Abstract
Introduction:
The efficacy of different antiplatelet treatment in minor strokes (MSs) or transient ischemic attacks (TIAs) and that of antiplatelet and intravenous thrombolysis (IVT) in MSs remain controversial.
Methods:
We searched PubMed, Embase, Web of Science and the Cochrane Library to identify all eligible articles until April 12, 2024. Efficacy outcomes were all-cause mortality, excellent outcome, functional independence and recurrent stroke. Safety outcomes were any types of bleeding and intracerebral hemorrhage (ICH). The associations were calculated for the overall data by using odds ratios (ORs).
Results:
Twenty three high-quality studies with 10 RCTs and 13 non-RCTs were included, involving 47,135 patients with MSs or TIAs. In MSs or TIAs, dual antiplatelet therapies (DAPTs) significantly improved the modified Rankin Scale (mRS) scores for patients with recurrent stroke, major vascular events and ischemic stroke although it was associated with an increased risk of ICH and bleeding when compared to aspirin. In MSs, compared to IVT, DAPT had a significant advantage in improving the mRMS scores and SAPT and DAPT significantly reduced the risk of any bleeding or sICH. IVT significantly reduced all-cause mortality, although it also increased the risk of sICH and ICH compared to no IVT.
Conclusions:
In MSs or TIAs, compared to aspirin, DAPTs can effectively prevent the recurrence of post-stroke neurological dysfunction and ischemic events, but it may increase the risk of ICH together with moderate or severe bleeding. Dipyridamole + aspirin resulted in the lowest risk of bleeding. In MSs, compared to IVT, DAPT may be associated with better improvements in neurological function, and it may not increase the risk of bleeding.

Introduction
Minor strokes and transient ischemic attacks (TIAs) account for 65% of all ischemic cerebrovascular events, 1 and the risk of recurrent stroke and other vascular events during the first 2 weeks is high, ranging from 3.7% to 11.7%.2–4 Without active intervention, there is a high risk of progressing to a more severe and disabling stroke. The MATCH trial showed that aspirin combined with clopidogrel was not superior to clopidogrel alone in patients with either a stroke or TIA. 5 The CHANCE and POINT trials confirmed that early use of dual antibody therapy in patients with mild strokes and high-risk TIAs could reduce the risk of further stroke within the first 90 days. 6
In addition, the use of intravenous thrombolysis for rapid improvement of acute mild symptoms in patients with any type of cerebral infarction is still controversial.7–10 In actual clinical practice, intravenous thrombolytic therapy may not be used or delayed for various reasons.11,12 Studies have shown that alteplase can improve cognitive function in patients with minor strokes. 13
Existing studies are still controversial with respect to the efficacy of different antiplatelet treatments in patients with minor stroke or TIA and the efficacy of antiplatelet and thrombolysis in patients with minor strokes. Therefore, we attempted to include RCTs and high-quality retrospective studies, and divided the meta-analysis into two sections based on the included population. These were patients with either minor strokes or TIAs, and only those ones with minor strokes. In addition, we determined whether intravenous thrombolysis in minor strokes was beneficial to patients, and the effectiveness and safety of different antiplatelet therapies for patients with minor strokes or TIAs.
Materials and methods
The study protocol was registered with PROSPERO (CRD42024537462).
Data sources and searches
We searched the databases of PubMed, Embase, Web of Science and the Cochrane Library in order to identify studies that evaluated the effects of antiplatelet and thrombolytic in minor strokes and TIAs from inception until April 12, 2024. We used only published articles in the English language. The search strategy covered three main concepts following the PICOS format: (“thrombolytic” OR “tenecteplase” OR “alteplase” OR “intravenous thrombolysis” OR “platelet aggregation inhibitors”) AND (“minor stroke” OR “transient ischemic attack”) AND “human” AND “clinical trial.” Medical subject headings and related keywords were employed to find potential articles. The search strategy is displayed in Table S1. The references of relevant studies were inspected to identify extra and follow-up studies. Two authors independently carried out database searches, and they excluded duplicates, examined titles and abstracts, and comprehensively evaluated the full texts to select potentially suitable studies. A third author joined the discussion and resolution when there were any discrepancies.
Eligibility criteria
The inclusion criteria were: (1) the type of studies were randomized and non-randomized clinical trials. (2) The types of participants involved: adults with a National Institutes of Health Stroke Scale (NIHSS) score of 5 or below or at a high-risk of TIA with a score of 4 or more on the ABCD 2 score and started on either antiplatelet or thrombolytic therapy within 72 h or 4.5 h of onset, respectively. (3) The interventions used: dual antiplatelet therapy (DAPT) (aspirin in combination with a second antiplatelet drug, such as clopidogrel, ticagrelor, prasugrel, dipyridamole, ticlopidine, or indobufen) versus aspirin alone or IVT with alteplase/tenecteplase or no IVT. (4) The reporting on one or more clinical outcomes is as detailed below.
The exclusion criteria were as follows: (1) studies without control groups, (2) non-cohort studies, such as case reports and cross-sectional studies, (3) studies evaluating irrelevant outcomes, and (4) studies with incomplete primary data records that were not suitable for statistical analysis and from which comprehensive information were unobtainable from the corresponding authors.
Data extraction
A pre-defined form was employed to extract information from the included studies. The following details were gathered: publication year, study design, subject characteristics (age and sex), sample size, intervention and control protocol, severity of stroke and TIA, dosages, duration of treatment, and follow up and analysis. The information was independently extracted by two authors, and all the extracted data were cross-checked for accuracy by a third author.
Clinical outcomes
The efficacy outcomes were all-cause mortality, excellent outcomes (with modified Rankin Scale scores (mRS) of 0 to 1), functional independence (mRS 0–2), recurrent strokes, major vascular events, ischemic strokes, functional disabilities (mRS 2–6), TIA and vascular causes of mortality. Safety outcomes included any bleeding events such as mild, moderate, moderate-severe and severe ones as well as intracerebral hemorrhage (ICH), symptomatic ICH (sICH) and asymptomatic ICH (aICH). The definitions of all-cause mortality, bleeding and major vascular events for each study are shown in Tables S3–S5.
Quality assessment
We strictly followed the Cochrane Collaboration’s criteria to evaluate the risk of bias in RCTs. We used the R 4.3.1 software package to assess each RCT for seven types of bias: selection, performance, detection, attrition, reporting and other potential biases. Each bias criterion was classified as either “low,” “high,” or “unclear.”
