Abstract
Purpose:
About 20%–25% of all intracerebral haemorrhages are associated with oral anticoagulation therapy. Reflecting changings prescription patterns in the general population, the spectrum of oral anticoagulation-associated intracerebral haemorrhage has substantially changed in the last decade. In many European countries, direct oral anticoagulant-associated intracerebral haemorrhage is now more frequent than vitamin K antagonist-associated intracerebral haemorrhage. Outcome in patients with anticoagulation-associated intracerebral haemorrhage is poor, likely mediated by a high incidence of haematoma expansion. Reversal of anticoagulation is an essential part of current care pathways for hyperacute treatment of intracerebral haemorrhage aiming to limit haematoma expansion and thereby improving outcome.
Methods:
In this review, we summarise the latest evidence regarding reversal therapy for vitamin K antagonist-, direct thrombin inhibitor- and factor Xa inhibitor-associated intracerebral haemorrhage.
Findings:
Two randomised controlled trials have shown that the use of prothrombin complex concentrate (compared to fresh frozen plasma) for reversing vitamin K antagonist-associated intracerebral haemorrhage and andexanet alfa (compared to usual care, mainly prothrombin complex concentrate) for factor Xa inhibitor-associated intracerebral haemorrhage had superior haemostatic efficacy. However, the incidence of thromboembolic complications was high in both trials. For reversal of Vitamin K antagonist-associated intracerebral haemorrhage, the overall rate was 18% but due to crossovers, it is impossible to determine the rate for any specific treatment. For factor-Xa inhibitor associated intracerebral haemorrhage, andexanet alfa led to an increase in the incidence of thromboembolic events. Moreover, these two randomised controlled trials were not powered to detect differences in mortality or functional outcomes and lacked long-term follow-up. Idarucizumab has shown promising results in a single-arm case series of patients with intracerebral haemorrhage associated with the direct thrombin inhibitor dabigatran, yet no randomised controlled trial is available to support these findings.
Conclusion:
Given that haematoma expansion is strongly associated with poor outcome, current evidence underlines the importance of rapid, targeted and effective reversal of anticoagulation in patients with anticoagulation-associated intracerebral haemorrhage. While haematoma expansion is a key prognostic factor, no randomised controlled trial has demonstrated a clear improvement in functional outcome. Future research should weigh the advantages of preventing haematoma expansion against the risks of increased thromboembolic events, and aim to identify the patients who would derive the most benefit from reversal treatments.

Keywords
Introduction
Intracerebral haemorrhage exerts a significant healthcare burden worldwide and is a major contributor to morbidity and mortality. 1 The incidence of intracerebral haemorrhage in Europe is projected to increase significantly by 2050 driven by an ageing population and and consequently prevalence of atrial fibrillation resulting in treatment with oral anticoagulants. 2 Oral anticoagulation is effective for the treatment and prevention of various thromboembolic diseases including the prevention of ischaemic stroke in patients with non-valvular atrial fibrillation and venous thromboembolism. Atrial fibrillation is a frequent comorbidity in up to 1/3 of patients with intracerebral haemorrhage.3–5
Treatment with oral anticoagulants increases the risk of intracerebral haemorrhage, 6 most likely mediated by the concomitant presence of bleeding-prone cerebral small vessel disease.7,8 Data from 2013 to 2019 from Switzerland and Norway found that approximately 20%–25% of all intracerebral haemorrhages were associated with prior intake of oral anticoagulants. 3 Moreover, throughout the last decade, the spectrum of anticoagulant use has shifted from vitamin K antagonists to a higher proportion of direct oral anticoagulants, mainly the direct factor Xa inhibitors, apixaban and rivaroxaban.9,10
