Abstract
Background:
Atrial fibrillation (AF) and congestive heart failure often coexist due to their shared risk factors leading to potential worse outcome, particularly cerebrovascular events. The aims of this study were to calculate the rates of ischemic and severe bleeding events in ischemic stroke patients having both AF and reduced ejection fraction (rEF) (⩽40%), compared to ischemic stroke patients with AF but without rEF.
Methods:
We performed a retrospective analysis that drew data from prospective studies. The primary outcome was the composite of either ischemic (stroke or systemic embolism), or hemorrhagic events (symptomatic intracranial bleeding and severe extracranial bleeding).
Results:
The cohort for this analysis comprised 3477 patients with ischemic stroke and AF, of which, 643 (18.3%) had also rEF. After a mean follow-up of 7.5 ± 9.1 months, 375 (10.8%) patients had 382 recorded outcome events, for an annual rate of 18.0%. While the number of primary outcome events in patients with rEF was 86 (13.4%), compared to 289 (10.2%) for the patients without rEF; on multivariable analysis rEF was not associated with the primary outcome (OR 1.25; 95% CI 0.84–1.88). At the end of follow-up, 321 (49.9%) patients with rEF were deceased or disabled (mRS ⩾3), compared with 1145 (40.4%) of those without rEF; on multivariable analysis, rEF was correlated with mortality or disability (OR 1.35; 95% CI 1.03–1.77).
Conclusions:
In patients with ischemic stroke and AF, the presence of rEF was not associated with the composite outcome of ischemic or hemorrhagic events over short-term follow-up but was associated with increased mortality or disability.
Introduction
Atrial fibrillation (AF) is the most common arrhythmia observed in clinical practice, accounting for approximately one-third of in-hospital admissions due to cardiac rhythm disturbances, the most common cardiac arrhythmia in the elderly, and is recognized as a major risk factor for stroke. 1 The coupling of AF and congestive heart failure (CHF) was demonstrated over 70 years ago. 2 Both conditions often coexist due to their shared risk factors, including hypertension, diabetes mellitus, ischemic heart disease, obesity, and valvular disease. The development of AF in patients with CHF has been reported to be associated with worse prognosis. Likewise, the development CHF in AF patients has also been associated with worse prognosis, 1 and is significantly associated with all-cause mortality. 3 Furthermore, an analysis of the Randomized Evaluation of Long-Term Anticoagulant Therapy (RE-LY) Study identified CHF as an independent predictor of all-cause mortality in AF patients and the strongest predictor of cardiac death. 4 Regarding the risk of stroke, Sartipy et al. 5 reported that in patients with reduced ejection fraction (rEF) this risk combined with death was higher in AF patients, compared to sinus rhythm patients (HR 1.19; 95% CI 1.14–1.26). Furthermore, current risk stratification schemes for AF focus on preventing stroke and systemic embolism by identifying those patients having major risk, that would require oral anticoagulation. The most commonly used score in this setting is the CHA2DS2-VASc score which incorporates a history of CHF as a risk marker. 6 However, a meta-analysis has reported a higher risk of mortality in patients with AF and rEF, compared to those with AF and preserved ejection fraction; the risks of stroke were reported to be similar in the two groups. 1 Another meta-analysis of the subgroup of patients with CHF enrolled in the phase III non-vitamin K antagonist (NOAC) trials, reported that the rates of systemic embolism, bleeding, or death, while on NOACs, were comparable to those of patients without CHF on NOACs. 7
In light of the above, the aims of this study were to calculate and compare the rates of ischemic and severe bleeding events in ischemic stroke patients having both AF with reduced ejection fraction (rEF) (⩽40%), and ischemic stroke patients with AF but without rEF. Moreover, functional clinical outcomes were assessed for both groups.
Study design and study population
We performed a retrospective study based on pooled individual data from an international collaboration of investigator-initiated prospective cohort studies. The following studies were included: the multicenter cohort studies Early Recurrence and Cerebral Bleeding in Patients with Acute Ischemic Stroke and Atrial Fibrillation (RAF and RAF-NOAC; 29 centers in Europe, Asia, and North America), Timing of initiation of oral anticoagulants in patients with acute ischemic stroke and atrial fibrillation comparing posterior and anterior circulation strokes (RAF-Posterior; 19 stroke units in Europe, North America, and Asia), and the Recurrent Ischemic Stroke and Bleeding in Patients with Atrial Fibrillation Who Suffered an Acute Stroke While on Treatment With NOACs (RENO-EXTEND; 43 centers in Europe and United States).