In this analysis, the Risk of Bias in Non-randomized Studies-1 of Intervention (ROBINS-1) 14 was used to evaluate the methodological quality of the included cohort studies. The ROB value for each observational study was evaluated in seven domains. The first three assessed pre- and at-intervention biases, and the last four dealt with post-intervention biases. Overall the ROB value was judged by the ROBINS-1 guidelines. Each domain was independently evaluated by two evaluators, and results were tallied. Any doubtful assessments were resolved with a third evaluator’s help.
Statistical analysis
We employed direct meta-analysis (DMA) combined with network meta-analysis (NMA) to compute the odds ratio (OR) for binary outcomes, by selecting the 95% confidence intervals (CIs). The fixed-effect pairwise meta-analysis was conducted for direct comparisons of ⩾2 RCTs. If there was judged to be substantial heterogeneity, then the sources of heterogeneity were explored, and sensitivity and subgroup analyses were performed. If heterogeneity among studies could not be reduced by this method, then the obtained results were only assessed by using a descriptive analytical method.
We performed the Bayesian NMA by using the “BUGSnet” and the “Gemtc” software packages in R, version 4.3.1. The optimal effect model was determined based on leverage plots and deviance information criterion (DIC). Convergence of iterations and stability of the model were assessed using trace and density plots, in addition to the potential scale reduction factor (PSRF). In cases of closed loops, we tested the assumption of transitivity, by examining whether the direct and indirect comparisons were consistent. Systematic analysis was conducted by using the node-splitting methods. Consistency was deemed to be good if p > 0.05, which indicated consistency, while a p < 0.05 indicated inconsistency among the nodes. In order to calculate the probability ranking of each intervention and assess the likelihood of each intervention being ranked first, the probabilities were aggregated and reported as the surface under the cumulative ranking curve (SUCRA). SUCRA values ranged from 100% for the most favorable intervention to 0% for the least favorable one. The criterion of publication bias was assessed when there was more than 10 clinical studies included in an evaluation.
Results
Study selection
20,938 records were initially screened from electronic databases through a comprehensive searches. After removing 3972 duplicates and screening the titles and abstracts of the remaining records, 16,521 studies were ruled out. Ten RCTs2,3,15–22 and 13 non-RCTs23–35 were included in this assessment, of which eight studies (seven RCTs and one non-RCT) included patients with minor stroke or TIAs, and fifteen studies (3 RCTs and 12 non-RCTs ) included only patients with minor strokes (Figure 1).
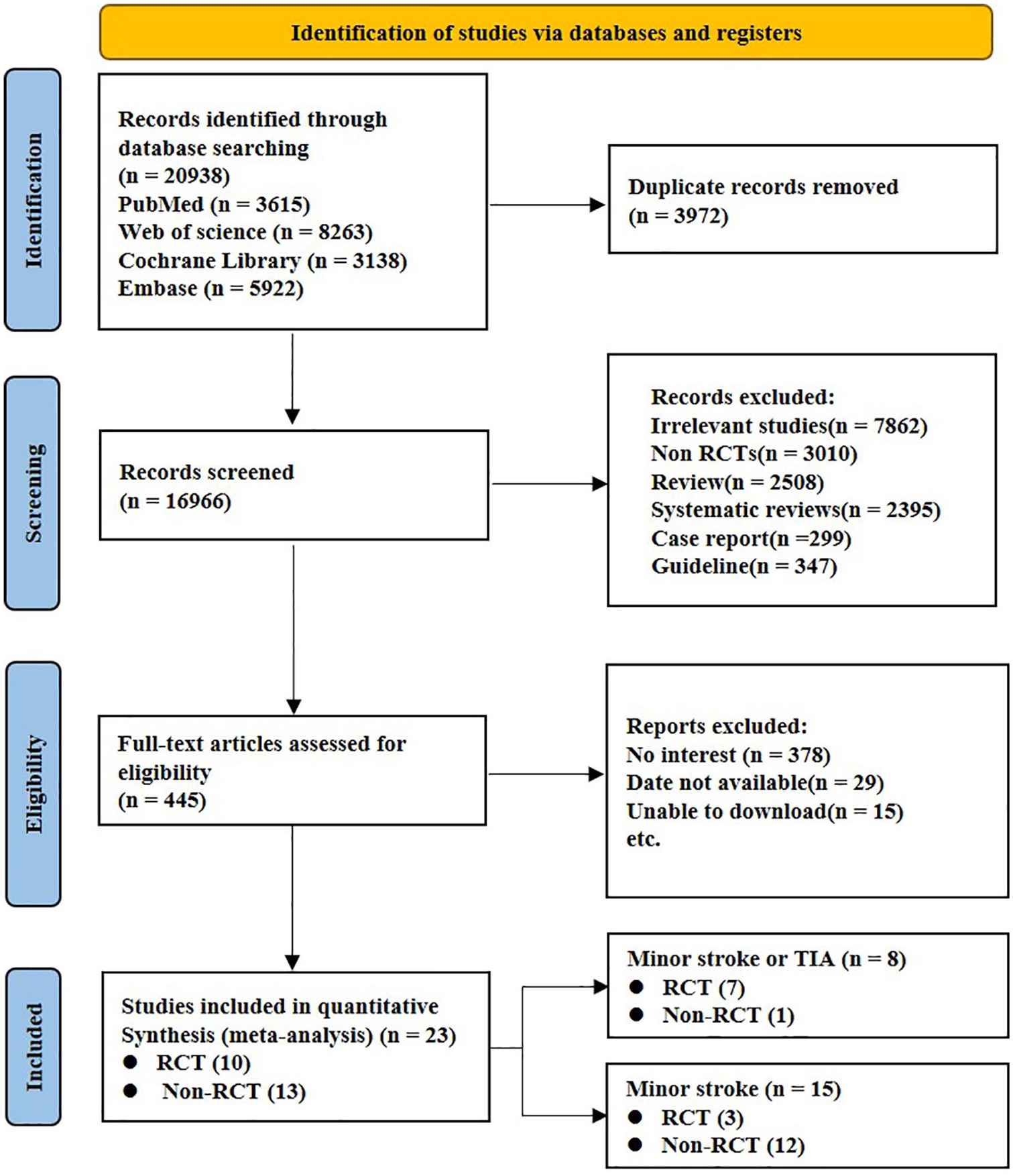
A flow diagram for selection of patients in this study.
