Abstract
Background:
Minor ischemic stroke, defined as National Institute of Health Stroke Scale score of 0–5 on admission, represents half of all acute ischemic strokes. The role of intravenous alteplase (IVA) among patients with minor stroke is inconclusive; therefore, we evaluated clinical outcomes of these patients treated with or without IVA.
Materials and methods:
We searched Medline, Embase, Scopus, and the Cochrane library until August 1, 2023. Inclusion was restricted to the English literature of studies that reported on minor nondisabling stroke patients treated with or without IVA. Odds ratios (ORs) with their corresponding 95% CIs were utilized using a random-effects model. Efficacy outcomes included rates of excellent (modified Rankin scale [mRS] of 0–1) and good (mRS of 0–2) functional outcome at 90 days. The main safety outcome was symptomatic intracerebral hemorrhage (sICH).
Results:
Five eligible studies, two RCTs and three observational studies, comprising 2764 patients (31.8% female) met inclusion criteria. IVA was administered to 1559 (56.4%) patients. Pooled analysis of the two RCTs revealed no difference between the two groups in terms of 90-days excellent functional outcomes (OR 0.76 [95% CI, 0.51–1.13]; I2 = 0%) and sICH rates (OR 3.76 [95% CI, 0.61–23.20]). No significant differences were observed between the groups in terms of good functional outcomes, 90-day mortality, and 90-day stroke recurrence.
Conclusion:
This meta-analysis of minor nondisabling stroke suggests that IVA did not prove more beneficial compared to no-IVA.

Keywords
Introduction
Minor stroke, defined as a National Institutes of Health Stroke Scale (NIHSS) score of 0–5, accounts for half of all acute ischemic stroke (AIS) cases. 1 It is considered as the most common reason for withholding reperfusion treatment to stroke patients presenting within the eligible treatment window. 2 However, this can be misleading as the prognosis of this group may not be benign. 3 Studies have shown that up to one-third of stroke patients who were considered too mild for thrombolysis were deceased or disabled at 90-day follow-up.4,5 Moreover, these patients might be at risk of stroke progression, and therefore treatment with intravenous alteplase (IVA) may offer a potential benefit. 6
Recent guidelines recommend treatment with IVA for minor disabling stroke and advocate against offering reperfusion therapy in the absence of disability.7,8 Yet, the recommendation in nondisabling stroke was primarily based on expert consensus and data from The Effect of Alteplase versus Aspirin on Functional Outcome for Patients with Acute Ischemic Stroke and Minor Nondisabling Neurologic Deficits (PRISMS) trial, from which its premature termination precluded a clear conclusion. 6 Recently, the Dual Antiplatelet Therapy versus Alteplase for Patients with Minor Nondisabling Acute Ischemic Stroke (ARAMIS) study suggested a non-inferiority of dual antiplatelet therapy (DAPT) compared to IVA in term of excellent functional outcomes at 90 days among patients with minor nondisabling stroke. 9
Evidence of IVA benefit in minor stroke without a clear disability is inconclusive, and the precise characterization of nondisabling deficits remains elusive.10–12 Hence, we aimed to investigate the benefits and risks of IVA in minor nondisabling stroke through a systematic review and meta-analysis.
Methods
This review was reported in compliance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 13 and the Meta-analysis of Observational Studies in Epidemiology (MOOSE) reporting guidelines. 14 This review was conducted according to a prespecified protocol registered with PROSPERO (CRD42023449038). As this work constitutes a systematic review and aggregate data meta-analysis of previously published studies, neither patient consent nor ethical approval was required. The data will be made available upon a reasonable request.
Search method and resources
We searched Medline, Embase, Scopus, and the Cochrane Library until August 1, 2023. To avoid missing any relevant study, we performed a manual search of the grey literature and reference list of included studies. The search algorithm included different combinations of possible keywords customized to each database. A detailed search strategy is provided in Appendix in the Supplement.