Outcome of anticoagulation-associated intracerebral haemorrhage is poorer compared to intracerebral haemorrhage not associated with prior anticoagulation therapy.11,12 This difference is most likely due to age, co-morbidities, as well as larger baseline haematoma volumes and a higher rate of haematoma expansion related to anticoagulation-associated intracerebral haemorrhage. 12 At present, this finding is mainly based on patients with vitamin K antagonist-associated intracerebral haemorrhage. However, more recent data from two large national stroke registries found that the prior use of vitamin K antagonist or direct oral anticoagulant therapy were both associated with higher mortality and poorer functional outcome compared to intracerebral haemorrhage in patients not taking oral anticoagulants. 3 Results directly comparing prognosis related to vitamin K antagonist-associated intracerebral haemorrhage and to direct oral anticoagulant-associated intracerebral haemorrhage demonstrated diverging results, depending on the time-point of assessment (early vs late) and the type of outcome (mortality vs functional outcome).3,4,11
The primary aim in the hyperacute treatment of anticoagulation-associated ICH is mainly focused on establishing effective haemostasis by reverting the anticoagulant effect of the drug in order to prevent haematoma expansion. 13 The principles of reversal therapy in patients with oral anticoagulant-associated intracerebral haemorrhage, including the available strategies according to the type of oral anticoagulant, are discussed in a separate paper from this special issue (Christensen al. ESJ). In addition to haematoma expansion affecting prognosis, the haematoma volume-specific direct effect on prognosis may differ according to ICH location.14,15 Given that anticoagulation-associated ICH differs slightly in location distribution compared to ICH unrelated to anticoagulation, including a significantly greater burden of intraventricular haemorrhage, this may, in part, contribute to its poorer outcome. 16 Haematoma expansion in intracerebral haemorrhage unrelated to the use of prior anticoagulation most commonly occurs within the first 3 h. 17 In patients with anticoagulation-associated intracerebral haemorrhage, altered haemostasis is hypothesised to prolong haematoma expansion compared to their non-anticoagulated counterparts, emphasising the potential benefit of anticoagulation reversal. A recent analysis from a large US-based registry found that the initiation of anticoagulant reversal within 60 min from hospital admission was associated with lower odds of mortality. 18 This finding was mainly based on data from patients with vitamin K antagonist-associated intracerebral haemorrhage. Although limited by a significant amount of missing data on anticoagulation reversal, this study advocated for a rapid and effective treatment strategy in anticoagulation associated intracerebral haemorrhage. Care bundle approaches for the treatment of intracerebral haemorrhage apply a combination of several acute interventions19,20 aiming to prevent haematoma expansion and improve outcome in patients with intracerebral haemorrhage. Reversal of oral anticoagulation is a major part of the care bundle approach. 21 In this review, we summarise and discuss the available clinical evidence for the efficacy and safety of reversal of oral anticoagulation in patients with intracerebral haemorrhage.
Literature search
We performed a systematic literature search using PubMed on 1 April 2023 and again on 25 February 2024 using the following search terms: ((intracerebral haemorrhage OR ICH OR intracerebral hemorrhage) AND (oral anticoagulation OR OAC OR warfarin OR phenprocoumon OR acenocoumarol OR rivaroxaban OR xarelto OR dabigatran OR pradaxa OR edoxaban OR lixiana OR apixaban OR eliquis)) AND (reversal OR prothrombin complex concentrate OR PCC OR fresh frozen plasma OR FFP OR andexan alfa OR idarucizumab). In addition, we searched the reference lists of relevant papers and searched for unpublished results from relevant studies recently presented at major conferences (2023–2024). We screened titles and abstracts followed by a full-text review of relevant articles. We applied the following inclusion criteria: (a) clinical trial (randomised controlled trials, prospective and retrospective cohort studies), (b) adult patients (⩾18 years) with anticoagulation-associated intracerebral haemorrhage, (c) any reversal therapy (with/-out a comparator) and (d) clinical or radiological outcomes of interest (haematoma expansion, mortality, functional outcome, thromboembolic complications). We excluded studies reporting only experimental data from rodent models/in-vitro studies, or studies reporting heterogeneous patient groups (i.e. where cases of intracerebral haemorrhage were indistinguishable from cases of other types of haemorrhages22,23). The literature search yielded 415 and 454 results, respectively. Finally, 12 studies were included.
