Abstract
Health misinformation is a growing problem in today’s world, reflecting an increasingly polarizing discourse in social and news media as well as declining trust in institutions. It is a particularly concerning issue in public health, where its effects can range from individually damaging to disastrous on a population level. In this article, we present and investigate one approach for addressing health misinformation—inoculation strategies—focusing specifically on tobacco marketing, which frequently uses suggestive descriptors that imply reduced harm. We theorized that inoculation strategies, that is, exposing people to weakened versions of opposing arguments to make them more resistant to persuasion or influence, can prevent misperceptions about a topic while preserving accurate favorable beliefs. We found that inoculation messages generally prevented lower-harm misperceptions without affecting lower-addictiveness beliefs for reduced nicotine content cigarettes or opinions about other modified risk tobacco products. We also found that information about prior industry deception promoted industry skepticism. These findings expand the evidentiary base for inoculating against misinformation, exploring strategies for nuanced communication of potentially competing messages. They also can inform policy and messaging strategies for health campaign designers and tobacco regulators.
In 2025, the United States saw multiple deaths from measles, a disease previously considered eliminated in 2000. 1 In the early 2020s, at the height of the COVID-19 pandemic, questioning of scientific evidence spurred opposition and noncompliance with mask and vaccine mandates. 2 And in 2009, regulators banned advertising for “light” and “mild” cigarettes because it led people to wrongly believe that these cigarettes delivered less tar and nicotine and were therefore safer to smoke.3,4 These ostensibly distinct issues are linked by the common thread of health misinformation—information that is “false, inaccurate, or misleading according to the best available evidence at the time.” 5 Public health officials have battled the spread of misinformation throughout history. 6 Misinformation is especially concerning today because of increasingly polarizing discourse in social and news media as well as declining trust in institutions, including medical systems, and its effects range from individually damaging to disastrous on a population level.7,8
Misinformation is increasingly problematic in the tobacco control field, the focus of our research. Despite the tobacco industry’s poor reputation and general public understanding that cigarettes are deadly, misinformation persists about the risks associated with different types of tobacco products, in part because clever marketing tactics obscure these risks. For instance, advertising for “organic” and “natural” cigarette varieties gives rise to misperceptions that these cigarettes are less harmful,9 –12 which can be further propagated through common sources of health information (and misinformation) like social media. 13 For public health advocates, responding to new marketing ploys and the resulting misperceptions resembles the carnival game of whack-a-mole—address one problem, and another almost immediately pops up. As the tobacco market has grown, this task has become even more complex, fueled by a new and complicated truth: Some tobacco products really are better (or worse) than others.
In 2019, the Food and Drug Administration (FDA) began authorizing marketing for modified risk tobacco products (MRTPs), which are considered lower risk than regular tobacco products. The manufacturers of General Snus (a smokeless tobacco product placed under the upper lip) and IQOS (a heated tobacco product that produces an aerosol instead of smoke), for example, were respectively authorized to advertise that these products pose lower risks of disease or lower exposure to harmful chemicals14,15 than cigarettes. The public health rationale here is that adults who use more harmful tobacco products may benefit by switching to these products and therefore reduce their risk of harm or chemical exposure. But messaging around MRTPs must straddle the line of communicating their benefits to current tobacco users without unintentionally attracting new users like youth, creating misunderstandings about product attributes, or sparking confusion about which products actually are modified risk.
VLN King and VLN Menthol King, reduced nicotine content (RNC) cigarettes made by 22nd Century Group, received MRTP authorization in 2021 for marketing claims of “95% less nicotine”/“greatly reduces nicotine consumption.”16,17 While RNC cigarettes lower exposure to nicotine (the chemical that makes cigarettes so addictive), they do not otherwise reduce risk of smoking-related disease,18 –21 so they are less addictive but not inherently less harmful. In fact, when the FDA considered the MRTP application for VLN King, a key question was whether consumers would accurately understand the product’s addiction and disease risks based on its marketing. Specific concerns were that smokers could mistakenly think these cigarettes are less harmful and be less motivated to quit, or that people who use other less harmful tobacco products could instead be attracted to these cigarettes. In some prior research, consumers accurately recognized the lower addictiveness of these cigarettes but also incorrectly perceived them as less harmful,22,23 and different message strategies produced different degrees of misperception. 24
The need for communication strategies about RNC cigarettes and preventing associated misinformation is particularly timely because in January 2025, the FDA proposed an historic rule to reduce nicotine content to minimally or nonaddictive levels in most combusted tobacco products—in effect, requiring all currently available cigarettes and most cigars to be RNC products. 25 Given the possible impact of this rule, communication strategies to prevent misperceptions about RNC tobacco products may be crucial for encouraging their appropriate use in support of public health and may in turn inform efforts to combat other types of health-related misinformation. One strategy in particular—inoculation messaging (or countering a future persuasive attempt by first exposing an audience to a weakened version of the persuasion in order to generate a sense of threat) —holds particular promise and was the focus of our research.
