Abstract
Introduction
Prediabetes is a condition that, if left unaddressed, can lead to various complications, such as neuropathy, retinopathy, and nephropathy. Among the critical factors contributing to the development of type 2 diabetes mellitus, dietary choices stand out as particularly significant.
Objective
Consequently, our objective is to examine the latest research findings concerning dietary consumption and its impact on glycemic control in individuals with prediabetes.
Methods
A literature review of randomized controlled trials was performed using databases such as PubMed, Scopus, and ScienceDirect with searches conducted from January 2019 to 2023. The primary reviewer assessed the quality of the selected studies for bias risk using the Joanna Briggs Institute critical appraisal method for randomized controlled trials. Initially, 975 articles were identified through the search, but after applying the inclusion criteria, only 9 articles were ultimately selected.
Results
The review found that a carrageenan-free diet, yogurt with Lactobacillus plantarum OLL2712, Allium hookeri extract (AHE), and delta-tocotrienol improve HbA1C levels. However, salmon, zinc supplement, and balanced deep-sea water were not effective on HbA1C. In addition, studies on the effectiveness of vitamin D in controlling blood glucose levels are inconsistent.
Conclusion
Nurses can enhance patient outcomes through collaborative efforts to create individualized dietary strategies. These strategies may encompass the adoption of a carrageenan-free diet, the inclusion of L plantarum OLL2712-enriched yogurt, the utilization of AHE, and the integration of delta-tocotrienol into the dietary plan. This approach is particularly applicable to ambulatory care nurses, health supervisors, and primary care providers.
Introduction
According to the Centers for Disease Control and Prevention (CDC), approximately 96 million American adults and more than 1 in 3 people have prediabetes (Centers for Disease Control and Prevention [CDC], 2022). According to a published study, the prevalence of prediabetes in adults aged 18 years and older in the United States stands at 33.9%, which equates to approximately 84.1 million adults. Furthermore, the research indicated a higher prevalence among older adults, males, and individuals with obesity (Bullard et al., 2018). In another study that examined data from 49 countries, the worldwide prevalence of prediabetes was estimated to be approximately 47%, accounting for nearly half of the adult population. Additionally, this research revealed a higher prevalence of prediabetes in low- and middle-income countries (Zhang et al., 2020). Nonetheless, these studies underscore the importance of recognizing prediabetes as a substantial public health concern and emphasize the necessity for concerted efforts in both the prevention and treatment of this condition.
According to the American Diabetes Association (ADA), prediabetes is a condition in which blood glucose levels are higher than normal but not high enough to be diagnosed as type 2 diabetes mellitus (T2DM) (American Diabetes Association [ADA], 2022). As per ADA, prediabetes is defined by an impaired fasting glucose (IFG) level ranging from 100 to 125 mg/dL, impaired glucose tolerance (IGT) with values between 140 and 199 mg/dL measured 2 hours after a glucose challenge, or a glycated hemoglobin (HbA1c) level within the range of 5.7%–6.4% (ADA, 2022). Furthermore, the study established that prediabetes constitutes a risk factor for the subsequent development of both T2DM and cardiovascular disease (ADA, 2022; Sims et al., 2021). Prediabetes is a condition that can lead to a number of complications if left untreated (Awuchi et al., 2020). A previous study found that people with prediabetes have a 15% higher risk of cardiovascular disease than people with normal blood glucose levels (Brannick & Dagogo-Jack, 2018; Mando et al., 2021). In addition, high blood glucose levels can damage nerves throughout the body, which is called neuropathy. One study found that prediabetes is associated with an increased risk of neuropathy (Kirthi et al., 2021). Moreover, prediabetes has been positively associated with the risk of renal dysfunction and an increased likelihood of developing kidney disease (Echouffo–Tcheugui et al., 2016; Chen et al., 2020). Furthermore, elevated blood glucose levels have the potential to harm blood vessels within the eyes, leading to visual impairments and vision-related issues (Priyadarshi et al., 2022). Diabetic retinopathy was more common in individuals with IGT and also occurred in 6.3% of Asian Indians with prediabetes (Rajalakshmi et al., 2022).
Management of prediabetes usually involves lifestyle changes, such as a modified diet, increased physical activity, and weight loss (Jadhav et al., 2017). Research revealed that adopting a dietary approach characterized by low fat, high fiber, adherence to the Mediterranean diet, and a plant-based focus, along with incorporating moderate exercise, effectively deterred or postponed the onset of T2DM in individuals with prediabetes (Santos et al., 2012; Satija et al., 2016). ADA suggests a dietary approach that prioritizes a high intake of fiber, whole grains, fruits, and vegetables, while keeping saturated and trans fats at a minimum as a treatment strategy for prediabetes (ADA, 2022). An additional research investigation revealed that engaging in physical activity, encompassing both aerobic exercises and resistance training, has the potential to enhance glucose tolerance and increase insulin sensitivity among individuals with prediabetes (Bouchard et al., 2012). Furthermore, when compared to a placebo, metformin demonstrated a notable reduction in diabetes incidence by 31% (95% CI 17%–43%), whereas lifestyle intervention exhibited a substantial reduction in incidence by 58% (95% CI 48%–66%). (Aroda & Ratner, 2018). Additionally, a recent comprehensive review of the literature has demonstrated that lifestyle interventions, frequently involving consistent dietary counseling and guidance on physical activity, possess the capacity to postpone or forestall the emergence of T2DM in prediabetes (Howells et al., 2016). The findings from these studies indicate that a blend of lifestyle adjustments, dietary modifications, and physical activity could prove to be efficacious in managing prediabetes and thwarting the onset of T2DM. Hence, it is imperative to promptly identify and address prediabetes to avert the progression of severe complications.
