Abstract
Introduction:
Reperfusion therapies represent promising treatments for patients with Central Retinal Artery Occlusion (CRAO), but access is limited due to low incidence and lack of protocols. We aimed to describe the benefit of implementing a Retinal Stroke-Code protocol regarding access to reperfusion, visual acuity and aetiological assessment.
Patients and methods:
Prospective cohort study performed at a Comprehensive Stroke Centre. Criteria for activation were sudden monocular, painless vision loss within 6 h from onset. Eligible patients received IAT when immediately available and IVT otherwise. All patients were followed by ophthalmologists to assess best-corrected visual acuity (BCVA) and visual complications, and by neurologists for aetiological workup. Visual amelioration was defined as improvement of at least one Early Treatment Diabetic Retinopathy Study (ETDRS) letter from baseline to 1 week.
Results:
Of 49 patients with CRAO, 15 (30.6%) received reperfusion therapies (12 IVT, 3 IAT). Presentation beyond 6 h was the main contraindication. Patients receiving reperfusion therapies had better rates of visual improvement (33.3% vs 5.9%, p = 0.022). There were no complications related to reperfusion therapies. Rates of neovascular glaucoma were non-significantly lower in patients receiving reperfusion therapies (13.3% vs 20.6%, p = 0.701). Similar rates of atherosclerotic, cardioembolic and undetermined aetiologies were observed, leading to 10 new diagnosed atrial fibrillation and five carotid revascularizations.
Conclusion:
A comprehensive acute management of CRAO is feasible despite low incidence. In our study, reperfusion therapies were safe and associated with higher rates of visual recovery. A similar etiological workup than ischemic stroke led to of high proportion of underlying aetiologies.
Keywords
Introduction
Central retinal artery occlusion (CRAO) represents a rare event with reported incidence that varies between different countries from 1 to 5.8 cases per 100,000 inhabitants per year, according to different reports.1–3 It has been published to represent around one in 10,000 ophthalmological outpatients’ visits. 4 CRAO diagnosis is mainly done by ocular fundus examination, which typically shows retinal whitening, cherry red spot in fovea, and arteriolar attenuation with segmentation of blood in retinal arterioles. In the earlier stages of CRAO the fundus examination may appear normal, as ischemic intracellular oedema causes a time-dependent increase in retinal thickness. 5 Classic findings in fundus examination are shown in Supplemental Figure 1.
CRAO shares risk factors and aetiology with stroke. 6 Therefore, investigation of these patients may result in diagnosis of unknown vascular risk factors and consequent optimization of secondary prevention, 7 which has especial relevance due to the high risk of suffering a stroke, especially within the first month, that CRAO entails.8,9 A recent meta-analysis showed that up to 30% of patients with acute CRAO and 25% of patients with acute BRAO (branch retinal artery occlusion) presented acute cerebral ischemia on MRI, reflecting the importance of early assessment of these patients. 10 CRAO aetiology resembles ischemic stroke, being large artery atherosclerosis (LAA) the most frequent underlying cause, followed by cardiogenic embolism (CE) and other uncommon causes. 11 Despite a complete etiological workup, the underlying CRAO aetiology remains undetermined in around 40% of the cases.8,9
Standard ocular management has not demonstrated efficacy and could even be harmful. 12 As an alternative, intravenous thrombolysis (IVT) within the first 4.5 h symptoms onset has demonstrated to improve visual recovery in small clinical trials and meta-analysis. 13 Intraarterial thrombolysis (IAT) (consisting in microcatheterization of the ophthalmic artery and in situ infusion of alteplase) remains an alternative for patients with contraindications for systemic IVT, providing also potential advantages, such a lower total dose and a presumed longer time-window for effectivity.14,15 For both therapies, an early and accurate diagnosis of CRAO is crucial to improve visual outcomes. To facilitate the conduction of future clinical trials and the later implementation of those therapies, algorithms and strategies for rapid and accurate diagnosis are lacking. Therefore, we aimed to describe evolution of visual acuity, complications and assessment of underlying aetiology in daily clinical practice, after the implantation of a retinal stroke-code protocol in a comprehensive stroke centre with ophthalmologic emergencies.
