Abstract
Introduction
Cancer imposes substantial physical and psychological burdens that negatively affect patients’ health-related quality of life (HRQoL). Assessing HRQoL prior to chemotherapy initiation is essential for tailoring early supportive care strategies.
Objective
To describe the characteristics and identify the factors associated with HRQoL in newly diagnosed cancer patients immediately before initiating outpatient chemotherapy.
Method
This cross-sectional study included adults with newly diagnosed stage I–III cancer scheduled to begin outpatient chemotherapy at a Brazilian cancer referral center. Sociodemographic and tumor-related data were obtained using a structured questionnaire. HRQoL was assessed with the EQ-5D-3L instrument. Descriptive and bivariate analyses were performed using Fisher's exact test.
Results
Eighty-four patients were included (mean age: 57.68 years); most were female (72.62%), of mixed ethnicity (40.48%), had completed only primary education (48.81%), and were married (51.19%). The average time from diagnosis to treatment initiation was 120 days, and 60.71% reported comorbidities. Breast cancer was the most common diagnosis (57.14%), predominantly at Stage II (57.14%). The mean EQ-5D-3L index score was 77.79. Comorbidities were significantly associated with impaired mobility (p = .009), and female patients reported significantly more anxiety and depression than males (p = .005). Inflammatory markers revealed that most patients had low neutrophil-to-lymphocyte ratio (76.54%), low platelet-to-lymphocyte ratio (72.84%), and elevated C-reactive protein (CRP; 84.52%). In the multivariate analysis, the presence of comorbidities remained significantly associated with worse mobility (OR = 5.456; 95% CI [1.205–24.707]; p = .028), and male sex was independently associated with lower levels of anxiety and depression (OR = 0.206; [0.067–0.634]; p = .006). A nonsignificant trend was also observed between higher CRP levels and increased pain (OR = 2.800; [0.824–9.510]; p = .099).
Conclusions
Patients with newly diagnosed cancer experience impaired HRQoL even before starting chemotherapy. Comorbidities were associated with worse mobility, and female patients reported higher levels of anxiety and depression. These findings support the need for early multidisciplinary care to mitigate functional and emotional burdens prior to treatment.
Introduction
Cancer is the disease with the highest incidence in developed countries and is the first or second cause of premature death after the age of 70 in many parts of the world (Primo, 2023; Siegel et al., 2024). Age, environmental factors such as exposure to solar radiation, and eating habits are some of the main causes of sporadic cancers (Primo, 2023). In Brazil, more than 2 million chemotherapy treatments are carried out every year and cost more than 48% of all costs related to cancer therapies in the Unified Health System (Sistema Único de Saúde—SUS; Ministério da Saúde, 2024). Even though chemotherapy is widely used to treat malignant neoplasms, the physical and mental problems related to treatment toxicity are still reported as detrimental to the patients’ health-related quality of life (HRQoL; Battisti et al., 2021; Giordano et al., 2002).
The World Health Organization defines quality of life (QoL) as an individual's perception of his position in life, within the context of the culture and value systems in which he lives and with their goals, expectations, standards, and concerns (Dehkordi et al., 2009). Based on these issues and the multidimensionality of QoL, the concept of HRQoL emerged, as well as a proliferation of instruments to measure it (Guyatt et al., 1993).
The term HRQoL can be understood as how health is empirically estimated as affecting QoL or even the term HRQoL is used to mean only its usefulness associated with a health state (Karimi & Brazier, 2016). HRQoL therefore measures self-perceived health status and how health can interfere with QoL (Ascef et al., 2017; Karimi & Brazier, 2016). Thus, there are several HRQoL questionnaires available, including the EuroQol 5 Dimensions (EQ-5D), which is useful for describing health through the patient's functionality and well-being (Karimi & Brazier, 2016; Rennen & Oppe, 2015).
The use of tools that assess HRQoL is reported in the scientific literature as an important tool in the general assessment of the impact of specific cancers on QoL since QoL is directly related to tumor staging (Casso et al., 2004; Dehkordi et al., 2009). However, in the cases of patients with a survival longer than one expected, there are problems in health habits, social support, changes in the emotional and spiritual dimensions of life, and concerns about self-image, in addition to experiencing numerous and complex symptoms, many of them caused by adverse effects associated with chemotherapy, which together greatly impact the HRQoL of these patients (Casso et al., 2004; Ganz et al., 2002; Mauer et al., 2008; Vitorino et al., 2018).
Inflammatory biomarkers such as the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR) and C-reactive protein (CRP) it's accessible and cost-effective indicators derived from routine laboratory tests. These biomarkers provide insights into tumor-related systemic inflammation and have demonstrated prognostic relevance across various cancer types. Some studies suggest that NLR and PLR may offer greater sensitivity than CRP in predicting adverse clinical outcomes and treatment response (Hung et al., 2011; Kim et al., 2020; Łukasiewicz et al., 2021).
NLR, PLR, and CRP ratios have gained prominence as useful tools for predicting prognosis and assessing treatment response in cancer patients. These indicators help personalize treatment and clinical management, since they reflect the balance between the systemic immune response and chronic inflammation—factors closely related to tumor progression and metastasis (Araújo et al., 2024; Zamora-Obando et al., 2022).
Recent studies have reinforced the relationship between systemic inflammation and HRQoL in cancer patients, highlighting the role of inflammatory biomarkers—such as CRP, NLR, and PLR—as potential predictors of disease burden and treatment-related impact. Chung et al. (2024) demonstrated that patients with advanced cancer exhibit significantly elevated levels of these inflammatory markers, which are associated with poorer symptom control, diminished global functioning, and consequently lower HRQoL. These findings support the hypothesis that chronic inflammation in cancer may exacerbate symptoms such as fatigue, pain, and impaired mobility—key domains in HRQoL assessment. Inflammatory biomarkers may thus provide an objective measure of systemic inflammation and aid in identifying patients at higher risk for HRQoL deterioration prior to chemotherapy initiation.
Another critical aspect involves the interplay between inflammation, body composition, and functional decline. Medeiros et al. (2024) emphasized that sarcopenia and cachexia, commonly observed in cancer patients, are closely linked to a heightened inflammatory profile, contributing to a decline in HRQoL during chemotherapy. Elevated CRP levels and increased NLR and PLR have been associated with reduced muscle mass and greater treatment toxicity, suggesting that systemic inflammation plays a pivotal role in both physical and emotional deterioration. Consequently, incorporating these biomarkers into clinical assessments may enable early identification of individuals at increased risk for functional decline and reduced QoL, allowing for the implementation of more personalized therapeutic strategies, such as nutritional support and antiinflammatory interventions.
