Abstract
Purpose:
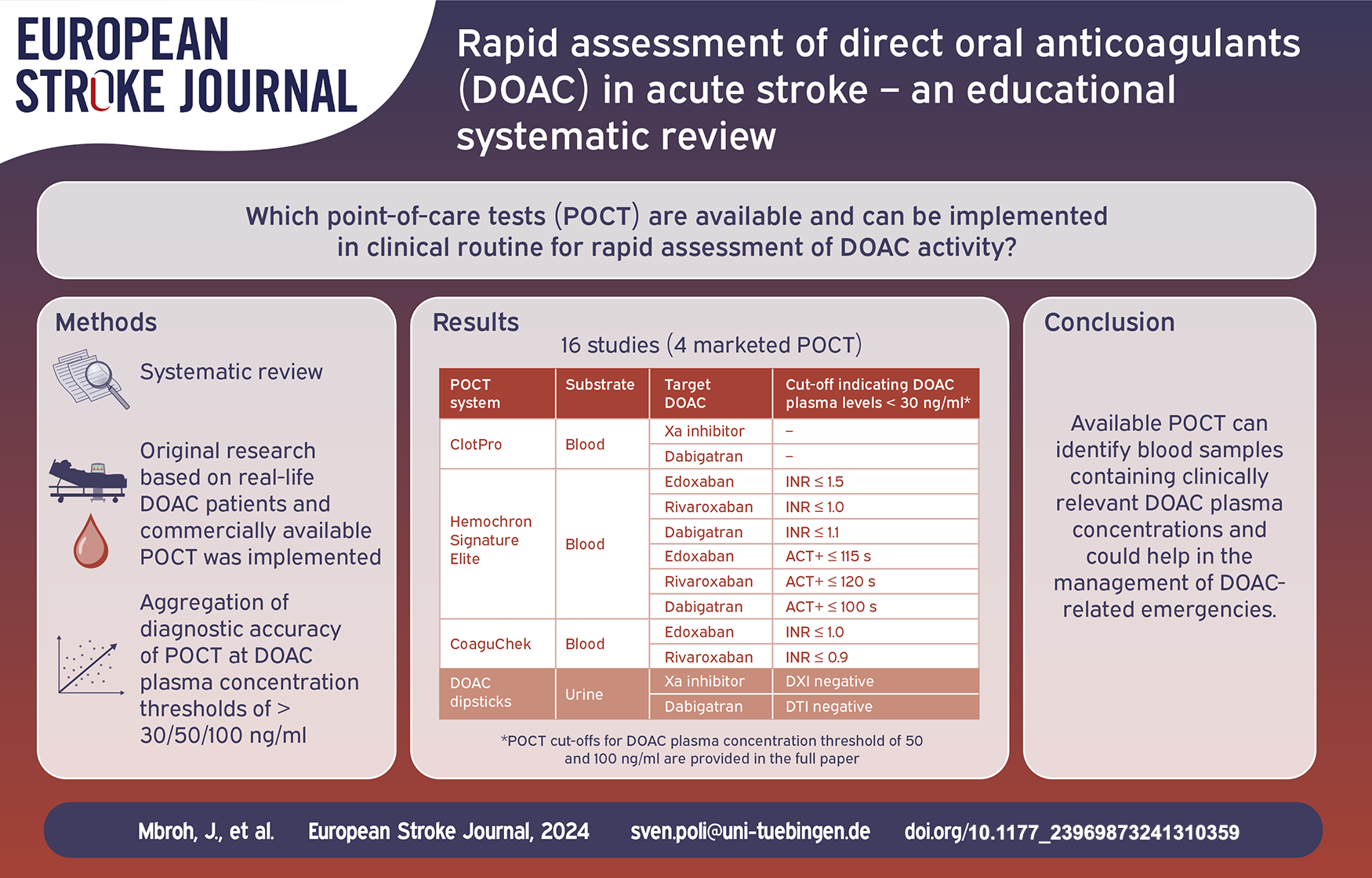
To provide an overview on commercially available point-of-care tests (POCT) for rapid assessment of direct oral anticoagulant (DOAC) activity, their diagnostic accuracy in identifying blood samples containing clinically relevant DOAC concentrations, and guidance on selecting the appropriate POCT system/assay for decision-making in emergencies.
Methods:
Systematic review with PubMed and Cochrane library search for published studies until September 6, 2024. Papers were included if in English, original research using blood samples from DOAC patients and POCT was implemented. Two reviewers assessed studies for eligibility and quality. Data from studies were aggregated and diagnostic accuracy at DOAC plasma thresholds of 30/50/100 ng/ml was recalculated.
Findings:
16 studies involving four commercially available POCT systems/assays were identified. Eleven studies evaluated blood-based POCT and five used urine for indirect estimation of DOAC plasma levels.
Discussion:
DOAC Dipsticks do not require knowledge about the taken DOAC, but measure urine and, due to low specificity, require blood testing in case of positive results. Hemochron Signature Elite (HSE) and CoaguChek use global coagulation assays and provide high sensitivity for edoxaban and rivaroxaban, HSE additionally for dabigatran. Due to insufficient correlation, both POCT are not recommended for apixaban, CoaguChek also not for dabigatran. The thromboelastometric ClotPro performs well for all DOAC, does not require knowledge about the DOAC, but was taken off the market recently.