Inoculating Against Misinformation
Once misinformation is accepted, it is very difficult to correct,26 –28 as a result of belief persistence (not believing a correction to misinformation), belief echoes, (affective consequences of corrected misinformation), and motivated reasoning (the desire to believe misinformation consistent with one’s own perspectives).29 –31 Because of this, strategies that can prevent misinformation before it is spread may be especially vital, and one strategy is leveraging inoculation theory.
Inoculation theory posits it is possible to build resistance to a future persuasive attempt by first exposing people to a weakened version of that persuasion and generating a sense of threat.32,33 Inoculation messages thus warn about the anticipated persuasive attempt (forewarning) and then provide counterarguments to that persuasion (refutation). Threat is a key mechanism in this process: The forewarning induces a sense of threat to one’s existing attitudes and prompts the desire to counterargue while the refutation offers particular counterarguments.32 –36 Refutations using arguments that do not appear in the subsequent persuasive message (refutational different) can be effective,37,38 although some studies have found mixed effectiveness of these inoculations compared to those that refute the exact same arguments raised in the persuasive message (refutational same). 39
Inoculation approaches have been used to counter science and political misinformation,40 –47 and in some health information contexts,48,49 where they often induced resistance to antivaccine propaganda.50 –52 Yet some research has found varying effectiveness in the latter contexts,53 –55 underscoring the importance of testing specific inoculation strategies for health misinformation.
One inoculation strategy involves flagging prior deception by the persuasive source, which may promote resistance to future misinformation from that source43,44,56,57 or encourage openness to a corrective message.58,59 Prior deception is especially relevant to tobacco-related misperceptions: A 2006 federal court ruling 60 found that the tobacco industry purposely misled the public regarding health risks and addictiveness of tobacco use. The initial court ruling mandated that the industry publicly correct misperceptions about tobacco and report their intentional deception. However, on appeal, tobacco companies were no longer required to disclose their prior deception, as such disclosures could be considered more emotional or punitive than factual or effective in correcting misinformation. 61
Identifying deceptive tobacco industry tactics can help promote misinformation correction 62 as well as foster industry “denormalization.” Messaging that highlights such deception has prompted anger toward the industry and boosted intentions to quit. 63 Promoting source distrust may be a useful approach for preventing misperceptions arising from marketing from knowingly deceptive sources, such as thinking that RNC cigarettes are less harmful after seeing their marketing. For instance, in prior research, inoculation messages mentioning deceptive tobacco marketing prevented misperceptions about “natural” cigarettes and promoted skepticism toward industry messaging. 64 But those inoculations did not also have to promote accurate beliefs about product benefits, as messaging about RNC cigarettes does.
Some prior research has shown that inoculation messages warning about misinformation or fake news can inadvertently decrease the credibility of other accurate information. 65 So here’s the tricky part: It can be problematic if source skepticism induced by inoculations also extends to accurate, health-serving messages from the same or related sources—for example, if skepticism extends to beliefs about reduced addictiveness of RNC cigarettes or reduced harm of other MRTPs. Therefore, how inoculation strategies can be leveraged to prevent health misperceptions and promote accurate beliefs about product risks and benefits remains to be seen.
Here, we assessed whether inoculation strategies can be used to (a) prevent misperceptions that arise from misleading advertising while (b) avoiding unintended spillover effects on other beliefs about this topic and related ones. We also aimed to determine whether inoculations are more effective in preventing misperceptions and sparking skepticism toward a persuasive source if they include information about prior deceptive marketing from that source. We explored these questions in the context of VLN-brand RNC cigarettes, using the following hypotheses:
H1: Inoculation messages will prevent implicitly arising misperceptions about lower health risks of VLN cigarettes.
H2: Inoculations that identify prior deceptive marketing by the tobacco industry will be more effective in (a) addressing misperceptions about VLN cigarettes and (b) promoting skepticism of tobacco marketing.
H3: Inoculations will not yield unintended consequences on beliefs about other MRTPs.
Since prior research has not demonstrated whether inoculations can combat misperceptions while preserving accurate, favorable beliefs about the same topic (and in some cases inoculations have had unintended effects on public opinions on other topics), 65 we posed this research question: Will inoculations promote accurate beliefs about reduced addictiveness of RNC cigarettes while simultaneously preventing reduced-harm misperceptions?
Methods and Analysis
Methods
In 2022, we completed an online randomized experiment recruited via Qualtrics research panels with 1,295 adults who were current or former daily smokers. Participants had to be at least 18 years old and current smokers (i.e., smoking either daily or on some days) or have formerly smoked on a daily basis but quit at least six months prior.
We adapted our methods from another study on inoculating against tobacco misperceptions. 64 Participants were randomized to an ad-only control condition or one of four inoculation conditions. All participants saw two marketing messages for VLN-brand cigarettes after completing a distraction task. Participants in the inoculation conditions saw one inoculation message prior to the distraction task and VLN ads. Outcome measures followed ad exposure. Figure 1 shows the experimental procedure.