Although the relationship between HRQoL and cancer has been widely studied, limited research has focused on newly diagnosed patients prior to their first outpatient chemotherapy session. Most available studies evaluate HRQoL after treatment has commenced, when the side effects of chemotherapy may already be present, making it difficult to distinguish between disease-related and treatment-related impacts. Moreover, clinical and biological factors influencing HRQoL at this early stage remain underexplored. This study seeks to address this gap by offering a comprehensive assessment of HRQoL before chemotherapy initiation and identifying key determinants that may influence it.
Review of Literature
Inflammatory cells and their mediators present in the tumor microenvironment influence cancer progression (Jiang et al., 2021; Kim et al., 2020; Ostan et al., 2015). The analysis of inflammatory biomarker response, such as CRP levels and neutrophil, lymphocyte, and platelet counts in the blood, contributes to defining the patient's prognosis (McMillan, 2013; Sun & Zhang, 2018). In this context, elevated NLR and PLR values have been related to poor prognostic outcomes, including overall survival and disease-free survival (Łukasiewicz et al., 2021; Silva et al., 2024; Templeton et al., 2014).
Several inflammatory biomarkers and their relationship with symptoms are known to be exacerbated in patients diagnosed with cancer, before, during, and after chemotherapy compared to noncancer patients (Hoogland et al., 2023). Although elevated CRP may be associated with several factors, such as infections or coexisting diseases, it is necessary to pay attention to pretreatment CRP levels that may reflect an overexpression of proinflammatory cytokines leading to the emergence of chemotherapy intolerance (Mikoshiba et al., 2019). CRP levels are considerably associated with HRQoL and can be negatively correlated with the physical, cognitive, and social dimensions. In addition, increased inflammatory biomarker levels are associated with poorer HRQoL (Li et al., 2019).
The comparison of NLR as a factor related to systemic inflammation is associated with tumor growth (Silva et al., 2024). Systemic inflammation can lead to tumor proliferation through mutations, genomic instability, and epigenetic modifications which induce the proliferation of premalignant cells. In addition, the increase in the N/L ratio during chemotherapy has been correlated with poor response to systemic treatment (Nemoto et al., 2021).
QoL in cancer patients is an essential aspect to support the practice of clinical professionals, especially in Oncology Nursing, and is directly linked to the patient's experience with the diagnosis of cancer and the treatment given (Lopes-Júnior & Lima, 2019; Lopes-Júnior et al., 2022). A systematic review study demonstrated the impact of chemotherapy-induced symptoms, such as vomiting on cancer patients (Sommariva et al., 2016), with repercussions on HRQoL. Therefore, the formulation of studies that address the impact of chemotherapy, considering the identification of symptoms related to both the disease and the adverse reactions of the therapy, allows to improve the healthcare of these patients. In addition, it makes it possible to develop personalized interventions that not only mitigate the adverse effects of chemotherapy treatment but can also promote a better experience for the individual (Lewandowska et al., 2020; Lopes-Júnior, 2021b, 2022, 2025; Lopes-Júnior & Veronez, 2023).
It should be noted that the early detection of indicators of poor HRQoL, such as the measurement of inflammatory biomarker levels in patients with advanced cancer, can make it possible to identify patients who are suitable for early referral to palliative care, to improve their QoL during cancer treatment (Daly et al., 2020; Velasco Yanez et al., 2024). In this context, the main objective of this study was to describe the characteristics and identify the factors associated with the HRQoL of newly diagnosed cancer patients immediately prior to first outpatient chemotherapy. In addition, secondary objectives were: (a) To analyze what sociodemographic and clinical characteristics affect the HRQoL of these cancer patients; (b) To investigate if there is an association between the HRQoL of newly diagnosed cancer patients and inflammation biomarkers immediately before the administration of the first chemotherapy; (c) To verify which dimensions of HRQoL are affected in newly diagnosed adult cancer patients immediately prior to first outpatient chemotherapy.
Method
Study Design
A cross-sectional observational study was carried out at Afecc-Hospital Santa Rita de Cássia (HSRC), a reference institution for cancer treatment in Brazil. The study adhered to STROBE guidelines, to enhance the reporting of observational studies.
Setting
The study was conducted at Afecc-Hospital Santa Rita de Cássia (Afecc-HSRC), a prominent reference center for cancer treatment located in Vitória, Espírito Santo, Brazil. Data collection took place between November 2022 and December 2023. The hospital provides comprehensive oncology services, including outpatient and inpatient care, chemotherapy, radiotherapy, and surgical oncology, serving a diverse patient population across the state and neighboring regions. Data collection occurred in the chemotherapy sector, where adult patients with newly diagnosed malignant neoplasms (stages I–III) were approached during their initial consultation. This setting allowed for the recruitment of patients prior to the start of the first chemotherapy, ensuring the assessment of baseline sociodemographic, clinical, and HRQoL parameters.
Participants
The sample selection followed the following inclusion criteria: age over 18 years, an anatomopathological diagnosis of malignant neoplasm at stages I, II, or III, and only newly diagnosed cancer patients undergoing curative chemotherapy on an outpatient basis. Patients in stage IV of the disease, those receiving exclusive palliative care, and those with more than one primary tumor were excluded. Initially, the medical records of clinical oncologists at Afecc-HSRC were analyzed to identify potential participants with a first-time indication for chemotherapy. After consultation with the clinical oncologist, patients were referred to the chemotherapy sector for an evaluation, during which they were approached and invited to participate in the study, in accordance with the eligibility criteria. During this initial approach, the research objectives were presented, and the patients signed the informed consent form (ICF).
Variables
A sociodemographic and clinical questionnaire was developed by the principal investigators based on an extensive systematic review of the literature on the topic, as well as based on the clinical-epidemiological variables in the Tumor Registration Form of the Afecc-HSRC; in addition, medical records were accessed to obtain more detailed information on clinical issues and cancer treatment, as well as to access blood count data.
