Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.1
Keywords
Primary Objective
BR2.3: Ductal Carcinoma In-Situ (DCIS). Compare and contrast ductal carcinoma in situ (DCIS) and lobular carcinoma in situ (LCIS) in terms of incidence, clinical presentation, morphology, biomarker expression, pattern of spread, natural history, treatment, and prognosis.
Competency 2: Organ System Pathology, Topic Breast (BR). Learning Goal 2: Molecular Basis of Breast Neoplasia
Secondary Objective
BR2.6: Categories of Breast Cancer. Construct a table to compare and contrast invasive ductal carcinoma (no special type), invasive lobular carcinoma, medullary carcinoma, colloid (mucinous) carcinoma, tubular carcinoma, and metaplastic carcinoma of the breast in terms of incidence, age predilection, etiology, pathogenesis, clinical presentation, gross and microscopic morphology, grade, molecular classification, patterns of spread, clinical course, prognostic indicators, treatment options, and survival rates and to indicate which are more common in males versus females.
Competency 2: Organ System Pathology, Topic Breast (BR). Learning Goal 2: Molecular Basis of Breast Neoplasia
Patient Presentation
A 50-year-old female presents to the mammography suite for an annual mammogram. She has no personal history of breast cancer and her family history is also unremarkable. She reports no discharge from either breast. Bilateral breast and axillary lymph node examinations do not demonstrate any palpable masses, lumps, or skin changes.
Diagnostic Findings, Part 1
An image of the left breast mammogram is shown (Figure 1). The left breast demonstrates a 50-mm grouping of calcifications in a segmental distribution at the 10-o’clock axis, 5 cm from the nipple (circled area, Figure 1). The calcifications are coarse in appearance and have a pleomorphic morphology with varying shape, size, and density. There were no other significant abnormalities in the left breast; the right breast was unremarkable.
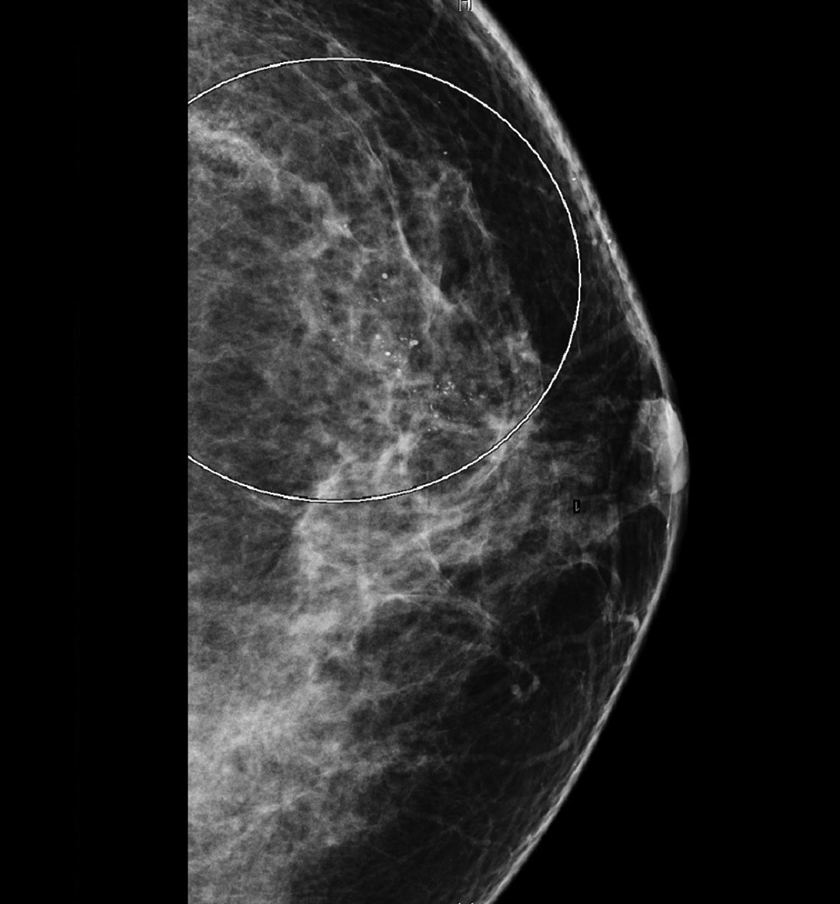
Screening mammogram of this patient’s breast demonstrates a grouping of coarse pleomorphic calcifications (within the circled area).
Questions/Discussion Points, Part 1
What Is the Differential Diagnosis of Calcifications Identified in the Breast on Mammogram?
Breast calcifications are a common finding on screening mammogram. Although they are usually associated with benign processes (fibrocystic changes, hyalinized fibroadenoma, fat necrosis, etc), certain types and patterns of calcifications may be a sign of breast cancer. One form of breast cancer most often detected on mammogram as calcifications is DCIS. In this case, the radiographic findings of the left breast were deemed suspicious.
Why Were the Calcifications on Mammogram Deemed Suspicious?
