Abstract
Purpose
Many daily-life clinical decisions in patients with cerebrovascular disease and cognitive impairment are complex. Evidence-based information sustaining these decisions is frequently lacking. The aim of this paper is to propose a practical clinical approach to cognitive impairments in patients with known cerebrovascular disease.
Methods
The document was produced by the Dementia Committee of the European Stroke Organisation (ESO), based on evidence from the literature where available and on the clinical experience of the Committee members. This paper was endorsed by the ESO.
Findings
Many patients with stroke or other cerebrovascular disease have cognitive impairment, but this is often not recognized. With improvement in acute stroke care, and with the ageing of populations, it is expected that more stroke survivors and more patients with cerebrovascular disease will need adequate management of cognitive impairment of vascular etiology. This document was conceived for the use of strokologists and for those clinicians involved in cerebrovascular disease, with specific and practical hints concerning diagnostic tools, cognitive impairment management and decision on some therapeutic options.
Discussion and conclusions: It is essential to consider a possible cognitive deterioration in every patient who experiences a stroke. Neuropsychological evaluation should be adapted to the clinical status. Brain imaging is the most informative biomarker concerning prognosis. Treatment should always include adequate secondary prevention.
Introduction
Vascular risk factors are recognized as one of the main determinants of cognitive impairment associated with ageing.1,2 Cognitive impairment (CI) due to cerebrovascular disease (CVD) can exist after stroke or in the context of chronic CVD without previous stroke, representing a leading concern of patients and caregivers. 3 Although acute stroke care has evolved substantially over the last decades, post-stroke cognitive impairment (PSCI) remains frequently underdiagnosed as it may be overlooked in the presence of other distressing signs (for instance motor or visual symptoms). Consequently, cognitive impact of acute stroke is often underestimated. Moreover, subtle and progressive decline might also be caused by vascular lesions (e.g. either lesions related to small vessel disease (SVD), repetitive minor injuries, or vascular consequences of systemic failure as for instance cardiac insufficiency). Stroke clinicians are well trained in the identification of stroke, but do not always recognize the myriad of cognitive and behavioural symptoms that accompany stroke in the acute and chronic phases.
Methods
This paper is a result of an effort of the ESO Dementia Committee (2018-2020), under the approval of the ESO Executive Committee, aiming to produce some practical clinical suggestions on the identification, diagnosis and management of CI for clinicians involved in the management of patients with stroke. Its use is not only for strokologists, but also for others professionals involved in the management of patients with CI due to vascular pathology. Several comprehensive and updated reviews are available on the topic, acknowledged throughout this paper, and we did not aim to do an exhaustive or systematic review or to cover all current evidence. We tried to incorporate differences of approach and access to ancillary investigations, keeping in mind the standard usual best practice.
Concerning CI in the context of CVD, different terminologies exist,4–6 and consensus is missing, although those terms refer broadly to the same or quite similar entities. In order to be practical, for the purpose of this paper, we will use vascular dementia (VD), 4 major vascular cognitive impairment/disorder 5 and the more recent major vascular neurocognitive disorder (NCD) 6 interchangeably, and where less severely affected, we use mild cognitive impairment/disorder or mild NCD.5,6 Post-stroke dementia (PSD) or post- stroke cognitive impairment (PSCI) will be used when it refers to stroke patients, irrespectively of the time elapsed since stroke.
