Abstract
The incidence of intracerebral hemorrhage (ICH) associated with oral anticoagulants (OAC) is about one in five cases of ICH and associated with severe clinical presentation, frequently rapid clinical deterioration, and 30-days mortality of app 50%. This narrative review gives an overview of presentation and acute treatment of OAC-ICH. Oral anticoagulants do not cause ICH but lead to prolongation of bleeding and higher risk of hematoma expansion (HE). Clinicoradiological characteristics of oral anticoagulant associated ICH are not different from ICH in general. The therapeutic principle of reversal is to prevent or limit HE. The mode of action of the reversal agents for vitamin K antagonists, direct oral thrombin inhibitor and direct oral factor Xa inhibitors are described in the main text. We also discuss the principles of blood pressure lowering in the setting of acute OAC-ICH as it may be the second driving force of HE. Stroke unit care is needed to prevent further complications. Data from randomized controlled trials and observational data from unselected patients are needed to make stronger and more precise recommendations on acute therapy.
Keywords
Introduction
The incidence of intracerebral hemorrhage (ICH) in Europe in 2019 was approximately 15.3 cases per 100,000 people per year (with highest rate in Georgia (41.1 cases per 100,000 people per year) and Turkey (33.5 cases per 100,000 people per year), and lowest rates in Finland (6.9 cases per 100,000 people per year) and Denmark (7.3 cases per 100,000 people per year 1 ). The incidence of ICH associated with the intake of oral anticoagulation (OAC) was found to be approximately one-fifth of all ICH cases, thus about 3 per 100.000 in Europe.2,3 The prognosis of patients with OAC-ICH is poor 4 with the main driving predictor being bleeding volume that can be influenced by minimizing expansion, which relates to parenchymal as well as to intraventricular expansion. In recent years, there have been significant advancements in the development of reversal agents for OACs, remains unclear as data from randomized controlled trials comparing treatment approaches are lacking. This article reviews the current principles of reversal of anticoagulation in patients with ICH related to OACs, including the available agents, their pharmacology, and the evidence for their use.
Mode of action of OACs and clinically relevant reversal agents – Coagulation
The OACs include the following
Vitamin K antagonists: warfarin, phenprocoumon, acenocoumarol, fluindione, and others
Direct oral thrombin inhibitor: dabigatran etexilate
Direct oral factor Xa inhibitors: rivaroxaban, apixaban and edoxaban
Reversal agents for OAC in patients with intracerebral hemorrhage related to OACs include the following.
Prothrombin complex concentrate (PCC) mainly for repletion of Vitamin-K dependent coagulation factors (3-factor PCC: II, VII, IX; 4-factor PCC: II, VII, IX, X)
Idarucizumab developed as specific reversal agent for the effect of dabigatran
Andexanet alfa developed as specific reversal agent for the effect of direct factor Xa-inhibitors (apixaban, edoxaban, rivaroxaban and enoxaparine)
There is a difference concerning the mode of action: agents like andexanet alfa and idarucizumab bind to the specific OAC (factor Xa-inhibitors or dabigatran), whereas agents like PCC replace depleted coagulation factors. In acute treatment the mechanisms of action of antidotes including andexanet alfa and idarucizumab are sometimes termed reversal in contrast to describing the mode of action of PCC, FFP or vitamin-K repletion.
