Abstract
Keywords
Primary Objective
Competency 2: Organ System Pathology; Topic RS: Respiratory System; Learning Goal 4: Obstructive Diseases of the Lung
Secondary Objective
Competency 1: Disease Mechanisms and Processes; Topic EM: Environmental Mechanism; Learning Goal 1: Cell Injury
Patient Presentation
A 75-year-old male patient presents to the emergency department with a dry cough and shortness of breath. The patient has been experiencing the shortness of breath for 2 months, and the dry cough persistently for 6 months. The patient reports feeling winded when at rest and especially when walking up the stairs in his home. Additionally, he has had unexplained and sudden weight loss in the past 6 months.
Diagnostic Findings, Part 1
The patient is overweight (body mass index of 28.0) and has a history of high blood pressure which has been under control for years with medication. Patient is a nonsmoker. Physical examination reveals the patient has a temperature of 98.9 °F and a pulse oximetry reading of 92% SpO2. Physical examination of the lungs reveals crackling sounds. He also has clubbed fingers, but cannot confirm when the symptom began.
Question/Discussion Points, Part 1
What Is Your Differential Diagnosis at This Point?
Generally, symptoms which include dyspnea and cough over a prolonged period of time, with or without an occupational risk factor may suggest chronic obstructive pulmonary disease, asbestosis, coal workers, pneumoconiosis, silicosis, lung cancer, bacterial infection, amyloidosis, progressive massive fibrosis (PMF), or sarcoidosis.
What Is an Important Factor to Consider When a Patient Presents With Lung Disease?
Exposure to certain agents over time can increase risk for developing lung disease. With that in mind, exposure due to occupation and environment must be taken into consideration when analyzing the differential diagnoses. When asking the patient about his occupation, he reports he was a shipyard worker for 30 years from 1965 to 1995, and has been retired ever since.
Diagnostic Findings, Part 2
The forced vital capacity of the lungs is 77% of the predicted value. Spirometry shows that total lung capacity is 76%, and the diffusing capacity for carbon monoxide (DLCO) is decreased. A sputum culture is negative for microorganisms. A chest X-ray shows irregular opacities in the lungs, and there is evidence of noncalcified pleural plaques. Computed tomography (CT) images show pleural disease and pleural plaques. Magnetic resonance imaging fluorodeoxyglucose positron emission tomography (MRI FDG-PET) scan shows negative results in the region of the lesions.
Question/Discussion Points, Part 2
Given the Additional Diagnostic Findings, What Is the Most Likely Diagnosis for the Patient?
The patient’s occupational history along with presentation of shortness of breath, a dry cough, wheezing over months, and clubbed fingers suggests the patient has a form of pneumoconiosis caused by inhaling asbestos fibers, called asbestosis. 2 The diagnostic evidence of asbestosis is as follows. Altogether, the pulmonary function tests (PFT) reveal a mild restrictive pattern. Note that the PFT’s role is to demonstrate the severity of lung disease, as opposed to playing a key role in diagnosing the lung disease itself. A negative sputum culture suggests the patient does not have tuberculosis (TB) nor any other bacterial infection. The presence of opacities and plaques on the chest X-ray is evidence there is scar tissue in the lungs, and pleural plaques are a common indicator of asbestos-related disease. 3 The chest X-ray and CT scan provide evidence of fibrotic lesions, likely resulting from asbestos fibers in the lungs, and the MRI FDG-PET scan was negative, which suggests the diagnosis is unrelated to lung cancer. In summation, the diagnosis therefore must not be related to a bacterial infection, ruling out TB and pneumonia, not related to lung cancer nor the other types of pneumoconiosis because of the specific occupational exposure to asbestos.
What Are the Risk Factors for Pneumoconiosis?
