Abstract
Primary Objective
Competency 2: Organ System Pathology; Topic: Cardiovascular – Heart (CH): Learning Goal 5: Valvular Dysfunction.
Secondary Objective
Competency 1: Disease Mechanisms and Processes, Topic: Adaptation and Cell Death (ACD): Learning Goal 1: Cellular Response to Injury
Patient Presentation
A 51-year-old African American male presented to his primary care physician complaining of not being able to “catch his breath” after going up the stairs in his home. He has developed a cough with worsening exercise tolerance and dizziness over the last 6 months, which he had attributed to “getting older.” The patient also reported occasional nausea and loss of appetite over the last couple weeks, which was not normal for him. He reported not scheduling any primary care visits for the last several years due to a busy schedule but came in today at the request of his daughter. His most recent visit, 10 years ago, discussed a family history of heart disease (his grandfather died in his 60’s of a “bad heart”), but the complete physical examination had been reportedly unremarkable. During his current visit, his blood pressure was recorded as 125/105 mm Hg, and auscultation of a new murmur, which radiated to the neck, led to a cardiology referral.
Diagnostic Findings, Part 1
During the referral physical examination, the murmur heard was a crescendo-decrescendo systolic murmur that peaked early in systole and was heard best in the right upper sternal border but also radiated bilaterally to the carotids. The Valsalva maneuver strain and handgrip bedside maneuvers reduced the intensity of the murmur, and releasing the Valsalva maneuver increased the intensity of the murmur after several beats. During pulmonary examination, the patient was found to have diffuse fine rales in both lung fields, slight jugular venous distention, and a small amount of edema in both legs.
Questions/Discussion Points, Part 1
What Is the Differential Diagnosis Based on the Clinical History and Physical Examination?
The patient’s complaints are consistent with some degree of heart failure, and the physical examination findings are consistent with a valvular cause. The differential diagnosis for systolic murmurs would include most commonly aortic stenosis and mitral regurgitation but also hypertrophic cardiomyopathy, a ventricular septal defect, pulmonary stenosis, and tricuspid valve disease. 2,3
The location where the murmur was heard best during physical examination would be consistent with the aortic valve as the source. In addition, the results of the bedside maneuvers like the Valsalva and handgrip reduce the likelihood of hypertrophic cardiomyopathy and mitral regurgitation, respectively. 2 -4
What Further Testing Is Indicated for the Patient?
Aortic valve stenosis is a narrowing of the aortic valve orifice, which prevents the effective flow of blood from the heart into the aorta, and based on the history and physical examination, is one of the most likely causes of the patient’s complaints. Imaging using transthoracic echocardiography (TTE) is recommended for initial evaluation of suspected aortic valve stenosis. 2,3 The TTE will also be useful for ruling out the other suspected differential diagnoses listed above. The TTE, in the case of aortic stenosis, measures aortic jet velocity and mean transvalvular pressure gradient, which can be used to assign a degree and stage that will best guide management. According to American College of Cardiology/American Heart Association (ACC/AHA) guidelines, symptomatic patients often have severe aortic stenosis, which is often quantified by flow velocities ≥4 m/s or mean ΔP ≥40 mm. Severe aortic stenosis should be medically treated, and due to the high mortality, surgical replacement has also received Class I Level B recommendation (ie, benefit greatly exceeds risk in the limited population trials that have been performed). 2 Transthoracic echocardiography along with chest X-ray could also be helpful in determining if left ventricle hypertrophy or heart failure are the cause of increased central venous pressure and fluid in the lungs or if it is due to another cause like infection.
Cardiac catheterization is also recommended for assessment in symptomatic patients when the noninvasive tests are inconclusive or some discrepancy between testing and physical examination findings exists. 5,6 The catheterization can confirm diagnosis and estimate severity as well as assess associated mitral valve and coronary artery disease that are also often present.