Patients and methods
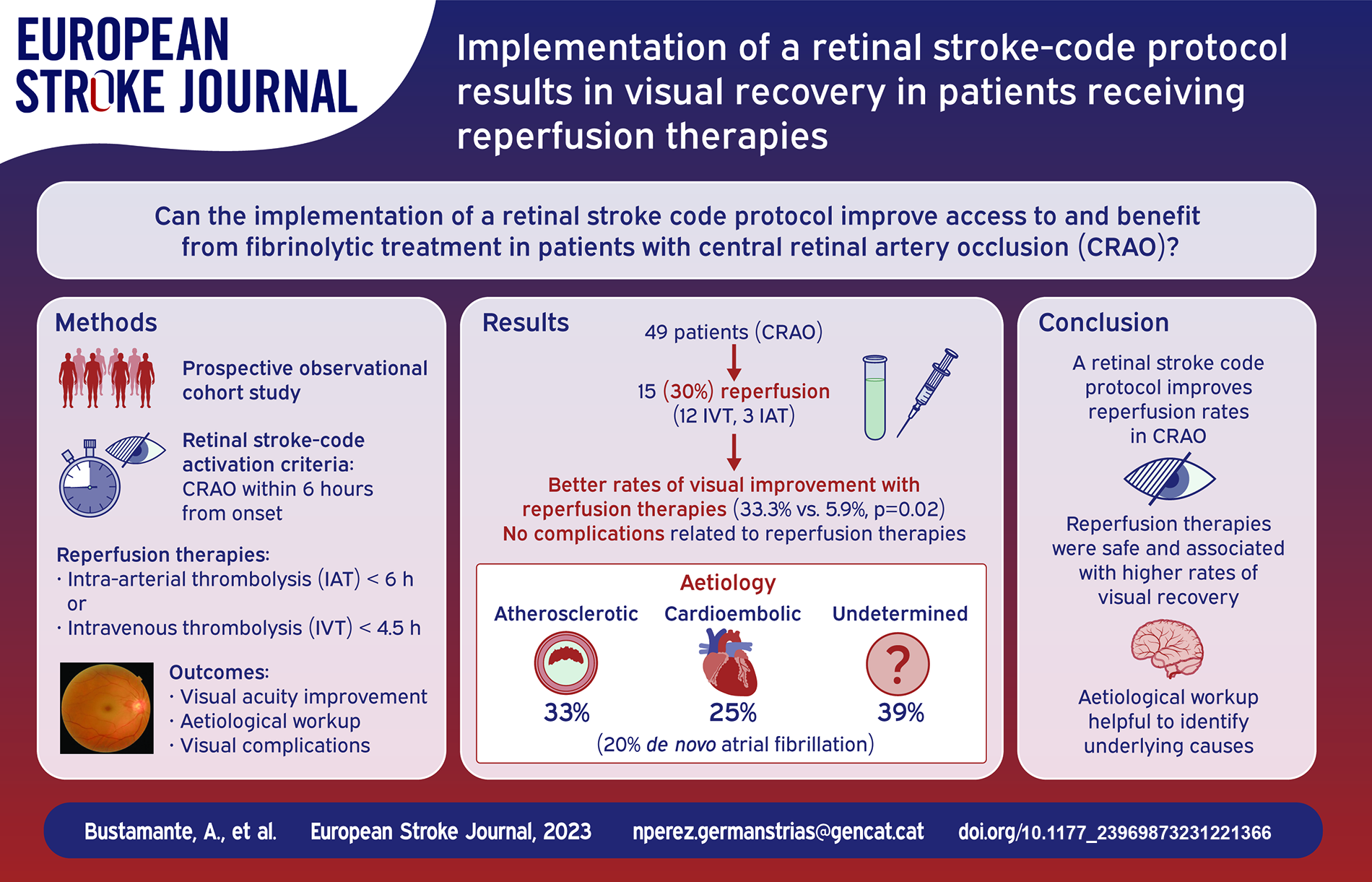
Based on the accumulated evidence of acute management of CRAO, a consensual multidisciplinary protocol was developed by Neurology, Ophthalmology, Radiology and Emergency departments, for emergent care assessment of patients presenting with a suspected CRAO. The protocol was evaluated and approved by the internal Quality Department of Hospital Universitari Germans Trias i Pujol (UIT-PT-013) and is summarized in Figure 1. Patients were considered for reperfusion when CRAO diagnosis was made with a baseline best corrected visual acuity (BCVA) ⩽ 20/400, in the absence of other underlying ophthalmological diseases justifying visual loss. Time window was considered within 4.5 h after symptom onset for IVT and within 6 h for IAT. Criteria for retinal stroke-code activation are summarized in Supplemental Table 1.
Standardized protocol for retinal stroke-code activation.