Dietary elements stand as a primary driver in the emergence of T2DM among individuals with prediabetes (Nathan et al., 2007; Roncero-Ramos et al., 2020), and an inappropriate diet can pose challenges for those seeking to manage their blood sugar levels effectively. Consuming a diet rich in calories, refined carbohydrates, and saturated as well as trans fats may play a role in fostering insulin resistance, a significant characteristic of prediabetes (Ley et al., 2014). Insulin resistance hinders the body's ability to utilize insulin efficiently, leading to increased levels of blood glucose (Martín-Timón, 2014). Conversely, a diet rich in fiber, whole grains, fruits, and vegetables, while being low in saturated and trans fats, may enhance insulin sensitivity and help regulate blood sugar levels in individuals with prediabetes. These food choices aid in decelerating the absorption of glucose into the bloodstream, thereby averting abrupt spikes in blood sugar levels (McMacken & Shah, 2017). Furthermore, a prior article recommended measures such as eliminating sugar-sweetened beverages, consuming a minimum of 5 servings of fruits and vegetables daily, and being mindful of portion sizes (using the palm for meat and the fist for other foods) (Magge et al., 2020). Nonetheless, it did not furnish precise dietary guidelines for lowering blood glucose levels in individuals with prediabetes. Consequently, our aim is to investigate the latest research findings concerning the influence of particular dietary regimens on glycemic control among individuals with prediabetes.
Methods
Search Strategy
The researcher conducted a comprehensive literature search across widely used online databases, including PubMed, Scopus, and ScienceDirect. The search encompassed English-language articles and human studies published between January 2019 and April 2023. The researcher used the following search terms: "prediabetes," "diet," "nutrients," "supplements," "glycemic control," "dietary behaviors," "impaired glucose tolerance," "impaired fasting blood glucose." The searching terms were combined as the Medical Subject Headings terms, text words, and Boolean operators. Additionally, other relevant publications were hand-searched in the reference lists for additional papers of interest.
Inclusion and Exclusion Criteria
To be considered for inclusion in this review, studies had to meet the following criteria: (a) They were full-text randomized controlled trials investigating the impact of foods, nutrients, or dietary supplements on the enhancement of HbA1C; (b) they involved individuals with a prediabetes diagnosis, with a focus on blood glucose parameters (IGT, IFG, or HbA1C); and (c) they were available in the English language. Studies that (a) encompassed children and adolescents, (b) involved patients with type 2 diabetes (T2DM), (c) lacked an HbA1C parameter, (d) utilized a food assessment questionnaire, or (e) incorporated research on probiotics were excluded from consideration. Our search identified a total of 13 previously published studies meeting these criteria.
Data Extraction
The author conducted a systematic screening process for the retrieved studies, categorizing them as either definitely meeting the inclusion criteria, potentially meeting the inclusion criteria, or clearly not meeting the inclusion criteria. This screening encompassed both the titles and abstracts, followed by a comprehensive examination of the complete articles using predefined inclusion and exclusion criteria. Articles that did not meet the relevance criteria were excluded from further consideration. Studies that passed the initial screening and were deemed sufficiently relevant underwent a secondary screening. During this stage, the authors thoroughly reviewed the full-text papers to determine if they met the established inclusion requirements. The included articles were then systematically documented in a structured table, which included information such as the author's name, year of publication, trial type, sample quality, sample size, research objective, intervention details, glycemic control aspects, type of food, supplement, or nutrient studied, research outcomes, and conclusions. A visual representation of the selection process is presented in Figure 1.
