Abstract
Introduction
Non-vitamin K-antagonist oral anticoagulants (NOAC) have become first choice oral anticoagulant (OAC) with decreasing use of vitamin K antagonists (VKA), partly due to lower risk of intracerebral hemorrhage (ICH). Aim: to identify trends in sale of OACs and relate them to trends in OAC-related ICH (OAC-ICH).
Patients and methods
Study was based on the population in the Capital Region of Denmark (1.8 million inhabitants). We identified all patients admitted with a non-traumatic OAC-ICH in 2010–2017 and ascertained diagnosis and drug use through medical charts. We used information available in the public domain on sale of defined daily doses (DDD) of OAC in the Capital Region of Denmark.
Results
453 patients with OAC-ICH out of a total of 2877 ICH-events were identified. From 2010 to 2017 sale of NOAC rose from 0.1 to 11.8 DDD/1000 inhabitants/day (p < 0.001); while VKA sale decreased from 7.6 to 5.2 DDD/1000 inhabitants/day (p < 0.001). The total number of ICH events was stable between 2010 and 2017, but the proportion of OAC-ICH events increased from 13% in 2010 to 22% in 2017 (p < 0.001). The proportion of ICH events related to NOAC had a significant increasing trend (p < 0.001), whereas a decreasing trend was observed for VKA (p = 0.04).
Discussion
In Denmark, the population on OACs has increased; resulting from increased use of NOACs. Parallel to this development, the proportion of OAC-ICH overall has increased based on an increasing trend in NOAC-related ICH.
Conclusion
Our findings document a need for further research on prevention and treatment of this complication.
Keywords
Introduction
Non-vitamin K-antagonist oral anticoagulants (NOACs) are non-inferior or superior to vitamin K-antagonists (VKA) in prevention of stroke among patients with atrial fibrillation (AF) and are associated with a 50% reduction in risk of oral anticoagulant (OAC) related intracerebral hemorrhage (ICH), in OAC treated AF-patients. 1 Further, NOACs are far easier to use for patients and healthcare professionals as dosing is fixed, and repeated blood testing, and food restrictions are not needed.
Based on reported superior risk/benefit profiles of NOACs in comparison to VKA, NOACs have become the drug-of-choice and the use has gradually been extended, also to high risk populations. 2
In spite of the present widespread use of NOACs, there are still little conclusive data on the presentation and prognosis of NOAC-related ICH (NOAC-ICH). Especially population-based data is lacking. Most studies are small and based on selected patients, and thereby have large confidence intervals or inconclusive results.3–11 This is illustrated by the mortality rates reported in NOAC-ICH, which range from 16% to 70%, most likely due to the fact that the studies are looking at small sample sizes, and at both acute and non-acute stages of the disease.3,5–8,10,11
So far, no data has been published on the trends in OAC-related ICH (OAC-ICH) over time and the temporal relationship to the exposure of the population to OAC. In this study we provide such data for OAC overall and by type of OAC (VKA vs NOAC) for the Capital Region of Denmark, a geographically well-defined area (population 1.7–1.8 million in the study period).