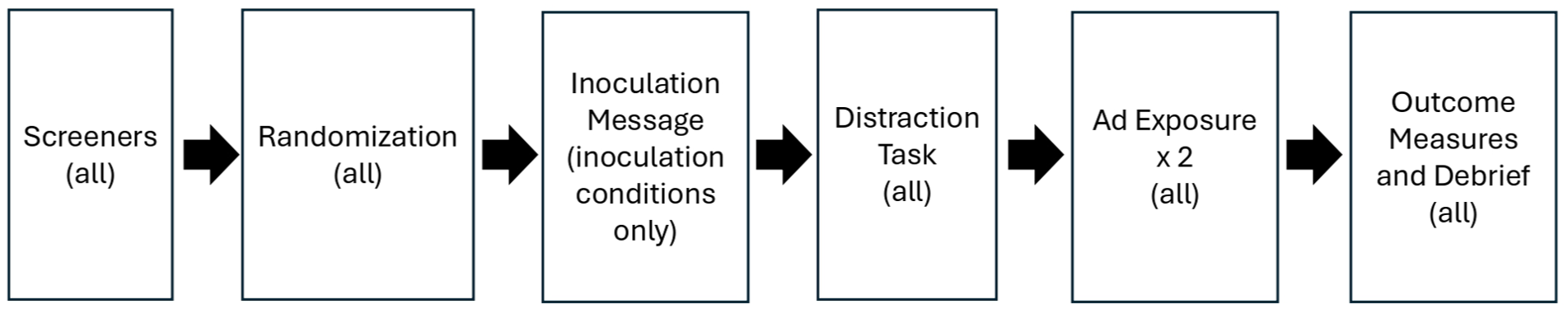
Experimental Procedure
Materials
All participants saw two marketing stimuli (the persuasive message): a VLN ad with minor graphic edits from the brand’s MRTP application and a textual description compiled from the brand’s website and its modified risk claims, alongside an image of VLN packs. Supplemental Figure 1 shows the marketing stimuli.
The study conditions were one control condition that did not receive an inoculation message prior to the marketing stimuli and four inoculation conditions that did. Inoculation stimuli varied by the presence or absence of explicit forewarning and refutation components (primary inoculation dimensions) and the presence or absence of information about prior deceptive marketing practices by the tobacco industry. The explicit forewarning cautioned the audience to be wary of VLN-brand marketing. Here, the forewarning was operationalized to warn about potentially misleading information rather than caution against information that might change the audience’s preexisting position, as would be seen in more traditional inoculation forewarnings.
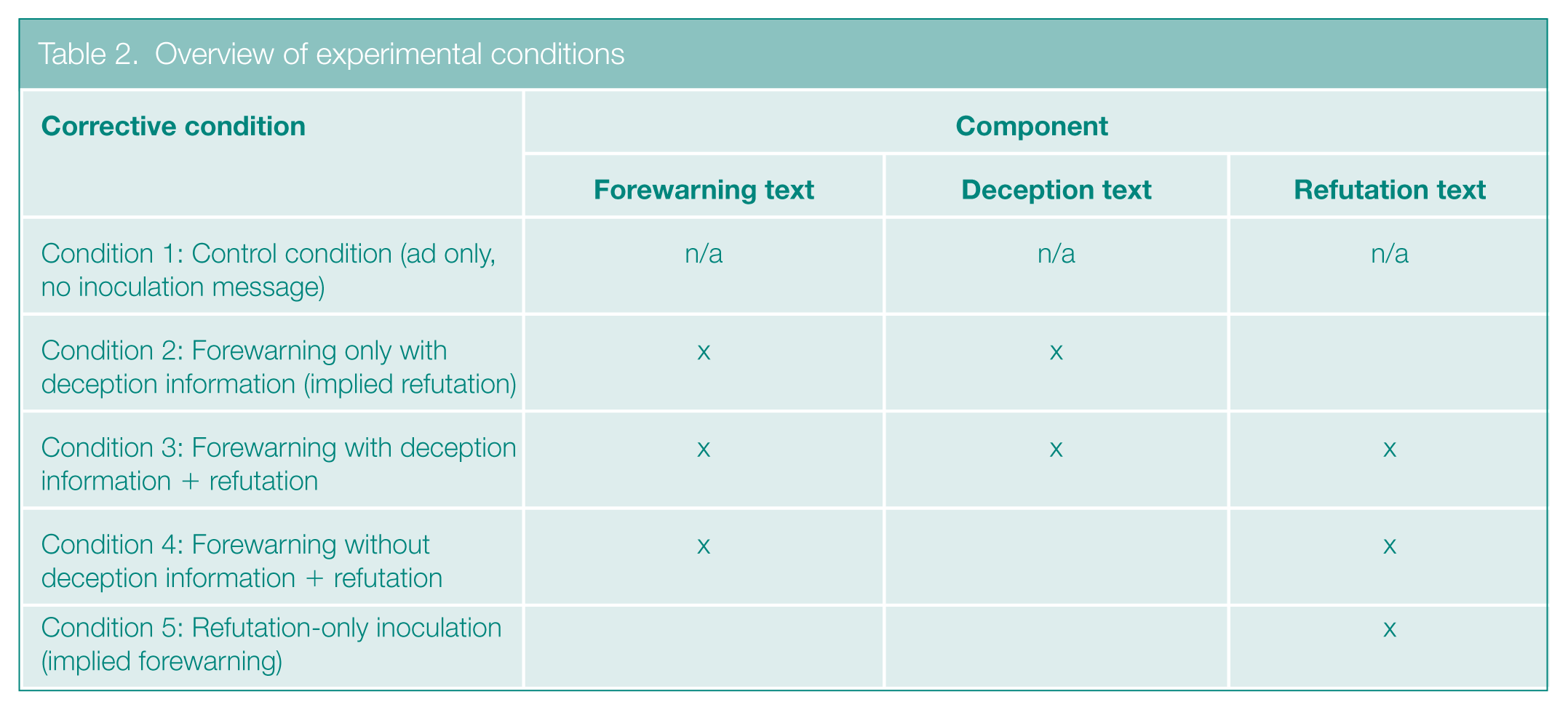
The deception information, a component of the forewarning in some conditions, detailed the tobacco industry’s history of deceptive messaging to conceal the harm from smoking cigarettes. The explicit refutation included corrective information specifying that RNC cigarettes are less addictive but not less harmful. Table 1 shows sample text for each of these components. Table 2 identifies the components included in each inoculation condition: (a) forewarning with deception, (b) forewarning with deception + refutation, (c) forewarning without deception + refutation, and (d) refutation only. In each inoculation condition, participants were randomized to see one of two inoculation messages that were written in expository or in third-person narrative style.66,67
Sample text of inoculation components

Overview of experimental conditions
Data Analyses and Research
We assessed beliefs about relative harm and addictiveness of VLN-brand cigarettes compared to other cigarettes, using measures adapted from the Population Assessment of Tobacco and Health (PATH) Study, 68 as well as other beliefs about VLN addictiveness such as “easier to quit”. We also assessed other potential health misperceptions 69 about VLN-brand cigarettes such as “less damaging to your lungs”.