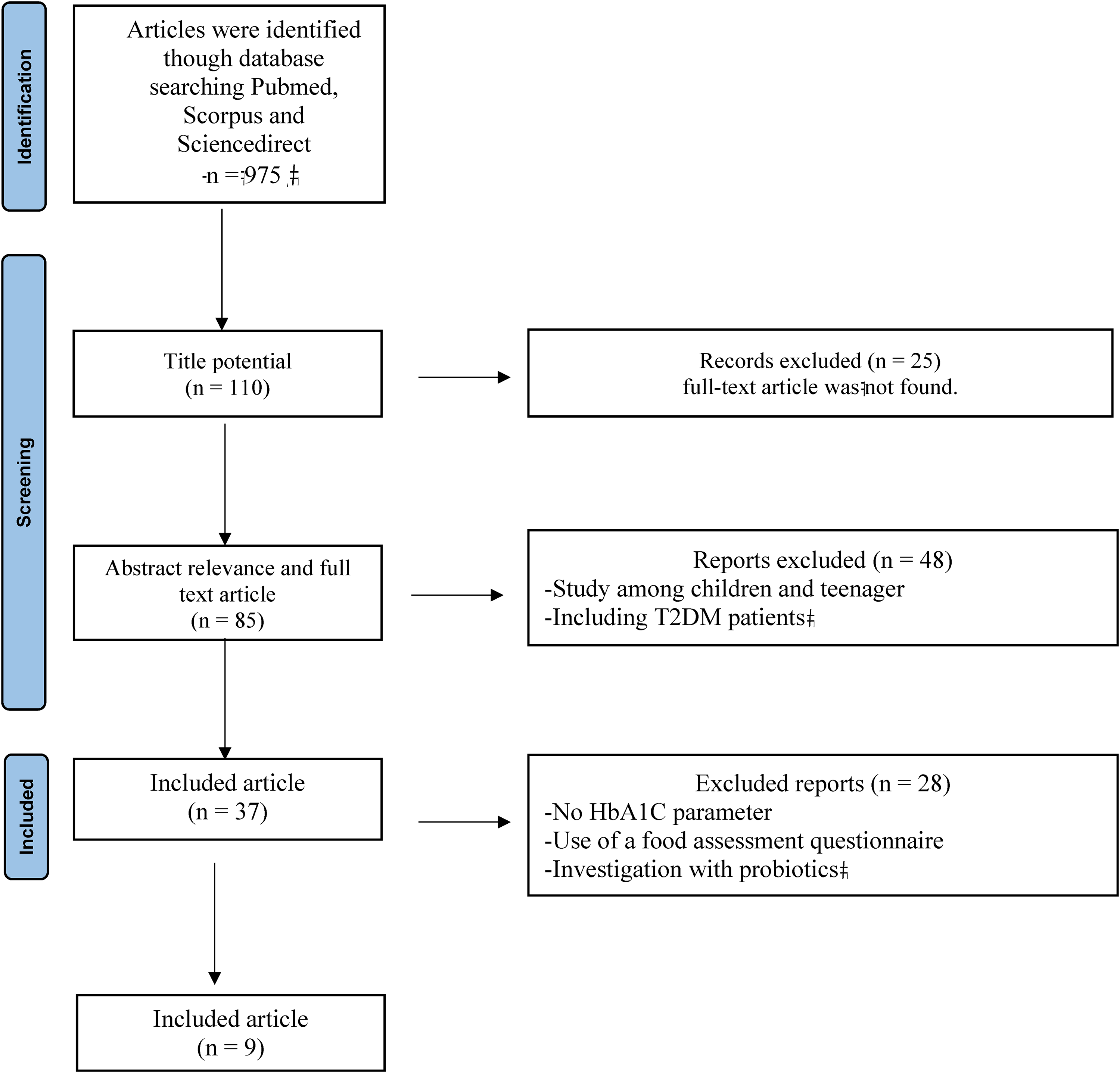
Summary of literature review process according to preferred reporting items for systematic reviews and meta-analyses flow diagram (Page et al., 2021).
Quality Appraisal
The studies included in this review were randomized controlled trials assessed for bias risk by the primary reviewer using the Joanna Briggs Institute critical appraisal method for Randomized Controlled Trials (Tufanaru et al., 2017). All articles successfully met at least 60% of the Joanna Briggs Institute critical appraisal criteria.
Results
In the initial database search, 975 studies were identified, and 9 articles were selected for review (Figure 1). These selected studies were randomized controlled trials with participant numbers ranging from 13 to 130. Recent research has highlighted the efficacy of various foods in reducing glycemic parameters, as summarized in 'Table 1' and 'Table 2'. The research explored different aspects of food, supplements, and nutrients, including vitamin D (Bhatt et al., 2020; Wallace et al., 2019), a no-carrageenan diet (Feferman et al., 2020), balanced deep-sea water (DSW) analysis (Ham & Shon, 2020), Allium hookeri extract (AHE) (Park et al., 2020), yogurt containing Lactobacillus plantarum OLL2712 (Toshimitsu et al., 2020), salmon (Hustad et al., 2021), delta-tocotrienol (Suleman et al., 2022), and zinc supplementation (Attia et al., 2022).
Summary of the Characteristics of the Selected Studies.