Certain features of calcifications seen on mammogram suggest a benign etiology, such as presence of multiple similar clusters in more than one quadrant of the breast or in both breasts, stability of the calcifications over time, and uniformity to the sizes and shapes of the calcifications. Other features of calcifications are deemed suspicious and warrant further investigation including calcifications with pleomorphic morphology (varying sizes, shapes, and densities), linear/branching patterns, segmental distribution within a lobe of the breast, or calcifications that are changing over time. 2 These suspicious features are often seen in DCIS, a nonobligate precursor of invasive ductal carcinoma that does not usually form a mass lesion, and calcifications are often the only evidence of the lesion on mammogram. Relatively, specific findings on mammogram for DCIS include calcifications that are pleomorphic, coarse, and in a linear or granular segmental pattern. 3
In this case, the calcifications were considered suspicious because of their coarse and pleomorphic nature and because of their segmental distribution. The patient underwent a stereotactic-guided needle core biopsy of the pleomorphic calcifications, which was sent to the surgical pathology laboratory.
Diagnostic Findings, Part 2
The core biopsy samples were fixed in formalin, embedded in paraffin, and subsequently tissue sections were prepared at a standard 4-µm thickness and the slides were stained with routine hematoxylin and eosin (H&E) stain, and delivered to the pathologist for microscopic examination.
First, the pathologist scanned the biopsy at low power, then at high power. Low- and high-power views of the tissue are shown in Figures 2 and 3.
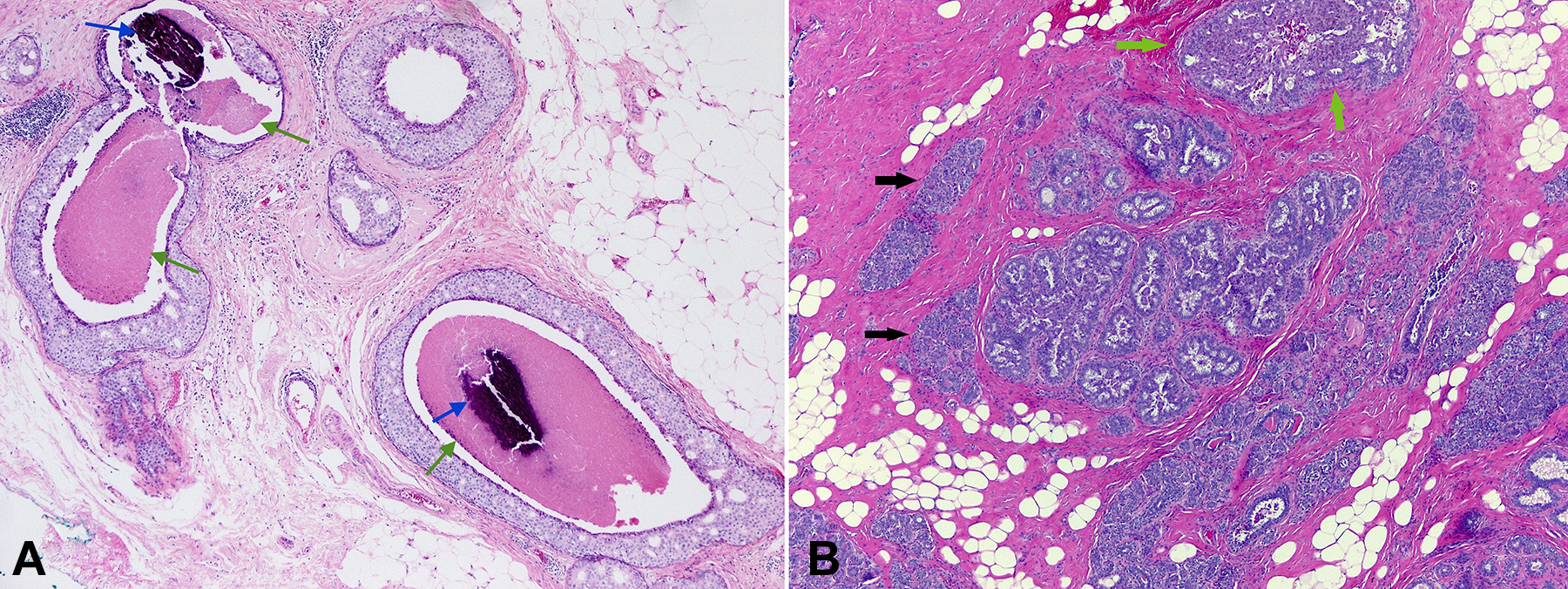
A, Low-power overview of a monomorphic appearing epithelial proliferation expanding but confined within mammary ducts, with large areas of central necrosis (green arrows) and microcalcifications (blue arrows). There is no invasive carcinoma seen. B, Low-power overview of unremarkable breast parenchyma (black arrows), where there is no expansion of the terminal duct lobular units (TDLUs) and no epithelial proliferation. Also seen is usual ductal hyperplasia (UDH; green arrows), where the TDLUs are expanded by a benign proliferation containing peripherally located and irregular shaped fenestrated spaces (hematoxylin and eosin stain, magnification ×40).