Pneumoconiosis is a group of interstitial lung diseases caused by breathing in certain kinds of dust particles, characterized by the presence of scar tissue in the lungs and reduced lung function. 4 -6 This means that routine occupational exposure to inhaled agents as listed in Table 1 is a risk factor for development of pneumoconiosis. The 3 most common types of dust exposure leading to pneumoconiosis include asbestos fibers, crystalline silica, and coal dust. 8,12 Asbestosis and silicosis are the more common types of pneumoconiosis, while coal worker pneumoconiosis (CWP) is more rare. In developing countries, silicosis is more frequent than asbestosis, although this trend is reversed in more developed countries. 13
Risk Factors for the Most Common Types of Pneumoconiosis, Including Pneumoconiosis Due to Asbestos Fibers, Crystalline Silica, and Coal Dust.

Of the 2 basic types of mining (surface mining and underground mining), underground miners are exposed to higher dust levels in the underground environment and as such are at greater risk of developing CWP. 14 Though surface miners have a reduced risk of developing CWP because coal dust is diluted by outdoor air, they are more at risk of developing silicosis. 14 According to the US Mine Safety and Health Administration, in 2019, there were 38 250 underground coal miners and 40 212 surface miners in the United States. 12
Workplace asbestos exposure affects 125 million people worldwide, and it is estimated that at least 107 000 people die each year from lung cancer, mesothelioma, and asbestosis resulting from occupational exposure to asbestos. 15 In the United States, the Occupational Safety and Health Administration (OSHA) did not have regulations regarding asbestos exposure in the workplace until 1971. 2 Although regulations set in the 1980s and 1990s now limit the risk of workers developing asbestos-related disease, the impacts of lack of regulation continue because it takes decades for asbestos-related diseases to develop. 2
Long-term occupational exposure to agents such as talc (used in the leather, rubber, textiles, ceramics, and paints industries), beryllium (used in the electronic, aerospace, and nuclear industries), iron dust (a common dust for welders), 7 and indium (common for dental technicians) can also lead to pneumoconiosis, with less frequency.
How Can You Make a Diagnosis of Pneumoconiosis?
Dust burden can only be measured indirectly. 7 Therefore, a diagnosis of pneumoconiosis is made through a combination of presentation of lung symptoms, physical examination, CT scan, or chest X-ray with evidence of lung nodules and masses, and sometimes, biopsy. 6,8 Diagnosis of pneumoconiosis can be greatly strengthened when there has been exposure to agents known to be associated with increased risk of disease. 7 However, a diagnosis of pneumoconiosis is not always straightforward. Many of the diagnostic findings will have characteristics that are shared with other lung diseases. Table 2 lists the 3 most common types of dust agents causing pneumoconiosis and their respective chest radiograph, CT, and pathologic findings.
Table of Common Pneumoconiosis Chest Radiograph, Computed Tomography, and Pathologic Findings of the 3 Most Common Agents of Pneumoconiosis: Asbestos Fibers, Silica, Coal Dust.

Abbreviation: CWP, coal worker pneumoconiosis.
What Is the Mechanism of Pneumoconiosis, Specifically Pneumoconiosis Resulting From Coal Dust, Asbestos, Silica?
The biological mechanism of pneumoconiosis is characterized largely by the properties of the inhaled dust particles themselves, amount, and duration of dust. The exact mechanism of pneumoconiosis is not fully understood, however, it is largely agreed upon that the tissue response to the dust depends on particle size and its biological activity. Inhaling coal dust, among other dust agents that are relatively inert, can lead to a considerable accumulation of dust in the lungs without much tissue response. 7 The characteristic pathologic finding related to CWP is the coal macule, which is made up of dust and dust-laden macrophages around respiratory bronchioles, with reticulin fibers and some collagen. 7 Coal dust alone contains mostly carbon, but coal mine dust contains hydrogen, nitrogen, trace metals, inorganic minerals, and crystalline silica. 14 Trace metals like boron, cadmium, copper, nickel, iron, lead, and zinc can be cytotoxic and carcinogenic. 14 Other fibrogenic dusts like silica and asbestos generate responses in the form of nodular fibrosis and diffuse fibrosis, respectively. 13,16
Asbestos exposure can lead to the formation of pleural plaques and evidence of asbestos bodies in lung tissue. Over an extended period of time, the build-up of particles in the lungs causes inflammation, leading to the development of scar tissue and reduced lung function. 5,19 This can be seen in Figure 2, a cross sectional view of lung tissue with a well-defined plaque present as a result of asbestos exposure. Another characteristic pathological finding in the lungs due to asbestos exposure is asbestos bodies, which are fibers that can be seen easily via light microscopy. 13 Figure 3 is a histological image of interstitial lung tissue with an embedded asbestos fiber. They are coated in an iron-rich proteinaceous material, and their presence in a lung biopsy is a helpful indication of considerable asbestos exposure. However, their absence in a biopsy is not enough to disprove asbestosis. 13 Asbestos plaques themselves are benign, so the greater concern is that the presence of plaque is indicative of exposure to asbestos, along with all of its associated risks, like mesothelioma and lung cancer. 2 This is an important distinction to make because asbestosis will not lead to mesothelioma in its progression, but the exposure that led to them does. 13 Figure 1A shows a circumferential pleural-based mass of mesothelioma.