Study selection and outcomes of interest
Two authors executed an initial title and abstract screening, which was followed by a full-text screening stage. Inclusion was restricted to randomized clinical trials (RCTs) or observational studies reporting safety and efficacy outcomes following treatment with IVA in patients with minor nondisabling stroke as defined by the study’s definition. Definition of a minor nondisabling stroke was predefined at study start as NIHSS score 0–5 upon patients’ initial presentation.6,9–12 Since there is no consensus regarding a standardized definition for nondisabling deficit, we restricted our inclusion criteria to studies reporting sufficient characterization of nondisabling minor stroke. This entails the inclusion of comprehensive criteria that specifically delineate the items from the NIHSS score and clinical assessments that were considered in defining nondisabling deficits and therefore patient selection. The primary efficacy outcome was the excellent functional outcome (defined as mRS 0–1 at 90 days) based on RCT data. The main safety outcome was the rate of sICH based on RCT data. Secondary outcomes included excellent functional outcome, good functional outcome (defined as mRS 0–2 at 90 days), sICH, mortality at 90 days, and recurrent stroke at 90 days based on data from both RCTs and observational studies.
Data extraction and quality assessment
Two independent authors (B.A.A. and M.A.) extracted data about the study and patients’ characteristics from each eligible study. Two internal reviewers (A.F.A. and H.S.) assessed the quality of included studies using the Cochrane’s Risk of Bias 2 (RoB2) 15 and the Risk of Bias in Non-Randomized Studies of Interventions (ROBINS-I) 16 tools for randomized and non-randomized studies, respectively. Conflicts were resolved via consensus or consultation with a third author (A.A.).
Statistical analysis
We analyzed the data using RevMan software, and statistical significance was set at p < 0.05. Data were pooled using a random-effects model, and a forest plot was established for each outcome. We computed odd ratios (ORs) and their corresponding 95% confidence intervals (CIs) for dichotomous variables. Additionally, we computed 95% prediction intervals (PIs) using the Comprehensive Meta-Analysis software and reported them alongside the corresponding 95% CIs for syntheses with three or more contributing studies. For contextual relevance, we stratified the studies in each synthesis based on their design, distinguishing between RCTs and observational studies. For RCTs, we sought measures derived from the intention-to-treat (ITT) model. In cases where observational studies did not provide adjusted data in the form of event/total or reported it using alternative formats, such as adjusted OR or risk ratios, we made the decision to pool unadjusted crude data. Currently, there is no agreement on how estimates from adjusted and unadjusted analyses from non-randomized studies can be combined in a way that does not compromise the sample size or introduce bias to the estimate. 17 Consequently, we conducted a sensitivity analysis of the excellent functional outcome by excluding observational studies that did not provide adjusted data. For publication bias, assessment methods such as Egger et al.’s test and meta-regression were not feasible as we examined fewer than 10 publications.18,19 Cochrane’s Q test and the Higgins index (I2) were used to measure heterogeneity, where p < 0.05 or I2 values greater than 50% were regarded as significant. Additionally, we performed subgroup analyses of the primary outcome among different NIHSS score (0–3 and 4–5), age (using 65 as a cutoff), and onset to treatment (OTT) before or after 2 h.
Results
Search results
Of 2149 citations identified, we excluded 378 as duplicates. 1765 reports were then excluded by screening of titles and abstracts. We retrieved 15 potentially eligible studies for full-text screening; of those, 5 eligible studies6,9–12 satisfied our inclusion criteria (Figure S1 in Supplement).
Study characteristics and quality of evidence
A summary of the included studies is shown in Table 1. We included two RCTs6,9, three observational studies,10–12 with a total of 2764 patients (31.8% female). There were 1559 patients who received IVA, while 1205 patients were in the control group. The treatment regimen in the control groups of included studies included aspirin, 6 DAPT, 9 or guideline-based treatment of aspirin, DAPT or anticoagulants.11,12 One study 10 did not report the treatment regimen in the control arm. Patient selection criteria and outcome definitions (including minor nondisabling stroke and sICH) across studies are detailed in Table S1 in the Supplement. Baseline NIHSS scores were comparable between the two arms within included RCTs, whereas the IVA group had higher baseline NIHSS scores compared to control within the observational studies (Table 1).
