Abstract
Aims
The bleeding risk of current antithrombotic strategies in clinical settings, including recently developed agents, needs to be clarified.
Methods and Design
In an investigator-initiated, prospective, multicentre, observational study, patients with cerebrovascular or cardiovascular diseases who were taking oral antiplatelet or anticoagulant agents were enrolled. Compulsory multimodal magnetic resonance images were acquired at baseline to assess cerebral small vessel disease. Six-month follow-up will be performed for two years. The primary outcome is major bleeding as defined by the International Society on Thrombosis and Hemostasis.
Results
Between October 2016 and March 2019, 5306 patients (71.7 ± 11.2 years old, 1762 women) were enrolled. Previous intracranial haemorrhage was documented in 181 patients (3.4%), cerebrovascular disease (including asymptomatic) requiring antithrombotic therapy in 5006 patients (94.3%), and atrial fibrillation in 1061 patients (20.0%). At entry, 3726 patients (70.2%) were taking antiplatelet agents alone, including 551 (10.4%) using dual antiplatelet agents, 1317 (24.8%) taking anticoagulants alone, and the remaining 263 (5.0%) taking both. The leading antiplatelet agent was clopidogrel (2014 patients), and the leading combination of dual antiplatelet medication was clopidogrel plus aspirin (362). Use of direct oral anticoagulants (1029 patients, 19.4%) exceeded warfarin use (554, 10.4%). The number of pivotal bleeding events exceeded 200 in April 2020.
Conclusions
This study is expected to provide the incidence of bleeding complications of recent oral antithrombotics in clinical practice and identify their associations with underlying small vessel disease and other biomarkers. Novel risk stratification models for bleeding risk will be able to be created based on the study results.
Keywords
Introduction
As oral antithrombotic agents have been increasingly used for the prevention of stroke and cardiovascular diseases, bleeding complications including intracranial haemorrhage (ICH) have become serious. Antithrombotic-related ICH reportedly accounts for more than 30% of overall ICHs potentially resulting in severe outcomes.1–3 We performed the multicentre observational Bleeding with Antithrombotic Therapy (BAT) study a dozen years ago and elucidated the increased risk of bleeding with dual antithrombotic use and the association of high blood pressure levels in the outpatient clinic with later ICH occurrence.
1
Of several biomarkers, cerebral small vessel disease (SVD) visible on magnetic resonance imaging (MRI) seems to be associated with future intracerebral haemorrhage. 16 In particular, cerebral microbleeds are known predictors of intracerebral haemorrhage, as well as of ischaemic stroke, especially in stroke patients on antithrombotic therapy.17–19 A total SVD score combining individual MRI features of SVD, including lacunes, cerebral microbleeds, perivascular spaces, and white matter hyperintensities, better describes the impact of SVD on the brain than do the individual features separately. 20 Other pathologies, such as chronic kidney disease, seem to be also predictive of bleeding events. 21
This study was designed to determine the incidence and details of bleeding complications in patients with cerebrovascular and cardiovascular diseases treated with oral antithrombotic therapy and to develop bleeding risk prediction models including laboratory and imaging biomarkers using a prospective registry involving institutes across Japan from the Network for Clinical Stroke Trials, a nationwide hospital-based clinical research network. 22
Methods
Design
The BAT2 study is an investigator-initiated, prospective, multicentre, observational study. The study is registered with ClinicalTrials.gov (NCT02889653) and the University Hospital Medical Information Network clinical trial registry in Japan (UMIN 000023669). A total of 52 hospital sites across Japan are participating in the study (see Supplemental Material Table 1). A flowchart of the study design is shown in Figure 1.