Characteristics of the included trials
Ten RCTs and thirteen non-RCTs with 47,135 minor stroke and TIA patients were included. The patients were grouped into four treatment groups in accordance with the treatment strategies they received: SAPT, DAPTs (clopidogrel + aspirin, dipyridamole + aspirin and ticagrelor + aspirin), IVT and No IVT. Among the 47,135 participants, 62.5% were men and 37.5% were women, with ages ranging from 61.5 to 71.0 years old. The reports originated from various countries including China, the United States, Australia, and others, and were published between 2006 and 2023 (Table S2). Figures S1–S4 show the networks of eligible treatment comparisons for the efficacy and safety outcomes considered in this study.
Risk of bias
As demonstrated in Figure S5, the Cochrane bias risk assessment tool was employed to assess the bias risk and methodological quality of the 10 RCTs included. Three studies were identified to have certain concerns regarding one or two items, such as allocation concealment selection bias or blinding of participants and personnel performance bias, but the risk of other biases was judged to be low, suggesting that the overall quality of the included studies was high. Figure S6 shows that out of the 13 ROBIN, seven studies were found to have some concerns regarding ROB and six had low ones.
All-cause mortality
In patients with minor strokes or TIAs, six studies reported on the all-cause mortality, with the network of evidence mapped in Figures S1A and S1B, involving 33,114 patients. The intervening drugs used led to direct and indirect comparisons (Figures S1A and S1B). There was no significant difference in all-cause mortality with four of the antiplatelet interventions (Figure 2(a) and (b)). Dipyridamole + Aspirin was associated with the lowest risk of all-cause mortality, followed by aspirin, and clopidogrel + aspirin and ticagrelor + aspirin, respectively (Figure S7A).
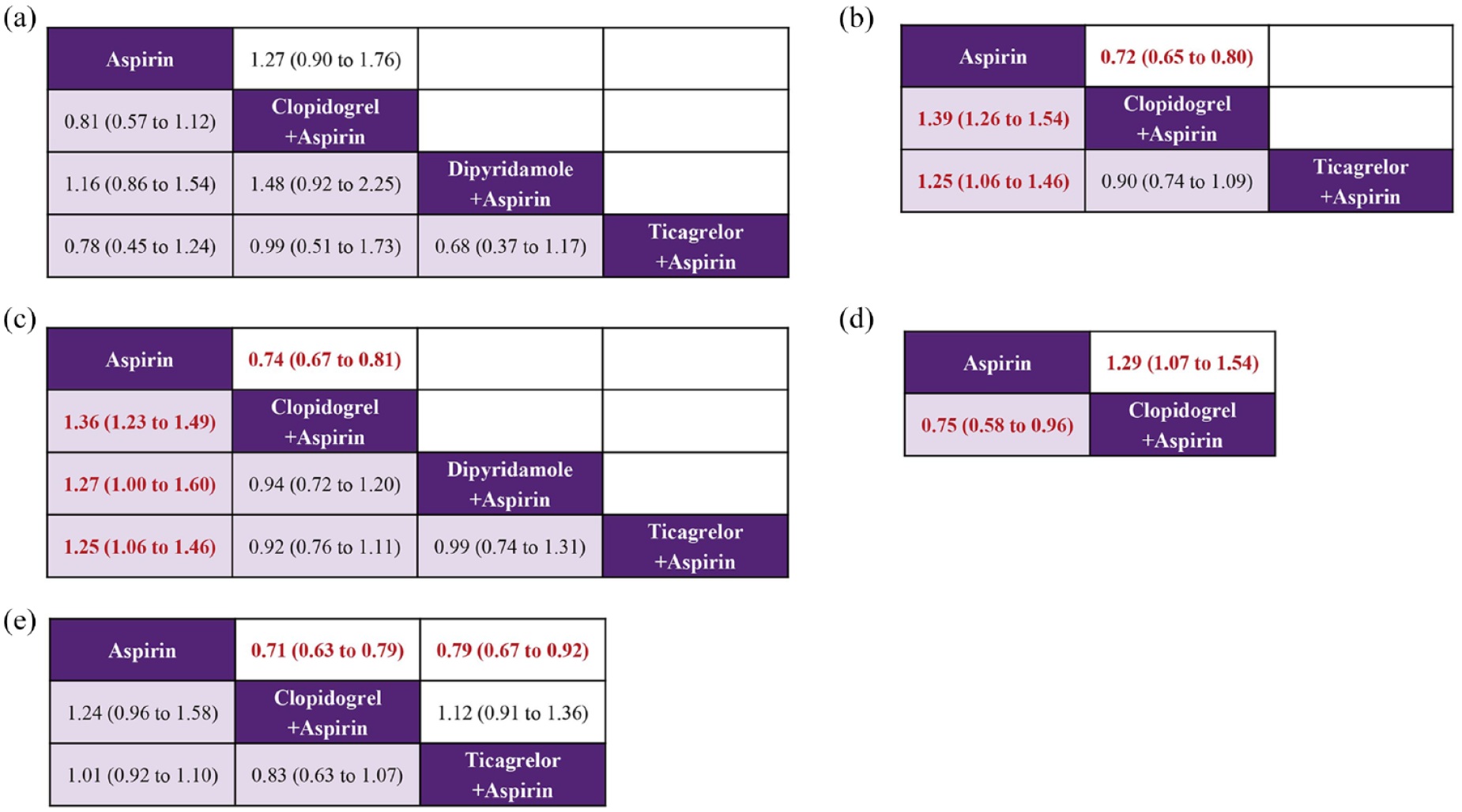
Summary of the efficacy outcomes of network meta-analysis in minor strokes or TIAs. The effect sizes are presented as odds ratio of the means with 95% credible intervals. The diagram should be read from left to right with OR < 1 favoring the column-defining treatment and this meant that the treatment in the column was associated with a lower risk for the outcome than the treatment in the row. The significant results are presented in red bold fonts: (a) all-cause mortality at 90 days (upper white fields) and all-cause mortality (lower shaded fields), (b) recurrent stroke at 90 days (upper white fields) and recurrent stroke (lower shaded fields), (c) major vascular events at 90 days (upper white fields) and major vascular events (lower shaded fields), (d) mRs 0–1 at 90 days (upper white fields) and mRs 0–2 at 90 days (lower shaded fields), and (e) ischemic stroke (upper white fields) and mRs 2–6 at 90 days (lower shaded fields).