At baseline, BCVA was measured by early treatment diabetic retinopathy study (ETDRS) chart at 20 feet in letters for all patients, except for those unable to see any letter, that were evaluated on an ordinal categorical scale from counting fingers (3 letters) and hand movement (2 letters), light perception (1 letter) and to non-light perception (0 letters), giving to each one a numerical punctuation according to the Freiburg Visual Acuity Test as described by Kilian Schulze-Bonsel et al. 16
After CRAO diagnosis, neurological and systemic clinical examination, routine blood tests, electrocardiogram, brain computed tomography (CT) and CT-angiography were performed. Written informed consent was obtained from all patients or representatives before reperfusion. Regarding treatment modality, IAT was preferred when immediately available or in patients not eligible for IVT. Alteplase dose and contraindications for IVT were the same as used for ischemic stroke. Exclusion criteria for IAT are summarized in Supplemental Table 2. IAT was performed by experienced neurointerventionists under conscious sedation. Alteplase was locally administered through a microcatheter placed in the ophthalmic artery by infusion of 3 mg bolus every 5 min up to a maximum dose of 20 mg. In all CRAO patients, an etiological workup like ischemic stroke was done during hospital admission and continued later if necessary. Initial workout included a vascular imaging of the neck and intracranial vessels, electrocardiogram, echocardiography, 24-h cardiac rhythm monitoring, and conventional blood tests. Whenever the basic workup was inconclusive, further diagnostic tests such as patent foramen ovale assessment, extensive blood tests including coagulopathies, prolonged cardiac monitoring with wearable devices or transesophageal echocardiography were performed. Stroke aetiology was classified according to the trial of ORG 10172 in acute stroke treatment (TOAST) classification system. 17
All patients included were followed by ophthalmologists in 24 h, 1 week, 2 weeks, 1 month, 3 months, 6 months and 1 year in order to detect long term ocular neovascularization presentation. In each visit, the data recorded were BCVA in ETDRS chart, intraocular pressure, relative afferent pupillary defect (RAPD), funduscopic examination, gonioscopy, angiography-OCT (OCT-A) for retinal perfusion, and finally at month three fundus fluorescein angiography (FA) or previously if signs of ischemia were found. Visual amelioration was defined as improvement of at least one ETDRS letter from baseline