Abstract
Introduction:
Restricted retinal diffusion (RDR) has recently been recognized as a frequent finding on standard diffusion-weighted imaging (DWI) in central retinal artery occlusion (CRAO). However, data on early DWI signal evolution are missing.
Patients and methods:
Consecutive CRAO patients with DWI performed within 24 h after onset of visual impairment were included in a bicentric, retrospective cross-sectional study. Two blinded neuroradiologists assessed randomized DWI scans for the presence of retinal ischemia. RDR detection rates, false positive ratings, and interrater agreement were evaluated for predefined time groups.
Results:
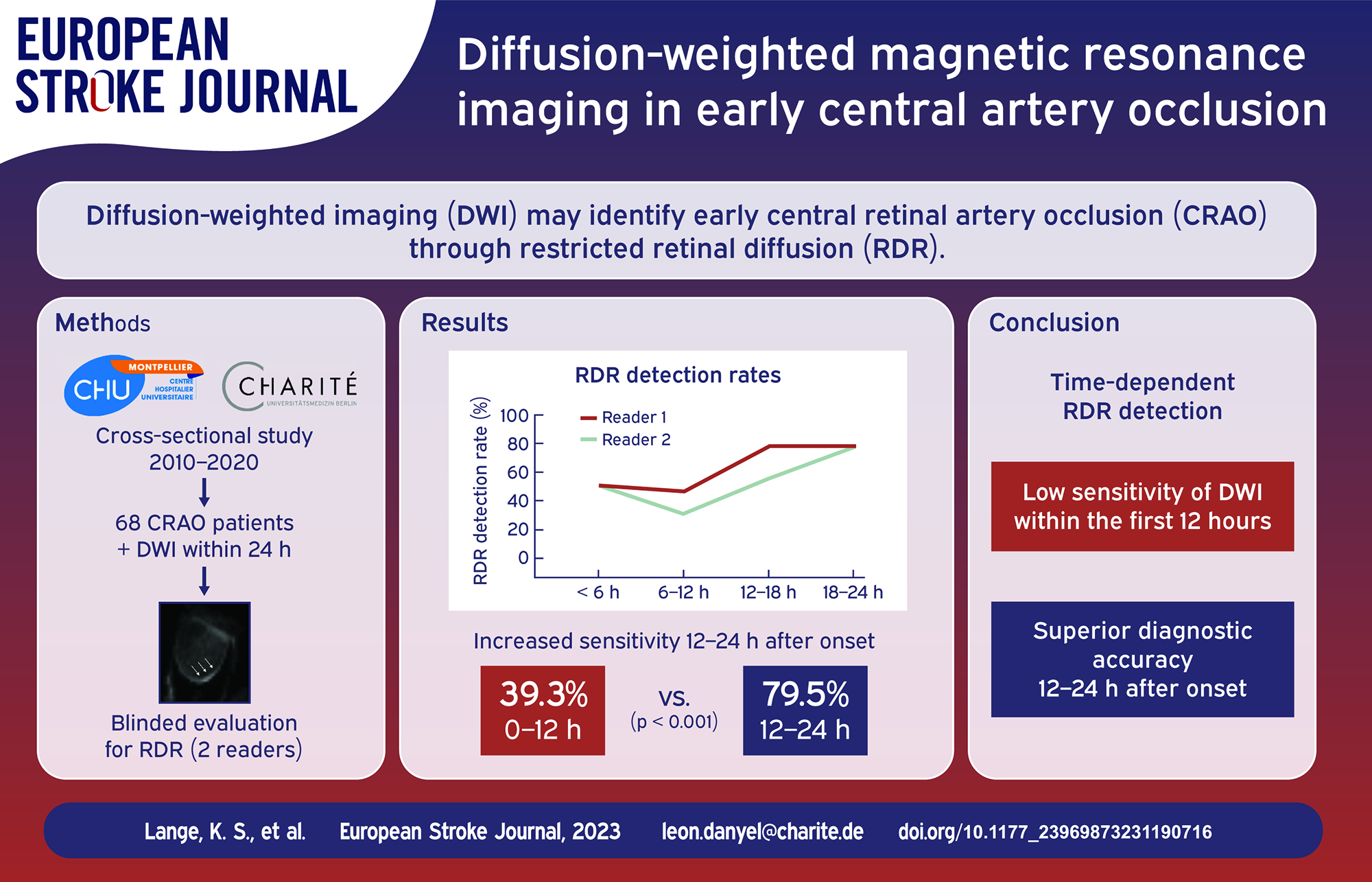
Sixty eight CRAO patients (68.4 ± 16.8 years; 25 female) with 72 DWI scans (76.4% 3 T, 23.6% 1.5 T) were included. Mean time-delay between onset of CRAO and DWI acquisition was 13.4 ± 7.0 h. Overall RDR detection rates ranged from 52.8% to 62.5% with false positive ratings in 4.2%–8.3% of cases. RDR detection rates were higher in DWI performed 12–24 h after onset, when compared with DWI acquired within the first 12 h (79.5%vs 39.3%, p < 0.001). The share of false positive ratings was highest for DWI performed within the first 6 h of symptom onset (up to 14.3%). Interrater reliability was “moderate” for DWI performed within the first 18 h (κ = 0.57–0.58), but improved for DWI acquired between 18 and 24 h (κ = 0.94).
Conclusion:
DWI-based detection of retinal ischemia in early CRAO is likely to be time-dependent with superior diagnostic accuracy for DWI performed 12–24 h after onset of visual impairment.