In minor strokes, eight studies reported on the all-cause mortality at 90 days, with the network of evidence mapped in Figures S2A and S2B, involving 4598 patients. The intervening drugs used led to direct and indirect comparisons, partially completing the loop (Figure S2A). Compared with SAPT, DAPT was significantly decreased in all-cause mortality at 90 days (Figure 3(a)). The results of meta-analysis for RCTs and non-RCTs studies were consistent (Figure S8A). Compared with no IVT, IVT was significantly decreased in all-cause mortality, with all the data being obtained from non-RCTs (Figures 3(a) and S8A). DAPT was associated with the lowest risk of all-cause mortality at 90 days, followed by IVT, no IVT and SAPT, respectively (Figure S9A).
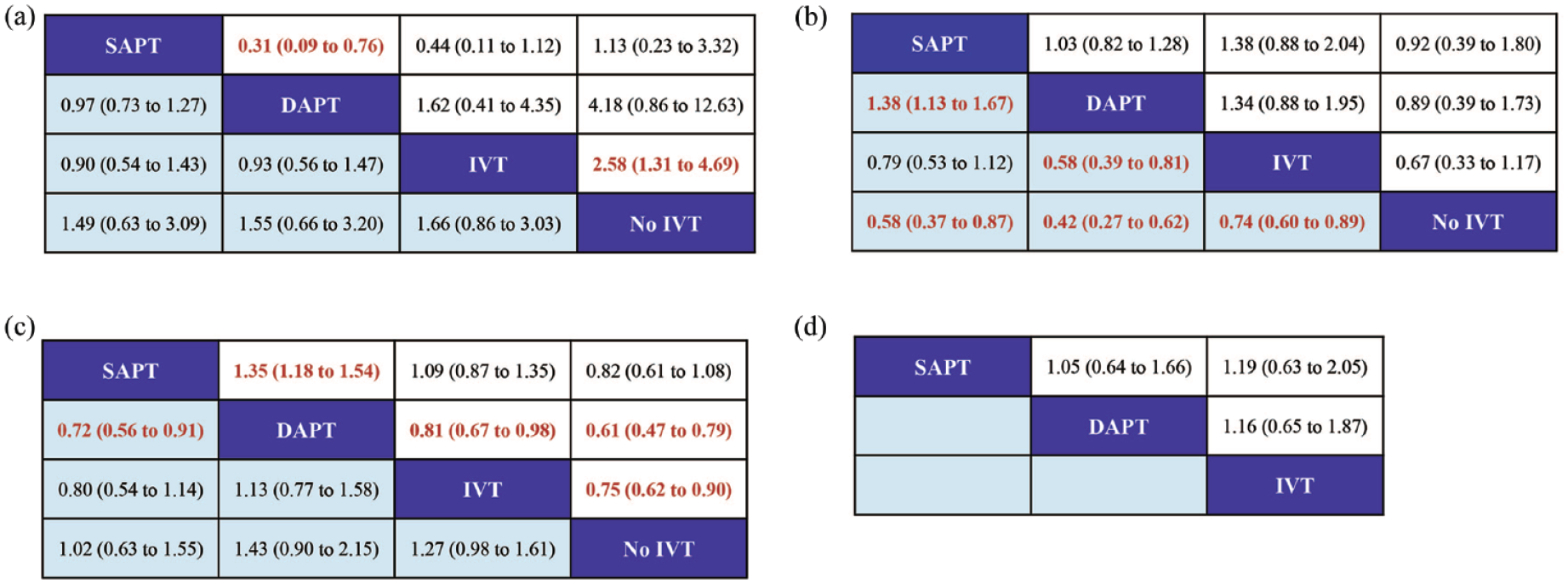
Summary of the efficacy outcomes of network meta-analysis in minor strokes. The effect sizes are presented as odds ratio of the means with 95% credible intervals. The diagram should be read from left to right with OR < 1 favoring the column-defining treatment and this meant that the treatment in the column was associated with a lower risk for the outcome than the treatment in the row. The significant results are presented in red bold fonts: (a) all-cause mortality at 90 days (upper white fields) and recurrent stroke (lower shaded fields), (b) major vascular events at 90 days (upper white fields) and mRs 2–6 at 90 days (lower shaded fields), (c) mRs 0–1 at 90 days (upper white fields) and mRs 0–2 at 90 days (lower shaded fields), and (d) ischemic stroke (upper white fields).
Recurrent stroke
In minor strokes or TIAs, six studies reported on the occurrence events of stroke, and the network of evidence is depicted in the evidence relationship diagram involving 30,767 patients. The intervening drugs used formed both direct and indirect comparisons (Figures S1C and S1D). In comparison with aspirin alone, clopidogrel + aspirin (OR = 0.72; 95% CI 0.65–0.80) was significantly associated with lower stroke recurrence at 9 days (Figure 2(b)). Aspirin was significantly associated with higher stroke recurrence during follow-up, compared with clopidogrel + aspirin (OR = 1.39; 95% CI 1.26–1.54) and ticagrelor + Aspirin (OR = 1.25; 95% CI 1.06–1.46; Figure 2(b)). Analytical results of SUCRA cumulative probability revealed that clopidogrel + aspirin had the lowest number of stroke recurrence, followed by ticagrelor + aspirin and aspirin, respectively (Figure S7B).
In minor strokes, eight studies reported on the occurrence events of stroke at 90 days, and the network of evidence is depicted in the evidence relationship diagram involving 7977 patients. The intervening drugs used formed both direct and indirect comparisons, partially completing the loop (Figure S2B). There was no significant difference in all-cause mortality with four of the interventions (Figure 3(a)). The results of meta-analysis of RCT and non-RCT studies were consistent (Figure S8B). Analytical results of SUCRA cumulative probability revealed that no IVT had the lowest number of stroke recurrence, followed by SAPT, DAPT, and IVT, respectively (Figure S9B).
Major vascular events
For minor strokes or TIAs, seven studies involving four different interventions and a combined participant population of 43,426 were included. The intervening drugs used formed both direct and indirect comparisons (Figures S1E and S1F). In comparison with aspirin, clopidogrel + aspirin (OR = 0.74; 95% CI 0.67–0.81) was significantly associated with lower major vascular events at 90 days (Figure 2(c)). Aspirin was significantly associated with higher major vascular events during follow-up, compared with clopidogrel + aspirin, dipyridamole + aspirin and ticagrelor + aspirin (Figure 2(b)). The SUCRA analysis cumulative probability revealed that patients being given clopidogrel + aspirin had the lowest number of major vascular events, followed by dipyridamole + aspirin, ticagrelor + aspirin and aspirin, respectively (Figure S7C).