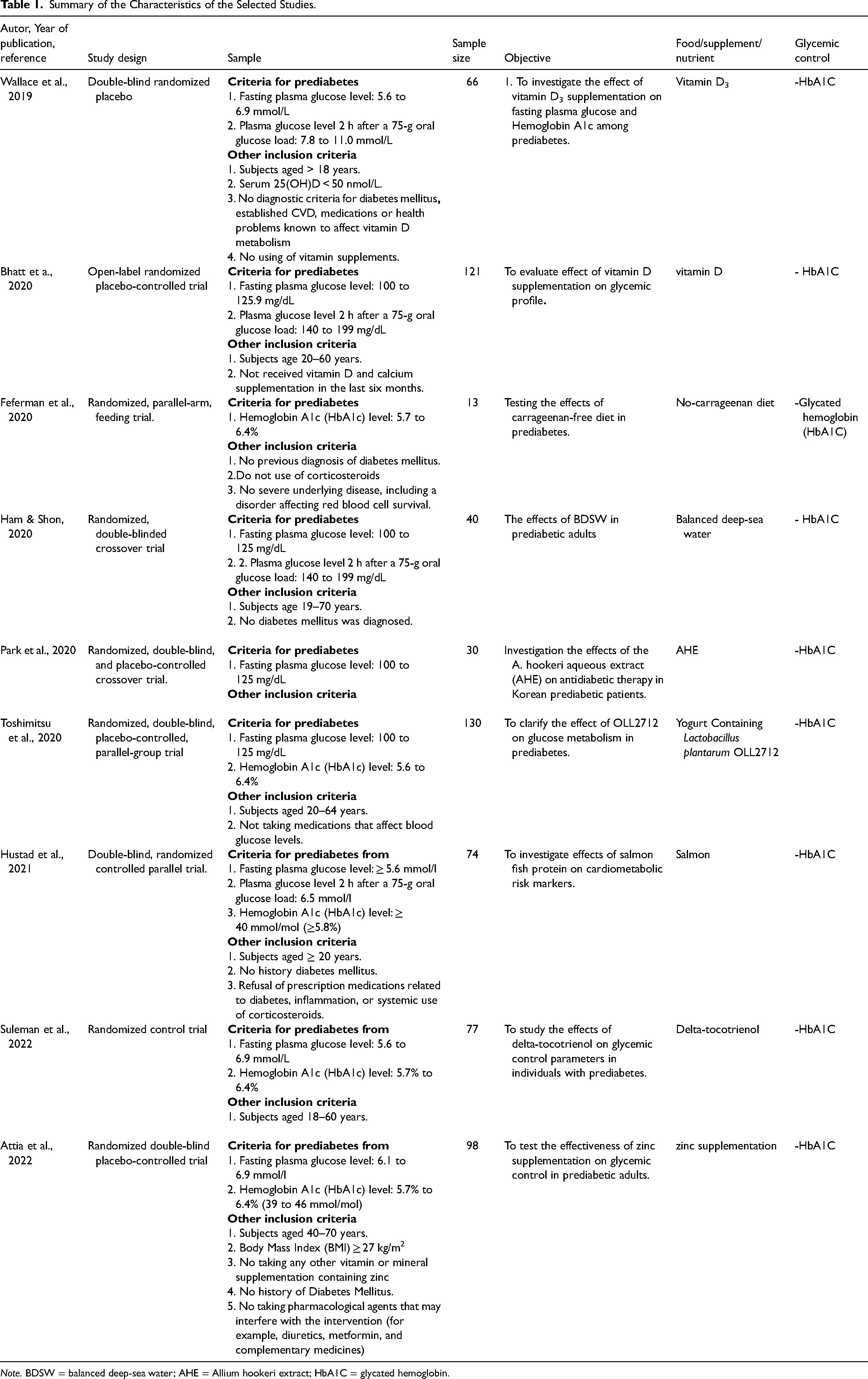
Note. BDSW = balanced deep-sea water; AHE = Allium hookeri extract; HbA1C = glycated hemoglobin.
Summary of the Intervention, Result and Conclusion of the Selected Studies.
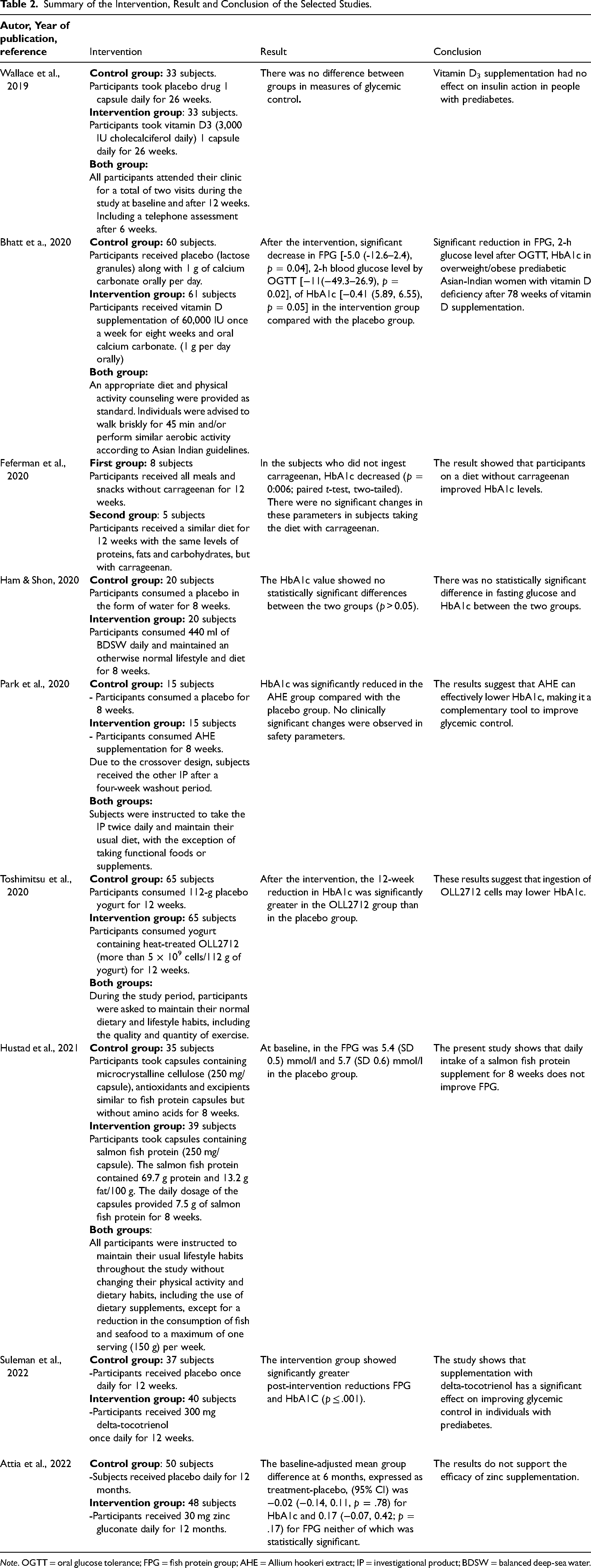
Note. OGTT = oral glucose tolerance; FPG = fish protein group; AHE = Allium hookeri extract; IP = investigational product; BDSW = balanced deep-sea water.
