Abstract
Keywords
Primary Objective
Competency 2: Organ System Pathology; Topic CH: Cardiovascular-Heart; Learning Goal 2: Atherosclerosis in Heart Disease
Secondary Objective
Competency 2: Organ System Pathology; Topic CH: Cardiovascular-Heart; Learning Goal 2: Atherosclerosis in Heart Disease
Patient Presentation
A 59-year-old man with a significant previous history of hypertension and right carotid endarterectomy presented to the emergency department (ED) with chest pain radiating to the left arm accompanied by diaphoresis and dyspnea.
Diagnostic Findings, Part 1
His electrocardiogram (ECG) was performed that showed elevation in leads 1, augmented Vector Left (aVL) and V1-V6 (Figure 1), consistent with the diagnosis of extensive anterolateral myocardial infarction (MI). His troponin I levels were found to be elevated, 20 ng/mL (normal reference: cardiac troponin I <0.03 ng/mL, cardiac troponin T <0.1 ng/mL, creatine kinase MB isoenzyme <10 ng/mL, myoglobin <170 ng/mL).
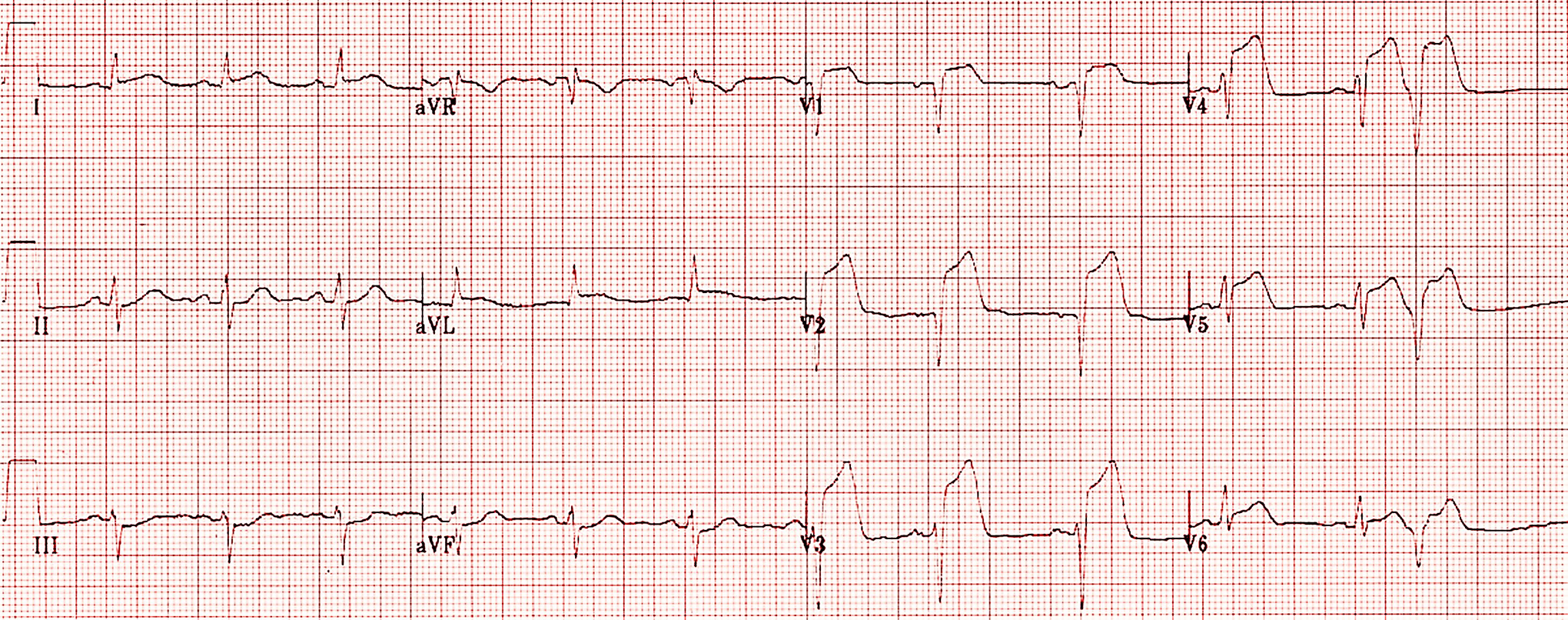
ECG showing ST elevation in leads I, aVL and V1-V6, consistent with acute anterolateral MI. ECG indicates electrocardiogram; MI, myocardial infarction.