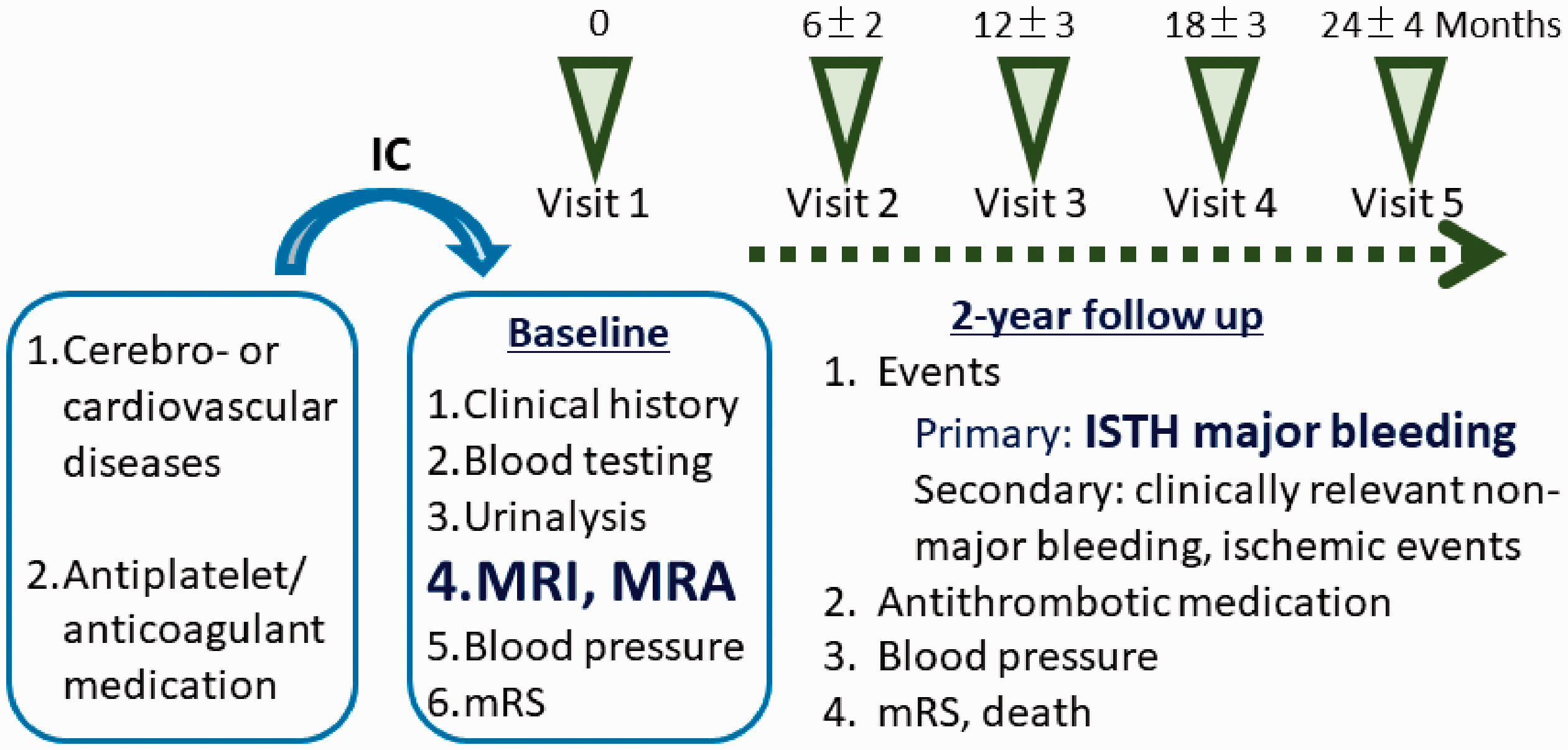
Study design flow chart. mRS: modified Rankin Scale; MRI: magnetic resonance imaging; MRA: magnetic resonance angiography; ISTH: International Society on Thrombosis and Haemostasis.
Participants
Patients with cerebrovascular or cardiovascular diseases (either symptomatic or asymptomatic) who newly started or continued taking oral antiplatelet agents or anticoagulants were enrolled. As typical cerebrovascular or cardiovascular diseases requiring antithrombotic therapy, ischaemic stroke, transient ischaemic attack, steno-occlusive artery disease (either cerebral or systemic), atrial fibrillation, other emboligenic heart disease, coronary artery disease, peripheral artery disease, venous thromboembolism, and post-stent placement were listed in the case report form. However, patients with any other cerebrovascular or cardiovascular diseases were included if each investigator in charge recognised the necessity of antithrombotic therapy. Any single use or combination of antiplatelet agents (aspirin, clopidogrel, cilostazol, ticlopidine, prasugrel, ticagrelor, eicoaspentaenoic acid, limaprost alfadex, and beraprost sodium) and anticoagulants (warfarin, dabigatran, rivaroxaban, apixaban, and edoxaban) that were clinically usable in Japan was eligible. The content and dosage of the agents depended on the choice of each investigator in charge. Inclusion and exclusion criteria are listed in Table 1. The study was approved by the local ethics committee at each participating centre. The investigators obtained written informed consent from patients or their family members before registration.