We assessed skepticism about the deceptive source, 70 such as the belief that tobacco companies are not a trustworthy information source. We included a broader attitude measure to distinguish general attitudes toward the industry from the more specific trust or skepticism measures. Finally, we explored unintended spillover effects onto beliefs about other current/prospective MRTPs (snus, e-cigarettes, heated tobacco products) that, unlike RNC cigarettes, may pose lower disease risk because they do not burn tobacco. We assessed perceived harm of other MRTPs 68 and intentions 71 to try them. Supplemental Figure 2 lists the outcome measures.
For harmfulness and addictiveness measures adapted from PATH, we used chi-square tests to compare percentages of respondents indicating the target products are “somewhat” or “a lot less” harmful or addictive among inoculation conditions versus control and to compare among inoculation conditions. For intention measures, we compared percentages by condition for those indicating any willingness to use the product. For Likert-agreement items, we created the following scales: Lower Harm Misbeliefs (n = 6, α = .97), Reduced Addictiveness Beliefs (n = 3, α = .81) and Skepticism Toward the Persuasive Source (n = 4, α = .76) using one-way analysis of variance with Tukey correction to assess differences among conditions.
Results
Participants
The sample (N = 1,295) was 51% male, primarily White (79%), and non-Hispanic (87%), with education levels of some college or more (64%), and 68% reporting current daily smoking. Supplemental Table 1 lists participant characteristics and condition assignment.
Beliefs
Table 3 shows the results for H1, H2, and the research question. H1, that inoculations would prevent misbeliefs relative to control, was supported: Significantly fewer inoculation condition participants than control participants (33.1%–38.7% versus 49.1%, p < .05) reported believing VLN-brand cigarettes are less harmful (PATH measure). Most inoculations also prompted lower misperceptions relative to control on the additional health misperception scale. Results did not support our second hypothesis about superior effectiveness of inoculations with information about prior source deception, as there were no significant differences among inoculation conditions for the harm belief measure or scale.