Vitamin K antagonists (VKAs) inhibit regeneration of vitamin K in the vitamin K cycle. This leads to a partial deficiency of vitamin K and production of non-functional acarboxy versions of the vitamin K-dependent coagulation factors (FII, FVII, FIX and FX) instead of the normal transcarboxylated factors 5 (Figures 1 and 2). Since VKA only inhibits the regeneration of vitamin K and not the vitamin itself, administration of vitamin K will always be able to neutralize the effect of VKA regardless of the VKA dose, but it takes > 24 h before the full effect of vitamin K occurs.6,7 For rapid reversal of the effect of VKA, prothrombin complex concentrate (PCC) is used. So-called 4-factor PCCs contain all the vitamin K-dependent coagulation factors as well as protein C and S, and by infusion a balanced substitution of all the decreased factors is achieved.8–10 Recommended dosage is 25–50 IU/kg body weight, depending on the INR before the infusion. To prevent a rebound effect, vitamin K is given at the same time. 11
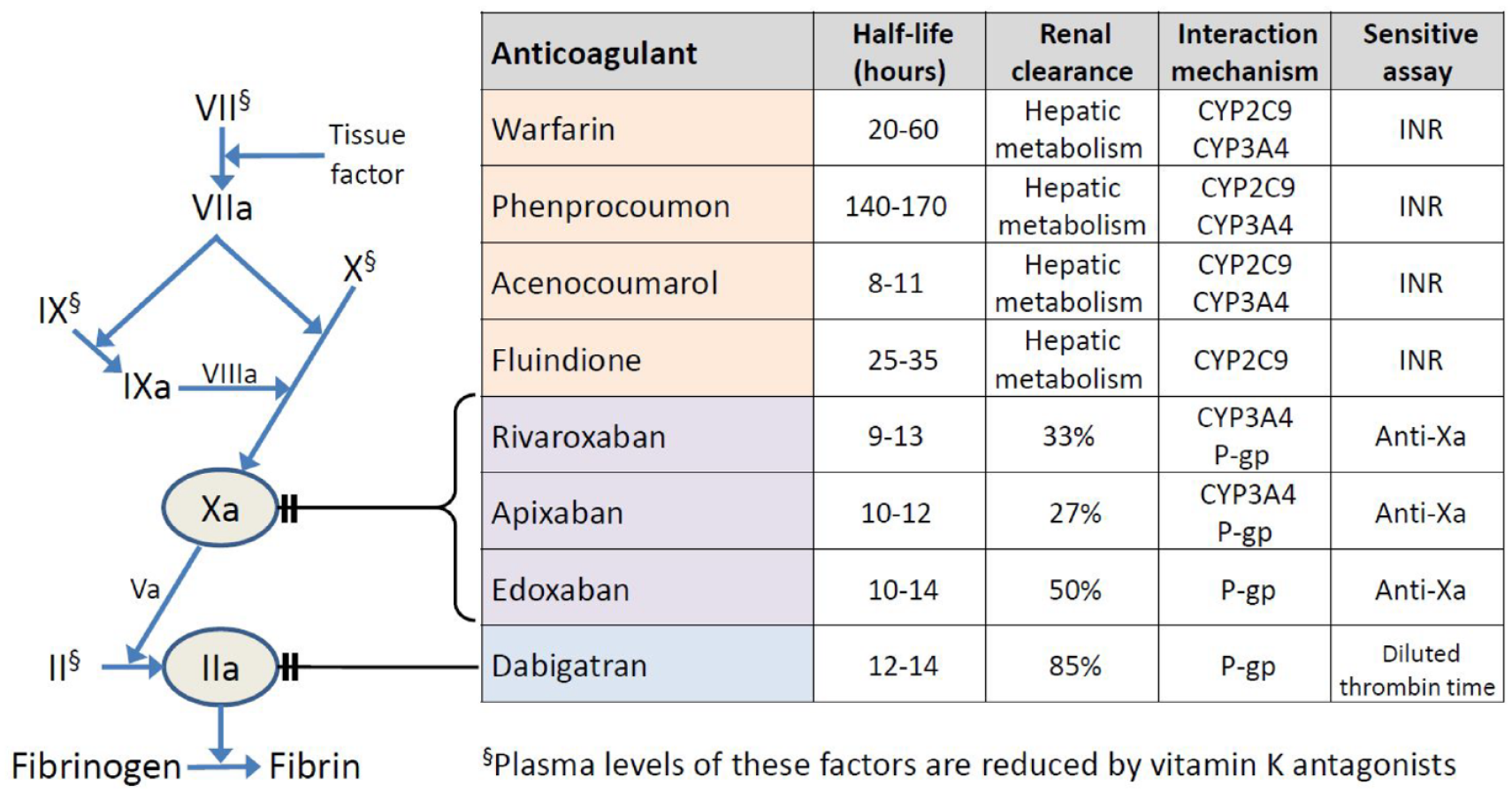
Mode of action and characteristics of oral anticoagulants.