Details regarding the included studies can be obtained from the previously published studies.8–11
Data collection
Data was collected as described in prior publications: local investigators filled in standardized forms with predefined variables using individual patient data from their respective study databases. Completed forms were sent to the coordinating center in Perugia, Italy, where the pooled analysis was performed.8–11
Baseline data
The following baseline variables were investigated: age, sex; antithrombotic treatment (no treatment, antiplatelet agents, vitamin K antagonist (VKA), or NOAC including type of NOAC) before and after index event. NOAC therapy was defined as: apixaban 2.5 mg or 5 mg twice daily; dabigatran 110 mg or 150 mg twice daily; edoxaban 30 mg or 60 mg once daily; or rivaroxaban 15 mg or 20 mg once daily. VKA therapy was defined as treatment with acenocoumarol/warfarin. The choice of treatment was decided by the treating physician in accordance with international guidelines.
On admission, stroke severity was assessed using the National Institute of Health Stroke Scale (NIHSS).
The following risk factors were collected: history of hypertension (blood pressure of ⩾140/90 mmHg at least twice before stroke or treatment with antihypertensive drugs), history of diabetes mellitus (fasting glucose level ⩾126 mg/dL preprandial on two measurements, glucose level ⩾200 mg/dL post-prandial, or HbA1c ⩾6.5%, or diabetic treatment), current cigarette smoking, hyperlipidemia (total cholesterol ⩾200 mg/dL or triglyceride ⩾140 mg/dL or lipid lowering therapy), history of symptomatic ischemic heart disease (myocardial infarction, angina, existence of multiple lesions on thallium heart isotope scan, or evidence of coronary disease on coronary angiography), current alcohol abuse (⩾300 g per week), treatment with statins after the index stroke or previous stroke/transient ischemic attack (TIA). On admission, a cerebral CT without contrast or an MRI scan was performed on all patients to exclude intracranial hemorrhage. We assessed the presence of white matter changes (leukoaraiosis defined on the first computed tomography examination as ill-defined and moderately hypodense areas of ⩾5 mm according to published criteria). Leukoaraiosis in the deep white matter was dichotomized into absent versus present (mild, moderate, or severe). 12
AF was classified as paroxysmal when episodes terminated spontaneously within 7 days.
The CHA2DS2-VASc score (2 points for history of stroke or age >75 years and 1 point each for CHF, hypertension, diabetes mellitus, vascular disease, age between 65 and 74 years, and female sex) was calculated before and after the index event. 6
Any concurrent antiplatelet use prior to and after the index stroke was also recorded. Patients treated with antiplatelets, had been taking aspirin 75–300 mg per day or clopidogrel 75 mg per day.
Classification of ejection fraction
Transthoracic echocardiogram (TTE) was performed within 7 days of stroke onset by an in-house cardiologist using a standardized protocol. Patients were imaged while in a left lateral decubitus position. Images were obtained using a 3.5 MHz transducer, at a depth of 16 cm in the parasternal (standard long- and short-axis images) and apical views (standard long-axis, two- and four-chamber images). Standard two-dimensional and color Doppler data, triggered to the QRS complex, were saved in cine loop format. Pulsed and continuous wave Doppler data were also stored digitally. On TTE, increased end-diastolic diameter and volume (LV diameter over 60 mm or 32 mm/m2 with LV volume exceeding 97 mL/m2) and end-systolic diameter and volume (LV diameter greater than 45 mm or 25 mm/m2 with LV volume over 43 mL/m2) were measured. Ejection fraction was calculated using the modified Simpson or the modified Quinones method. 13 Patients were classified as being with rEF (⩽40%) or without rEF (>40%). 14
Outcome evaluation
Patients were followed-up prospectively either in person or by telephone interviews over a duration of at least 3 months from the time of the index event. The initiation of oral anticoagulation followed international guideline recommendations. 15
The primary outcome was the composite of ischemic stroke, systemic embolism, intracranial bleeding, and major extracranial bleeding. Recurrent stroke was defined as the sudden onset of a new focal neurological deficit of vascular origin in a site consistent with the territory of a major cerebral artery and was categorized as ischemic or hemorrhagic. Regarding systemic embolism, it was defined as an acute vascular occlusion of an extremity or organ confirmed by imaging, surgery, or autopsy. Intracranial bleeding was defined as a spontaneous hemorrhagic stroke (intraparenchymal bleeding), subdural, or subarachnoid hemorrhage, whereas traumatic intracranial bleeding was not considered as an outcome event. Major extracranial bleeding was defined as either a reduction in the hemoglobin level of 2 g/dL or more, the requirement of a blood transfusion of at least two units, or symptomatic bleeding in either a critical area or organ such as intraspinal, intraocular, retroperitoneal, intra-articular or pericardial, or intramuscular with compartment syndrome.
