Abstract
Introduction. Patient decision aid (PDA) certification standards recommend including the positive and negative features of each option of the decision. This review describes the inclusion of concepts related to overdiagnosis and overtreatment, negative features often ambiguously defined, in cancer screening PDAs. Methods. Our process followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. We reviewed 1) current systematic reviews of decision aids, 2) the Ottawa Hospital Research Institute Decision Aid Library Inventory, and 3) a web-based, gray literature search. Two independent reviewers identified and evaluated PDAs using content analysis. Reviewers coded whether overdiagnosis/overtreatment was described as 1) detecting cancer that would not lead to death, 2) detecting cancer that would not cause symptoms, and/or 3) a potential harm or consequence of screening. Coding discrepancies were resolved through consensus. Results. A total of 904 records (e.g., articles, PDAs) were reviewed and 85 PDAs were identified: prostate (n = 36), breast (n = 26), lung (n = 10), colorectal (n = 10), and other (n = 3). Sixty-seven PDAs included concepts related to overdiagnosis/overtreatment; 57 (67.1%) used a term other than overdiagnosis/overtreatment, 23 (27.1%) used the specific term “overdiagnosis,” and 13 (15.3%) used “overtreatment.” PDAs described overdiagnosis/overtreatment as a potential harm or consequence of screening (n = 62) and/or a detection of a cancer that would not cause symptoms (n = 49). Thirty-six described overdiagnosis as the detection of a cancer that would not result in death. Twenty PDAs described the probabilities associated with overdiagnosis/overtreatment. Conclusions. Over three quarters of cancer screening PDAs addressed concepts related to overdiagnosis/overtreatment, yet terminology was inconsistent and few included probability estimates. Consistent terminology and minimum standards to describe overdiagnosis/overtreatment would help guide the design and certification of cancer screening PDAs.
Patient decision aids (PDAs) are tools designed to support patient engagement in making high-quality, informed health-related decisions.1,2 With features to improve knowledge, clarify values, and prepare patients to participate in the decision-making process, PDAs facilitate decision-making concordance with patient’s informed values. 1 The use of PDAs is particularly instrumental for patients making medical decisions when there is equipoise between the decision options and patient preferences are central for identifying the “best” decision.1,3 PDAs are not designed to replace the patient-physician conversation; rather, they provide tools and strategies to enhance shared decision-making conversations.1,3,4 As the number of PDAs being developed continues to increase, there is a need for a way to ensure the quality of their content.
There has been a push for certification standards of decision aids in the United States to ensure that these tools meet the requirements needed to facilitate quality patient decision making.1,5 The use of PDAs in preference sensitive care was introduced as a priority in The Patient Protection and Affordable Care Act. 6 Additionally, one component of the Centers for Medicare & Medicaid Services (CMS) reimbursement requirement for lung cancer screening with low-dose computed tomography is the documentation of a shared decision making visit with the use of a PDA. 7 Moreover, Washington State has propelled public policy forward by endorsing the use of PDAs that meet their state certification standards. 8
Based largely on standards from the International Patient Decision Aid Standards Collaboration,9,10 the National Quality Forum introduced certification standards recommending the inclusion of both positive and negative features of the options addressed in a PDA, including a description of the “consequences of detection that would not have caused problems if the screen was not done.” 11 For cancer screening decision making, the inclusion of positive and negative features is needed for patients to have realistic expectations for what the test may reveal, especially if there is concern for the negative features of screening, like overdiagnosis.12,13
In many instances, cancer screening is a preference-driven decision due to differing guidelines, insufficient evidence, and patient valuation of the benefits versus the harms.12,14,15 The ideal cancer screening test would identify harmful cancers earlier when curative treatments are more effective; however, among breast, prostate, thyroid, and melanoma cancers, screening efforts have increased the detection and treatment of potentially indolent or insignificant cancers. 16 The identification of indolent cancers raises concerns about overdiagnosis, defined as a diagnosis that does not benefit the screened individual and includes detecting disease that would not cause harm, would not progress, and/or would resolve itself.17–20 Overtreatment is the subsequent treatment of an overdiagnosed condition that does not provide a benefit.17–20
Overdiagnosis and overtreatment are harms of cancer screening that are not frequently discussed between patients and providers.12,13 Wegwarth and Gigerenzer observed that less than 10% of patients reported discussing overdiagnosis or overtreatment with their physician when discussing cancer screening. 13 Overdiagnosis in cancer is a serious concern because it can initiate treatment in a healthy person who would not benefit from the treatment. Additionally, it is a challenging and poorly understood concept to discuss in part because those who are overdiagnosed will likely never know.13,21 Thus, efforts to increase patient understanding of overdiagnosis as a potential downside of cancer screening are needed.