In the ED, his blood pressure dropped and he was placed on vasopressors. After appropriate initial management, he was sent to the cardiac catheterization lab. He was found to have 90% occlusion of the left anterior descending (LAD) coronary artery, and a drug-eluting stent was placed in the LAD. The patient eventually stabilized and was weaned off of vasopressors, and he was transferred to the general cardiology service. His ECG results were stable, and troponins were trending down. The patient was discharged.
Questions/Discussion, Part 1
What Is the Differential Diagnosis in a Patient Presenting With Chest Pain?
The differential diagnosis of a patient presenting with chest pain includes MI, acute gastritis, pulmonary embolism, anxiety, aortic dissection, and musculoskeletal chest pain. Based on the clinical features, a number of differentials can be enlisted in this case:
Acute gastritis
The chest pain in MI may mimic heartburn. However, the typical ECG changes of MI together with elevated cardiac enzymes, in this case makes MI more likely.
Pulmonary embolism
The patient has chest pain associated with tachypnea and dyspnea which are symptoms seen in pulmonary embolism. However, a lack of risk factors and the gradual onset of his pain makes this diagnosis unlikely. The ECG changes together with elevated cardiac enzymes also favor MI.
Musculoskeletal chest pain
It may present with severe chest pain but is associated with point tenderness and does not radiate. Also, it is not associated with ECG changes and elevated cardiac enzymes.
Aortic dissection
The presenting complaint of aortic dissection is also chest pain; however, it starts abruptly with maximal intensity at onset, tearing in character, radiates to the back (interscapular area), and does not show the classic ECG findings of an MI.
Anxiety
Patients with anxiety (panic attack) present with chest pain, sweating, and palpitations. However, the classic ECG findings are absent.
What Is the Workup in a Patient With Chest Pain?
The first and foremost step to evaluate the exact cause of chest pain and the artery involved is to do an ECG. Table 1 describes the findings of ECG in MI in detail. 2
Arteries involved in MI with respective ECG findings.

Abbreviations: LAD, left anterior descending artery; RCA, right coronary artery; LCX, left circumflex artery.
Cardiac markers (creatine kinase [CK-MB], troponin I, troponin T, and myoglobin) are also done to diagnose MI. Since cardiac troponin has high sensitivity and high specificity for myocardial injury, it is the gold standard marker to diagnose MI. The time when the different markers begin to peak and normalize is shown in Figure 2.

Timing of changes of cardiac markers.
What Is the Definitive Diagnosis in This Patient?
The patient had chest pain radiating to the left arm, accompanied by diaphoresis, dyspnea, and tachycardia, raising a strong suspicion for MI. The ECG changes together with elevated cardiac enzymes confirm the diagnosis.
Two weeks later, he returned with the complaint of mild chest pain radiating to the left arm, associated with diaphoresis, nausea, and shortness of breath and was hypoxic.
Diagnostic Findings, Part 2
Pulse: 112 bpm, irregularly irregular
Blood pressure: 100/60 mm Hg
Respiratory rate: 22 per minute
Temperature: 98 °F
On this second visit, his ECG showed Q waves, consistent with old infarct, while his troponin I was elevated, 5 ng/mL. The patient progressed to ventricular fibrillation the same day. Chest compression and resuscitation efforts were performed, but his condition deteriorated and he was found to be unresponsive and pulseless. The patient was intubated and required multiple vasopressors. Despite these interventions, the patient expired 13 to 15 hours after the presentation to ED. At the request of the patient’s family, an autopsy was performed.