Reversal of vitamin K antagonist therapy in patients with intracerebral haemorrhage
The vitamin K antagonists warfarin, phenprocoumon and acenocoumarol induce a clotting factor deficiency including factors II, VII, IX and X by blocking vitamin K epoxide reductase.24,25 Reversal therapy therefore focuses on the repletion of the depleted coagulation factors (Christensen al. ESJ). Two randomised controlled trials have compared fresh frozen plasma with 4-factor prothrombin complex concentrate in patients with vitamin K antagonist-related major bleedings, 26 and in patients requiring urgent major surgery. 27 However, these trials included all types of vitamin K antagonist-related major bleedings and only a small number of patients with intracerebral haemorrhage. There are two large-scale retrospective cohort studies28,29 and one randomised controlled trial 30 that provide current evidence comparing fresh frozen plasma and 4-factor prothrombin complex concentrate in patients with vitamin K antagonist-associated intracerebral haemorrhage. Table 1 summarises their results. The major findings from these studies showed that rapidly reversing anticoagulation to achieve an International Normalized Ratio (INR) of <1.3 within 4 h of onset was associated with a lower incidence of haematoma expansion and a significantly reduced mortality rate.28,30 Moreover, the administration of 4-factor prothrombin complex concentrate alone, or in combination with fresh frozen plasma, demonstrated superior efficacy to fresh frozen plasma alone in achieving significant reversal of anticoagulation (i.e. normalising the INR <1.3) following vitamin K antagonist-associated intracerebral haemorrhage. 30 In the large, randomised trials comparing 4-factor prothrombin complex concentrate to fresh frozen plasma within a general bleeding population (i.e. not restricted to patients with intracerebral haemorrhage), no significant difference in the rate of thromboembolic complications (7%–8%) was observed.26,27 However, in the randomised controlled trial restricted to patients with intracerebral haemorrhage, the overall rate of thromboembolic complications associated with both fresh frozen plasma and 4-factor prothrombin complex concentrate was very high (Table 1). 30 A meta-analysis comparing 4-factor and 3-factor prothrombin complex concentrates for emergent warfarin reversal found that 4-factor prothrombin complex concentrate was superior in terms of INR reversal while maintaining similar safety profiles. 31 Finally, the effect of reversal with 4-factor prothrombin complex concentrate and/or fresh frozen plasma on long-term functional outcome in vitamin K antagonist-associated intracerebral haemorrhage remains unclear.
Clinical trials on anticoagulation reversal in patients with vitamin K antagonist-associated intracerebral haemorrhage.
