Abstract
Rationale:
Meta-analyses of case series of non-arteritic central retinal artery occlusion (CRAO) indicate beneficial effects of intravenous thrombolysis when initiated early after symptom onset. Randomized data are lacking to address this question.
Aims:
The REperfusion therapy with intravenous alteplase for recovery of VISION in acute central retinal artery occlusion (REVISION) investigates intravenous alteplase within 4.5 h of monocular vision loss due to acute CRAO.
Methods:
This study is the randomized (1:1), double-blind, placebo-controlled, multicenter adaptive phase III trial.
Study outcomes:
Primary outcome is functional recovery to normal or mildly impaired vision in the affected eye defined as best-corrected visual acuity of the Logarithm of the Minimum Angle of Resolution of 0.5 or less at 30 days (intention-to-treat analysis). Secondary efficacy outcomes include modified Rankin Score at 90 days and quality of life. Safety outcomes include symptomatic intracranial hemorrhage, major bleeding (International Society on Thrombosis and Haemostasis definition) and mortality. Exploratory analyses of optical coherence tomography/angiography, ultrasound and magnetic resonance imaging (MRI) biomarkers will be conducted.
Sample size:
Using an adaptive design with interim analysis at 120 patients, up to 422 participants (211 per arm) would be needed for 80% power (one-sided alpha = 0.025) to detect a difference of 15%, assuming functional recovery rates of 10% in the placebo arm and 25% in the alteplase arm.
Discussion:
By enrolling patients within 4.5 h of CRAO onset, REVISION uses insights from meta-analyses of CRAO case series and randomized thrombolysis trials in acute ischemic stroke. Increased rates of early reperfusion and good neurological outcomes in stroke may translate to CRAO with its similar pathophysiology.
Trial registration:
ClinicalTrials.gov: NCT04965038; EU Trial Number: 2023-507388-21-00.
Introduction and rationale
Thromboembolic central retinal artery occlusion (CRAO) is an acute neurovascular-ophthalmological emergency that leads to severe and permanent vision loss in 90% of affected eyes. 1 Non-arteritic etiology accounts for 95% of CRAO cases with no evidence-based therapy being available despite widely practiced treatments including hemodilution, ocular massage, or paracentesis.2,3 Animal models have proven limited tolerance of the retina to ischemia with irreversible damage occurring within 2–4 h after cessation of arterial blood supply.4,5 Therefore, as with cerebral ischemic stroke, 6 establishing early reperfusion seems the logical therapeutic approach.
Two randomized controlled trials tested intra-arterial 7 and intravenous thrombolysis (IVT) 8 in non-arteritic CRAO but could not demonstrate beneficial treatment effects. 3 Enrolment time windows in these trials, however, were 20 and 24 h and only one patient received thrombolytic therapy within less than 4.5 h of vision loss.7,8 Moreover, symptomatic intracranial hemorrhage rates in the thrombolysis arms were 5% and 12.5%.7,8
In contrast, recent individual patient-level meta-analyses comprising data from retrospective and prospective observational cohort studies, case series and the two randomized controlled trials found a low (1%) risk of symptomatic intracranial hemorrhage and indicate enhanced vision recovery associated with early thrombolytic therapy.9,10 Comparison of 238 CRAO patients who underwent IVT to 396 natural history patients revealed higher rates of vision recovery only in those in whom thrombolysis had been initiated within 4.5 h after symptom onset. 9 Data in these meta-analyses, however, stems from small case series, with the largest comprising 30 thrombolysed patients, and only 67 patients had received intravenous alteplase, which is best established for treatment of acute stroke, within 4.5 h. 9
Yet, neither efficacy nor safety of early IVT in general nor specifically of alteplase can be considered as established for treatment of CRAO. Ophthalmological and stroke societies have expressed the need for high-quality clinical trials.2,11
The REVISION randomized trial consequently investigates IVT with alteplase within 4.5 h of CRAO onset for achieving recovery of vision.
Methods
The study protocol is provided as Supplemental Material.
Design
This is a randomized (1:1), double-blind, placebo-controlled, multicenter adaptive phase III trial.
Patient population
Adults with acute painless monocular vision loss to the level of functional blindness due to non-arteritic CRAO and previously near normal vision that allowed reading with the affected eye.