Keywords
Introduction
Patients with central retinal artery occlusion (CRAO) develop progressive, irreversible ischemic damage of the inner retinal layers, leading to persisting visual impairment with poor overall prognosis for visual recovery. 1 Since CRAO patients may benefit from intravenous tissue-type plasminogen activator treatment administered within 4.5 h of symptom onset, 2 there is growing interest to assess the utility of various diagnostic modalities in acute CRAO. Restricted diffusion of the retina (RDR) has recently been recognized as a frequent finding on standard diffusion-weighted magnetic resonance imaging (DWI MRI) in CRAO.3–6 So far, only one study has investigated the time-dependency of RDR, 5 which reported a peak sensitivity of 79% in DWI acquired within 24 h after onset retinal ischemia. However, data on DWI signal evolution in early CRAO are missing.
Notably, two recent studies employing optical coherence tomography demonstrated a near-linear progression of retinal edema within the first 5 h of CRAO, as well as a gradual deceleration and plateauing of edema progression 10 h after onset of visual impairment.7,8 Since visibility of RDR is likely to be dependent upon the degree of ischemic retinal swelling, 9 sensitivity of DWI-MRI in early CRAO may be lower than previously reported. Consequently, the aim of this study was to investigate the occurrence of RDR for DWI-MRI acquired in early CRAO.
Patients and methods
This bicentric cross-sectional study retrospectively evaluated early DWI scans of CRAO patients for the presence of retinal restricted diffusion at the Charité Universitätsmedizin Berlin, Germany (Center 1), and the University Hospital Montpellier, France (Center 2). Study approval was obtained from the local ethics committees (registration numbers EA1/177/19 and IRB-MTP 2020-11-202000626), who waived the requirement to obtain informed consent for this retrospective analysis. All methods were performed in accordance with relevant guidelines and/or regulations. Our report complies with the “Strengthening the Reporting of Observational Studies in Epidemiology” (STROBE) guidelines.
Patients
Non-arteritic CRAO patients treated at the participating institutions between January 2010 and December 2020 with available diffusion-weighted imaging performed within 24 h after onset of visual impairment were included in the study. Initial identification of patients was achieved through systematic database inquiries at the respective study centers. Subjects were included in the study, if individual patient records met CRAO diagnostic criteria of sudden and painless, persisting monocular visual loss and evidence of retinal ischemia on fundoscopic exam
10
and/or optical coherence tomography.11–13 Patients with amaurosis fugax or branch retinal arteriolar occlusion, as well as patients with symptoms or diagnostic findings suggestive of giant cell arteritis,
14
were not included in this study. Data records were retrieved and assessed for medical history, including common cardiovascular risk factors (arterial hypertension, diabetes mellitus, dyslipidemia, active smoking, and atrial fibrillation), initial visual acuity (VA), and administration of intravenous or intra-arterial tissue plasminogen activator therapy. Grade of visual impairment was adapted from the World Health Organization International Statistical Classification of Diseases and Related Health Problems (10th revision, 2016): Mild visual impairment: VA ⩾6/18 (⩽0.48 logMAR) Moderate visual impairment: VA <6/18 – ⩾6/60 (>0.48 – ⩽1.00 logMAR) Severe visual impairment: VA <6/60 – ⩾3/60 (>1.00 – ⩽1.30 logMAR) Blindness: VA <3/60 (>1.30 logMAR)
Imaging acquisition and analysis
Diffusion-weighted imaging was performed at the two study centers, utilizing their respective clinical MRI scanners and sequence protocols. Center 1 used two 1.5 T (Aera, Siemens, Erlangen, Germany) and one 3 T scanner (Skyra, Siemens, Erlangen, Germany), each equipped with a 20-channel head coil. Another 3 T scanner with a 32-channel head coil was used for EPI-DTI sequence acquisition (Trio, Siemens, Erlangen, Germany). Trace DWI b = 1000 s/mm2 images were acquired from EPI-DWI-or calculated from EPI-DTI-sequences with slice thicknesses ranging from 2.5 to 5 mm. Center 2 used one 1.5 T (Aera Siemens Erlangen, Germany) and two 3 T scanners (Skyra and Prisma, Siemens, Erlangen, Germany), each equipped with a 20-channel head coil. Trace DWI b = 1000 s/mm2 images were acquired from EPI-DWI sequences, from readout-segmented DWI sequences (RESOLVE) with or without Simultaneous Multi-Slice (SMS) imaging with slice thicknesses ranging from 2 to 5 mm.
Acquired images from both centers were pseudonymized and merged into a randomized Digital Imaging and Communications in Medicine (DICOM) data set. Two radiologists (Reader 1, ES: board-certified neuroradiologist with >15 years of experience in MR stroke imaging; Reader 2, AC: board-certified neuroradiologist with >8 years of experience in MR stroke imaging), blinded for patient data, including lateralization of CRAO, separately assessed DWI sequences from the dataset for the presence of RDR (localized signal increase of the inner wall of the affected globe, visible on at least two adjacent slices). In the case of a positive rating, assessment confidence was graded according to a five-level Likert-scale: (1) “not confident at all,” (2) “slightly confident,” (3) “somewhat confident,” (4) “fairly confident,” (5) “completely confident.” In addition, both readers categorized the image quality of each individual imaging set as: (1) “extremely poor,” (2) “poor,” (3) “average,” (4) “good,” or (5) “excellent.”