The data in this questionnaire included: age, gender, self-reported skin color, education, marital status, time from diagnosis to start of treatment (in days), previous history and presence of comorbidities, tobacco use, alcohol use, International Classification of Diseases 10th Revision (ICD-10), and staging. In parallel with the collection of sociodemographic and clinical data, a blood sample was collected immediately before the administration of the first chemotherapy, and each participant donated a sample of peripheral venous blood (4 mL), collected using venipuncture in the cubital fossa (Monkhouse, 2001). For this purpose, vacuum tubes were used Vacutainer® Ethylenediaminetetraacetic acid, Becton Dickinson, Franklin Lakes, USA. To obtain the plasma, the biological samples collected were processed according to the following protocol: the Vacutainer tubes containing blood were centrifuged for 10 min at 4 °C, 581.2 g/2,000 r/min (Eppendorf Centrifuge 5810R). The plasma was then aliquoted using a pipette and transferred to storage microtubes. All the aliquoted material was stored in the freezer at −80 °C until the biomarker quantification stage. The analyses were processed by the Tommasi Laboratory, which is located on the premises of the Afecc-HSRC.
The ultra-sensitive CRP (US-CRP) was obtained using the MULTIGENT Vario ARCHITECT® assay (Abbott Laboratories, Abbott Park, IL), which is a latex immunoassay. The lower limit of detection is 0.01 mg/dL. The total coefficient of variation of the assay is ≤ 6%. For this study, values lower than 0.5 mg/dL were considered normal (Aguiar et al., 2013; Calixto-Lima et al., 2012; Martins, 2013). It should be noted that the blood count was routine at the hospital and, therefore, the information was collected directly from the patients’ medical records at a date prior to the first chemotherapy infusion.
Data Sources/Measurement
Sociodemographic and clinical data were collected using a structured questionnaire designed by the principal investigators, incorporating variables from the Tumor Registration Form of the Afecc-HSRC. Additional clinical and treatment-related information was obtained directly from patient medical records.
The EQ-5D is a generic instrument for measuring HRQoL that allows the generation of a representative index of the value of an individual's health status. It is based on a classification system that describes health in five dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. Each of these dimensions has three levels of severity associated with it, corresponding to “no problems” (Level 1), “some problems” (Level 2), and “extreme problems” (Level 3) experienced or felt by the individual. Thus, this system makes it possible to describe a total of 35 = 243 distinct health conditions (EuroQol Group, 1990). The questionnaire has two versions that vary according to the number of options for grading the severity of each dimension: the EQ-5D-5L and the EQ-5D-3L (version used in our study; Rennen & Oppe, 2015). The questionnaire consists of a descriptive system and a visual analog scale. Parameters of different nationalities for health statuses were evaluated, allowing adjustments in terms of preferences of each population, including psychometrically validated for the Brazilian population (Ferreira et al., 2013). The visual analog scale, on the other hand, records the interviewee's self-perception of health on a scale in which his extremities are displayed as “best imaginable health status” and “worst imaginable health status” (Ferreira et al., 2013; Rennen & Oppe, 2015). This instrument, along with the sociodemographic and clinical questionnaire, was administered immediately prior to the first chemotherapy session for cancer patients.
In addition, inflammatory biomarkers were assessed through the quantification of US-CRP levels. Peripheral venous blood samples were collected prior to the first chemotherapy session using standardized venipuncture techniques. Samples were processed in the hospital laboratory using the MULTIGENT Vario ARCHITECT® assay (Abbott Laboratories), a latex immunoassay with a detection limit of 0.01 mg/dL and a coefficient of variation ≤ 6%. Blood count data were obtained from routine pretreatment laboratory tests documented in patient medical records.
Ethical Approval
The study was approved by the Research Ethics Committee of the Center for Health Sciences (CEP/CCS/UFES), under Opinion No. 4122342. The permission of the hospital (Afecc-HSRC) was also approved in the instances involved for data collection. After voluntarily expressing their desire to participate in the study, the participants signed an ICF, safeguarding the confidentiality of the data.
Bias
Selection bias was mitigated by applying strict inclusion and exclusion criteria to ensure a homogeneous sample of patients with newly diagnosed malignant neoplasms (stages I–III) undergoing curative outpatient chemotherapy. The identification of eligible patients was conducted through systematic review of the medical records at Afecc-HSRC, ensuring that only first-time chemotherapy cases were included.
Information bias was reduced by utilizing validated instruments and standardized methods for data collection. Sociodemographic and clinical data were obtained through a structured questionnaire based on the Tumor Registration Form of the Afecc-HSRC Cancer Registry and supplemented with data from patient medical records. Laboratory data, including inflammatory biomarker levels, were processed under stringent protocols using calibrated equipment and standardized assays, ensuring consistency and accuracy.
Study Size
The sample size was estimated based on the number of newly diagnosed cancer patients treated at the cancer referral hospital where the study was conducted as well as based on previous studies held at the aforementioned Hospital (Marcarini et al., 2024; Silva et al., 2023). Data from the hospital's 2022 caseload (N = 3,513) were used, as this was the most recent year unaffected by the COVID-19 pandemic. The following formula for finite populations was applied:
n = [N × Z² × p × (1 − p)]/[Z² × p × (1 − p) + e² × (N − 1)] (Borges et al., 2020)
where n is the calculated sample size, N is the population size, Z is the z-score for the desired confidence level (1.96 for 95%), p is the expected prevalence (set at 0.5 for maximum variability), and e is the acceptable margin of error (set at 5%). Assuming a 95% confidence level and a power of 80% (β = 0.20), the minimum required sample size was determined to be 84 patients. This sample allows the detection of a moderate effect size (Cohen's w ≈ 0.3) in bivariate analyses such as Fisher's exact test. Given the exploratory nature of the study and the primary objective of identifying sociodemographic, clinical, and inflammatory factors associated with HRQoL prior to chemotherapy, the sample size is considered adequate. It offers sufficient power to detect statistically and clinically meaningful associations that can inform early supportive care strategies for this patient population.
Statistical Methods
Categorical variables were presented based on absolute and relative frequencies, while numerical variables were summarized using measures of central tendency and dispersion. Fisher's exact test was used to investigate associations between variables, based on the small sample size and categorical nature of the variables. Regarding biomarker, the CRP was measured in plasma using a high-sensitivity immunoturbidimetric assay performed on an automated biochemical analyzer (e.g., Cobas 6000, Roche Diagnostics), following manufacturer instructions and analytical precision guidelines for clinical assays (Rifai et al., 2018; Thomas, 2005). The analytical sensitivity of the method is generally below 0.3 mg/L, with a reference range of 0.0–5.0 mg/L. All analyses were performed in duplicate, maintaining intra-assay coefficients of variation below 5%.