Inclusion criteria were as follows:
age ⩾ 18 years;
acute non-arteritic CRAO confirmed by an experienced ophthalmologist through assessment of visual acuity, intraocular pressure, relative afferent pupil defect, slit-lamp biomicroscopy, and fundoscopy;
optical coherence tomography (OCT) of the macula of both eyes if feasible within 10 min;
functional blindness of the affected eye defined as best-corrected visual acuity of the Logarithm of the Minimum Angle of Resolution (LogMAR) ⩾ 1.3;
reading was possible with the affected eye before CRAO (LogMAR ⩽ 0.5);
neurological assessment including the National Institutes of Health Stroke Scale (NIHSS) score performed by an experienced stroke physician;
cranial CT or magnetic resonance imaging (MRI);
study treatment can be initiated within 4.5 h of symptom onset;
written informed consent from patient.
Key exclusion criteria were as follows:
suspected giant cell arteritis;
rapidly improving vision in the affected eye;
acute cerebral stroke with indication for on-label thrombolysis;
contraindication for thrombolysis.
Randomization
Patients are randomized (1:1) using block randomization without stratification. The randomization list is provided by Technical University Munich Trial Center, Germany. Randomization is completed by an investigator opening the study treatment package with the lowest randomization number that is available at the respective trial site.
Study intervention
IVT: alteplase (Actilyse; Boehringer Ingelheim, Germany) is administered intravenously at 0.9 mg/kg of bodyweight (maximum 90 mg). 10% of the dose are given as bolus and the remaining 90% as continuous infusion over 1 h (see Figure 1).
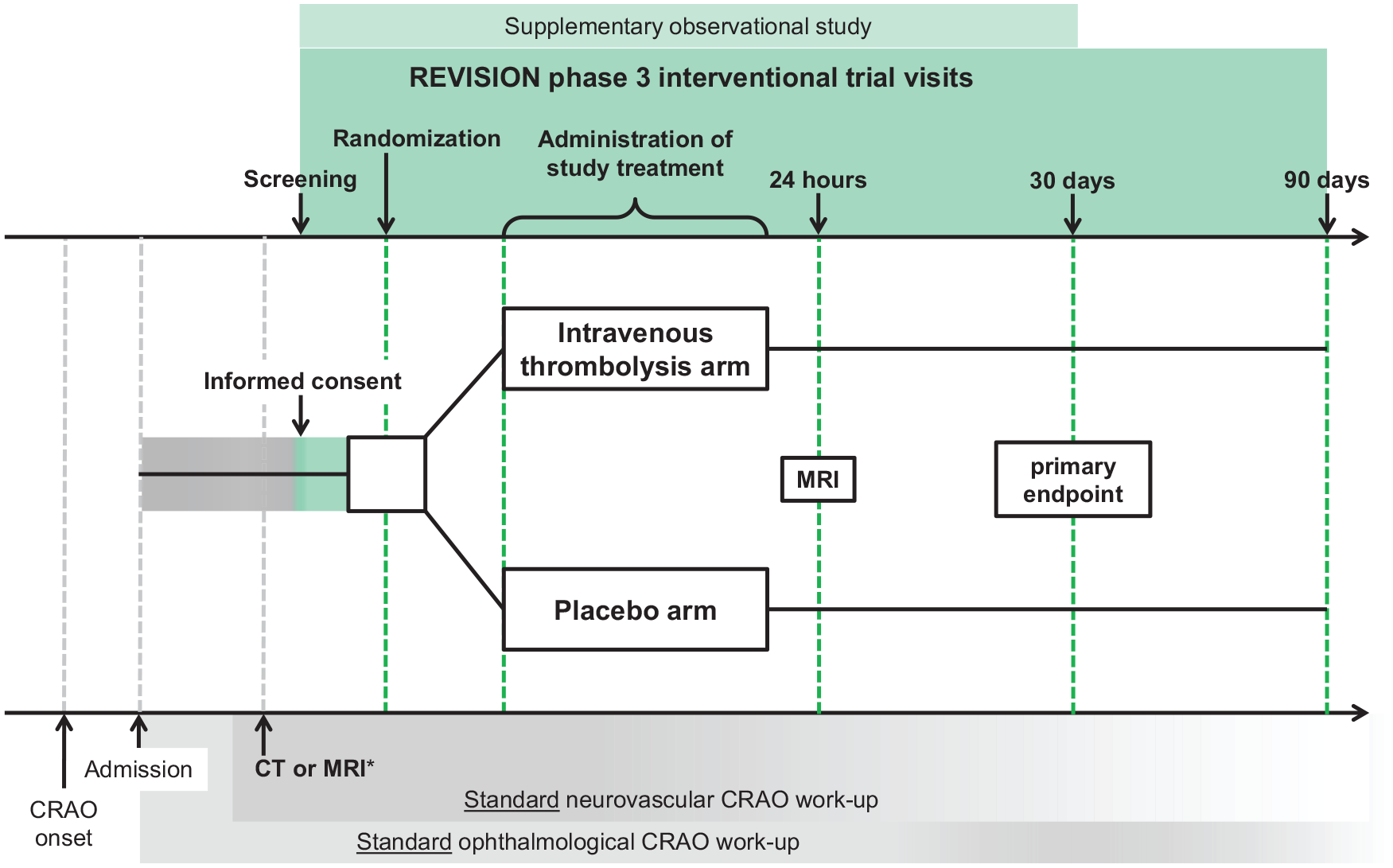
Study flow. CRAO: central retinal artery occlusion.