Patients and methods
The Danish stroke registry
The Danish Stroke Registry (DSR) is a nationwide registry. 12 All danish citizens have access to tax-supported healthcare free of charge. Expenses to most prescribed drugs, including all OACs, are partially reimbursed. All departments treating patients with acute stroke are required by law to prospectively report data to DSR. 12 DSR has been validated, in terms of credibility in use of the ICH diagnosis, in the Region of Southern Denmark (positive predictive value of 85% (95% CI: 81–88) for spontaneous ICH). 13
COOL-ICH
The Capital Region Anticoagulation-related ICH study (COOL-ICH) was a retrospective observational study including all adult patients admitted in the Capital Region of Denmark within 24 hours of onset of a spontaneous ICH related to oral anticoagulant therapy (OAC-ICH) between 2010 and 2017. This analysis included 79 patients with symptom onset over 24 hours before admission, in order to make the cohort as complete as possible. Patients were identified through discharge lists retrieved from the five departments providing designated stroke care in the Capital Region (four neurological and one neurosurgical department); from the same wards, to enhance completeness, we also retrieved patient-specific data on ICH discharges reported to the Danish Stroke Registry. 12 We included eligible patients with an International Classification of Diseases, 10th edition (ICD-10) code for ICH (I61) identified through either of the two sources. In Denmark, all patients are admitted based on hospital catchment areas and all patients with recognized symptoms of stroke are admitted to designated neurology or neurosurgery departments caring for patients with stroke according to a national stroke plan. Patients not admitted to hospital, patients admitted at other departments, but not transferred to the stroke care departments, or patients not identified as having an ICH after admission, will consequently have been missed based on our approach. All hospitals in the Capital Region use electronic health records (EHRs). A patient’s EHR holds information on drug use during hospital stays. In addition, each patient’s EHR holds continuously updated data on prescriptions presented at community pharmacies anywhere in Denmark. Due to this unique feature, it is possible to obtain a virtually complete medication history for patients, which covers both pre-hospital and in-hospital use of drugs. Data on clinical characteristics, management and outcome were extracted from individual electronic patient files. A protocol for data acquisition and handling was published before we initiated extracting data from patients’ files. 14
Statistics Denmark
Statistics Denmark is a public institution under the Ministry of Social Affairs and the Interior. Data regarding number of inhabitants in the Capital Region was retrieved through “StatBank”. 15
Medstat
Data regarding sale of OAC in the general population were retrieved from the Danish Registry of Medicinal Product Statistics, through the publicly available MEDical STATistics (Medstat), published by the Danish Health Data Authority. 16 We obtained sales data measured in Defined Daily Doses (DDD) for drugs with Anatomical Therapeutic Chemical (ATC) codes of B01AA (03-04), B01AE07 and B01AF (01-03), corresponding to warfarin (DDD = 7.5 mg), phenprocoumon (3 mg), dabigatran (300 mg), rivaroxaban (20 mg), apixaban (10 mg) and edoxaban (60 mg), 17 sold in the Capital Region from 2010 until 2017, in both the primary healthcare sector (dispensed prescriptions) and the hospital sector (bulk sales). We used DDD per 1,000 inhabitants per day as a measure of drug consumption in the population, which enabled us to compare drug utilization patterns over time at the population level, but not at the level of the individual patient.
Data analysis
Frequency distributions of OAC-ICHs in the Capital Region, distributed by year of admission and type of drug, were based on data from COOL-ICH. Frequency distribution of all ICH events in the Capital Region by year of admission was based on data from DSR. We retrieved census data from Statistics Denmark on the number of inhabitants on the first day of the third quarter of each year in the study period, which we used as an estimate of person years at risk. Based on these data and the annual number of incident cases of ICH in the Capital Region, we calculated incidence rates of ICH with a 95% confidence interval. Poisson regression was used to evaluate trends in number of ICH-events from 2010 to 2017. Calendar year was the independent variable and was incorporated in the analyses as an ordered variable and as a numeric variable (trend analysis). The trend of the proportion of OAC-related, NOAC-related and VKA-related ICH (of the total number of ICH) was tested with a two-sided Cochran-Armitage test for trend.
Aggregate sales data of NOAC and VKA were calculated based on data from MEDSTAT. Results were graphically illustrated using line plots and bar charts. Linear regression analysis was used to detect significant trends in the annual sale of OAC, where calendar year was both incorporated as an ordered variable and as a numeric variable (trend analysis). All data-analyses were made using R version 3.5.1.
Ethics
The study was approved by the Danish Data Protection Agency (2012-58-0004) and by the Danish Patient Safety Authority (3-3013-2102/1). Patient data were collected from patient files without patient contact nor informed consent.