We also employed NLR and PLR, which are widely used as systemic inflammatory markers and have shown prognostic value in cancer—particularly breast cancer (Mathur et al., 2019; Guo et al., 2019; Zhang et al., 2017; Shi et al., 2017). NLR was calculated as the absolute neutrophil count divided by the absolute lymphocyte count and was classified as high when ≥3.0 and low when <3.0 (Guo et al., 2019; Shi et al., 2017). PLR was computed by dividing the absolute platelet count by the absolute lymphocyte count, and values >200 were classified as high, indicating poor antitumor activity and prognosis (Cao et al., 2019; Mathur et al., 2019; Ming-Sheng et al., 2022; Zhang et al., 2017; Shi et al., 2017).
The analyses were conducted using the R environment (4.3.2) and the RStudio software (2023.09.1 Build 494), considering a 5% significance level (Daniel & Cross, 2018).
To investigate factors associated with the QoL domains, binary logistic regression and ordinal regression models were employed, based on the nature of each outcome variable. All models included the following independent variables: age group, sex, presence of comorbidities, clinical stage, and CRP levels. Results were reported as odds ratios (ORs) with corresponding 95% confidence intervals (95% CI). A two-tailed significance level of .05 was used for all statistical tests.
Results
Table 1 shows sociodemographic and clinical characterization and the prior history of cancer patients with indications for outpatient chemotherapy. The mean age of the participants was 57.68 years and standard deviation was 11.81 years, and 72.62% were women. Regarding self-reported skin color, 40.4% were brown (parda), 38.10% White, and 11.90% Black; 48.18% had completed elementary school and the majority (51.19%) were married. The mean time from diagnosis to the start of treatment was 120 days, and 60.71% of the sample had comorbidities. In addition, 91.67% did not use tobacco and 84.52% did not use alcohol.
Sociodemographic, Clinical, and Past History Characteristics of Cancer Patients Undergoing Outpatient Chemotherapy (n = 84).

Regarding diagnoses, the types of malignant neoplasms included in this study were breast, colorectal, endometrium, bladder, penis, bronchi and lungs, sinuses, cardia, undifferentiated malignant neoplasm (Merkel's carcinoma), cholangiocarcinoma, ovary, stomach, malignant neoplasm of the soft tissues of the retroperitoneum and peritoneum, and esophagus. The most prevalent were breast tumors (57.14%), followed by colorectal cancer (22.62%). The most prevalent clinical stage was Stage II (57.14%), followed by Stage III (28.57%).
Table 2 illustrates the descriptive relationship of the information obtained from the complete blood count and biomarkers of cancer patients with indications for outpatient chemotherapy. The mean red blood cells were 4.3 million/mm3 (SD = 0.56), the mean leukocytes were 7.38 million/mm3 (SD = 2.57), the mean neutrophils were 66.36 million/mm3 (68.54%), typical lymphocytes were 28.46% (SD = 9.55), and platelets were 312 thousand/mm3 (SD = 108.47). The mean NLR was 2.91 thousand/mm3 (SD = 3.71), and the mean PLR was 175.81 thousand/mm3 (SD = 90.86). Most patients had low NLR (76.54%) as well as low PLR (72.84%), while the mean CRP levels of the participants were 6.83 mg/dL (SD = 18.84), and the majority (84.52%) of the sample had an increased CRP.
Blood Count Data and Inflammatory Biomarkers of Cancer Patients Receiving Outpatient Chemotherapy (n = 84).

Note. NLR = neutrophil-to-lymphocyte ratio; PLR = platelet-to-lymphocyte ratio; CRP = C-reactive protein.
Figure 1 illustrates the distribution of inflammatory biomarkers—NLR, PLR, and CRP—among cancer patients scheduled for outpatient chemotherapy.

Distribution of Inflammatory Biomarkers Among Cancer Patients Undergoing Outpatient Chemotherapy (n = 84). Horizontal bars Represent the Absolute Number and Percentage of Patients Categorized as Having Low or High Neutrophil-to-Lymphocyte Ratio (NLR), Platelet-to-Lymphocyte Ratio (PLR), and Normal or Increased C-Reactive Protein (CRP). Most Patients Exhibited a Low NLR (76.5%) and PLR (72.8%), While the Majority (84.5%) had Increased CRP Levels, Indicating a Prevalent Inflammatory State Even Before Chemotherapy Initiation.
Table 3 shows the information obtained from the EQ-5D-3L questionnaire, based on its five domains, and the final score. For Domain 1, referring to Mobility, 25% of the participants reported having problems walking. For Domain 2, which concerns self-care, 3.57% had some problems washing or dressing. About the usual activities present in Domain 3 of the instrument, 17.86% had some problems performing usual activities and 3.75% were unable to perform usual activities. In Domain 4, related to pain/discomfort, the participants who had moderate or extreme pain/discomfort amounted to 22.62% and 4.76%, respectively. In Domain 5, on anxiety/depression, most of the sample (52.38%) were moderately anxious or depressed, while 14.29% were extremely anxious or depressed. The final average score of the questionnaire was 77.79.
Dimensions of the EQ-5D-3L Questionnaire of Cancer Patients Receiving Outpatient Chemotherapy (n = 84).

Note. QoL = quality of life.
Table 4 shows the results of the association tests of the EQ-5D-3L questionnaire with sociodemographic and clinical variables and biomarkers. Domains 1 and 5 showed statistical significance, and Domain 1 referring to mobility, when impaired, was associated with the presence of patient comorbidities (p = .009), while Domain 5, which deals with anxiety/depression, was associated with the gender variable (p = .005), indicating that females experienced more symptoms of anxiety and depression than males.
Associations Between the EQ-5D-3L Questionnaire and Sociodemographic and Clinical Variables in Cancer Patients Receiving Outpatient Chemotherapy (n = 84).

Note. *Fisher's exact test.
Table 5 shows the results of this comparative analysis between NLR and PLR groups of patients, which revealed that there were no significant differences in terms of mobility (D1), self-care (D2), usual activities (D3), pain/discomfort (D4), and anxiety/depression (D5).
