Abstract
Highlights
Most patient decision aids for prostate cancer screening lacks important information on overdiagnosis.
Specific guidance is needed on how to communicate the risks of overdiagnosis.
Introduction
Cancer screening may be in the form of screening (population based or opportunistic), in which asymptomatic people get tested for cancer, or case finding, in which screening is usually offered as part of a clinical encounter for a different health condition. For prostate cancer, the latter is more common.1,2 Decisions on cancer screening seldom have one option that will be clearly preferred by all individuals. The value that each patient gives to the relevant outcomes (including benefits, harms, and costs) can differ considerably. 3 Shared decision making is a process in which clinicians and patients work together to make a health decision by discussing the options and their benefits and harms and considering the patient’s values, preferences, expectations, and circumstances. 4 It is especially useful to guide decisions about screening, investigations, and treatments, where a preference-sensitive decision is involved. When there is uncertainty as to the preferred option, the benefits and harms of each option need to be carefully discussed in the context of individual preferences and values.5–8
Patient decision aids are tools to support the shared decision-making process. 9 In general, they help in improving knowledge, clarifying personal values and needs, and supporting the patients to make an informed decision on screening. 9 The International Patient Decision Aid Standard Collaboration (IPDAS) recommend that patient decision aids should provide balanced information including harms as well as benefits of screening. This conforms with ethical and legal obligations of informed consent for screening. 10
Overdiagnosis, detecting cancer that will not cause symptoms or death during one’s lifetime, is one well-known harm of prostate cancer screening.11–13 Often, the tradeoff between averting a potential cancer death (benefit) versus the risk of overdiagnosis (harm) is not straightforward; hence, a decision to screen or not to screen should be clearly based on the specific circumstances and personal values and preferences of the patient. 14 It is therefore important that all patients are provided with clear, up-to-date evidence on the risks of overdiagnosis as well as potential mortality benefits, associated with prostate cancer screening. 11 Previous reviews have examined how overdiagnosis is reported in patient decision aids for prostate cancer screening.14,15 However, these did not include a detailed analysis of the information on overdiagnosis provided, including estimates of its frequency, concepts of competing mortality risks, and the existence of a reservoir of indolent cancers, all of which are important contextual factors for communicating prostate cancer overdiagnosis.
In this study, we aimed to address these gaps in knowledge by conducting a comprehensive review of information on overdiagnosis provided in prostate cancer screening decision aids.
Method
All publicly available decision aids on prostate cancer screening, published (or translatable through the publisher’s webpage) in the English language and without date restrictions, were included. A decision aid was defined as a tool that helped men participate in the decision making on whether to undergo prostate cancer screening, provided information on the options available to them, and helped them to clarify and communicate how they personally valued prostate cancer–specific outcomes, including the risks of dying, urinary or bowel symptoms, and erectile dysfunction.16,17
The search included 3 components. First, we included all decision aids from included studies in a recent systematic review co-authored by 2 of us (J.M.R. and K.A.O.T.). 16 This review used a comprehensive search strategy including 5 databases (MEDLINE, Embase, CINAHL, PsychINFO, and Cochrane Central Register of Controlled Trials [CENTRAL]) without language limits. Second, because this systematic review used fairly strict inclusion criteria for decision aids (defined as decision aids if the material helped men making individual choices and included information regarding the association of screening with the following patient-important outcomes: risk of dying, risk of urinary or bowel symptoms, and risk of erectile dysfunction), 16 we also screened the list of excluded studies (eTable 3 of the published systematic review) 16 to identify any further decision aids relevant to the current review. Third, to identify decision aids that were not published as part of a research study, we searched on Google using the search terms (“Decision Support” OR “Decision Aids” OR “Decision Aid”) AND (“Prostate cancer” OR “Prostate cancers” OR “Prostatic cancers” OR “PSA test” OR “PSA testing”). This led to “about 272,000 results” in total (as displayed on the Google search result page). Screening of these results was done by 2 authors (T.P. and K.P.) who independently screened the search results in each web page. This process was repeated until 2 consecutive Google search results web pages with no relevant search results were found (for both authors, this was achieved by screening up to the first 8 web pages of Google search results).