Statistical analysis
Statistical analyses were performed with IBM SPSS Statistics software (IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY: IBM Corp.) and GraphPad Prism (Version 8.0.0 for Windows, GraphPad Software, San Diego, CA, USA). RDR detection rates and interrater agreement were assessed for each reader and predefined “onset-to-DWI”-time group (according to 6-h-intervals, e.g. <6, ⩾6–12, ⩾12–18, ⩾18–24 h). RDR detection rates were compared among time groups using chi-squared testing. Additionally, standard test quality criteria (sensitivity, specificity, positive and negative predictive value, positive and negative likelihood ratio) for DWI-MRI in early CRAO were calculated. Interrater agreement, according to unweighted Cohen’s κ, was calculated through the observed Pr(a) and expected percentage of agreement Pr(e):
Results
Sixty eight CRAO patients (68.4 ± 16.8 years; 25 [36.8%] female) with a total of 72 DWI scans performed within the first 24 h after onset of visual impairment were included in the study. Lateralization of CRAO was right-sided in 35 (51.5%) and left-sided in 33 (48.5%) patients. In 61 patients, visual acuity on initial examination was >1.3 logMAR (“blindness,” 89.7%). One patient had “severe” visual impairment, while “moderate” and “mild” visual impairment was observed in two patients for each group. For two patients, no information on presenting visual acuity was available. In 13 (19.1%) cases, tissue plasminogen activator therapy was administered within the first 4.5 h.Table 1 summarizes CRAO patient characteristics.
CRAO patient data.
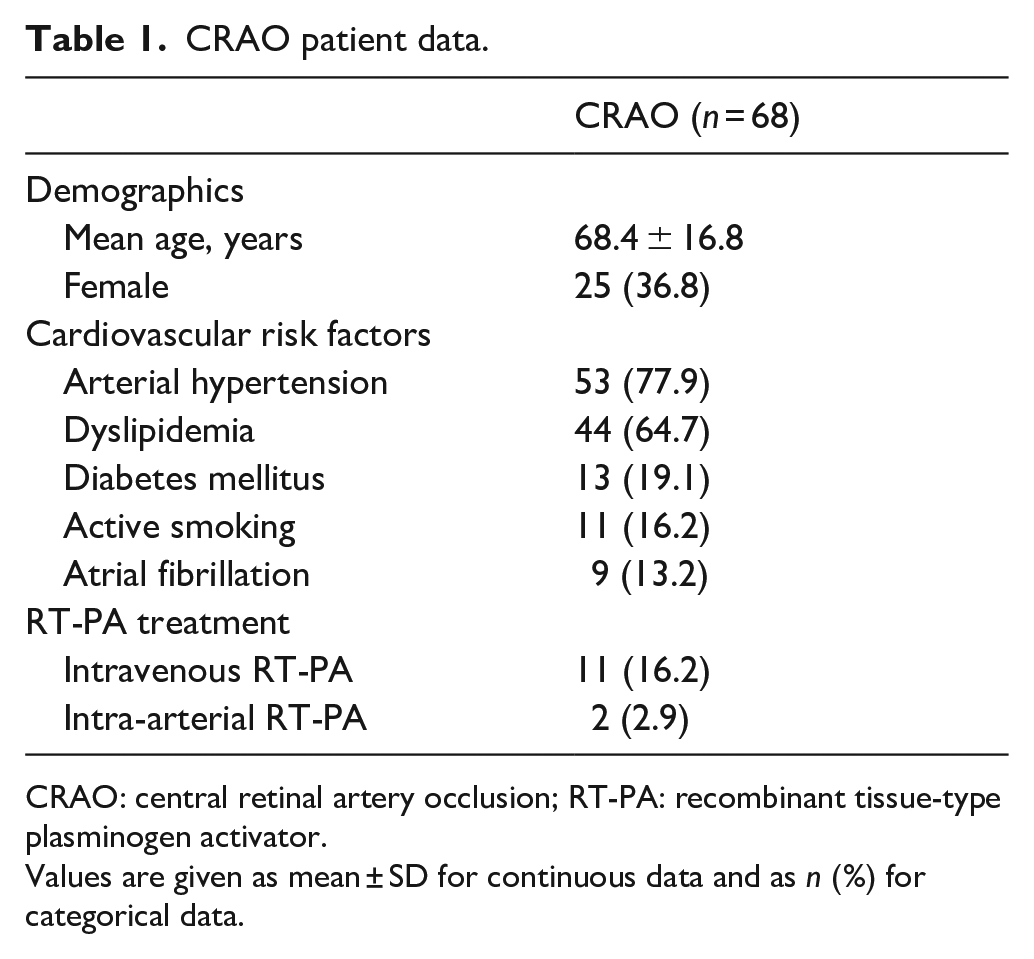
CRAO: central retinal artery occlusion; RT-PA: recombinant tissue-type plasminogen activator.
Values are given as mean ± SD for continuous data and as n (%) for categorical data.
DWI-MRI imaging characteristics are detailed in Table 2. Field strengths employed for DWI acquisition were 3 T in 55 (76.4%) and 1.5 T in 17 (23.6%) of scans with slice thicknesses ranging between 2 and 5 mm. The most common sequence types used were RESOLVE in 28 (38.9%), DTI-EPI in 22 (30.6%), and DWI-EPI TRACE in 12 (16.7%) of scans. Overall image quality was rated 3.3 ± 0.8 (joint readers). Figure 1(a) illustrates the image quality scores of the most commonly applied DWI-sequence protocols.
Technical DWI-MRI parameters.