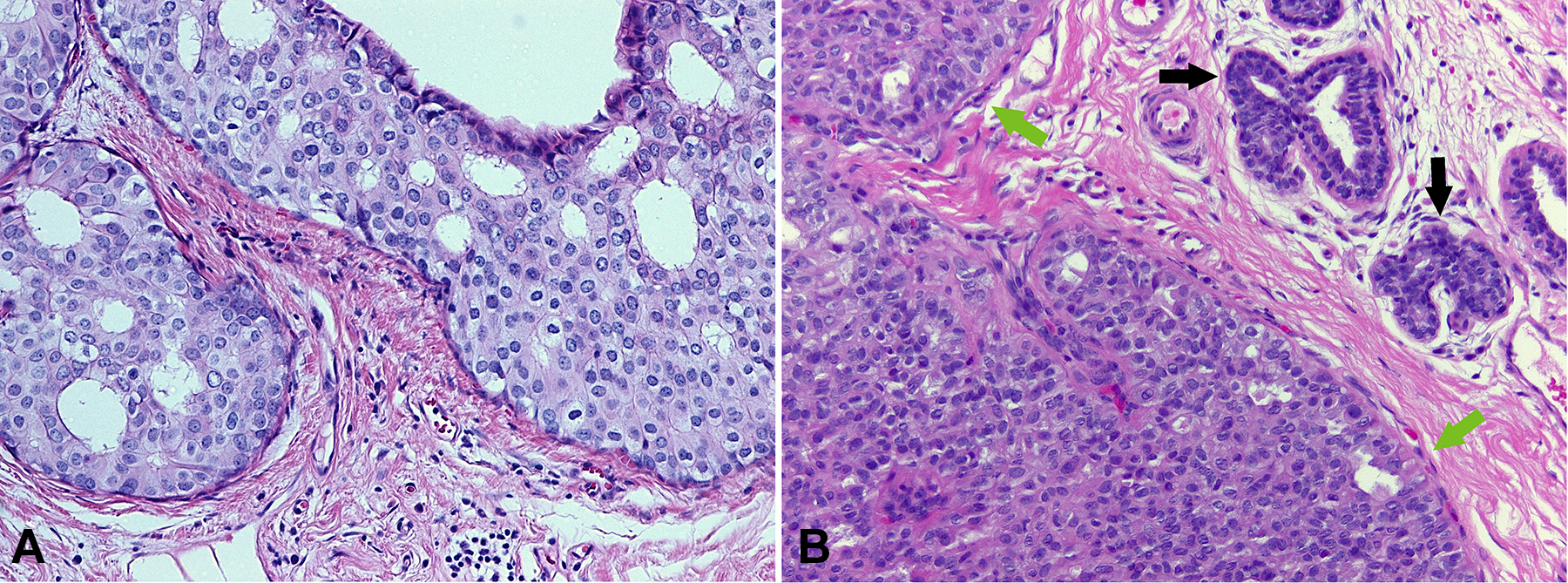
A, Higher power view of the enlarged neoplastic cells, with round nuclei with open chromatin and prominent nucleoli. The neoplastic cells also have prominent cytoplasmic borders, are respecting each other’s space, and look very similar to each other. The architectural pattern is cribriform, as one can see the punched-out rigid spaces within the proliferation. B, Higher power view of the unremarkable terminal duct lobular unit (TDLUs; black arrows) with luminal epithelial cells forming 1 to 2 layers. Also seen is a higher power view of the usual ductal hyperplasia (UDH; green arrow), where cells are small, irregularly shaped, with scattered nuclear grooves, and with nuclear overlapping. Cytoplasmic borders are inconspicuous, nucleoli are not present, and the chromatin is inactive (or closed), illustrated by its dark color (hematoxylin and eosin stain, magnification ×200).
Questions/Discussion Points, Part 2
Where Is the Abnormality Located Within the Mammary Tissue (Best Appreciated in the Low-Power View)?
At low magnification, an architecturally atypical proliferation of cohesive ductal epithelial cells is seen, confined within terminal duct lobular units (TDLUs; Figure 2A). Even at this low magnification, one can appreciate the cribriform rigid “punched out” spaces created by the neoplastic cells (Figure 2A) as compared to nondistended unremarkable TDLUs (black arrows, Figure 2B) and to TDLUs expanded by usual type ductal hyperplasia (green arrows, Figure 2B), which is a common nonneoplastic epithelial proliferation within TDLUs. The usual type ductal hyperplasia contains peripheral and irregular fenestrated spaces within the proliferation, as opposed to the more evenly distributed punched-out rigid spaces seen within the neoplastic proliferation. The neoplastic cells have a tendency to demonstrate a polarity with the long axis of the nucleus oriented toward the center of the punched-out luminal spaces. The centers of some of the ducts involved by the neoplastic proliferation show areas of necrotic epithelium (green arrow, Figure 2A) and associated microcalcifications (blue arrow, Figure 2A). Lastly, the TDLUs involved by the neoplastic proliferation demonstrate rounded smooth peripheral borders that supports that the neoplastic cells are contained within the TDLU; there is no evidence of invasion into the surrounding stroma.