Association of the EQ-5d-3L Questionnaire with Inflammation Biomarkers in Cancer Patients Receiving Outpatient Chemotherapy (n = 84).
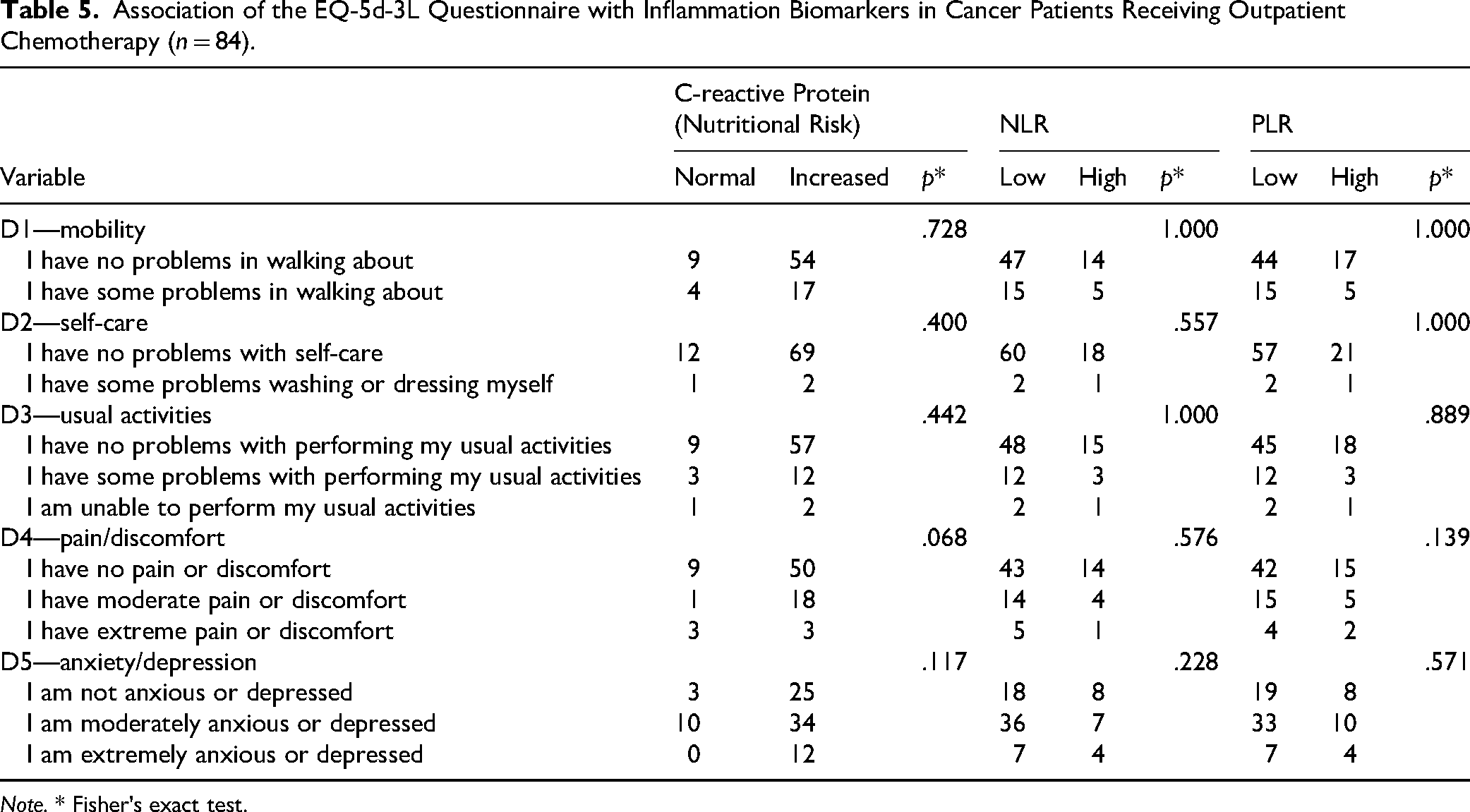
Note. * Fisher's exact test.
Table 6 presents the adjusted regression models for the EQ-5D-3L questionnaire domains. Domain D1—Mobility was analyzed using binary logistic regression, with the presence or absence of mobility problems as the dependent variable. Domains D3—Usual Activities, D4—Pain/Discomfort, and D5—Anxiety/Depression, which are ordinal in nature, were analyzed using ordinal regression with the proportional odds model. Domain D2—Self-Care was excluded from multivariate modeling due to a substantial imbalance in category distribution.
Regression Models for the Dimensions of Quality of Life.
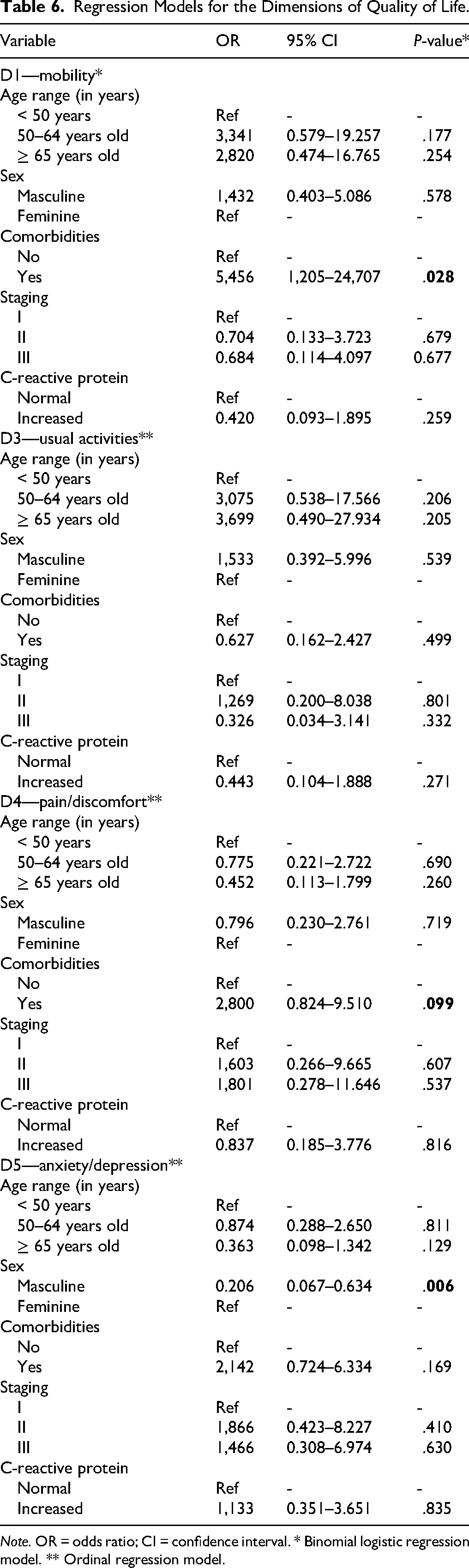
Note. OR = odds ratio; CI = confidence interval. * Binomial logistic regression model. ** Ordinal regression model.