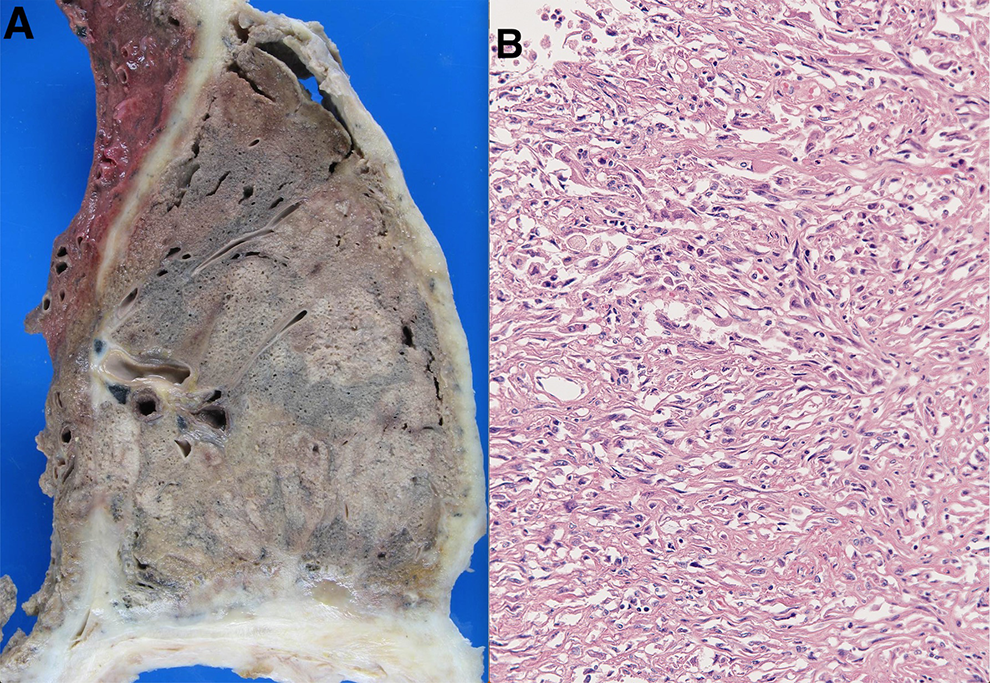
A, Circumferential pleural-based mass of mesothelioma. B, Histologic section of the mass showing disorganized pattern of highly pleomorphic spindle cells (H&E, ×200).