No-Carrageenan Diet
Carrageenans are sulfated polysaccharides, typically with a substantial molecular weight ranging from 100 to 1000 kDa. They are extracted from the cell walls of various species of red seaweeds, particularly from species such as Chondrus crispus, Gymnogongrus fucellatus, Soleria chordalis, Cystoclonium purpureum, and Kappaphycus alverezii. (Ahmadi et al., 2015; David et al., 2018; Martino et al., 2017; Martín-del-Campo et al., 2021; Neamtu et al., 2022; Noor, 2018).
Previous studies have shown that carrageenan exhibits various effects, such as antiviral properties (Rodríguez et al., 2014; Wang et al., 2012), anticoagulant activity (de Araújo et al., 2013; Liang et al., 2014; Yermak et al., 2012), antitumor effects (Prasedya et al., 2016; Yuan et al., 2011), antioxidant activity (Thevanayagam et al., 2014), and anti-inflammatory properties (Matsumoto et al., 2015). Furthermore, studies have shown that the oral ingestion of carrageenan induces inflammation in mice, leading to glucose intolerance. Mice exposed to tap water containing carrageenan developed glucose intolerance within six days. Carrageenan consumption can impair insulin signaling in animals, especially in mice, affecting hepatic phospho-insulin receptor substrate 1 and growth factor receptor-bound protein 10 in the liver (Bhattacharyya et al., 2012). Therefore, if carrageenan indeed contributes to glucose intolerance, adopting a carrageenan-free diet may potentially mitigate glucose intolerance and lead to reduced blood glucose levels. A recent randomized trial delved into the effects of a carrageenan-free diet on parameters such as HbA1c, oral glucose tolerance, and homeostatic model assessment-insulin resistance (HOMA-IR) among individuals with prediabetes. The study participants were divided into two groups: the first group followed a regimen devoid of carrageenan for a span of 12 weeks, while the second group adhered to a diet that included carrageenan. The outcomes indicate an amelioration in HbA1c levels among participants following the carrageenan-free diet (Feferman et al., 2020).
Yogurt Containing L Plantarum OLL2712
Chronic inflammation leads to insulin resistance, disrupting glucose and lipid metabolism in adipose, muscle, and liver tissues (Fazakerley et al., 2019). Inflammation triggers the release of pro-inflammatory substances, such as interleukin (IL-6) and monocyte chemotactic protein-1 (MCP-1). Lowering IL-6 and MCP-1 may prevent insulin resistance and affect blood glucose levels. IL-10 is known to suppress the production of pro-inflammatory cytokines, including MCP-1 and IL-6 (Lumeng et al., 2007; Winer et al., 2009). Previous research has demonstrated that stimulating IL-10 prevents insulin resistance and enhances glucose metabolism in obese mice (Cintra et al., 2008; Hong et al., 2009). L plantarum OLL2712 (OLL2712) is notable for its high ability to induce IL-10 production in murine immune cells (Toshimitsu et al., 2016). A study reported that taking Lactobacillus acidophilus NCFM for 4 weeks preserved insulin sensitivity in T2DM patients compared with placebo (Andreasen et al., 2010). Moreover, recent investigation revealed the effectiveness of L plantarum OLL2712 in individuals with prediabetes. Consuming OLL2712 cells significantly reduced fasting plasma glucose levels by mitigating chronic inflammation (Toshimitsu et al., 2018). Therefore, the researchers conducted a study to examine the effects of 12-week consumption of a test yogurt containing heat-treated OLL2712 cells compared with placebo yogurt on parameters associated with glucose metabolism in prediabetes. After the intervention, the 12-week reduction in HbA1c was significantly greater in the OLL2712 group than in the placebo group, suggesting that OLL2712 cell ingestion can reduce HbA1c levels (Toshimitsu et al., 2020).
Salmon
Fish, recommended by the Food-Based Dietary Guidelines, is a high-protein, high-fat food source (Cámara et al., 2021) Omega-3 polyunsaturated fatty acids (n-3 PUFAs) are found in three main forms: Alpha-linolenic acid, eicosapentaenoic acid, and docosahexaenoic acid (Shin & Chang, 2023). Earlier animal studies suggest that n-3 PUFAs positively impact renal function; however, limited evidence from human studies supports this claim (Ong et al., 2023). n-3 PUFAs are primarily sourced from seafood, such as salmon. An animal study indicated that incorporating a diet rich in hydrolyzed salmon protein significantly enhanced postprandial glucose regulation (Drotningsvik et al., 2016). Despite this, there is a scarcity of clinical trials involving protein supplements derived from fatty fish, particularly salmon, in humans. A recent study investigated the effects of consuming a fish protein supplement derived from salmon over an 8-week period. The study revealed that fasting plasma glucose (FPG) levels was 5.4 mmol/l (SD 0.5) in the fish protein group and 5.7 mmol/l (SD 0.6) in the placebo group. The research concluded that supplementing with salmon fish protein did not significantly impact glucose tolerance when compared to the placebo group (Hustad et al., 2021).