Criteria for patient registration and definition of endpoints.

MRI: magnetic resonance imaging.
Data acquisition and management
At registration, baseline clinical information and blood test and urinalysis results were collected. The Research Electronic Data Capture system is being used for the collection and management of the data from each participating site through a secured network connection with authentication.
Brain MRI and magnetic resonance angiography (MRA) at 3-T or 1.5-T were mandatory for all patients at registration and allowed to be obtained from 90 days before to 14 days after registration. Sequences of axial MRI, parallel to the anterior commissure-posterior commissure line or the orbitomeatal line, included T1-weighted imaging, T2-weighted imaging, fluid-attenuated inversion recovery image, and T2*-weighted imaging, and the detailed conditions are described in Supplemental Material Table 2.
The anonymised Digital Imaging and Communications in Medicine data of MRI were uploaded through a secure network Medical Imaging Cloud Communication and Knowledge System, which is an integrated diagnosis support platform using a next-generation medical cloud system. All MRI examinations were interpreted by the central diagnostic radiology committee (Supplemental Material Table 3) for cerebral microbleeds, superficial siderosis, white matter hyperintensity, perivascular spaces, lacunes, and other infarctions according to the criteria of STandards for ReportIng Vascular changes on nEuroimaging. 23 Findings on MRA were evaluated at every participating site according to a standardised method for measuring intracranial arterial stenosis. 24 Detailed definitions and baseline findings of MRI abnormalities will be reported separately.
Patients are to be followed up by clinic visit (or by telephone survey for patients with severe physical conditions) every six months for two years, with ischaemic and bleeding events recorded during the follow-up period (Figure 1). Other than the scheduled follow-up, patients are asked to report events to the investigators in charge soon after the occurrence for better data collection regarding events.
Primary and secondary outcomes
The primary outcome is major bleeding as defined by the International Society on Thrombosis and Haemostasis. 25 The secondary outcomes are clinically relevant non-major bleeding and ischaemic events. The definition and additionally collected clinical information for each event are listed in Table 1.
Data monitoring body
Data monitoring is conducted centrally. In some of participating sites, on-site monitoring was performed through site visits.
Sample size calculation
The annual incidence of life-threatening bleeding (including ICH) was 0.98%, that of ICH was 0.53%, and that of major bleeding was 0.88% in the overall participants of BAT.
4
Compared to BAT, BAT2 had some reasons for estimating lower incidences of bleeding, including the reported lower risks of bleeding of recently developed antithrombotics such as DOACs
7
To perform multivariate analysis with about 10 variables, 100 cases of pivotal bleeding, either life-threating or major (estimated combined incidence of 1.9%), would be needed. If the number of patients were set to 6000, the probability to observe 100 events was calculated to be 91.5%, assuming the probability shows Poisson’s distribution.
Statistical analysis
All registered cases will be analysed in this study. Major bleeding as the primary outcome will be evaluated using Kaplan–Meier curves. A risk prediction model of major bleeding will be prepared using Cox regression analysis. 28 Covariates include the type of antithrombotic agent (antiplatelet agents, anticoagulants, or both), MRI results, and clinical biomarkers selected according to previous studies. In order to confirm the robustness of the results, analysis based on propensity scores calculated from the covariates will be carried out. The same method as the analysis of the primary outcome will be used to establish the risk prediction models of secondary outcomes. Two-sided testing will be performed unless otherwise noted, with a significance level of 5%. The confidence interval will be two-sided, with a confidence interval of 95%. In this study, SAS version 9.3 and JMP version 14.0 (all from SAS Institute, Cary, NC) will be used.
Study organisation and funding
The BAT2 was organised by a central coordinating centre located at the National Cerebral and Cardiovascular Center, with funding support from the governmental Japan Agency for Medical Research and Development (18ek0210055h0003, 20lk0201094h0002, and 20lk0201109h0001) and from the Japan Society for the Promotion of Science (Grants-in-Aid for Scientific Research 19K17023).
