Abstract
Background:
Severe left ventricular (LV) systolic dysfunction (ejection fraction [EF] < 30%) is a known cardiovascular risk factor and a major cause of cardioembolism. However, less severe forms of LV disease (LVD), such as mild-to-moderate LV dysfunction and LV wall motion abnormalities (LVWMAs), are considered potential minor cardiac sources in Embolic Stroke of Undetermined Source (ESUS), but their role is underexplored. This study aims to evaluate the prevalence of LVD in ESUS and its association with adverse vascular events and mortality.
Methods:
Retrospective, single-center study including consecutive ESUS patients admitted from January 2016 to May 2024. LVD was defined as either global systolic dysfunction (LV ejection fraction 30%–49%) or regional LVWMAs, unrelated to acute or recent (within 4 weeks) myocardial infarction. Univariate and multivariate Cox regression analyses evaluated the association of LVD with a primary composite outcome (including ischemic stroke recurrence, acute coronary events, and all-cause mortality), and its components separately.
Results:
Among the 556 ESUS patients (median age 71 years [IQR 60–80], 44.6% female), 95 (17.1%) had LVD, including 51 (53.7%) with reduced LVEF (30%–49%), and 81 (85.3%) presenting LVWMAs. During follow-up (median 30 months), LVD(+) patients had significantly higher rates of the composite outcome (41.0% vs 21.3%,
Conclusions:
LVD is significantly associated with an increased risk of ischemic stroke recurrence and adverse outcomes in ESUS patients. These findings highlight the clinical importance of identifying and optimizing LVD management among ESUS to improve long-term outcomes in this population.
Keywords
Introduction
Left ventricular (LV) systolic dysfunction is a well-documented risk factor for ischemic stroke, systemic embolism, and adverse cardiovascular outcomes.1–4 Patients with acute ischemic stroke and severely reduced ejection fraction (EF < 30%) are generally considered at high-risk for stroke and are included under the “cardioembolic” subtype in most ischemic stroke classifications.5–7 This categorization is based on the pathophysiological theory that low cardiac output, dilated chambers, poor contractility, and endothelial dysfunction in these patients may promote thrombus formation and subsequent embolic events. 8 While severe LV dysfunction has been widely studied, other less severe forms of LV disease (LVD) – such as mildly to moderately reduced EF (30%–49%) or LV wall motion abnormalities (LVWMAs) – may also contribute to embolic events. These conditions are recognized as potential sources of cardio-embolism, and are therefore included in the embolic stroke of undetermined source (ESUS) definiton.9–11 This inclusion is supported by evidence linking mild to moderate LVD to ESUS, compared to other non-cardioembolic stroke etiologies.12–14 Indeed, studies have demonstrated that patients with mild to moderate LV systolic dysfunction face an increased risk of ischemic stroke, and LVWMAs have been associated with a higher risk of ischemic stroke recurrence in the general stroke population.15–17 Despite the well-recognized cardiovascular risk associated with severe LV dysfunction, the broader spectrum of LVD – including mild to moderate impairments and LVWMAs – within the ESUS population remains inadequately explored and warrants investigation. The aim of this study is to shed light on this ESUS subgroup by assessing the prevalence of LVD within the ESUS population and exploring its relationship with adverse clinical outcomes including the risk of ischemic stroke recurrence, acute coronary events, and mortality.
Material and methods
Study design and population
This retrospective, single-center study included all consecutive patients diagnosed with ESUS, who were admitted to the Stroke Unit of Niguarda Hospital in Milan (Italy) between January 2016 and May 2024. Patients were initially classified as ESUS at hospital discharge. A thorough retrospective review of patient records and diagnostic evaluations conducted during admission was subsequently performed to confirm the ESUS classification.
ESUS was defined according to standard criteria
9
as a non-lacunar stroke, in the absence of extracranial or intracranial atherosclerosis causing ⩾ 50% luminal stenosis of the artery supplying the area of ischemia, no major-risk cardioembolic source of embolism, and no other specific cause of stroke identified. As specified in the ESUS criteria, patients with major-risk cardio-embolic sources of embolism (including permanent or paroxysmal atrial fibrillation [AF], sustained atrial flutter, intra-cardiac thrombus, prosthetic cardiac valve, atrial myxoma or other cardiac tumors, mitral stenosis, recent [<4 weeks] myocardial infarction [MI], LV EF less than 30%, valvular vegetations, or infective endocarditis) were excluded. All patients underwent a comprehensive minimum diagnostic assessment according to ESUS criteria.