All references were collated using a reference management system (Endnote x9) and duplicates removed. Where decision aids were not provided in a publication (as links or appendices), we attempted to search for these online, and the authors were contacted through email or via online networking channels (LinkedIn, ResearchGate).
The included decision aids were then reviewed by 2 authors (T.P. and K.P.), who independently extracted data on whether and how information on overdiagnosis was reported using a standardized data extraction template (Supplementary Text 2). This included information on whether the word
This study had no external funding source. Ethics approval was not required as this study was based on a review of published decision aids readily available online.
Results
A total of 80 records on prostate cancer screening were collectively identified via the search undertaken in the previous systematic review (
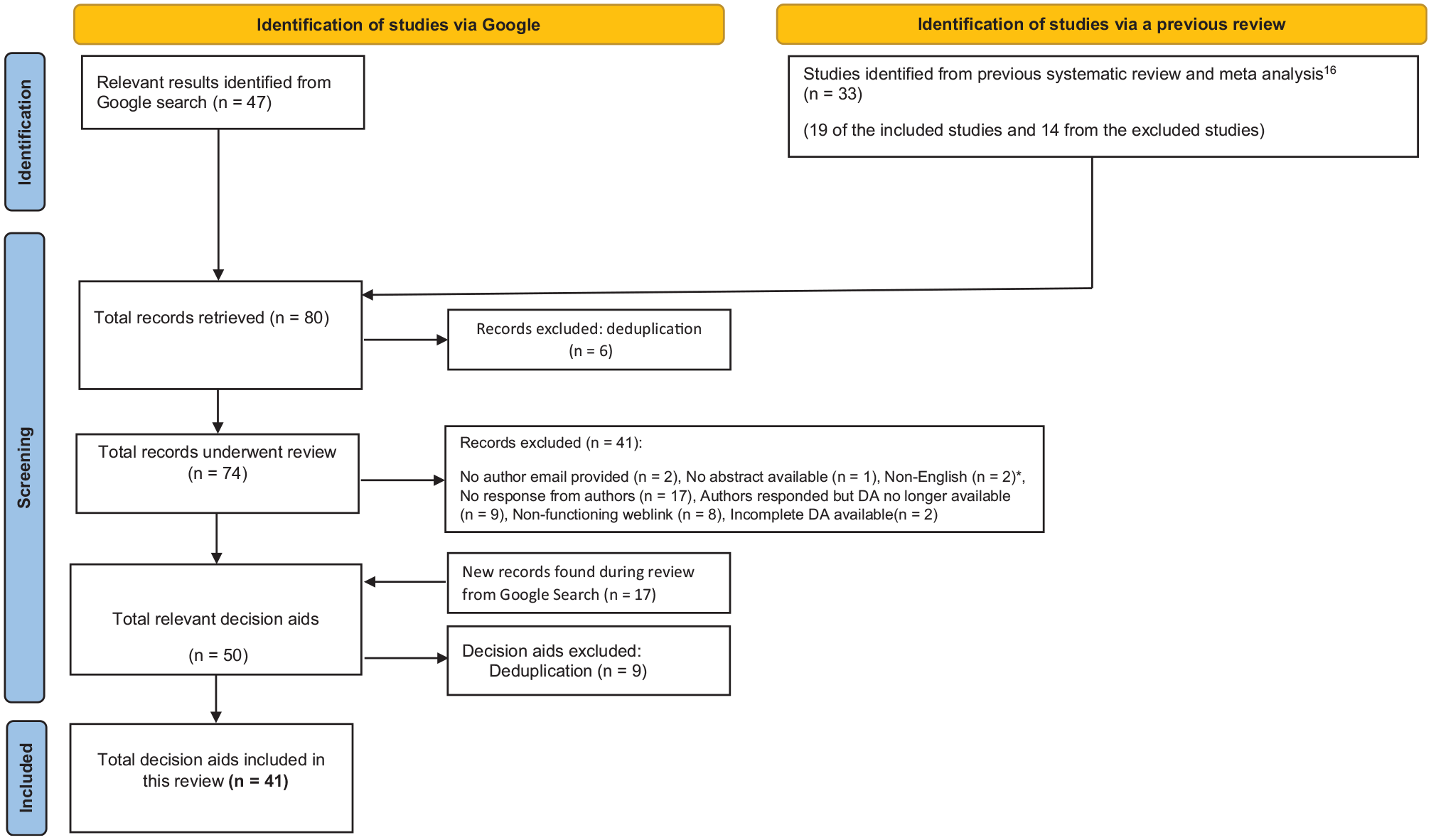
Flow chart outlining the search.