Results
Sales of OAC
Sales of NOAC rose from 0.1 to 11.8 DDD/1000 inhabitants/day from 2010 to 2017; while sales of VKA decreased from 7.6 to 5.2 DDD/1000 inhabitants/day. From 2015 to 2017, more NOAC than VKA was sold (Figure 1). In 2017, sales of apixaban surpassed sales of rivaroxaban (Figure 2). The difference between sales of warfarin, apixaban and rivaroxaban in 2017, was within a range of 1 DDD/1000 inhabitants/day (the sales being 5.1, 4.7 and 4.4 DDD/1000 inhabitants/day, respectively). Neither sales of phenprocoumon nor edoxaban exceeded 0.2 DDD/1000 inhabitants/day. Sales of VKA decreased (p < 0.001) and sales of NOAC increased (p < 0.001) during the period.
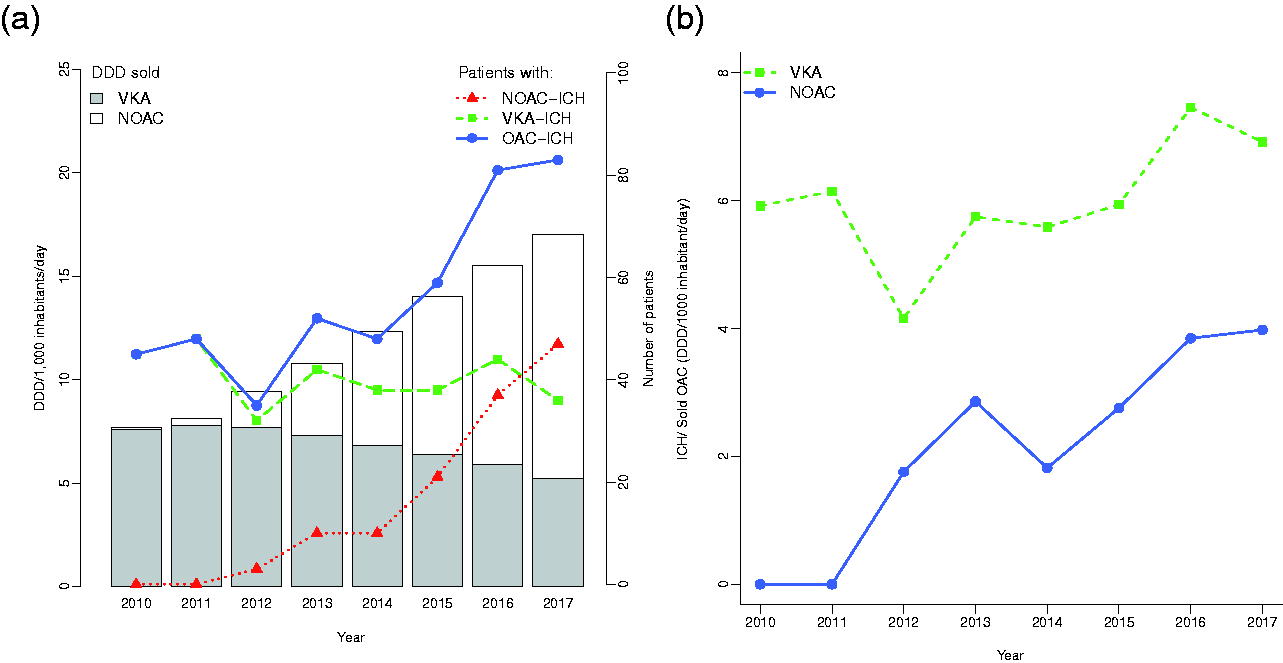
Annual numbers of OAC-ICH, and corresponding annual sales in Daily Defined Doses (DDD) per 1,000 inhabitants per day in Capital Region, Denmark 2010–2017. ICH = intracerebral hemorrhage. NOAC = non-vitamin K-antagonist oral anticoagulants. VKA = vitamin K-antagonist. OAC = oral anticoagulants.