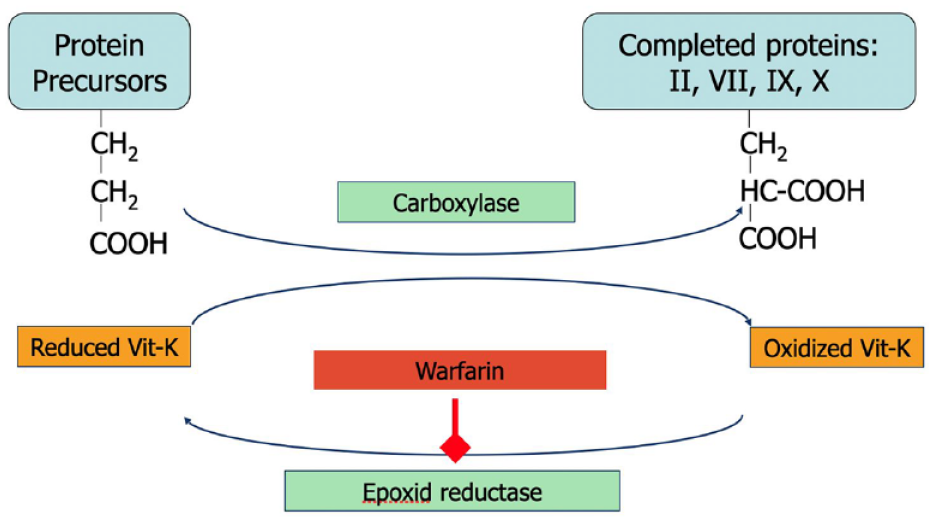
Vitamin-K and action of vitamin-K-antagonists (example: warfarin).
Dabigatran etexilate is a prodrug which after oral administration is converted in the liver to its active form dabigatran. Dabigatran is a small molecule that binds reversibly to the active site in both free and fibrin-bound thrombin, whereby fibrin formation is inhibited. 12 When dabigatran etexilate was launched in 2008, there was no specific antidote for reversing the anticoagulant effect. The half-life of dabigatran is 12–14 h and even longer in patients with impaired renal function. In case of life-threatening bleeding, pausing the treatment is therefore not sufficient. The anticoagulant effect of dabigatran can be antagonized by infusion of concentrate of coagulation factors in non-activated form (PCC) or activated form (FEIBA, eptacog alfa), but this may lead to hypercoagulation and a high risk of thrombosis.13,14 Idarucizumab is a specific, neutralizing, monoclonal antibody fragment, which adheres to the thrombin-binding site of dabigatran with a binding affinity which is approximately 350 times higher than dabigatran to thrombin. Intravenous infusion of idarucizumab 5 g completely reverses the anticoagulant effect of dabigatran within minutes with high affinity and specificity.15,16 The REVERSE-AD study 15 was a prospective case series (n = 504) that evaluated the effect of idarucizumab (praxbind®) in patients with cerebral hemorrhage on the thrombin inhibitor dabigatran (Pradaxa®) (subdural n = 39 [13.0%], sub-arachnoid n = 26 [8.6%], intracerebral n = 53 [17.6%]. 1 Idarucizumab (two times 2.5 g i.v.) was given either in patients with major bleeding (n = 301, of which n = 98 with ICB) or as an emergency procedure (n = 202) when the study physicians saw the need to neutralize the effect of dabigatran to prevent secondary bleeding or to allow emergency procedures to be performed. The median time of the last intake of dabigatran before idarucizumab was 15.5 h. The primary endpoint was the maximum percentage cancelation of the effect of dabigatran within 4 h of administration of idarucizumab and was determined by the coagulation parameters dTT (diluted thrombin time) and ECT (ecarin clotting time). In all patients, dTT and ECT were normalized after idaruzumab administration. No serious side effects were observed. Within the first 30 days, thromboembolic events occurred in 4.8% (24 of 503) of the patients, which, in conjunction with the mode of action, indicates no intrinsic prothrombotic effect. Therapy with idarucizumab has been approved worldwide since 2016. Idarucizumab is used in some centers to reverse the effects of dabigatran before providing thrombolytic therapy to dabigatran treated patients with ischemic stroke. 17
Direct oral factor Xa inhibitors cause a dose-dependent inactivation of free and clot-associated factor Xa. 18 When the factor Xa inhibitors were introduced, no specific antidote was available. As with dabigatran, it was therefore recommended to use agents for inducing hypercoagulation in case of major and potentially life-threatening bleeding.14,19 Andexanet alfa, marketed in 2019, is a recombinant truncated form of endogenous factor Xa without the ability to activate prothrombin, but with high affinity for factor Xa inhibitors. 20 As it has a short pharmacodynamic half-life of about 1 h, it is given as an intravenous infusion over 2 h. After cessation of the infusion, anti-FXa activity rapidly returns to the same level as after normal elimination of the FXa inhibitor. 