The presence of comorbidities was significantly associated with impaired mobility. Individuals with comorbid conditions were 5.46 times more likely to report mobility difficulties compared to those without comorbidities (OR = 5.456; 95% CI [1.205–24.707]; p = .028). No other variable showed a statistically significant association with mobility.
A trend toward an association between clinical stage and the presence of pain or discomfort was observed. Patients in more advanced stages were more likely to report pain, although this association did not reach statistical significance (OR = 2.800; 95% CI [0.824–9.510]; p = .099).
Male sex was significantly associated with lower levels of anxiety and depression. Men were 79.4% less likely to report higher levels of these symptoms compared to women (OR = 0.206; 95% CI [0.067–0.634]; p = .006). Other variables, including age, comorbidities, clinical stage, and CRP levels, were not significantly associated with this domain.
Chart 1 provides an integrated summary of the study's main findings, encompassing sociodemographic and clinical characteristics, biomarker profiles, QoL domains, and significant statistical associations.
Integrated Summary of Main Study Findings.

Note. NLR = neutrophil-to-lymphocyte ratio; PLR = platelet-to-lymphocyte ratio; CRP = C-reactive protein; OR = odds ratio.
Discussion
QoL is a subjective assessment made by the patient concerning his general well-being in the context of the culture in which he lives and his goals, expectations, standards, and concerns (EuroQol Group, 1990). For a more comprehensive understanding, this study investigated the HRQoL of cancer patients with indication for chemotherapy treatment, in addition to potentially associated factors such as sociodemographic and clinical variables and inflammatory biomarkers that may impact their HRQoL. In general, our findings confirm that HRQoL was associated with clinical and sociodemographic characteristics of cancer patients undergoing chemotherapy.
Patients with comorbidities may have their HRQoL affected in the mobility dimension. We found that gender can influence the state of anxiety and depression, suggesting possible disparities in the perception of these aspects of health between men and women. In our study, when compared with males, we found that women reported a higher frequency of anxiety/depression on a moderate to extreme scale. However, these findings should be interpreted with caution, as the observed association with gender may not be genuine due to the unequal distribution of females and males in the overall sample. In different studies, authors have reported that the overall QoL of patients before starting cancer treatment is an important predictor of survival (Lewandowska et al., 2020; Li et al., 2012; Montazeri et al., 2001). The parameters used to assess QoL may be impaired throughout cancer treatment. The diagnosis of malignant neoplasm can cause severe anxiety, feelings of insecurity, and danger. One study showed that 38% of participants were in shock when they received the news of the diagnosis, and 22% had a nervous breakdown (Lewandowska et al., 2020). Another 35% experienced anger, 12% shame, and 30% developed depression. In addition, other studies reported that chronic illnesses can cause guilt, loss of control, anger, sadness, and confusion (Stanton et al., 2007; Zabora et al., 2001).
The Pain/Discomfort dimension (D4) was the only domain that demonstrated a trend toward statistical significance (p = .068) in relation to CRP levels. Although this result does not meet the conventional threshold for significance, it suggests a potential association between systemic inflammation and increased pain perception. This relationship is biologically plausible, given CRP's role as a systemic inflammatory marker. Prior studies, such as Amano et al. (2021), have reported significant correlations between elevated CRP levels and higher pain intensity in patients with advanced cancer, supporting this association. In the present study, the absence of statistical significance may be attributable to the limited sample size, the predominance of early-stage cancers, and the timing of data collection, which occurred prior to the initiation of chemotherapy.
No statistically significant association was observed between the inflammation biomarkers (NLR and PLR) and the health domains assessed by the EQ-5D-3L questionnaire. This lack of association suggests that the inflammation biomarkers may not have been affected by the health status of the patients since it precedes the initiation of chemotherapy treatment, however it's important to consider the sample size and timing of biomarker collection, predominance of early-stage cancers. It is important to note that blood collection was performed prior to the administration of the first chemotherapy. Such findings point out the complexity of the relationship between these biomarkers and the subjective perception of health, highlighting the need to consider multiple factors in the interpretation of clinical outcomes (Lewandowska et al., 2020).
In this sense, research confirms that inflammatory biomarkers are an accessible method that can be easily obtained from the complete blood count and that it can monitor tumor-induced inflammatory or immune reactions, playing a crucial role as prognostic factors in various types of cancer, potentially more effective than CRP (Forget et al., 2015; Hung et al., 2011; Kim et al., 2020; Łukasiewicz et al., 2021; McMillan, 2009). Early detection of indicators of poor HRQoL, such as inflammatory reactions, in patients with advanced cancer, can make it possible to identify patients suitable for early referral to palliative and supportive care, leading to better HRQoL outcomes (Abrahão et al., 2019; Daly et al., 2020; Lima et al., 2023; Nunes et al., 2017).
However, in our study, no associations were found with inflammatory biomarkers, while scientific evidence has indicated that peripheral inflammatory markers such as interleukin-6 (IL-6), CRP, and tumor necrosis factor (TNF-α) are associated with depressive symptoms in various cancer settings (McFarland et al., 2022). Undeniably, CRP, an acute phase protein, has previously been evaluated for its relationship to a variety of symptoms, including pain, fatigue, sleep disturbances, and depression, reflecting innate inflammation (Aamdal et al., 2022; Kwekkeboom et al., 2018). Studies have identified a positive association between systemic inflammation and worse HRQoL through CRP (Aamdal et al., 2022; Daly et al., 2020; Manigault et al., 2021; McFarland et al., 2021). High inflammation, characterized by CRP ≥10 mg/dL, is associated with significantly poorer HRQoL, covering aspects such as global health, functionality, social interaction, fatigue, pain, and loss of appetite (Aamdal et al., 2022).