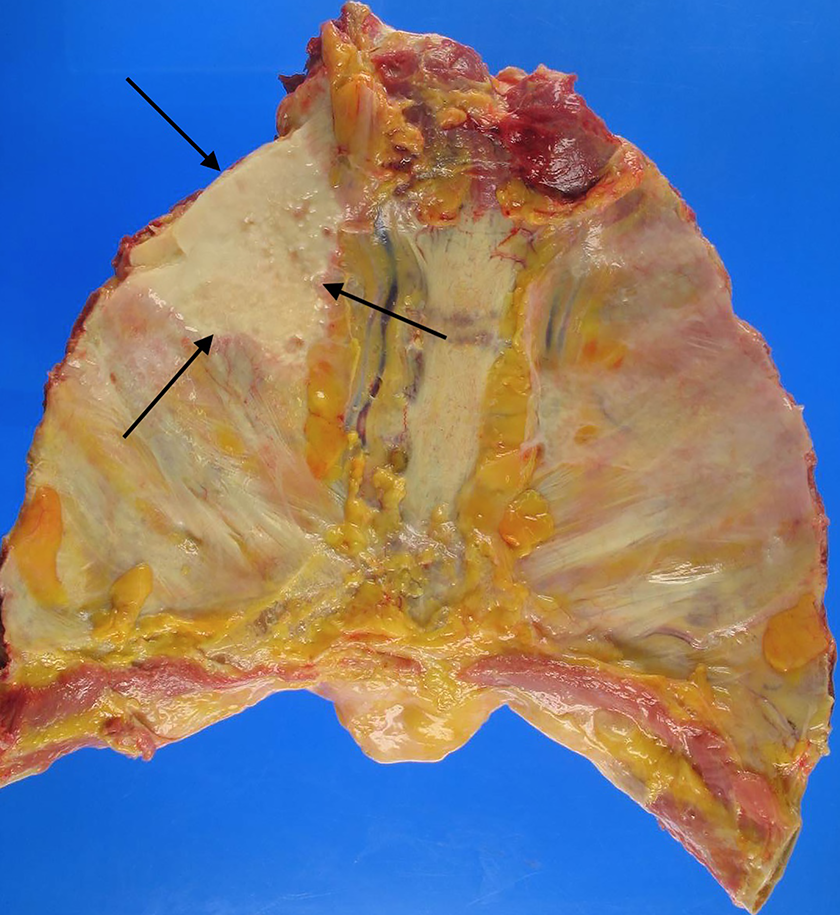
The arrows indicate the borders of a well-circumscribed plaque on the parietal pleural surface of a patient with asbestos exposure. Such plaques are fibrotic and often calcified but lack asbestos bodies.
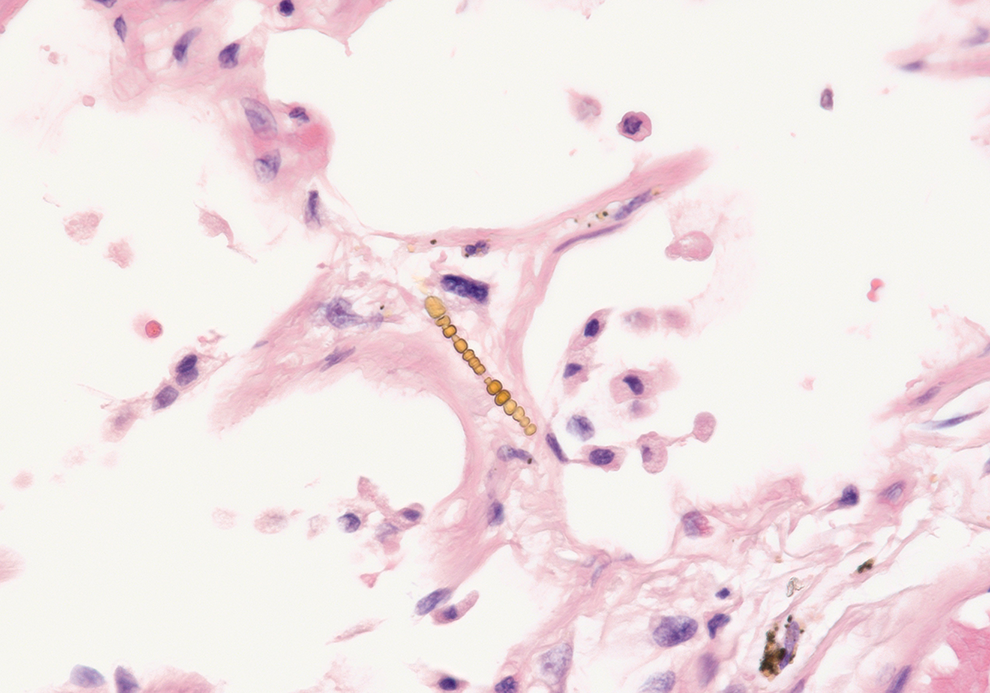
Asbestos body in the lung of a patient with asbestos exposure. Note the gold-brown beaded rod in the interstitium (H&E, ×400).
