Abstract
Introduction
The aim of this study in patients with acute posterior ischaemic stroke (PS) and atrial fibrillation (AF) was to evaluate (1) the risks of recurrent ischaemic event and severe bleeding and (2) these risks in relation with oral anticoagulant therapy (OAT) and its timing.
Materials and Methods
Patients with PS were prospectively included; the outcome events of these patients were compared with those of patients with anterior stroke (AS) which were taken from previous registries. The primary outcome was the composite of stroke recurrence, transient ischaemic attack, symptomatic systemic embolism, symptomatic cerebral bleeding and major extracranial bleeding occurring within 90 days from acute stroke.
Results
A total of 2470 patients were available for the analysis: 473 (19.1%) with PS and 1997 (80.9%) with AS. Over 90 days, 213 (8.6%) primary outcome events were recorded: 175 (8.7%) in patients with AS and 38 (8.0%) in those with PS. In patients who initiated OAT within 2 days, the primary outcome occurred in 5 out of 95 patients (5.3%) with PS compared to 21 out of 373 patients (4.3%) with AS (OR 1.07; 95% CI 0.39–2.94). In patients who initiated OAT between days 3 and 7, the primary outcome occurred in 3 out of 103 patients (2.9%) with PS compared to 26 out of 490 patients (5.3%) with AS (OR 0.54; 95% CI 0.16–1.80).
Discussion
our findings suggest that, when deciding the time to initiate oral anticoagulation, the location of stroke, either anterior or posterior, does not predict the risk of outcome events.
Conclusions
Patients with PS or AS and AF appear to have similar risks of ischaemic or haemorrhagic events at 90 days with no difference concerning the timing of initiation of OAT.
Introduction
Anticoagulant therapy has been proven to be highly effective for secondary stroke prevention in patients with atrial fibrillation (AF). 1 Deciding when to initiate this therapy in patients with acute ischaemic stroke and AF is a challenge because the risks of both early recurrent ischaemic stroke and haemorrhagic transformation (HT) are highest in the days immediately following the acute event. 2 Indeed, there are no definitive data on this issue, and current guidelines do not adequately address when to best start oral anticoagulation with even greater uncertainties in patients with acute posterior circulation stroke. 3 In fact, it has been described that posterior circulation stroke was associated with a higher early risk of subsequent stroke compared to carotid events, while there was a lower risk of HT.4–6
This international prospective multicentre study included patients with acute ischaemic posterior circulation stroke and AF, with the principle aims of evaluating at 90 days from an acute event for (1) the risks of recurrent ischaemic embolic event and severe bleeding (both intra and extracranial) as well as (2) the risks of recurrent ischaemic embolic event and bleeding in relation to the timing of the start of anticoagulant therapy. Moreover, the rates of ischaemic events for patients with anterior or posterior circulation stroke were compared. The data for patients with anterior circulation stroke came from previously published studies. The protocol and the statistical analyses were discussed and approved by all the co-authors before initiating the collect of the data.
Methods
This observational study was performed between October 2018 and June 2019 and included consecutive patients with acute ischaemic posterior circulation stroke and previous or newly diagnosed AF without contraindications to anticoagulation. Overall, 19 Stroke Units across Europe, North America and Asia participated. The study was approved by the local institutional review boards, whenever required.
Posterior circulation stroke was defined as any acute stroke occurring in the territories of the posterior cerebral arteries (deep and/or superficial), brainstem or cerebellum 7 visible on neuroimaging.
On admission, stroke severity was assessed using the National Institutes of Health Stroke Scale (NIHSS) and a cerebral computed tomography examination without contrast or cerebral magnetic resonance imaging (MRI) was performed on all patients to exclude for intracranial haemorrhage. Thrombolysis treatment and/or mechanical thrombectomy were performed as per standard local protocol, in accordance with international guidelines. All participating Stroke Units provided standard Stroke Unit care and monitoring. Physicians were free to decide on any administration of anticoagulant treatment (warfarin, apixaban, edoxaban, dabigatran or rivaroxaban), as well as their timing.