21 ANNEXA-I was a phase four randomized clinical trial of andexanet alfa for injection in patients with acute intracranial hemorrhage associated receiving an oral factor Xa inhibiton. Inclusion criteria are: age > 17 years, imaging confirmed (CCT or MRI) ICH < 2 hours prior to ramdomisation, ICH volume ⩾ 0.5 to ⩽60 mL, treatment with apixaban, rivaroxaban, or edoxaban, last intake < 15 hours or if > 15 hours or unknown time of last dose, if documented anti FXa activity is > 100 ng/mL for direct fXa inhibitors ⩽ 35 at the time of consent. 22 A press release 23 from Astra-Zeneca reported that the ANNEXA-I trial has met its efficacy endpoint at the planned interim analysis, showing improved control of bleeding with targeted anticoagulation reversal, compared to usual care; patient related endpoints including functional outcome or death will be covered in the planned scientific publication.as well as further information on the reported risk of thromboembolic events after treatment with the FXa inhibitor, a high.20,24,25
Epidemiology of OAC-ICH
Since introducing the direct oral anticoagulants (DOACs), the overall use of OACs has increased by 30–50 %.2,26,27 This is partially explained by the increasing age of the general population, 28 leading to an increased number of atrial fibrillation patients,29–31 but also by the ease of use and better safety profile of the DOACs.32–36 In the pivotal trials examining the efficacy and safety of DOACs in atrial fibrillation patients, haemorrhagic stroke occurred in 0.44% of patients on DOAC annually versus 0.9% vitamin K-antagonist-treated. 37 Following the increased use of OAC, numbers of ICHs related to OAC2,3 also increased and in an unselected, catchment area defined Danish cohort, patients with OAC-ICH now constitute a fifth of the ICH patients.
For patients who experience an OAC-related ICH (OAC-ICH) prognosis is poor: In the RE-LY trial, the 30-day mortality was 52% in participants with non-traumatic ICH, 38 in ROCKET-AF 53% 39 for Rivaroxaban and 59% for warfarin, and in ARISTOTLE 45,4% for Apixaban and 42,6% for Warfarin. 40 However, these reports on outcomes present very inhomogeneous results, largely depending on the follow-up time (from 24 h to 3 months), the need for patients consent before study inclusion, as well as the type of study population (e.g. tertiary hospital admissions vs overall admissions/ registry-based data), Table 1. However, the reported level of mortality in the pivotal trials are comparable to reports based on unselected patients in the Swedish Stroke Registry. 41
Reported outcomes after OAC-ICH.
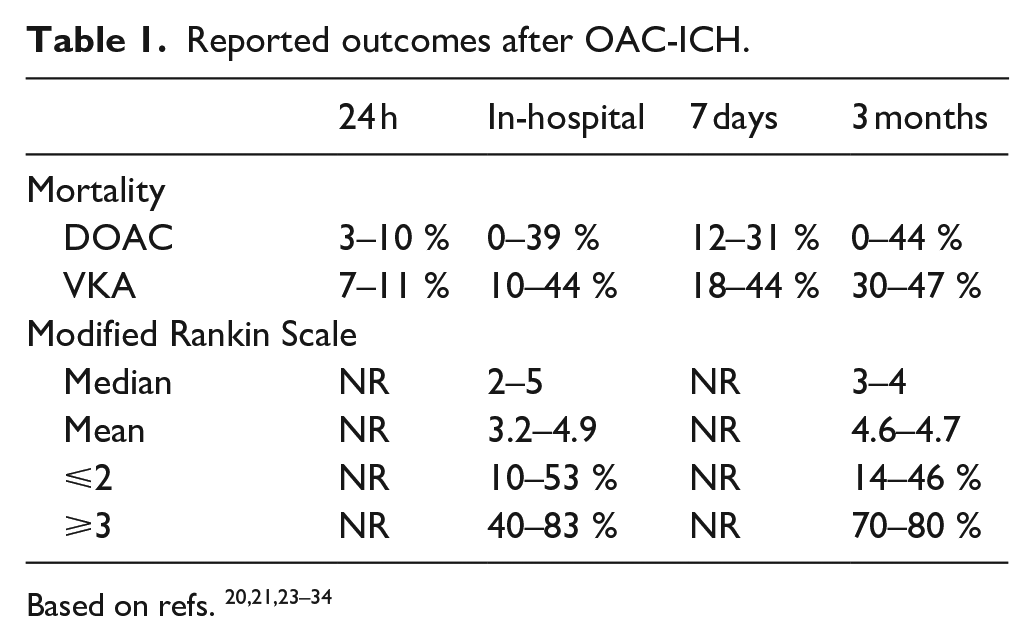
When comparing patients with DOAC-ICH to patients with VKA-ICH, a meta-analysis reported that DOAC was related to a reduced hematoma volume (weighted mean difference of −9.59 (95% confidence interval: −15.33; −3.85)) and lower mortality at discharge (relative risk of 0.82 (95% confidence interval: 0.71; 0.96)), with no significant differences in hematoma expansion, unfavorable functional outcome or mortality at 3 months. The lower mortality rate at discharge, was not significant in any of the individual studies, only in the meta-analysis. 42 However, one large study was not included 43 and another large study has been published since. 44 Other meta-analyses found similar outcomes for patients with DOAC-ICH versus VKA-ICH.