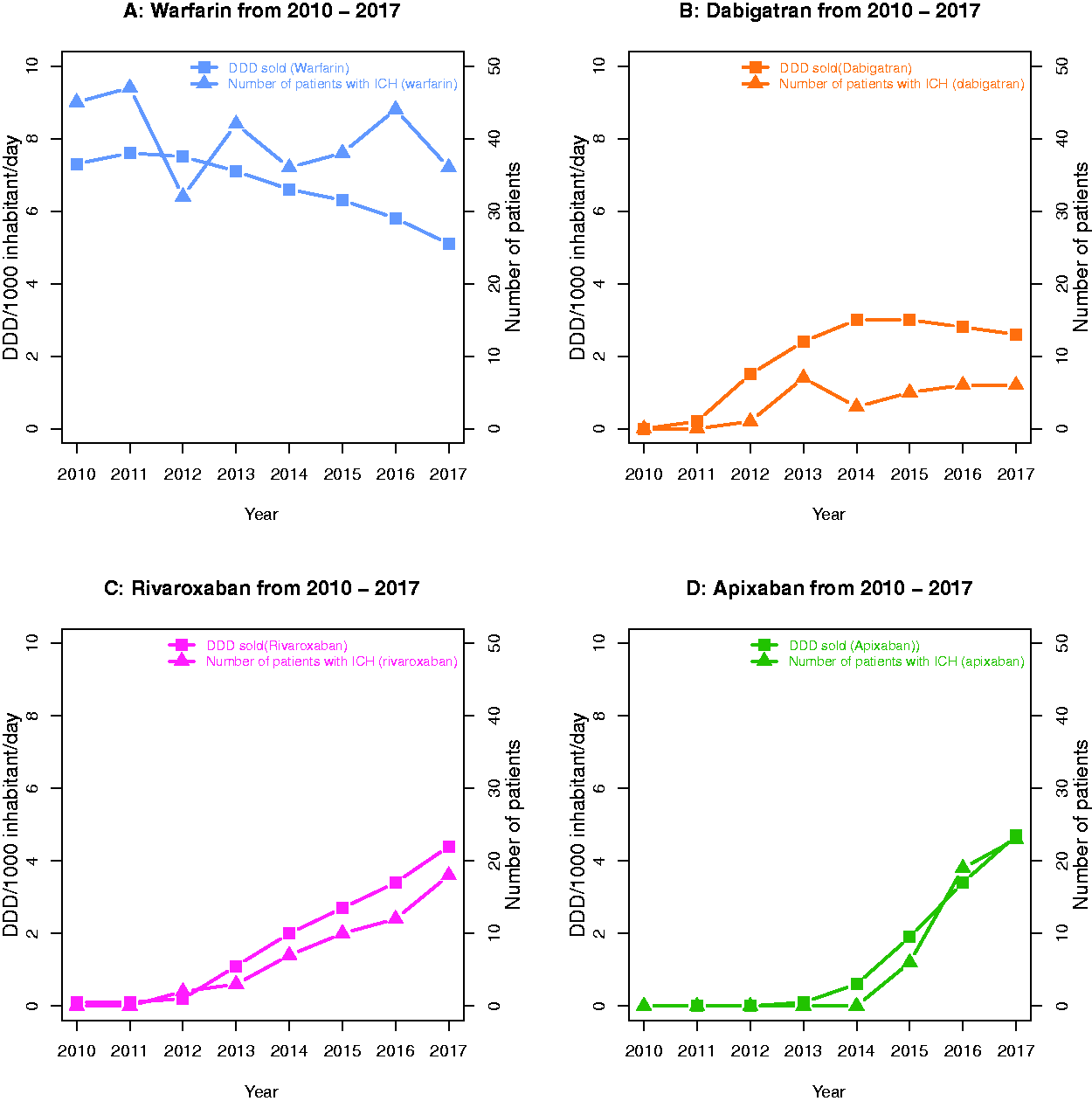
Number of patients with ICH related to specific oral anticoagulant drugs and the corresponding sales of the drugs in the population over the years from 2010 to 2017 for each individual oral anticoagulant. DDD = Defined Daily Doses. ICH = intracerebral hemorrhage.
