Abstract
Rationale
Atrial fibrillation causes one-fifth of ischemic strokes, with a high risk of early recurrence. Although long-term anticoagulation is highly effective for stroke prevention in atrial fibrillation, initiation after stroke is usually delayed by concerns over intracranial hemorrhage risk. Direct oral anticoagulants offer a significantly lower risk of intracranial hemorrhage than other anticoagulants, potentially allowing earlier anticoagulation and prevention of recurrence, but the safety and efficacy of this approach has not been established.
Aim
Optimal timing of anticoagulation after acute ischemic stroke with atrial fibrillation (OPTIMAS) will investigate whether early treatment with a direct oral anticoagulant, within four days of stroke onset, is as effective or better than delayed initiation, 7 to 14 days from onset, in atrial fibrillation patients with acute ischemic stroke.
Methods and design
OPTIMAS is a multicenter randomized controlled trial with blinded outcome adjudication. Participants with acute ischemic stroke and atrial fibrillation eligible for anticoagulation with a direct oral anticoagulant are randomized 1:1 to early or delayed initiation. As of December 2021, 88 centers in the United Kingdom have opened.
Study outcomes
The primary outcome is a composite of recurrent stroke (ischemic stroke or symptomatic intracranial hemorrhage) and systemic arterial embolism within 90 days. Secondary outcomes include major bleeding, functional status, anticoagulant adherence, quality of life, health and social care resource use, and length of hospital stay.
Sample size target
A total of 3478 participants assuming event rates of 11.5% in the control arm and 8% in the intervention arm, 90% power and 5% alpha. We will follow a non-inferiority gatekeeper analysis approach with a non-inferiority margin of 2 percentage points.
Discussion
OPTIMAS aims to provide high-quality evidence on the safety and efficacy of early direct oral anticoagulant initiation after atrial fibrillation-associated ischemic stroke.
Introduction and rationale
Atrial fibrillation (AF) causes at least 20% of all ischemic strokes. 1 Oral anticoagulation reduces the risk of stroke in AF by around two-thirds,2,3 but its safety and benefit in acute ischemic stroke is unclear. Current practice—based on limited observational data and expert opinion—is to delay anticoagulation by up to two weeks, during which the daily risk of recurrence is 0.4–1.3%.4–7 Earlier anticoagulation might prevent many recurrent ischemic strokes but might cause intracranial hemorrhage (ICH), including intracerebral hemorrhage due to hemorrhagic transformation of the infarct (Supplementary Figure 1), which is associated with poor outcome if severe. 8 The optimal timing of anticoagulation after AF-associated ischemic stroke is thus a frequent dilemma in stroke medicine.
Historical evidence suggests that anticoagulation within 48 h of stroke onset with vitamin K antagonists (VKAs) or heparins is not beneficial because an increased risk of symptomatic ICH outweighs any reduction in ischemic stroke. 9 However, direct oral anticoagulants (DOACs) are as effective in ischemic stroke prevention, but with half the risk of ICH of VKAs. 3 Although the large randomized trials of DOACs excluded participants with acute ischemic stroke, 10 increasing observational data suggest they might be used safely in acute AF-associated stroke (Supplementary Table 1). However, this evidence is limited by selection bias and may not generalize beyond mild–moderate stroke. 10 Randomized controlled trials (RCTs) are therefore needed, are supported by stroke physicians, who report clinical uncertainty regarding optimal anticoagulation timing, 11 and have been called for by international guidelines.12,13
The OPtimal TIMing of Anticoagulation after acute ischemic Stroke trial (OPTIMAS) aims to establish the safety and efficacy of early anticoagulation with a DOAC after AF-associated stroke.
Methods and design
OPTIMAS is a phase IV multicenter RCT with an open-label intervention, blinded end-point adjudication, and hierarchical non-inferiority/superiority gatekeeper design, comparing a policy of early DOAC initiation, within four days of stroke onset, to delayed initiation, 7 to 14 days from onset, in patients with AF and acute ischemic stroke.
Patient population
OPTIMAS will recruit 3478 participants within four days of stroke onset, from UK stroke units. Sites are listed at https://optimas.org.uk.