Conclusion:
Studies have shown feasibility of POCT in identifying blood samples containing clinically relevant DOAC plasma concentrations. Each POCT device has its own unique limitations. Knowledge about the POCT assays, time since last intake and type of DOAC enhances confidence in making appropriate therapeutic decisions. We propose an algorithm that could help guiding physicians in selecting an appropriate POCT system/assay in DOAC-related emergencies.
Keywords
Introduction
The global prevalence of atrial fibrillation was 60 million people in 2019, 1 and is expected to increase exponentially over the next decades due to extended life expectancy and associated increase of vascular risk factors. 2 This increase translates to more and more patients receiving direct oral anticoagulants (DOAC), which are the antithrombotic treatment of choice to prevent atrial fibrillation-associated cardioembolism including stroke.3–6 Prescriptions of DOAC, that is, of the factor Xa inhibitors apixaban, edoxaban or rivaroxaban and of the thrombin inhibitor dabigatran, increased by 416% from 2015 to 2020. 7 Despite DOAC therapy, around 1% of atrial fibrillation patients will suffer ischemic stroke per year,3–6 and up to 3.6% of DOAC-treated atrial fibrillation patients will develop a major bleeding per year and up to 0.5% an intracranial hemorrhage.3–6
To identify patients who qualify for thrombolysis and/or anticoagulation reversal, a timely assessment of the DOAC activity would be advantageous because relevant anticoagulation may not be present due to DOAC’s pharmacokinetic properties with plasma concentrations changing significantly throughout the day. 8 Importantly, no residual anticoagulation is found in up to one third of DOAC emergency patients.8–10 Reversal therapy should be avoided in these latter cases even in the most critical intracranial hemorrhage since unnecessary anticoagulation reversal not only comes at considerable financial costs but, more importantly, in case of andexanet alfa and prothrombin complex, additionally with potentially lethal thromboembolic complications.10–13 On the other hand, in acute ischemic stroke, if relevant anticoagulant activity cannot be ruled out by coagulation testing and DOAC intake is assumed within the last 48 h, applying international guideline recommendations would lead to withholding thrombolysis in up to 28% of otherwise eligible patients. 14
Although ultra-performance liquid chromatography-tandem mass spectrometry represents the gold standard for DOAC assessment, this method is time-consuming and not readily available rendering it unsuitable for emergency decision-making. 15 The widely implemented coagulation testing in DOAC patients in clinical routine is the quantitative assessment of DOAC plasma levels using laboratory assays that are calibrated with specific DOAC calibrators.16,17 These assays measure the anti-Xa activity in case of factor Xa inhibitors or the ecarin clotting time, diluted thrombin time, or direct anti-thrombin activity, if dabigatran was applied.16,17 Importantly, diagnostic accuracy of these assays critically varies at low plasma concentrations.16,17 Furthermore, availability of DOAC specific coagulation assays is still limited to selected laboratories. 18 The more widely available anti-factor Xa activity assays that are calibrated to unfractionated or low-molecular weight heparins could be an alternative when it comes to identify blood samples containing low but relevant concentrations of a direct oral factor Xa inhibitor.16,18 Despite workflow optimization, the turn-around times associated with any laboratory-based coagulation testing of 30 to 60 min due to sample collection, transport, and centrifugation are inconvenient in the case of acute ischemic or hemorrhagic stroke where time is brain. 19
Rapid bedside point-of-care coagulation testing (POCT) of the prothrombin time (PT) and the derived international normalized ratio is nowadays well established for guiding decision-making in vitamin K antagonist-related emergencies and was shown to reduce admission to anticoagulation reversal or thrombolysis times by more than 20 min compared to a laboratory-based approach for confirmation or exclusion of relevant anticoaguation.20,21
Global coagulation tests such as the PT or the activated partial thromboplastin time (aPTT) may be prolonged in the presence of DOAC, and a test result above the normal range could indicate a clinically relevant DOAC plasma concentration.22,23 However, the reverse conclusion—that a “normal” PT or aPTT excludes relevant DOAC plasma concentrations—is not valid, even with the use of a DOAC-sensitive assay. Only the establishment and application of assay-specific cut-offs (typically below the respective test’s upper limit of normal) allow for the reliable exclusion of even low DOAC plasma levels.23,24
International guidelines suggest DOAC plasma concentrations of >30 ng/ml to provide clinically relevant anticoagulant effects.25,26 Plasma concentrations below 30 ng/ml are generally considered safe for thrombolysis or surgery and to withhold reversal therapy in case of intracranial hemorrhage and life-threatening bleeding.25,26 Additionally, 50 ng/ml have been suggested in case of a high benefit-risk ratio, surgery with a low bleeding risk, and less critical bleeding.26,27 Observational thrombolysis in acute ischemic stroke studies however indicate that even higher DOAC plasma concentrations of up to 100 ng/ml could be safe, or even completely reject the necessity of coagulation assessment for thrombolysis decision-making. 14 However, none of these thresholds have been supported by randomized clinical trials.
By means of qualitative, dichotomous assessment (i.e., is the DOAC plasma concentration above or below the currently recommended thresholds of 30/50/100 ng/ml), POCT could guide emergency treatment in DOAC-treated patients (i.e., can thrombolysis or surgery be initiated or should a reversal agent be administered in case of life-threatening bleeding?). 28
This systematic study therefore aims at
(1) identifying commercially available POCT systems that have been evaluated using real-life biosamples from DOAC patients to directly or indirectly rule out DOAC concentrations in blood samples that are currently considered relevant for emergency decision-making, that is, 30/50/100 ng/ml,
(2) providing information on their diagnostic accuracy to identify blood samples containing DOAC plasma concentrations >30/50/100 ng/ml and
(3) suggesting algorithms on how these could be implemented in clinical routine.