What Are the Histopathologic Features of Ductal Carcinoma In Situ in the Biopsy? Describe Both Cellular and Architectural Features
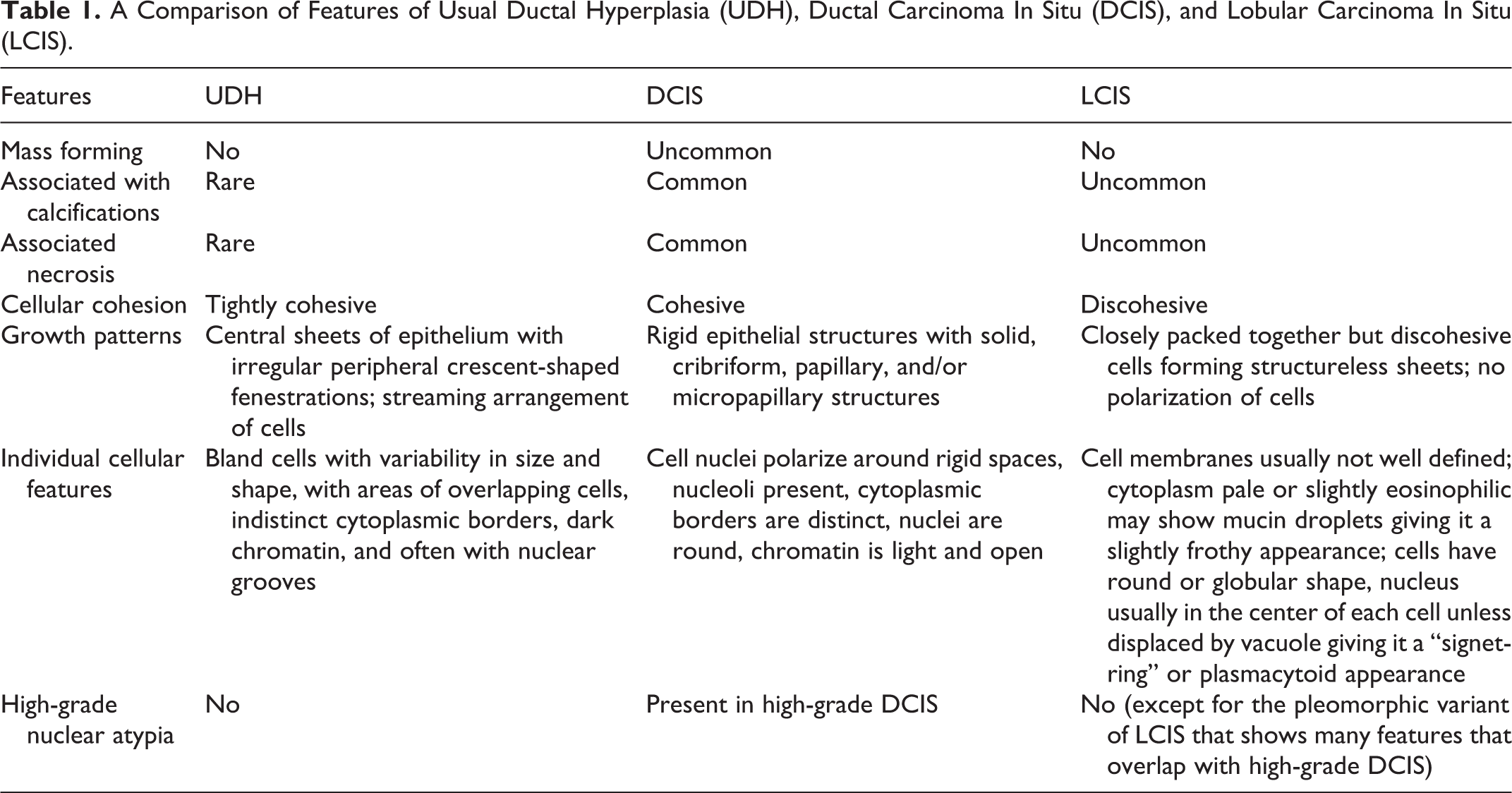
High-magnification examination demonstrates that the neoplastic epithelial cells resemble each other with a monomorphic appearance. Additionally, the cells are seen respecting each other’s spaces and not overlapping, with each cell’s cytoplasmic border being evident (Figure 3A). The cells are enlarged compared with the patient’s normal background mammary epithelial cells (black arrows, Figure 3B) and the benign epithelial cells within the usual type ductal hyperplasia (green arrows, Figure 3B). The neoplastic cells have round nuclei with open chromatin and prominent nucleoli rather than the variable and irregularly shaped nuclei with scattered grooves and dark inactive chromatin within the nonneoplastic epithelial cells. The epithelial cells of the usual type ductal hyperplasia show streaming patterns and areas with nuclear overlapping, without polarizing toward the spaces within the proliferation. Necrosis, mitoses, and calcifications are less likely to be seen within usual type ductal hyperplasia (see Table 1 for a comparison of features of benign and neoplastic epithelial proliferations of the breast).
A Comparison of Features of Usual Ductal Hyperplasia (UDH), Ductal Carcinoma In Situ (DCIS), and Lobular Carcinoma In Situ (LCIS).
What Is the Differential Diagnosis Based on the Histologic Features Seen?