In minor strokes, 10 studies involving four different interventions and a combined participant population of 9414 were included. The intervening drugs used formed both direct and indirect comparisons, partially completing the loop (Figure S2C). There was no significant difference in all-cause mortality with four of the interventions (Figure 3(c)). The SUCRA analysis cumulative probability revealed that no IVT patients had the lowest number of major vascular events, followed by SAPT, DAPT, and IVT, respectively (Figure S9C).
Excellent outcomes (mRS 0–1)
In minor strokes or TIAs, only one study was included with 6100 participants accounted for excellent outcomes (mRS 0–1) at 90 days. This outcome measure involved only two interventions and did not form a network (Figure S1G). Compared to aspirin alone, clopidogrel + aspirin (OR = 1.29; 95% CI 1.07–1.54) was associated with a significant difference in excellent outcomes (mRS 0–1) at 90 days (Figure 2(d)).
In minor strokes, 13 included studies with 9949 participants accounted for excellent outcomes (mRS 0–1) at 90 days. Direct and indirect comparisons were established for each interventional drug and this formed a partially closed loop (Figure S2D). Compared to SAPT, DAPT (OR = 1.35; 95% CI 1.18–1.54) was associated with a significant difference in excellent outcomes (mRS 0–1) at 90 days (Figure 3(c)). The results of meta-analysis for RCT and non-RCT studies were consistent (Figure S8D). IVT (OR = 0.81; 95% CI 0.67–0.98) and no IVT (OR = 0.61; 95% CI 0.47–0.79) with a 90-day excellent outcome (mRS 0–1) was significantly lower than in the DAPT group (Figure 3(c)). DAPT was associated with the highest occurrence of excellent outcomes (mRS 0–1) at 90 days, followed by IVT, and for SAPT and it was no IVT (Figure S9D).
Functional independence (mRS 0–2)
In minor strokes or TIAs, one study with 6100 participants was studied and this accounted for excellent outcomes (mRS 0–2) at 90 days. This outcome measure involved only two interventions and did not form a network (Figure S1H). Compared to clopidogrel + aspirin, aspirin (OR = 0.75; 95% CI 0.58–0.96) was associated with a significant difference in excellent outcomes (mRS 0–2) at 90 days (Figure 2(d)).
In minor strokes, the occurrence of functional independence at 90 days was reported in 10 studies, including 7569 patients with 6900 patients with mRS scores of 0–2. For each interventional drug used, direct and indirect comparisons were performed, thereby partially constituting a closed loop (Figure S2E). DAPT can significantly increase the incidence of functional independence (mRS 0–2) at 90 days compare to SAPT. The results of meta-analysis for RCT and non-RCT studies were consistent (Figures 3(c) and S8E). These results indicated that DAPT was the most effective regimen in improving the functional independence (mRS 0–2) of patients at 90 days with the biggest SUCRA, followed by IVT, SAPT, and no IVT (Figure S9E).
Functional disability (mRS 2–6)
In minor strokes or TIAs, functional disability (mRS 2–6) at 90 days were reported in two studies that enrolled 17,116 patients incurring a combined total of 2825 events. The intervening drugs used formed direct and indirect comparisons (Figure S1I). There was no significant difference in all-cause mortality with three of the interventions (Figure 2(e)). The results of SUCRA analysis showed that the cumulative probability with clopidogrel + aspirin had the lowest functional disability (mRS 2–6) at 90 days, followed by ticagrelor + aspirin and aspirin, respectively (Figure S7D).
In minor strokes, functional disability (mRS 2–6) at 90 days were reported in nine studies that enrolled 6310 patients with a combined total of 1170 events. The intervening drugs used formed direct and indirect comparisons, partially closing the loop (Figure S2F). In comparison with no IVT, SAPT (OR = 0.58; 95% CI 0.37–0.87), DAPT (OR = 0.42; 95% CI 0.27–0.62) and IVT (OR = 0.74; 95% CI 0.60–0.89) was significantly associated with lower functional disability (mRS 2–6) at 90 days (Figure 3(b)). The significant difference between no IVT and other interventions mentioned above was obtained from non-RCTs (Figure S8F). The estimated effects indicated that DAPT was significantly associated with a lower functional disability (mRS 2–6) at 90 days, compared with IVT (OR = 0.58; 95% CI 0.39–0.81; Figure 3(b)). The results of SUCRA analysis showed that the cumulative probability with DAPT had the lowest functional disability (mRS 2–6) at 90 days, followed by SAPT, IVT, and no IVT (Figure S9F).
Ischemic stroke
In minor strokes or TIAs, ischemic stroke was reported in four studies involving three different interventions, encompassing 21,167 patients who collectively experienced 1869 events. The intervening drugs used established both direct and indirect comparisons (Figure S1J). Compared to aspirin alone, clopidogrel + aspirin (OR = 0.71; 95% CI 0.63–0.79) and ticagrelor + aspirin (OR = 0.79; 95% CI 0.67–0.92) were significantly associated with lower ischemic stroke during follow-up (Figure 2(e)). Clopidogrel + aspirin was associated with the lowest occurrence of ischemic stroke, followed by ticagrelor + aspirin and aspirin, respectively (Figure S7D).
In minor strokes, ischemic stroke was reported in two studies involving three different interventions, encompassing 1619 patients who collectively experienced 103 events. The intervening drugs used established both direct and indirect comparisons, partially closing the loop (Figure S2G). There was no significant difference in ischemic strokes with three of the interventions (Figure 3(d)). SAPT was associated with the lowest occurrence of ischemic stroke, followed by DAPT and IVT (Figure S9G).
Any type of bleeding events
In minor strokes or TIAs, seven studies reported bleeding events during follow-up, and these included 30,945 participants. The involved drugs established direct and indirect comparisons (Figure S3A). The estimated effects indicated that clopidogrel + aspirin (OR = 1.94; 95% CI 1.58–2.36) and ticagrelor + aspirin (OR = 3.59; 95% CI 1.74–6.86) were significantly associated with a higher bleeding risk, when compared with aspirin (Figure 4(a)). Compared to clopidogrel + aspirin and ticagrelor + aspirin, the risk of bleeding was significantly reduced in dipyridamole + aspirin (Figure 4(a)). The SUCRA cumulative probability revealed that dipyridamole + aspirin had the lowest bleeding risk, followed by aspirin, clopidogrel + aspirin and ticagrelor + aspirin (Figure S10A).