Death was recorded and functional recovery was assessed by the modified Rankin Scale (mRS), dichotomizing between functional independence (mRS score 0–2) and disability (mRS score ⩾3). 16 Follow-up visits and outcome adjudication were performed by local investigators, in a non-blinded manner.
Statistical analysis
For patients with or without outcome events, differences in clinical characteristics and risk factors were calculated using the χ 2 test of proportions (with a two-sided α level of 5%). To this regard, 95% confidence intervals (CIs) were calculated for odds ratio (OR). Moreover, multivariable logistic regression analysis was performed to identify independent associations for outcome events and the variables included in this analysis were the following: age, sex, hypertension, diabetes, mellitus, hyperlipidemia, current alcohol abuse, current smoking habit, paroxysmal AF, history of stroke or TIA, history of myocardial infarction, therapy with oral anticoagulants, therapy with statins, presence of leukoaraiosis, antiplatelet therapy in addition to oral anticoagulant therapy after the index stroke and rEF. For mortality and disability, the same analyses were performed. Furthermore, for outcome events, survival and empirical cumulative hazard functions were estimated via the Kaplan-Meier estimator for the two groups. Patients were censored at the time of an outcome event or death.
In order to compare the risk of outcome events in those patients who had rEF treated with NOACs, with patients who had rEF treated with vitamin K antagonist (VKA), the relation between the survival function and the set of explanatory variables was calculated by Cox proportional hazard model.
Data were analyzed using the SPSS/PC Win Package 25.0.
Results