The differential diagnosis of a noninvasive epithelial proliferation occurring within TDLUs includes benign processes (such as usual type ductal hyperplasia), atypical lesions that imply an increased risk for the development of breast cancer (such as atypical ductal hyperplasia and atypical lobular hyperplasia), and noninvasive (in situ) carcinomas (DCIS and LCIS).
What Is the Diagnosis Based on the Histopathologic Examination of the Biopsy?
The described histologic findings in the biopsy material are diagnostic of DCIS, intermediate nuclear grade, with cribriform architectural pattern, and associated central necrosis and microcalcifications. Although DCIS may be a precursor to invasive carcinoma, there is no evidence of invasive carcinoma in this biopsy sample.
What Are the Diagnostic Criteria for Ductal Carcinoma In Situ?
The ductal epithelial proliferation must meet the following diagnostic criteria to be classified as DCIS:
must demonstrate both cytologic and architectural atypia
must either measure greater than 2 mm in size or involve at least 2 ducts if the proliferation shows low or intermediate nuclear grade; proliferations with high nuclear grade are diagnosed with DCIS regardless of size.
How Is Ductal Carcinoma In Situ Classified?
Ductal carcinoma in situ is classified as low-, intermediate-, or high-grade based on the nuclear features of the neoplastic cells. The pathology report will also include information about the architectural pattern, the presence or absence of calcifications, and necrosis. However, the nuclear features are most important, as nuclear features determine the grade of DCIS and thus its classification.
Low-grade DCIS is represented by a monomorphic neoplastic proliferation of epithelial cells that are cytologically similar to benign luminal epithelial cells with only a subtle increase in nuclear:cytoplasmic ratio. The nuclei have smooth contours, diffuse fine chromatin, no or indistinct nucleoli, and no or minimal mitotic figures. The cells of low-grade DCIS often show cribriform architecture with the epithelial cells showing polarization around the luminal spaces (the epithelial cells are oriented perpendicularly to the lumen). High-grade DCIS (nuclear grade 3) shows obvious cytologic atypia with large, markedly pleomorphic and misshapen nuclei, with irregular nuclear membranes, coarse chromatin, and prominent nucleoli; mitoses are often seen and can be atypical. Necrosis is commonly identified in high-grade DCIS. Intermediate-grade DCIS (nuclear grade 2) has features between those of grades 1 and 3. The architectural patterns of the DCIS (most frequently cribriform or solid pattern with or without associated necrosis) are also included in the diagnostic report. The architectural patterns are useful in recognizing DCIS during microscopic examination (particularly in differentiating DCIS from LCIS), but the grade of the DCIS is of more clinical relevance with higher grade lesions showing increased propensity to progress to invasive carcinoma.