Of these 41 decision aids, 26 (63%) were published in the United States (Supplementary Table 1). The dates of publication were reported in only 32 of the 41 (78%) decision aids; 22 decision aids (54%) were published after 2010, and 14 (34%) were published after 2015. The publication year ranged from 1993 to 2017, where this was reported. Only 7 (17%) decision aids reported the year they had been developed. Of the 41 included decision aids, 40 were published in English, and one was translated through publisher website. All decision aids were accessible online, with 22 also being available in print (54%) and 16 as leaflet or pamphlet (39%). Approximately half of all decision aids had not been evaluated in a research study (
While all decision aids included general information about benefits and harms, 22 (54%) presented the benefits of PSA screening before the harms (Supplementary Table 2). Sixteen (39%) decision aids presented information on competing risks of mortality, for example, “many men will develop prostate cancer in their lifetime but most men die of something else other than prostate cancer.” A very small minority presented information of the prevalence pool of undiagnosed prostate cancer in the general population (2.5%), where this was described as “autopsies on men who have died from causes other than prostate cancer have shown that up to 60% of men over the age of 60 had prostate cancer and did not know it” or “researchers have looked at the prostate glands of men who died from causes other than prostate cancer. Around 25% of men in their 50’s, 35% of men in their 60’s, and 40% of men aged 70 years or older had cancer cells in their prostate glands.”
Information on overdiagnosis was mentioned in most decision aids (Table 1). Although 38 decision aids (92%) included information about overdiagnosis, only 9 (21%) of the decision aids specifically used the word
Information Reported on Overdiagnosis
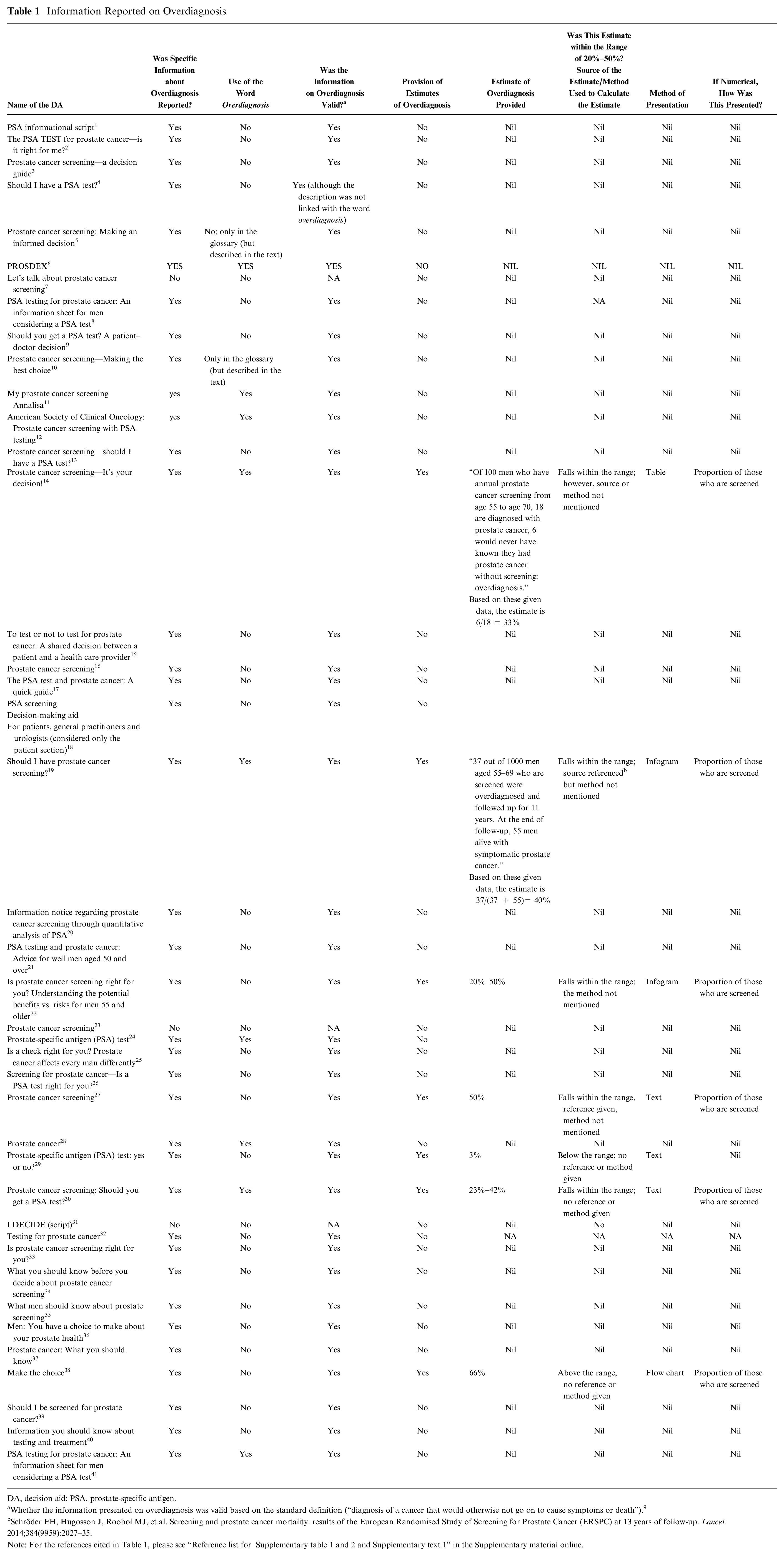
DA, decision aid; PSA, prostate-specific antigen.