ICH-events
A total of 451 patients with OAC-ICH-events were identified in the study period out of a total of 2877 ICH-events. The incidence rate of ICH in the population varied from 19.5/100,000 person-years to 21.9/100,000 person-years with no significant variations or trends during the period (test of trend p = 0.87). Figure 3 illustrates the stable ICH frequency through the study period, but a relative increase in the proportion of OAC-ICH.
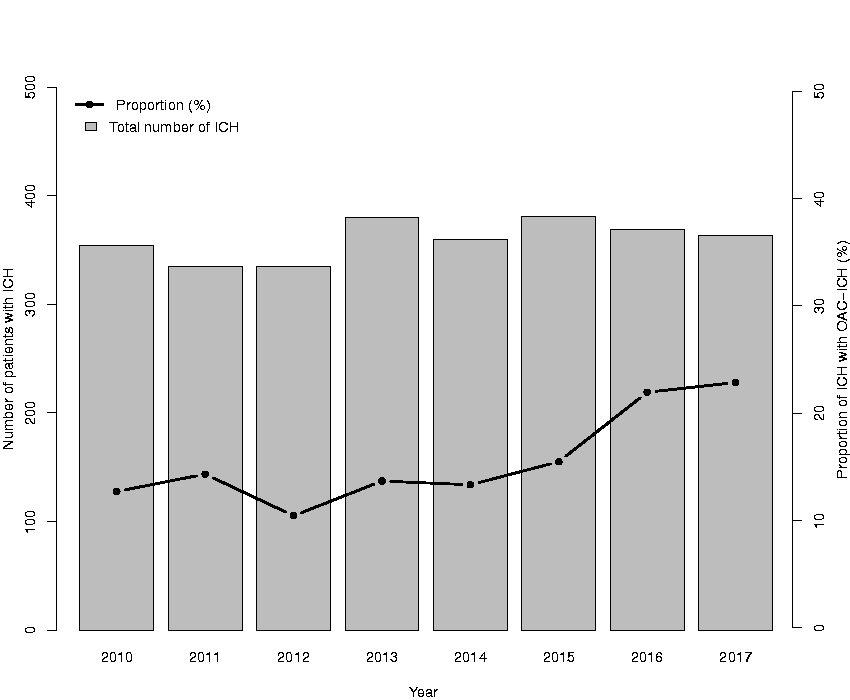
Total number of patients with spontaneous ICH, OAC related included (columns), and proportion of OAC-ICH (line) per year. OAC = oral anticoagulants. ICH = intracerebral hemorrhage.
OAC-ICH
Of all OAC-ICH events identified (n = 451), 323 occurred in patients on VKA and 128 in patients on a NOAC. The total number of patients with OAC-ICH was lower in the first half of the study period (2010-2013: n = 180), compared with 2014-2017 (n = 271). Correspondingly, the proportion of OAC-ICH increased from 12% to 23% of the total number of ICH events from 2010 to 2017 (Cochran-Armitage test of trend, p < 0.001); illustrated in Figure 3. The first NOAC-related ICH-event was in 2012; by 2017, NOAC-ICH constituted 13% of all ICH-events. VKA-related ICH (VKA-ICH) decreased from 12% in 2010 to 10% in 2017 of all ICH-events. In 2017, NOAC-ICH was more frequent than VKA-ICH (Figure 1). During the study period, the proportion of ICH events related to NOAC-treatment increased (p < 0.001), whereas the absolute number of ICH events related to VKA-treatment did not change significantly (p < 0.25). Demographic data of OAC-ICH patients is presented in Supplementary Table 1.