The purpose of this investigation was to examine if and how concepts related to overdiagnosis and overtreatment were included in cancer screening PDAs. Specifically, we identified whether these concepts were described as the detection and/or treatment of a cancer 1) that would not cause death, 2) that would not cause symptoms, and 3) a potential harm or consequence of screening.
Methods
We conducted a systematic review of literature to identify and evaluate cancer screening PDAs that included concepts related to overdiagnosis and overtreatment, and how these concepts were described. Our process for obtaining PDAs included in this review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Figure 1). 22 The search terms are available in Appendix 1.

PRISMA diagram.
Data Source
We used three primary search strategies: 1) reviewing current systematic reviews of decision aids, 2) reviewing decision aids from the source document of the Ottawa Hospital Research Institute (OHRI) Decision Aid Library Inventory, and 3) a web-based, gray literature search.
For the first strategy, a research librarian with expertise in systematic reviews (GP) reviewed the 2014 Cochrane Systematic Review 23 and the 2014 Agency for Healthcare Research and Quality (AHRQ) Systematic Review for Cancer Screening Decision Aids. 24 Both these systematic reviews used highly sensitive research strategies (i.e., 38,000 citations for Cochrane and 16,700 for AHRQ), which informed our approach. We began with the individual database strategies used in the 2014 Cochrane Systematic Review 23 (i.e., Medline, Embase, PsycINFO, CINAHL, and Cochrane) and added a search module of cancer and cancer screening–related terms (e.g., neoplasms, mammography, colonoscopy, PSA [prostate-sensitive antigen]), including all cancers, all years, and both treatment and screening decisions. However, this approach yielded almost 10,000 records (approximately 3,800 Medline records and 5,800 Embase records, all years).
A search module of decision aid–focused and overdiagnosis-related terms was added to the strategies to identify the database records most likely to be relevant to this review (Appendix 1).
Our search strategy built on that of the 2014 Cochrane and 2014 AHRQ database searching efforts by adding to the identification of all cancer screening decision aids that can be found through database searches up to 2012, a time period covered by both reviews. We then initiated and completed identifying all of the records meeting these criteria from 2012 through October 25, 2016. We repeated the search using the updated 2017 Cochrane Systematic Review 1 published on April 12, 2017.
In our second strategy, to ensure inclusion, we reviewed the OHRI source document to identify all decision aids incorporated in the 2017 Cochrane Systematic Review as of March 3, 2017. We also included the records from the 2013 Jimbo and colleagues review of cancer screening decision aids, completing the review of these aids as of July 15, 2018. 25
Third, an online gray literature search was completed on January 25, 2017. In this search, terms (e.g., [“decision aid” OR “screening aid” OR “patient screening aid”] AND [“cancer screening aid” OR “cancer decision aid”]) were used to identify cancer screening patient decision aids. Records were captured through the first 10 pages listed through the Google search. This was an iterative process and any additional decision aids found online during the review process were included in the gray literature search. This search was updated on October 14, 2017, and the final decision aid added to the database for this review was on December 1, 2017.