Results
A total of 5374 patients were registered from October 2016 through March 2019. The follow-up is ongoing through March 2021. To date, the verification of data is proceeding, and 32 patients proved to be ineligible, 3 were duplicate registrations, and the baseline dataset was incomplete in 33. Thus, a total of 5306 patients (71.7 ± 11.2 years old, 1762 women) were eligible. Of these, 4783 patients had a history of ischaemic stroke or transient ischaemic attack in median 72 days (interquartile range 15–1462 days for 4354 patients with available data) after symptom onset and were taking oral antithrombotic agents as secondary stroke prevention; the remaining 523 patients as primary prevention of stroke or cardiovascular disease. Baseline clinical characteristics of these patients are shown in Table 2, and baseline laboratory data are shown in Supplemental Material Table 4. Atrial fibrillation was documented in 1061 patients (20.0%) with the median CHADS2, CHA2DS2-VASc, and HAS-BLED scores of 4, 5, and 4, respectively. Past histories of major or clinically relevant non-major bleeding events were documented in 374 patients (7.0%) including 181 (3.4%) with ICH (Supplemental Material Table 5).
Baseline clinical characteristics.
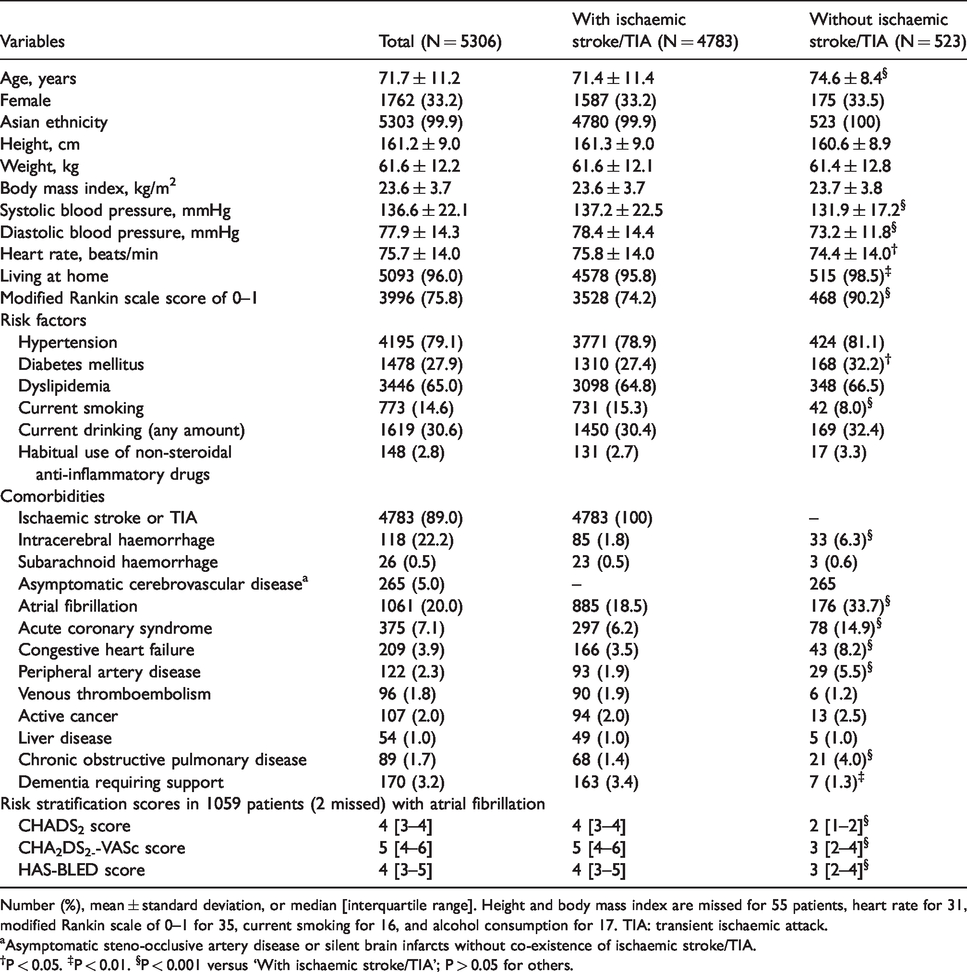
Number (%), mean ± standard deviation, or median [interquartile range]. Height and body mass index are missed for 55 patients, heart rate for 31, modified Rankin scale of 0–1 for 35, current smoking for 16, and alcohol consumption for 17. TIA: transient ischaemic attack.
aAsymptomatic steno-occlusive artery disease or silent brain infarcts without co-existence of ischaemic stroke/TIA.
†P < 0.05. ‡P < 0.01. §P < 0.001 versus ‘With ischaemic stroke/TIA’; P > 0.05 for others.