Summary of the safety outcomes of network meta-analysis in minor strokes or TIAs. The effect sizes are presented as the odds ratio of means with 95% credible intervals. The diagram should be read from left to right with OR < 1 favoring the column-defining treatment and this meant that the treatment in the column was associated with a lower risk for the outcome than the treatment in the row. The significant results are presented in red bold fonts: (a) any bleeding (upper shaded fields) and ICH (lower shaded fields), (b) sICH (lower shaded fields), (c) mild bleeding events at 90 days (upper shaded fields) and moderate-severe bleeding events at 90 days (lower shaded fields), and (d) moderate bleeding events at 90 days (upper shaded fields) and severe bleeding events at 90 days (lower shaded fields).
In minor strokes, 13 studies reported bleeding events during follow-up, and these included 9547 participants. The involved drugs established direct and indirect comparisons, partially completing the loop (Figure S4A). The estimated effects indicated that IVT was significantly associated with a higher risk of bleeding, when compared with SAPT (OR = 5.51; 95% CI 2.27–11.93) and DAPT (OR = 5.15; 95% CI 2.21–10.92; Figure 5(a)). The results were basically consistent between those of RCTs and no-RCTs (Figure S11A). Compared to IVT, the risk of bleeding was significantly reduced in no IVT (Figure 5(a)), and the relevant data were obtained from no-RCTs (Figure S11A). The SUCRA cumulative probability revealed that SAPT had the lowest bleeding risk, followed by DAPT, no IVT and IVT, respectively (Figure S12A).
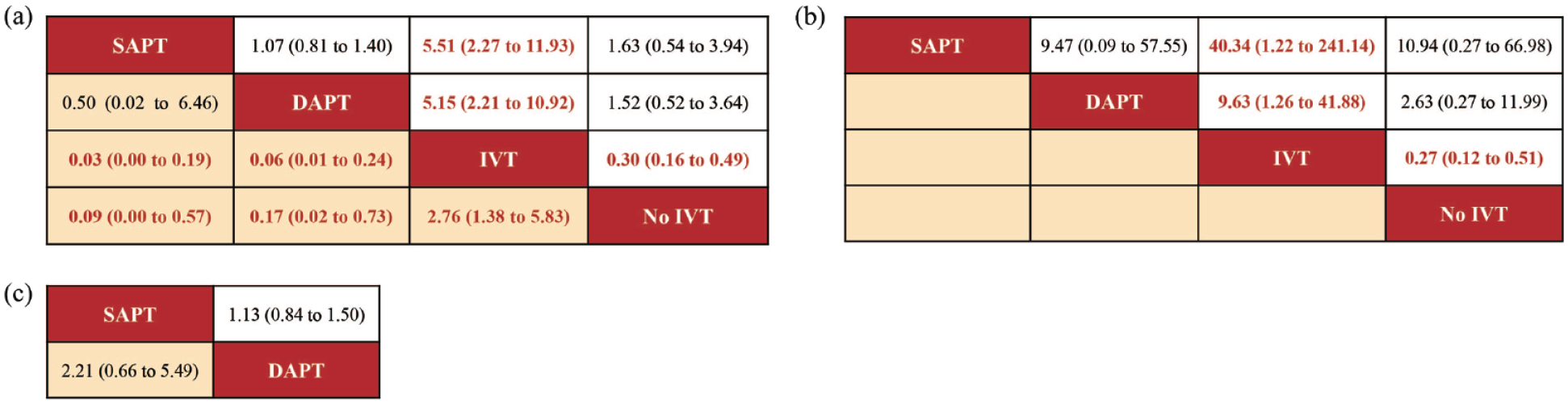
Summary of the safety outcomes of network meta-analysis in minor strokes. The effect sizes are presented as the odds ratio of means with 95% credible intervals. The diagram should be read from left to right with OR < 1 favoring the column-defining treatment and this meant that the treatment in the column was associated with a lower risk for the outcome than the treatment in the row. The significant results are presented in red bold fonts: (a) any bleeding (upper shaded fields) and ICH (lower shaded fields), (b) sICH (lower shaded fields), and (c) mild bleeding events at 90 days (upper shaded fields) and moderate-severe bleeding events at 90 days (lower shaded fields).
ICH
In minor strokes or TIAs, three RCTs reported the occurrence of ICH at 90 days, encompassing 17,510 patients who collectively experienced 55 events. Direct and indirect comparisons were made for each intervention drug used (Figure S3B). The estimated effects indicated that aspirin was significantly associated with a lower ICH risk at 90 days, compared with clopidogrel + aspirin (OR = 0.48; 95% CI 0.19–0.95) and ticagrelor + aspirin (OR = 0.32; 95% CI 0.11–0.68; Figure 4(a)). The SUCRA cumulative probability results revealed that aspirin had an ICH risk at 90 days, followed by clopidogrel + aspirin and ticagrelor + aspirin, respectively (Figure S10B).
In minor strokes, the occurrence of ICH at 90 days was reported in six studies, encompassing 3209 patients who collectively experienced 62 events. Direct and indirect comparisons were made for each intervention drug used (Figure S4B). The estimated effects indicated that IVT was significantly associated with a higher ICH risk at 90 days, compared with SAPT (OR = 0.03; 95% CI 0.00–0.19), DAPT (OR = 0.06; 95% CI 0.01–0.24) and no IVT (OR = 2.76; 95% CI 1.38–5.83; Figure 5(a)). Compared to no IVT, the risk of ICH was significantly reduced in SAPT and DAPT (Figure 5(a)). The SUCRA cumulative probability results revealed that SAPT had an ICH risk at 90 days, followed by DAPT, no IVT, and IVT, respectively (Figure S12B).
SICH
In minor strokes or TIAs, only one trial accounted for sICH at 90 days. The estimated effects indicated that there was no significant difference in sICH risk at 90 days with clopidogrel + aspirin and aspirin (Figure 4(b)).