Whether the information presented on overdiagnosis was valid based on the standard definition (“diagnosis of a cancer that would otherwise not go on to cause symptoms or death”). 9
Schröder FH, Hugosson J, Roobol MJ, et al. Screening and prostate cancer mortality: results of the European Randomised Study of Screening for Prostate Cancer (ERSPC) at 13 years of follow-up.
Note: For the references cited in Table 1, please see “Reference list for Supplementary table 1 and 2 and Supplementary text 1” in the Supplementary material online.
Examples of Descriptions of Overdiagnosis in Prostate Cancer Screening Decision Aids a

“Quantification” terminology is underlined.
Note: For the references cited in Box 1, please see “Reference list forSupplementary table 1 and 2 and Supplementary text 1” in the Supplementary material online.
Discussion
Our review found that most decision aids (
A previous review by Housten and colleagues
14
on decision aids for several different cancers (
We found that only a minority of the decision aids (
We also found that the estimates of overdiagnosis were reported in various ways, including the use of text (
Presenting the benefits before the harms may result in a framing effect. A framing effect could occur when people are influenced by different semantic descriptions of the same issue, which may lead to different risk perceptions affecting their decision making.31,32 Visual aids such as infographics can minimize framing effects by showing the number of patients experiencing the outcome and the number of patients not experiencing the outcome for each option being considered all at once. 10 Most of the decision aids included in our review that did not present benefits before the harms presented the information using infograms (a visual representation of information/data) or flow charts to show outcomes of screening. Future studies may explore how such visual aids may effectively minimize framing effects further, as well as explore how any framing effects may influence the ultimate decisions made on screening.