A second brain computed tomography scan or MRI was to be performed 24 to 72 h from stroke onset. HT was defined as any degree of hyperdensity within the area of low attenuation and was classified as either haemorrhagic infarction or parenchymal hematoma.8,9 HT was considered symptomatic whenever it was associated with a decline in neurological status (an increase of 4 points in NIHSS) in the absence of any bleeding evidence on the first computed tomography. 10 The sites and sizes of the qualifying infarcts were determined according to standard templates (7): small, when a lesion was ≤1.5 cm and large, when a lesion was >1.5 cm.
Risk factors and the outcome events of patients with posterior circulation stroke were compared with those of patients with anterior circulation stroke. The latter data came from the RAF and RAF NOACs (Non-vitamin K oral anticoagulants) studies11,12 that were prospective observational studies carried out between January 2012 and March 2014 at 29 Stroke Units and between April 2014 and June 2016 at 35 stroke units respectively, across Europe, United States and Asia. Both the RAF studies enrolled consecutive patients with acute ischaemic stroke and known or newly diagnosed AF without contraindications to anticoagulation. The same inclusion criteria used for RAF and RAF NOACs were used for the present study.
Risk factors
Data on risk factors were collected: age, sex, history of hypertension (blood pressure ≥140/90 mm Hg at least twice before stroke or already under treatment with antihypertensive drugs), history of diabetes mellitus (fasting glucose level ≥126 mg/dL (7 mmol/L) preprandial on two examinations, glucose level ≥200 mg/dL (11.1 mmol/L) postprandial, or HbA1c ≥6.5%, or under antidiabetic treatment), current cigarette smoking, hyperlipidaemia (total cholesterol ≥200 mg/dL (11.1 mmol/L) or triglycerides ≥140 mg/dL (7.8 mmol/L) or already under lipid lowering therapy), history of symptomatic ischaemic heart disease (myocardial infarction, history of angina pectoris, or existence of multiple lesions on thallium heart isotope scan or evidence of coronary disease on coronary angiography), history of symptomatic peripheral arterial disease (intermittent claudication of presumed atherosclerotic origin; or ankle/arm systolic blood pressure ratio <0.85 in either leg at rest; or history of intermittent claudication with previous leg amputation, reconstructive surgery or angioplasty), alcohol abuse (≥300 g per week), obesity (body mass index ≥30 kg/m2) or previous stroke/transient ischaemic attack (TIA). White matter changes (leukoaraiosis defined on the first computed tomography examination as ill-defined and moderately hypodense areas of ≥5 mm according to published criteria) were investigated. 13 Leukoaraiosis in the deep white matter was dichotomised into absent versus mild, moderate or severe. Other baseline variables obtained at admission for all patients included fasting serum glucose, fasting serum cholesterol (total, high-density lipoprotein and low-density lipoprotein), platelet count, International Normalised Ratio, activated partial thromboplastin time, systolic blood pressure and diastolic blood pressure. Data on the use of any antiplatelet, anticoagulants or revascularisation therapy (thrombolytic agents and/or mechanical thrombectomy), before admission, at baseline and during the follow-up period, were recorded. The CHA2DS2-VASc score (2 points for a history of stroke or age ≥75 years and 1 point each for age 65–74 years, a history of hypertension, diabetes, cardiac failure, female sex and vascular disease) before the index event was also calculated. 14
AF was categorised as paroxysmal (episodes terminating spontaneously within 7 days), persistent (episodes lasting more than 7 days or requiring pharmacological and/or electrical cardioversion) and permanent (persisting for more than 1 year, either because cardioversion failed or was not pursued.) 15 For the purpose of the present study, AF was categorised into two types: paroxysmal AF or sustained (persistent or permanent) AF.