9
In line with the recent proposed revision to the concept of ESUS,
18
all diagnostic evaluations were reviewed to exclude: (1) patients aged < 60 years with
Data collected comprised baseline demographics (age, sex, prior ischemic stroke history, and stroke severity measured by NIHSS), vascular risk factors (current smoking, arterial hypertension, diabetes, dyslipidemia, obesity), antithrombotic therapy prior to admission and acute reperfusion therapies. Coronary artery disease (CAD) was defined as any previous MI or unstable angina, stable angina, coronary revascularization (percutaneous coronary intervention, coronary artery bypass grafting, and other arterial revascularization procedures), obstructive coronary atherosclerosis (such as significant plaque on coronary angiography or CT scan, multivessel coronary disease with > 50% stenosis in two major epicardial arteries). Functional status was assessed on admission and at 90 days using the modified Rankin Scale (mRS). Outpatient cardiac monitoring, including Holter monitoring (from 24 h to 30 days) and/or an implantable loop recorder (ILR), was performed at the discretion of the treating physician. During follow-up, data on AF detection after stroke (AFDAS) were collected. AFDAS was defined as any AF detected after stroke, occurring in patients without known AF (and not considering AF detected during admission as AFDAS). 19
This study received approval from the local Ethical Committee (Comitato Etico Milano Area 3). Upon admission, patients were informed that all data obtained during routine clinical practice would be used for research purposes, and they provided written informed consent. This study is reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline.
Echocardiographic parameters and LV disease definition
Transthoracic echocardiography (TTE; and additional transesophageal echocardiography [TEE] at the discretion of the treating physician) was performed on each patient during hospitalization. All tests were conducted, and measurements were acquired in accordance to the American Society of Echocardiography guidelines. 20 All data were reviewed by a team of two cardiologists (AB, AM). Measurements extracted from the reports included LA volume index (LAVI), LVEF, LV diastolic dysfunction (LVDD), and LV end-diastolic volume index (LV EDVI). LVDD was defined according to American Society of Echocardiography guidelines 21 using mitral valve inflow pattern with pulsed-wave Doppler, e’ wave at Tissue Doppler of lateral and septal mitral anulus, tricuspid regurgitation velocity and LAVI. LA enlargement (LAE) was defined based on standard criteria as LAVI > 34 ml/m2. LV dilatation was defined based on LV EDVI according to the established guidelines. 20 LVD was defined as global systolic dysfunction (reduced EF 30–49%) and/or regional LVWMAs, which were defined as any hypokinetic, akinetic or dyskinetic segment of the LV wall, that was not associated with acute or recent (i.e. within 4 weeks) MI, based on standard criteria given by the American Society of Echocardiography. 20 The entire cohort was then stratified into two groups based on the presence [LVD(+)] or absence [LVD(−)] of LVD.
Follow-up and outcomes
The primary outcome was a composite outcome that included ischemic stroke recurrence, acute coronary events, and all-cause mortality during follow-up. Each component was also analyzed individually as secondary outcome. Ischemic stroke recurrence was defined as the development of new neurologic symptoms with corresponding lesions on brain imaging after hospital discharge. Acute coronary events included MI, cardiac revascularization, and hospitalization for unstable angina. All-cause mortality was defined as any death from any cause. Participants in the study were followed up until the last available follow-up (death or loss to contact). Data were retrospectively collected from outpatient visits, medical records, or telephone interviews. For patients lost to follow-up, data were censored at the last time known to be alive. Patients who experienced multiple events (such as recurrent ischemic strokes, acute coronary events, and death), were censored at the time of the first event.