Inclusion criteria
≥18 years old AF (confirmed by ECG recording or medical records) Clinical diagnosis of acute ischemic stroke Eligible for anticoagulation with DOAC Treating physician is uncertain regarding the potential participant’s optimal time to start DOAC
Exclusion criteria
Coagulopathy or current anticoagulation with International Normalised Ratio (INR) ≥1.7 at randomization (anticoagulant-treated patients not excluded if INR <1.7); clinically significant thrombocytopenia (platelets <75 × 109/L) Severe hemorrhagic transformation of the infarct (ECASS PH2)
14
or acute ICH unrelated to infarct DOAC use contraindicated: severe renal impairment (creatinine clearance <15 ml/min); cirrhosis (Child Pugh B or C) or ALT > 2 × upper limit of normal; concurrent medication with significant interaction (e.g. strong CYP3A4 inducers); definite indication for VKA use Pregnancy or breastfeeding
Randomization
Participants will be randomized in a 1:1 ratio using an independent online randomization service with random permuted blocks and randomly varying block lengths, stratified by National Institutes of Health Stroke Scale (NIHSS) at randomization (0–4, 5–10, 11–15, 16–21, > 21). The participant and treating clinicians will not be blinded to allocation, but outcomes will be adjudicated blind.
Intervention
The intervention group will initiate a DOAC within four days of stroke onset (or the time symptoms were first noted, if the onset time cannot be determined). The control group will initiate a DOAC 7 to 14 days after onset, an interval selected based on a 2018 survey of UK practice. 11 The treating clinician determines the exact timing of anticoagulation in each group. Any DOAC licensed for stroke prevention in AF (i.e. apixaban, dabigatran, edoxaban, rivaroxaban) may be used. The dose may be reduced if recommended by the relevant Summary of Product Characteristics.15–18 All other clinical care follows current UK best practice. 19
