (e.g. lower ASPECTS, clot burden, hematoma volume or expansion, spot sign and parenchymal lesion) baseline characteristics. This is in line with our hypothesis that a combination of clinical and imaging findings is necessary to better predict this complex outcome.
While our study benefits from being conducted at a high-volume tertiary referral centre, which ensures centralized care and standardized procedures, it also has several limitations. Firstly, its retrospective design, based on a decade of medical records, introduces variability in treatment practices due to evolving guidelines and potential biases in data recording. The inclusion of patients referred from other hospitals might have skewed the observed severity of clinical presentations, possibly overestimating the incidence of severe outcomes. Also, patients who experienced early death in the ICU prior to transfer to the stroke unit may not have been captured in the registry. However, differential loss to follow-up between the early deterioration and no deterioration groups is unlikely to have introduced significant bias, as the large majority of patients classified as lacking follow-up in the early deterioration group had documented neurological death during admission. We acknowledge the broad range of severity encompassed in our definition of early deterioration and suggest that future targeted analyses of subgroups could provide a deeper understanding of its clinical implications. The possibility of subclinical radiological deterioration in patients without follow-up imaging may have introduced selection bias. However, given that most of those patients without follow-up imaging exhibited a benign clinical course, usually with isolated headache, we believe that the impact on the main findings should be minimal. We did not include occlusion location in the model due to the occurrence of multiple simultaneous occlusions in most patients, which complicates interpretation. Additionally, thrombus load was not associated with deterioration during admission. Cognitive complaints were self-reported and formal neuropsychological assessment was not mandatory. Finally, the sample size might limit the statistical power. To prevent model overfitting and maintain statistical validity, variables with very few cases were excluded, which might have narrowed the scope of our predictive model. These factors may hinder the generalizability of our findings to settings with different patient demographics or treatment approaches, particularly since most patients were selected for endovascular treatment after showing signs of deterioration. Also, we could not determine whether clinical deterioration during admission was an independent predictor of long-term outcomes due to the relatively small sample size. However, this is an important aspect that future studies with larger cohorts could explore through multivariate analysis.
Conclusion
Our study identifies specific predictors of early deterioration in patients with CVT, namely the presence of aphasia and motor deficits and the evidence of parenchymal brain lesions on imaging. These findings underscore the importance of vigilant monitoring for this subgroup, which appears to be at greater risk. Given the potential worse prognosis, these patients may benefit from proactive management strategies, including the consideration of early endovascular interventions. These predictors could enhance the design and targeting of future clinical trials aimed at evaluating additional therapeutic interventions in patients with CVT.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251315340 – Supplemental material for Predictors and outcome of deterioration during admission in patients with cerebral venous thrombosis
Supplemental material, sj-docx-1-eso-10.1177_23969873251315340 for Predictors and outcome of deterioration during admission in patients with cerebral venous thrombosis by Margarida Ferro, Sofia Bettencourt, Mafalda Soares, Mariana Baptista, Cláudia Marques-Matos, Isabel Fragata, Ana Paiva Nunes and Diana Aguiar de Sousa in European Stroke Journal
Footnotes
Acknowledgements
None
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was not sought for the present study because of its retrospective nature.
Ethical approval
Ethical approval for this study was obtained from the local ethical committee and Conselho de Administração, Centro Hospitalar Universitário Lisboa Central - INV 399.
Guarantor
M.F.
Contributorship
M.F. wrote the first draft of the manuscript. M.F., M.S. and S.B. worked on data collection. M.F., M.S., D.A.S., A.P.N. and C.M.M. contributed to the clinical data analysis. S.B., M.B. and I.F. contributed to the imaging data analysis. M.S. and D.A.S. contributed to the statistical analysis. D.A.S., M.B., A.P.N., I.F. and C.M.M. contributed to the supervision. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