Findings
How to recognize cognitive complaints/impairment
Cognitive impairment due to CVD can occur in different settings: after a stroke (PSCI), in the acute stage, in the recovery stage (while other stroke symptoms improve), or delayed until months/years after stroke. When CI due to CVD follows repetitive or chronic vascular lesions, identification of those symptoms may be difficult as they might be quite subtle and misleading. Characteristically, and apart from focal cognitive symptoms due to stroke itself (such as aphasia and hemineglect), the initial symptoms may be hard to identify. These symptoms might include reduced initiative for usual tasks, slowness, and higher latency to start an answer/action. Patients may accept undertaking actions if externally motivated and initiated and more time may be needed. Because attention is impaired, patients are easily distracted even by irrelevant stimuli. Multi-tasking can be difficult, not only due to attention shifting difficulties, but also to difficulty in alternating between different tasks and patients may have difficulties in making decisions. Sometimes, behaviour is predominantly affected and proxies/families acknowledge some “personality” changes. Behaviour changes can co-exist or even be the only initial manifestation, such as more inflexible behaviour, with reduced tolerance to changes of routine activities and repetition of the same mistakes (as patients may not be able to correct themselves). Control of inhibition may be disturbed, loss of control of emotional expression, as well as socially inappropriate manifestations (even sexually inappropriate behaviour), although these latter are usually less frequent and occur in more advanced stages. Patients may be labelled as “depressed” although usually do not complain of sadness, and other key aspects of depression are not present. As a result of the symptoms above, patients reduce their level of social interaction, quit usual hobbies and sometimes relatives/caregivers takeover tasks intuitively. The keystone for considering the above symptoms as a manifestation of CVD is that they represent a change from a previous way of functioning, implicate an adaptation in daily-life, and finally, CVD is the presumed etiology. Evolution might be stepwise, progressive, or fluctuant. If only the patient is interviewed, it is possible to miss the picture. Interview of a proxy(ies) may be necessary, but beware of the patient who always looks to the partner to answer and the obliging partner who provides all the responses. Separate interview of the informant/relatives should be considered whenever interview of both patient and relative becomes a sensitive point, as relatives/caregivers might be uncomfortable giving some information or describing some details in the presence of the patient. This separate interview should, nevertheless, follow usual good practice approaches.
Criteria for CI and for CI due to CVD
Several sets of criteria for CI due to CVD have been proposed,4,5 most of them requiring to demonstrate the presence of CI, the presence of cerebral vascular lesions and a relationship between them. The VASCOG criteria 5 have the advantage to define criteria for both mild and major CI (based on the DSM-V) 6 and for both patients with stroke and those without stroke. This is especially important considering studies showing that a large proportion of patients with CI related to a cerebrovascular lesion did not have a clinically-evident stroke. 7
The criterion of CI is operationalized in the diagnostic criteria of CI due to CVD,4,5 although this operationalization still lacks consensus. Diagnosis of mild CI frequently uses the 1.5 standard deviation threshold on cognitive testing following criteria of Winblad et al. 8 two thresholds have been proposed in the DSM-V (1 and 2 standard deviations for mild CI and major CI, respectively). 6 In addition, some teams and studies applied these thresholds to each performance score or to each domain summary scores. Moreover, different results can be due to the chosen operationalized criteria 9 since normative data depend on selection of the controls (volunteers, community or not community, with or without brain imaging), which can, per se, limit interpretation of findings. A strict and explicit harmonization is needed as the use of different procedures deeply influences the interpretation (at least in patients with mild impairment) and the false positive rate.10,11 The use of a global cognitive score summarizing all domains and the fifth percentile threshold has been shown to improve sensitivity while controlling for specificity [i.e. false positive rate]. 10 Whatever the chosen procedure, it is essential to ensure that it provides an optimal sensitivity and controls specificity adequately. In addition, the selection (volunteers vs general population), demographic characteristics (representation of older and low education subjects) and size sample of normative population influence the determination of cognitive test cutoff scores.
The characteristics of vascular lesions in the brain are detailed in the subsection ‘
How to evaluate the neuropsychological status in stroke patients
Regarding stroke patients, we will focus on the post-acute phase, i.e., 3 to 6 months post-stroke. Cognitive assessment at the acute stroke onset should be performed as part of the neurological examination and contributes to the diagnosis of the acute condition in the emergency room; in the stroke unit it usually consists of clinical assessment and screening tests with, when needed, language or hemineglect tests to manage early rehabilitation. 12 More detailed information is already published. 12 Although most post-stroke assessments are now performed within 3-6 months, timing of neuropsychological assessment may influence the profile of CI: marked improvement in speed and attention, frontal executive functions, perceptual and nominal skills can occur over time, compared to stable findings in verbal and visual memory.13,14
We propose that the initial full neuropsychological evaluation should only be conducted after some stabilization was achieved (possibly as late as 6 months after a severe stroke), unless specific cognitive training could be advised earlier (for instance cognitive intervention for neglect). We do not advise to test and re-test repeatedly, unless specific questions arise (search for associated degenerative disease, driven ability or other legal reason, or working difficulties and need for retirement evaluation, for instance). In case re-test is needed for clinical clarification, an ideal interval of 12 months should be considered to avoid learning bias between evaluations.