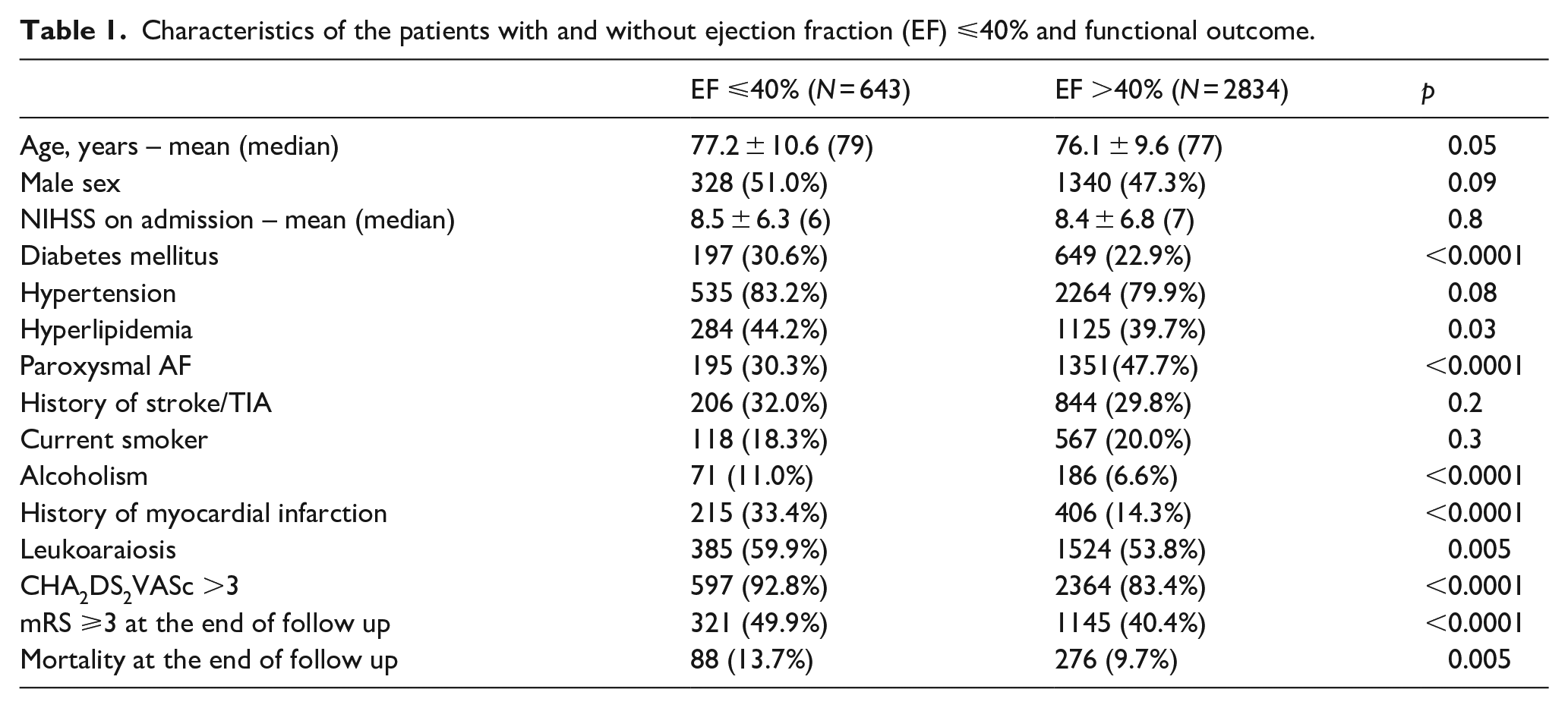
The final cohort comprised of 3477 of the original 3869 patients (89.9%) with ischemic stroke and AF from the pooled data sets: 252 patients were excluded due to duplication (patients present in more than one database of the cohort populations considered) and 140 had not undergone a TTE during hospitalization. In this cohort, 643 patients (18.3%) had rEF and their characteristics are summarized in Table 1. Independent associations of rEF included history of myocardial infarction (OR 3.74; 95% CI 2.79–5.02), alcohol abuse (OR 2.18; 95% CI 1.40–3.38), and diabetes mellitus (OR 1.37; 95% CI 1.04–1.80) whereas, the presence of paroxysmal AF was inversely associated with rEF (OR 0.35; 95% CI 0.27–0.47).
Characteristics of the patients with and without ejection fraction (EF) ⩽40% and functional outcome.
Factors associated with ischemic and bleeding events
After a mean follow-up of 7.5 ± 9.1 months (2173 patient-years), in 375 patients (10.8%) 382 outcome events were recorded, corresponding to an annual rate of 18.0%. The following outcome events were observed: 199 ischemic strokes, 31 systemic embolisms, 79 intracranial bleedings, and 73 major extracranial bleedings. The demographic and clinical characteristics of the patients with or without outcome events are summarized in Tables S1–S3.
The number of recorded outcome events in patients with rEF was 86 (13.4%) combined, 58 (9.0%) ischemic, and 29 (4.5%) hemorrhagic compared to patients without rEF, where there were 289 (10.2%) combined events, 172 (6.1%) ischemic events, and 123 (4.4%) hemorrhagic events (OR 1.36; 95% CI 1.05–1.64, OR 1.53; 95% CI 1.12–2.09, and OR 1.04; 95% CI 0.69–1.57, respectively) (Table 2).
Outcome events in patients with or without ejection fraction (EF) ⩽40%.