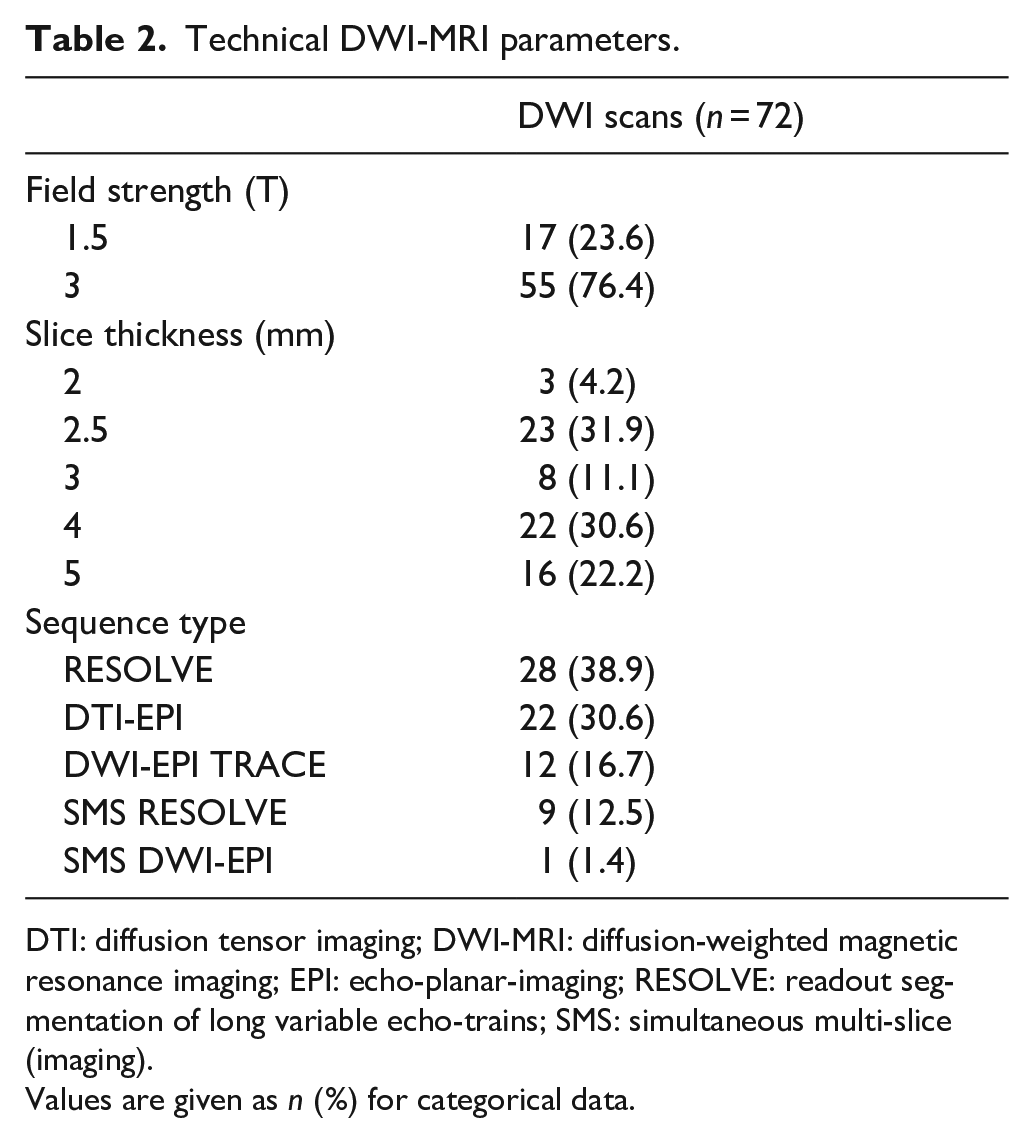
DTI: diffusion tensor imaging; DWI-MRI: diffusion-weighted magnetic resonance imaging; EPI: echo-planar-imaging; RESOLVE: readout segmentation of long variable echo-trains; SMS: simultaneous multi-slice (imaging).
Values are given as n (%) for categorical data.
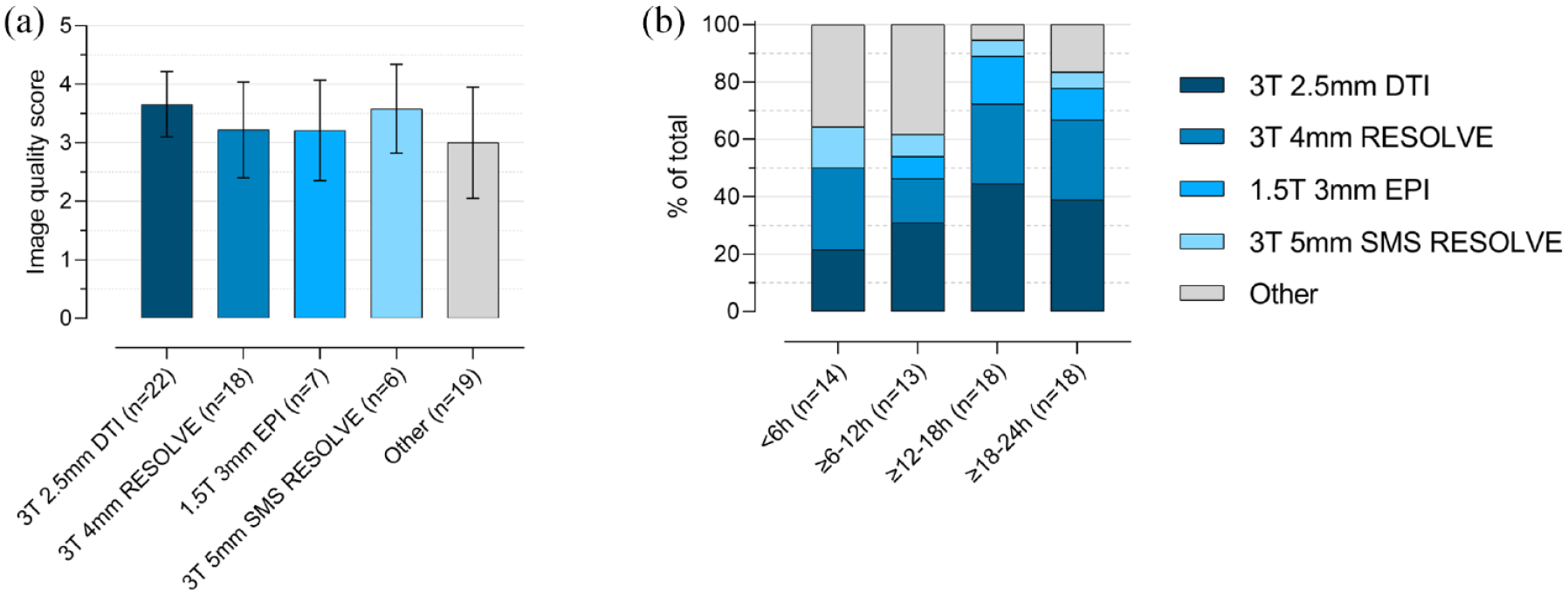
Image quality and frequency distribution of employed DWI-MRI sequences: (a) image quality scores of the most commonly applied DWI-sequence protocols (mean ± SD) and (b) stacked bar graphs indicating the DWI-MRI sequence distribution among predefined “onset-to-DWI”-time groups. DWI-MRI: diffusion-weighted magnetic resonance imaging.