Overview of included studies.
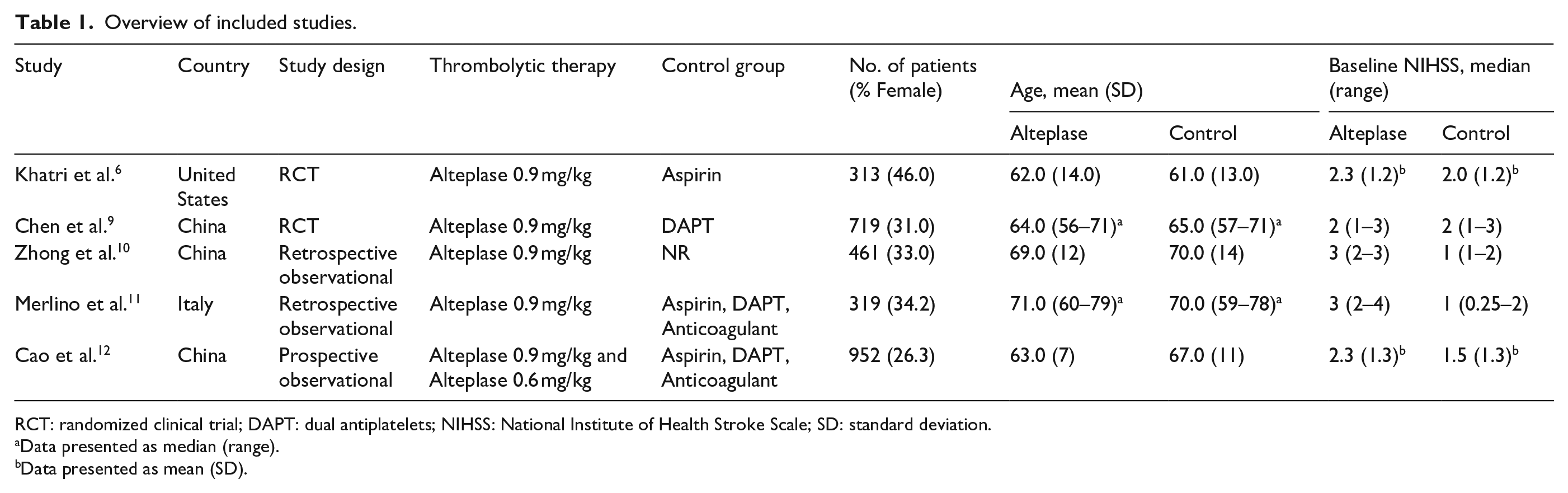
RCT: randomized clinical trial; DAPT: dual antiplatelets; NIHSS: National Institute of Health Stroke Scale; SD: standard deviation.
Data presented as median (range).
Data presented as mean (SD).
Details on the risk of bias assessments of included studies are provided in the Supplement. In short, the two included RCTs6,9 were judged to have a low risk of bias across all domains (Table S2). However, the observational studies varied in terms of quality. Cao et al. 12 study showed moderate overall risk of bias, while Merlinoet al. 10 and Zhong et al. 11 had serious overall risk of bias. It is crucial to recognize that non-randomized studies, as observed in most of our assessments, are unlikely to exhibit low risk of bias in the pre- and at-intervention domains. Detailed risk of bias assessment is provided in the supplement. (Table S3).