The main differences between women and men resemble those observed in stroke patients in general. Women are older than men at the time of ICH, have slightly worse pre-stroke functional level, while men are more likely smokers and have a higher alcohol consumption. 45 Women with OAC-ICH may be less likely to receive reversal agents, and more likely to receive a do not resuscitate within the first week. 45
Pathophysiology of OAC-ICH
Despite recent advances, our knowledge about pathophysiological mechanisms, clinico-radiological predictors, and long-term prognosis of OAC-ICH is still limited. Indeed, in patients with OAC-ICH many competing risk factors co-exist, and the independent contribution of OAC has not yet been elucidated.46–48 Previous ICH etiological classification systems included anticoagulation as a mechanistic potential ICH cause,49,50 however, up to date, there is no evidence showing that OAC can directly increase vascular fragility or cause vascular rupture.51,52 Accordingly, most OAC-ICH cases occur within VKA therapeutic range,53,54 and available literature on DOAC-ICHs found consistent results.55,56 Moreover, clinico-radiological characteristics of OAC-ICH are similar as ICH without prior anticoagulation, supporting the hypothesis that OAC likely acts as a risk or exacerbating factor.48,57–60 In this context, OAC intake should neither be regarded itself as the direct cause of bleeding nor preclude the diagnostic etiological work-up for underlying cerebral small vessel disease (SVD), that is, arteriolosclerosis, also called lypohialinosis or deep perforating arteriopathy, and sporadic cerebral amyloid angiopathy (CAA).61,62
Main clinical identified predictors of OAC-ICH include increasing age, arterial hypertension, diabetes, prior stroke, higher intensity and lability of anticoagulation, antiplatelet use in addition to anticoagulation. 60 Genetic risk scores were also recently shown to improve ICH risk stratification for patients with OAC intake.60,63,64 Type of OAC also matters, as patients with DOACs have a 50% reduction in ICH risk compared to VKA.37,65–67
From a radiological point of view, presence, burden, and location of SVD biomarkers have a pivotal clinical relevance in OAC-ICH, for both supporting the diagnosis of the subtype of underlying microangiopathy (with deep perforating arteriopathy and CAA often co-existing) and for future ICH risk stratification. High global neuroimaging biomarker burden, expressed by SVD scores, has been shown to be more prevalent in patients with OAC-ICH compared to patients without previous OAC. 48 Moreover, presence and burden of individual MRI SVD biomarkers have been consistently associated with increased VKA- and DOAC-ICH risk, both haemorrhagic ones, in particular cerebral microbleeds (CMB) in lobar location and cortical superficial siderosis (CSS), and those of presumed ischemic nature, such as white matter hyperintensities (WMH) and basal ganglia enlarged perivascular space (EPVS),.60,68–72 Accordingly, in patients with AF taking OAC, clinical score implementation with radiological biomarkers (CMB and CSS) has been shown to improve haemorrhagic risk stratification at the individual level in patients without previous stroke.73–77
Despite that OAC is unlikely to trigger the bleeding, OAC-ICH is associated with poorer prognosis, mainly because of a larger initial volume78,79 and a threefold increase in ICH expansion rate. 80 Data concerning clinico-radiological differences between DOAC-ICH versus non-OAC ICH and differences in long-term functional outcomes in patients with VKA-ICH versus DOAC-ICH are still lacking. However, recent studies comparing VKA-ICH with DOAC-ICH showed that ICHs occurring on DOACs have milder initial clinico-radiological severity than on VKA, with a lower risk of expansion.4,44,59,81–85 Indeed, hematoma expansion (HE) results from persistent bleeding along the ruptured perforating artery and secondary extravasation sites in the surrounding branches.67,86 OAC intake is likely to sustain HE over time through the persistent coagulopathy, and together with other mechanisms, including increased vessel-tissue pressure gradient and shear forces, it promotes diseased adjacent vessel rupture. In this context, arterial hypertension is likely to further increase vessel-tissue pressure gradient, worsening HE rates especially in patients with OAC intake. Consistently with this hypothesis, arterial hypertension during the acute ICH phase worsens ICH prognosis in patients with previous OAC intake, strengthen the hypothesis that a bundle of care, including both rapid blood pressure lowering, and anticoagulant reversal could improve clinical impact of future clinical trials of OAC reversal agents.87,88