Endorsement of beliefs, by condition
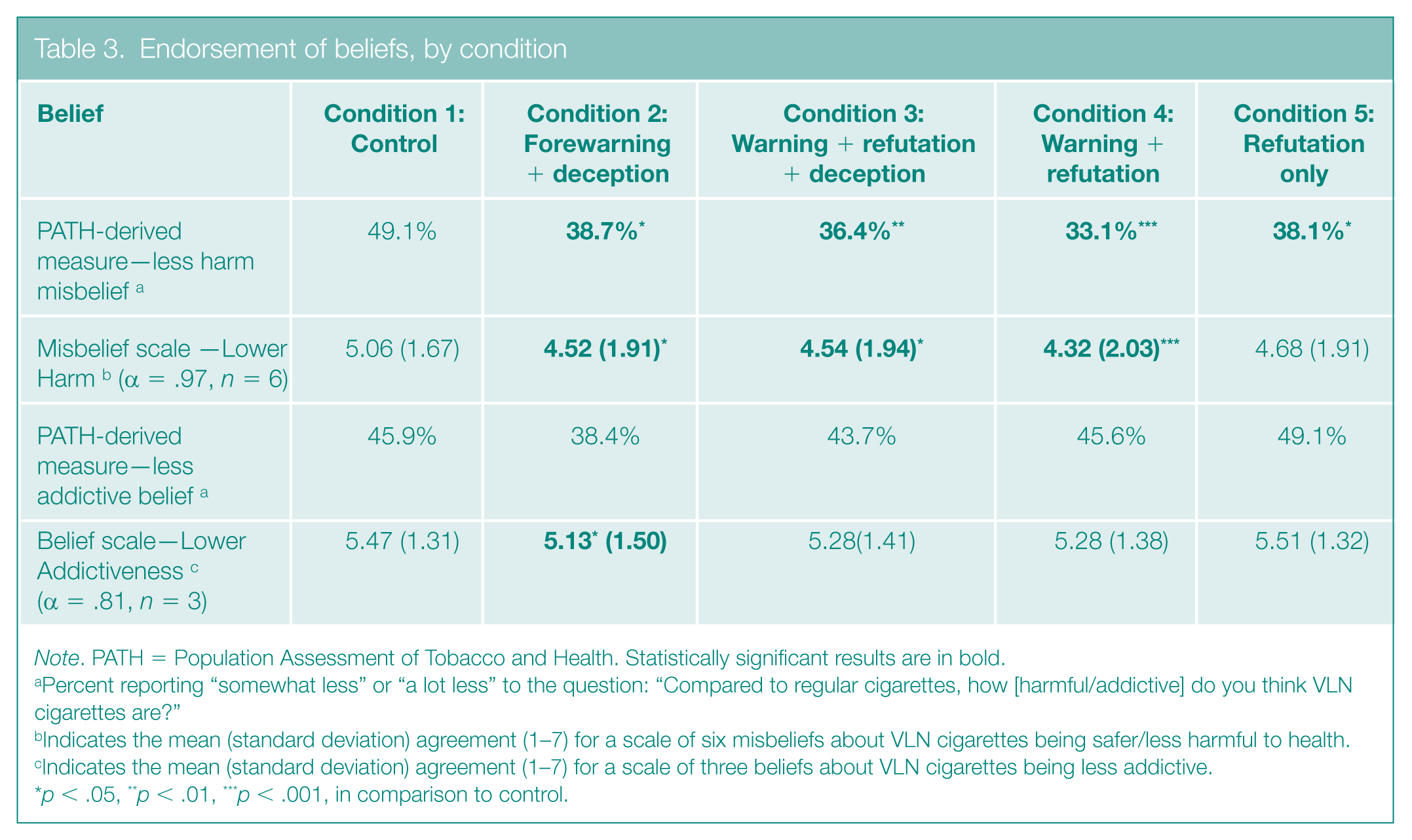
Note. PATH = Population Assessment of Tobacco and Health. Statistically significant results are in bold.
Percent reporting “somewhat less” or “a lot less” to the question: “Compared to regular cigarettes, how [harmful/addictive] do you think VLN cigarettes are?”
Indicates the mean (standard deviation) agreement (1–7) for a scale of six misbeliefs about VLN cigarettes being safer/less harmful to health.
Indicates the mean (standard deviation) agreement (1–7) for a scale of three beliefs about VLN cigarettes being less addictive.
p < .05, **p < .01, ***p < .001, in comparison to control.
Regarding our research question, inoculations promoted accurate beliefs about reduced VLN-brand cigarette addictiveness in most but not all cases. For each inoculation condition, the percentage of participants who reported VLN-brand cigarettes are less addictive than regular cigarettes did not significantly differ from control, meaning the inoculation messaging did not detract from this belief. While most inoculations also prompted comparable results to control on the addictiveness belief scale, the forewarning + deception condition produced lower levels of belief in reduced VLN-brand cigarette addictiveness, relative to control (Condition 1: M = 5.13, SD = 1.50; control: M = 5.47, SD = 1.31, p < .05).
Skepticism
Results did not support our hypothesis that inoculations with information about prior source deception would promote higher source skepticism than those without deception information, as there were no significant differences among inoculation conditions on the Skepticism scale (see Table 4). Yet, it is worth noting that only the inoculations with deception information reduced trust/increased skepticism in industry messaging relative to control (Condition 2: M = 3.81, SD = 1.42; Condition 3: M = 3.83, SD = 1.52; control: M = 4.21, SD = 1.42, p < .05).