Control treatment: placebo (Boehringer Ingelheim) includes all ingredients of Actilyse except alteplase, is indistinguishable from the active drug, and administered in the same way.
Primary outcome
Primary efficacy of IVT is determined by the functional recovery to normal or mildly impaired vision in the affected eye (LogMAR ⩽ 0.5) in the thrombolysis and control arms at 30 days.
Secondary outcomes
Main secondary efficacy outcomes are the scores on the modified Rankin Scale and National Eye Institute Visual Function Questionnaire-25 at 30 and 90 days, and vision recovery at 24 h and 90 days. Further secondary efficacy outcomes include number and volume of ischemic lesions on MRI at 24 h, central retinal artery recanalization on OCT/angiography at 24 h, 30 and 90 days, retinal perfusion on fluorescein angiography, and the kinetic visual field at 30 and 90 days.
Safety outcomes include symptomatic and any intracranial hemorrhage, intraocular hemorrhage and major bleeding (International Society on Thrombosis and Haemostasis definition) at 24 h, treatment-requiring retinal neovascularization, and mortality until 90 days. Exploratory analyses of OCT/angiography, transorbital ultrasound, and MRI biomarkers for outcome and treatment response prediction will be conducted. OCT/angiography and MRI are core lab-adjudicated.
Blinding
The trial is double-blinded. Boehringer Ingelheim provides alteplase and placebo to the pharmacy at University Medical Center Hamburg-Eppendorf, Germany, which labels, packs, and distributes study medication to trial sites along with respective unblinding envelopes. Labeling follows the randomization list which is kept in safe and confidential custody at the provider, the pharmacy, an independent unblinded statistician, and Data Safety Monitoring Board (DSMB). Unblinding envelopes may be opened by an investigator if medically imperative.
Data Safety Monitoring Board
Two independent clinical CRAO/thrombolysis trial experts and one statistician ensure ethical conduct of the trial and protect the rights and welfare of participants. DSMB receives periodic reports (see Supplemental Material) and meets in semiannual intervals.
Sample size estimates
Based on recent meta-analyses9,10 and our case series, 12 we conservatively estimated the rate of functional recovery in affected eyes to LogMAR ⩽ 0.5 to be 10% in the placebo arm and 25% in the alteplase arm. Expecting a 10% drop-out rate, a maximum of 211 patients per arm would be needed for a one-sided test at alpha 0.025 to detect a treatment effect with overall 80% power. Adaptive design 13 allows the trial to be stopped for success (p < 0.0102) or futility (p ⩾ 0.5) after interim analysis at 96 patients or to be continued with up to additional 244 patients considering a conditional power of ~75% for the second stage. Assuming 10% missing values, total sample size would be 422 according to Lachin 14 with interim analysis at 120 patients and maximally 302 additional patients.
Statistical analyses
Primary analysis will test the null hypothesis that the rate of functional recovery to LogMAR ⩽ 0.5 at 30 days is at least as high in the placebo arm as it is in the alteplase arm (intention-to-treat analysis), at a one-sided alpha 0.025 level using a logistic regression model with functional recovery as the response variable and best corrected visual acuity at baseline (i.e. screening) and treatment as explanatory variables. Visual acuity is assessed at baseline, 24 h, 30 and 90 days. Missing values will be imputed conditionally on previous values, age and sex. Primary analysis will be performed for the full analysis set which comprises all randomized patients unless <18 years, no written informed consent provided and/or intracranial hemorrhage visible on screening CT/MRI (except microbleeds on MRI).