Methods
This study was conducted in accordance with the Preferred Reporting Item for Systematic Reviews and Meta-Analysis (PRISMA). 29 We conducted a full text database search in PubMed and Cochrane library for published studies until September 6, 2024, which evaluated commercially available POCT for assessment of DOAC (see Supplemental Methods). The PubMed search strategy was adapted for use in Cochrane library. Studies were included in the systematic review if they met the following criteria: publication in English, original studies based on real-life patients under DOAC and point of care testing was implemented. Outcomes of the search are summarized in Figure 1 and Supplementary Table S1. Quality assessment by means of Risk of Bias Assessment tool for Non-randomized Studies (RoBANS) was performed to assess the methodological quality of all included studies under which studies were rated as having either a high, a low, or an uncertain risk of bias. 30 Two reviewers (JM and YW) screened and assessed the relevant studies for eligibility and disagreements were resolved through a third reviewer (JT).
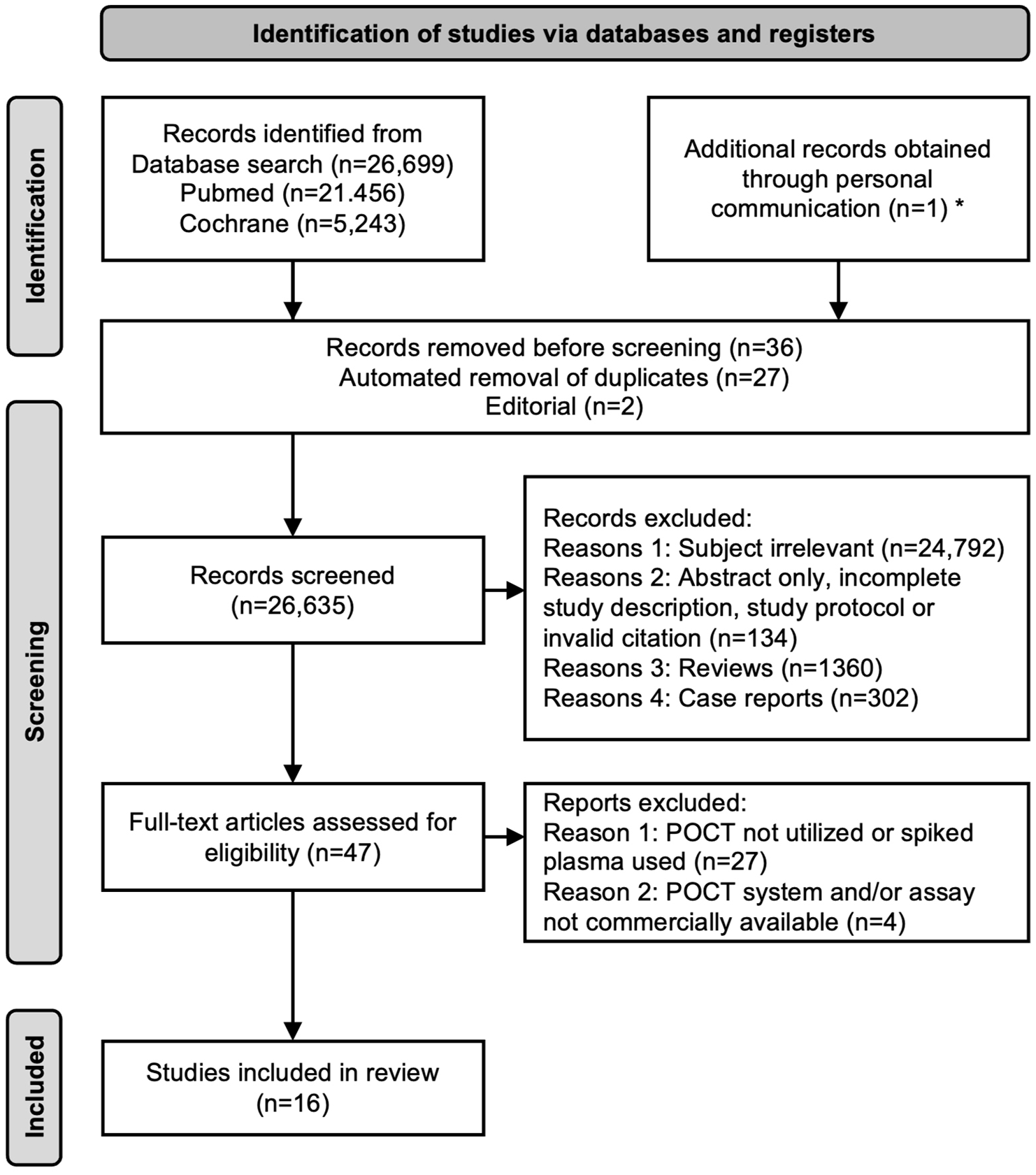
Preferred reporting items for systematic reviews and meta-analyses (PRISMAs) flow diagram.