The cutoff values adopted for CRP, NLR, and PLR were based on thresholds commonly reported in the literature with clinical relevance and prognostic significance in oncological populations (Cao et al., 2019; Mathur et al., 2019; Ming-Sheng et al., 2022; Zhang et al., 2017; Shi et al., 2017). However, in our study, the lack of statistically significant associations between these biomarkers and HRQoL domains may reflect several limitations. First, the sample size may not have provided sufficient statistical power to detect modest associations, especially given the variability in biomarker levels and patient-reported outcomes. Second, residual confounding by unmeasured variables—such as nutritional status, functional capacity, or concurrent treatments—could have attenuated potential associations. Additionally, the sample consisted of newly diagnosed cancer patients who had not yet initiated chemotherapy; therefore, systemic inflammation may not have reached the levels typically observed during or after treatment, when disease progression, cytotoxic effects, and cachexia are more pronounced. Also, the predominance of early-stage cancer in our sample (stages I–II) may have contributed to a relatively preserved inflammatory profile, limiting the ability to detect strong correlations with HRQoL.
Our results differ from findings reported in studies involving patients with more advanced disease or those undergoing treatment. For example, Laird et al. (2016) demonstrated that elevated systemic inflammation, as measured by the modified Glasgow Prognostic Score, was independently associated with poorer HRQoL in patients with advanced cancer, regardless of performance status. Similarly, Zhang et al. (2023) found that higher PLR and NLR values were significantly associated with increased cancer-related fatigue and lower QoL in patients receiving chemotherapy or radiotherapy.
A possible explanation for the discrepancy lies in the timing of HRQoL assessment. While previous studies often assessed patients during or after treatment—when inflammatory responses and treatment-related toxicity are heightened—our study captured baseline HRQoL and inflammatory markers prior to chemotherapy initiation. These findings suggest that although inflammatory biomarkers may be valuable in predicting HRQoL deterioration over the course of treatment, their predictive utility in the pretreatment phase, particularly among early-stage patients, may be limited. Future longitudinal studies are warranted to explore whether baseline inflammation can predict subsequent declines in specific HRQoL domains during treatment.
When it comes to comparing HRQoL with the demographic characteristics of cancer patients, significant disparities have been observed in the levels of physical, functional, and social well-being among patients of different age groups (Ayub et al., 2023). In our study, there was no significant relationship between age group and negative implications for HRQoL. Besides, there is a relationship between QoL and the frequency of chemotherapy cycles (CT) (Ayub et al., 2023). Patients undergoing CT may have an average QoL even in the first cycles (Ayub et al., 2023), but it was not possible to estimate this association in our study since it is of the cross-sectional type.
In a prospective multicenter study, the authors explain the benefits of using tools that assess QoL, which can be a predictor of improved survival in >6 months (Anwar et al., 2014). In comparison, blood biomarkers or radiological imaging biomarkers can be invasive and costly, while measuring HRQoL is fast, free, and allows patients to understand their health status and contribute significantly to their care. In addition, blood biomarkers do not demonstrate predictive information in such a short period, such as 3 to 6 weeks (Anwar et al., 2014). Patients have typically already experienced toxicity and increased morbidity over months before traditional markers can predict negative outcomes (Anwar et al., 2014). On the other hand, the results of our study did not show relevance in the association between the biomarkers and the QoL assessed. However, there was statistical significance in the association of individuals who had some comorbidity and who also had mobility problems.
Anxiety and depression to some degree were the most negatively reported dimensions in the EQ-5D-3L questionnaire. These are typical symptoms associated with a cancer diagnosis (Ayub et al., 2023; Hamood et al., 2018). Although the HRQoL assessment took place before chemotherapy, the results found may be related to the prognostic awareness of the patients, as well as the symptoms experienced until the diagnosis and start of treatment. Ensuring an improvement in HRQoL is essential to offer holistic and excellent care to people facing a cancer diagnosis (Pinheiro & Rosenberg, 2023). It is also important to note that assessing HRQoL at a single point in time may not fully encompass the patient's experience throughout the entire course of cancer treatment (Pinheiro & Rosenberg, 2023).
The anxiety/depression variable associated with gender was statistically significant with results pointing to women having greater emotional challenges with complaints of anxiety and moderate depression. Aligned with this finding is a study that showed the existence of mental impairment with reports of restlessness and tension with moderate to severe expression related to the stages of discovery of the disease and the therapy administered (Grusdat et al., 2022). In addition, a study conducted in Spain showed that men have greater stoicism, suppression of all emotional expression, and increased tolerance to suffering, which leads them to seek coping strategies that reveal a greater understanding of psychological suffering (Gomez et al., 2022). It is important to highlight that emotional well-being is affected at all ages and genders, which worsens patients’ QoL (Velasco-Durantez et al., 2024).
A systematic review of the QoL in breast cancer patients undergoing neoadjuvant chemotherapy demonstrated in some studies that depression was more severe during neoadjuvant chemotherapy when compared to before and after treatment, and anxiety was more severe before chemotherapy treatment (Zhao et al., 2022). In our study, it was observed that before the first cycle of treatment, 52.38% of the patients were also moderately anxious or depressed. Besides, this review concluded that there were no significant changes in overall HRQoL in the period before and after neoadjuvant chemotherapy treatment in patients with breast cancer. However, patients’ HRQoL decreased during chemotherapy cycles due to drug toxicities, and after treatment HRQoL tended to increase as a result of decreased adverse effects, corroborating the literature (Zhao et al., 2022).
The finding that comorbidities increase the likelihood of reporting difficulty in walking by 5.46 times confirms and reinforces the finding of this study in cancer survivors, according to which the presence of additional conditions (such as cardiometabolic) increases the risk of functional deficit. This effect is plausible when considering that multiple pathologies can aggravate structural lesions, reduce physiological reserve and amplify symptoms such as pain, fatigue and muscle weakness. Therefore, the rehabilitation approach in oncology needs to go beyond focusing on cancer and integrate the appropriate management of comorbidities, prevention of functional decline and promotion of active mobility, in order to reduce disparities and improve QoL in the posttreatment period (Cao et al., 2024).