Tobacco industry trust/skepticism, by condition
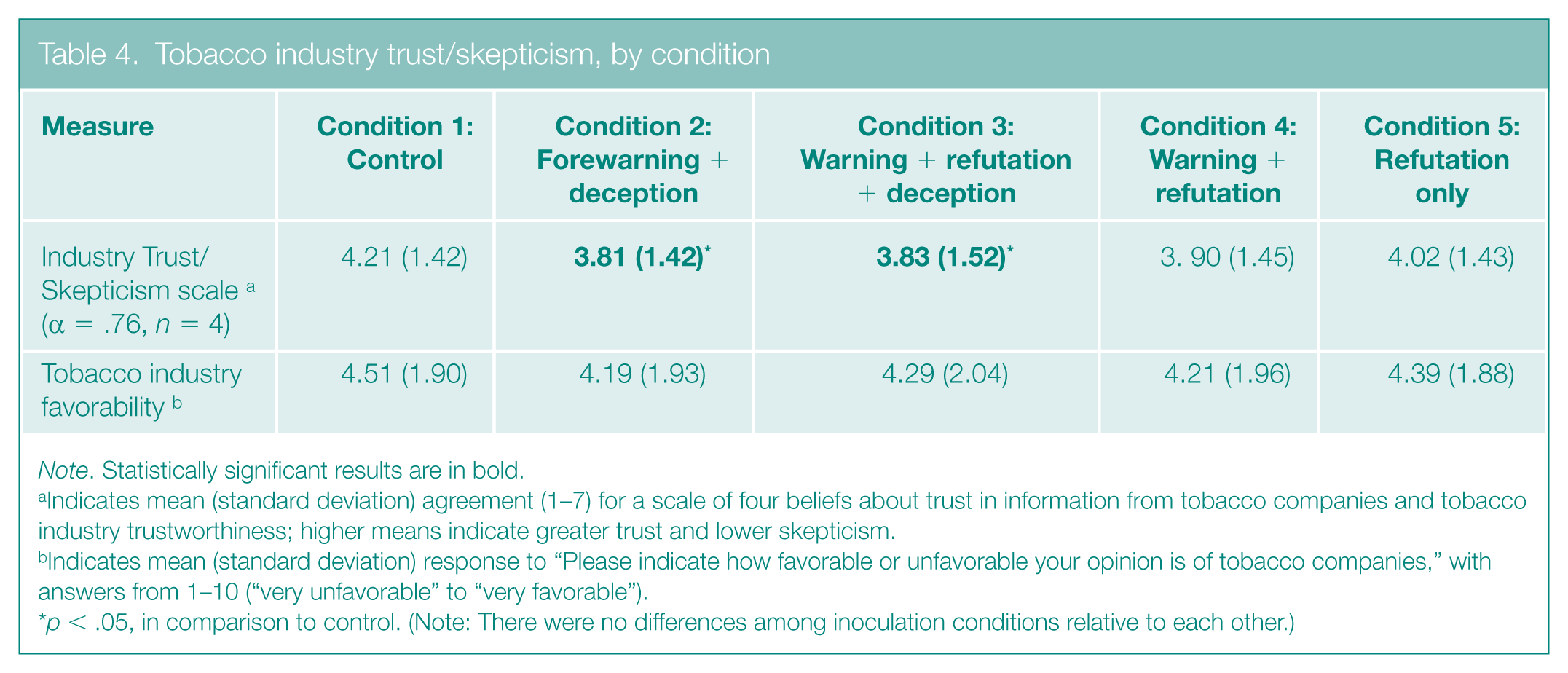
Note. Statistically significant results are in bold.
Indicates mean (standard deviation) agreement (1–7) for a scale of four beliefs about trust in information from tobacco companies and tobacco industry trustworthiness; higher means indicate greater trust and lower skepticism.
Indicates mean (standard deviation) response to “Please indicate how favorable or unfavorable your opinion is of tobacco companies,” with answers from 1–10 (“very unfavorable” to “very favorable”).
p < .05, in comparison to control. (Note: There were no differences among inoculation conditions relative to each other.)
Spillover Effects
The results supported H3, that inoculations would not have unintended spillover effects on beliefs or intentions for other MRTPs. None of the inoculations lowered beliefs about reduced harm of other MRTPs or reduced interest in them relative to control (see Table 5).