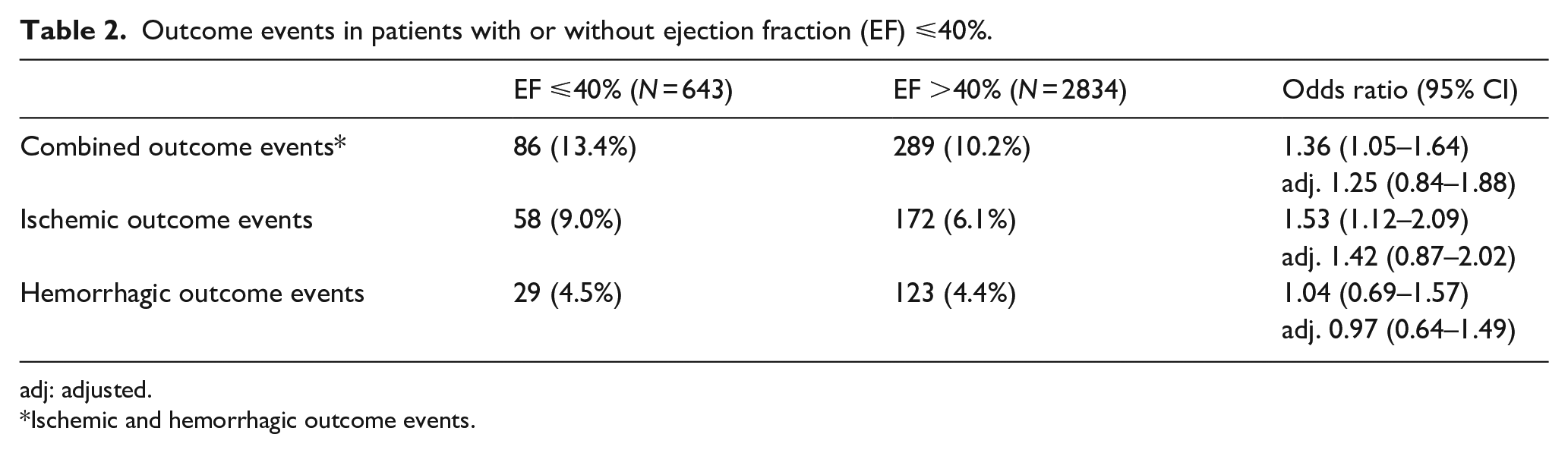
adj: adjusted.
Ischemic and hemorrhagic outcome events.
Factors independently associated with the different outcomes are reported in Table 3; rEF was not associated with any outcome: OR 1.25; 95% CI 0.84–1.88 for combined outcome, OR 1.42; 95% CI 0.87–2.02 for ischemic outcome, and OR 0.97; 95% CI 0.64–1.49 for hemorrhagic outcome.
Factors independently associated with the different outcomes.
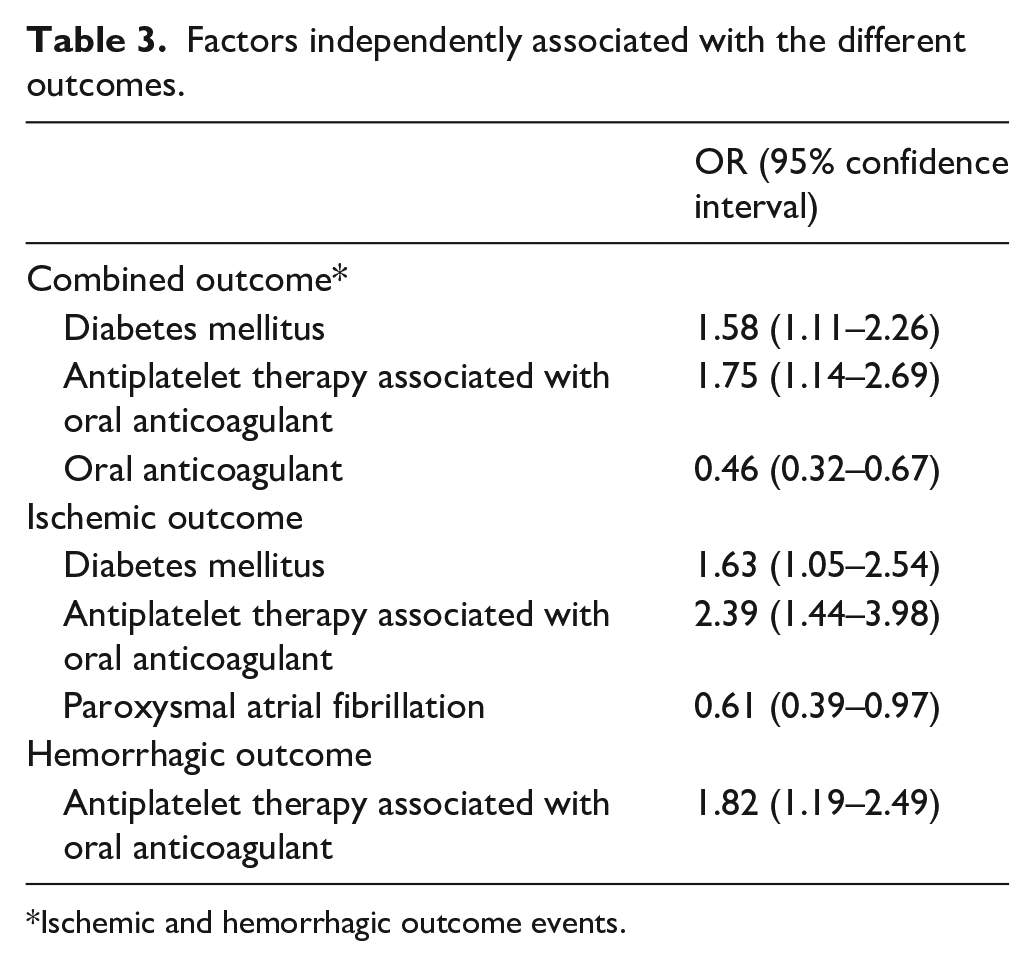
Ischemic and hemorrhagic outcome events.