to 1 week. In addition to any visual improvement, we also classified improvement as moderate (>5 letters) and notable (>15 letters). These categories were not exclusive, and therefore patients with notable visual recovery were also classified as moderate and any visual improvement. Complications such as neovascular glaucoma (NVG) by gonioscopy and slit-lamp examination were registered as well.
All patients were included in a local prospective registry of acute stroke patients, including retinal occlusion linked to the electronic medical system of the hospital and including baseline and follow-up data.
Statistical analysis
Statistical analysis was performed with Statistical Packages for Social Sciences (SPSS) software, version 24. Categorical variables were expressed as number (percentage), and continuous variables as mean ± standard deviation or median (interquartile range) depending on data distribution, which was assessed using Kolmogorov-Smirnoff test. Intergroup comparisons were performed between patients receiving or not any reperfusion treatment, as well as and between patients experiencing or not visual recovery improvement at any degree. For intergroup comparisons, Chi-squared test for categorical variables and Student’s t test or Mann–Whitney U test for continuous variables, depending on data distribution, were used.
Results
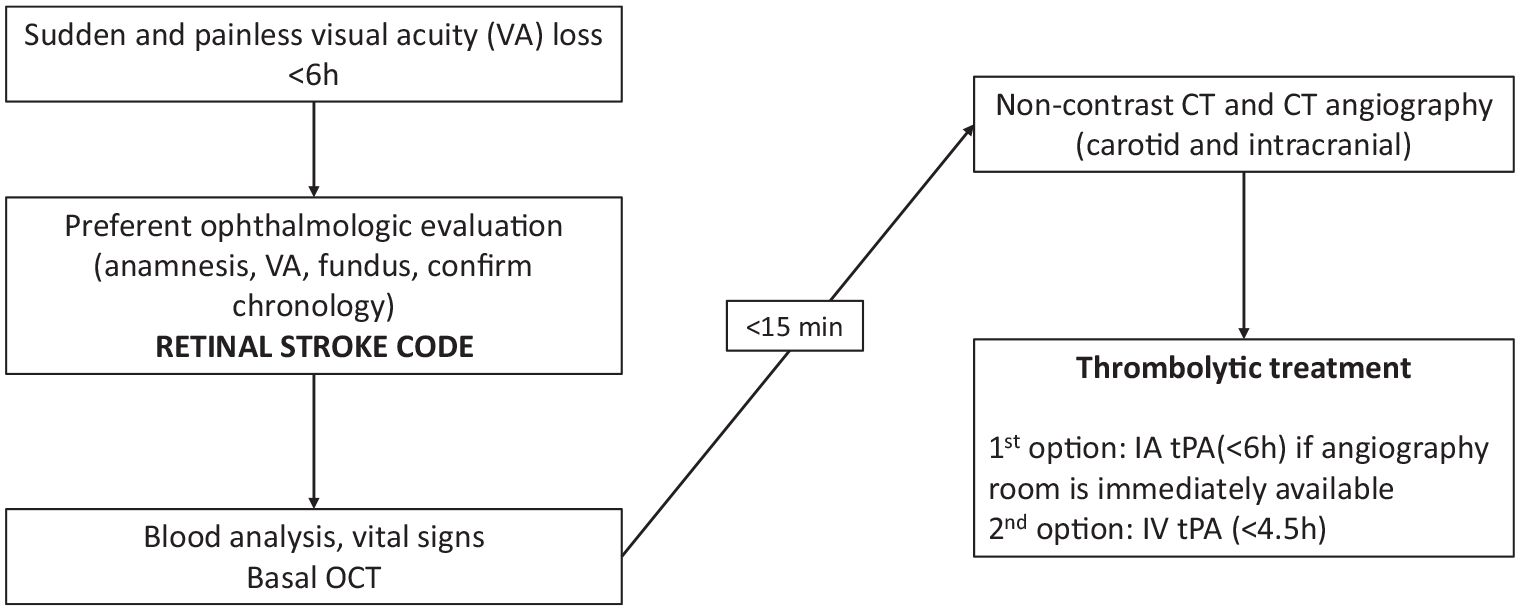
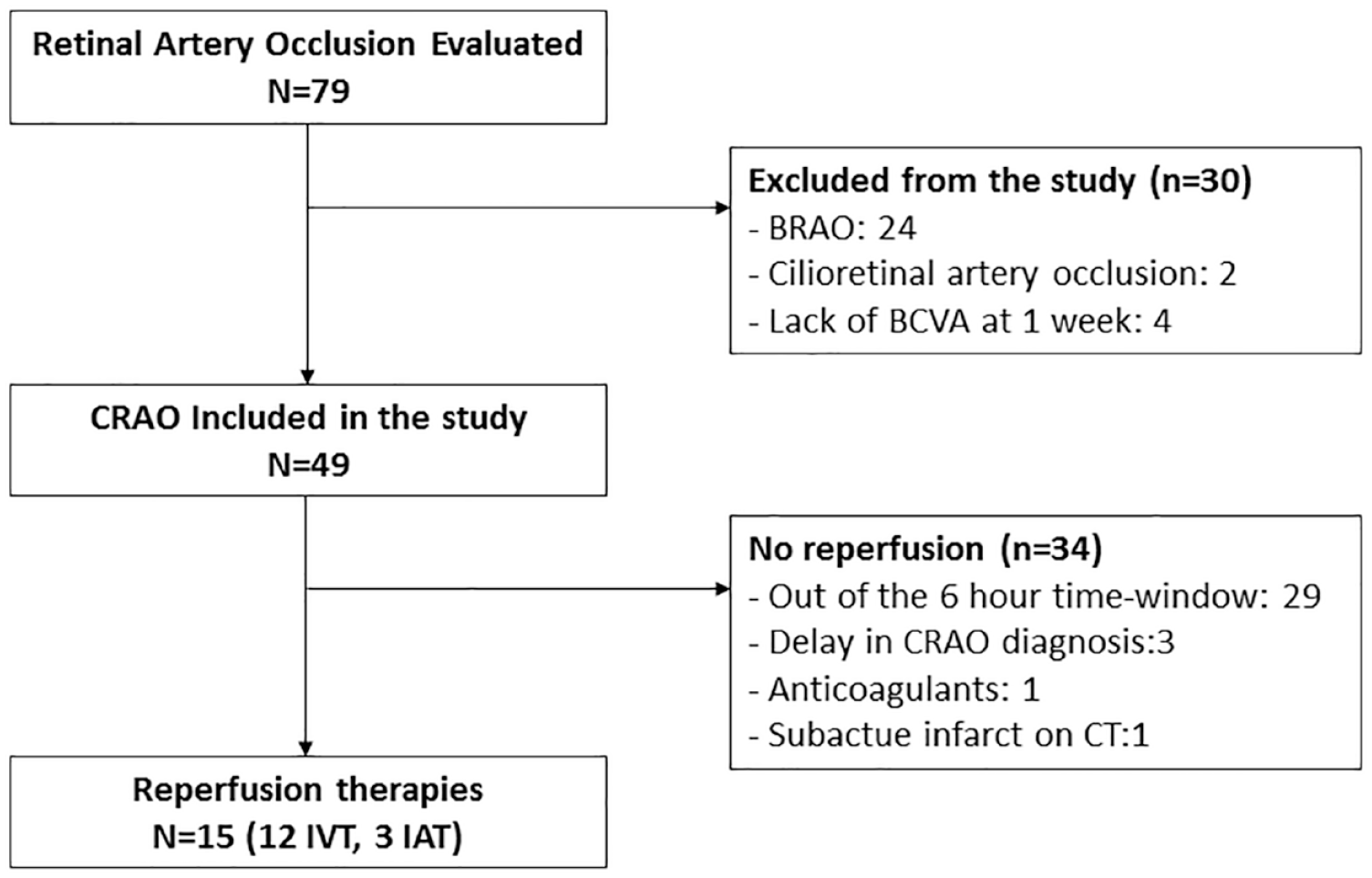
From March-2018 to February-2023, 79 patients with a diagnosis of CRAO or BRAO were enrolled among 79,268 ophthalmologic emergencies. After excluding patients with branch or ciliorretinal artery occlusions and those lost to follow-up, BCVA at 1 week was available for 49 patients, and those were finally included in the analyses (Figure 2). Mean age was 75 years and 49% were women. Hypertension (69.4%) and dyslipidaemia (59.2%) were the most prevalent vascular risk factors (Table 1). Mean time from symptoms onset to hospital arrival was 315 (123–1409) min. Of the included patients, 15 (30.6%) received reperfusion therapies (12 IVT, 3 IAT), being more than 6 h from symptom onset the main contraindication. Median door-to-needle time in patients receiving IVT was 113.5 (65.5–137) min and median door-to-groin puncture time in patients receiving IAT was 177 (143.5–203) min. Overall, 1 week after the presentation, BCVA improved from baseline in seven cases and worsened in six. Improvement was moderated (>5 letters) in five patients and notable (>15 letters) in four patients, which improved 60, 60, 75 and 75 letters respectively.