Clinical presentation and complications
Spontaneous ICH in the presence of anticoagulation is often devastating coupled with hematoma expansion, ensuing rise in intracranial pressure and/or IVH. Clinical signs are typical of stroke with sudden onset of focal neurological impairment, for example, varying degrees of hemiparesis, hemisensory defect, aphasia and/or ataxia. General signs of cerebral affection are common including nausea, vomiting, headache, and lowered level of consciousness. Thunderclap headache is not typical in ICH and should raise suspicion of aneurysmal SAH. It should be noted that aneurysmal bleeds can expand into brain tissue and thus imitate a spontaneous ICH on radiological examination. Early deterioration is common in severe ICH and patients may already be drowsy or comatose at hospital arrival. With raised intracranial pressure but also depending on localization, cranial nerve signs such as forced gaze deviation and defect pupillary reflexes are typical clinical signs.
Hematoma expansion typically occurs early and is most often seen in the first 2 h after the ictus.89,90 HE is more common in larger hematomas 80 in particular with volumes of at least 6 mL. It is not entirely clear why hematomas attain a certain volume, but local tissue properties likely play a role as well as size of vascular defect, physiological repair mechanisms including platelet function and coagulant factors. Blood pressure is thought to be an important factor for development of HE. Patients on anticoagulant or antiplatelet therapy have higher risks of large hematoma volume 80 which indicates a role for early vascular repair mechanisms and prognosis is similar in these groups. 41 Based on propensity-score adjusted analyses reduced HE (7 mL) was reported in patients treated with andexanet alfa in comparison to patients given usual care only, 91 this suggest that reversal may attenuate HE. IVH may occur regardless of hematoma size and is an independent predictor of bad outcome.
Rapid neurological deterioration, in particular lowered level of consciousness should always raise the possibility of ongoing HE. Acute radiological examination is key in stroke and apart from confirmation of a diagnosis of ICH there are radiological signs which suggest that HE is likely. Simple measurement of size (A + B+C)/2 (with C being the product of the number of slices and slice thickness) will provide an indication with likelihood of HE being low in hematoma volumes below 6 mL and conversely higher and rising with larger volumes up to approximately 60 mL where the risk for HE seems to reach a plateau. 80 Presence and expansion of intraventricular hemorrhage (ICH) is an independent predictor of higher risk of mortality. 92 As such IVH it should be integrated in the definition of HE. 93 With a contrast CT, small extravasations of contrast within or close to the hematoma often described as “spot sign” have been described to predict HE. 94 The swirl sign, which is seen within the hematoma with a non-contrast CT predicts bad outcome 95 and has also been coupled with a high risk of HE. 96 Imaging biomarkers on native CCT such as the “Black Hole Sign,” “Heterogeneous Density Sign,” “Blend Sign,” “Hypodensity Sign,” “Irregular Shape Sign,” or “Island Sign” may also indicate continued active bleeding. 97 This should prompt performing a CTA or CMRA. It is however important to note that the absence of these signs does not predict a benign course with any certainty. In summary, presence of anticoagulation, early imaging preferably within 2 h of ictus and large hematoma size are simple and reliable predictors of HE.