PSCI is observed in about 50% of stroke survivors, two thirds of them corresponding to mild CI, and one third to major CI according to present CI criteria (see previous section).11,15,16 PSCI has a marked effect on functional prognosis, risk of institutionalization15,17,18 and risk of recurrence of a major vascular event.19,20
Optimal diagnosis of PSCI should be based on comprehensive cognitive assessment in patients at risk of CI. Although this is always a clinical indication, and should, in the end, based on the individual level, some cues can be given: this objective can be achieved using a recently explored strategy based on risk factors of PSCI (Table 1 provided in supplementary material, and “Predictors” section). 21 Several factors have been found to be associated with PSCI, major CI, in particular.10,16,22 A recent study has identified a minimal set of factors for selecting patients at risk of full-spectrum PSCI. 21 The Rankin score represents an important step provided it is graded with a reliable informant, using a structured interview (including difficulties in instrumental activities of daily living). 23 Except in specific situations (e.g. return to a complex occupation), a comprehensive assessment might be considered to be futile in patients having regained all pre-stroke activities without any concerns (i.e., Rankin score=0), and in bedridden patients (i.e., Rankin score=5). In the same vein, comprehensive assessment is usually unnecessary for diagnosis in patients with substantial impairment on screening tests.
Diagnostic evaluation of patients with CI-CVD.
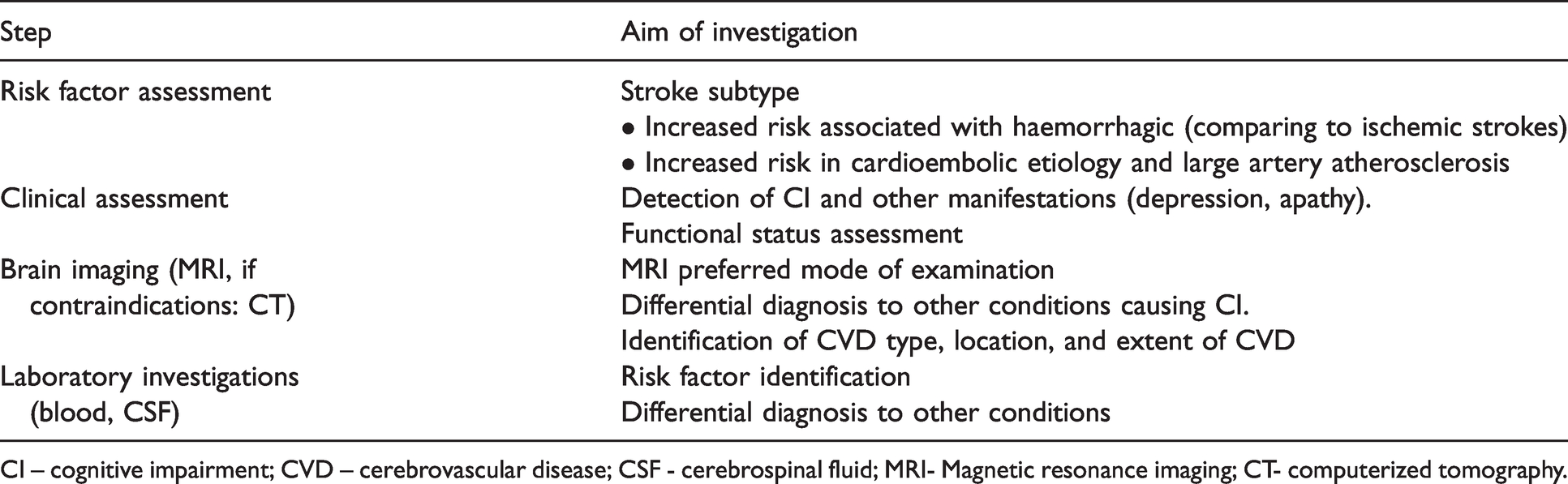
CI – cognitive impairment; CVD – cerebrovascular disease; CSF - cerebrospinal fluid; MRI- Magnetic resonance imaging; CT- computerized tomography.