Allium Hookeri Extract
Allium hookeri Thwaites, a traditional herb belonging to the Liliaceae family, falls under the Alliaceae family's subgenus Amerallium. It is native to regions including Ceylon, Greece, Yunnan in southern China, Bhutan, Sri Lanka, and India (Sharma et al., 2011). Mainly used as a dietary supplement and medicine (Kim et al., 2016). Allium hookeri is notable for its elevated levels of total protein, sugar, fiber, phytosterol, ascorbic acid, and total phenolics, along with comparatively lower total fat content when compared to Allium cepa (Ayam, 2011). A. hookeri has been described as anti-inflammatory (Kim et al., 2017), antiobesity (Parket al., 2018), and osteoblastic (Park et al., 2016). Additionally, the root and leaf of Allium hookeri have demonstrated antidiabetic effects (Roh et al., 2016; Singh et al., 2013). In a recent study, researchers examined the impact of Allium hookeri by administering an extract of the herb over an 8-week period, comparing it to a placebo. Following the intervention, the group receiving the AHE exhibited a significant decrease in HbA1c levels compared to the placebo group (Park et al., 2020).
Vitamin D
Vitamin D, a crucial fat-soluble nutrient, plays a vital role in maintaining human health. Primary sources of vitamin D include natural foods such as fatty fish (sardines, herring, tuna, mackerel, salmon, and cod liver oil), egg yolk, shiitake mushrooms, and liver or organ meats (Antonucci et al., 2018; Herrick et al., 2019). Additionally, 7-dehydrocholesterol in the skin absorbs ultraviolet B rays from sunlight, triggering the body's vitamin D production (Herrick et al., 2019), constituting 90% of the body's intake (Antonucci et al., 2018). Vitamin D acts as a hormone, playing a significant role in maintaining calcium and phosphorus balance. (Holick, 2005; Mostafa, & Hegazy, 2015; Pittas et al., 2007). Its deficiency is associated with a higher risk of type 1 diabetes mellitus, (Holick, 2005; Pittas et al., 2007) and T2DM (Pittas et al., 2007). Vitamin D may impact peripheral insulin sensitivity through its effect on VDRs expressed in human skeletal muscle and adipose tissue cells (Alvarez & Ashraf, 2010) and glucose homeostasis. Vitamin D deficiency has been linked to insulin resistance, affecting beta cell function, insulin sensitivity, and inflammatory markers in obesity (Ashraf et al., 2009; Miao et al., 2020). Previous cross-sectional studies have associated vitamin D deficiency with T2DM development (Butler et al., 2020; Xiao et al., 2020). Cohort studies have shown an inverse association between serum levels of 25-OH-vitamin D [25(OH) D] and future risk of hyperglycemia, insulin resistance, T2DM, and diabetic complications (Ahmed et al., 2020; Mattila et al., 2007; Wang et al., 2020). Vitamin D supplementation has been found to reduce FPG, HbA1c, and HOMA-IR in T2DM with deficient vitamin D status (Farahmand et al., 2023). A recent study demonstrated that taking vitamin D supplementation of 60,000 IU once a week for 8 weeks, along with oral calcium carbonate (1 g per day orally), led to a significant decrease in FBG [−5.0 (−12.6–2.4), p = .04] and HbA1c [−0.41 (5.89, 6.55), p = .05] in the intervention group compared to the placebo group (Bhatt et al., 2020). Another study, involving daily intake of vitamin D3 (3000 IU cholecalciferol) in the form of one capsule daily for a period of twenty-six weeks, found no disparity in FPG and HbA1C measurements between the groups (Wallace et al., 2019).
Delta-Tocotrienol
Tocotrienols, which are natural subtypes of vitamin E consist of four homologs: alpha (α), beta (β), gamma (γ), and delta (δ) (Che et al., 2017). Previous animal studies have demonstrated that tocotrienol-rich fraction (TRF), particularly γ- and δ-tocotrienol, can significantly improve FPG and HbA1c levels by enhancing insulin sensitivity in animal models (Pang & Chin, 2019). However, human studies in this area are limited. A recent study investigated the effects of 300 mg δ-tocotrienol supplementation on glycemic control parameters in individuals with prediabetes. The intervention group exhibited significantly greater reductions in post-intervention FPG and HbA1c levels (p ≤ 0.001). This study suggests that supplementation with delta-tocotrienol has a notable effect on improving glycemic control parameters in individuals with prediabetes (Suleman et al., 2022).