Flowchart.
Demographics, baseline clinical information and differences between patients receiving reperfusion therapies or not.
AF: atrial fibrillation; CHF: congestive heart failure; mRS: modified Rankin score; SBP: systolic blood pressure; DBP: diastolic blood pressure; TOAST: trial of ORG 10172 in acute stroke treatment; LAA: large artery arteriosclerosis; CE: cardiac embolism; UND: undetermined. BCVA: best corrected visual acuity.
Data are expressed as number (percentage), median (interquartile range) or mean ± standard deviation, as appropriated.
Moderate (>5 letters), Notable (>15 letters).
Comparison between patients receiving or not reperfusion therapies
There were no significant differences regarding demographics, risk factors, and baseline characteristics between patients receiving or not reperfusion therapies. Patients treated with reperfusion therapies arrived at the hospital earlier (103 (75–154) vs 845 (250–2406), p = 0.001) and trended to have worse baseline BCVA (2 (2–5) vs 4.5 (2–18), p = 0.055). The rates of any visual improvement (33.3% vs 5.9%), as well as moderated (26.7% vs 2.9%) and notable (26.7% vs 0%) visual improvement were significantly higher in patients receiving reperfusion therapies (Table 1).
Factors related to visual improvement
There were no significant differences regarding demographics, risk factors or baseline data, between patients experiencing or not visual improvement at 1 week, but patients who improved trended to be older and to have higher rates of atrial fibrillation (AF). As mentioned above, patients who improved at 1 week received reperfusion therapies more frequently (71.5% vs 30.6%) and time from symptom onset to hospital admission trended to be shorter (180 (50–376) vs 489 (127–1686), p = 0.130) (Table 2).