After completing the search and deduplication process, 904 records of research articles and PDAs were identified. At least one research team member (AJH, LML, ASH, LEJ, ZZ) reviewed each article record to determine if the record: 1) described or included a PDA and 2) described a cancer screening PDA (i.e., identified a specific cancer type, decision aid designed for cancer screening [e.g., not treatment]). If there was a reported cancer screening PDA, research team members (AJH, ZZ) made significant efforts to search for and access the PDAs for evaluation. If the article described a PDA but a copy of the aid was not available, it was excluded from the study. Cancer screening decision aids were then reviewed by a minimum of two research team members (AJH, LML, ASH, LEJ, ZZ) to determine if they met the criteria for a PDA based on the National Quality Forum criteria (Figure 2). 11 PDAs meeting these criteria were included in this review.
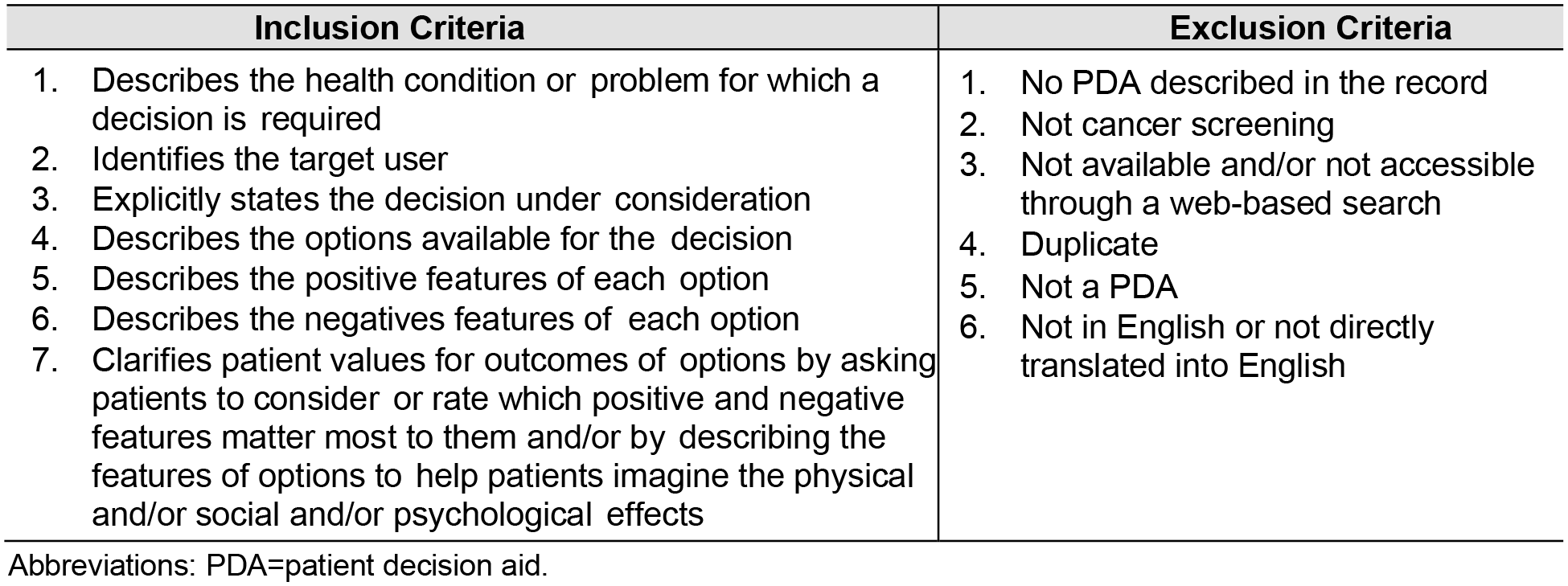
Patient decision aid (PDA) inclusion and exclusion criteria.