Excellent functional outcome (mRS 0-1 at 90 days)
All included studies6,9–12 reported on excellent functional outcome at 90 days. Pooled analysis of the two RCTs6,9 revealed no difference between the two groups in terms of excellent functional outcomes at 90 days (OR 0.76 [95% CI, 0.51–1.13]; p = 0.17) with no between-study heterogeneity (I2 = 0%; p = 0.73). Across both the RCTs and observational studies, IVA was associated with lower odds of excellent functional outcome compared to control (84.3% vs 87.9%; OR 0.75 [95% CI, 0.59–0.96; 95% PI, 0.50–1.12]; p = 0.02), with no evidence of heterogeneity (I2 = 0%; p = 0.98). (Figure 1(a)) Results from sensitivity analyses showed similar trends after excluding two observational studies10,11 that only reported crude unadjusted data. (Figure S2) Results of prespecified subgroup analyses suggest no significant difference between IVA and medical management in patients with NIHSS of 0–3 (OR 0.74 [95% CI, 0.45–1.20]; p = 0.22) or NIHSS of 4–5 (OR 0.97 [95% CI, 0.45–2.11]; p = 0.94). (Figure S3A) Similarly, no significant difference was observed between the two arms when stratified into different age or OTT strata (Figure S3B, S3C).
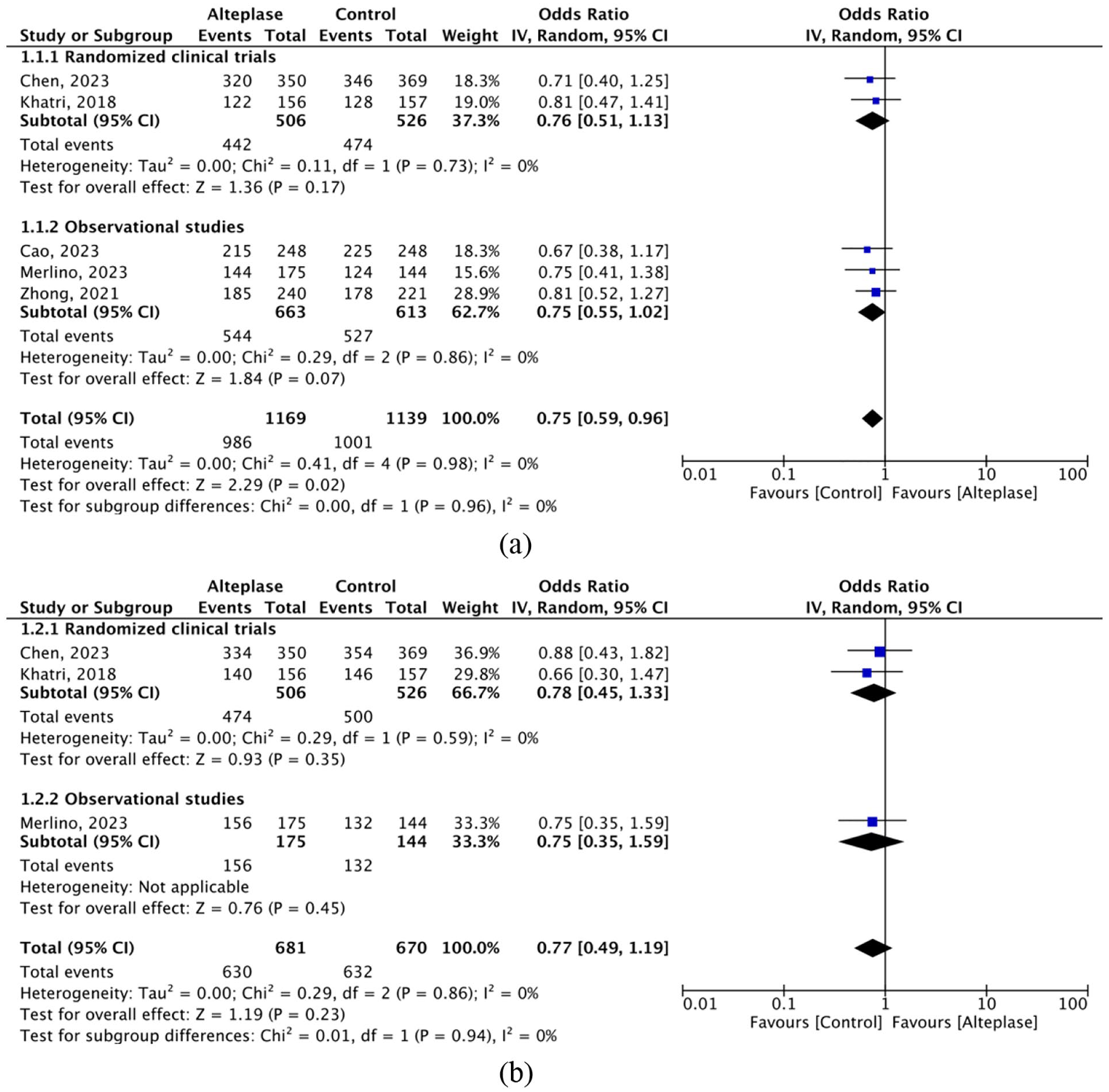
Forest plots of (a) excellent functional outcome (mRS 0–1) at 90 days and (b) and good functional outcome (mRS 0–2) at 90 days.