As oral antithrombotic medication, 3726 patients (70.2%) were taking antiplatelet agents alone including 551 (10.4%) using dual antiplatelet agents, 1317 (24.8%) were taking anticoagulants alone, and the remaining 263 (5.0%) were taking both; 17 patients (0.3%) were taking dual antiplatelet agents plus anticoagulants. Antiplatelet agents alone were more commonly chosen and anticoagulants alone were less commonly chosen (P < 0.001 for both) in patients with a history of ischaemic stroke or transient ischaemic attack than those without (Table 3). Figure 2 shows the types and daily dosages of antiplatelet agents. The leading agent was clopidogrel for 2014 patients (38.0%), followed by aspirin (1689, 31.8%) and cilostazol (784, 14.8%). The leading combination of dual antiplatelet medication was clopidogrel plus aspirin for 362 patients (6.8%), followed by clopidogrel plus cilostazol (105, 2.0%) and aspirin plus cilostazol (64, 1.2%). Clopidogrel was more commonly chosen (P < 0.001) and aspirin was less commonly chosen (P = 0.001) in patients with a history of ischaemic stroke or transient ischaemic attack than those without (Table 3). Figure 3 shows the types and daily dosages of anticoagulant agents. Warfarin was taken by 554 patients (10.4%), for whom the median international normalised ratio (INR) of prothrombin time (PT) was 1.82 at registration (Supplemental Material Table 4), and the most common target range of PT-INR was between 1.6 and 2.6 (336 patients). Data on time in the therapeutic range were available for 503 warfarin users, and it was ≥60% in 355 (70.6%). DOACs were taken by 1029 patients (19.4%). Apixaban was the most commonly used DOAC, for 437 patients (8.2%). DOACs were less commonly chosen (P < 0.001) in patients with a history of ischaemic stroke or transient ischaemic attack than those without (Table 3).
Choice of oral antithrombotic medication.
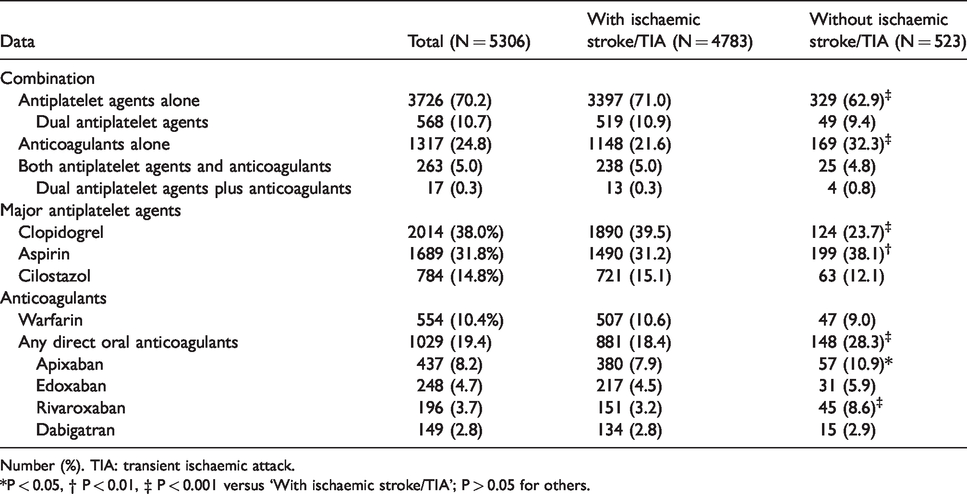
Number (%). TIA: transient ischaemic attack.
*P < 0.05, † P < 0.01, ‡ P < 0.001 versus ‘With ischaemic stroke/TIA’; P > 0.05 for others.