Lobular carcinoma in situ is the other common neoplastic proliferation of epithelial cells confined to TDLUs (Figure 4A). In contrast to DCIS, LCIS tends to not be associated with calcifications, and it is often detected as an incidental histologic finding in breast tissue that has been biopsied or resected for other reasons. The neoplastic cells of LCIS are characterized by the loss of expression of the cell-to-cell adhesion protein E-cadherin (Figure 4B), and this manifests histologically as a proliferation of discohesive epithelial cells filling TDLUs but not forming specific growth patterns. Conversely, DCIS maintains cohesive properties and forms various architectural patterns including solid, cribriform, papillary, and/or micropapillary structures.
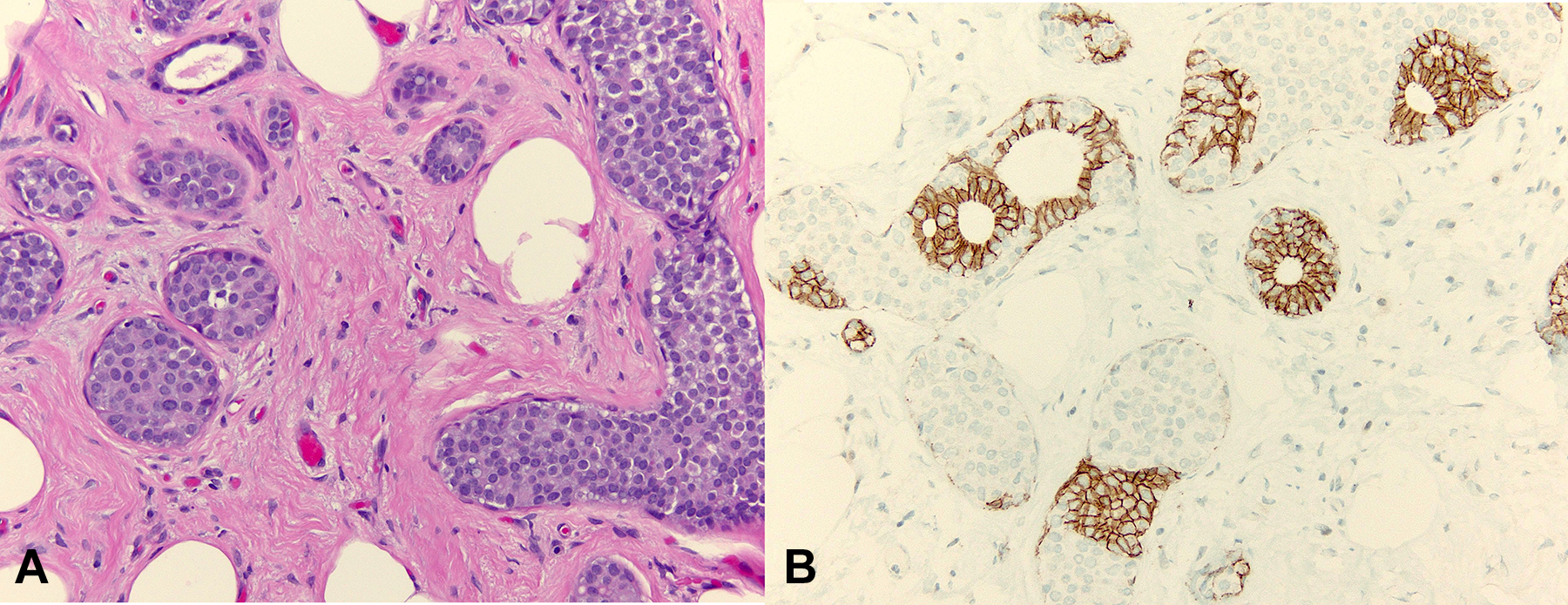
A, Lobular carcinoma in situ (LCIS) shows predominantly lobulocentric distention of the terminal duct lobular unit (TDLU). The neoplastic cells proliferate as patternless sheets of single cells without forming specific architecture (hematoxylin and eosin stain, magnification ×200). B, This is due to their lack of expression of E-cadherin anchoring protein at the cell membranes, shown here as loss of brown membranous chromogen in LCIS but retention in benign mammary parenchyma (E-cadherin immunohistochemical stain, magnification ×200).
Once a Diagnosis of Ductal Carcinoma In Situ Has Been Made, Why Is a Careful Microscopic Examination Required?
Once a diagnosis of DCIS has been established, careful microscopic examination is warranted to look for evidence of invasive carcinoma arising from the DCIS, as this would impact the prognosis and the clinical management for the patient. Particularly, close attention should be paid to any ducts demonstrating irregular borders and ducts showing adjacent stromal reactions that include inflammatory infiltrates and reactive fibrous stroma. If there is doubt about whether invasive carcinoma is present, immunohistochemical markers that highlight myoepithelial cells (p63, calponin, smooth muscle myosin, etc) can be helpful (Figure 5A and B). The myoepithelial layer of cells will be intact around ducts involved by in situ carcinoma. A myoepithelial layer will not be detected around nests of invasive carcinoma as the invasive carcinoma has invaded beyond the confines of the myoepithelial layer and basement membrane and into the surrounding stroma.
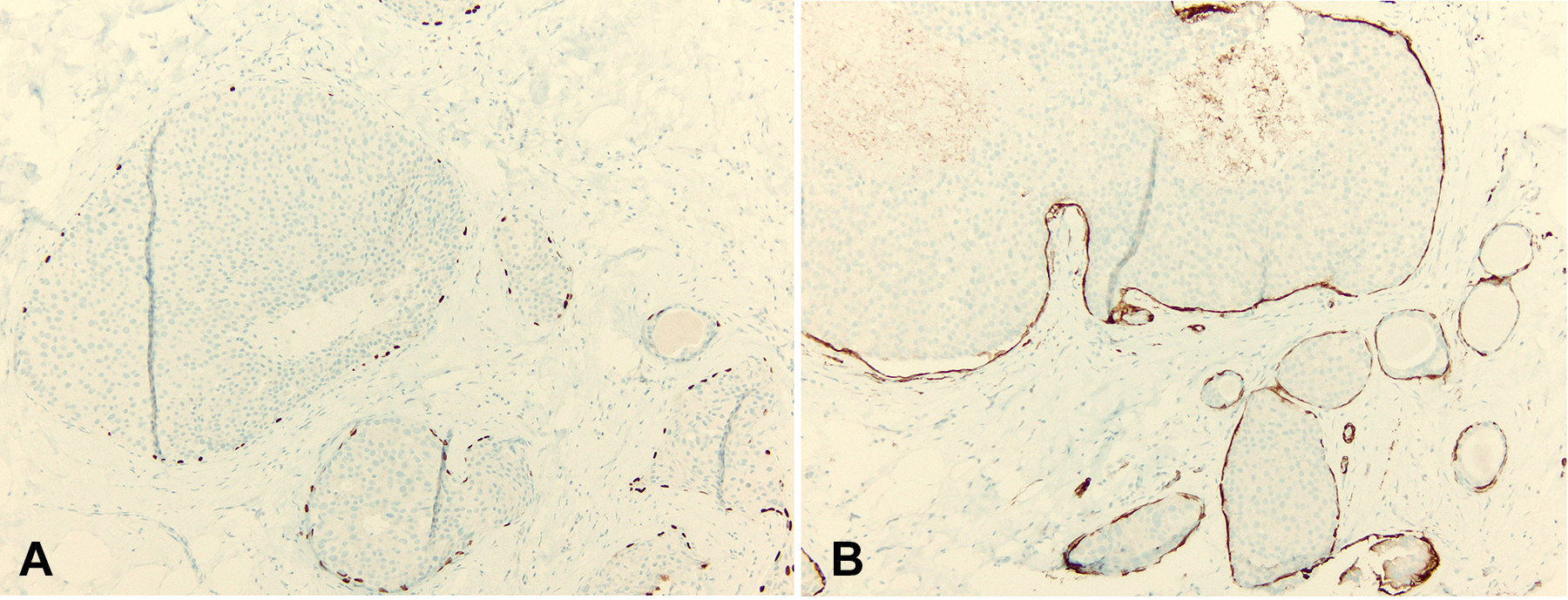
Myoepithelial cells are highlighted with immunohistochemical stains. A, p63, brown chromogen at the periphery of each terminal duct lobular unit (TDLU) decorates the nuclei of the myoepithelial cells; and, (B) smooth muscle myosin or SMMS-1, a cytoplasmic protein of myoepithelial cells, also at the periphery of each TDLU, is highlighted with brown chromogen (immunohistochemical stains, original magnification ×200).