Secondary analyses will include shift analyses of the modified Rankin Scale and vision outcome categories according to World Health Organization International Classification of Diseases 11 (normal vision (LogMAR ⩽ 0); mild (LogMAR > 0 to ⩽ 0.5); moderate (LogMAR > 0.5 to ⩽ 1.0); severe vision impairment (LogMAR > 1.0 to ⩽ 1.3); LogMAR > 1.3 to counting fingers; hand motion or light perception; no light perception). Continuous variables will be analyzed with linear regression methods. Subgroup analyses will be conducted based on presence/absence of the retrobulbar spot sign on ultrasound 15 and relative retinal thickness on OCT of the macula. 16 In these analyses, respective variables will be used as additional explanatory variables.
Study organization
Recruitment is planned at trial sites in Germany, Austria, and Switzerland. REVISION is carried out according to Good Clinical Practice E6(R2) guidelines and the current Declaration of Helsinki. Trial sponsor is Medical Faculty of Tübingen University, Germany. Center for Clinical Trials at Heidelberg University, Germany, coordinates and monitors the trial.
Study conduct
First patient was enrolled on 10 October 2022. Estimated study completion date is 31 December 2025.
Protocol amendments
Until first active version 3 (20 May 2022), regulatory requests of competent authorities were implemented.
Discussion
Fundamental considerations based on recent meta-analyses9,10 and our case series 12 which indicate enhanced vision recovery when IVT is initiated within 4.5 h of CRAO onset were implemented in the design of REVISION.
In CRAO, the size of the occluded vessel does not allow for endovascular mechanical thrombectomy. Invasiveness, only marginally higher recanalization rates and procedure-associated treatment delays made us decide against intra-arterial thrombolysis. 17
We adopted the therapeutic window, thrombolytic drug, and dose established for IVT in stroke. Although (intracranial) bleeding risk is increased with standard dose alteplase at 0.9 mg/kg of bodyweight, 6 low-dose alteplase (0.6 mg/kg) could not demonstrate non-inferiority. 18 Tenecteplase may be an excellent alternative but has not been evaluated in CRAO9,10 and has only recently been recommended by guidelines for stroke.19,20
Limiting enrollment to patients with massive vision loss in the affected eye to functional blindness (LogMAR > 1.3) at screening and at most midly impaired vision that permitted reading (LogMAR ⩽ 0.5) before CRAO justifies using a potentially harmful therapy with full recovery and severe disability as alternative outcomes. It also avoids bias through unbalanced visual acuity at baseline and most reliably indicates CRAO as opposed to branch artery occlusion, which is more likely to have better prognosis independent of treatment. 21
Recovery of vision to LogMAR ⩽ 0.5 in affected eyes at 30 days will be compared between treatment arms for primary efficacy assessment. Vision usually recovers within the first 7 days but not beyond 30 days after CRAO. 21 LogMAR ⩽ 0.5 permits reading with the affected eye and is thus best suited to distinguish favorable from unfavorable outcome. 12 Mean LogMAR changes, on the contrary, may not reflect relevant improvement although being statistically significant. 12 Moreover, recovery to LogMAR ⩽ 0.5 represents true recovery of retinal function rather than apparent recovery through eccentric fixation, a common phenomenon after CRAO. 21 LogMAR ⩽ 0.5 cannot be achieved through eccentric fixation due to low spatial resolution in the peripheral visual field. 22
Patients with suspected giant cell arteritis are excluded from trial participation. Arteritic CRAO features different pathophysiology and outcomes compared to thromboembolic CRAO, and evidence-based (corticosteroid) treatment is available. 21
Brain imaging before randomization and study treatment shall increase the participants’ safety by ruling out intracranial pathology including subacute infarcts that may increase the risk of intracranial hemorrhage. Follow-up MRI instead of CT the day after study treatment allows detection of even small (silent) infarcts which may be present in up to 37% of CRAO cases. 23 Incidence, numbers, and volumes of these may influence neurofunctional outcome and be reduced by thrombolysis. To facilitate outcome interpretation in participants with CRAO and additional minor non-disabling stroke symptoms, NIHSS is assessed before study treatment.
OCT/angiography, transorbital ultrasound, and retinal/optic nerve MRI will be conducted to non-invasively assess biomarkers for outcome and treatment response prediction. These include retinal thickness increase, 16 retrobulbar spot sign status, 15 and the extend of ischemic damage in deeper structures, 24 respectively. Safer selection of thrombolysis candidates may be expected in future.