The administration of a comprehensive neuropsychological battery is the gold standard for the diagnosis of CI but may be complex to perform and not feasible in all stroke survivors and requires suitable quiet and uninterrupted settings. Hence, the first line of cognitive assessment usually relies on clinical examination and screening tests such as Informant Questionnaire on Cognitive Decline in the Elderly (mainly used to identify pre-stroke CI), 24 MiniMental Status Examination (MMSE) 25 and Montréal Cognitive Assessment (MoCA). 26 These instruments are a first step and may identify different severities of CI. We must acknowledge that MMSE and MoCA do not have interchangeable results. MoCA tests included more nonverbal and non-memory items (namely visuospatial/executive functions and attention) compared to MMSE. A recent systematic review indicated good to excellent accuracy, good internal consistency and good reliability of MoCA in differentiating between both mild CI and major CI patients from controls. 27 Nevertheless, despite the mildly higher sensitivity of MoCA as compared to MMSE, 27 a low specificity28,29 still limits its use and both tests have only moderate to good sensitivity for the diagnosis of PSCI.28–31 Thus, both tests underestimate the impairment in a significant proportion of affected patients, i.e. they miss about one fifth of cognitively impaired patients, a proportion which increases in mild PSCI. In addition, their specificity is also lower than 100%,28,30,32 indicating that mildly decreased scores might be observed in subjects with normal comprehensive assessment. Score interpretation needs to take into account the first language, education level (for both tests) and age (for MoCA) 28,33; their scores might also be influenced by sensory-motor deficit, deficits in language and perception (hemi-neglect). Hence, it important to highlight that screening tests scores need always to be integrated in the clinical context and in the whole condition of the patient, in order not to over value results of the screening tests.
Assessment of cognitive abilities is difficult in patients with severe aphasia. In such cases diagnosis of PSCI is usually made on the basis of an aphasia battery and screening test. Further assessment might be necessary to determine the cognitive profile (i.e. associated memory disturbances, executive dysfunction and action slowing). When comprehension abilities allow the use of cognitive tests, further cognitive assessment is usually based in non-verbal tests including visual recognition tests (such as the Doors test, for more details Table 2 in supplementary material), reasoning on visual material (such as Progressive Matrices), visual-motor tests assessing attention and processing speed (such as cancellation test, digit symbol modalities subtest).
Which tests should be used in patients with suspected CI due to CVD?
Considering the profile of vascular CI, a comprehensive test/battery should assess attention, action speed (also called psychomotor speed or processing speed), cognitive and behavioural executive functions, episodic memory, language, and visuo-constructive abilities as well as depressive symptoms. When needed, this first line of tests should be followed by optional tests assessing aphasia, hemi-neglect, agnosia, etc. Cognitive testing should anyway be adapted for the age and sociocultural context, beyond specific stroke deficits. The battery of tests is now standardized owing to the Harmonization Standards protocol battery. 34 This battery has been adapted into multiple languages and cultures and interestingly it provides similar cognitive profiles across countries, which sustains evidence for the robustness and generalizability of the included tests (detailed tests and references provided in Table 2 of supplemental material). Other studies have used neuropsychological assessment, albeit different, that permitted pooled analysis, including the main cognitive domains identified by harmonization standards protocol. 35
Difficulties in activities of daily living (ADL) should be assessed using scales that can distinguish those difficulties due to CI (as needed for a diagnosis of major CI) from those due to sensory-motor deficit and less frequently, to psychiatric disorders,4,5 as physical impairment can be a confounder for diagnosis. 10 As this distinction (critical for the diagnosis of major CI) may be challenging, some studies have used an adaptation of instrumental activities of daily living assessment, with additional questions and examination that identify the mechanism (sensory-motor, cognitive or psychiatric depressive) accounting for the decline of each activity. 10 This poorly investigated area still requires additional validation studies.
Predictors of CI
Several factors have been identified as predictive of future mild or major CI in patients with CVD disease. These factors can be informative for clinicians regarding counselling of patients and relatives as well as selection of patients for more intensive follow-up and for clinical trials.
Neuroimaging predictors of cognitive impairment in small vessel disease
In patients with cerebral SVD (but not necessarily with history of stroke), clinical status and brain magnetic resonance imaging (MRI) aid in predicting cognitive deterioration. While age and initial clinical status (cognitive and functional assessments) already predict future cognitive decline and incident dementia to a large extent, brain MRI has added value. 36 Although volumetric measures, such as total brain volume, white matter volume and hippocampal volume, emerged as the most consistent imaging predictors,37,38 their practical use in non-specialized clinical settings is scarce. Baseline white matter hyperintensities (WMH) and lacunes (cavitated lesions) have also been identified as independent predictors.37,39 More novel markers, such as diffusion (tensor) imaging and structural network analysis, show potential,40,41 but still need further development and simplification to be applicable in clinical routine care.