Questions/Discussion, Part 2
What Is an MI?
Myocardial infarction, commonly called a heart attack, is the death of cardiac muscles as a result of prolonged severe ischemia. 2 Ischemia is defined as restricted blood supply to cells and tissues that cause shortage of oxygen, vital to cell metabolism.
What Is the Classic Clinical Presentation of an MI?
Patients with MI present with chest pain that typically radiates to the left arm, jaw, back or shoulder, dyspnea, diaphoresis, lightheadedness, and tachycardia. Additionally, inferior wall MI can present with bradycardia because of vagal stimulation. 2
What Is the Pathogenesis of MI?
An inability of coronary blood flow to meet the myocardial demand is the major cause of MI. Most commonly, there is a narrowing of the coronary arteries due to atherosclerotic plaques with varying degrees of superimposed changes, for example, thrombosis and vasospasm. Certain conditions, such as hypertrophy, tachycardia also lead to MI, in which demand outweighs supply. The sequence of events underlying MI is detailed below. 2
Most MIs are caused by coronary artery occlusion. The pathogenesis is shown in the Figure 3. 2
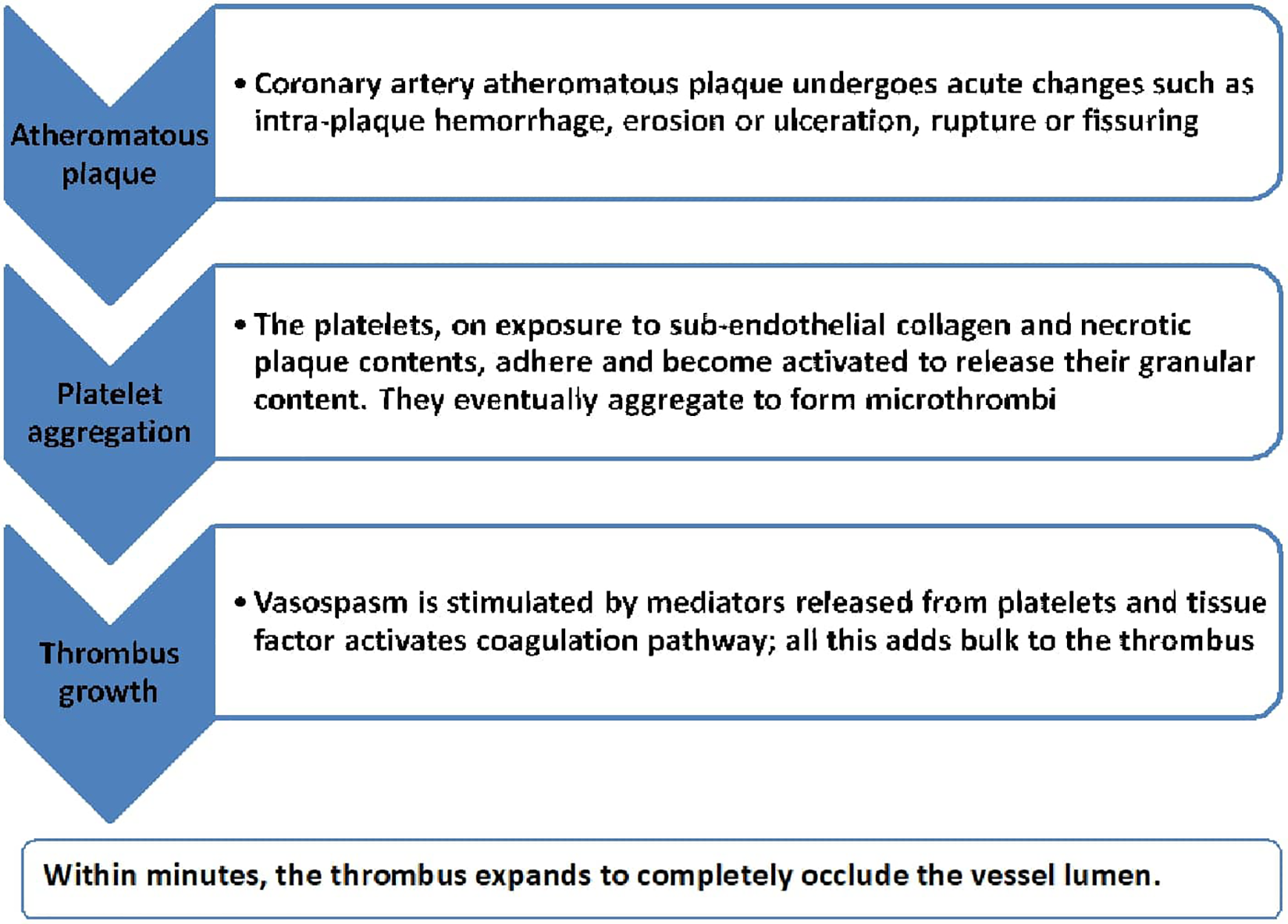
Pathogenesis of the vessel occlusion.