Numbers of participants, numbers of samples, DOAC plasma concentrations of samples, true and false negatives, and true and false positives were extracted from studies if provided to recalculate diagnostic accuracy parameters at the DOAC plasma concentration thresholds of 30/50/100 ng/ml for each POCT and study and for pooled study data. For studies including negative samples from DOAC-naïve participants, diagnostic accuracy parameters were additionally recalculated without the latter to provide diagnostic insights that are more useful for the clinical question which is to identify DOAC patients whose plasma concentrations waned just below a treatment threshold rather than to identify those who never took the DOAC in question. Sensitivity was defined as the percentage of samples containing DOAC plasma concentrations above a threshold that were correctly identified by POCT. Specificity was defined as the percentage of samples containing DOAC plasma concentrations below a threshold that were correctly identified as such and, thus, as theoretically belonging to a patient eligible for immediate thrombolysis or surgery and without need for anticoagulation reversal. Positive (PPV) and negative predictive values (NPV) as well as positive (LR+, sensitivity/(1–specificity)) and negative likelihood ratios (LR–, (1–sensitivity)/specificity) were calculated. 95% confidence intervals (CI) for all proportions were calculated according to the efficient-score method as described by Newcombe 31 using the free online VassarStats Clinical Calculator 1. 32 SPSS version 29 (IBM, Armonk, NY, USA) was used for all other statistical analyses.
Results
The search yielded 26,699 studies including one extra study obtained through personal communication (Figure 1). 33 After removal of duplicates, screening and assessment for eligibility, 16 studies involving four commercially available POCT systems/assays (i.e., the blood-based Hemochron Signature Elite (Werfen, Barcelona, Spain), CoaguChek (Roche, Rotkreuz, Switzerland), the ClotPro (Haemonetics, Boston, Massachusetts, USA) and the urine-based DOAC Dipsticks (DOASENSE, Heidelberg, Germany)) were included in this systematic review (Supplementary Table S1).33−48 Data on risk of bias are shown in Supplementary Figure S1. Six33,39−43 of the eleven studies evaluating blood-based POCT and five34−38 using urine for indirect estimation of DOAC plasma levels provided data on diagnostic accuracy at the DOAC plasma concentration treatment thresholds 30/50/100 ng/ml. Diagnostic accuracy of these POCT systems/assays with pooled data of the five studies evaluating the DOAC Dipsticks is shown in Supplementary Tables S2 and S3. Diagnostic accuracy for the individual DOAC Dipstick studies is shown in Supplementary Tables S5 and S6.34–38 Underlying numbers of true/false positive/negative results are displayed in Supplementary Tables S4 and S7. An algorithm on how POCT could be integrated into the emergency workup is shown in Figure 2.
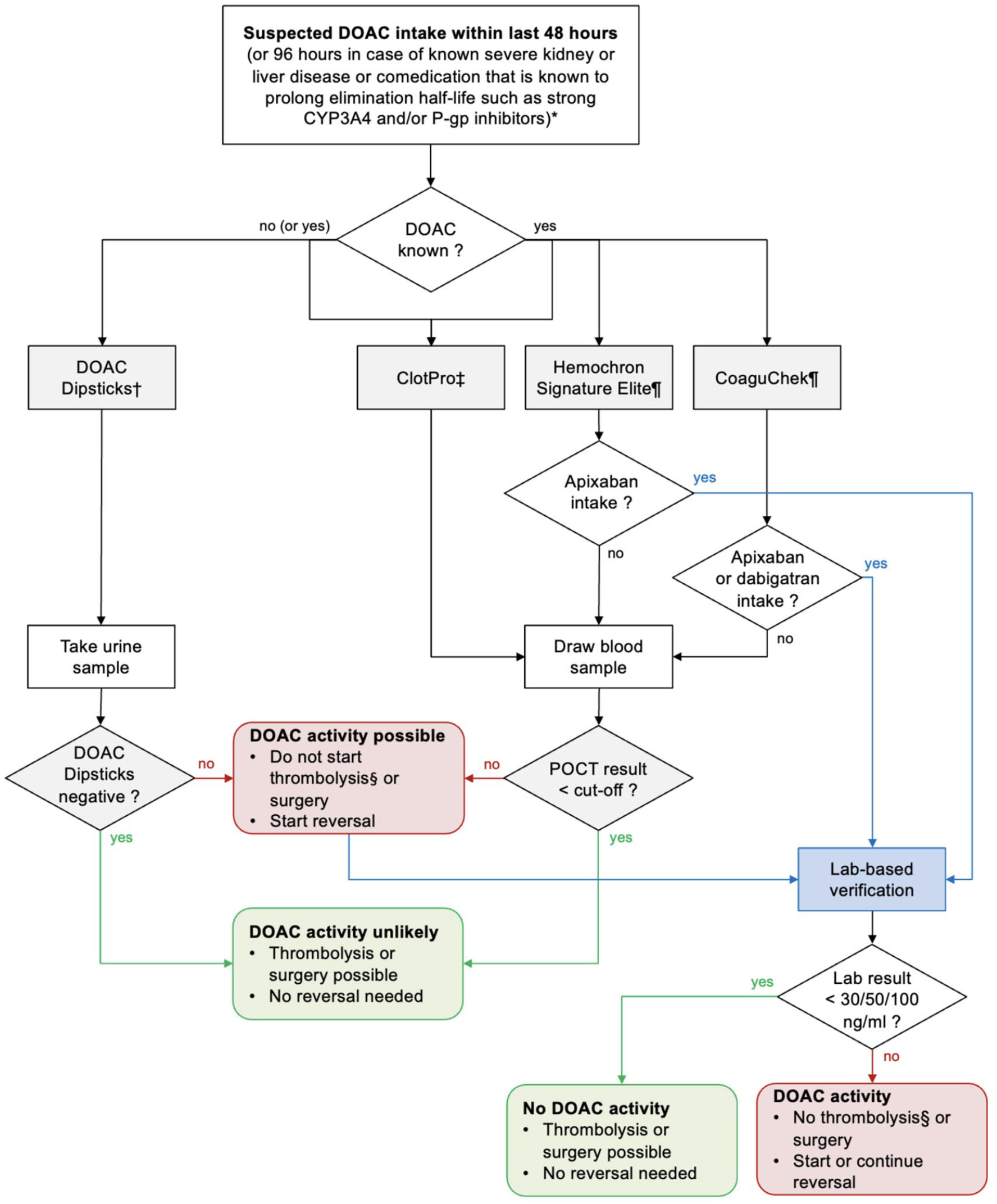
Algorithm to assess relevant direct oral anticoagulant (DOAC) activity in the blood using point-of-care testing (POCT).