Good functional outcome (mRS 0-2 at 90 days)
Three studies6,9,11 reported on good functional outcome. Pooled analysis of the two RCTs6,9 revealed no difference between the two groups in terms of good functional outcomes at 90 days (OR 0.78 [95% CI, 0.45–1.33]; p = 0.35) with no between-study heterogeneity (I2 = 0%; p = 0.59). Similarly, rates of good functional outcome across both the RCTs and observational studies were comparable between the two groups (OR 0.77 [95% CI, 0.49–1.19; 95% PI, 0.05–12.95]; p = 0.23), with no heterogeneity (I2 = 0%; p = 0.86) (Figure 1(b)).
Safety outcomes
Data from two RCTs6,9 showed no significant difference between groups in the odds of sICH (OR 3.76 [95% CI, 0.61–23.20]; p = 0.15). A meta-analysis of all five studies6,9–12 showed that IVA was associated with significantly higher odds of sICH compared to control (1.6% vs 0.3%; OR 4.10 [95% CI, 1.37–12.25; 95% PI, 0.69–24.24]), with no evidence of heterogeneity (I2 = 0%; p = 0.91). (Figure 2(a)) In the same context, four studies6,9,11,12 reported mortality data at 90-days. Mortality rates were similar between the two groups (OR 0.77 [95% CI, 0.34–1.72; 95% PI, 0.21–2.84]; p = 0.53), with no evidence of heterogeneity (I2 = 0%; p = 0.62) (Figure 2(b)). For stroke recurrence within 90 days, two RCTs6,9 reported relevant data. Rates of stroke recurrence were comparable between the two groups (OR 0.98 [95% CI, 0.33–2.90]; p = 0.98), with no heterogeneity (I2 = 0%; p = 0.55) (Figure 2(c)).
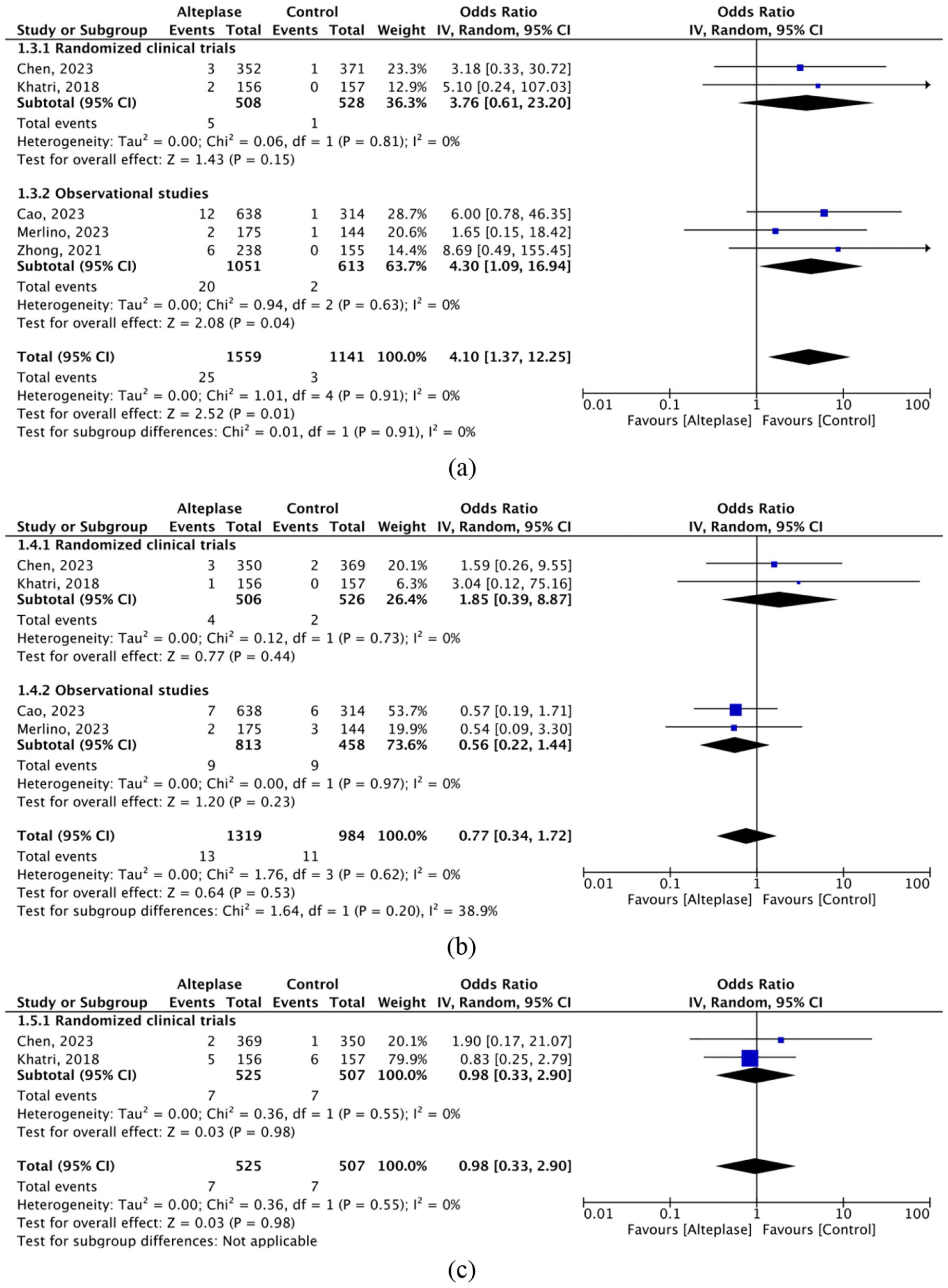
Forest plots for (a) sICH, (b) mortality, and (c) stroke recurrence.
Discussion
This systematic review and meta-analysis indicates that among patients with minor nondisabling stroke, use of IVA was not associated with added benefits in terms of excellent functional outcome. Although not statistically significant, there was a tendency toward higher rates of sICH in individuals who received IVA within RCTs. Similar rates of good functional outcome, mortality, and stroke recurrence were noted in both treated and untreated groups.