Demographics, baseline clinical information and differences between patients experiencing or not visual improvement at 1 week.
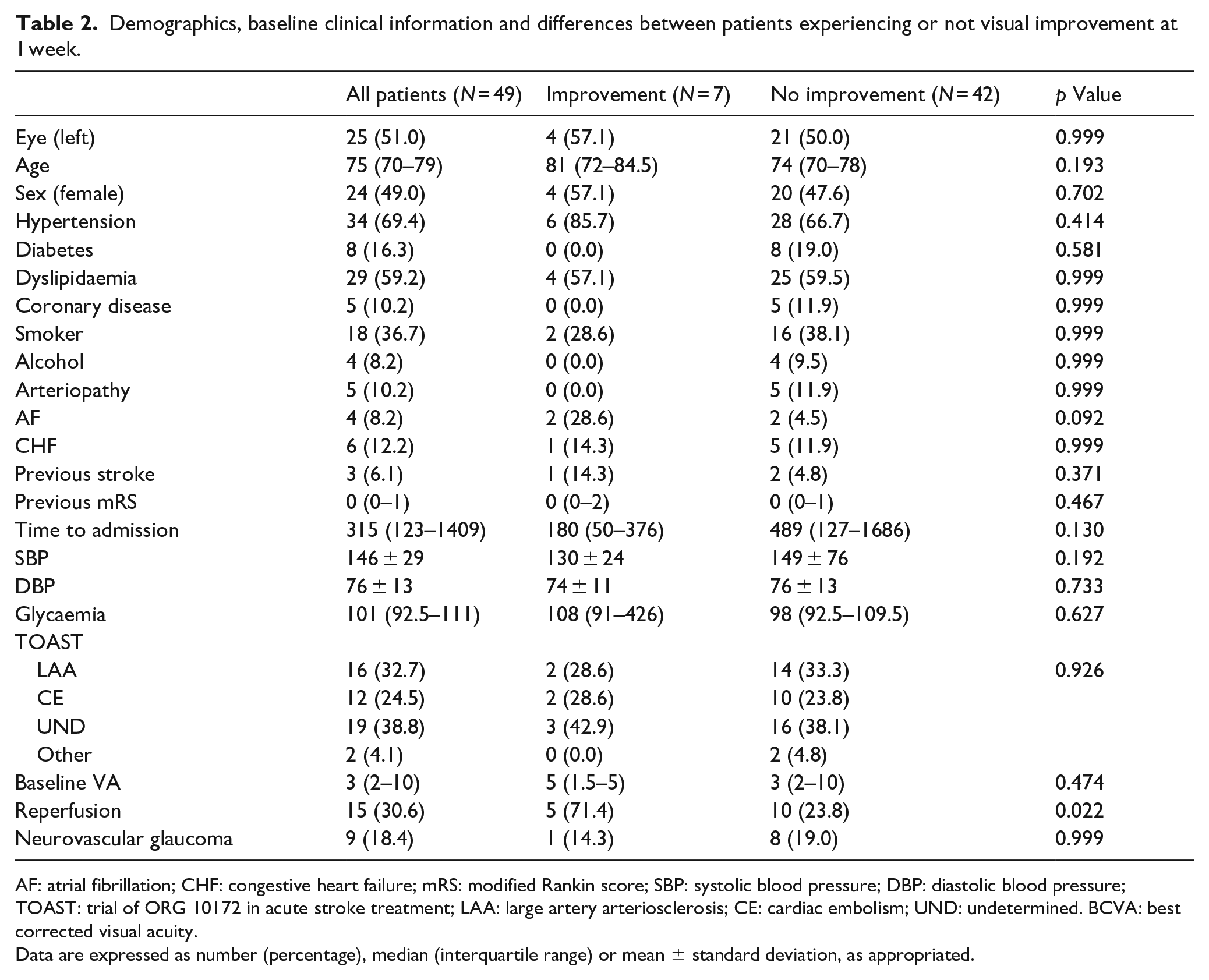
AF: atrial fibrillation; CHF: congestive heart failure; mRS: modified Rankin score; SBP: systolic blood pressure; DBP: diastolic blood pressure; TOAST: trial of ORG 10172 in acute stroke treatment; LAA: large artery arteriosclerosis; CE: cardiac embolism; UND: undetermined. BCVA: best corrected visual acuity.
Data are expressed as number (percentage), median (interquartile range) or mean ± standard deviation, as appropriated.
Aetiology of CRAO
After in- and outpatient etiological workup, we observed as causal factors similar rates of large vessel atherosclerosis, cardiac embolism and undetermined aetiologies. Out of 19 CRAO of undetermined origin, 14 cases were cryptogenic under complete evaluation. More than one causal factor was identified in three cases and the diagnostic workup was considered incomplete in two cases. Large-vessel atherosclerosis (16, 32.7%) was classified as probable in 12 cases and possible (due to <50% stenosis or aortic arch atheroma) in four cases. Cardiac embolism (12, 24.5%) was classified as probable in 11 cases, all of them due to AF, and possible in a case of severe mitral valve calcification. Just two patients had other determined causes, specifically a case of giant cell arteritis and a case of antiphospholypid syndrome. Figure 3 represents the distribution of the TOAST subtypes in the cohort. Of note, the etiological workup resulted in 10 cases of “de novo” AF diagnoses and five carotid revascularization procedures.
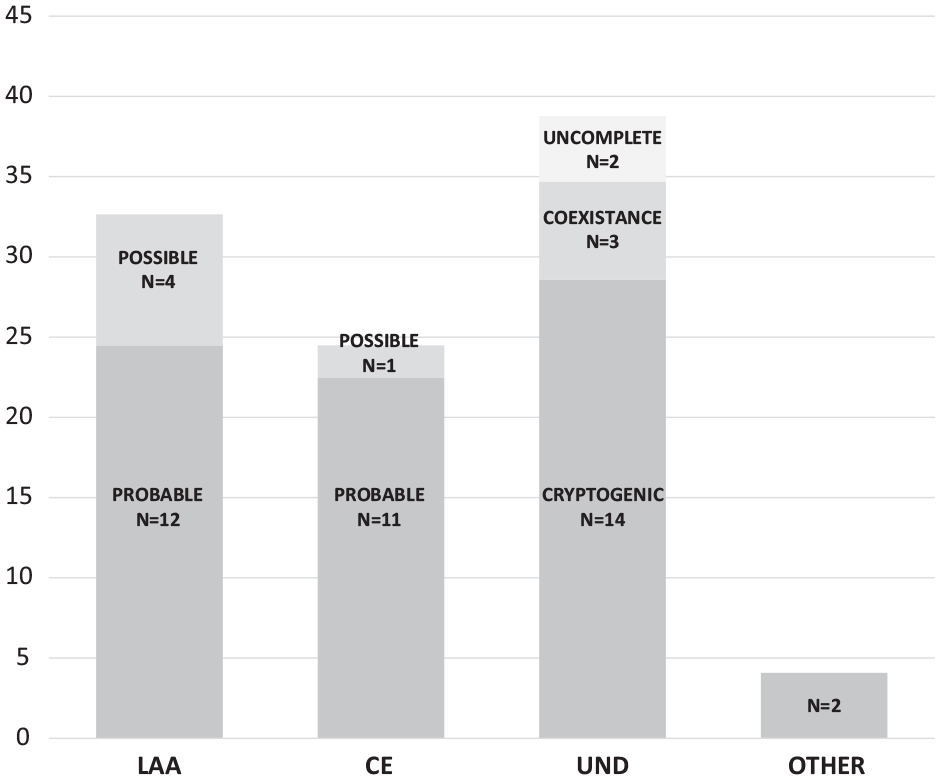
CRAO aetiologies.