We enroll CRAO patients with contraindications for alteplase and/or within up to 12 h after symptom onset into a supplementary observational study to gain insights on retinal pathology in the extended time window. This will also serve to maintain study momentum and thereby contribute to successful enrollment into the interventional trial which may be pivotal considering the low incidence of hospital admissions of CRAO patients within 4.5 h after symptom onset. 12
Limitation of the study is the large sample size in a setting with challenges in recruitment. On the contrary, conservative estimation accounts for non-responders and may avoid a negative trial due to insufficient power.
Summary and conclusion
Despite being a rare disease, non-arteritic CRAO causes considerable disability which justifies aggressive treatment. 11 Meta-analyses of CRAO case series indicate enhanced recovery of vision and low rates of critical bleeding complications associated with IVT within 4.5 h after symptom onset,9,10 which is the established treatment of acute ischemic stroke. 20
Together with the THrombolysis with alteplase in patients with acutE central retInal Artery occlusion study (THEIA, N = 70, ClinicalTrials.gov: NCT03197194) and the Tenecteplase in Central Retinal Artery Occlusion Study (TenCRAOS, N = 78, ClinicalTrials.gov: NCT04526951), the REVISION trial will generate evidence for the use of early IVT in CRAO. Whether results are positive, negative, or neutral, they will have an immediate, practice-changing impact on the field.
Supplemental Material
sj-docx-1-wso-10.1177_17474930241248516 – Supplemental material for Early REperfusion therapy with intravenous alteplase for recovery of VISION in acute central retinal artery occlusion (REVISION): Study protocol of a phase III trial
Supplemental material, sj-docx-1-wso-10.1177_17474930241248516 for Early REperfusion therapy with intravenous alteplase for recovery of VISION in acute central retinal artery occlusion (REVISION): Study protocol of a phase III trial by Sven Poli, Carsten Grohmann, Daniel A Wenzel, Khouloud Poli, Johannes Tünnerhoff, Max Nedelmann, Jens Fiehler, Ina Burghaus, Monika Lehmann, Monika Glauch, Hauke M Schadwinkel, Pia Kalmbach, Julia Zeller, Tobias Peters, Christoph Eschenfelder, Hansjürgen Agostini, Bruce CV Campbell, M Dominik Fischer, Marek Sykora, Brian Mac Grory, Nicolas Feltgen, Markus Kowarik, David Seiffge, Daniel Strbian, Michael Albrecht, Mohammad S Alzureiqi, Gerd Auffarth, Hansjörg Bäzner, Stefanie Behnke, Anne Berberich, Felix Bode, Ferdinand O Bohmann, Bastian Cheng, Michael Czihal, Leon A. Danyel, Spyridon Dimopoulos, João D Pinhal Ferreira de Pinho, Fabian N Fries, Maria-Andreea Gamulescu, Florian Gekeler, Alexandra Gomez-Exposito, Christoph Gumbinger, Rainer Guthoff, Lars-Olof Hattenbach, Lars Kellert, Ramin Khoramnia, Thomas Kohnen, David Kürten, Bernhard Lackner, Mona Laible, John-Ih Lee, Christoph Leithner, Raffael Liegl, Piergiorgio Lochner, Marc Mackert, Joshua Mbroh, Susanne Müller, Simon Nagel, Michelle Prasuhn, Jan Purrucker, Arno Reich, Sibu Mundiyanapurath, Georg Royl, Daniel J Salchow, Jan H Schäfer, Felix Schlachetzki, Ingo Schmack, Götz Thomalla, Maria P Tieck Fernandez, Philip Wakili, Peter Walter, Armin Wolf, Marc Wolf, Karl U Bartz-Schmidt, Maximilian Schultheiss and Martin S Spitzer in International Journal of Stroke
Footnotes
Acknowledgements
The authors thank the DSMB (Matthew Schrag, Andreas Stahl, and Diethelm Messinger), the independent statistician Jens Pryzbilla, and REVISION investigators for their diligence, guidance, and support.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Alteplase/placebo is provided by Boehringer Ingelheim at no costs. Boehringer Ingelheim was given opportunity to review the manuscript for medical/scientific accuracy and intellectual property considerations. The authors did not receive any payment related to trial/manuscript development.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: German Federal Ministry of Education and Research (01KG2014 grant).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