However, in approximately 10% of cases of transmural MI, coronary atherosclerosis is absent. In these cases, other mechanisms are responsible for the reduced myocardial perfusion including
2
vasospasm, which might be associated with platelet aggregation or resulting from drug ingestion (eg, cocaine or ephedrine); atrial fibrillation resulting in emboli formation, infective endocarditis vegetations, and prosthetic material inside the heart; and an embolus from the right side of the heart or the peripheral veins, traversing a patent foramen ovale into the coronary arteries.
Ischemia caused by disorders of small intramural coronary vessels (eg, vasculitis), hematologic abnormalities (eg, sickle cell disease), amyloid deposition in the walls of the vessels, and arterial dissection hypertrophy (eg, aortic stenosis), hypotension (eg, shock), or inadequate myocardial protection during cardiac surgery.
There is a characteristic myocardial response associated with MI. The occlusion of a coronary artery reduces blood flow to an area of the myocardium, causing ischemia, dysfunction of the myocardium, and eventually myocyte death due to prolonged vascular compromise. The duration and severity of blood flow reduction are the 2 factors that determine the outcome. Inadequate production of high-energy phosphates (eg, ATP and creatinine phosphate) and accumulation of potentially noxious metabolites (eg, lactic acid) are a result of the cessation of aerobic metabolism. The contractility of the myocardium stops soon after the ischemic insult begins because the myocardial function is largely dependent on oxygen and nutrients. Ultimately, heart failure pursues even before myocyte necrosis.
Within a few seconds of the onset, changes in the ultrastructure develop (including relaxation of myofibrils, depletion of glycogen, cell, and mitochondrial swelling). However, the early manifestations of ischemic injury are potentially reversible. 2
How Is Pathophysiology Related to Laboratory Diagnosis?
The disruption of sarcolemmal membranes of necrotic myocytes allows leakage of intracellular macromolecules into the interstitial tissue and finally into microvasculature and lymphatics. This forms the basis of elevated cardiac enzymes in MI. 2
The cardiac enzymes include creatine kinase (CK-MB), troponin I, and troponin T. The CK-MB peaks at 24 hours and normalizes in 48 to 72 hours. Troponin I peaks in 24 hours and begins to normalize in 5 to 10 days, whereas troponin T peaks in 12 to 48 hours and normal in 5 to 14 days. These cardiac enzymes are used for diagnosis and following up the patient.
What Are the Morphological Features of MI?