What Ancillary Tests Are Performed Subsequent to the Diagnosis of Ductal Carcinoma In Situ?
Once the diagnosis of DCIS is made, the pathologist will order estrogen receptor (ER) and progesterone receptor (PgR) immunostains (Figure 6A and B). These biomarkers have prognostic and predictive significance. Ductal carcinoma in situ retaining nuclear expression of ER and PR tends to be of lower grade, behaves less aggressively, and will likely respond to endocrine therapy (such as tamoxifen or aromatase inhibitor), thus reducing the risk of ipsilateral and contralateral recurrence 4,5 ; while those that are ER- and PgR-negative do not benefit from endocrine therapy.
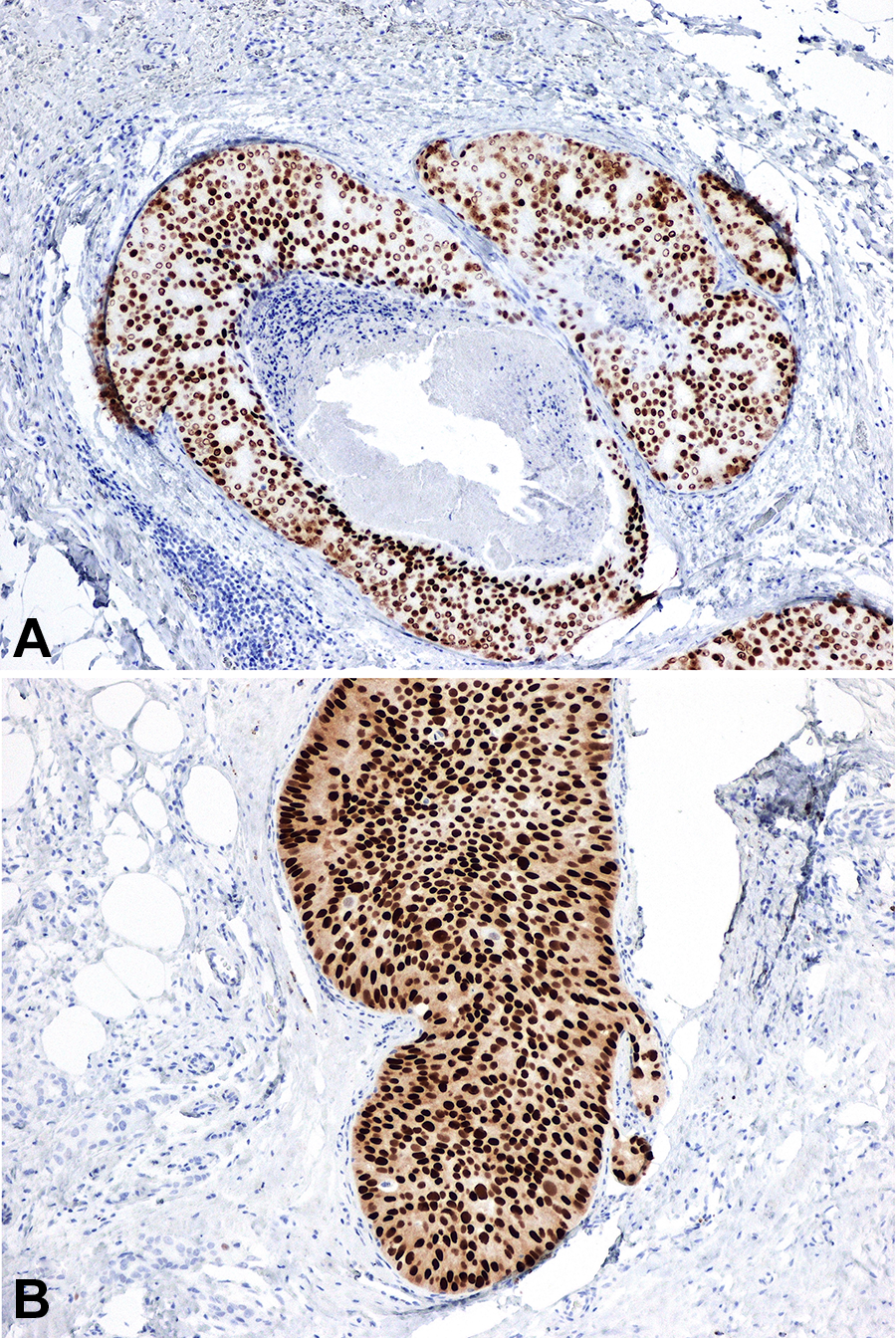
Immunohistochemical stains for biomarkers estrogen receptor (ER; A) and progesterone receptor (PgR; B) show diffuse strong nuclear reactivity, as illustrated by the brown chromogen, within the neoplastic proliferation (immunohistochemical stains, original magnification ×200).