Primary outcome
The primary outcome is a composite of recurrent ischemic stroke, symptomatic ICH (including hemorrhagic transformation), unclassifiable stroke syndromes, and systemic arterial embolism incidence at 90 days post-randomization. Primary outcome events will be adjudicated centrally by independent expert clinicians blinded to treatment allocation.
Secondary outcomes
Secondary efficacy outcomes include all-cause and cardiovascular mortality, recurrent ischemic stroke, systemic embolism, and venous thromboembolism. Safety outcomes include symptomatic ICH, its anatomical subtypes, major extracranial bleeding, 20 and clinically relevant non-major bleeding. 21 We will record functional (modified Rankin Scale), cognitive (MoCA), and patient-reported (EQ-5D-5 L, PROMIS-10) outcomes, length of hospital stay, medication adherence, and health and social care resource use.
Data collection
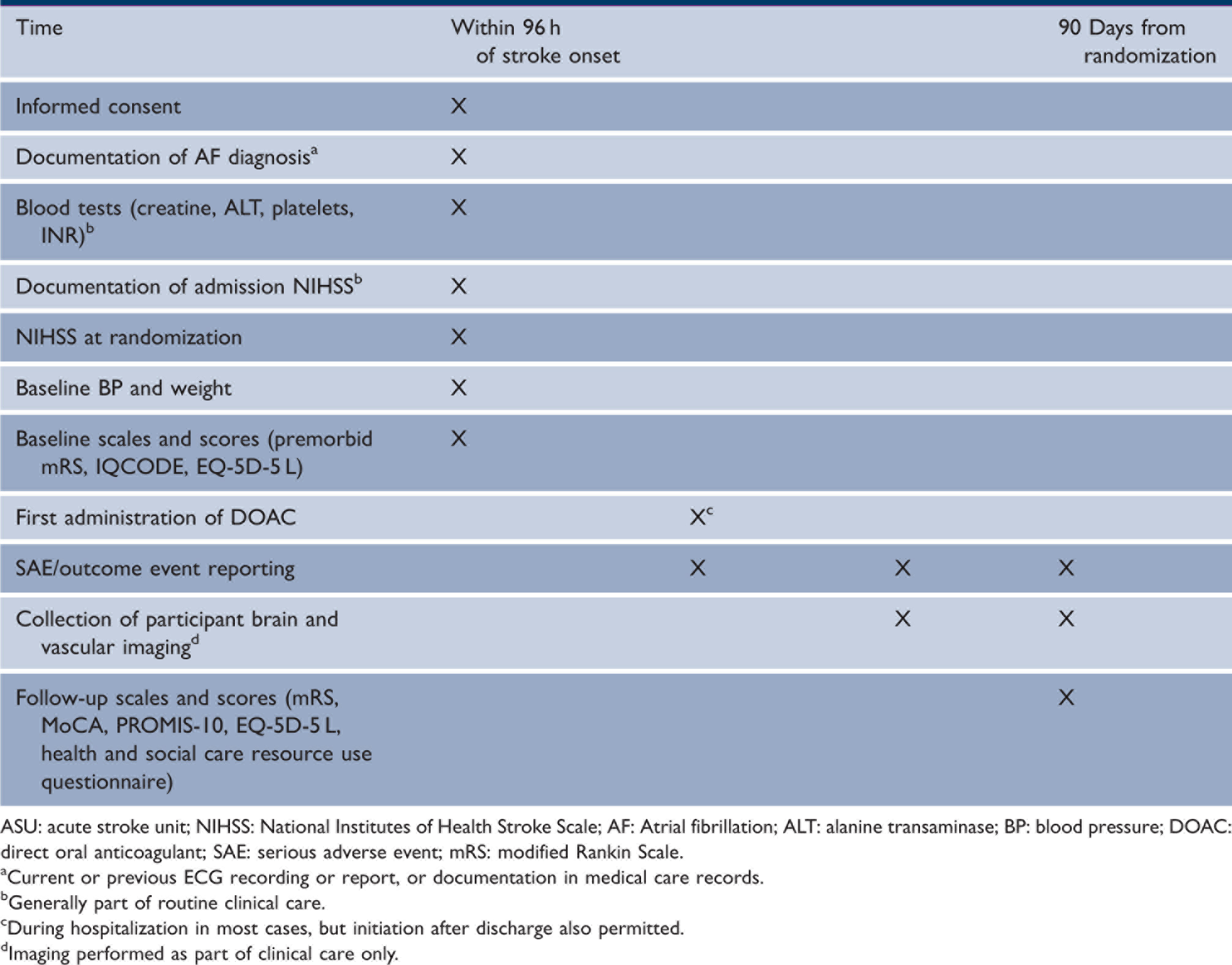
Trial data are collected via a secure online electronic data capture system, and pseudonymized clinical imaging data via a secure file transfer portal. Table 1 shows the schedule of study assessments and interventions, and Figure 1 shows the study flowchart.
Study flowchart. Schedule of study assessments and interventions. ASU: acute stroke unit; NIHSS: National Institutes of Health Stroke Scale; AF: Atrial fibrillation; ALT: alanine transaminase; BP: blood pressure; DOAC: direct oral anticoagulant; SAE: serious adverse event; mRS: modified Rankin Scale. Current or previous ECG recording or report, or documentation in medical care records. Generally part of routine clinical care. During hospitalization in most cases, but initiation after discharge also permitted. Imaging performed as part of clinical care only.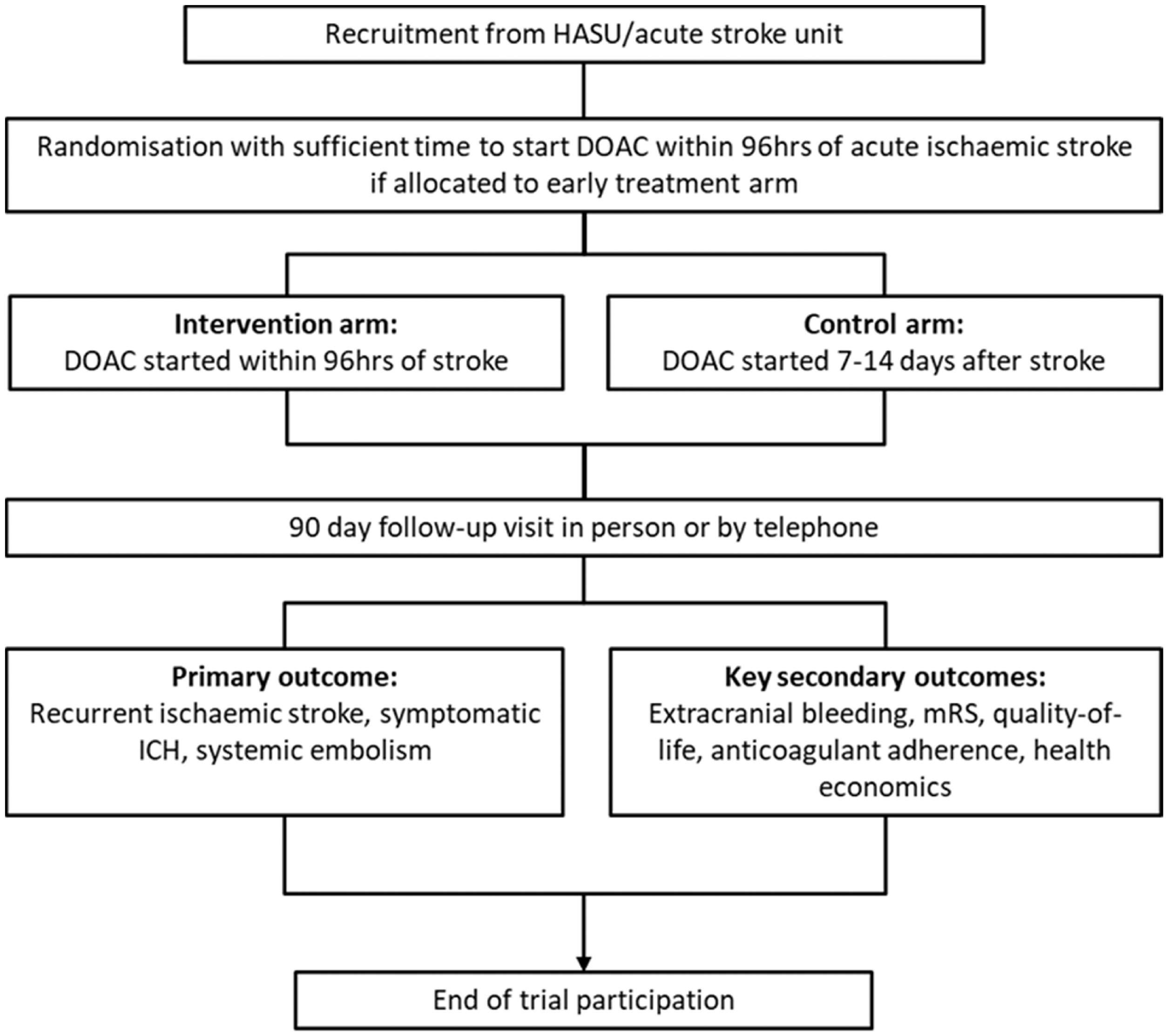
Sample size estimates