Statistical analysis
Baseline characteristics, echocardiographic parameters, and outcomes were summarized using descriptive statistics: categorical data as counts and proportions, while continuous data as medians with IQR ranges. Differences between LVD(+)
Results
Of the 674 initially enrolled ESUS patients, 119 were excluded based on predefined criteria, resulting in a final cohort of 556 patients. Among these, 95 patients (17.1%) had LVD, with 51 (53.7%) showing reduced LVEF 30%–49%, and 81 (85.3%) presenting LVWMAs. The general characteristics of the final ESUS cohort, stratified by the presence of LVD, are detailed in Table 1. Median age was 71 years (IQR 61–80), female 44.6%, baseline NIHSS 5 (IQR 2–11). LVD(+) patients were less likely to be women compared to LVD(−) (22.1% vs 49.2%,
Clinical characteristics of ESUS patients, stratified by the presence of LVD.
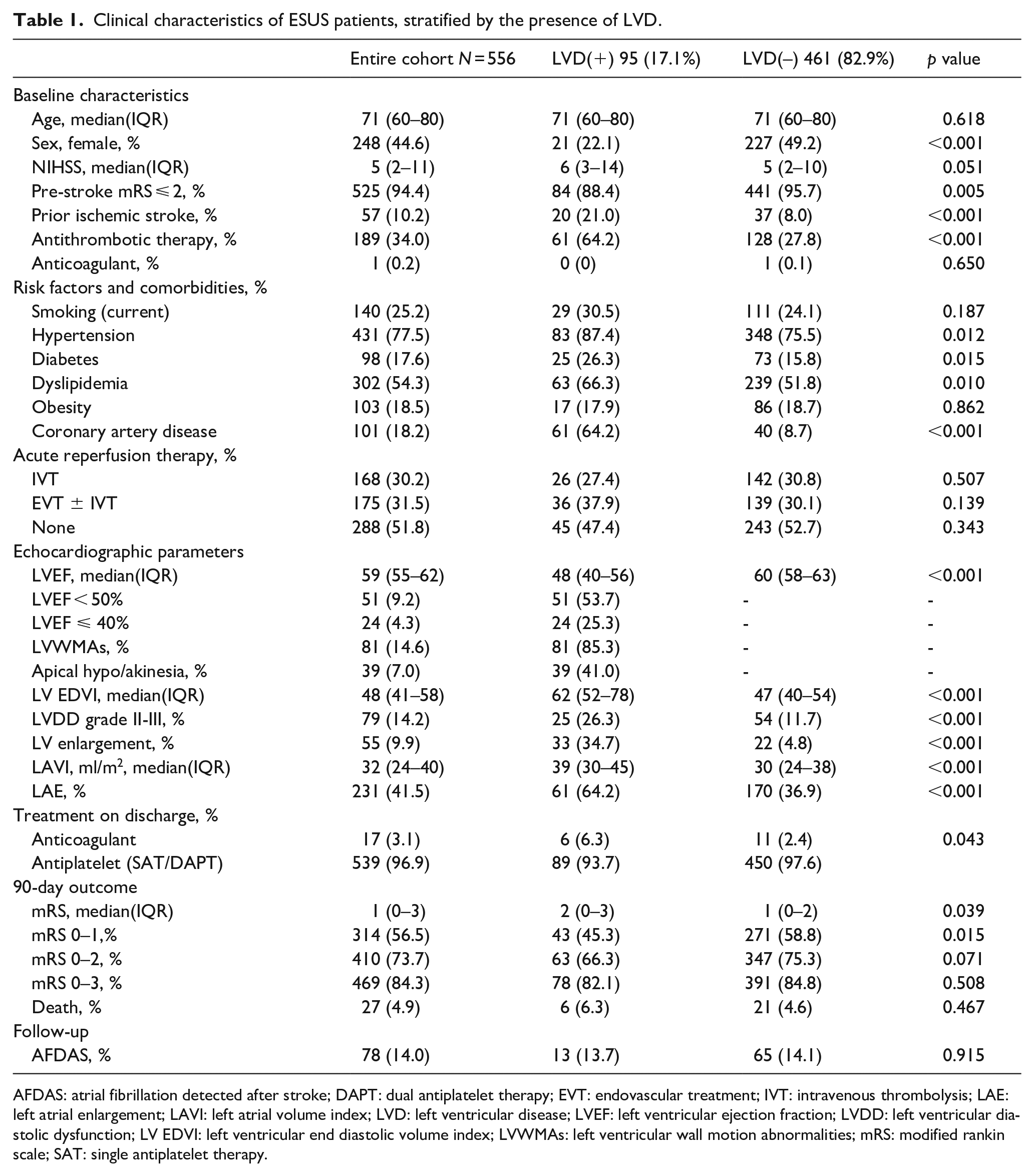
AFDAS: atrial fibrillation detected after stroke; DAPT: dual antiplatelet therapy; EVT: endovascular treatment; IVT: intravenous thrombolysis; LAE: left atrial enlargement; LAVI: left atrial volume index; LVD: left ventricular disease; LVEF: left ventricular ejection fraction; LVDD: left ventricular diastolic dysfunction; LV EDVI: left ventricular end diastolic volume index; LVWMAs: left ventricular wall motion abnormalities; mRS: modified rankin scale; SAT: single antiplatelet therapy.