Data Collection and Categorization
Reviewers evaluated the decision aids to assess whether they included concepts related to overdiagnosis and overtreatment, and how these concepts were described (Table 1). The definition for overdiagnosis used in this review was from Carter et al., describing overdiagnosis as “an (asymptomatic) person is diagnosed with a condition; that diagnosis does not produce a net benefit for that person.” 17 We interpreted and applied this definition to include descriptions of overdiagnosis and overdetection that may identify those diagnosed with a disease that would not cause harm, would not progress or is indolent and would not cause harm during their lifetime, and/or would resolve spontaneously (Figure 3).18–20 The definition used for overtreatment was, “provision of treatment with no net benefit by individual clinicians to their patients.” 17 We interpreted and applied this definition to include descriptions of overtreatment for those that may have been diagnosed and treated for a disease that would not cause harm, would not progress or is indolent and would not cause harm during their lifetime, and/or would resolve spontaneously.18–20
Presentation of Overdiagnosis and/or Overtreatment in Patient Decision Aids Across Cancer Screening Contexts a

Percentages may exceed 100% as there were multiple response options.
Breast includes patient decision aids with a combination of breast and ovarian cancer (n = 4/26).
Colorectal cancer = colon and rectum.
Other cancer types include cervical (n = 1), thyroid (n = 1), and cancers associated with Lynch syndrome (e.g., colorectal, endometrial, ovarian, stomach, kidney; n = 1).
Percentage calculated of decision aids including overdiagnosis/overtreatment (includes probabilities/number of decision aids including overdiagnosis/overtreatment).
Study data were collected using a questionnaire hosted on Research Electronic Data Capture (REDCap) at The University of Texas MD Anderson Cancer Center. 