Our planned sample size assumed a reduction in the primary outcome event rate from 11.5% in the control group to 8% in the intervention group (a relative risk reduction of 30%). We judged this to be a clinically meaningful benefit likely to influence guidelines and practice. The assumed composite event rate and hypothesized effect size were derived from the Virtual International Stroke Trials Archive of trials in patients with ischemic stroke and AF. 22 The sample size calculation used 90% power for superiority, significance level 5%, and was inflated by 10% for loss to follow-up or other challenges; we anticipate a much lower rate than this. Based on the expected event rate and a non-inferiority margin of 3%, a sample size of 3478 evaluable participants would have had 80% power for non-inferiority.
We re-evaluated study power in November 2021 at the request of our Independent Data Monitoring Committee, due to a lower-than-expected interim adjudicated primary outcome rate of 4.3%. Given this event rate, our planned sample size has 80% power to show non-inferiority based on a non-inferiority margin of 2%, and 80% power for superiority assuming an odds ratio reduction of 38%.
Statistical analyses
Our main analysis will follow the intention-to-treat principle. Outcome data will be collected from all participants enrolled, unless consent is specifically withdrawn (in which case data will be included up to the point of withdrawal). We will first test for non-inferiority of the intervention, using a non-inferiority margin of 2 percentage points identified as clinically acceptable. If non-inferiority is established, we will test for superiority. For our primary outcome, we will use mixed-effects logistic regression including an independent variable indicating treatment allocation, with adjustment for stroke severity (NIHSS) at randomization. Sites will be included as random intercept terms.