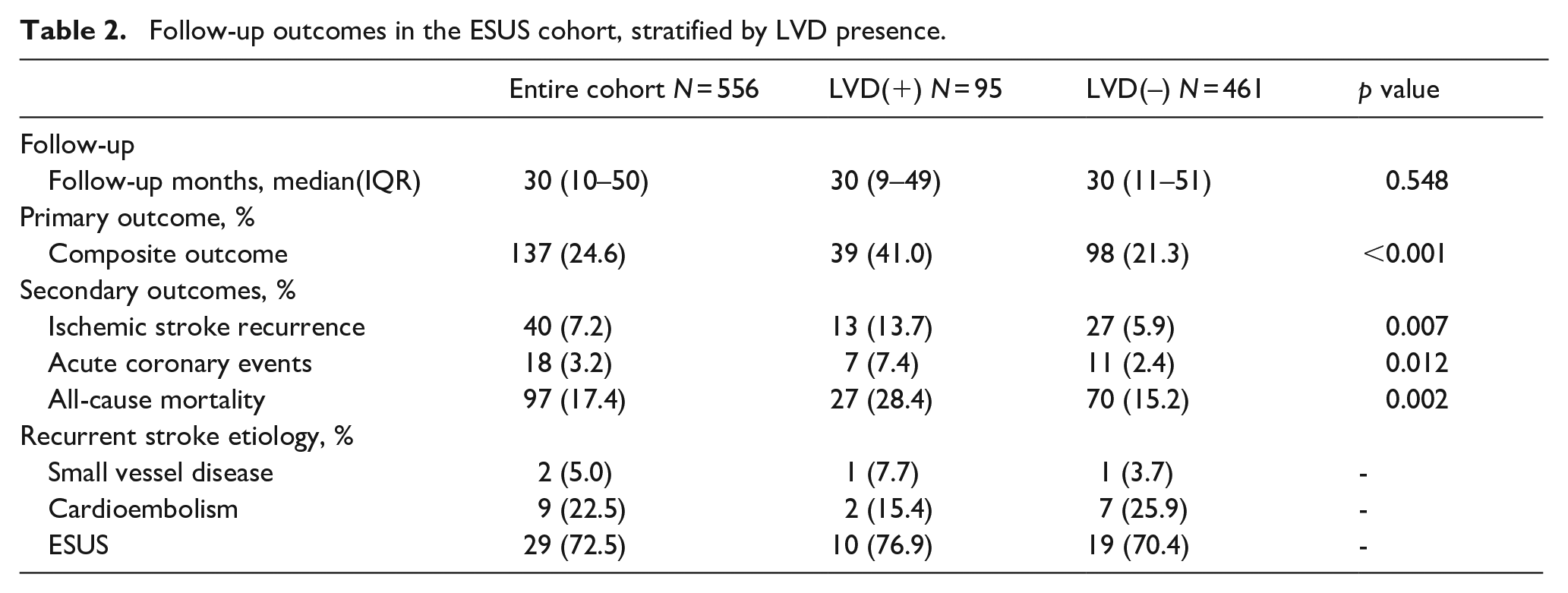
Follow-up data were obtained for all patients at least 3 months from the ischemic stroke. Over a median of 30 months (IQR 10–50), the composite outcome occurred in 137 patients (24.6%): 40 patients (7.2%) had ischemic stroke recurrence, 18 (3.2%) an acute coronary event, and 97 (17.4%) died. LVD(+) patients had significantly higher rates of the composite outcome (41.0% vs 21.3%,
Follow-up outcomes in the ESUS cohort, stratified by LVD presence.