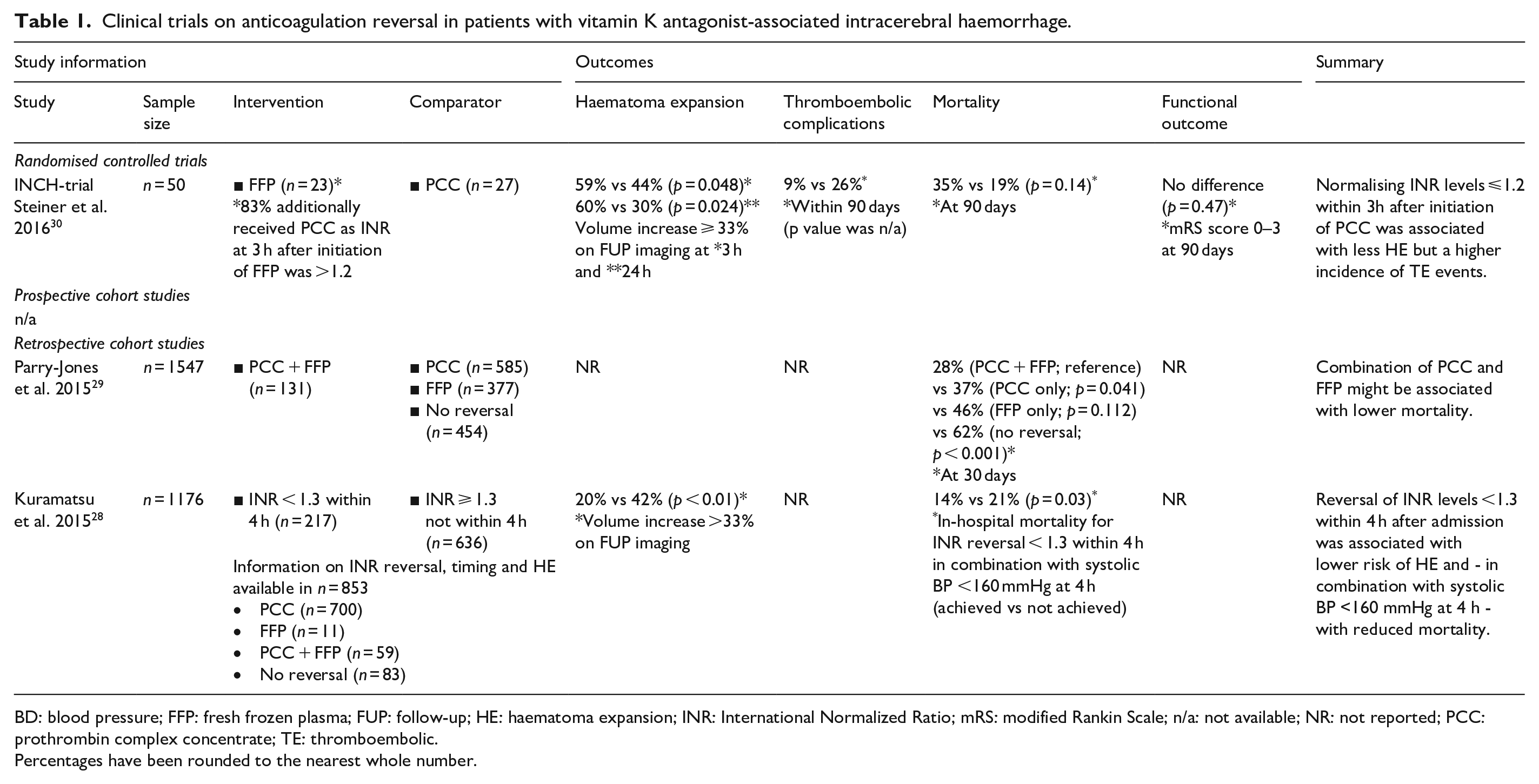
BD: blood pressure; FFP: fresh frozen plasma; FUP: follow-up; HE: haematoma expansion; INR: International Normalized Ratio; mRS: modified Rankin Scale; n/a: not available; NR: not reported; PCC: prothrombin complex concentrate; TE: thromboembolic.
Percentages have been rounded to the nearest whole number.
Reversal of direct thrombin inhibitor therapy in patients with intracerebral haemorrhage
Dabigatran is a direct thrombin inhibitor 32 used for the prevention of stroke and systemic embolism in patients with atrial fibrillation as well as for the prevention and treatment of venous thromboembolism and other thromboembolic conditions. Idarucizumab is a monoclonal antibody, developed to reverse the anticoagulant effect of dabigatran. 33 Its mechanism of action is described elsewhere (Christensen al. ESJ).