Discussion
Urine-based point-of-care coagulation testing of direct oral anticoagulant activity
DOAC dipsticks
Approximately 30% to 85% of DOAC are excreted in urine due to their hydrophilic nature and their small molecular weight. The DOAC Dipsticks (DOASENSE, Heidelberg, Germany) were developed on the basis of its ability to detect traces of DOAC in urine. 50 They have two test pads for the detection of the presence of the direct oral thrombin inhibitor dabigatran and of direct oral factor Xa inhibitors. To confirm the validity of the test, two additional test pads indicate abnormal urine color and creatinine. 50 Hence, with the DOAC Dipsticks, DOAC detection is only based on the identification or exclusion of a DOAC in the urine and thus, any conclusions about the actual in vivo DOAC plasma concentration can only be drawn indirectly. Nonetheless, DOAC Dipsticks do not require knowledge about the taken DOAC and are so far the only commercially available POCT that is CE (Conformité Européenne)-certified for DOAC testing and has found its way into some national and international guidelines for ruling out clinically relevant DOAC levels in the surgical enviroment.51,52 Currently, the DOAC Dipsticks are not approved by the United States Food and Drug Administration (FDA). The pooled analysis of DOAC Dipstick studies indicates sensitivities of a positive urine test result between 95% and 100% for the detection of factor Xa inhibitor plasma concentrations >30 ng/ml34–38 and of 98% for the detection of dabigatran plasma concentrations >30 ng/ml (Supplementary Tables S2 and S3).34–36,38 Importantly, however, the individual DOAC Dipstick studies showed a great disparity in correctly identifying samples containing DOAC plasma concentrations of less than 30 ng/ml. Papageorgiou et al. only reported very low specificities. 37 However, specificities in the other studies on urine test-based identification of patients with low plasma concentrations ranged from low to very high.34–36,38 This might be due to the individual study design and the samples used as “controls” (i.e., samples below the investigated DOAC threshold): Overall, there was a very low number of included DOAC samples with plasma concentrations below the 30 ng/ml threshold, many of which tested false positive (Supplementary Table S7). Instead, the majority of control samples were acquired from patients naïve to the tested DOAC (i.e., from DOAC naïve patients, from dabigatran patients for testing factor Xa inhibitors, or from factor Xa inhibitor patients for testing dabigatran), most of which tested “true” negative and consequently enhanced NPV and specificity. Hence, metrics of test accuracy in some studies are artificially improved but cannot be translated into the clinical emergency setting in which the main objective is to differentiate patients with a low and clinically irrelevant residual DOAC activity from those with plasma concentrations above the 30 ng/ml threshold (compare recalculated test accuracy parameters with and without samples from patients naïve to the tested DOAC in Supplementary Tables S2 and S3).34–38,53 How urine testing with the DOAC Dipsticks performs at the 50 and 100 ng/ml DOAC plasma concentration thresholds has yet to be studied. Algorithms for the use of the DOAC dipsticks to guide thrombolysis or DOAC reversal therapy have been proposed, however, in case of a positive urine test result, only in conjunction with blood-based coagulation testing.49,53 Consequently, the high false positive rate, especially when analyzing urine from patients who actually took a DOAC, necessitates additional plasma-based coagulation testing in basically all positively tested cases, which represents a significant limitation of this POCT system. Moreover, urine sampling as compared to blood sampling may be a hurdle in the emergency setting contributing to a prolonged turn-around time.
Blood-based point-of-care coagulation testing of direct oral anticoagulant activity
Hemochron signature elite
The Hemochron Signature Elite POCT measures plasmatic coagulation within less than five minutes using a drop of whole blood without need for processing. Currently, test cartridges for estimating the PT, aPTT, activated clotting time low-range (ACT-LR) and high-range (ACT+) are commercially available. These test cards are not approved for DOAC assessment (off-label) and no DOAC-specific test cards exist. Studies using samples from real-life patients found a correlation between test results obtained with the Hemochron Signature Elite POCT and therapeutic dabigatran or rivaroxaban concentrations. The authors encountered a significant overlap between test results of samples with low DOAC plasma levels and samples obtained from healthy subjects containing no DOAC at all.46–48 However, based on qualitative testing, test cartridge-specific cut-offs which might be different from the respective assay’s intrinsic normal range have been established for each DOAC for the Hemochron Signature Elite POCT and all its commercially available test cartridges.40,43 Hemochron® Signature Elite POCT together with the PT and ACT+ test cartridges were shown to correlate with actual dabigatran, edoxaban and rivaroxaban concentrations.40,43 At a sensitivity of >95%, Hemochron Signature Elite POCT could detect dabigatran, edoxaban and rivaroxaban plasma levels >30/50/100 ng/ml (Supplementary Tables S2 and S3).40,43 These data show a corresponding high specificity for edoxaban and moderate specificities for rivaroxaban and dabigatran at these thresholds. None of the Hemochron Signature Elite POCT assays was capable of safely identifying samples with clinically relevant apixaban levels (Supplementary Tables S2 and S3).40,43 As a guide for emergency physicians for the rapid assessment of anticoagulation status in DOAC patients, the authors have proposed primarily the PT cartridge of the Hemochron Signature Elite POCT for the qualitative assessment of dabigatran and edoxaban and the ACT+ cartridge for the qualitative assessment of rivaroxaban.40,43 Although cartridges may also be used vice versa, interpretation of results requires knowledge about the taken DOAC.