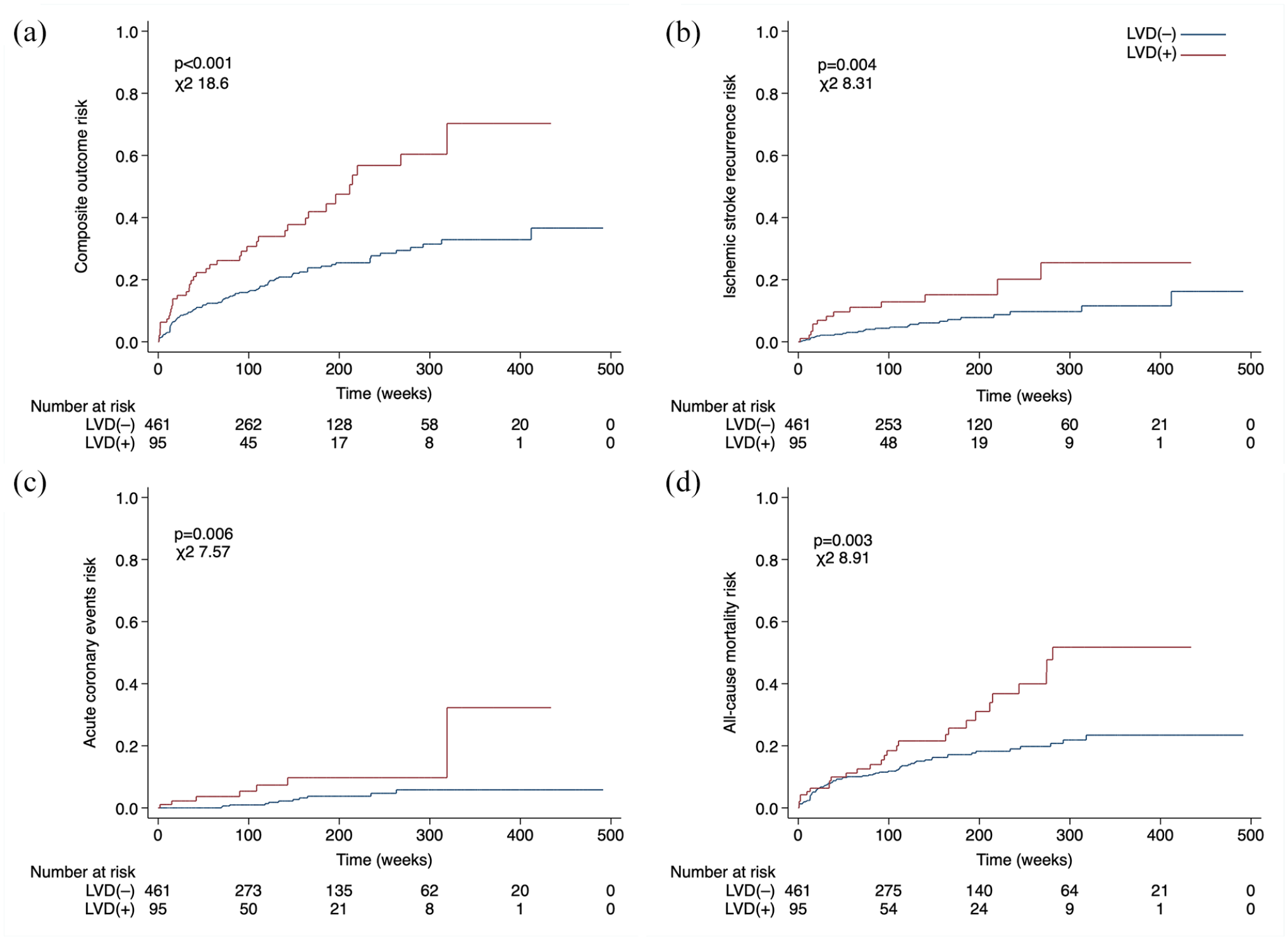
Kaplan-Meier survival curves illustrate the effect of LVD on the risk of the (a) composite outcome, (b) ischemic stroke recurrence, (c) acute coronary events, and (d) all-cause mortality.