Evaluation of outcome
Patients were followed-up prospectively by face-to-face, telephone interviews or a review of available medical charts whenever a patient had died during follow-up. Whenever an outcome event occurred, patients were requested to consign any related documentation during a face-to-face visit. Study outcomes were (1) recurrent ischaemic cerebrovascular events (stroke or TIA) and symptomatic systemic embolisms;(2) symptomatic cerebral bleedings (intracerebral haemorrhage) and major extracerebral bleeding at 90 days. The primary study outcome was the composite of stroke, TIA, symptomatic systemic embolism, symptomatic cerebral bleeding and major extracerebral bleeding. Disability and mortality at 90 days were also assessed using the modified Rankin Scale score (mRS). Functional outcome was defined as either non-disabling (mRS 0–2) or disabling (mRS 3–5).
Stroke was defined as the sudden onset of a new focal neurological deficit of vascular origin in a site consistent with the territory of a major cerebral artery and categorised as ischaemic or haemorrhagic. HTs found on neuroimaging 24–72 h after onset were not considered outcome events, unless they were symptomatic. TIA was defined as a transient episode of neurological dysfunction caused by focal brain ischaemia without acute infarction. Systemic embolism was defined as an acute vascular occlusion of an extremity or organ, confirmed by imaging, surgery or autopsy. Major extra-cerebral bleeding was defined as a reduction in the haemoglobin level of at least 2 g per decilitre, requiring blood transfusion of at least 2 U, or symptomatic bleeding in a critical area or organ. 16
Statistical analysis
Patient characteristics are presented as mean and standard deviations for continuous variables and as absolute numbers and percentages for categorical variables. Differences in the characteristics of patients with posterior or anterior circulation stroke were calculated using Chi-square test for nominal variables and ANOVA for continuous variables. Furthermore, differences in the characteristics of patients with or without outcome event at 90 days were calculated using Chi-square test for nominal variables whereas ANOVA was used for continuous variables. A multivariable analysis was performed using logistic regression to seek independent predictors of outcome events. The variables included in the model were age, sex, vascular risk factors, type of anticoagulant, leukoaraiosis, lesion size, type of AF and pacemaker.
Given that data relevant to anterior and posterior circulation strokes were collected over different time periods, a multivariable analysis that adjusted for these time period differences was also performed. For this analysis, the variable ‘time period of data collection’ was classified as 0 for the period 2014–2016 and 1 for the period 2018–2019, and then included in the multivariable model. Moreover, a multivariable model adjusted for any possible effects associated with the different times of initiating the oral anticoagulants was utilised.
Survival function and empirical cumulative hazards function were estimated using Kaplan–Meier estimators for the investigated subgroups of patients; the differences between survival functions were tested using the Log rank statistic (or Mantel–Haenszel test) that in the case of large samples has an asymptotic Chi-square distribution. 17 Patients were censored at the time of an outcome event, death or if they were lost to follow-up. In order to best estimate associations, we used odds ratios (OR) for multivariable logistic models; a two-sided p-value < 0.05 was considered significant. All statistical analyses were performed using the IBM SPSS Statistics version 25.0 (IBM Corporation, Somers, NY, USA).
The study fulfilled 20/22 items listed in the STROBE for observational studies. 18
Results
A total of 473 patients with posterior circulation stroke were prospectively included; the data of these patients were compared with the data of 1997 patients with anterior circulation stroke taken from the RAF (969) and RAF NOACs (1028) studies. This population of 2470 patients (1306 females, 52.9%) were available for the analysis.
The baseline characteristics of the patients are listed in Table 1. Posterior circulation stroke patients were more likely to have the following: lower baseline NIHSS score, diabetes mellitus, hypertension, hyperlipidaemia, paroxysmal AF, congestive heart failure, implanted pacemaker, history of myocardial infarction and peripheral arterial disease, CHA2DS2-VASc > 4 and large lesion size. Moreover, patients with posterior circulation stroke were also more likely to be on treatment with direct oral anticoagulant after the index stroke; 22 patients with posterior circulation stroke had stenosis ≥50% or occlusion of the basilar artery.
Baseline characteristics stratified by stroke location.