In minor strokes, 8 studies were included in the NMA data and these accounted for sICH at 90 days. Each intervention drug used formed direct and indirect comparisons (Figure S4C). The estimated effects indicated that IVT was significantly associated with a higher sICH risk at 90 days, compared with SAPT (OR = 40.34; 95% CI 1.22–241.14), DAPT (OR = 9.63; 95% CI 1.26–41.88) and no IVT (OR = 0.27; 95% CI 0.12–0.51; Figure 5(b)). The significant difference obtained between SAPT and IVT were all from RCT results, while those of DAPT and IVT may have been partially affected by the non-RCT results (Figure S11C). The SUCRA cumulative probability results revealed that SAPT had the lowest sICH at 90 days, followed by DAPT, no IVT, and IVT, respectively (Figure S12C).
Mild bleeding events
In minor strokes or TIAs, four studies accounted for mild bleeding events observed at 90 days with 16,798 participants included in the study. Compared to clopidogrel + aspirin, the risk of mild bleeding events at 90 days was significantly reduced in aspirin (Figure 4(c)). With respect to minor strokes, two non-RCT accounted for mild bleeding events observed at 90 days with 3328 participants included in the study. The estimated effects indicated that there was no significant difference in mild bleeding events at 90 days with SAPT and DAPT (Figure 5(c)).
Moderate-severe bleeding events
In minor strokes or TIAs, three studies reported moderate-severe bleeding events at 90 days. Direct and indirect comparisons were made for each intervention drug used (Figure S3E). Compared to aspirin alone, the risk of moderate-severe bleeding at 90 days was significantly increased in ticagrelor + aspirin (OR = 0.32; 95% CI 0.15–0.58), and this may have been affected by the RCT results (Figure 4(c)). The SUCRA cumulative probability results revealed that aspirin had the lowest sICH at 90 days, followed by clopidogrel + aspirin and ticagrelor + aspirin, respectively (Figure S10C). With respect to minor strokes, two non-RCT reported moderate-severe bleeding events at 90 days. There was no significant difference in moderate-severe events at 90 days with SAPT and DAPT (Figure 5(c)).
Moderate bleeding events
In minor strokes or TIAs, only one RCT reported moderate bleeding events at 90 days. The estimated effects indicated that there was no significant difference in moderate bleeding events at 90 days with clopidogrel + aspirin and aspirin (Figure 4(d)).
Severe bleeding events
In minor strokes or TIAs, three RCTs accounted for severe bleeding events at 90 days and involved 21,067 participants who were included in the NMA. Direct and indirect comparisons were made for each intervention drug used (Figure S3G). When compared to clopidogrel + aspirin and ticagrelor + aspirin, the risk of severe bleeding at 90 days was significantly reduced in patients given aspirin (OR = 0.53; 95% CI 0.26–0.97, OR = 0.26; 95% CI 0.10–0.53; Figure 4(d)). The SUCRA cumulative probability results revealed that aspirin had the lowest severe bleeding risk at 90 days, followed by clopidogrel + aspirin and ticagrelor + aspirin, respectively (Figure S10D).
Network consistency and heterogeneity
In terms of all outcomes, the comparison of the total posterior residual deviance and the quantity of unconstrained data points for all FE and RE NMAs suggested that the fit was appropriate. The heterogeneity results illustrated that most of the comparisons across the included studies had minimal or median heterogeneity, and comparisons of the DIC showed no major variations in the fit between the FE and RE consistency models (Figures S13 and S14). In order to make the results more reliable, the RE model was used in all meta-analyses performed in this study. The comparisons of DIC and the examination of leverage plots gave no indications of a violation of the consistency assumption. The detailed information of model fit statistics and checks for inconsistency are provided in Figures S15–S20.
Publication bias
We utilized a comparison-adjusted funnel plot to examine whether there was publication bias and small-study effects. The determination that the funnel plot was approximately symmetrical suggested no sign of publication bias (Figures S21 and S22). However, it is important to realize that the evaluation of publication bias through funnel plots may be unreliable for clinical outcomes when the number of included studies are small.
Discussion
This systematic review and NMA included data from 10 RCTs and 13 on-RCTs involving 47,135 minor stroke or TIA patients. Among these, seven RCTs and one non-RCT included patients with minor strokes or TIAs and three RCTs and twelve non-RCTs included patients with only minor strokes. In minor strokes or TIAs, we found that DAPT (using clopidogrel + aspirin, dipyridamole + aspirin and ticagrelor + aspirin) was able to significantly preventing post-stroke neurological dysfunctions (mRS 0–1 and mRS 0–2), recurrent strokes, major vascular events and ischemic strokes for up to 90 days after patients were initially subjected to aspirin. There was no significant difference between the three different types of DAPT. However, DAPTs were associated with a significantly increased risk of any type of bleeding and ICH as well as moderate-to-severe and severe hemorrhage when compared to aspirin alone. It should be noted that compared to clopidogrel + aspirin and ticagrelor + aspirin, the risk of any bleeding events was significantly decreased in the patients who received dipyridamole + aspirin. With respect to other safety outcomes, there was no significant difference between different the different DAPTs.
With minor strokes, when compared to SAPT, DAPT had a significant advantage in decreasing all-cause mortality and major vascular events at 90 days. In addition, it improved neurological dysfunctions (mRS 0–1 and mRS 0–2) and there was no significant difference in the risk of bleeding. Compared to IVT, DAPT may be associated with improvements in patients’ neurological functions as well as reducing the lower risk of bleeding.
In our NMA, there were 12 studies with DAPT and SAPT in the intervention and control groups, and these included eight RCTs and four non-RCTs. Nine of the studies used clopidogrel and aspirin as DAPTs, while in the rest, cilostazol, ticagrelor and dipyridamole were combined with aspirin and given to patients. Consistent with current international guidelines for minor strokes and TIAs, our meta-analysis also showed that DAPT was more effective than SAPT in stroke prevention and neurological function improvements, but it was observed to be less safe than SAPT. The above results were also consistent with the results of the RCT meta-analysis, which improved the reliability of the results. It is noteworthy that those studies with larger sample sizes may contribute more to the meta-analysis. The four studies with the largest sample sizes in this study were the THALES, 20 INSPIRES, 17 CHANCE, 3 and POINT 2 trials, which greatly influenced the efficacy and safety of the outcome indicators. However, in addition to the CHANCE 3 trial, the difference in DAPT loading dose in the other three studies probably had a greater impact on the results of the meta-analysis. Specifically, the loading dose of 300–325 mg aspirin in the THALES 20 trial, 100–300 mg aspirin in the INSPIRES 17 trial and 600 mg clopidogrel in the POINT 2 trial, all of which may have magnified the efficacy of DAPT and the risk of bleeding.