Univariate and multivariate Cox regression analyses to evaluate the association between LVD and the risk of the composite outcome, as well as each individual component (ischemic stroke recurrence, acute coronary events, and all-cause mortality).
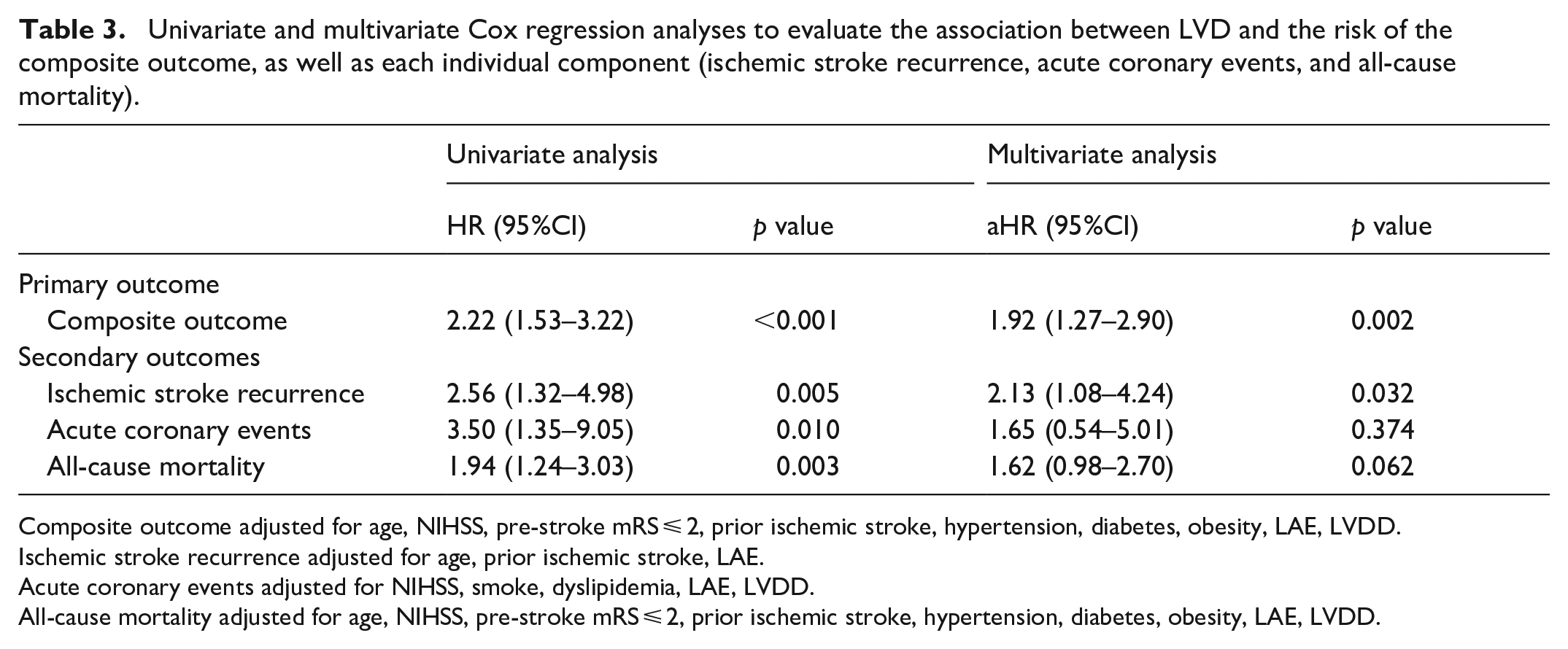
Composite outcome adjusted for age, NIHSS, pre-stroke mRS ⩽ 2, prior ischemic stroke, hypertension, diabetes, obesity, LAE, LVDD.