Mean time-delay between onset of visual impairment and DWI acquisition was 13.4 ± 7.0 h. Restricted diffusion of the retina was present in 45 (62.5%, Reader 1) and 38 DWI scans (52.8%, Reader 2) with a corresponding mean RDR assessment confidence score of 3.9 ± 1.2 and 3.5 ± 1,0, respectively. In six (8.3%, Reader 1) and three cases (4.2%, Reader 2) RDR were falsely attributed to the healthy eye (false positives, RDR rated as absent for the affected eye). Overall interrater agreement was “substantial” with κ = 0.71 (95% CI 0.59–0.84). Distribution of DWI-MRI among predefined “onset-to-DWI”-time groups was as follows: <6 h in 14 (19.4%), ⩾6–12 h in 13 (18.1%), ⩾12–18 h in 18 (25.0%), and ⩾18–24 h in 18 (25.0%). For nine scans (12.5%), an unambiguous assignment to one of the predefined “onset-to-DWI”-time groups was not possible. The distribution of different sequence types among predefined “onset-to-DWI”-time groups is illustrated in Figure 1(b).
RDR detection rates were significantly higher for DWI performed 12–24 h after onset of visual impairment, when compared with DWI acquired within the first 12 h (Reader 1: 31/39 or 79.5% vs 11/28 or 39.3%, p < 0.001; Reader 2: 27/39 or 69.2% vs 9/28 or 32.1%, p = 0.003). In addition, joint RDR assessment confidence was significantly higher for DWI performed 12–24 h after onset of visual impairment, when compared with DWI acquired within the first 12 h (4.0 ± 1.1 vs 3.3 ± 0.9, p = 0.006). Notably, RDR detection rates were highest for DWI-performed between 18 and 24 h after onset (14/18 or 77.8%, both readers). Interrater agreement was “moderate” for DWI performed within the first 18 h after CRAO with κ values ranging between 0.57 and 0.58. However, agreement substantially improved for DWI performed between 18 and 24 h after onset with unweighted kappa κ = 0.94 (95% CI 0.83–1.00), indicating “almost perfect” interrater reliability. Table 3 summarizes standard test quality criteria (sensitivity, specificity, positive and negative predictive value, positive and negative likelihood ratio) for DWI-MRI in early CRAO. Figure 2 details DWI assessment characteristics among CRAO patients in different “onset-to-DWI”-time groups, while Figure 3 shows representative DWI-MRI examples in CRAO with differing “onset-to-DWI”-time intervals.
Standard test quality criteria for DWI-MRI in early CRAO.
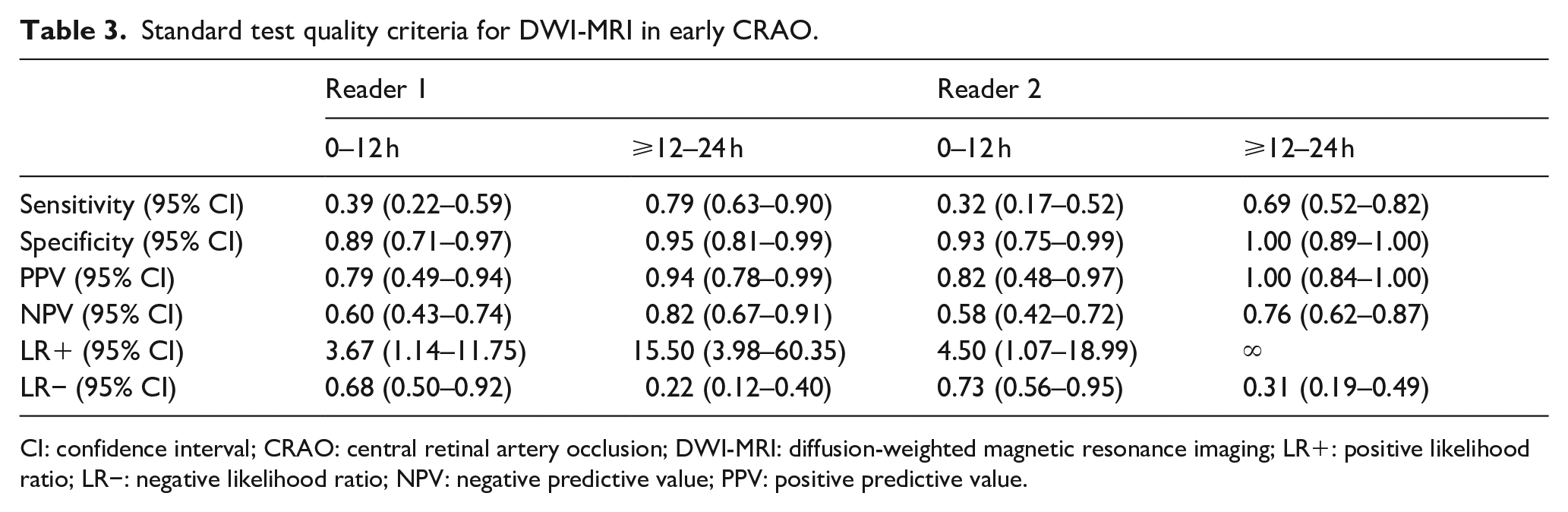
CI: confidence interval; CRAO: central retinal artery occlusion; DWI-MRI: diffusion-weighted magnetic resonance imaging; LR+: positive likelihood ratio; LR−: negative likelihood ratio; NPV: negative predictive value; PPV: positive predictive value.