Complications
There were no complications related with reperfusion therapies, such as haemorrhagic transformation, angioedema, or procedure related. One patient had a recurrent stroke during hospitalization. During follow-up, NVG was diagnosed in nine cases (18.4%). The median time for NVG development was 70 days, ranging from 19 to 210 days. A total of 66.6% of NVGs appeared in patients with previous history of diabetes, despite none of them had proliferative diabetic retinopathy hypoxia. Rates of NVG were non-significantly lower in patients receiving reperfusion therapies (13.3% vs 20.6%), p = 0.701) (Table 1).
Discussion
The present study represents a 5-year experience in a comprehensive stroke centre with 24-h ophthalmologic emergencies. The main findings of the present study, based on real-life clinical practice, can be summarized as follows: first, a comprehensive management of CRAO patients, similar to acute ischemic stroke, is feasible despite the low incidence. Second, reperfusion therapies were safe and associated with higher rates of visual recovery in our study, and seems to have a protective role of NVG development. Third, a similar etiological workup that ischemic stroke led to the identification of the underlying aetiology in a high proportion of the cases.
Despite increasing evidence of the usefulness of reperfusion therapies in the acute phase of CRAO, usual systems of care able to rapidly identify and treat CRAO patients are lacking. In fact, the main reason against the application of reperfusion therapies in our study was a first evaluation beyond 6 h. However, in our study we were able to perform higher rates of thrombolysis than previous studies. 18 The use of pre-existing code stroke systems represents a unique opportunity for the early identification of this condition at hospital arrival, integrating a multidisciplinary team including ophthalmologists, stroke neurologists and radiologists, as well as trained emergency nurses able to identify unilateral, painless vision loss a guiding symptom. 19 Also, pre-hospital stroke code circuits could be also adapted to CRAO, triaging and allocating patients with suspected CRAO to stroke centres able to provide at least IVT in a 24/7 manner and stroke unit admission, as well as ophthalmologic emergencies 24/7. To increase also the awareness of the general population about the condition, adding a “B” for balance and an “E” for eyes to the FAST (Face, Arm, Speech, Time) acronym, resulting in “BE-FAST,” has been proposed to improve the recognition of this entity. 20
The most interesting result of the present study is the magnitude of the effect of reperfusion on visual recovery, despite the small sample size. Despite a relative lack of evidence and phase III clinical trials, the efficacy of IVT in the treatment of CRAO has been demonstrated through phase II clinical trials, and meta-analyses at patient-level based on small cohorts. 13 These studies have reported safety, as well as higher rates of visual recovery in patients receiving IVT, especially within 4.5 h after symptom onset, although a recent multicentre, retrospective study with a large cohort of 47 patients treated with IVT showed no evidence of superiority for IVT. 21 In addition to the retrospective character of this study, it include also important differences with our design, with different inclusion criteria regarding VA at baseline and longer therapeutic window (median onset-to-treatment time around 4.5 h). The development of a coordinated protocol with strict criteria, as well as the use of pre-existing code-stroke networks and systems to provide acute care to CRAO patients might be the one of the reasons for the positive results observed in our experience. Regarding endovascular treatment, the EAGLE clinical trial evaluated the efficacy and safety of IAT in the treatment of OACR. 15 However, the study was interrupted for safety concerns such major bleedings in the IAT group. A large (12 h) time-window for enrolment and the concomitant use of heparin were the main criticisms to this study. A recent meta-analysis suggests that a 6-h time-window has the potential of improving BCVA in CRAO, which should be balanced against procedure-related complications. 19 A short time window, a mandatory neuroimaging protocol, admission to a specialized stroke unit and a mandatory fulfilment of the reperfusion protocol that do not allow the use of any antithrombotic within the first 24 h after reperfusion might explain the absence of haemorrhagic complications in our study.