26 The questionnaire provided definitions for the terms overdiagnosis and overtreatment because these terms are frequently used interchangeably and in an overlapping way.17–20 Specifically, reviewers coded whether 1) overdiagnosis, 2) overtreatment, and/or 3) another term describing concepts of overdiagnosis/overtreatment was used in the PDA, and if it described it as 1) the detection and/or treatment of a cancer that would not cause death, 2) the detection and/or treatment of a cancer that would not cause symptoms, and 3) a potential harm or consequence of screening. If the decision aid included topics related to overdiagnosis, coders recorded if overdiagnosis was mentioned as 1) a key point or take-away message, 2) part of the fact-check section (e.g., knowledge self-test, executive summary), and/or 3) included in the values clarification component of the PDA. When a PDA presented lay terms for concepts related to overdiagnosis and/or overtreatment, reviewers coded these as another term related to overdiagnosis/overtreatment in efforts to avoid conflation or misappropriation of the two terms with specific definitions. Finally, coders identified if the description of overdiagnosis was quantified (e.g., probabilities, frequencies). Any coding discrepancies were resolved through consensus coding.
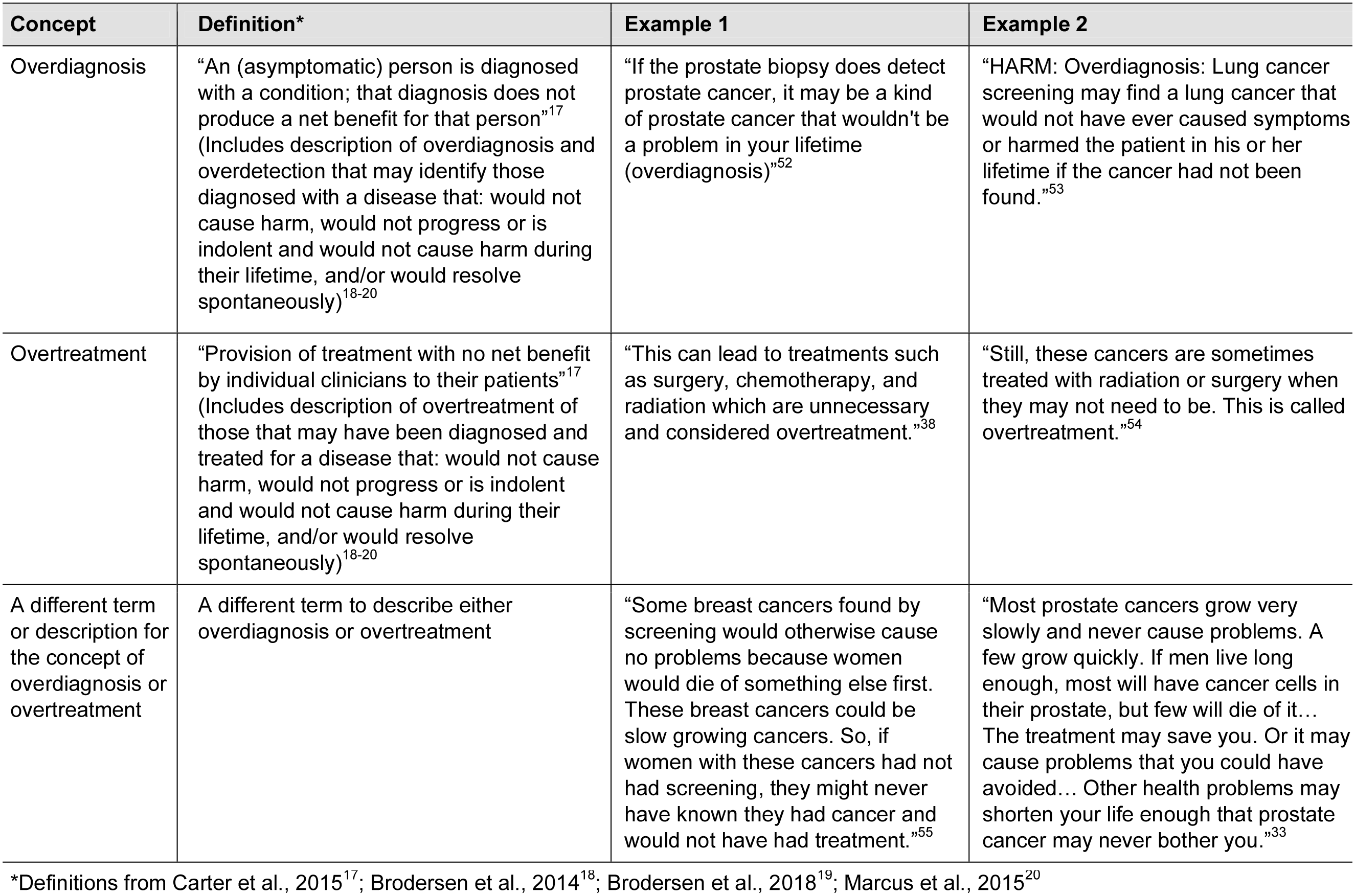
Definitions of overdiagnosis and overtreatment used for data extraction for analysis.
The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report. The content is solely the responsibility of the authors and does not necessarily represent the official views of the funders.
Results
A total of 904 records (e.g., articles, PDAs) were identified, and from these records 85 PDAs were identified and met inclusion criteria. Reasons for exclusion were the following: no PDA described in the record (n = 260), not cancer screening (n = 216), not available and/or not accessible through a web-based search (n = 237), duplicate (n = 40), not a PDA (n = 10), or not in English or not directly translated into English (n = 3). The 85 PDAs that met inclusion criteria focused on cancer screening for the following: prostate (n = 36), breast (n = 26), lung (n = 10), colorectal (n = 10), and other (cervical, n = 1; thyroid, n = 1; and cancers associated with Lynch Syndrome [e.g., colorectal, endometrial, ovarian, stomach, kidney, n = 1]). Appendix 2 is a comprehensive list of PDAs coded for this review.