Principles of interventions
The primary goal of early intervention following OAC-ICH is to prevent HE. Coagulopathy reversal and blood pressure management are the key components of acute therapy. HE is frequent in OAC-ICH and a meta-analysis of 77 studies and 5425 patients found that anticoagulation leads to a 3.5-fold increased odds of massive ICH expansion, even after controlling for confounding factors such as initial ICH volume, time from ictus to imaging and concomitant antiplatelet therapy. 80 Furthermore, anticoagulation has been associated with higher mortality rates and worse outcomes among ICH patients,53,98,99 while, conversely, antithrombotic reversal is associated with higher chances of survival in some observational reports.100,101
Generally, all patients with anticoagulant-related intracranial hemorrhage should receive a reversal agent urgently, irrespective of the original indication for anticoagulation (e.g. patients with mechanical heart valves, mechanical cardiac assist devices, venous thromboembolism, or atrial fibrillation should all be considered similarly for reversal), or the size or location of hemorrhage (e.g. subdural, subarachnoid or intraparenchymal compartment)14,102, Figure 3. This maxim is based on data demonstrating that: (1) ICH expansion risk is highest in the first few hours after initial haemorrhage,80,89 (2) rapid reversal has been associated with significantly reduced risk of hemorrhage expansion,10,98,103 (3) even small hemorrhages can expand massively; (4) small haemorrhagic expansion can be catastrophic in eloquent areas; and (5) the risk of early, acute recurrent hemorrhage is substantially higher than the risk of acute ischemic or thromboembolic events that could occur with reversal.103–108 Exceptions to the principle of early, rapid reversal, include patients with ICH due to cerebral sinus thrombosis (in which case anticoagulation is the preferred treatment), or patients with severe concomitant acute life-threatening thrombosis. In the later circumstance, the risks and benefits of reversal should be weighed on a case-by-case basis and discussed with the patient/family.
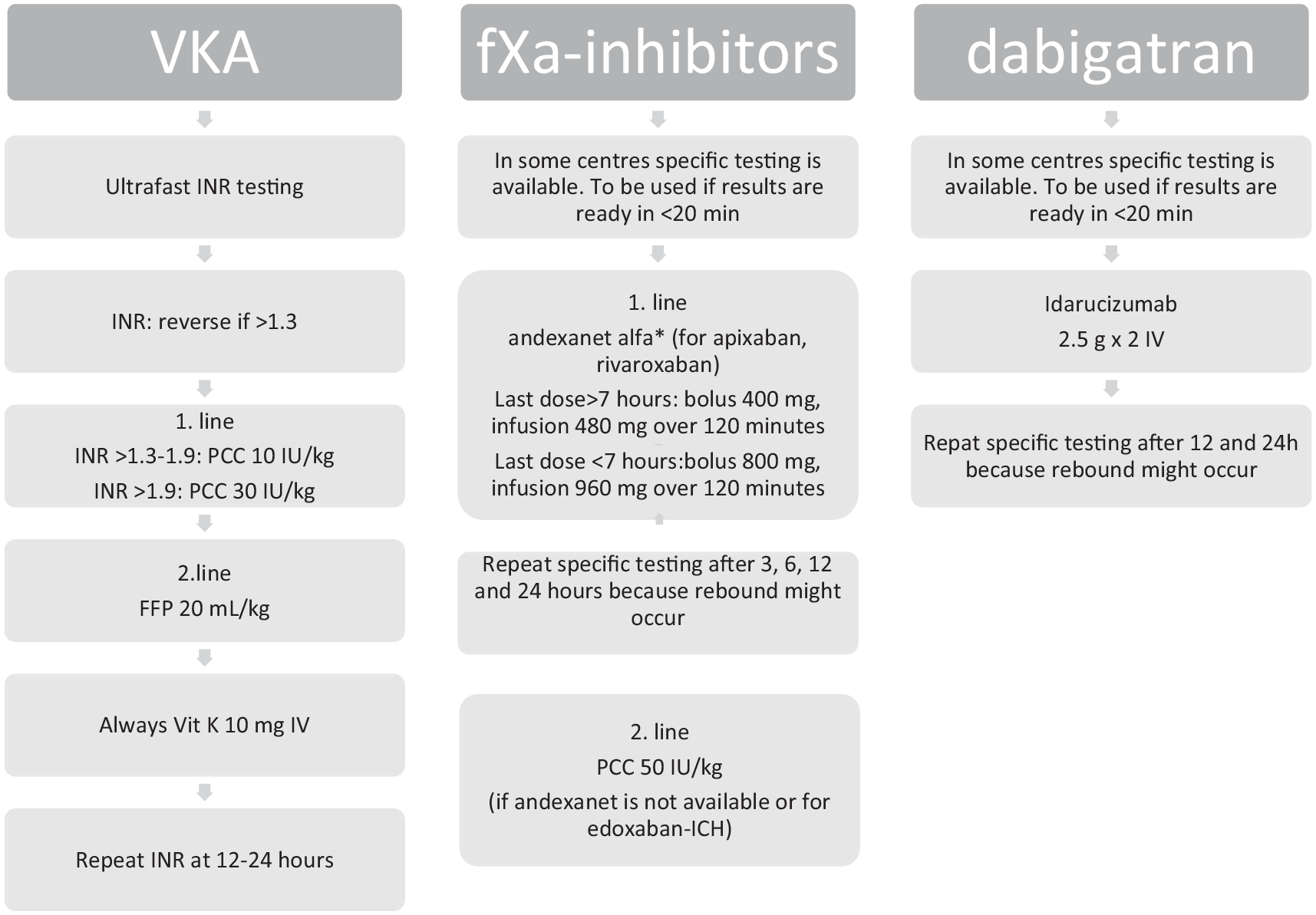
Flowchart from ’ European Stroke Organisation Guideline on Reversal of Oral Anticoagulants in acute Intracerebral Hemorrhage. Christensen et al. ESJ 2019. 14
Acute blood pressure management must be part of early bundled care for ICH patients, in addition to coagulopathy reversal. Indeed, achieving a systolic blood pressure (SBP) <160 mmHg within 4 h of admission in addition to anticoagulation reversal, was associated with significantly lower rates of ICH expansion, compared to INR correction alone, among patients with warfarin-associated ICH. 88 . The American Heart Association/American Stroke Association guidelines have suggested a SBP target of 130–150 mmHg, 61 while the European Stroke Association recommends a SBP target of 110–140 mmHg 109 within 6 h of ICH. Though the targets may differ slightly, it is important to note that there is a lower threshold for SBP that should be respected, and overly aggressive drops in SBP should be avoided. The pre-planned patient-level meta-analysis of INTERACT2 and ATACH-II found that early and stable SBP lowering (mean achieved SBP 147 mmHg, mean blood pressure reduction of 29 mmHg) was safe and associated with significantly better 90-day mRS scores. 110