The morphologic findings in MI vary with the time since infarction, as shown in Table 2. 2 How long the patient survives following an MI determines the gross and microscopic features seen thereafter. Coagulative necrosis is the cell death that occurs due to ischemia, leading to denaturation of structural proteins. It is the first change that occurs in a cell after an MI. After the necrosis, neutrophilic influx is seen in around 12 to 24 hours. This is followed by loss of nuclei on days 1 to 3, phagocytosis by macrophages on days 3 to 7, and granulation tissue formation at the margins. Fibrosis, or scarring, begins in around 2 weeks, and eventually, complete scar formation occurs in 2 months. Changes on gross examination are not apparent before 12 hours. It can be difficult to histologically recognize MI early as myocardium takes 6 to 12 hours to undergo secondary changes of necrosis. 3
Timing of Gross and Microscopic Changes After Myocardial Infarction.

The postmortem autopsy of the patient described above revealed an extensive MI, involving the right and left ventricles, papillary muscles, and interventricular septum. Gross examination shows 2 lesions, dark, mottled area corresponds with an MI 12 to 24 hours old, and the yellow, softened lesion with red-tan borders is grossly consistent with a 10 to 14 days old MI (Figure 4A and B, respectively). The microscopic examination shows coagulative necrosis of myocytes and neutrophilic infiltration (Figure 5) corresponding with 12 to 24 hours of infarction. Figure 6 shows granulation tissue at the margins with extensive collagen deposition, corresponding with an approximate age of infarction of 10 to 14 days prior to death.
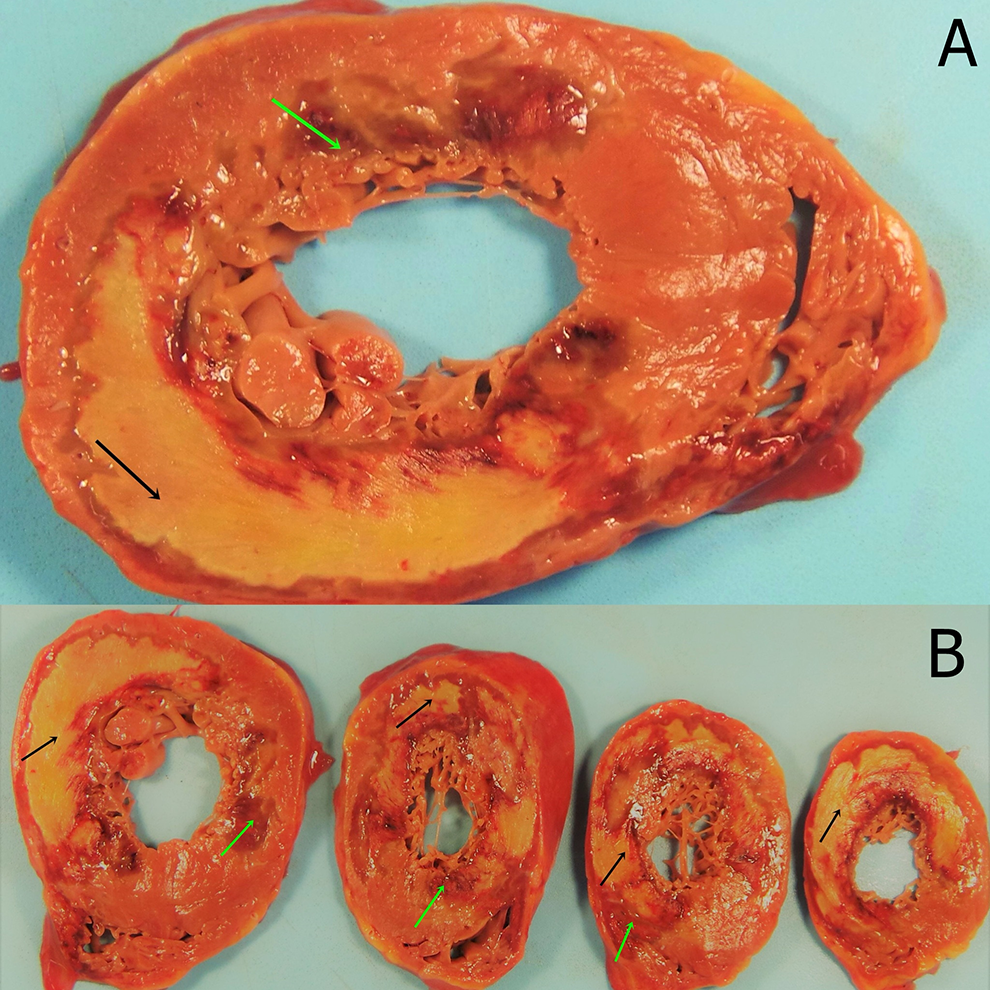
A and B, On gross examination, 2 lesions involving the myocardium and the papillary muscles are seen. Green arrows point to dark mottling, and the black arrows point to a yellow, softened lesion with red-tan borders; these correspond to a myocardial infarction in between 12 to 24 hours and 10 to 14 days, respectively.