The pathogenesis of silicosis is similarly not entirely understood. However, it is understood that silicosis can rapidly progress to its more serious forms after short periods of intense exposure, and therefore those with occupational exposure to silica should be monitored closely. 7,17 It is also known that silica used or prepared in different ways can have varying toxicity based on its biological activity. For example, freshly fractured silica generated during sandblasting is more toxic to alveolar macrophages than “aged” silica because of its increased redox potential. 7
What Is the Difference Between Simple and Complicated Pneumoconiosis?
Pneumoconiosis is categorized according to the severity of the disease. Broadly, there are 2 general categories of pneumoconiosis: simple and complicated. 4 Simple pneumoconiosis causes a relatively small amount of scar tissue in the lungs, but it can, due to radiographic severity of the disease, dust exposure level, and total dust burden, progress to become PMF. 6 Progressive massive fibrosis is characterized by the presence of damaged and scarred tissue surrounding alveoli in the lungs. 5,6,14 This scarring makes it more difficult for oxygen to pass into the bloodstream, which can raise pulmonary artery pressure in the lungs. 19 Eventually, further complications can arise from pneumoconiosis including: respiratory failure when blood oxygen levels are extremely low, lung cancer, blood clots in the lungs, a collapsed lung, and lung infections. 5,6
What Is the Treatment for Pneumoconiosis?
While there is no cure for pneumoconiosis, the goal of treatment is to limit further damage to the lungs. Treatments include inhalers, pulmonary rehabilitation, oxygen. 8 Patients who smoke should be strongly advised to quit smoking 6,8 because certain types of pneumoconiosis can compound and increase the risk of lung cancer. Patients will also be advised to stop their exposure to dusts as well. 16
How Can Pneumoconiosis Be Prevented?
Pneumoconiosis should be approached with an emphasis on prevention because it cannot be cured. 6 The OSHA sets standards for workers at risk for pneumoconiosis and some of these include wearing a mask, washing areas of skin that come into contact with dust, removal of dust from clothing, washing hands and face before eating, drinking, and taking medications, not smoking, getting regular chest X-rays and physical examinations, 20 and informing a doctor or employer of pneumoconiosis symptoms. 5,6
Teaching Points
Pneumoconiosis is a group of interstitial lung diseases caused by breathing in certain kinds of dust particles, characterized by the presence of scar tissue in the lungs and reduced lung function.
The 3 most common types of dust exposure leading to pneumoconiosis include asbestos fibers, crystalline silica, and coal dust; oftentimes these are occupational exposures that occur over many years.
A diagnosis of pneumoconiosis is made through a combination of presentation of lung symptoms, physical examination (including pulmonary function tests), CT scan or chest X-ray with evidence of lung nodules and masses, personal history of occupational exposure to agents causing pneumoconiosis, and biopsy.
There are 2 types of pneumoconiosis: Simple pneumoconiosis, which usually only causes a small amount of scar tissue in the lungs, and complicated pneumoconiosis or progressive massive fibrosis (PMF), which is characterized by the presence of damaged and scarred tissue surrounding alveoli in the lungs.
Treatment of pneumoconiosis seeks to limit further damage to the lungs and includes inhalers, pulmonary rehabilitation, and oxygen.
There is no cure for pneumoconiosis, so there is a focus on prevention and management of exposure through OSHA standards like wearing a mask, washing areas of skin that come into contact with dust, removing dust from clothing, washing hands and face before eating, drinking, and taking medications, not smoking, getting regular chest X-rays and physical examinations, and informing a doctor or employer of pneumoconiosis symptoms.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