The Kaplan-Meier curves that compared the overall outcome events in those patients with and without rEF are shown in Figure 1.
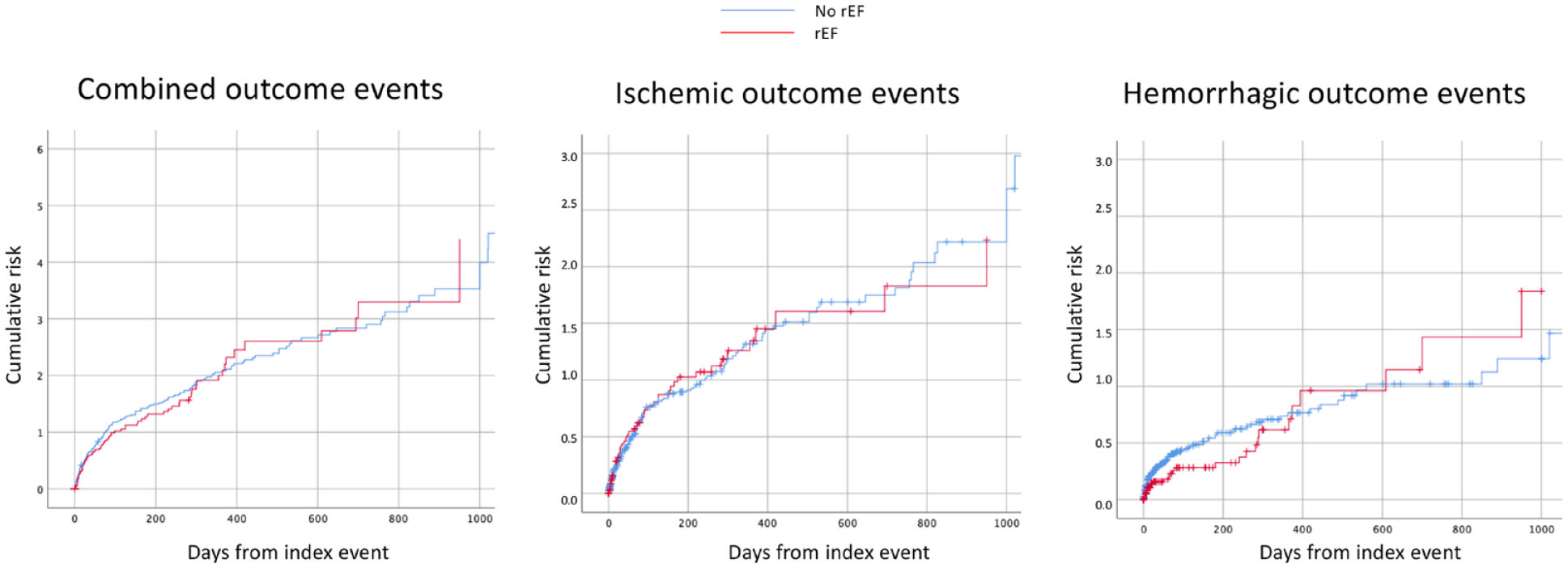
Cumulative risk of combined, ischemic, or hemorrhagic endpoint events in patients with and without reduced ejection fraction.
Rates and associated factors of mortality or disability
Regarding functional outcome, at the end of follow-up, 1466 (42.2%) patients were deceased or disabled (mRS ⩾ 3), whereas 364 (10.5%) were deceased. Comparison-wise, 321 (49.9%) with rEF were deceased or disabled, compared to 1145 (40.4%) without (OR 1.47; 95% CI 1.24–1.75), while 88 (13.7%) with rEF were deceased, compared to 276 (9.7%) without (OR 1.47; 95% CI 1.14–1.90). On multivariable analysis, rEF was correlated with mortality or disability (OR 1.35; 95% CI 1.03–1.77) but not with mortality (OR 1.13; 95% CI 0.69–1.87).