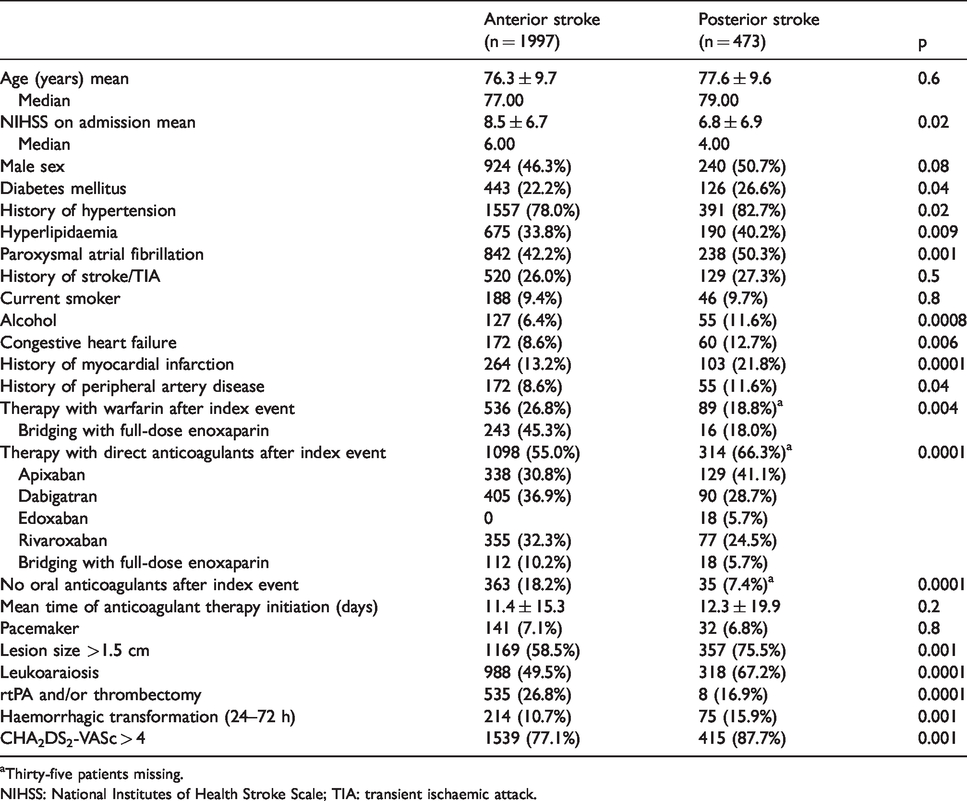
aThirty-five patients missing.
NIHSS: National Institutes of Health Stroke Scale; TIA: transient ischaemic attack.
Outcome events
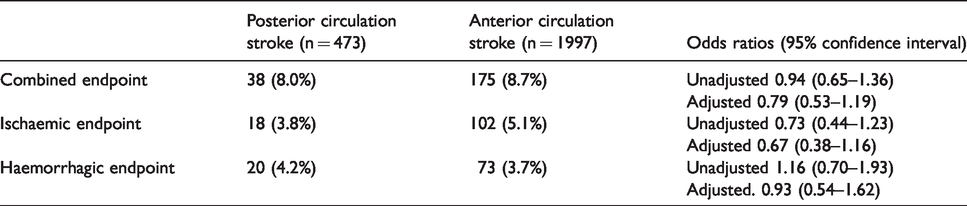
Over 90 days of follow-up, 213 (8.6%) outcome events were recorded: 175/1997 (8.7%) in patients with anterior circulation stroke and 38/473 (8.0%) in patients with posterior circulation stroke. In Table 2 the outcome events are summarised according to anterior or posterior circulation. There were no significant differences between the two groups regarding all types of outcome events (combined, ischaemic or haemorrhagic). Considering the entire population, on multivariable analysis, diabetes mellitus (OR 1.66; 95% CI 1.20–2.31), pacemaker (OR 1.73; 95% CI 1.06–2.82), large lesion size (OR 1.81; 95% CI 1.26–2.63) and treatment with warfarin versus NOACs (OR 2.76; 95% CI 1.77–4.29) were associated with combined outcome events, while diabetes mellitus (OR 1.60; 95% CI 1.06–2.44), treatment with warfarin versus NOACs (OR 2.90; 95% CI 1.68–5.03) and NIHSS on admission (OR 1.02 for each point increase; 95% CI 1.00–1.05) were associated with ischaemic outcome events. Furthermore, diabetes mellitus (OR 1.63; 95% CI 1.01–2.62), treatment with warfarin versus NOACs (OR 2.23; 95% CI 1.18–4.23) and large lesion size (OR 2.91; 95% CI 1.61–5.20) were associated with haemorrhagic outcome events. Adjusting for different time period collections, no significant differences were evidenced between anterior and posterior circulation strokes, regarding all types of outcome events: OR 0.79 (95% CI 0.10–6.11) for combined endpoint, OR 1.23 (95% CI 0.16–9.55) for ischaemic endpoint and OR 2.11 (95% CI 0.27–16.53) for haemorrhagic endpoint.