What Is the Next Step in Treatment? How Does the Knowledge of the Biologic Behavior of Ductal Carcinoma In Situ Influence the Treatment Choices?
A diagnosis of DCIS on core biopsy is an indication for surgical excision. If based on radiologic studies (such as mammogram and magnetic resonance imaging), the area of DCIS appears relatively localized, a breast-conserving excision (lumpectomy) can be performed. The goal is to excise the area of DCIS with a rim of uninvolved adjacent breast tissue (clear margins) while conserving the remainder of the breast. Radiation therapy after the lumpectomy is often indicated as this may reduce the risk of recurrence by approximately 50% in patients undergoing breast conservation. 6,7 In cases where a large area of DCIS is anticipated based on the preoperative radiologic imaging, or if the imaging findings indicate involvement of multiple quadrants of the breast (multicentric disease), a mastectomy may be recommended. Mastectomy may also be indicated for patients when attempts at breast conservation surgery were unable to attain clear margins.
Ductal carcinoma in situ is considered a preinvasive disease, so cases of pure DCIS are not expected to spread to regional lymph nodes. However, for patients diagnosed with DCIS on core biopsy, subsequent surgical excision reveals associated invasive carcinoma in approximately 25% of cases. 8 Sentinel lymph node biopsy is generally not performed during breast-conserving excisions for DCIS; however, if the final pathology reveals invasive carcinoma the patient can be brought back for a sentinel node procedure. A mastectomy alters the lymphatic drainage and the anatomy and makes a subsequent return trip to the operating room for sentinel lymph node biopsy unreliable. Thus, for patients undergoing mastectomy for DCIS, sentinel lymph node biopsy is often performed at the time of mastectomy, particularly in patients with radiologic evidence of a large burden of DCIS and patients considered as high risk for occult invasive carcinoma. 9,10
The management of LCIS and DCIS is substantially different. One reason for this is that while DCIS is often a localized process amenable to excision, LCIS is more frequently diffusely distributed including multicentric involvement of a breast (in approximately 50% of cases) and bilateral breast involvement (approximately 30% of cases), and thus LCIS is less amenable to cure by a breast-conserving excision. Although LCIS is considered a precursor lesion to invasive lobular carcinoma, a diagnosis of LCIS implies an increased risk for developing invasive carcinoma not only in the area of the diagnosed LCIS but also in other quadrants of that breast and in the contralateral breast. When DCIS is identified in a needle core biopsy, a surgical excision is performed. In contrast, a diagnosis of LCIS in a core biopsy does not usually trigger an excision unless other high-risk lesions are also identified in the same specimen (such as atypical ductal hyperplasia or DCIS). If DCIS is present at a margin of a surgical excision specimen, an additional surgery is usually performed to obtain a clean margin. In contrast, if LCIS is present at a margin of excision, an additional procedure to obtain a clean margin is not performed. Lobular carcinoma in situ increases the risk of an invasive recurrence in the ipsilateral and contralateral breasts, but its presence at a surgical margin does not substantially influence the recurrence risks. In these ways, LCIS is approached like a proliferative disease associated with increased risk for development of a future cancer.
Teaching Points
Most calcifications seen mammographically correspond to benign conditions, but certain calcification patterns can be suggestive of DCIS.
On histology, DCIS characteristically shows a monomorphic neoplastic proliferation of epithelial cells, expanding the TDLUs, but not invading beyond the confines of the myoepithelial cells and basement membrane.
Ductal carcinoma in situ is classified as low, intermediate, or high nuclear grade, with higher grade lesions associated with higher risk of progression to invasive carcinoma.
Ductal carcinoma in situ retaining nuclear expression of ER and PgR tends to be of lower grade, behaves less aggressively, and will likely respond to endocrine therapy, thus reducing the risk of recurrence.
After a diagnosis of DCIS on core biopsy, surgical excision is indicated (lumpectomy or mastectomy), and the risk of finding invasive carcinoma in the excision specimen is approximately 25%.
Radiation therapy after breast-conserving surgery is often indicated as this may reduce the risk of recurrence by approximately 50% in patients undergoing breast conservation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article processing fee for this article was funded by an Open Access Award given by the Society of ‘67, which supports the mission of the Association of Pathology Chairs to produce the next generation of outstanding investigators and educational scholars in the field of pathology. This award helps to promote the publication of high-quality original scholarship in Academic Pathology by authors at an early stage of academic development.