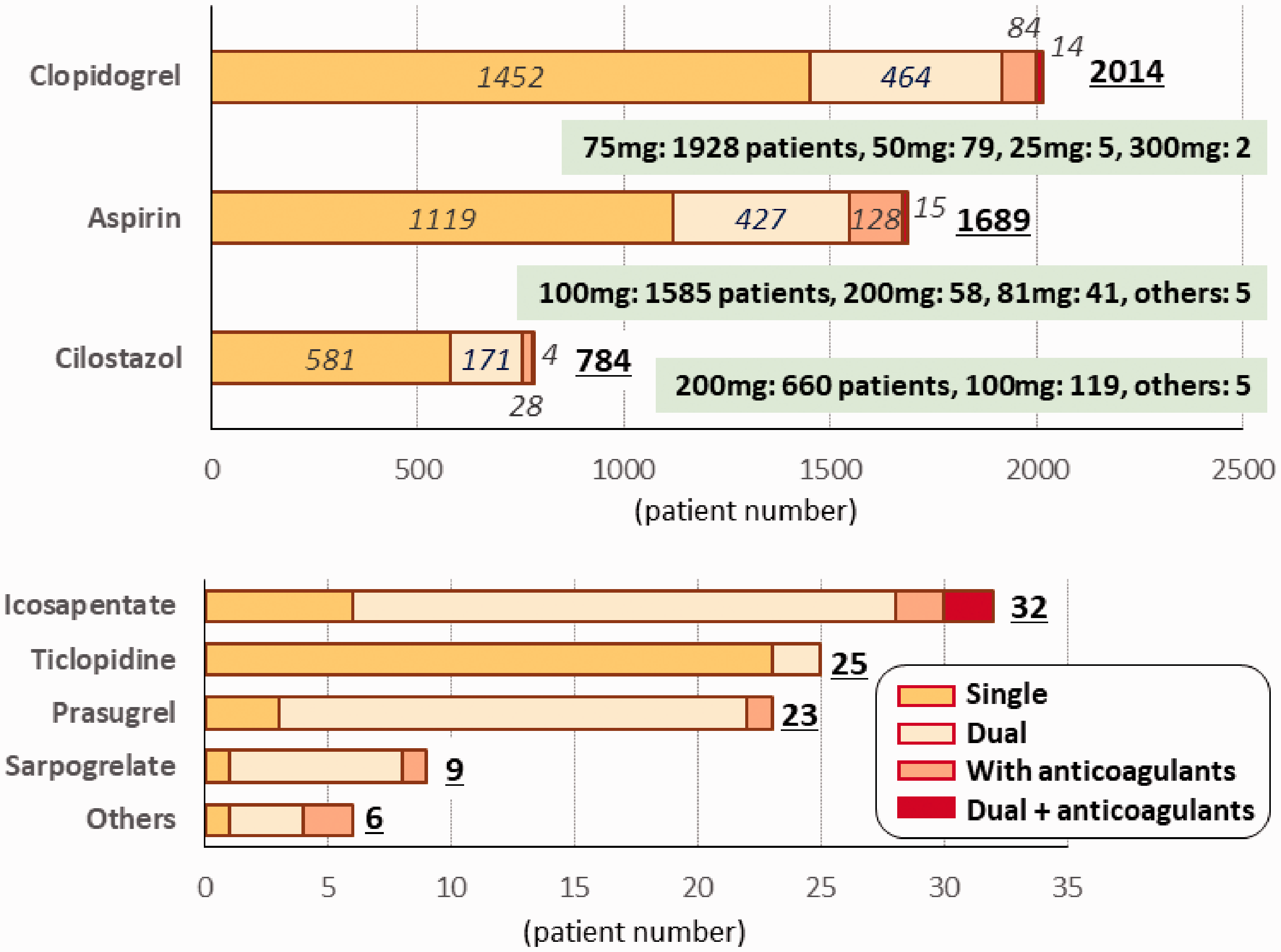
Types and daily dosages of antiplatelet agents.