Association of stroke location (anterior vs posterior) with outcome events.
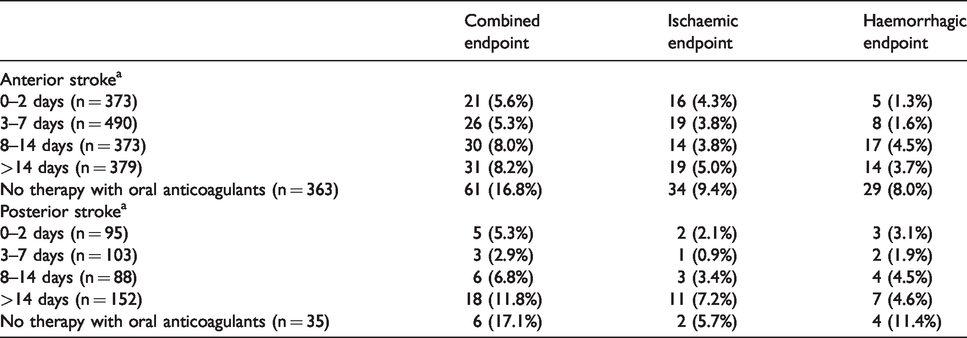
The outcome events according to the time of oral anticoagulant therapy (OAT) initiation in patients with anterior or posterior circulation stroke are reported in both Table 3 and Figure 1: 2 out of 95 patients (2.1%) with posterior circulation stroke who initiated OAT within 2 days from index stroke had an ischaemic endpoint, compared to 16 out of 373 patients (4.3%) with anterior circulation stroke who initiated OAT within the same time period (OR 0.48; 95% CI 0.11–2.12). While 1 of the 103 patients (0.9%) with posterior circulation stroke who initiated OAT between day 3 and 7 from index stroke, had an ischaemic endpoint, compared to 19 out of 490 patients (3.8%) with anterior circulation stroke who initiated OAT within the same time period (OR 0.24; 95% CI 0.03–1.84). Furthermore, 3 out of 95 patients (3.1%) with posterior circulation stroke who initiated OAT within 2 days from index stroke had a haemorrhagic endpoint compared to 5 out of 373 patients (1.3%) with anterior circulation stroke who initiated OAT within the same time period (OR 2.37; 95% CI 0.56–10.11). Whereas 2 of 103 patients (1.9%) with posterior circulation stroke who initiated OAT between days 3 and 7 from index stroke had a haemorrhagic endpoint, compared to 8 out of 490 patients (1.6%) with anterior circulation stroke who initiated OAT within the same time period (OR 1.19; 95% CI 0.25–5.70). Regarding the combined endpoint (ischaemic and haemorrhagic), 5 out of 95 patients (5.3%) with posterior circulation stroke who initiated OAT within 2 days from index stroke had an endpoint, compared to 21 out of 373 patients (4.3%) with anterior circulation stroke who initiated OAT within the same time period (OR 0.93; 95% CI 0.34–2.54). While 3 out of 103 patients (2.9%) with posterior circulation stroke who initiated OAT between days 3 and 7 from index stroke had an endpoint, compared to 26 out of 490 patients (5.3%) with anterior circulation stroke who initiated OAT within the same time period (OR 0.54; 95% CI 0.16–1.80). The risk of any outcome event appeared to be higher after starting anticoagulant therapy >7 days from index stroke for both groups. Adjusting for the different times of oral anticoagulant initiation from index stroke, no significant difference was observed between anterior and posterior circulation strokes regarding combined outcome event (OR 8.31; 95% CI 0.68–100.88). This analysis was not performed for either ischaemic or haemorrhagic outcome events, due to the low number of events in both arms.