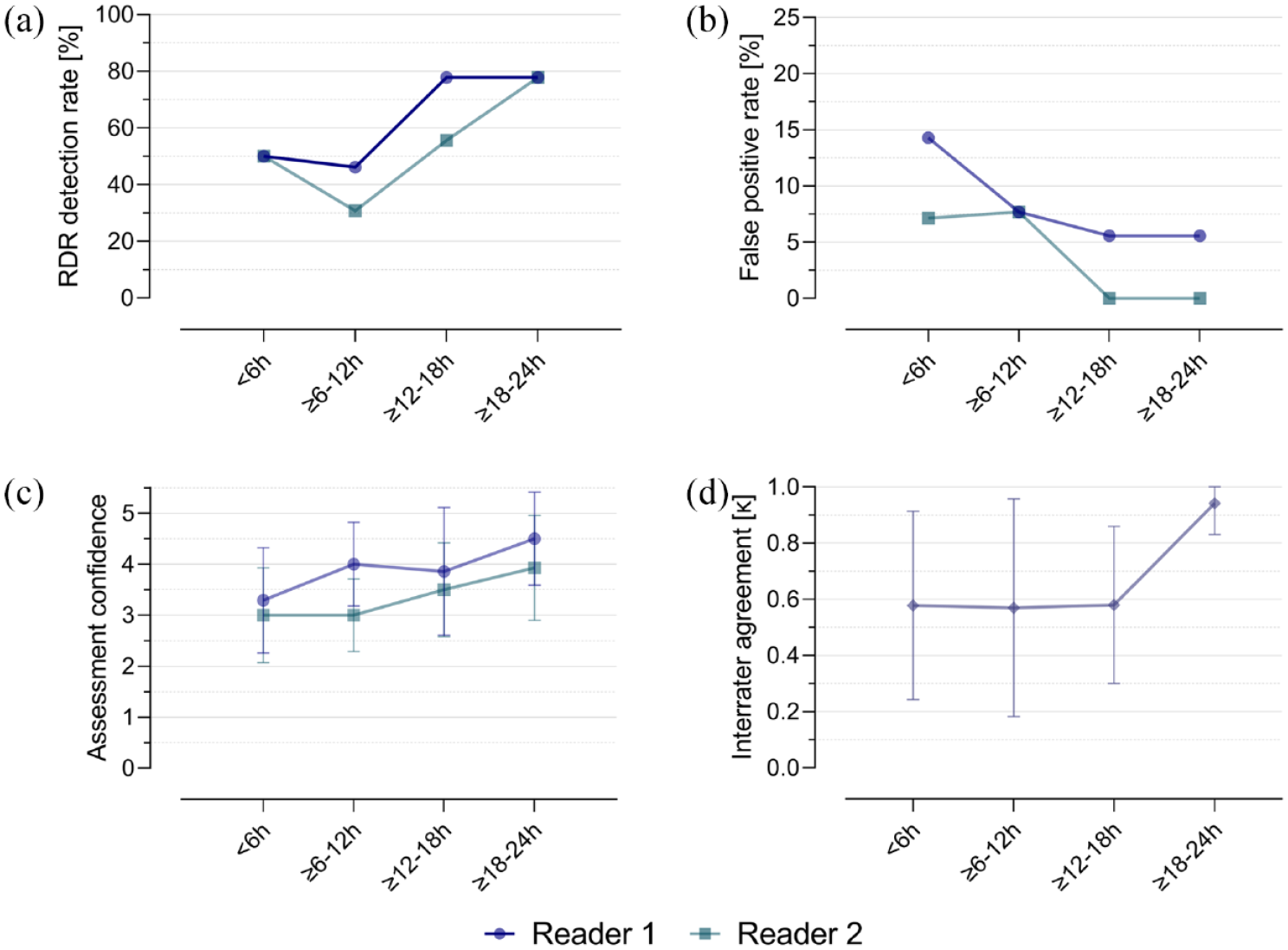
DWI-MRI assessment characteristics among “onset-to-DWI”-time groups. RDR was more frequently identified in DWI-MRI performed within 12–24 h after onset of CRAO (a) with a reduction in the overall share of false positive ratings (RDR falsely attributed to the healthy eye; (b)). Similarly, assessment confidence score ratings were higher in the later time groups (c). Interrater agreement (d) was “moderate” for DWI performed within the first 18 h after CRAO (Cohen’s kappa: 0.57–0.58), but substantially improved for DWI performed between 18 and 24 h after onset (Cohen’s kappa: 0.94). CRAO: central retinal artery occlusion; DWI-MRI: diffusion-weighted magnetic resonance imaging; RDR: retinal diffusion restriction.