Frequency of Inclusion of Topics Related to Overdiagnosis
Of 85 PDAs, 67 (78.8%) of PDAs included concepts related to overdiagnosis and/or overtreatment. Fifty-seven (67.1%) used a term other than overdiagnosis/overtreatment, for example, “A high PSA test may find a prostate cancer that is slow-growing and never would have caused you problems” 27 and “. . . unnecessary diagnosis (being diagnosed and treated for cancer that might not be harmful).” 28 Twenty-three (27.1%) PDAs used the specific term “overdiagnosis”: “. . . over-diagnosis is when an abnormality is diagnosed and treated as ‘cancer’ but would never have caused symptoms or death if it were never detected by a screening test,” 29 and “What does overdiagnosis mean? This means the diagnosis of breast cancer that, in hindsight, would not have caused harm.” 30 Thirteen (15.3%) PDAs used the exact term “overtreatment;” for example, “. . . treatment for a slow-growing lung cancer that would not have caused problems even if you never got treatment (sometimes called ‘overdiagnosis’ or ‘overtreatment’).” 31
All 10 (100.0%) of the lung cancer screening PDAs and 34 of 36 (94.4%) prostate cancer screening PDAs included topics related to overdiagnosis and/or overtreatment. In contrast, 6 out of 10 (60.0%) colorectal cancer screening PDAs and 16 (61.5%) breast cancer screening PDAs included topics related to overdiagnosis. For the PDAs that include both breast and ovarian cancer screening (n = 4/26), one of the four (25%) PDAs included topics related to overdiagnosis.
Description of Topics Related to Overdiagnosis in PDAs
Of 85 PDAs, 62 (72.9%) described overdiagnosis/overtreatment as a potential harm or consequence of screening. For example, “Pap tests can have risk: The test may show something that does not look normal but would go away on its own. Abnormal results cause anxiety. And they can lead to repeat Pap tests and follow-up treatment that you may not need.” 32 Forty-nine (57.6%) described overdiagnosis/overtreatment as the detection of a cancer that would not cause symptoms, such as, “. . . the older you are, the less likely prostate cancer will cause problems in your lifetime . . . other health problems may shorten your life enough that prostate cancer may never bother you.” 33 Thirty-six (42.4%) PDAs described overdiagnosis/overtreatment as the detection of a cancer that would not result in death, for instance, “Limitations of Regular Mammograms: some breast cancers detected may not be life threatening, but may still lead to invasive treatment.” 34 and “. . . in some cases, prostate cancer never causes problems or threaten life.” 35 Appendix 3 includes abridged language from PDAs that incorporated these three concepts (i.e., detection/treatment of a cancer that would not cause death, detection/treatment of a cancer that would not cause symptoms, a potential harm or consequence of screening) and associated probabilities in the description of overdiagnosis/overtreatment.
Topics related to overdiagnosis were covered in various sections of PDAs. Twenty-two of 85 (25.9%) PDAs covered overdiagnosis/overtreatment in the values clarification section of the PDA. Additionally, 17 of 85 (20.0%) addressed overdiagnosis/overtreatment as a key point or take-away message, and three (3.5%) covered these topics in a fact-check section.
Probabilities
Of the 67 PDAs that included overdiagnosis/overtreatment, 20 (29.9%) of PDAs gave specific probabilities associated with overdiagnosis. For example, However, out of 1,000 people screened yearly for 3 years, around 40 may need a biopsy or surgery. About 17 of those will turn out not to have lung cancer. There is a small chance that you could find and treat a lung cancer that would not have become a problem for you.
36
and For women . . . who have breast screening over 20 years: 4 out of 1000 women avoid dying from breast cancer, and 19 out of 1000 women experience over-detection. So that means more women experience over-detection than avoid dying from breast cancer.
37
Of 10 lung cancer screening PDAs, five included probabilities of overdiagnosis/overtreatment, for instance, “Of the lung cancers found by screening, about 1 in 10 would have never harmed you (overdiagnosis).”
38
Nine of the 34 (26.5%) prostate PDAs and 6 of the 16 (37.5%) breast PDAs that included overdiagnosis/overtreatment included probabilities associated with overdiagnosis/overtreatment. Some examples are, “It is estimated that half of men treated for PSA detected prostate cancer would not have had clinical symptoms during their lifetime.”
39
and . . . it was concluded that for the 10,000 women screened from when they are 50 to 70, 129 women would be over-diagnosed . . . [another review] found that for the 2,000 women screened over 10 years, 10 women would have unnecessary treatment. In this analysis, for every life saved, ten women would have treatment which was not necessary . . .