Ischemic stroke recurrence adjusted for age, prior ischemic stroke, LAE.
Acute coronary events adjusted for NIHSS, smoke, dyslipidemia, LAE, LVDD.
All-cause mortality adjusted for age, NIHSS, pre-stroke mRS ⩽ 2, prior ischemic stroke, hypertension, diabetes, obesity, LAE, LVDD.
Other covariates associated with the outcome of interest (
Discussion
In this single-center, retrospective study, we found that LVD represents a significant ESUS subtype associated with an elevated risk of adverse events (including ischemic stroke recurrence, acute coronary events, and all-cause mortality). In particular LVD was associated with a twofold higher risk of ischemic stroke recurrence compared to the rest of the ESUS population, highlighting its critical role in increasing stroke risk among ESUS patients.
LVD is recognized as a potential source of embolism in ESUS population, 11 with prevalence rates ranging from 4% to over 50%, depending on the definitions used across studies.10,22 In our cohort, 17.1% of patients had LVD, a rate consistent with studies that employed similar criteria23,24 and demonstrated a significant association between LVD and increased stroke risk in the general population.12,17 Although LVD constitutes a significant ESUS subgroup, current research on the role of LVD in ESUS population is scarce compared to more widely studied ESUS sources like silent AF. This research gap is particularly notable given our findings that ESUS patients with LVD represent a vulnerable population at increased risk of vascular events and mortality.
Our study revealed a significant independent association between LVD and the composite outcome of ischemic stroke recurrence, acute coronary events, and death, reflecting the elevated clinical risk of this ESUS subgroup. Approximately 41% of patients with LVD experienced an adverse event during follow-up, emphasizing the multifactorial cerebrovascular and cardiovascular risk these patients face, highlighting the need for tailored management strategies to address their complex clinical profile. Specifically, LVD emerged as a significant independent risk factor for ischemic stroke recurrence with a twofold increased risk compared to the rest of ESUS population, and an incidence rate of 13.7% over 30 months. Furthermore, approximately 21% of LVD(+) patients had a prior history of ischemic stroke. Paroxysmal thrombus formation within the LV may be the key mechanism driving stroke recurrence in these patients. Studies indicate that even mild LV dysfunction or the presence of WMAs 25 can increase the risk of cardiac thrombus formation. 26 Kaplan-Meier analysis shows a steeper initial slope in the survival curve for the LVD(+) group compared to LVD(–), indicating an higher risk of early stroke recurrence shortly after the index stroke. Moreover, most stroke recurrences continued to be classified as ESUS, suggesting a potentially similar underlying mechanism for these recurrences, such as paroxysmal LV thrombus formation. In our study, we found that AFDAS did not significantly differ between patients with and without LVD, reinforcing the hypothesis that stroke recurrence in these patients may primarily related to LVD itself, independently of AFDAS.
Previous randomized trials27,28 have failed to demonstrate that Vitamin K antagonists (VKAs) are superior to antiplatelet therapy for preventing ischemic strokes in patients with severe LVD (EF⩽35%) and sinus rhythm, due to a higher bleeding risk. However, a recent subgroup analysis of the NAVIGATE-ESUS study suggested that anticoagulation with direct oral anticoagulants (DOACs) may be more effective than aspirin in reducing the risk of recurrent stroke 24 in patients with LV dysfunction. Additionally, some evidences come from trials that used a combination of aspirin and low-dose DOACs in patients with heart failure and sinus rhythm.29,30 To date anticoagulation is not routinely prescribed in these patients: current guidelines recommend anticoagulation only in the presence of LV thrombus (class I, Level B), while individualized decisions are advised in patients with reduced EF < 50%, without evidence of LV thrombus (class IIb, Level C).31,32 No ongoing studies are evaluating anticoagulant therapy for secondary prevention in ESUS patients with LVD. Given the insufficient evidence regarding anticoagulation and considering the potential thrombogenic risk of LVD, a rational approach to better manage these patients, would be to enhance thrombus detection by implementing more advanced cardiac imaging. Standard TTE, routinely used, has low sensitivity for detecting LV thrombi, compared to superior cardiac imaging modalities such as c-TTE, cardiac CT or MRI. Studies have shown that cardiac CT or MRI significantly improves the detection of intra-cardiac thrombi in ESUS patients, especially those with global or regional LV dysfunction.25,33 Despite this, advanced cardiac imaging is not routinely incorporated into the ESUS diagnostic work-up in the absence of clear guidelines recommendations.31,32
In our study, LVD was primarily attributed to CAD in approximately 60% of cases. Given their underlying chronic ischemic heart disease, these patients were expected to have an elevated risk of recurrent coronary events. While our study observed a higher incidence of acute coronary events in the LVD(+) group, this association did not reach statistical significance in the multivariate analysis. These results are likely due to the relatively small number of events recorded, which may have limited the study’s power to detect a meaningful difference.