The presence of a large reservoir of subclinical cancer in the general population is a key prerequisite that contributes to overdiagnosis of cancers, specially prostate cancer.11,33 A systematic review of autopsy studies done on men who have died from other causes found that the estimated prevalence of subclinical prostate cancer increased from 5% (95% confidence interval [CI]: 3%–8%) at age <30 years to 59% (95% CI: 48%–71%) by age >79 years. 33 Including information about the prevalence pool of subclinical cancers in a prostate cancer screening decision aid is important because screening could lead to the detection of these dormant prostate cancers, which would otherwise go undiscovered and not cause any harm.
However, we found that only 2 of 41 (5%) of the decision aids included this information. Furthermore, because prostate cancer is a slow-growing disease, most people diagnosed with prostate cancer actually die with it (from other more common causes such as cardiovascular disease), rather than from it.11,12 However, only 16 (39%) of the decision aids included information on how the competing risks of mortality may lead to overdiagnosis.
To our knowledge, this is the first detailed review of prostate cancer screening decision aids to analyze the information on overdiagnosis including the estimates of overdiagnosis, concepts of competing mortality risks, and the presence of a silent reservoir of indolent cancers. Our comprehensive search strategy based on the search results from a published systematic review and complemented by a Google search is a strength.
Limitations
Our study had some limitations. We included only decision aids published in English or directly translatable to English through publisher websites. In addition, not all decision aids identified through our Google search were accessible in their complete form and hence had to be excluded. It is also important to note that due to the lack of consensus on a single definition for overdiagnosis, in this study we used one of the most common definitions.
Furthermore, because the key focus of our study was to review how information on overdiagnosis is reported in prostate cancer screening decision aids, we did not investigate reporting of other related issues. We did not evaluate reporting on risk of false-positive results and potential harms that may arise. A 2019 review of prostate cancer screening decision aids available in Germany found that less than one-third of the decision aids described the risk of psychological distress caused by false-positive results but did not provide further details. 15 We also did not evaluate whether decision aids mentioned active surveillance as one means of mitigating harms from overdiagnosis of low-risk cancers. While reporting on both of these issues was out of the scope for our review, future studies might usefully explore how they are reported in prostate cancer screening decision aids.
Conclusion
Most patient decision aids currently available for prostate cancer screening lack information on concepts important for understanding the risks of prostate cancer overdiagnosis. The presentation of benefits before the harms may lead to framing that overemphasizes benefit. Specific guidance is needed on how to communicate the risks of overdiagnosis in decision aids, including appropriate content, terminology, and graphical display.
Supplemental Material
sj-docx-1-mpp-10.1177_23814683221129875 – Supplemental material for Including Information on Overdiagnosis in Shared Decision Making: A Review of Prostate Cancer Screening Decision Aids
Supplemental material, sj-docx-1-mpp-10.1177_23814683221129875 for Including Information on Overdiagnosis in Shared Decision Making: A Review of Prostate Cancer Screening Decision Aids by Thanya I. Pathirana, Kristen Pickles, Jarno M. Riikonen, Kari A. O. Tikkinen, Katy J. L. Bell and Paul Glasziou in MDM Policy & Practice
Footnotes
Acknowledgements
We wish to thank Mr. Justin Clark, senior research information specialist at the Institute for Evidence Based Healthcare, for his expert input in formulating the Google strategy.
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PG was a co-author of one of the assessed decision tools from the Royal Australian College of General Practitioners. TIP, KP, JR, KAOT, KJLB declare no conflicts of interest.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: KAOT is supported by the Academy of Finland (309387), Competitive Research Funding of the Helsinki and Uusimaa Hospital District (TYH2019321; TYH2020248), and Sigrid Jusélius Foundation. KJLB is the recipient of an Australian National Health and Medical Research Council (NHMRC) Investigator Grant (No. 1174523). PG is funded by an Australian National Health and Medical Research Council NHMRC Fellowship APP1175487.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