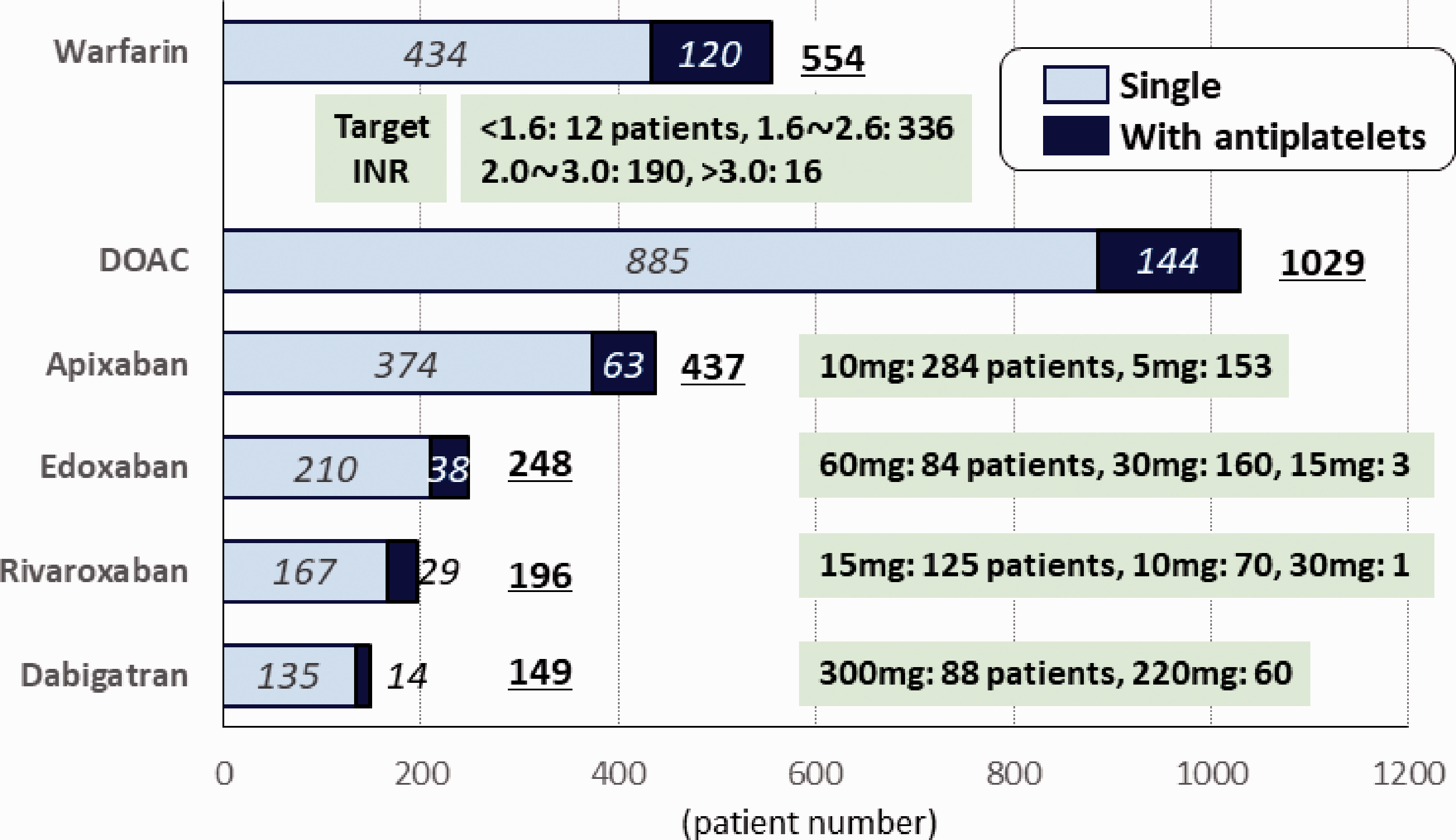
Types and daily dosages of anticoagulant agents. DOAC: direct oral anticoagulant; INR: international normalised ratio.
The number of pivotal bleeding events exceeded 200 in April 2020, round twice as many as expected at the day of expiration of all the follow-up in 2021.
Discussion
Our motivation in planning the previous BAT study was the strong demand for clarifying bleeding risk during antithrombotic therapy for ICH-prone Japanese patients, 4 in whom a lower target range of PT-INR for warfarin is used in elderly patients with non-valvular atrial fibrillation (NVAF), a lower dosage (0.6 mg/kg) of alteplase is used for stroke thrombolysis, and lower dosages of several antithrombotics such as rivaroxaban (ordinarily 15 mg once daily for NVAF) and prasugrel (3.75 mg once daily as the maintenance dose for coronary disease) are used than in the global population, mainly due to concern about bleeding complications. 29 After a dozen years, the mainstream antithrombotics have changed greatly. Clopidogrel and DOACs, as the leading antiplatelet and anticoagulant agents in the present BAT2, were not in clinical use in Japan during the enrolment period of BAT, when aspirin and warfarin were the leading agents.
The baseline characteristics of the present registry respect the above originality of Japanese patients, including the predominant use of cilostazol and the relatively low median PT-INR for warfarin users, according to recommendations based on the results of domestic clinical studies.
13
Emergent reversal of anticoagulant effects affects ICH outcomes. All of the new four-factor prothrombin complex concentrates for warfarin, 33 idarucizumab for dabigatran, 34 and andexanet alfa for factor Xa inhibitors 35 are recently developed strategies that can affect the choice of anticoagulants. Such data will be input in the present registry.