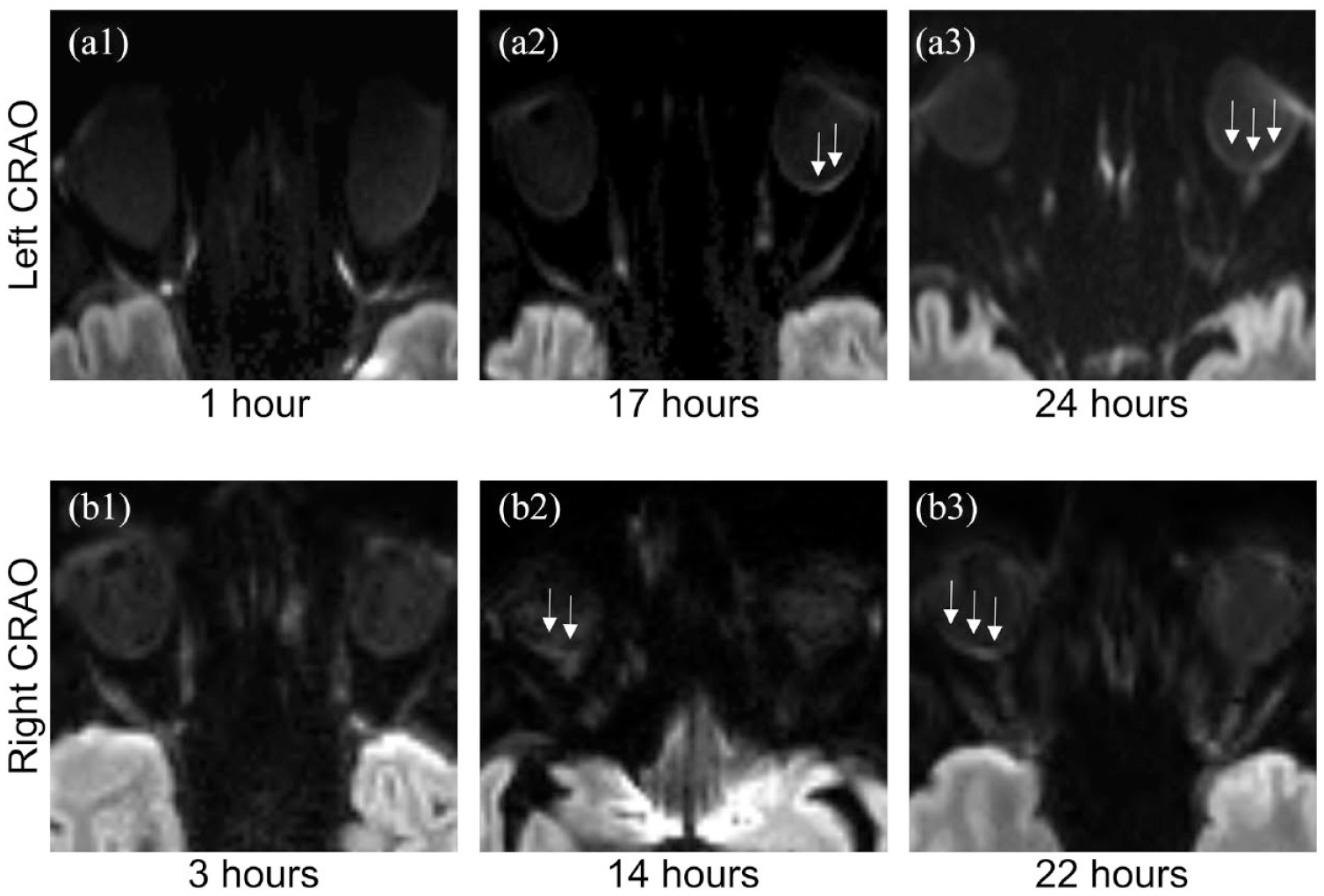
Examples of RDR in CRAO patients with variable onset-to-DWI time intervals. 3 T, 2.5 mm DWI-TRACE calculated from DTI-EPI (a1–a3) and 3 T, 4 mm RESOLVE-DWI (b1–b3) images. Restricted diffusion of the retina is not clearly discernable on early imaging, but becomes visible with longer delay between onset and acquisition of diffusion-weighted images. CRAO: central retinal artery occlusion; DWI: diffusion-weighted imaging.
Discussion
This first, systematic investigation in early CRAO indicates that detection of retinal ischemia by means of standard DWI-MRI may be unreliable if imaging is performed within the first 12 h after onset of retinal ischemia. In this sense, detection rates for RDR within the first 12 h were low (32.1%–39.3%) and accompanied by a substantial number of false positive ratings (RDR falsely attributed to the healthy eye), accounting for a share of up to 14.3% of cases in DWI performed within the first 6 h after onset. Similarly, interrater agreement for RDR in CRAO was only “moderate.” Conversely, in DWI performed between 12 and 24 h after onset, sensitivity for RDR improved up to 79.5%, while the overall number of false positive ratings decreased. Notably, the identification of retinal ischemia by MRI was the most reliable in DWI performed between 18 and 24 h after onset. Here, the highest sensitivity for RDR in CRAO was found with the lowest overall number of false positive ratings and an “almost perfect” interrater agreement of κ = 0.94. Based on their first systematic investigations on retinal DWI in CRAO, Danyel et al.4,5 suggested that RDR might constitute a common imaging finding in MRI performed within 24 h up to 1 week after the onset of visual impairment. However, the utility of DWI for the detection of early retinal ischemia remained elusive.