Association of outcome events with the timing of oral anticoagulant therapy initiation after index event stratified by stroke location.
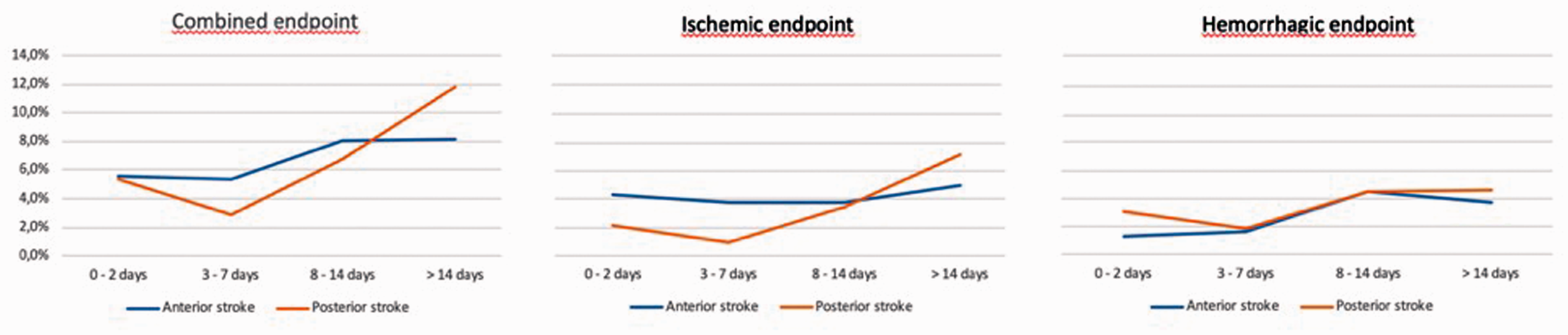
Kaplan–Meier curves of outcome events stratified by timing of initiation of oral anticoagulants in patients with anterior or posterior stroke.
The outcome events in patients with posterior circulation stroke associated with lesion size are reported in Table 4 and Figure 2: 2 out of 116 patients (1.7%) with small lesion had a haemorrhagic outcome event within 90 days, compared to 18 out of 352 (5.0%) of patients with large lesion (Log rank test, p = 0.05).
Association of outcome events with lesion size in patients with posterior circulation stroke.

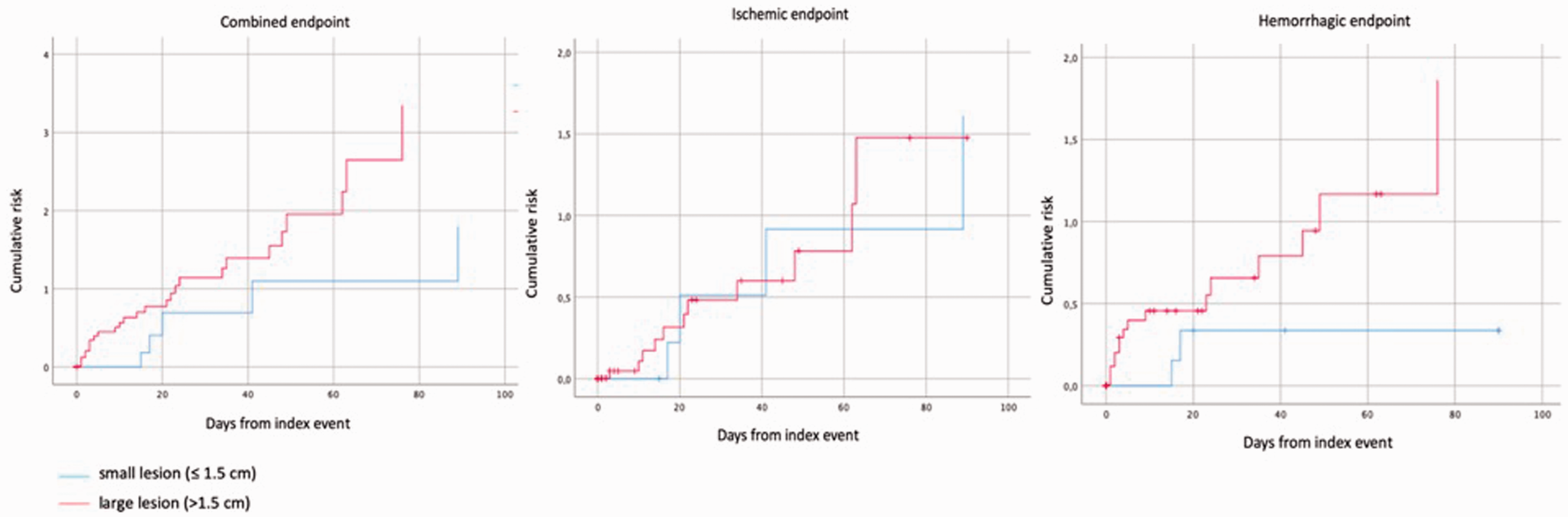
Kaplan–Meier curves of outcome events in patients with posterior stroke stratified by lesion size.
At 90 days, 763 out of 1997 patients (38.2%) with anterior circulation stroke and 187 out of 473 patients (39.5%) with posterior circulation stroke had an mRS > 2 (OR 0.95; 95% CI 0.77–1.16); 123 out of 1997 (6.2%) with anterior circulation stroke and 41 out of 473 patients (8.7%) with posterior circulation stroke died (OR 0.69; 95% CI 0.48–1.00).
Discussion
This study showed that patients with posterior circulation stroke and AF had, at 90 days, an 8.0% risk of combined outcome events that was similar to that of anterior circulation stroke patients (8.7%). The observed risk of ischaemic outcome events, though not statistically significant, was lower in patients with posterior circulation stroke, compared to those with anterior circulation stroke. Moreover, it seems that patients with posterior circulation stroke had a non-statistically significant higher risk for haemorrhagic outcome events, compared to