Early thrombolysis is the most effective measure to restore cerebral blood flow and improve nerve function in patients with acute ischemic strokes. It was feasible to determine whether patients with minor strokes benefited from thrombolytic therapy in this study. 36 Our results showed that compared with IVT, DAPT could only reduce the incidence of neurological dysfunction, while SAPT and DAPT had no significant advantages in improving other effective outcomes. In terms of safety, antiplatelet therapy was superior. It should be noted that antiplatelet strategies after thrombolytic therapy can have a direct impact on clinical outcomes. Unfortunately, in some of studies23,26,31 included here, there was a failure to specify the details of any follow-up antiplatelet therapy strategies. There may have been large differences in outcomes for patients but these were not reported. In addition, all included studies used alteplase for thrombolytic therapy, and although the TEMPO-1 37 trial compared intravenous thrombolytic therapy with different doses of tenecteplase in minor stroke patients, these data were excluded because the intervention did not meet the inclusion criteria. Currently, TEMPO-2 studies are underway to compare the efficacy of tenecteplase and antiplatelet therapy in minor stroke patients, and this may provide additional therapeutic possibilities in the future. Given these concerns, the results should be interpreted with caution, and further high-quality studies are needed to confirm the benefits of thrombolytic therapy in patients with minor strokes.
In the present study, IVT was found to be associated with a lower incidence of all-cause mortality and higher risks of sICH and ICH, when compared with no IVT. Additionally, patients in the no IVT group tended to have a lower odds of recurrent stroke than that observed in the non-RCT subgroup. The above results were all from retrospective studies. In the process of article screening, we only included studies that excluded the influence of confounding factors by using statistical methods such as propensity matching scores, so as to improve the quality of the included literature.
However, we found that seven of the interventions and controls included in this study were IVT and no IVT, and five of the studies included patients in the no IVT group who received unknown treatments (Table S2). There is no doubt that the differences of the no IVT group to prevent vascular re-occlusion in each study would directly affect the clinical outcome and reduce the credibility of the results of the meta-analysis. This may be the reason why patients in the no IVT group in our study tended to have the lowest odds of recurrent stroke, although this data were not statistically different. Additionally, an important factor in the absence of significant differences in other efficacy outcomes between IVT and no IVT may be due to the shorter half-life of intravenous alteplase (compared to oral antiplatelet agents) and the absence of antithrombotic coverage within 24 h after exponential stroke following plasma alteplase clearance. 38
We noted that the dose of alteplase included in the study was 0.9 mg/kg, which is the recommended dose for patients with acute ischemic stroke within 4.5 h of onset. 36 For patients with acute ischemic stroke receiving intravenous thrombolytic therapy, the occurrence of hemorrhage transformation is the most important concern for clinical decision-makers, and it is also one of the main reasons why thrombolysis was not previously recommended for patients with minor strokes. Compared with low dose of alteplase (0.6 mg/kg), the use of 0.9 mg/kg alteplase intravenous thrombolytic therapy is a risk factor that cannot be ignored as it is likely to increase bleeding transformation. However, only a few clinical studies were available on the use of low-dose alteplase in the treatment of minor strokes. This means that there is still a significant knowledge gap regarding the use of low-dose alteplase intravenous thrombolytic agents in patients with minor strokes. Late-stage trials have the potential to provide a promising and cost-effective thrombolytic therapy for patients with minor ischemic strokes.
This study had some limitations. Firstly, the synthesized data contained a certain inherent bias that was associated with the RCTs and non-RCTs included. Owing to the observational nature of some studies, the decision of patients to accept SAPT or DAPT or IVT or No IVT was not randomized. Secondly, there were inconsistencies among the included studies in terms of the time of treatment initiation, baseline NIHSS scores, definitions of minor stroke and dosing paradigms of anti-platelets usage. These inconsistencies could have potentially influenced the outcome assessment of the synthesized analysis. Thirdly, there was no closed loop in the network diagram of some outcome indicators, which indicated that there was some possible consistencies among all the studies and analytical results reported. Fourthly, in some of the included studies, the preventive measures within the no IVT group were unclear, and the possible differences could have directly affected the clinical outcome and reduce the credibility of the meta-analysis results to a certain extent. Lastly, there were subsets of outcomes with fewer than 10 studies. The validity of the tests used in the results from these may be relatively although the publication bias analysis in these cases was found to be low. Therefore, the results should be considered with some caution since the possibility of confounders must be considered. However, many of our conclusions were robust and clear and this novel study provides some enlightening insights into future interventions in the treatment of minor strokes or TIAs.
Conclusions
This study represents a comprehensive meta-analysis to evaluate the efficacy and safety of different antiplatelet therapies in patients with minor strokes or TIAs and antiplatelet and IVT in minor strokes. In minor strokes or TIAs, compared to aspirin, DAPTs (clopidogrel + aspirin, dipyridamole + aspirin and ticagrelor + aspirin) can effectively prevent the recurrence of post-stroke neurological dysfunction and ischemic events, but it may increase the risk of ICH together with either moderate or severe bleeding. Dipyridamole + aspirin had the lowest risk of any bleeding. In minor strokes, DAPT may be associated with better improvements in neurological function compared to IVT. In addition, DAPT was associated with a lower risk of bleeding.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241303686 – Supplemental material for Acute treatment and secondary prevention for patients with minor stroke or transient ischemic attack: A Bayesian network meta-analysis
Supplemental material, sj-docx-1-eso-10.1177_23969873241303686 for Acute treatment and secondary prevention for patients with minor stroke or transient ischemic attack: A Bayesian network meta-analysis by Sitong Guo, Shiran Qin, Dandan Xu, Chunxia Chen and Xiaoyu Chen in European Stroke Journal
Footnotes
Acknowledgements
The authors thank Dr Dev Sooranna of Imperial College London for editing the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Science Foundation of China (Nos. 82260258, 81960246, and 82160763) and Natural Science Foundation of Guangxi Zhuang Autonomous Region (No. 2024GXNSFBA010018).
Ethical approval
Not applicable.
Informed consent
Not applicable.
Guarantor
Xiaoyu Chen.
Contributorship
Both authors performed searching and screening of studies. Disagreements were resolved by discussion between two authors. All authors were involved in data analysis. Sitong Guo wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