Detection of retinal ischemia by DWI-MRI is likely contingent upon the extent of retinal cell edema, which progressively develops after the cessation of blood flow via the central retinal artery. Importantly, the severity and speed of structural damage to the retinal layers in CRAO may show marked inter-individual variability.8,13,15 Siebert et al. 9 suggested that mild edema of the inner retinal layers in CRAO may not suffice to produce a perceivable signal increase on standard DWI-MRI. Recent studies have investigated the temporal dynamics of ischemic retinal edema progression in CRAO.7,8 Using optical coherence tomography, Wenzel et al. 8 described a hyperbolic curve with a near-linear increase of retinal thickness within the first 5 h, as well as a subsequent plateauing of edema progression 10–20 h after ischemia onset. It is reasonable to infer that in DWI-MRI performed within the first 12 h after onset of CRAO, retinal edema is not fully developed in the majority of patients. Consequently, while discreet retinal ischemia in early CRAO may not be discernible, visible RDR on DWI-MRI may signify the presence of extensive and irreversible ischemic retinal edema.
Occlusion of central retinal artery constitutes a medical emergency that, if unremitting, leads to persisting blindness of the affected eye. 1 To date, there are no effective therapeutic agents for CRAO that have been successfully tested in randomized controlled trials. 16 However, intravenous tissue-type plasminogen activator treatment (rtPA) has emerged as a potential therapy for CRAO patients, but the potential treatment effect is limited to a narrow therapeutic window of up to 4.5 h after onset.2,17,18 Currently, three European randomized-controlled trials investigate the efficacy of intravenous tissue-type plasminogen activator treatment in CRAO (NCT04965038, NCT03197194, NCT04526951). In the context of a time-dependent treatment efficacy in CRAO, reliable early diagnosis of the disease is becoming increasingly important. However, up to 35% of CRAO patients report visual impairment at wake-up, and consequently have to be excluded from treatment with tissue-type plasminogen activator due to an unknown time of symptom onset. In ischemic stroke, MRI-guided estimation of the time of onset has been a crucial step for the identification of patients who benefit from thrombolysis in a “wake-up” setting.19,20 In theory, a standardized orbital DWI-MRI protocol may, through the absence RDR, exclude advanced retinal infarction and determine rtPA candidacy in wake-up retinal ischemia. As such, improving our understanding of MRI signal evolution in central retinal artery occlusion may help to inform treatment decisions in the future.
This study is limited by its retrospective and cross-sectional design. Our patient selection process was based on a medical database inquiry and may not have captured CRAO patients in cases of miscoding or documentation omissions. While we did not employ a control-cohort, all readers were blinded to patient clinical data, including the side of CRAO. As such, the healthy eye served as an intrinsic control during radiologic evaluation. With an incidence around 1.9 per 100,000 person-years in the United States, CRAO can be considered a rare disease. Consequently, although only a moderate number of CRAO patients was examined, this study constitutes the largest cohort of early DWI-MRI in CRAO up to date. There was notable heterogeneity with regards to the DWI-MRI sequences employed, which is likely due to the bicentric study design and owed to the fact that patients were included over a time period of 10 years in which scanners and imaging protocols were subject to change. Especially in MRI scans performed within the first 12 h, considerable DWI sequence heterogeneity was observed, which may have impacted the analysis. It is important to note, that this study employed DWI-MRI sequences originally developed for the detection of ischemic stroke. Diffusion-weighted imaging of the retina, however, poses a number of specific challenges, such as motion-artifacts caused by eye movements, as well as geometric distortion and signal loss due to susceptibility artifacts caused by inhomogeneity of the orbital magnetic field secondary to adjacent air – bone - soft tissue – interfaces 21 and low signal gain from a delicate structure of submillimetric thickness such as the retina. Optimization strategies to overcome these challenges have already been successfully implemented in other anatomic regions22–24 and may include innovative sequence designs, such as non-EPI DWI, small field-of-view DWI, and fast spin-echo radial acquisition DWI sequence designs. Furthermore, employing higher field strengths and surface coils may add to the diagnostic yield of DWI-MRI, as may the specific instruction of the patient to keep eyes focused on one point during the scan. Longitudinal studies with uniform imaging protocols are required to confirm our findings for retinal signal development on DWI-MRI.
Conclusion
In summary, our first systematic investigation in early CRAO indicates that DWI-based identification of retinal ischemia is time-dependent with superior diagnostic accuracy for DWI performed 12–24 h after onset of visual impairment, but limited by low sensitivity for the detection of CRAO within the first 12 h.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231190716 – Supplemental material for Diffusion-weighted magnetic resonance imaging in early central retinal artery occlusion
Supplemental material, sj-docx-1-eso-10.1177_23969873231190716 for Diffusion-weighted magnetic resonance imaging in early central retinal artery occlusion by Kristin Sophie Lange, Isabelle Mourand, Arthur Coget, Nicolas Menjot de Champfleur, Xavier Ayrignac, Caroline Arquizan, Michael Scheel, Georg Bohner, Kersten Villringer, Charlie Zagroun, Eberhard Siebert and Leon Alexander Danyel in European Stroke Journal
Footnotes
Acknowledgements
LAD is participant in the BIH-Charité Clinician Scientist Program funded by the Charité-Universitätsmedizin Berlin and the Berlin Institute of Health.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was waived because of the retrospective nature of the study and analysis of routinely acquired data.
Ethical approval
Institutional Review Board approval was obtained. Charité – Universitätsmedizin Berlin, Germany: registration number EA1/177/19. University Hospital Montpellier, France: registration number IRB-MTP 2020-11-202000626.
Guarantor
The scientific guarantor of this publication is LAD.
Contributorship
ES, KSL, and LAD researched literature and conceived the study. KSL, IM, ES, and LAD were involved in gaining ethical approval. KSL, IM, AC, NMC, XA, CA, KV, CZ, ES, and LAD were involved in data acquisition, KSL, AC, LAD, and ES in data analysis. LAD, KSL, and ES wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