Perceived harm & intentions toward modified risk tobacco products, by condition
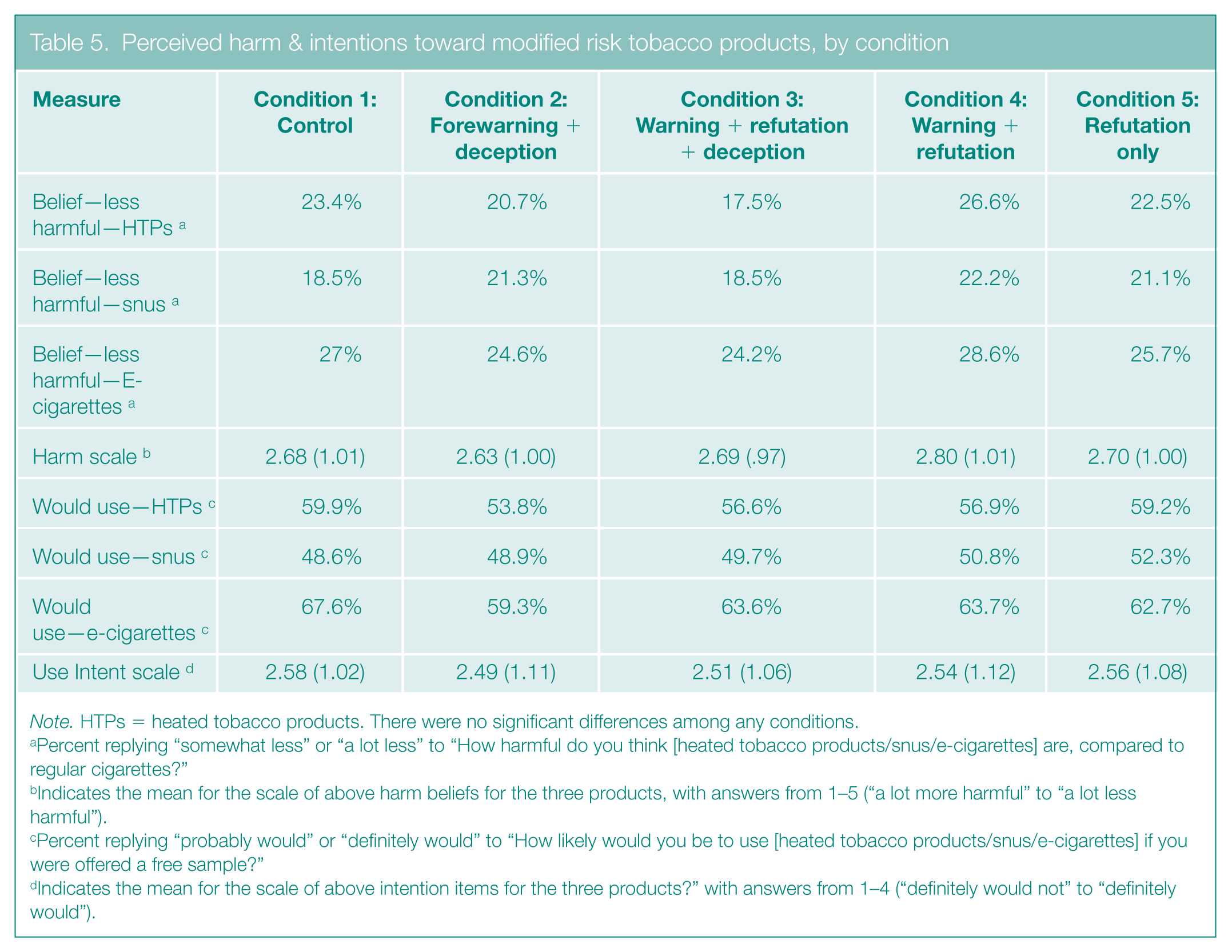
Note. HTPs = heated tobacco products. There were no significant differences among any conditions.
Percent replying “somewhat less” or “a lot less” to “How harmful do you think [heated tobacco products/snus/e-cigarettes] are, compared to regular cigarettes?”
Indicates the mean for the scale of above harm beliefs for the three products, with answers from 1–5 (“a lot more harmful” to “a lot less harmful”).
Percent replying “probably would” or “definitely would” to “How likely would you be to use [heated tobacco products/snus/e-cigarettes] if you were offered a free sample?”
Indicates the mean for the scale of above intention items for the three products?” with answers from 1–4 (“definitely would not” to “definitely would”).
Public Health and Policy Implications
These study findings are pertinent to public health practitioners as well as regulators as they may inform antitobacco and substance advocacy as well as public health campaigns more generally. Inoculation strategies can be incorporated into health campaign messages via social or news media,48,72 commercials, educational and classroom initiatives, 73 as well as press releases from health agencies. Our findings for H1 demonstrated that just as refutational-different inoculation arguments can effectively inoculate against subsequent persuasive attacks, inoculation techniques that forewarn about suggestive industry messaging (e.g., our forewarning-only inoculation) can be applied to prevent misperceptions about a topic or product, even without expressly refuting specific elements from that product’s advertising. This is important because in the context of suggestive marketing for tobacco and other addictive products like alcohol and sugar-sweetened beverages, it is hard to anticipate and counter the full spectrum of industry ad tactics. Thus, a key finding for public health practitioners is that public health messages with a strong forewarning cautioning people to question suggestive industry marketing have the potential to protect against health misperceptions and may disrupt industry persuasion.