There are no data from randomised controlled trials or cohort studies assessing the comparative safety or effectiveness of idarucizumab compared to placebo, standard of care or any other comparator in patients with dabigatran-related intracerebral haemorrhage. Idarucizumab was approved by the FDA, EMA and other regulatory bodies based on the results of a prospective case series (REVERSE-AD). 33 REVERSE-AD enrolled patients on dabigatran treatment who experienced major bleeding or who required major surgery, including 98 patients with intracranial bleeding, of whom 53 had intracerebral haemorrhage. All patients received reversal treatment with idarucizumab. The primary outcome was reversal of dabigatran, assessed by the change in diluted thrombin time or ecarin clotting time which was achieved in more than 98% of the patients. Secondary clinical endpoints, such as mortality and thromboembolic events, were generally favourable (see Table 2), although this assessment was not restricted to patients with intracerebral haemorrhage and lacked a control group. Therefore, it is impossible to conclude whether the observed outcomes were a result of effective reversal with idarucizumab or the natural outcome of patients with thrombin inhibitor-associated ICH. Following market approval, data from a retrospective case series from Germany 34 provided additional evidence supporting the use of idarucizumab (Table 2). Overall, the reversal of dabigatran using idarucizumab was rapid and sustainable, clinical outcomes were favourable, and the incidence of thromboembolic events was low. However, some of the outcomes were not reported separately for intracerebral haemorrhage and a treatment comparator was not present (including no reversal) making the interpretation of these results difficult. Moreover, data on haematoma expansion and long-term functional outcomes in patients with intracerebral haemorrhage were unavailable.
Clinical trials on anticoagulation reversal in patients with dabigatran-associated intracerebral haemorrhage.

HE: haematoma expansion; mRS: modified Rankin Scale; n/a: not available; NR: not reported; TE: thromboembolic.
Percentages have been rounded to the nearest whole number.
Reversal of factor Xa inhibitor therapy in patients with intracerebral haemorrhage
The factor Xa inhibitors apixaban, 35 edoxaban 36 and rivaroxaban 37 are approved for the prevention of stroke and systemic embolism in patients with non-valvular atrial fibrillation as well as for the prevention and treatment of other thromboembolic conditions. Expert opinion 13 and guidelines38–41 have recommended the use of 4-factor prothrombin complex concentrate for the reversal of factor Xa inhibitor-associated intracerebral haemorrhage since their market approval. Tranexamic acid, an antifibrinolytic drug used in different conditions of major bleeding, has been considered a potential add-on therapy for factor-Xa inhibitor associated haemorrhages. 41 Andexan alfa is a modified recombinant inactive form of human factor Xa developed for the reversal of factor Xa inhibitors. 42 Its mechanism of action is described elsewhere (Christensen al. ESJ).
Although 4-factor prothrombin complex concentrate is recommended for the treatment of factor Xa inhibitor-associated intracerebral haemorrhage, there is currently no evidence from randomised controlled trials to support this recommendation. Two recent retrospective cohort studies from Germany 43 and Hong Kong 44 assessed the association of 4-factor prothrombin complex concentrate with haematoma expansion and clinical outcomes following factor Xa inhibitor-associated intracerebral haemorrhage. Both studies found no evidence that 4-factor prothrombin complex concentrate treatment might improve radiological or clinical outcomes, yet it should be mentioned that the administered PCC dosages were lower than guideline-adherent recommendations (Table 3). Add-on treatment with tranexamic acid was tested in a small, phase-II, placebo-controlled, randomised controlled trial from Switzerland (TICH-NOAC), with 65% of patients receiving additional 4-factor prothrombin complex concentrate. 45 The trial did not identify any benefit or harm of tranexamic acid treatment, although it was likely underpowered. Andexan alfa is approved by the FDA, EMA and other regulatory bodies based on results of a prospective case series that included patients with major bleeding (ANNEXA-4; including 227 patients with intracranial bleedings of whom 99 had non-traumatic intracerebral haemorrhage) who had received andexan alfa.42,46 This study did not include any control group. Subsequently, matched comparisons of patients with intracerebral haemorrhage from the ANNEXA-4 population with a retrospective German cohort study 43 and with the TICH-NOAC 47 study population have been performed. Both studies showed a significantly lower incidence of haematoma expansion in patients who received andexan alfa without an increased risk for thromboembolic complications (Table 3). However, this did not translate to any difference in mortality or functional outcome between the matched cohorts. Finally, a randomised controlled trial (ANNEXA-I) compared the safety and efficacy of andexan alfa with usual care (86% of patients received 4-factor prothrombin complex concentrate in the usual care arm) in patients with intracerebral haemorrhage. 48 ANNEXA-I was stopped prematurely after a pre-planned interim analysis for efficacy. 48 In ANNEXA-I, treatment with andexan alfa resulted in a significantly higher haemostatic efficacy (i.e. a lower incidence of symptomatic haematoma expansion) by 13.4% (in the prespecified interim analysis of the primary efficacy end point) and 11.0% (in the full trial population) absolute difference to usual care. 48 However, treatment with andexan alfa resulted in a significant increase in thromboembolic events (10.3%) compared to usual care (5.6%). 48 There was no difference in mortality or functional outcome, but the trial was not powered for these endpoints, which were assessed after an observational period of only 30 days.