OAC-ICH compared to sale of OAC and total number of ICHs
Figure 2 illustrates trends related to the four most frequently used OACs. The pattern is highly similar for all four drugs (A–D): The development over time in the number of patients suffering from ICH while on a specific OAC mirrors trends in use of the specific OACs in the general population. No ICH events related to current use of edoxaban or phenprocoumon were observed in the study period; due to sparse data, trends regarding sales of edoxaban and phenprocoumon in the general population were not calculated.
Discussion
This study documents that the total number of ICH events in a geographically well-defined area in Denmark was stable between 2010 and 2017, but that the proportion of OAC-ICH events increased substantially from 12% to 23%. Importantly, the composition of OAC-ICH changed markedly over time with the observed increase being driven by more frequent occurrence of NOAC-ICH. By the end of the study period, NOAC-ICH was a considerably more frequent event than VKA-ICH. The observed changes occurred in parallel with similar changes in patterns of OAC use in the general population, i.e., an increase in OAC use due entirely to increased use of NOACs. 18
Results from Randomized Controlled Trials (RCTs) and post-marketing registry studies have led to an expectation of a relative decrease in number of patients presenting with OAC-ICH, if more patients were on NOACs rather than VKA. However, based on previous underuse of OAC, 19 and thanks to the more favorable safety profile of NOACs, 1 the number of patients in the general population on OACs has increased markedly. In this study, we observed an increase in NOAC use in the Capital Region of Denmark with the steepest increase observed in the last years of the study period. By 2017, 11.8 DDD/1000 inhabitants/day, corresponding to > 1% of the entire population were on a NOAC treatment with the total use of OAC that year being 17.0 DDD/1000 inhabitants/day. Our results indicate that higher use of NOACs in the general population – despite the lower risk of ICH – has translated into a larger proportion of ICH patients being classified as OAC-ICH.
In Denmark, OACs were mainly used in AF and venous thromboembolism (VTE). In Scandinavia, recent data suggests an incidence atrial fibrillation of 3.0/1000 person years 20 and an incidence of VTE of 1.4/1000 person years. 21 Recent Western European data reported rates ranging from 1.9–2.9%. 22 Consequently, the actual DDD/1000 inhabitants/day of OAC only corresponds to less than half of the population affected by atrial fibrillation or venous thromboembolism potentially leaving room for further increase in use of OAC.
With increasing age, risk of multi-morbidity and frailty increases,23–25 which may affect the risk/benefit balance during treatment with antithrombotics. However, in the RCTs comparing warfarin to NOACs, the average age of participants was below 75 years, and patients with multiple comorbidities or high risk of bleeding were excluded by protocols.26–35 Thus, RCTs only provide little data on frail patients and patients aged > 80 years.1,36 Likewise, there are substantial differences between populations in the four RCTs comparing NOACs to VKA in AF patients. 1 Further, no RCTs have directly compared NOACs. Because of this, it is difficult to ascertain if results are based on differences in populations or differences in the risk-profiles of the individual NOACs. Consequently, extending the target population may be associated with a difference in the adverse event profile. However, so far, observational data from registries confirmed the assumed safety profile of NOACs. 37
The development in choice of OAC observed in this study follows the Danish guidelines: Until 2016, Danish guidelines on anticoagulants in AF recommended VKA as first line therapy if time in therapeutic range (TTR) was above 70%. 38 However, the most recent Danish guideline from 2016 recommends NOACs as first line therapy. 39 We assume this change in guidelines explains the significant increase in use of NOACs observed in the later part of the study period. Observational data suggests a beneficial effect from resumption of OAC after ICH. Are these findings confirmed in ongoing randomized trials the number of patients treated with OAC might increase further. 40
So far, there is little evidence to support that OAC directly causes cerebral bleeding, it is more likely that OAC increases the clinical significance of an incident bleeding. One previous report explored the incidence of ICH following increased use of VKA in the 1990s. 41 The authors reported an increase not only in the proportion of ICHs which were OAC-related, but also in the total incidence of ICH based on data from five groups of patients in the Greater Cincinnati/Northern Kentucky area. 41 Of note, according to data from the Global Burden of Disease Project there was in contrast a decline in incidence of ICH in USA. 42 In Denmark, a marked decrease in rate of ICH was also observed until 2010. 42 However, according to data from the present study, there was no further decline in ICH incidence, in the Capital Region from 2010–17. Whether this halt in the decline of the incidence of ICH can be ascribed to the marked increased in use of OACs is open to speculation.