Vessel reperfusion with IVA within 4.5 h can reduce disability and therefore has been the standard of care for AIS management.20,21 Yet, evidence of IVA benefit in ischemic stroke with minor deficit is scarce6,22 as landmark alteplase trials in AIS excluded individuals with low NIHSS scores.20,21,23–25 The current body of evidence conveys conflicting results regarding the efficacy of alteplase in minor stroke.26–30 These inconsistencies could be attributed to the heterogeneous clinical presentation of the minor stroke population (disabling vs nondisabling). Moreover, appropriate characterization and definitions of nondisabling stroke have been a matter of debate with a lack of a universal approach. 12 In this meta-analysis, combined findings from RCTs and observational studies suggest that patients with minor nondisabling stroke who did not receive IVA were more likely to achieve excellent outcomes (87.9% vs 84.3%) and good functional outcome (94.3% vs 92.5%) compared to patients treated with IVA. Yet, this effect was primarily derived from mixed quality observational studies. In terms of RCT data, 90-days excellent functional outcome were similar between IVA and control. This aligns with the results of two previous RCTs indicating trends of lack of benefit in patients with nondisabling stroke treated with IVA.6,9 Although differences in baseline NIHSS scores, vascular risk factors, and treatment approach within the control groups between included studies might contribute to different rates of favorable long-term outcomes, all included studies showed similar trends.6,9–12 These results underscore the limited benefit of IVA in nondisabling stroke and further justify the use of DAPT as current best practice in these patients.