Rates of ischemic and bleeding events in patients with rEF treated with NOACS or with VKA
The treatment approaches for secondary stroke prevention in patients with rEF were the following: 421 (65.5%) NOACs, 129 (20.1%) VKA, and 93 (14.5%) received no oral anticoagulants. The demographic and clinical characteristics of the patients treated with NOACs or VKA are summarized in Table S4. The recorded outcome events in patients treated with NOACs were 46 (10.9%) combined, 32 (7.6%) ischemic, and 14 (3.3%) hemorrhagic compared to patients treated with VKA where there were 25 (19.4%) combined events, 17 (13.2%) ischemic events, and 8 (6.2%) hemorrhagic events (OR 0.51; 95% CI 0.30–0.87, OR 0.54; 95% CI 0.29–1.01, and OR 0.46; 95% CI 0.19–1.09, respectively). The Cox regression curve analysis that compared the overall outcome events in those patients treated with NOACs or VKA is reported in Figure 2. Within this analysis, VKA was associated with a non-statistically significant increase in combined outcome events compared to NOACs (HR 1.65; 95% CI, 0.95–2.86; p = 0.07). Finally, of those 93 patients who had not received oral anticoagulants, 14 (15.1%) had combined events, 9 (9.7%) ischemic events, and 5 (5.4%) hemorrhagic events while 37 (39.8%) were deceased.
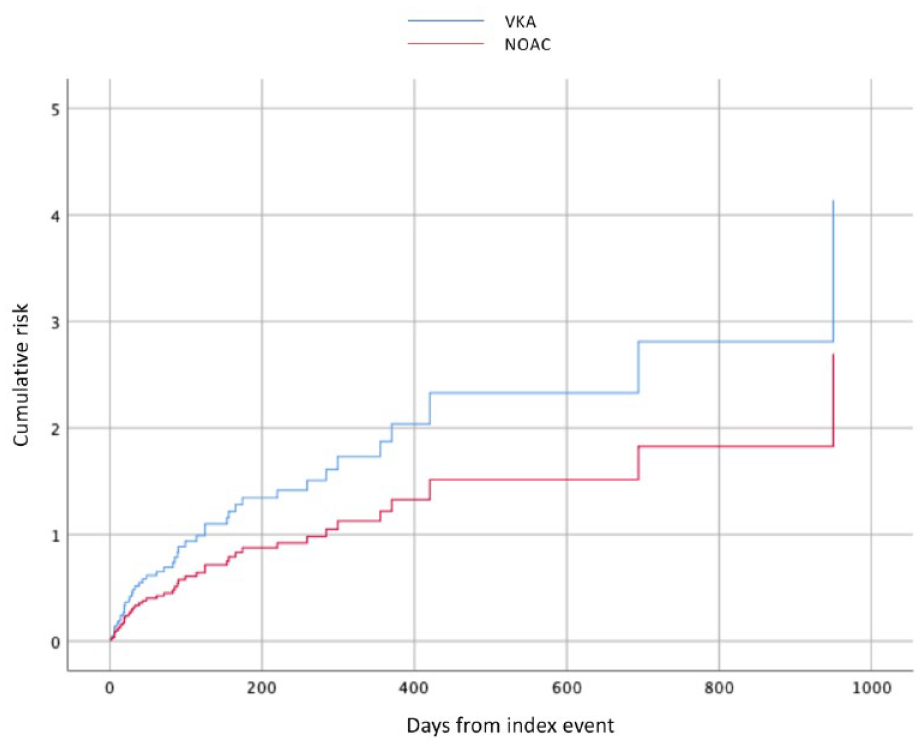
Cumulative risk of combined endpoint events in patients with reduced ejection fraction treated with NOAC or with vitamin-K antagonist.
Discussion
In this pooled retrospective analysis that drew upon prospective cohort data, about 18% of the patients with ischemic stroke and AF had rEF, and the latter was associated with a history of myocardial infarction, diabetes mellitus, and alcohol abuse, while it was inversely associated with the presence of paroxysmal AF; rEF was not associated with the composite outcome of ischemic or hemorrhagic events, but was associated with mortality or disability.