We observed a significant higher mortality among LVD(+) patients during follow-up, underscoring the increased vulnerability of this population, which faces an elevated vascular risk. However, LVD was not independently associated with the risk of death. This likely reflects the multifactorial nature of “all-cause mortality” definition, which includes a wide range of potential causes of death, and is influenced by the complex interplay of various contributing factors.
Our study has several limitations that should be acknowledged. As retrospective, single-center study, the generalizability of our findings may be limited. Furthermore, the underdiagnosis of small ventricular thrombi in patients with LVD remains possible, as advance cardiac imaging was not routinely used, potentially introducing some selection bias. In fact, this may have led to some patients being misclassified as ESUS rather than cardioembolic. Nevertheless, our study has notable strengths. To the best of our knowledge, it is the largest study evaluating the role of LVD in a large and well-selected ESUS cohort, with an extended follow-up period; this allowed us to assess the long-term prognosis of this ESUS subgroup, providing valuable insights into this specific stroke population. As in a previous study, 34 we adopted the recently proposed ESUS construct to have a highly selected cohort, excluding patients with probable alternative stroke etiologies, such as PFO-associated stroke, high-risk non stenotic plaques, and cancer. While the traditional ESUS concept has been pivotal in guiding clinical research and practice, advancements in stroke research and the failure of large therapeutic trials have highlighted the need to revise its original definition, allowing to reduce heterogeneity and minimizing confounding factors. Future studies using these refined criteria are likely to produce more accurate and clinically relevant findings, addressing limitations of earlier research and providing better insights into the pathophysiology and treatment of ESUS.
Conclusion
Our study finds that ESUS patients with LVD represent a critical subgroup at elevated risk for adverse outcomes including ischemic stroke recurrence, acute coronary events, and death. Notably, LVD is independently associated with a significantly higher risk of ischemic stroke recurrence compared to the overall ESUS population. These findings suggest a potential gap in optimal secondary stroke prevention. Further large-scale studies and randomized trials are needed to evaluate alternative antithrombotic strategies, incorporate the routine use of advanced cardiac imaging, and implement a comprehensive therapeutic approach to improve outcomes in this high-risk stroke population.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241311331 – Supplemental material for Left ventricular disease as a risk factor for adverse outcomes and stroke recurrence in patients with embolic stroke of undetermined source
Supplemental material, sj-docx-1-eso-10.1177_23969873241311331 for Left ventricular disease as a risk factor for adverse outcomes and stroke recurrence in patients with embolic stroke of undetermined source by Angelo Cascio Rizzo, Ghil Schwarz, Andrea Bonelli, Andrea Magi, Elio Clemente Agostoni, Antonella Moreo and Maria Sessa in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study received approval from the local Ethical Committee (Comitato Etico Milano Area 3).
Informed consent
All patients were informed that all data acquired during routine clinical practice could be used for research purposes.
Guarantor
Angelo Cascio Rizzo
Contributorship
ACR primarily conceived and designed the work, analyzed data and drafted a substantial portion of the manuscript. GS, ECA, AM, and MS contributed significantly to the study’s conception and design. ACR, GS, AB, AMa acquired the data. All authors revised the manuscript for intellectual content and approved the final version.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