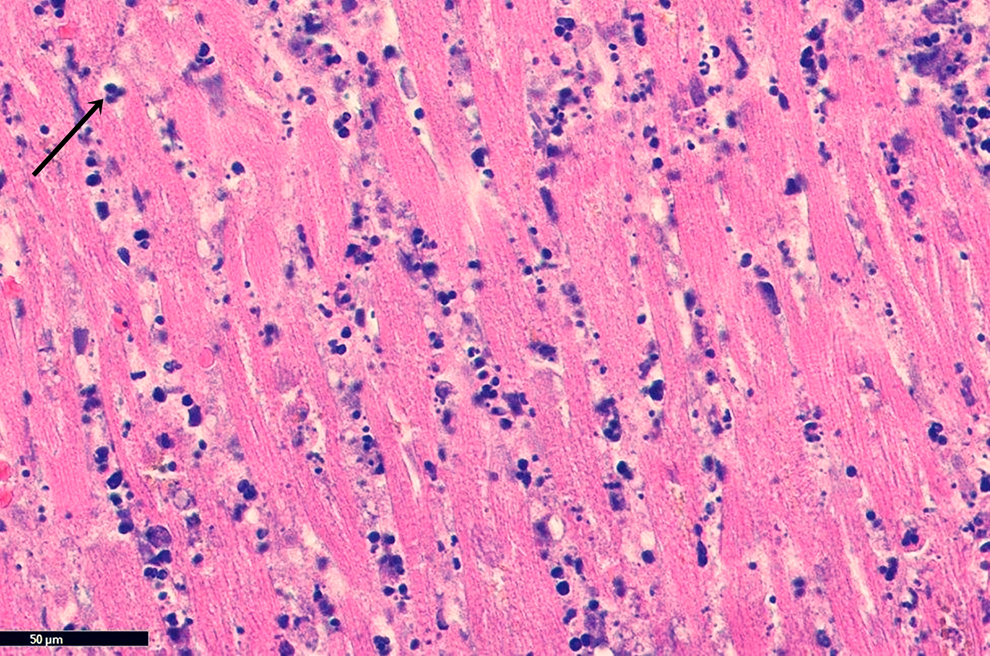
Coagulative necrosis of the myocytes with absent nuclei and an interstitial neutrophilic infiltrate (black arrow).