those patients with anterior circulation stroke. With regard to the timing of initiation of oral anticoagulant treatment, the combined outcome event risk in patients with posterior circulation stroke, who had initiated the therapy in the first 7 days, was 8.2%, compared to 10.9% in patients with anterior circulation stroke. Our findings suggest that, when deciding the time to initiate oral anticoagulation, the location of stroke, either anterior or posterior, does not predict the risk of outcome events. Also, the functional final outcome at 90 days using mRS was similar between the two groups, while the NIHSS revealed a less severe stroke syndrome in posterior circulation stroke patients compared to anterior circulation stroke patients. This finding can be attributed to the fact that NIHSS includes more symptoms of the anterior than the posterior circulation; patients with posterior circulation stroke have a lower score despite similar stroke severity. 19
In patients with posterior circulation stroke and AF, the risk of intracerebral bleeding in patients with small lesion was low, suggesting an earlier initiation of OAT. Several randomised trials are ongoing to further investigate this specific research question. 2
Several studies have reported that patients with posterior and anterior circulation stroke did not differ in terms of the prevalence of vascular risk factors.20–22 Other studies have reported differences in aetiology between the two groups.19,23–26 In our study, patients with posterior circulation stroke appeared to have a higher prevalence of risk factors. Of note, all of the above studies included patients with anterior or posterior circulation stroke without considering aetiology while our study included only patients with acute stroke and AF.