CoaguChek
The CoaguChek POCT similarly measures plasmatic coagulation within few minutes using a drop of whole blood without need for any processing. No specific test stripe for DOAC assessment exists for the device. Although not approved for DOAC testing (off-label), the commercially available global plasmatic coagulation test stripes (PT and aPTT) have been evaluated with respect to DOAC.33,41,42,46,48 The CoaguChek POCT has been tested in elective settings providing the basis for emergency decision making using test stripe- and DOAC-specific cut-offs.33,41,42 CoaguChek PT and aPTT test stripes could detect edoxaban and rivaroxaban plasma concentrations >30/50/100 ng/ml with a sensitivity of >95%, indicating high reliability of CoaguChek POCT-based patient selection.33,41,42 The specificities of CoaguChek PT and aPTT test results at the respective thresholds however differ considerably for both edoxaban and rivaroxaban between the studies (i.e., ranging from 12% to 86% for the PT test stripe and 23% to 84% for the aPTT test stripe) (Supplementary Tables S2 and S3).33,41,42 Neither the CoaguChek PT nor aPTT were found compatible for qualitative assessment of apixaban and dabigatran anticoagulant acitivity.33,41,42 As with other global plasmatic coagulation testing, interpretation of results requires knowledge about the taken DOAC. 24
ClotPro
The ClotPro POCT measures clotting time of blood by using an ecarin-based assay (ECA-test) for testing thrombin inhibitors including dabigatran or a Russell’s viper venom-based assay (RVV-test) for direct oral factor Xa inhibitors with a turn-around time for the measurement of less than 10 minutes. 44 The ClotPro ECA-test showed high correlation with actual dabigatran plasma concentrations (r = 0.8 to 1.0) measured by mass spectrometry or laboratory-based anti-thrombin activity assays.39,44,45 The manufacturer-defined cut-off of 180 s for the ECA-test clotting test for detecting dabigatran plasma concentrations above 50 or 100 ng/ml in a blood sample yielded a 100% sensitivity (Supplementary Table S3). Whereas specificity of the ECA-test assay for dabigatran at the 50 ng/ml threshold was above 90% with or without DOAC-naïve patients, specificity at the 100 ng/ml threshold dropped from 90% to 64% if samples from patients naïve to the tested DOAC were excluded. 39
Edoxaban levels strongly correlate with the results of the ClotPro RVV-test as shown in two studies (r = 0.93 and 0.94).39,45 For apixaban and rivaroxaban, correlations of 0.74 and 0.88, respectively, have been reported. 39 With the manufacturer’s cut-off of 100 s, the ClotPro RVV-test provided sensitivities of 100% for edoxaban and rivaroxaban at both, the 50 and 100 ng/ml plasma concentration thresholds, and 95% and 93% for apixaban, respectively (Supplementary Table S3). Specificity however was lower and ranged from 70% to 90% in case of all reported samples being considered, but only from 16% to 45% if samples from patients naïve to the tested DOAC were excluded (compare recalculated test accuracy parameters with and without samples from patients naïve to the tested DOAC in Supplementary Tables S2 and S3). 39
Despite promising data, the ClotPro is a semi-automated POCT system and requires sample pipetting which may be cumbersome in the emergency setting and contributes to the longer turn-around time. Unfortunately, the ClotPro has so far not been evaluated at the 30 ng/ml DOAC threshold which limits its applicability especially in the setting of intracranial hemorrhage or other life-threatening bleeding when reversal therapy may be indicated even if residual DOAC activity is very low.25,26 Moreover, the ClotPro POCT has recently been taken off the market in mid-2024. However, a re-launch is planned, and it remains the only CE-certified POCT system for blood-based DOAC coagulation testing. Currently, the ClotPro system and its specific RVV-test and ECA-test are not approved by the FDA.
Not commercially available point-of-care coagulation test systems for direct oral anticoagulant activity
The Haemonetics thromboelastography (TEG; Haemonetics, Boston, Massachusetts, USA) 6 s54,55 and the rotational thromboelastometry (ROTEM; Werfen, Barcelona, Spain)56–58 POCT systems use a multichannel testcard with standard or modified thromboelastometric assays which allows simultaneous testing to differentiate between dabigatran and factor Xa inhibitors. However, the prototype cartridges required for DOAC assessment,53,55,58 are so far not commercially available, and it remains unclear whether and when these cartridges will be brought to the market. Standard thromboelastometric assays showed insufficient correlation with DOAC activity at low plasma levels.56,57
The Cascade Abrazo (Helena Laboratories, Beaumont, Texas, USA) measures plasmatic coagulation within few minutes. 59 The ecarin-based direct thrombin inhibitor assay was originally developed to assess bivalirudin-mediated anticoagulation. 59 It showed excellent correlation with even lowest dabigatran plasma concentrations, thereby allowing for calibration and thus quantitative DOAC assessment similar to laboratory-based testing. 59 Unfortunately, this assay is not commercially available.