When considering reperfusion therapy for minor ischemic stroke, differentiating disabling from nondisabling deficits is imperative. 12 In the PRISMS trial, disabling deficit was defined as a deficit that, if unchanged, would impede basic activities of daily living (ADL). 6 The ARAMIS trial characterized disabling deficits based on elements of NIHSS scores and in consultation with the patient or their available family member. 9 Elements such as moderate-to-severe leg/arm weakness, total aphasia, and visual field cuts might be considered clearly disabling with NIHSS scores of less than 5. 6 However, the NIHSS score may not capture all disabling deficits, as immeasurable deficits do not necessarily represent a nondisabling presentation. Cognitive dysfunction, truncal ataxia, and depression can largely affect ADLs, yet would be overlooked by the presenting NIHSS score. 12 Therefore, when evaluating the level of disability in patients with minor ischemic stroke, it is crucial to consider a combination of factors including item-based assessment of the NIHSS score, impact on ADL, and professional judgment of treating practitioners in conjunction with the patient or their family regarding what constitutes a disabling deficit to the patient.
Data are limited concerning the role of IVA in nondisabling stroke characterized by high NIHSS scores (4–5). 9 In the current study, our pooled data from the PRISMS and ARAMIS trials revealed no additional benefits of IVA among patients with nondisabling stroke and NIHSS scores ranging from 4 to 5. This observation, however, is constrained by a relatively small sample size and a broad confidence interval, thus necessitating additional research.
The efficacy of IVA in minor stroke with different stroke etiologies and in the presence of cerebral vessel occlusion remains unclear.31,32 Among included studies in this analysis, we found a substantial difference in the distribution of stroke etiology. This could be explained by differences in patient selection, vascular risk factors, and ethnicity. Furthermore, results from a large prospective registry showed similar outcomes following reperfusion therapies in disabling and nondisabling stroke (characterized by PRISMS criteria) 6 in the presence of cerebral vessel occlusion. 32 Individuals with large or medium vessel occlusion are susceptible to an increased risk of neurological deterioration and the use of IVA in this subgroup might be justified.32–37 Although intravenous thrombolysis may benefit patients with minor stroke and large vessel occlusion, 38 it is important to note that the available data are limited in terms of differentiating between disabling and nondisabling deficits. Lack of sufficient data precludes a quantitative meta-analysis in this regard. Therefore, we highlight the need for high-quality studies addressing reperfusion strategies in patients presenting with nondisabling deficits with large or medium vessel occlusion.
One major disadvantage of IVA treatment is the risk of hemorrhagic complications. Based on RCT data, we found a non-significant trends of higher sICH rates among patients treated with IVA compared to control. In patients with nondisabling deficit, sICH can be an unforgiving complication. Rates of sICH for both RCTs and observational studies following IVA in this meta-analysis (1.6%) were comparable to that from a previous meta-analysis reporting a higher rate of sICH in minor stroke patients treated with IVA (1.9%). 39 This supports the existing notion of possible additional harm with IVA compared to medical management in nondisabling deficit, which was expected given the known interaction between alteplase and risk of hemorrhage.6,9 In addition, it is important to highlight that the overall outcome, which encompasses both the potential benefits and any deleterious effects such as sICH, is what determines the net benefit of alteplase. Other safety parameters including rates of mortality and stroke recurrence were comparable between the two groups.