Zinc Supplement
Zinc, a vital trace element in the human body, plays a crucial role in growth and development (Olechnowicz et al., 2018). It regulates chronic inflammation by decreasing inflammatory cytokines, mitigates oxidative stress by engaging in antioxidant enzyme synthesis, and acts as a catalyst for enzymes essential in lipid, carbohydrate, and protein metabolism (Barbara, & Mindikoglu, 2021). Additionally, zinc is essential for the regular synthesis, storage, and secretion of insulin within pancreatic beta cells (Chimienti, 2013; Yang et al., 2015). Previous studies have demonstrated that zinc supplementation improves glucose homeostasis in patients with diabetes mellitus (Jayawardena et al., 2012). Furthermore, the concentration of zinc shows an inverse relationship with insulin resistance (Ahn et al., 2014). Low zinc levels increase the risk of diabetes mellitus in women by 17% compared to women with adequate zinc levels (El Dib et al., 2015). A study revealed that zinc supplementation in prediabetes patients lowered blood glucose and insulin resistance while improving β-cell function (Ranasinghe et al., 2018). In a recent study, 48 individuals with prediabetes were given a daily dose of 30 mg of zinc gluconate over a 12-month period, and their outcomes were compared with a placebo group. The findings at the 6-month mark showed no statistically significant difference in HbA1C levels (treatment-placebo: −0.02, 95% CI −0.14 to 0.11, p = 0.78) and FPG (treatment-placebo: 0.17, 95% CI −0.07 to 0.42, p = 0.17) (Attia et al., 2022). contradict earlier research, further investigations involving larger patient populations are essential for future research.
Balanced Deep-sea Water
DSW, found at depths exceeding 200 meters, is distinguished by its purity, cold temperature, and rich concentration of distinctive minerals (Hwang et al., 2009; Miyamura et al., 2004). Previous research has demonstrated that balanced deep-sea water (BDSW), a blend of mineral extracts from DSW and desalinated water, possesses antidiabetic properties. It achieves this by curbing hyperglycemia, enhancing glucose intolerance, and augmenting glucose uptake in both type 1 and type 2 diabetic mice (Ha et al., 2013; Haase et al., 2014). In a prior human study, consuming 440 ml of BDSW daily while maintaining a normal lifestyle and diet for eight weeks led to a statistically significant difference between the two groups. However, the HbA1c value and FPG showed no statistically significant differences between the two groups (p > 0.05) (Ham & Shon, 2020).
Discussion
The aim of this study was to review the current evidence on dietary intervention aimed at reducing HbA1C in prediabetes. Several years ago, researchers endeavored to investigate the efficacy of various dietary patterns, incorporating vegetables, herbs, meats, nutrients, and acidic extracts from vegetables, plants, or herbs, in reducing blood glucose levels in individuals at risk of developing T2DM, particularly those with prediabetes. The carrageenan-free diet stands out as a compelling dietary pattern capable of disrupting insulin signaling in animals, including mice. Consequently, adopting a carrageenan-free diet can mitigate glucose intolerance and result in decreased blood glucose levels. However, there is limited research on the impact of a carrageenan-free diet in humans. The findings from this study indicate that a carrageenan-free diet has a beneficial effect on HbA1c levels in individuals with prediabetes (Feferman et al., 2020). Nonetheless, additional research is necessary to validate the effectiveness of a carrageenan-free diet, as this study was limited to a small sample size.
Furthermore, research is ongoing to explore the potential of dairy products, including yogurt containing L plantarum OLL2712, in reducing sugar levels. A prior study has already indicated the effectiveness of L plantarum OLL2712 in individuals with prediabetes (Toshimitsu et al., 2018). The findings from this study indicate that the ingestion of OLL2712 cells can lead to a reduction in HbA1c levels (Toshimitsu et al., 2020). Nevertheless, there is a limited body of research on the effectiveness of sugar reduction through yogurt containing L plantarum OLL2712, highlighting the need for more robust and reliable investigations. These should encompass studies involving a substantial and diverse participant pool.
Consuming fish may play a role in the prevention of developing T2DM (Nanri et al., 2011). Among fish species, salmon is notably the fattiest and the richest in n-3 PUFAs (Gormley et al., 2007). A diet abundant in hydrolyzed salmon protein has shown the ability to enhance postprandial glucose control (Drotningsvik et al., 2016). In a more recent investigation involving the consumption of a salmon fish protein supplement over an 8-week period, fasting serum glucose levels were 5.4 mmol/l (SD 0.5) in the fish protein group and 5.7 mmol/l (SD 0.6) in the placebo group (Hustad et al., 2021). Nevertheless, additional research is necessary to validate the effectiveness of salmon in managing blood glucose levels in individuals with prediabetes. Furthermore, certain vegetables have demonstrated potential in reducing blood sugar levels. One such example is Allium hookeri Thwaites, known for its antidiabetic properties. Both the root and leaf of Allium hookeri have been identified as having beneficial effects on diabetes management (Singh et al., 2013; Roh et al., 2016). The results of a recent study suggest that the AHE can effectively lower HbA1c levels, offering a complementary method to improve glycemic control (Park et al., 2020).