Clinical trials on anticoagulation reversal in patients with factor Xa inhibitor-associated intracerebral haemorrhage.
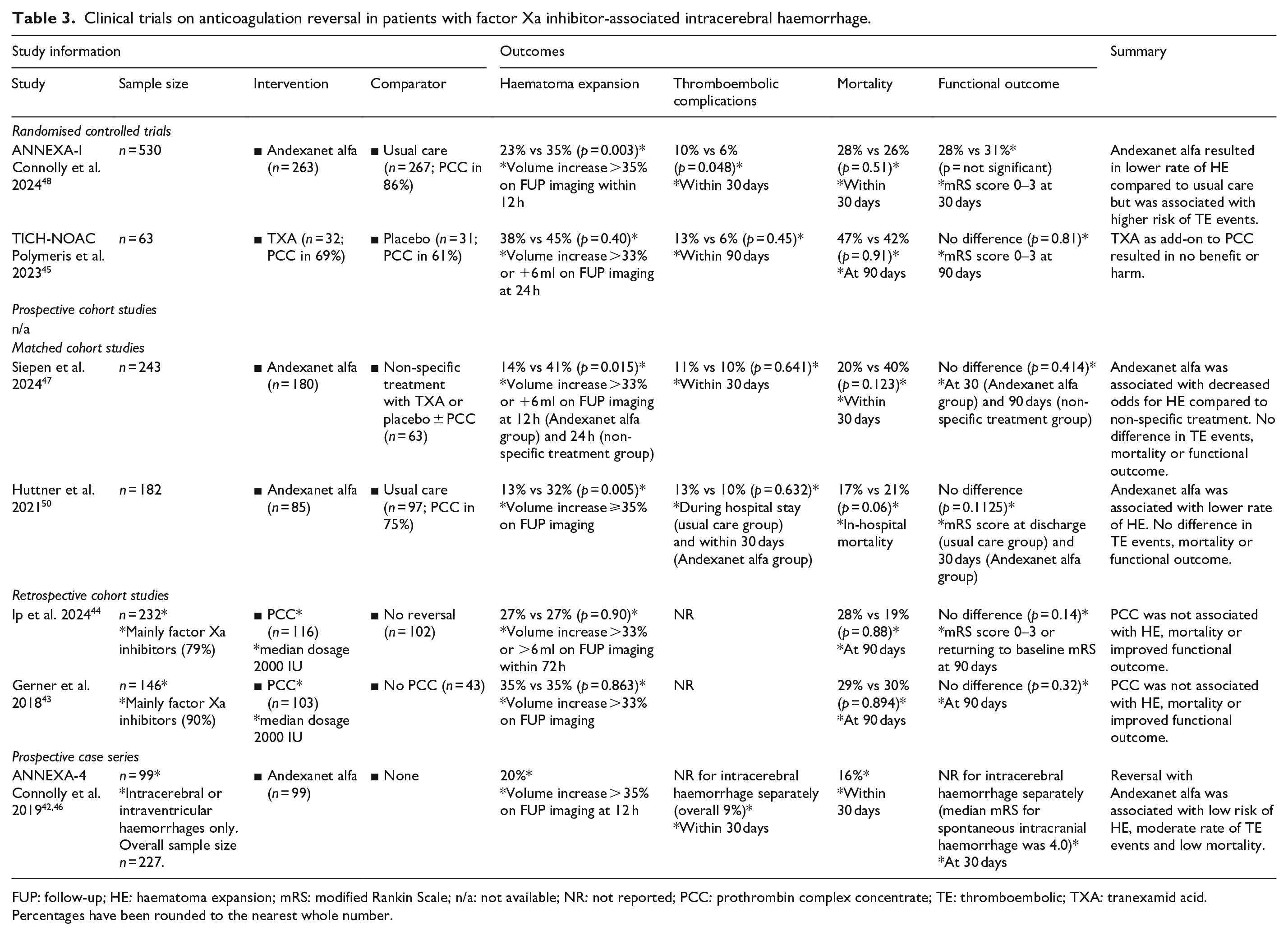
FUP: follow-up; HE: haematoma expansion; mRS: modified Rankin Scale; n/a: not available; NR: not reported; PCC: prothrombin complex concentrate; TE: thromboembolic; TXA: tranexamid acid.
Percentages have been rounded to the nearest whole number.
Summary and conclusions
Rapid, targeted and effective reversal of anticoagulation is a pivotal component in the hyperacute bundle of care aimed at preventing haematoma expansion in patients with anticoagulation-associated intracerebral haemorrhage. 21 In summary, there is a growing body of evidence supporting the benefits of the various reversal agents for the different types of anticoagulation-associated intracerebral haemorrhage (Figure 1). While two randomised controlled trials have provided support for the reversal treatment of vitamin K antagonist-associated and factor Xa-associated intracerebral haemorrhage, their findings are constrained by small sample sizes and reliance on surrogate