In this study, to our knowledge, we present the first meta-analysis examining the use of IVA in cases of minor nondisabling stroke. Treatment with alteplase in this patient population did not demonstrate beneficial effects and was associated with potential increase in the risk of sICH. Importantly, our synthesis demonstrates a lack of heterogeneity across the two RCTs, highlighting the generalizability of these findings across various definitions of nondisabling stroke. Moreover, the incorporation of real-world data provides further support.
Despite the strength and the clinical implication of our meta-analysis, some limitations need to be acknowledged. First, we did not incorporate an individual participant meta-analysis, which could potentially offer a more nuanced examination and would have allowed us to explore potential interactions between variables. Moreover, the pooling of aggregate adjusted data is constrained by the existence of variations in the adjustment factors among the studies included in the analysis. Second, the inclusion of only five studies may not be representative of the entire population of patients with minor non-disabling stroke, and the small sample size may limit the power to detect meaningful differences between groups. Third, varying definitions across the studies of important parameters, such as what constitutes nondisabling deficits and sICH, could have affected the interpretation of our results. Fourth, comparator arms among included studies varied or were not specified 10 which limited performing subgroup analysis. Fifth, a proportion of patients in Cao et al. 12 analysis were begun on a low dose alteplase 0.6 mg/kg. Yet, this treatment strategy was consistent with their local guidelines, and it was supported by previous studies that demonstrated no difference between the standard 0.9 mg/kg dose and low dose alteplase in Chinese individuals. Lastly, it is worth noting that while the inclusion of observational studies in our analysis may have enriched the sample size and provided additional supportive data, the mixed quality of these studies could impact the rigor of our findings.
Conclusion
In conclusion, our meta-analysis of minor nondisabling stroke literature suggests that IVA did not prove more beneficial to no-IVA. These findings support the current practice guidelines that recommend dual antiplatelet therapy, rather than thrombolysis, as the standard treatment for patients with minor nondisabling stroke presenting within 4.5 h. Further well-designed studies are needed to evaluate precision medicine strategies and to identify clinical or imaging characteristics that could help select appropriate treatments tailored to individual patients.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241237312 – Supplemental material for Intravenous alteplase in minor nondisabling ischemic stroke: A systematic review and meta-analysis
Supplemental material, sj-docx-1-eso-10.1177_23969873241237312 for Intravenous alteplase in minor nondisabling ischemic stroke: A systematic review and meta-analysis by Adel Alhazzani, Fahad S Al-Ajlan, Ahmed Alkhiri, Ahmed A Almaghrabi, Aser F Alamri, Basil A Alghamdi, Hassan K Salamatullah, Abdullah R Alharbi, Maher B Almutairi, Hui-Sheng Chen, Yongjun Wang, Mohamad Abdalkader, Guillaume Turc, Pooja Khatri and Thanh N Nguyen in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873241237312 – Supplemental material for Intravenous alteplase in minor nondisabling ischemic stroke: A systematic review and meta-analysis
Supplemental material, sj-docx-2-eso-10.1177_23969873241237312 for Intravenous alteplase in minor nondisabling ischemic stroke: A systematic review and meta-analysis by Adel Alhazzani, Fahad S Al-Ajlan, Ahmed Alkhiri, Ahmed A Almaghrabi, Aser F Alamri, Basil A Alghamdi, Hassan K Salamatullah, Abdullah R Alharbi, Maher B Almutairi, Hui-Sheng Chen, Yongjun Wang, Mohamad Abdalkader, Guillaume Turc, Pooja Khatri and Thanh N Nguyen in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Guarantor
Ahmed Alkhiri (AA).
Contributorship
AA, FSA, AA, TNN, PK, GT, MA, YW, and HC contributed to the conception and design of the study; AA, FSA, AA, TNN, PK, AAA, AFA, BAA, HKS, ARA, and MBA contributed to the acquisition and analysis of data; AA, FSA, AA, TNN, PK, AAA, AFA, BAA, HKS, ARA, and MBA contributed to drafting the text or preparing the figures.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