Our study has several limitations. First, due to the non-randomised nature of the study, our results may be influenced by several confounders including a selection bias for treatment, even if adjusted statistical models were used in the attempt to control them. Second, a central adjudication of the outcome events was not performed but rather, these events were assessed by the local investigators. Third, in observational studies it is not possible to adjust for physician biases associated with prescription of oral anticoagulant timings and agents. Another potentially important factor is the difference in time period between posterior strokes and anterior strokes; the former being recent and the latter being from previously published studies with enrolment before 2014 (the RAF and RAF-DOAC studies). There could therefore be important secular trends accounting for different event rates (e.g. changes in NOAC use), improved general stroke care (e.g. possibly less bridging) or different OAT timing. In fact, regarding changes in oral anticoagulant type use, in the posterior stroke population, which was more recently collected, 66% of the patients were treated with NOACs compared to 55% of the patients in the anterior stroke population. Conversely, 27% of the patients with anterior stroke were treated with warfarin, compared to 19% of the patients in the posterior stroke population.
Furthermore, in the posterior circulation stroke group, compared to the anterior circulation stroke group, a lower number of patients were treated with bridging therapy with full dose of enoxaparin (18% versus 45%). It is plausible that this disparity was influenced by the fact that, in the RAF studies, patients treated with bridging therapy had a worse outcome, compared to those treated without bridging therapy. 27 These differences might have influenced the results, even if multivariable models were used in the attempt to adjust them.
There are several ongoing randomised controlled trials to overcome the limitations of these observational data, 2 and furthermore, subgroup analyses of these trials are needed to determine how the timing of anticoagulation might interact with ischaemic stroke location.
The strengths of our study include its prospective design, the multi-centre nature and relatively large sample size.
In conclusion, we observed that patients with posterior or anterior stroke and AF had similar risks of ischaemic and haemorrhagic events at 90 days. The timing of oral anticoagulant initiation did not appear to influence the association between stroke location and outcome events.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Maurizio Paciaroni received honoraria as a member of the speaker bureau of Aspen, Sanofi-Aventis, Boehringer Ingelheim, Bayer, Bristol Meyer Squibb, Daiiki Sankyo and Pfizer.
Giancarlo Agnelli received honoraria as a member of the speaker bureau of Boehringer Ingelheim and Bayer.
Valeria Caso received honoraria as a member of the speaker bureau and as consultant or advisory board of Boehringer Ingelheim.
Jesse Dawson has received speaker fees from Boehringer Ingelheim, Bayer, Bristol Meyer Squibb, Daiiki Sankyo and Pfizer.
Jukka Putaala received honoraria for lectures related to atrial fibrillation and anticoagulants from Orion Pharma, Bristol Meyer Squibb, Pfizer, Bayer and Boehringer Ingelheim.
Turgut Tatlisumak received honoraria as consultant or advisory relationship from Bayer, Boehringer Ingelheim, Bristol Meyer Squibb, Lumosa Pharm and Portola Pharma.
Kennedy Lees reports fees and expenses for data monitoring committee work and lectures from Boehringer Ingelheim.
Walter Ageno has received speaker’s honoraria from, and participated in scientific advisory boards for, Boehringer Ingelheim, Bayer, Bristol-Myers Squibb/Pfizer, and Daiichi Sankyo, and has received research support from Bayer and Boehringer Ingelheim.
Danilo Toni received speaker’s honoraria and participated in scientific advisory boards for Abbott, Bayer, Boehringer Ingelheim, Daiichi Sankyo, Medtronic and Pfizer.
George Ntaios reports speaker fees/advisory board/research support from Bayer, Pfizer, Boehringer Ingelheim and Elpen. All fees are paid to his institution.
Simona Sacco has received honoraria as speaker or as member of advisory boards from Abbott, Allergan, Eli Lilly, Novartis and Teva.
Peter Vanacker received honoraria as a member of the speaker bureau and as advisory board of Boehringer Ingelheim, Bristol Meyer Squibb, Daiichi Sankyo, Medtronic, EG and Pfizer.
The other authors have nothing to disclose.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
The study was approved by the local institutional review boards.
Informed consent
Written or verbal informed consent was obteined from the patients for their anonymised information to be published in this article.
Guarantor
MP.
Contributorship
MP researched literature, conceived the study, were responsible for data analysis and wrote the first draft of the manuscript. All the authors were involved in protocol development, gaining ethical approval and patient recruitment. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