imaging outcomes as primary endpoints. Additionally, variations in imaging definitions, haematoma volume assessment, study design, and laboratory endpoints across reversal studies further limit comparability (see explanatory notes in Tables 1–3). Despite these factors, haematoma expansion has consistently been shown to be significantly associated with poor outcome 49 and therefore seems a valid endpoint for haemostatic trials.
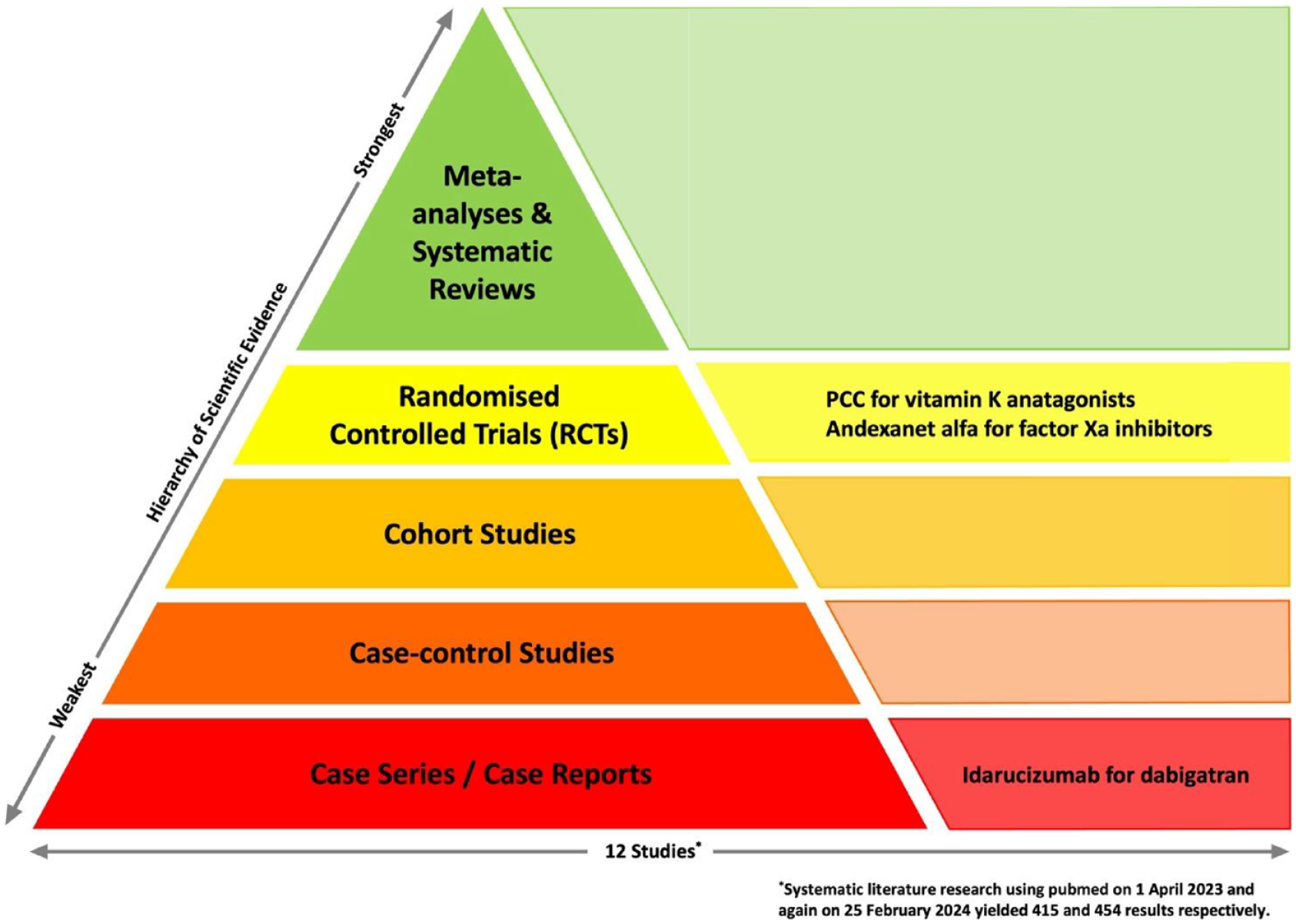
Pyramid of evidence for reversal treatment of different types of anticoagulation-associated intracerebral haemorrhage.
Finally, no trial was sufficiently powered to show an effect of anticoagulation reversal on meaningful clinical outcomes such as functional outcome or death. Given that reversal of anticoagulation may result in an increased rate of thromboembolic events, further research needs to focus on the clinical impact of competing events associated with reversal treatments, ideally offering a balanced appraisal of the net treatment effect, namely the benefit from prevention of haematoma expansion versus the potential harm from a possibly increased risk of thromboembolic events. There is a clear need to identify patients who are most likely to benefit from anticoagulation reversal and those who are more vulnerable to experience thromboembolic complications without gaining significant benefits.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DS received research funding from the Swiss National Science Foundation, the Swiss Heart Foundation, the Bangerter-Rhyner Foundation and AstraZeneca. DS received fees paid to his institution for consulting and speaker for AstraZeneca, Pfizer, VarmX and Bioxodes. BMS received research funding from the Swiss Academy of Medical Sciences/Bangerter-Rhyner Foundation, the Swiss Heart Foundation and the Inselspital Department of Teaching and Research. All other co-authors have nothing to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
No ethics approval needed.
Informed consent
No informed consent needed.
Guarantor
DS
Contributorship
DS and BMS researched literature, conceived the paper and wrote the first draft manuscipt. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