The health economic evaluation will calculate the mean incremental cost per quality adjusted life year (QALY) gained by early initiation of a DOAC. Cost-effectiveness will primarily be evaluated using cost-effectiveness acceptability curves generated from bootstrapped results to calculate the probability that early initiation of DOAC is cost-effective compared to late initiation for a range of values of willingness to pay for a QALY gained.
Further details of our statistical and health economic analyses are presented in online Appendix. A full statistical and health economic analysis plan will be published before the end of recruitment. Prespecified secondary statistical analyses will include: a per-protocol analysis; a mediation analysis; and analyses by stroke severity (NIHSS) and neuroimaging features including infarct location, volume, hemorrhagic transformation (including subtypes of hemorrhagic infarction and parenchymal hematoma 14 ), and cerebral small vessel disease markers.
Study organization and funding
The study is funded by the British Heart Foundation (CS/17/6/333561) and sponsored by University College London. Online Appendix describes study approvals and governance, key study committees (Supplementary Tables 2 to 7), and arrangements for data monitoring, safety reporting, and quality assurance. Study results will be presented at scientific meetings and published in peer-reviewed journals.
Discussion
OPTIMAS is a prospective RCT based in routine clinical practice throughout the UK, addressing the important clinical uncertainty of the optimal timing of anticoagulation for secondary prevention of cardioembolic stroke, with broad eligibility criteria intended to give a representative study sample and results readily applicable to clinical practice.
We decided against imaging-based eligibility criteria. Although infarct size is a risk factor for hemorrhagic transformation, 8 to our knowledge, it has not been shown that anticoagulation timing and infarct size interact with respect to the risks of clinically significant hemorrhagic transformation and adverse clinical outcomes, although these considerations often feature in expert guidance. 23 Furthermore, larger infarct size is also a risk factor for recurrent ischemic stroke in AF patients.24,25 Visual classifications of infarct size are based mainly on vascular anatomy and expert opinion, 8 and accurate measurement requires diffusion-weighted magnetic resonance imaging (or a delayed computed tomography) and trained raters, increasing the time and complexity of establishing eligibility, an important consideration in a time-sensitive trial. Our approach concurs with that of the Swedish TIMING trial (ClinicalTrials.gov NCT02961348), 26 facilitating pooled individual patient data analyses and will complement the international imaging-based ELAN trial (which varies anticoagulation timing according to infarct size; NCT03148457) and the United States’ START trial (which excludes participants with large infarct volumes; NCT03021928), 27 with the possibility of aligning imaging-based subgroup analyses.
We chose a hierarchical non-inferiority then superiority gatekeeper design because: (1) a finding of non-inferiority but not superiority at our chosen margin of 3 percentage points would give confidence that early anticoagulation with a DOAC is of similar clinical benefit to delayed anticoagulation, so could reasonably be chosen for its practical advantages and (2) early anticoagulation might have advantages beyond prevention of stroke and systemic arterial embolism, such as improved adherence and reduced length of stay.
No participants in OPTIMAS will be randomized to start anticoagulation between four and seven days after onset per protocol. This separation between treatment groups aims to minimize crossovers and ensure that the two groups receive different timings of treatment onset. Our best judgement was that these advantages would outweigh the possibility that the optimal timing of anticoagulation might be within this period. A large-scale pooled analysis with other anticoagulation timing trials is planned, giving full coverage of the first two weeks after stroke onset and power to explore this issue in detail.
Summary and conclusions
OPTIMAS will determine the efficacy and safety of early anticoagulation with a DOAC in patients with acute ischemic stroke and AF, a strategy with the potential to prevent early recurrent ischemic strokes, shorten hospital stays, and improve quality of life.