Moreover, after a long-term follow-up, our study shows a trend towards lower rates of NVG in patients receiving reperfusion therapies (13.3%). The rates of NVG are within previous reported rates, such as Rudkin et al. (15.2%) and Mason et al. (12%).7,22 In accordance with previous reports, we also found an association with diabetes mellitus as a risk factor of NVG development, suggesting that a close follow-up of long-term complications after CRAO might be recommended in patients with diabetes. After careful review, we noticed that NVG was presented in two patients in which this complication could not be attributed to reperfusion. The first patient received endovascular treatment; however, the ophthalmic artery could not be catheterized and, therefore, IAT was not administered. The second case suffered severe ischemic ocular arterial syndrome secondary to short posterior ciliary artery occlusion 10 days after the thrombolysis in the CRAO presentation and was judged to be responsible for the complication rather than the initial CRAO. Therefore, this could support the fact that reperfusion therapy could be protective of development of long-term ocular complications, even in the coexistence of diabetes mellitus. Future clinical trials and registries should include assessment of retinal ischemia and NVG in the long term to explore the association with acute reperfusion therapies.
The last interesting finding of our study is given by the high rates of cardioembolic aetiology (up to 28.3%), higher than previous reports. In fact, in a recent meta-analysis, pooled prevalence of AF in CRA has been estimated in 11.5%. 23 A recent observational study showed that, in people wearing cardiac monitoring devices in situ after CRAO, the rate of atrial fibrillation detection at 2 years was significantly higher than in matched controls, and comparable to patients with ischemic stroke. 24 An early stroke unit admission and an etiological workup similar to stroke patients allowed us to identify the underlying cause in around 2/3 of the cases, including 10 cases of “de novo” AF diagnoses and five carotid revascularization procedures, therefore improving stroke secondary prevention in this population.
The present study is not free of limitations. First, the small sample size, especially in the group of patients treated with IAT, did not allow us to explore the effect of IVT and IAT independently. The small sample size also precluded us from conducting logistic regression analyses on parameters such as visual improvement. Second, we cannot forget that the present study does not represent a randomized double blinded clinical trial, and treatments were applied based on clinical indications from a consensus protocol. Therefore, the validity of the comparisons is just relative.
Conclusion
In conclusion, our experience supports the establishment of hospital-based systems of care for CRAO, able to provide safe reperfusion therapies in a timely manner leading to higher rates of visual recovery, as well as an etiological workup.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231221366 – Supplemental material for Implementation of a retinal stroke-code protocol results in visual recovery in patients receiving reperfusion therapies
Supplemental material, sj-docx-1-eso-10.1177_23969873231221366 for Implementation of a retinal stroke-code protocol results in visual recovery in patients receiving reperfusion therapies by Alejandro Bustamante, Marta Balboa, Garbiñe Ezcurra, Adrián Sánchez-Fortún, Judith Ruiz, Jordi Castellví, Susana Castillo-Acedo, Èric Matas, Rachid Bouchikh, Marina Martínez-Sánchez, Carlos Castaño, Sebastiá Remollo, Mariano Werner, Maria Carmen Salgado, Samuel Villodres, Mireia Gea, Mònica Millán, Natàlia Pérez de la Ossa and Susana Ruiz-Bilbao in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873231221366 – Supplemental material for Implementation of a retinal stroke-code protocol results in visual recovery in patients receiving reperfusion therapies
Supplemental material, sj-docx-2-eso-10.1177_23969873231221366 for Implementation of a retinal stroke-code protocol results in visual recovery in patients receiving reperfusion therapies by Alejandro Bustamante, Marta Balboa, Garbiñe Ezcurra, Adrián Sánchez-Fortún, Judith Ruiz, Jordi Castellví, Susana Castillo-Acedo, Èric Matas, Rachid Bouchikh, Marina Martínez-Sánchez, Carlos Castaño, Sebastiá Remollo, Mariano Werner, Maria Carmen Salgado, Samuel Villodres, Mireia Gea, Mònica Millán, Natàlia Pérez de la Ossa and Susana Ruiz-Bilbao in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Stroke Unit from Hospital Germans Trias i Pujol takes part in the stroke research network RICORS-ICTUS (RD21/0006/0024) from Instituto de Salud Carlos III.
Ethical approval
Ethical approval was not sought for the present study because it was part of clinical routine care in our centre. The study protocol was evaluated and approved by the internal Quality Department of Hospital Universitari Germans Trias i Pujol (UIT-PT-013). This study was completed in accordance with the Helsinki Declaration as revised in 2013.
Informed consent
Informed consent was not sought for the present study because it was part of the clinical routine care. Written informed consent was obtained from all subjects receiving reperfusion therapies (or legally authorized representatives) before administration.
Guarantor
Natàlia Pérez de la Ossa,Susana Ruiz-Bilbao
Trial registration
Not applicable.
Contributorship
NPO and SRB researched literature and conceived the study. AB, MB, GE, ASF, JR, JC, SCA, EM, RB, MM, CC, SR, MW, MCS, SV, MG, MM, NPO and SRB were involved in protocol development, gaining ethical approval, patient recruitment and data collection. AB wrote the first draft of the manuscript and analysed data. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Data sharing statement
The data that support the findings of this study are available on request from the corresponding author, NPO. The data are not publicly available due to their containing information that could compromise the privacy of research participants.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