40
Discussion
In this review of cancer screening PDAs, over three quarters of PDAs addressed concepts related to overdiagnosis; however, terminology used to describe overdiagnosis and overtreatment was inconsistent. Topics related to overdiagnosis and overtreatment were described using a variety of terms and presented in different components of the PDA. For example, overdiagnosis and overtreatment were more often described using lay terms rather than using the specific terms. Only 30% of PDAs included probability estimates associated with overdiagnosis/overtreatment.
Several factors may contribute to if and how overdiagnosis and overtreatment are included in a PDA. Challenges associated with the availability and strength of evidence for overdiagnosis and overtreatment may influence whether these topics and associated probabilities are included. Estimates of overdiagnosis resulting from cancer screening range from 0% to 50% of diagnoses depending on cancer type and screening method. 20 Screening for specific cancer types, like prostate (PSA testing),41,42 lung (low-dose computed tomography),43,44 and breast (mammography)45,46 have available evidence and potential risks associated with overdiagnosis; however, the magnitude of risk associated with overdiagnosis remains highly variable.41–46 This magnitude of risk may be of particular concern for cancer types with conflicting recommendations and guidelines regarding initiation and frequency of screening. 18 Communicating overdiagnosis in PDAs may not be as high of a concern among other cancer types that have lower magnitude of risk for overdiagnosis, such as with colorectal cancer screening based on US recommendations and guidelines.47,48 The variability of inclusion of topics related to overdiagnosis may reflect the quality of evidence and magnitude of risk.
How overdiagnosis and overtreatment are described in PDAs may also be a reflection of the underlying variability in use of these complex terms. While overdiagnosis and overtreatment are related, they are not interchangeable. Overdiagnosis often leads to overtreatment, but patients may elect for less invasive interventions after a positive finding (e.g., active surveillance for low-risk prostate cancer). Similarly, overtreatment can occur without overdiagnosis when the treatment intervention provided does not provide effective treatment (e.g., antibiotics for certain infections). 19 Another related term, overdetection, is the detection through a screening method of a cancer that would not have caused harm, and again, may or may not lead to overtreatment. 17 These terms may also confound with descriptions of false-positives, although distinct concepts. 19 Clear descriptions of these key concepts are needed to support effective communication and inform PDA certification criteria.17–20
It is also possible that PDA developers are using alternative terms and phrases to improve understanding across health literacy levels. Evidence shows that there is poor lay understanding of these concepts in the context of cancer screening and descriptions such as “unnecessary diagnosis” or “diagnosis of a condition that would never have caused harm” may be a better method to explain overdiagnosis.37,49 Avoiding the use of the specific terms “overdiagnosis” and “overtreatment” may be purposeful. By describing these concepts rather than using the specific terms, PDA developers may be able to create aids with plain language communication and designed for the cultural context that may assist patients as they work to understand concepts related to overdiagnosis. Public policy may be a contributing factor to the inclusion of these concepts in PDAs, like in the CMS reimbursement requirement for low-dose computed tomography in lung cancer screening. 7 In our review, all (10/10) of the lung cancer screening PDAs included topics related to overdiagnosis. Moreover, 7 of the 10 PDAs were developed and/or updated in 2015 or after. The 2015 CMS coverage determination may have played a role in PDA developers including these topics. Adopting standardized strategies for communication (e.g., plain language, magnitude of risk) and the inclusion of approaches to elicit values and priorities may enable a more functional conversation with regard to discussing overdiagnosis and why one may consider this harm when making a cancer screening decision. 50 An additional consideration is the potential to address skepticism regarding the discussion of the harms, like overdiagnosis, in the context of cancer screening. 48 The perception that services are being withheld by key stakeholders, like insurers, can trigger a strong reaction that can result in push back and distrust in discussions surrounding harms. 48 Thus, the discussion of harms, including overdiagnosis and overtreatment, may merit providing more detailed descriptions of the consequences of screening harms (e.g., financial, quality of life) to ameliorate skepticism that may emerge from discussing harms. In addition, there may be concerns for the populations used in developing cancer screening guidelines and whether certain populations are represented in the data with which the guidelines were based. 50 This may contribute to how to communicate the magnitude of risk associated with overdiagnosis. 50 These considerations may be more critical for those who are older, from racial/ethnic minorities, and those with low socioeconomic status. 48 The trade-offs regarding cancer screening and how to communicate them persist and warrant the development of effective communication strategies to support decision making.18,50 Moreover, there is a strong ethical need to include the potential harms of cancer screening, including overdiagnosis, that can be understood by a broad population. 51
There are some limitations to consider. While a comprehensive search was completed, we were unable to gain access to all PDAs and they were excluded from the review. Some PDAs were still under evaluation and therefore embargoed and unable to be included in our review. We only included PDAs in English or those directly translated into English. Finally, overdiagnosis and overtreatment exist within a context of multiple definitions and consensus lacks for a single definition. Therefore, we interpreted and applied our definition to be inclusive of explicit descriptions of overdiagnosis and overtreatment. An additional potential limitation is the inclusion of only explicit terms, not implicit terms, used to describe topics related to overdiagnosis and overtreatment for coding in our review.