Supplemental Material
sj-jpg-1-wso-10.1177_17474930211057022 – Supplemental material for Optimal timing of anticoagulation after acute ischemic stroke with atrial fibrillation (OPTIMAS): Protocol for a randomized controlled trial
Supplemental material, sj-jpg-1-wso-10.1177_17474930211057022 for Optimal timing of anticoagulation after acute ischemic stroke with atrial fibrillation (OPTIMAS): Protocol for a randomized controlled trial by Jonathan G Best, Liz Arram, Norin Ahmed, Maryam Balogun, Kate Bennett, Ekaterina Bordea, Marta G Campos, Emilia Caverly, Marisa Chau, Hannah Cohen, Hakim-Moulay Dehbi, Caroline J Doré, Stefan T Engelter, Robert Fenner, Nick Freemantle, Rachael Hunter, Martin James, Gregory YH Lip, Macey L Murray, Bo Norrving, Nikola Sprigg, Roland Veltkamp, Iwona Zaczyk and David J Werring in International Journal of Stroke
Supplemental Material
sj-jpg-2-wso-10.1177_17474930211057022 – Supplemental material for Optimal timing of anticoagulation after acute ischemic stroke with atrial fibrillation (OPTIMAS): Protocol for a randomized controlled trial
Supplemental material, sj-jpg-2-wso-10.1177_17474930211057022 for Optimal timing of anticoagulation after acute ischemic stroke with atrial fibrillation (OPTIMAS): Protocol for a randomized controlled trial by Jonathan G Best, Liz Arram, Norin Ahmed, Maryam Balogun, Kate Bennett, Ekaterina Bordea, Marta G Campos, Emilia Caverly, Marisa Chau, Hannah Cohen, Hakim-Moulay Dehbi, Caroline J Doré, Stefan T Engelter, Robert Fenner, Nick Freemantle, Rachael Hunter, Martin James, Gregory YH Lip, Macey L Murray, Bo Norrving, Nikola Sprigg, Roland Veltkamp, Iwona Zaczyk and David J Werring in International Journal of Stroke
Supplemental Material
sj-pdf-3-wso-10.1177_17474930211057022 – Supplemental material for Optimal timing of anticoagulation after acute ischemic stroke with atrial fibrillation (OPTIMAS): Protocol for a randomized controlled trial
Supplemental material, sj-pdf-3-wso-10.1177_17474930211057022 for Optimal timing of anticoagulation after acute ischemic stroke with atrial fibrillation (OPTIMAS): Protocol for a randomized controlled trial by Jonathan G Best, Liz Arram, Norin Ahmed, Maryam Balogun, Kate Bennett, Ekaterina Bordea, Marta G Campos, Emilia Caverly, Marisa Chau, Hannah Cohen, Hakim-Moulay Dehbi, Caroline J Doré, Stefan T Engelter, Robert Fenner, Nick Freemantle, Rachael Hunter, Martin James, Gregory YH Lip, Macey L Murray, Bo Norrving, Nikola Sprigg, Roland Veltkamp, Iwona Zaczyk and David J Werring in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DJW reports personal fees from Bayer, Portola and Alnylam. STE has received funding for travel or speaker honoraria from Bayer, Boehringer Ingelheim, BMS/Pfizer and Daiichi-Sankyo. His institution has received an educational grant from Pfizer and research support from Daiichi-Sankyo. BN report personal fees from Astra Zeneca and Bayer. MJ reports travel or speaker honoraria from Daiichi-Sankyo, Portola, Boehringer Ingelheim. RV reports research funding from Bayer, Boehringer Ingelheim, BMS, Pfizer, Daiichi Sankyo, Medtronic, and has received speaker honoraria or personal fees from Bayer, BMS, Pfizer, Abbott, Astra Zeneca, Javelin. HC reports, outside the submitted work, institutional research support and support to attend scientific meetings from Bayer Healthcare, and travel support from UCB Biopharma, with honoraria for lectures from Bayer Healthcare and consultancy fees from UCB Biopharma paid to University College London Hospitals Charity. GYHL has been a consultant and speaker for BMS/Pfizer, Boehringer Ingelheim and Daiichi-Sankyo. No fees are received personally. All other authors declare no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: HC, CD, STE, MJ, GYHL, BN, NS, RV, and DJW report a grant from the British Heart Foundation for OPTIMAS. MJ is supported by the NIHR South West Peninsula Applied Research Collaboration. RV is partially supported by the European Union's Horizon 2020 research and innovation programme under grant agreement No. 754517 (PRESTIGE-AF). All other authors declare no grants or funding in respect of the work.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