Another finding applicable for practice was that when an inoculation forewarning was bolstered with information about prior deceptive industry practices, the inoculations promoted skepticism toward messaging from that source. This research provides evidence of a corrective and protective value of inoculations with information about industry deception, which may be relevant from a legal or regulatory standpoint. Prior deception or efforts to conceal product risks can be used as a theme in public health campaigns targeting industries like tobacco and updated with specific counterarguments to industry marketing as appropriate. Another takeaway for practitioners is that messages highlighting past industry deception can serve a protective function in prompting people to be more skeptical of future messages from that industry.
Our study can also inform tobacco regulators, who must balance the need for messaging about RNC cigarettes or other MRTPs to communicate the benefit of moving away from worse products without simultaneously creating misperceptions that could stimulate tobacco use initiation. Our inoculations, which highlighted reduced product addictiveness but cautioned against assumptions of reduced harm, were successful in preventing misperceptions without depressing correct beliefs of lower addictiveness or causing unintended effects regarding other MRTPs. This underscores the potential of inoculation strategies in achieving more nuanced public health messaging tasks. Although needing further study, a possible regulatory implication is that MRTP communications, and perhaps product labels, may enhance effectiveness by juxtaposing product benefits and product risks.
Our findings may directly inform public health messaging related to MRTP authorization of RNC cigarettes, the new nicotine product standard, and other MRTPs, in order to minimize risk from false beliefs and maximize protection of public health. They can also inform how to apply inoculation strategies to prevent misperceptions that arise from misleading marketing or communications by any persuasive source with a history of deceptive messaging.
Discussion
We explored the application of inoculation message strategies to prevent harmful misperceptions about a topic without stifling accurate positive beliefs, in this case regarding RNC cigarettes, a type of MRTP. MRTPs occupy a unique market position, authorized solely to protect public health by encouraging switching from other products that are worse for human health. Yet regulators walk a fine line with MRTP marketing as it must convey benefits to the intended audience of current smokers without dissuading cessation or attracting unintended audiences like youth, a vulnerable population the tobacco industry intentionally targets. 74
Our findings support and extend prior tests of inoculation theory showing the effectiveness of using inoculation strategies to combat political and science-related misinformation41,43,58 and also to inspire more global source skepticism that engenders continued resistance to suggestive marketing. 64 Perhaps our most distinct finding that extends prior research was that besides preventing harm-related misperceptions, our inoculations preserved accurate beliefs about reduced addictiveness in almost all cases, except the forewarning-only inoculation, which could indicate the importance of the explicit refutation in clarifying specific benefits versus risks. This more nuanced task has not been a focus in inoculation studies, and prior research about RNC cigarettes showed that other messaging strategies may unintentionally promote inaccurate reduced harm beliefs.22,24 Our findings generally show that inoculation strategies can achieve these potentially competing communication goals of combatting misperceptions while also preserving accurate, favorable beliefs.
Limitations of our study include its focus on a single issue, so the techniques explored here should be tested in additional health contexts. Moreover, we did not use a full factorial design; that is, the forewarning-only inoculation was only presented with deception information, in order to have a more parsimonious design and because the forewarning-only inoculation already lacked specific refutational arguments. Also, in our experimental context, the inoculations were shortly before the persuasive message, so whether they are similarly effective with longer time delays between inoculation and persuasive attack would be worth exploring in future research. Lastly, we did not measure threat, which is considered a key mechanism of inoculation effects, so we cannot determine which inoculations elicited the strongest threat response.
Broadly speaking, our results indicated that unlike other messaging approaches, 22 inoculation messages achieved what they were intended to do: They prevented misperceptions about RNC cigarettes and promoted source skepticism without yielding unintended effects such as depressing accurate beliefs about RNC cigarettes or affecting opinions of other MRTPs. This research expands the evidentiary base for applying inoculation strategies to health misinformation and directly informs messaging strategies for public health practitioners and regulators.
Supplemental Material
sj-docx-1-bsx-10.1177_23794607251384736 – Supplemental material for Leveraging inoculation theory to combat the spread of health misinformation
Supplemental material, sj-docx-1-bsx-10.1177_23794607251384736 for Leveraging inoculation theory to combat the spread of health misinformation by Stefanie K. Gratale, Melissa Mercincavage, Matthew Joy and Olivia A. Wackowski in Behavioral Science & Policy
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Cancer Institute of the National Institutes of Health and the U.S. Food and Drug Administration Center for Tobacco Products under Grant #U54CA229973, via the University of Pennsylvania-Rutgers Tobacco Center of Regulatory Science Pilot Project Program. Contributions by SG were also supported in part by K01CA281062. The content is solely the responsibility of the authors and does not necessarily represent the views of the NIH or FDA.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