The Intensive Care Bundle with Blood Pressure Reduction in Acute Cerebral Hemorrhage Trial (INTERACT3) was an international, multicentre, blinded endpoint, stepped wedge cluster randomized controlled trial conducted in nine low- and middle-income countries. The INTERACT3 trial studied the effect of a care bundle consisting of correction of systolic blood pressure (SBP < 140mmHg and > 130 mmHg), hyper- or hypoglycemia, pyrexia (temperature < 37.5 C°), and coagulopathy (PCC and or FFP incase of an INR > 1.5) in patient with an acute intracerebral hemorrhage (ICH) compared to usual standard of care did improve functional outcome. 111 The primary endpoint – the modified Rankin Scale score (mRS) at 6 months – was significantly better in the care bundle group (common odds ratio (OR) 0.86 (95% CI 0.76–0.97; p = 0.015). Interestingly, the difference in reduction of mean hematoma expansion between both groups was very small (0.22 mL), and there was no significant influence from neither glucose nor temperature change. However, only very few patients with OAC-ICH were included:0.8% in intervention group and 1,6% in control group.
Conclusion
Based on the well justified increasing use of OAC the numbers of patients presenting with OAC-ICH have been growing. Consequently, admission of these patients is not infrequent in everyday clinical practice and knowledge on best treatment and care is crucial both for neurologists, neurosurgeons and emergency physicians. Presently, evidence confirming that reversal of OAC improves prognosis is not yet existing, but it is reasonable to assume that early reversal reduces hematoma expansion. Further, blood pressure lowering remains a corner stone in prevention/reduction of HE. Finally, the importance of supportive stroke unit care to prevent further complications cannot be underestimated. Better knowledge based on randomized controlled trials and observational studies in unselected patients is needed to be able to make strong recommendations on acute therapy as well as to describe the clinical course and prognosis with precision. Further, fXIa inhibitors – a new generation of OACs – have reached phase three trials with data supporting a lower bleeding risk.
Footnotes
Acknowledgements
We would like to thank David Bach-Nielsen, PA to HC for technical assistance with the MS.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HC is National Lead and Steering group member in ANNEXA-I trial (Astra-Zeneca). TS has received consultancy fees from Bayer, BMS, Pfizer, Boehringer Ingelheim, Bayer, Daiichy Sankyo, and Octapharma. JP is Local PI for the FAST trial NCT00127283. JF is local PI for ANNEXA-I (Astra Zeneca) and ASPIRE (NIH) and SATURN (NIH). JG, BC, NJD has declared no conflicting interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
NA
Informed consent
NA
Guarantor
HC
Contributorship
HC and TS drafted the outline of the review, BC, JF, JDN, JP, JG, TS and HC drafted the sections of the review. All authors commented on the MS in two rounds.