Vitamin D has the capacity to reduce blood sugar levels, and its deficiency is linked to an increased risk of elevated blood sugar and T2DM (Pittas et al., 2007). Insufficient vitamin D levels have been characterized as contributing to insulin resistance, impacting beta cell function and insulin sensitivity, especially in individuals with obesity (Ashraf et al., 2009; Miao et al., 2020). A recent study demonstrated that individuals who received vitamin D supplementation of 60,000 IU once a week for eight weeks, in addition to oral calcium carbonate (1 g per day orally), experienced reductions in both FPG and HbA1c levels when compared to the placebo group (Bhatt et al., 2020). Conversely, a trial involving the daily consumption of vitamin D3 (3,000 IU cholecalciferol daily) through a single capsule for a duration of 26 weeks did not reveal any significant differences in FPG and HbA1C measurements between the groups (Wallace et al., 2019). Nonetheless, there is a disparity in the outcomes of studies regarding the effectiveness of vitamin D in individuals with prediabetes. This discrepancy could be attributed to variations in dosage or experimental methods. Consequently, further research is warranted in the future.
Tocotrienols have shown significant potential, with research indicating that the TRF, especially γ- and δ-tocotrienol, can notably enhance FPG and HbA1c levels by improving insulin sensitivity in animal models (Pang & Chin, 2019). Consequently, tocotrienols have been investigated in human studies. The most recent study demonstrated significantly more substantial reductions in both FPG levels and HbA1c levels among individuals taking a 300 mg δ-tocotrienol supplementation. This study underscores the potential of delta-tocotrienol supplementation as a beneficial option for individuals with prediabetes (Suleman et al., 2022). Nonetheless, there is a scarcity of studies in this area, highlighting the necessity for additional research in the future.
Furthermore, previous research has suggested that zinc supplementation can improve glucose homeostasis in individuals with diabetes mellitus (Jayawardena et al., 2012). However, a recent study investigating the effects of a daily 30 mg intake of zinc gluconate over a 12-month period in individuals with prediabetes did not yield results consistent with prior research findings (Attia et al., 2022). Therefore, further studies involving larger patient populations are necessary for future investigations.
DSW, found at depths greater than 200 m, (Hwang et al., 2009; Miyamura et al., 2004). is characterized by its high concentrations of essential minerals such as magnesium (Mg), calcium (Ca), potassium (K), as well as trace elements like chromium (Cr), selenium (Se), zinc (Zn), and vanadium (V) that are significant for human health (Sheu et al., 2013). Previous research, involving the daily consumption of 440 ml of BDSW over an 8-week period alongside a regular lifestyle and diet, showed no statistically significant difference in FPG and HbA1c levels between the BDSW group and the control group (Ham & Shon, 2020). While the results did not demonstrate significant impacts on glucose parameters, BDSW remains an intriguing area for future exploration. Further investigations could focus on reducing its sugar content to evaluate potential effects on glycemic control.
Implications for Practice
Prediabetes is a condition characterized by elevated blood glucose levels that surpass the normal range but do not meet the threshold for a T2DM diagnosis (ADA, 2022). Left unaddressed, prediabetes can lead to various complications, highlighting the importance of adopting a diabetes-friendly diet. The literature review emphasizes that one of the most challenging aspects hindering patients’ ability to regulate blood glucose levels is related to dietary habits, particularly excessive carbohydrate consumption (Thongpootorn & Lowirakorn, 2020). Nurses play a crucial role in identifying individuals showing signs of prediabetes, intervention before the onset of T2DM. Since obesity is a prevalent risk factor, vigilant monitoring of blood glucose levels is essential for these individuals. Patients categorized as overweight also require careful attention. Collaborative efforts between nurses, ambulatory care nurses, health supervisors, and primary care providers are essential. By utilizing the insights from this study, healthcare professionals can develop customized dietary plans, which may involve adopting a carrageenan-free diet, incorporating yogurt enriched with L plantarum OLL2712, utilizing AHE, and integrating delta-tocotrienol into the nutritional regimen. These tailored nutrition and exercise plans can significantly improve patient outcomes for individuals with prediabetes.
Limitations of This Study
While an extensive search was conducted across substantial electronic databases, there is a possibility that studies from alternative databases were inadvertently omitted. Additionally, it is important to note that this review was carried out by a single author, which could introduce bias into the process of selecting articles for the literature review.
Conclusion
In recent times, the world has grappled with a global epidemic of T2DM. Research suggests that the development of T2DM in individuals with prediabetes can potentially be thwarted through modifications to their lifestyle and dietary choices. Within the scope of this review, certain dietary interventions have shown promise in improving HbA1C levels. These include adopting a carrageenan-free diet, consuming yogurt enriched with L plantarum OLL2712, using AHE, and incorporating delta-tocotrienol into the diet. Conversely, interventions involving salmon, zinc supplementation, and BDSW did not yield significant improvements in HbA1C levels. Moreover, studies examining the effectiveness of vitamin D in regulating blood glucose levels have produced inconsistent findings, necessitating further investigation. As we progress, it is crucial to continue exploring the potential of these dietary strategies in the context of glycemic control for individuals with prediabetes. Additionally, there is a need to investigate and potentially incorporate new foods into the spectrum of approaches aimed at managing prediabetes.
In summary, it is imperative for multidisciplinary healthcare systems, including nurses, to acknowledge the significance of "prediabetes" as a critical health condition. They should implement suitable interventions, particularly focusing on nutrition, to prevent the progression of T2DM in individuals with prediabetes. Additionally, further research is needed to substantiate and reevaluate the potential of various dietary supplements in enhancing glycemic control.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Excellence Center of Community Health Promotion, School of Nursing, Walailak University, Nakhon Si Thammarat, Thailand.