Neuroimaging predictors of post-stroke cognitive impairment
Specific MRI markers as post-ischemic event predictors have been summarized in a recent review. 42 The most consistent neuroimaging predictors of PSCI, in addition to clinical predictors, were global and medial temporal lobe atrophy.42,43 These data suggest that it might be beneficial to use brain imaging (computerized tomography - CT- or MRI) to identify stroke patients with these atrophy patterns. Volume and location of the infarct (including lacunes) and strategically-located infarcts were also found to be major predictors. 43 Interestingly, data from the large STRIDE study suggests that imaging predictors for PSCI may differ depending on the time point of CI symptom onset. 44 While early PSCI showed the strongest association with infarct features (mostly size and location), delayed PSCI was strongly associated with (pre-existing) SVD on MRI, 45 although these findings await replication in other studies. PSCI risk may differ according to stroke subtype, with an increased risk of CI for cardioembolic etiology and large artery atherosclerosis,46,47 while others reported no differences after adjustment for other factors such as stroke severity and premorbid status, 48 or noted a significant progressive trend of CI among patients with small vessel disease and lacunes up to 5 years after stroke 47 (Table 1).
Pre-stroke brain pathology may contribute to cognitive decline after stroke by increasing the susceptibility to CI. Because of their high prevalence in the elderly, SVD and neurodegenerative pathology, in particular of the Alzheimer‘s disease (AD) type, are the most obvious candidate predictors. An association between pre-existing AD pathology detected by amyloid positron emission tomography (PET) and PSD early after stroke has indeed been shown. 49 However, several studies do not support a prominent role of amyloid pathology in delayed PSCI 50 or PSD,45,51 i.e., CI occurring months to years after stroke.
MRI markers of SVD, such as WMH, lacunes, and cerebral microbleeds should be assessed since these all increase the risk of PSCI. 52 A large comprehensive systematic review and meta-analysis clearly demonstrates a strong association between increasing severity of WMH (on MRI or CT) and several adverse outcomes including subsequent dementia. 52 Nevertheless, this association becomes less strong with aging, when degenerative pathology (AD type) probably superimposed on the impact of WMH. 53 However, many studies did not account for factors such as premorbid cognitive ability or resilience/reserve (discussed below), which may partly account for the apparent ‘looseness’ of the association between WMH burden and cognition. 54 Given these results, the effect of some other predictors of delayed PSCI, such as diabetes, might at least in part be mediated by cerebral SVD, and is potentially modifiable through better risk factor control. The fact that delayed CI occurs months to years after the initial stroke might open a time window for therapeutic interventions, again emphasizing the importance of risk factor treatment after the acute event.
From a practical point of view, infarct volume and location, in combination with WMH, microbleeds and atrophy (globally and medial temporal lobe), may be the most important neuroimaging predictors of PSCI, 51 providing added value on top of clinical variables.
Finally, it should be mentioned that predictors of minor and major CI after ischemic stroke and after intracerebral haemorrhage appear to be largely similar, 22 with haemorrhagic stroke associated with an increased risk of PSCI compared with ischemic stroke.48,55
Clinical predictors
Predictors are of particular interest in the context of PSCI, to identify patients at high-risk for CI promptly identified after the acute event. Multiple studies on PSCI identified predictors related to the concept of brain resilience or reserve. 44 This concept addresses the phenomenon that the same level of brain pathology leads to different levels of CI depending on the premorbid condition of the brain and presumably its ability to actively compensate for the damage. 56 Or. in other words, lower resilience leads to a greater susceptibility for PSCI. Predictive factors attributable to the concept of resilience or reserve are level of education, early-life intelligence (also reflected in type of job), leisure activities, as well as employment and relationship status pre-stroke.10,57–60
The Oxford Vascular Study 22 is at present the largest prospective incidence study for PSD. Stroke severity as measured by the National Health Institutes Stroke Scale (NIHSS) 61 score was one of the strongest predictors of PSD. Other factors were age, previous stroke, recurrent stroke, dysphasia, baseline cognition, low education, pre-morbid dependency, leukoaraiosis - on brain imaging-, and diabetes. The latter is of particular interest for clinicians, since it was the only vascular risk factor associated with PSD. This suggests that intensified risk factor management post-stroke might be most effective in the case of diabetes, or reflect that hypertension and hyperlipidaemia are already now well managed. Recent data from the same study found that APOE e4 homozygosity was associated with PSD, reinforcing the conviction of the influence of a previous neurodegenerative pathology. 62
Still concerning stroke survivors, a combined cognitive risk score based on four easily documented factors (severity of neurological deficit, presence of multiple strokes, multiple deep WMH corresponding to Fazekas score ≥2 and a mild decrease of MMSE score, i.e., adjusted MMSE score from 21 to 27) provided a very good screening strategy 21 but remains to be tested independently and more widely in other cohorts before adoption into practice.