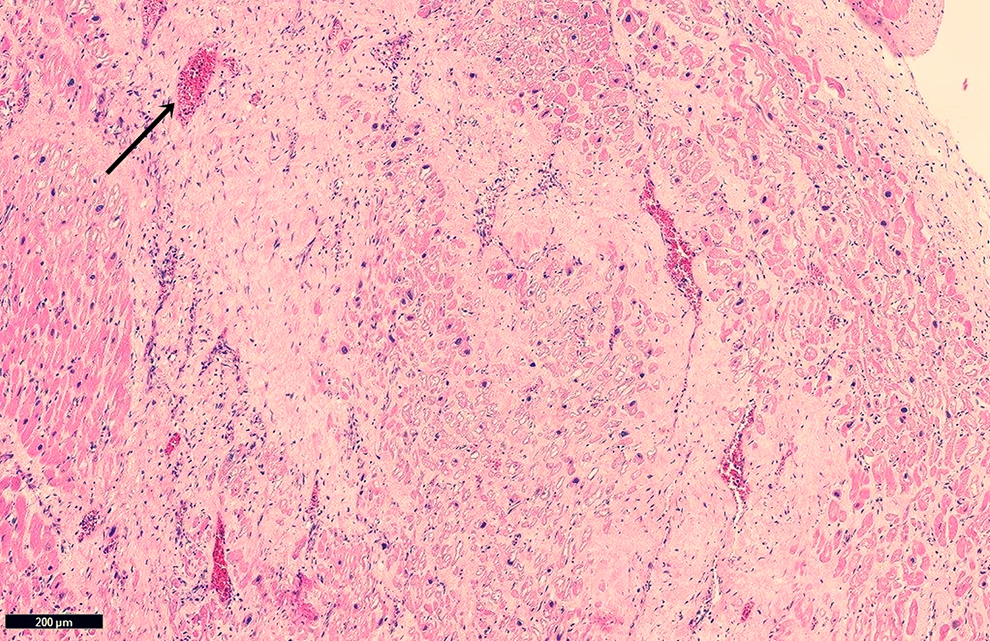
Well-formed granulation tissue with collagen deposition and neovascularization (black arrow).
What Pathophysiologic Mechanisms Underlie the Histologic Changes? 2
The waviness of fibers seen at 30 minutes to 4 hours following infarction results from the forceful pull of the viable fibers on adjacent dead fibers that cannot contract during systole. The margins of infarcts may show an additional ischemic change known as myocyte vacuolization or myocytolysis, which occurs as a consequence of intracellular salt and water retention in the sarcoplasmic reticulum. The necrotic muscle evokes an acute inflammatory response followed by the removal of the dead myocytes by macrophages. Granulation tissue replaces the damaged area leading to a “fibrous scar” formation. Mostly, a well-developed scar is formed in 6 weeks. After an infarct is healed, it becomes impossible to assess its age.
How Is MI Managed?
As the patient approaches the ED with acute coronary syndrome (a term that encompasses unstable angina as well as MI), management includes
4
: immediate ECG and troponins oxygen and rhythm monitoring analgesics antiplatelet; aspirin 300 mg orally anticoagulant intravenous nitrates
The management of ST-elevation MI (STEMI) differs from that of Non-ST-elevation MI (NSTEMI). In STEMI, there is complete occlusion by the thrombus, ECG shows ST elevation, and cardiac enzymes are elevated. While in NSTEMI, the occluding thrombus causes either partial occlusion of a major artery or complete occlusion of a minor artery, ECG shows ST depression, and cardiac enzymes are found to be elevated.
If the patient has STEMI, percutaneous coronary intervention (PCI) is preferred to restore the patency of the coronary artery. The standard of care is to perform PCI within 90 minutes after the patient comes to the ED. If PCI is unachievable, thrombolytic therapy should be administered. In case of NSTEMI, PCI does not prove to be helpful.
Teaching Points
Myocardial infarction is the death of cardiac muscles due to prolonged severe ischemia.
The primary diagnostic tests are ECG, followed by serum cardiac enzymes, normal reference range being: cardiac troponin I <0.03 ng/mL, cardiac troponin T <0.1 ng/mL, creatine kinase MB isoenzyme <10 ng/mL, myoglobin <170 ng/mL).
Cardiac troponin has high sensitivity and high specificity for myocardial injury.
The main pathophysiologic steps include coronary artery occlusion leading to myocardial ischemia due to which myocardial contractility ceases.
Gross changes appear only after 12 hours showing characteristic dark mottling.
Histological changes take 6 to 12 hours to develop with the first apparent change being coagulative necrosis.
After the necrosis, neutrophilic influx is seen in around 12 to 24 hours.
Loss of nuclei occurs on days 1 to 3, phagocytosis by macrophages on days 3 to 7, and granulation tissue formation at the margins.
Eventually complete scar takes around 2 months to form.
The primary goal is to manage MI as early as possible.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