The trend observed between advanced clinical staging and an increased likelihood of pain (OR = 2.800; 95% CI [0.824–9.510]; p = .099) suggests that patients with more advanced tumors may experience more intense pain, although the association did not reach statistical significance. This finding aligns with existing clinical literature indicating that pain and discomfort typically increase as tumors progress and involve deeper or more extensive tissue, particularly in cases of infiltrative or compressive neuropathy. Despite the wide CI and p-value above .05, the elevated OR points to a clinically relevant trend that warrants attention. This highlights the importance of active pain surveillance in patients with advanced-stage cancer, as well as the need for studies with greater statistical power to confirm this association and inform evidence-based strategies for early and effective symptom management (Concepción Pérez et al., 2024).
The finding that men were 79.4% less likely to report high levels of anxiety and depression (OR = 0.206; 95% CI [0.067–0.634]; p = .006) is consistent with prior evidence indicating a higher prevalence of depressive symptoms among women in oncology settings. Literature, including a meta-analysis involving 6,317 cancer patients, suggests that this disparity reflects both biological mechanisms (e.g., immune response, treatment-related toxicity) and psychosocial dimensions (e.g., social roles, emotional expression). The fact that other variables—such as age, comorbidities, clinical stage, and CRP levels—were not significantly associated with anxiety/depression further underscores the central role of gender. These findings support the implementation of gender-sensitive psychosocial interventions, with targeted screening and early psychological support particularly for women, while also addressing gender-specific manifestations of psychological distress in men, in order to reduce disparities in mental health outcomes throughout the cancer care continuum (Vitale et al., 2024).
Strengths and Limitations
The restrictions of our research include its cross-sectional methodological design, which makes it impossible to establish the cause–effect relationships investigated here. In addition, we consider it a limitation that the study was carried out at a single point in time, its cutoff point, before chemotherapy treatment, thus making it impossible to assess HRQoL after chemotherapy treatment. In this study, it was not possible to estimate the association between HRQoL and chemotherapy cycles because the collection took place before administering the first cycle of chemotherapy, thus increasing the importance of longitudinal studies for the monitoring and evaluation of patients’ HRQoL throughout the chemotherapy cycle.
Despite its limitations, this pioneering study in Brazil, using an instrument validated for the Portuguese of Brazil, the EQ-5D-3L, demonstrated that for newly diagnosed cancer patients with chemotherapy indication, domain 5 was already compromised for more than half of the cancer patients who reported being moderately anxious and depressed. It is noteworthy that the early detection of indicators of poor HRQoL, associated with inflammatory biomarkers levels, in cancer patients, can enable risk stratification of patients, to design effective personalized interventions for a better prognosis. Patients undergoing chemotherapy may have an average QoL even in the first few cycles (Ayub et al., 2023).
Implications for Practice
Cancer imposes a substantial burden on patients’ physical, psychological, and family well-being. Therefore, the use of validated instruments to assess HRQoL is essential to promote better adherence to chemotherapy and guide supportive care. In clinical practice, oncologic evaluations often focus on the frequency of adverse effects and laboratory biomarkers, while the psychosocial consequences of the diagnosis and treatment complications are underrecognized. However, the decline in health status commonly observed in patients undergoing invasive cancer treatments significantly reduces their functional capacity, ultimately leading to poorer HRQoL (Nunes et al., 2024).
This study represents a significant advancement by demonstrating the feasibility and clinical value of incorporating HRQoL assessments—particularly by nurses—into routine care within High Complexity Oncology Care Centers. These assessments can inform the development of personalized nursing interventions tailored to the diverse profiles and needs of cancer patients undergoing chemotherapy (Amorim & Lopes-Júnior, 2021; Lopes-Júnior, 2021a).
Furthermore, they offer a critical opportunity to identify patients at increased risk for physical and emotional decline early in the treatment trajectory. The use of brief, validated instruments such as the EQ-5D-3L, in combination with inflammatory biomarkers (CRP, NLR, PLR), may support a more comprehensive and individualized evaluation, enhancing the quality and responsiveness of care.
Nurses in High Complexity Oncology Care Centers are uniquely positioned to lead the implementation of targeted, evidence-based interventions tailored to patient needs, including symptom management, psychosocial support, and referrals to physical therapy or nutritional services. These actions can be initiated proactively, even before chemotherapy, to enhance treatment readiness, reduce the burden of symptoms, and improve overall QoL.
By emphasizing early detection and multidisciplinary care planning, this study supports a person-centered approach in oncology and reinforces the critical role of nurses in delivering comprehensive, responsive, and impactful cancer care from the outset of treatment.
Conclusions
In summary, this study provides a novel contribution by examining HRQoL in newly diagnosed cancer patients immediately prior to the initiation of outpatient chemotherapy. Patients with newly diagnosed cancer already exhibit reduced functional capacity and compromised HRQoL prior to initiating chemotherapy. Multivariate analysis revealed that comorbidities significantly increase the likelihood of impaired mobility, while female sex is independently associated with higher levels of anxiety and depression. Although systemic inflammation, as measured by CRP, did not reach statistical significance, a clinically relevant trend toward greater pain perception was observed. These findings highlight the need for early, tailored multidisciplinary interventions—particularly for individuals with comorbidities and for women—to address both physical limitations and emotional distress at the outset of oncologic treatment. Future studies with larger samples and longitudinal follow-up are warranted to confirm these associations and guide more personalized supportive care strategies.
Footnotes
Acknowledgments
The authors would like to thank the Afecc-Hospital Santa Rita de Cássia for supporting this research.
ORCID iDs
Author Contributions
All the authors made substantial contributions to the manuscript. LCLJ conceived and designed the study. EJCSM, RMP, SIPCS, NSA, VOS, JG, WRG, and KZN were responsible for data collection; WRG and LCLJ analyzed data; EJCSM, RMP, SIPCS, NSA, VOS, JG, WRG, and KZN drafted the manuscript; and LCLJ supervised the study and also made critical revisions on the paper. All the authors have approved the final version of this manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received funding from the Espírito Santo Research and Innovation Support Foundation (Fundação de Amparo à Pesquisa e Inovação do Espírito Santo—FAPES; FAPES N° 03/2021—UNIVERSAL; Process No. 2021-5BDLS) and from the National Council for Scientific and Technological Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPq; Research Productivity Fellowship—(PQ2), Process No. 311427/2023-5) to Dr Luís Carlos Lopes-Júnior.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