A last word considering age. Although age is an important predictor, PSCI, both acute and delayed, is not infrequent in young stroke survivors, and considering relative risk (although not absolute risk), the dementia risk is greater in younger populations.16,22 Inspite of that, predictors of post-stroke cognitive status in this subpopulation are largely understudied.63,64
Complementary investigations not to be missed
The large clinical and neuroimaging heterogeneity of CI due to CVD explains the difficulty of developing a standardized medical evaluation in the clinical setting for all types of CI due to CVD.65,66
It should go without saying that all patients who are seen in a CVD clinic have a comprehensive evidence-based vascular risk factors assessment67,68 and a work-up for determining the stroke subtype and potential underlying mechanism.69–71 The underlying source of vascular brain damage should be pursued in all CI due to CVD patients 72 in order to prevent subsequent/recurrent strokes.
Clinical assessment of patients with CI due to CVD should include the analysis of typical cognitive changes (described above) but also the recognition of non-cognitive manifestations of CVD such as depression, apathy, motor disability, gait difficulties, balance problems, sensorimotor deficit(s), sphincter control dysfunction, parkinsonism, pseudobulbar palsy and all their possible functional consequences in daily life (Table 1).
While functional outcome in patients surviving acute stroke is well-established, comprising measures of disability (modified Rankin scale score) 73 and functional independence (Barthel Index), 74 other aspects of activities and functional disturbances in daily living are multifaceted, nuanced, difficult to delineate and not well assessed using specific tools.65,66,75–77 Cognitive impairment and executive dysfunction, in particular, as well as depression and apathy, may all have a significant impact on patients’ functional abilities and independence. 78 One practical way to assess this impact is using the interview, with a relative/caregiver. The interview should include aspects mentioned before in “how to recognize cognitive impairment/complaints” such as abandonment of leisure activities, change of habits.
Laboratory analysis in CI due to CVD
No specific laboratory analysis or biomarker in the blood or cerebrospinal fluid (CSF) is available yet for determining the exact vascular injury responsible for CI due to CVD. 67 However, blood laboratory tests can help identify and monitor vascular risk factors.
In patients with SVD, CSF studies may help in differential diagnosis of inflammatory myelin disorders or to exclude vasculitis. 79 CSF protein examination can provide evidence of blood-brain barrier disruption (increased albuminCSF to albuminblood ratio).60,79
Analysis of CSF markers of cortical neuronal degeneration and amyloid pathology may help in detecting mixed etiologies (namely with AD -reduced amyloid β1-42 - also detected in amyloid angiopathy - associated with increased phosphorylated-tau). 79 Other multiple markers are so far of limited value in clinical practice,80–83 such as serum and CSF inflammatory markers, markers of extracellular matrix breakdown (matrix metalloproteinases) or of neuroaxonal damage (serum neurofilament light chain), markers of hypercoagulable state, oxidative stress as well as other metabolic markers (e.g., homocysteine).
Neuroimaging in CI due to CVD
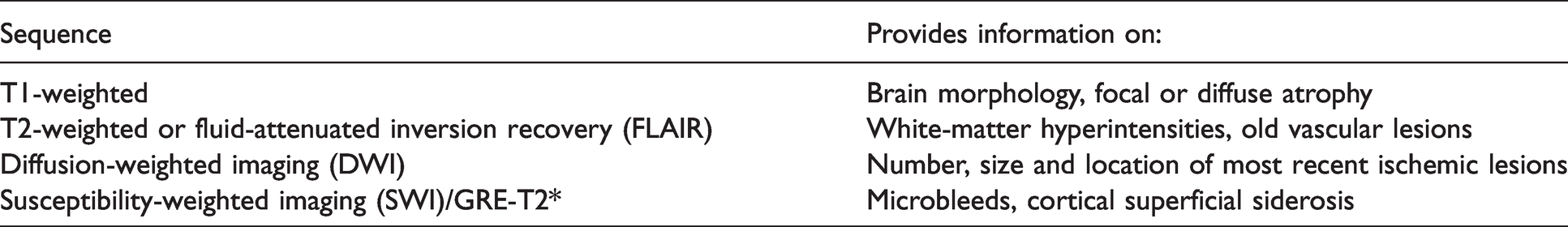
Neuroimaging will have been performed in most patients in the acute setting to assess the stroke subtype, and to plan the secondary prevention strategy at individual level (Table 3 in supplementary material).66,72,84,85 This imaging can also support the evaluation of the likely cause of CI. In this context, the best imaging tool is brain MRI, which can be considered as the gold standard for diagnosis of CI due to CVD, 65 although CT scanning is the most widely available method and provides relevant information on stroke type and pre-stroke brain changes including leukoaraiosis and atrophy. MRI examination should include sequences shown in Table 2.