Conclusion
Most PDAs include attention to overdiagnosis and overtreatment. PDAs more often describe these concepts using lay language and do not explicitly state either term. Few PDAs include probability estimates associated with overdiagnosis and/or overtreatment. To guide the design and certification of PDAs, consensus is needed in the terminology and minimum standards used to describe overdiagnosis as a potential downside of cancer screening.
Supplemental Material
Online_supplemental_material_1 – Supplemental material for A Review of the Presentation of Overdiagnosis in Cancer Screening Patient Decision Aids
Supplemental material, Online_supplemental_material_1 for A Review of the Presentation of Overdiagnosis in Cancer Screening Patient Decision Aids by Ashley J. Housten, Lisa M. Lowenstein, Aubri Hoffman, Lianne E. Jacobs, Zineb Zirari, Diana S. Hoover, Dawn Stacey, Greg Pratt, Therese B. Bevers and Robert J. Volk in MDM Policy & Practice
Supplemental Material
Online_supplemental_material_2 – Supplemental material for A Review of the Presentation of Overdiagnosis in Cancer Screening Patient Decision Aids
Supplemental material, Online_supplemental_material_2 for A Review of the Presentation of Overdiagnosis in Cancer Screening Patient Decision Aids by Ashley J. Housten, Lisa M. Lowenstein, Aubri Hoffman, Lianne E. Jacobs, Zineb Zirari, Diana S. Hoover, Dawn Stacey, Greg Pratt, Therese B. Bevers and Robert J. Volk in MDM Policy & Practice
Supplemental Material
Online_supplemental_material_3 – Supplemental material for A Review of the Presentation of Overdiagnosis in Cancer Screening Patient Decision Aids
Supplemental material, Online_supplemental_material_3 for A Review of the Presentation of Overdiagnosis in Cancer Screening Patient Decision Aids by Ashley J. Housten, Lisa M. Lowenstein, Aubri Hoffman, Lianne E. Jacobs, Zineb Zirari, Diana S. Hoover, Dawn Stacey, Greg Pratt, Therese B. Bevers and Robert J. Volk in MDM Policy & Practice
Footnotes
Acknowledgements
We thank Gary M. Deyter, PhD, for his editorial assistance.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The University of Texas MD Anderson Cancer Center Duncan Family Institute for Cancer Prevention and Risk Assessment; The National Cancer Institute at the National Institutes of Health (P30 CA016672 to University of Texas MD Anderson Cancer Center as a Cancer Center Support Grant and to use the Shared Decision Making Core; training under R25T CA57730 [PI: Shine Chang] to AJH); and The National Institute on Minority Health and Health Disparities of the National Institutes of Health (K99 MD011485 to AJH).
The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
The content is solely the responsibility of the authors and does not necessarily represent the official views of the funders.
Authors’ Note
PROSPERO International Prospective Register of Systematic Reviews: CRD42017072653.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