The strength of BAT2 is the mandatory performance of multimodal MRI including the gradient recalled echo T2*-weighted sequence at the baseline assessment. MRI devices are present in almost all stroke centres in Japan. Adding imaging biomarkers to conventional clinical biomarkers would enable us to create risk stratification models for bleeding risk during antithrombotic therapy including the influence of SVD. Another strength is the unforeseen quick accumulation of bleeding events, presumably partly because of a too conservative estimate of the incidence for bleeding; it will strengthen the statistical power.
The study has some limitations. First, patients recently initiating antithrombotic medication and those who had been on it for years were mixed although the risk of bleeding might differ between these two groups. Second, the antiplatelet agents listed in the present registry had somewhat different power of antiplatelet effect and safety one another. The mixture of timing of initiation and contents of agents, the same strategies with BAT, are partly due to compare the present results with those in BAT. However, such mixture may make the analysis of results complicated. We have a plan to analyse the results in separate groups classified by timings after index stroke. We will also analyse the results of each antiplatelet agent if the sufficient number of events for statistical analysis will occur.
BAT2 will provide data regarding bleeding events in this new era of antithrombotic medication.
Supplemental Material
sj-pdf-1-eso-10.1177_2396987320960618 - Supplemental material for The bleeding with antithrombotic therapy study 2: Rationale, design, and baseline characteristics of the participants
Supplemental material, sj-pdf-1-eso-10.1177_2396987320960618 for The bleeding with antithrombotic therapy study 2: Rationale, design, and baseline characteristics of the participants by Masahito Takagi, Kanta Tanaka, Kaori Miwa, Makoto Sasaki, Masatoshi Koga, Teruyuki Hirano, Kenji Kamiyama, Yoshiki Yagita, Yoshinari Nagakane, Haruhiko Hoshino, Tadashi Terasaki, Yusuke Yakushiji, Kohsuke Kudo, Masafumi Ihara, Sohei Yoshimura, Yoshitaka Yamaguchi, Masayuki Shiozawa, Kazunori Toyoda and for BAT2 Investigators in European Stroke Journal
Footnotes
Declaration of conflicting interests
All of the following conflicts are outside the submitted work. Toyoda received lecture honoraria from Daiichi-Sankyo, Bayer Yakuhin, Nippon Boehringer Ingelheim, and Bristol-Myers Squibb. Hirano received lecture honoraria from Daiichi-Sankyo, Bayer Yakuhin, Nippon Boehringer Ingelheim, Pfizer, and Bristol-Myers Squibb. Hoshino received lecture honoraria from Daiichi-Sankyo and Pfizer. Koga received lecture honoraria from Bayer Yakuhin. Ihara received lecture honoraria from Daiichi-Sankyo, Eisai, and Bayer Yakuhin and research grant from Bristol-Myers Squibb and Panasonic. The other authors had no conflicts of interest.
Funding
This work was supported by the governmental Japan Agency for Medical Research and Development (18ek0210055h0003, 20lk0201094h0002, 20lk0201109h0001) and the Japan Society for the Promotion of Science (Grants-in-Aid for Scientific Research 19K17023).
Ethical approval
The local ethics committee at each participating centre approved this study (M28-011 in National Cerebral and Cardiovascular Center).
Informed consent
Written informed consent was obtained from patients or their family members before registration.
Guarantor
KT.
Contributorship
Toyoda is the Chief Investigator and designed the trial and secured funding; Koga, Hirano, Ihara, and Yoshimura helped design the assessments, Takagi is the trial manager, responsible for daily running of the trial including regulatory compliance, and drafted the initial manuscript; Tanaka, Miwa, Yamaguchi, and Shiozawa helped the trial management; Tanaka and Miwa provide statistical expertise; Sasaki, Hirano, Yakushiji, and Kudo provide imaging expertise; Hirano, Kamiyama, Yagita, Nagakane, Hoshino, Terasaki, and Yakushiji are Principle Investigators; All authors approved the final version for submission.
Acknowledgements
The authors would like to thank Drs. Shoichiro Sato and Toshimitsu Hamasaki for help in study planning and Ms. Yoshiko Hayashida, Kie Yamaguchi, Satomi Yi, Kieko Fujita, and Kaori Minato for their contribution as research assistants.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