MRI can also show suggestive patterns of lesions in favor of specific underlying disorders; Cerebral Autosomal Dominant Arteriopathy with Subcortical Infarcts and Leukoencephalopathy (CADASIL) is often associated with temporal pole T2 hyperintensities; cerebral amyloid angiopathy (CAA) often leads to lobar macro- and microbleeds and cortical superficial siderosis.86,87 Diffusion tensor imaging that can probe the microstructure of white matter (even in otherwise normal appearing brain tissue), as well various refined MRI modalities (high-resolution MRI systems, proton NMR spectroscopy and dynamic contrast-enhanced MRI) can provide information about the tissue status but are not used in daily clinical practice.40,79,88 Neuroimaging acquisition, interpretation and reporting of cerebral SVD are now better standardized, and the Standards for ReportIng Vascular changes in nEuroimaging (STRIVE) criteria have been proposed to better define MRI lesions. 89 In patients with MRI contraindications, CT scans can depict atrophy, intracranial haemorrhage, acute and old infarcts, and, to a lesser degree, lacunes and extensive WMH. 67 The use of fluorodeoxyglucose -PET is not helpful for differentiating AD from patients with vascular pathology. 90 In a recent meta-analysis, PET amyloid positivity (a classical feature in presence of CAA or AD) has been reported in elderly APOE ε4 carriers meeting the criteria of VD, and a further increase may be observed in PSD subjects,50,67,91 suggesting a contribution from AD pathology, and a mixed etiology in older patients with PSD.
Integration of diagnostic information and diagnostic labels
Complementary investigations may be needed for the differential diagnosis of MRI-identified lesions (e.g. vascular versus demyelinating lesions in younger patients, or differential diagnosis of white matter lesions at different ages)92,93 or for identifying associated disorders, particularly neurodegenerative conditions that develop with aging. 79 In hereditary forms of CI due to CVD, the patient should be referred to a comprehensive center enabling diagnosis of genetic diseases which can help to reduce unnecessary diagnostic procedures and implement treatment strategies.
Treatment to improve cognition in patients with CI due to CVD
Currently, there is no specifically approved treatment for CI due to CVD. A systematic review of cholinesterase inhibitors (donepezil, rivastigmine, galantamine) and N-methyl-d-aspartate receptor antagonists (memantine) suggested that these drugs improved cognition in CI due to CVD, but did not improve behaviour or functional status.94,95 It should be noted, however, that due to the limitations of inclusion and diagnostic criteria, the vascular origin of cognitive impairment could not be determined in all participants in any of the trials. More dropouts and adverse events (anorexia, nausea, vomiting, diarrhea, and insomnia) occurred with cholinesterase inhibitors compared with memantine. In CADASIL, a pure form of VD, 96 the use of donepezil was also found to improve some executive performances but without improving activities of daily living. 97 Hence, these drugs are not recommended when CI or dementia is of purely vascular origin. However, they can be considered at individual level when the vascular component of dementia is associated with a degenerative disease such as AD, which might be the case in many patients seen in daily practice, particularly older patients.
No significant effect was detected on CI due to CVD using nimodipine, piracetam, huperzine A, cytidine di-phosphocholine and vinpocetine. Other molecules have shown a limited benefit in patients with CI due to CVD (dl-3-n-butylphthalide, gingko biloba extract, cerebrolysin, actogevin).72,98 The results were obtained in small samples or only in subgroups of individuals and were not replicated at large scale. Therefore, we see no evidence to recommend these drugs in patients with CI due to CVD.
In conclusion, the use of cholinesterase inhibitors and memantine might be considered in patients with CI due to CVD only very cautiously and on a case-by-case basis where AD is thought to contribute, depending on the authorization available in the country, the individual tolerance of the treatment and the perceived benefit during follow-up.
Prevention in patients with CI due to CVD
In patients with CI due to CVD or at risk of developing CI of vascular origin, it is obviously crucial to prevent the occurrence of any new stroke event or incident cerebrovascular lesion. The assessment of the underlying CVD and all measures to reduce its progression should be undertaken in all patients.