Regarding etiology, rEF has been reported to be a possible consequence of AF due to rapid pulse (tachycardia) that generally leads to atrial morphological changes with subsequent cardiomyopathy 17 and reduced left atrial “kick.” 18 About 70% of the rEF patients included in our study had persistent or permanent AF, compared to about 52% of patients without rEF. In fact, persistent or permanent AF have been reported to be more commonly associated with cardiac structural degeneration, compared to paroxysmal AF. This cardiac structural degeneration has been associated with subsequent stroke risk. 19 Conversely, structural changes associated with chronic CHF make AF much more prevalent. 17
Concerning functional outcome, patients with rEF had index strokes with similar severities, measured with NIHSS on admission, compared to those patients without rEF. Furthermore, the risks of either embolic recurrences or hemorrhagic complications were similar in patients with or without rEF. Despite the above findings, patients with rEF were more likely to be deceased or disabled at the end of follow-up. The principal cause of this worse functional outcome, might plausibly be explained by a progression of heart failure over time. To this regard, a retrospective analysis of the Studies of Left Ventricular Dysfunction (SOLVD) trial, which had enrolled 6500 patients with left ventricular ejection fraction ⩽35%, reported that the presence of AF was an independent predictor for both progressive pump failure, and the combined end point of death or hospitalization for heart failure. 20
As for treatment, a meta-analysis reported that in patients with AF and CHF, NOACs significantly reduced the risks of stroke, systemic embolism, and bleeding events, including major bleeding and intracranial hemorrhage, compared with warfarin. No interactions in the efficacy or safety profile of NOACs compared with warfarin were observed in patients either with or without heart failure. 7 In our study, the risk of combined events (ischemic and hemorrhagic) was 10.9% in patients having AF and reduced eEF and prescribed with NOACs, compared to 19.4% for those prescribed with warfarin. From Cox regression analysis, this difference did not result being statistically significant, despite a clear trend in favor of NOACs. Our study, however, was not designed to evaluate this aim.
The strengths of our study included its adequate sample size and its prospective design of the included studies. Moreover, our findings reflect real-life experiences and may provide critical observational information. In fact, in view of a lack of any randomized data, the use of predictive factors to assess prognosis has been shown to improve quality of life, as well as long-term outcome.
Our study had several limitations. First, the reported results could have been influenced by unmeasured confounders which could not be overcome in adjusted statistical models which were used to reduce their impact. Second, we did not collect data regarding any treatment for heart failure prescribed for each patient during follow-up, including therapies for rate control or rhythm restoration and rhythm control, ablation, or pacemaker. Third, we did not collect data on patients with a history of heart failure but with preserved ejection fraction, this is because we had dichotomized the cohort into patients with or without rEF. To this regard, studies have reported differing outcomes in patients with heart failure and rEF, compared to those with heart failure and preserved ejection fraction.1,21 Fourth, 4% of the patients were excluded because of lacking TTE, and these patients might have been severely disabled or terminal patients, which would have affected the composite outcome. Finally, the duration of follow-up was limited to a mean of 7.5 months which might not have been long enough to evidence a difference in stroke recurrence between the two groups, as there was evidence of a trend toward higher vascular events in the rEF group.
In conclusion, our study results suggest that, in patients with ischemic stroke and AF, the presence of rEF was not associated with combined, ischemic, or hemorrhagic events over short-term follow-up but was associated with mortality or disability at the end of follow-up.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231177625 – Supplemental material for The risk of stroke recurrence in patients with atrial fibrillation and reduced ejection fraction
Supplemental material, sj-docx-1-eso-10.1177_23969873231177625 for The risk of stroke recurrence in patients with atrial fibrillation and reduced ejection fraction in European Stroke Journal
Footnotes
Acknowledgements
None
Declaration of conflicting interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M. Paciaroni received honoraria as a member of the speaker bureau of Sanofi-Aventis, Bayer, Bristol Meyer Squibb, Daiiki Sankyo and Pfizer.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The studies had been approved by the pertinent institutional review boards if required.
Informed consent
Informed consent had been obtained according to local requirements.
Guarantor
Maurizio Paciaroni
Contributorship
Maurizio Paciaroni conceived the study and wrote the first draft of the manuscript. All Investigators were involved in protocol development, gaining ethical approval, patient recruitment, and data analysis. All Investigators reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