Important general considerations for any coagulation testing of direct oral anticoagulant activity
In the clinical setting, false positive POCT results are generally less critical than false negatives. A false positive leads to the assumption of relevant anticoagulation, prompting cautious treatment identical to the treatment chosen if no coagulation POCT is available (e.g., no immediate thrombolysis or surgery, antidote administration in case of bleeding). In contrast, a false negative falsely suggests intact coagulation and thus, could result in aggressive treatment with potentially life-threatening consequences. Therefore, all POCT approaches that are recommended in this study primarily focus on avoiding false negatives.
Physicians must consider the time since last DOAC intake when measuring DOAC activity: DOAC levels peak within two to three hours after intake and negative assessment before peak levels are reached carry the risk of a subsequent increase of anticoagulant activity.
When monitoring the success of DOAC reversal, it is important to know that residual anticoagulant activity of factor Xa inhibitors cannot be monitored after administration of andexanet alfa using conventional anti-factor Xa activity assays. 60 This is due to the loose binding of andexanet alfa to the factor Xa inhibitors which is disrupted in the test environment, releasing the active-factor Xa inhibitor and thereby leading to falsely high anti-factor Xa activity results despite application of andexanet alfa. 60 This is not an issue in case of dabigatran reversal with the monoclonal antibody idarucizumab which binds dabigatran irreversibly and does not affect coagulation parameters.
Eventually, emergency physicians must know that blood-based coagulation assessment of the direct oral factor Xa inhibitors could be influenced by heparins and heparinoids 61 and of dabigatran by other thrombin inhibitors such as bivalirudin or argatroban 59 which also inhibit factor Xa and thrombin, respectively.
Conclusions
POCT for DOAC is not widespread although individual studies have shown its feasibility in identifying blood samples containing clinically relevant DOAC plasma concentrations to guide emergency decision-making on thrombolysis, urgent surgery and DOAC reversal in intracranial hemorrhage or other life-threatening bleeding. Each device has its own unique limitations. Generally, for more reliable interpretation of POCT for DOAC, knowledge about the time of last intake and exact type of DOAC is advantageous. In view of these limitations, we propose an algorithm that could help guiding stroke and emergency physicians in selecting an appropriate POCT assay in DOAC-related emergencies (Figure 2 and Table 1).
Point-of-care test (POCT) systems and respective cut-offs for identifying patients with a direct oral anticoagulant (DOAC) plasma concentration >30, 50, or 100 ng/ml.
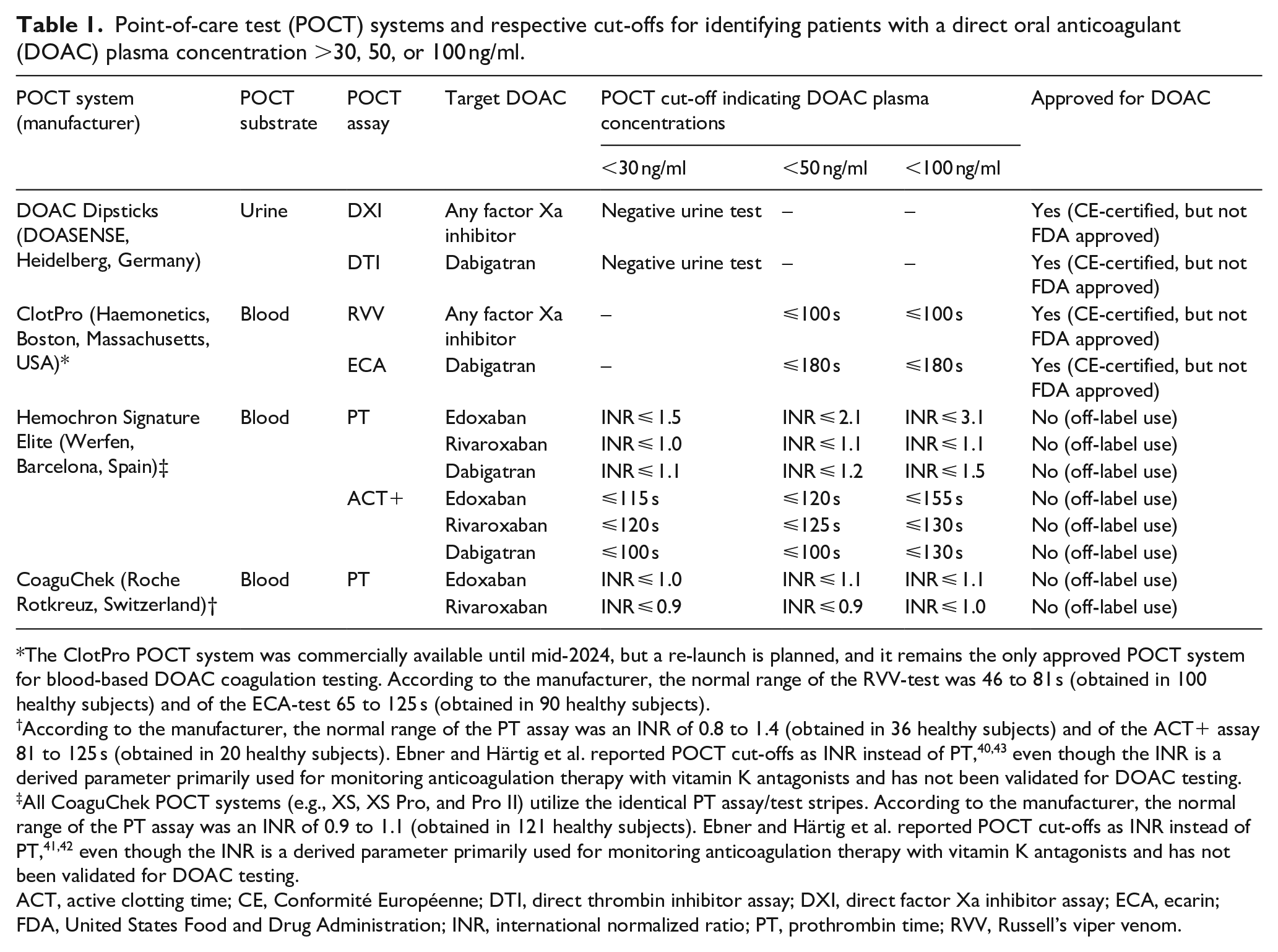
The ClotPro POCT system was commercially available until mid-2024, but a re-launch is planned, and it remains the only approved POCT system for blood-based DOAC coagulation testing. According to the manufacturer, the normal range of the RVV-test was 46 to 81 s (obtained in 100 healthy subjects) and of the ECA-test 65 to 125 s (obtained in 90 healthy subjects).