Control of vascular risk factors and lifestyle changes have limited effects at cognitive level, with exception of hypertension (with suggestions of some benefit from randomized studies), 99 but globally, multi-domain interventions, including non-pharmacologic and lifestyle modifications showed no consistent benefit in cognition in stroke survivors.100–103
Patients with CI due to CVD should be treated as usually recommended after the occurrence of an acute ischemic or haemorrhagic stroke. 104 In patients with a past history of ischemic stroke, there is accumulating evidence suggesting that the number of microbleeds on MRI imaging should no longer be considered as a contra-indication to antithrombotic drugs. 105 Recent data support that in the vast majority of cases, the absolute risk of ischemic events largely exceeds that of haemorrhages. Only the presence of lobar haemorrhage in probable CAA, anticoagulant should be thoroughly discussed dependent on the level of risk of ischemic events.
Particular attention must be paid to patients with CI due to CVD when cognitive deficits are severe, to assess the risk related to therapeutic compliance, including errors or misunderstanding regarding the use of antithrombotic treatments. 106 In some individuals, a caregiver may be needed to control the treatment administration. When in doubt, treatments that expose a high risk of complications might be avoided.
Reperfusion therapies in presence of CI
There is no study examining specifically the potential of thrombolysis or thrombectomy to treat acute ischemic stroke in patients with CI due to CVD. However, the risk of death and haemorrhage is not increased in persons suffering from dementia 107 and there is some evidence that persons with dementia may benefit as do other acute stroke patients from intravenous rt-PA. 108 Therefore, thrombolysis or thrombectomy should be considered in all acute stroke patients including those with CI due to CVD. However, the premorbid level of function, quality of life, social support and life expectancy should be weighted whenever possible before deciding to treat as they can be major determining factors in outcome. 107
Hence, the use of cerebral reperfusion therapies should not be ruled out in patients with CI. Individual decisions of not to treat maybe taken, namely in situations where autonomy is already severely affected and when large lesions cannot be significantly reduced by the treatment.
Discussion and conclusion
Additional investigations are needed to improve the management of cognitive disorders due to cerebrovascular pathology. The development of innovative preventive therapies in stroke patients that can further reduce the risk of vascular brain damage will remain the best guarantee for decreasing the risk of cognitive decline. Any progress in the management of all types of CVD will be essential in this context.
Since the benefit of some specific pharmacologic agents may vary depending on the distribution and severity of cerebral damage, importance of brain and cognitive reserve, but also on age, gender, metabolic or genetic factors, new strategies that could better integrate complex parameters at individual level should be considered in future clinical trials for developing a personalized approach to management.
The potential of various types of neuroprotective agents for reducing cerebral tissue damage in CVD needs further investigations. eHealth interventions for improving prevention, clinical follow-up and treatment will need specific studies. This approach might be also used in the near future to enable innovative numeric rehabilitation and regular counselling via internet platforms.
Supplemental Material
sj-pdf-1-eso-10.1177_23969873211000258 - Supplemental material for Cognitive impairment in patients with cerebrovascular disease: A white paper from the ESO Dementia Committee
Supplemental material, sj-pdf-1-eso-10.1177_23969873211000258 for Cognitive impairment in patients with cerebrovascular disease: A white paper from the ESO Dementia Committee by Ana Verdelho, Joanna Wardlaw, Aleksandra Pavlovic, Leonardo Pantoni, Olivier Godefroy, Marco Duering, Andreas Charidimou, Hugues Chabriat and Geert Jan Biessels in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Olivier Godefroy during the last five years has served on scientific advisory boards and speaker (Biogen, Astra Zeneca, Novartis) and received funding for travel and meetings from Bristol-Myers Squibb, Roche, Biogen, Teva-santé, Boehringer-Ingelheim, Covidien, Ipsen. The remaining authors declare that there are no conflict of interest concerning the paper.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Informed consent for patient information to be published in this article was not obtained because this was an audit.
Guarantor
AV.
Contributorship
All authors contributed equally to conceive the manuscript, the structure of paper, literature research and writing of the manuscript. All authors contributed to the different stages of the manuscript, reviewed and edited and finally approved the final version of the manuscript.
Acknowledgements
We thank Professor Franz Fazekas from the Medical University of Graz, Austria for his contribution on this paper and we thank the European Stroke Organization for supporting and for the endorsing this initiative.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.