The strengths of this study include that it describes a well-defined geographical population and that ICH events included have been validated through individual patient charts. The structure of the Danish health system enables rapid evaluation for stroke with no financial health barriers; patients suspected of suffering a stroke are swiftly admitted or transferred to dedicated stroke units. Limitations include the observational character of data, preventing straightforward conclusions on causality. Also, our population is a high-income region, in a high-income country, with ethnically homogenous population affecting the generalizability of the study. Some eligible patients may not have been included in the study due to our identification method, e.g., patients that died before reaching a stroke unit were not included in the study per design. This may have resulted in some degree of underestimation of the total number of ICHs in the Capital Region in the study period, particularly among the oldest old. However, we have little reason to believe this potential source of bias would be associated to a specific OAC. Our data on OAC use among OAC-ICH patients was based on evidence of presented prescriptions in patients’ EHRs, which may have led to some misclassifications, e.g. patients who stopped taking their OACs after their most recent prescription was presented, but before ICH onset without the admitting physician knowing it. Our estimates of OAC use in the general population were based on annual sales data of OAC retrieved from Medstat. 16 In spite of the inability to trace use of OAC in individual patients, we believe it is suited to describe trends in population use of drugs. 18
Conclusion
During the study period incidence of OAC-ICH has almost doubled, while sale of OAC has more than doubled. Based on the increasing number of patients presenting with NOAC-ICH, there is an obvious need for more research in course, treatment – including on reversal strategies – prognosis as well as care for patients with NOAC-ICH, which in 2017 constituted 13% of the ICH population. It is important to emphasize that there is strong evidence supporting the use of OAC as well as a more favorable risk/benefit ratio in NOACs in comparison to VKA. Therefore, our data do not challenge the existing recommendations on use of OAC or bring forward data on the individual patient risk/benefit ratio in relation to OAC treatment.
Supplemental Material
sj-pdf-1-eso-10.1177_23969873211008770 - Supplemental material for Trends in incidence of oral anticoagulant-related intracerebral hemorrhage and sales of oral anticoagulants in Capital Region of Denmark 2010–2017
Supplemental material, sj-pdf-1-eso-10.1177_23969873211008770 for Trends in incidence of oral anticoagulant-related intracerebral hemorrhage and sales of oral anticoagulants in Capital Region of Denmark 2010–2017 by Josefine Grundtvig, Christian Ovesen, Inger Havsteen, Thomas Christensen, David Gaist, Helle K Iversen, Christina Kruuse, Alexander Lilja-Cyron, Karen Ægidius, Sverre Rosenbaum, Per Meden, Jacob Marstrand, Louisa Christensen, Thorsten Steiner and Hanne Christensen: ; for the PHYS-Stroke study group in European Stroke Journal
Footnotes
Acknowledgements
No acknowledgements.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Christensen has received honoraria for duties as national lead from Bayer and Portola, and speakers honoraria from Daichi-Sanko, Bayer, Boehringer-Ingelheim and BMS. Dr. Gaist reported receiving honoraria from AstraZeneca (Sweden) for participation as a coinvestigator on a research project outside the submitted work; and receiving speaker honorarium from Bristol-Myers Squibb outside the submitted work. All other authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Lundbeck Foundation.
Informed consent
Informed consent not required.
Ethical approval
This study was not a biomedical study as to the definition in Danish law, and no ethical approval was required.
Guarantor
HC.
Contributorship
CO, HC and TS researched literature and conceived the study. JG, CO, HC and TS were involved in protocol development, data acquisition and data analysis, DG contributed to planning of data analysis. JG wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