According to the manufacturer, the normal range of the PT assay was an INR of 0.8 to 1.4 (obtained in 36 healthy subjects) and of the ACT+ assay 81 to 125 s (obtained in 20 healthy subjects). Ebner and Härtig et al. reported POCT cut-offs as INR instead of PT,40,43 even though the INR is a derived parameter primarily used for monitoring anticoagulation therapy with vitamin K antagonists and has not been validated for DOAC testing.
All CoaguChek POCT systems (e.g., XS, XS Pro, and Pro II) utilize the identical PT assay/test stripes. According to the manufacturer, the normal range of the PT assay was an INR of 0.9 to 1.1 (obtained in 121 healthy subjects). Ebner and Härtig et al. reported POCT cut-offs as INR instead of PT,41,42 even though the INR is a derived parameter primarily used for monitoring anticoagulation therapy with vitamin K antagonists and has not been validated for DOAC testing.
ACT, active clotting time; CE, Conformité Européenne; DTI, direct thrombin inhibitor assay; DXI, direct factor Xa inhibitor assay; ECA, ecarin; FDA, United States Food and Drug Administration; INR, international normalized ratio; PT, prothrombin time; RVV, Russell’s viper venom.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241310359 – Supplemental material for Rapid assessment of direct oral anticoagulants in acute stroke—An educational systematic review
Supplemental material, sj-docx-1-eso-10.1177_23969873241310359 for Rapid assessment of direct oral anticoagulants in acute stroke—An educational systematic review by Joshua Mbroh, Ingvild Birschmann, Matthias Ebner, Johanna A. Kremer Hovinga, Edelgard Lindhoff-Last, Jan Purrucker, Simon T. Schäfer, Johannes Tünnerhoff, Yi Wang and Sven Poli in European Stroke Journal
Footnotes
Acknowledgements
Not applicable.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JP has received consultation fees and travel expenses from Abbott, Akcea, Bayer, Boehringer Ingelheim, Daiichi Sankyo, and Pfizer (all outside the submitted work).
SP reports research support from BMS/Pfizer, Boehringer Ingelheim, Daiichi Sankyo, German Federal Joint Committee Innovation Fund, and German Federal Ministry of Education and Research, Helena Laboratories and Werfen as well as speakers’ honoraria/ consulting fees from Alexion, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol Myers Squibb/Pfizer, Daiichi Sankyo, Portola, and Werfen (all outside of the submitted work).
IB has acted as paid speaker in the past for CSL Behring, Siemens Healthcare Diagnostics Products, AstraZeneca, Octapharma. She has performed contract research for Siemens Healthcare Diagnostics Products and Behnk Elektronik and is a member of the advisory board of Alexion and of the expert group of CSL Behring and Siemens Healthcare Diagnostics Products.
JAKH has served on advisory boards of Ablynx, now part of Sanofi, CSL-Behring, NovoNordisk, Roche, Sobi and Takeda, and has received lecture fees from CSL-Behring, Roche, Sanofi and Takeda. All honoraria and fees are paid to her employer, Insel Gruppe AG, Bern, Switzerland.
ELL received lecture honoraria and advisory fees from Bayer, Boehringer Ingelheim, BMS/Pfizer, Daiichi Sankyo, Portola, CSL Behring, Norgine, Roche, Leo, AstraZeneca, and Aspen and institutional research support from Bayer, BMS /Pfizer, Daiichi Sankyo, Werfen, and CSL Behring.
The other authors report no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was not sought for this article because it is a systematic review based on published literature and does not contain data from any individual person.
Ethical approval
Ethical approval was not sought for this article because it is a systematic review based on published literature and does not report on or involve the use of any animal or individual human data or tissue.
Guarantor
SP takes full responsibility for the article, including for the accuracy and appropriateness of the reference list.
Contributorship
JM and SP conceived the study and developed the protocol. JM and YW assessed eligibility and quality of identified studies. SP aggregated and analyzed the data and supervised the study. JM, JT and SP interpreted the data. JM wrote the first draft of the manuscript. JT and SP refined and complemented the manuscript for critical review. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Data availability
The data supporting the findings of this study are available within the article and its supplementary materials.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