During either of these procedures, the presence of a bicuspid aortic valve is the underlying reason for up to 50% of aortic valve replacements and has been shown to calcify 20 to 30 years earlier than tricuspid valves and could explain this patient’s condition at a relatively young age. 7,8
Diagnostic Findings, Part 2
The patient is given the option of surgical replacement of the valve but chooses to consider his options for a period of time. Two weeks after seeing the cardiologist, he is found dead at home and an autopsy is performed which identifies the 2 findings illustrated in Figures 1 and 2. The weight of the heart at autopsy is 610 grams.
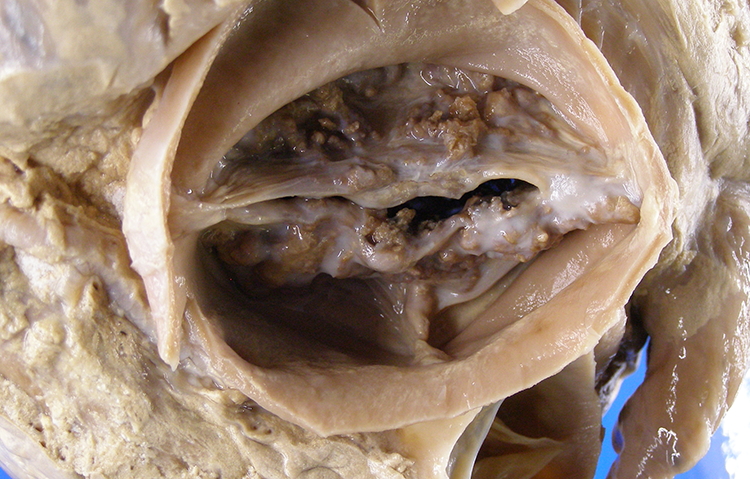
Aortic stenosis due to dystrophic calcification of a bicuspid aortic valve. At the 6 o’clock position is the midline raphe from incomplete separation at the commissure.
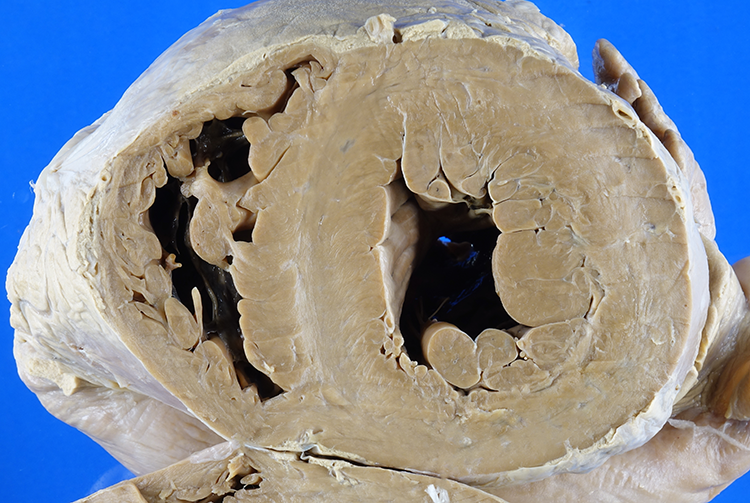
Left ventricular hypertrophy, overall. The interventricular septum measures 2.4 cm in thickness (with the upper range of normal being 1.5 cm).
Questions/Discussion Points, Part 2
What Is the Diagnosis?
The patient has aortic stenosis due to calcification of a bicuspid aortic valve (Figure 1), which has led to hypertrophy of the left ventricle (Figures 2 and 3).

Left ventricular hypertrophy, close-up. The interventricular septum measures 2.4 cm in thickness (with the upper range of normal being 1.5 cm).
What Is the Pathogenesis of Aortic Stenosis, and What Conditions Predispose Toward Its Development?
The most common pathologic forms of aortic stenosis are calcific aortic stenosis, either of a normal tricuspid valve or of a congenitally bicuspid aortic valve, and chronic rheumatic heart disease. Other more rare causes of aortic stenosis include Williams syndrome, alkaptonuria, systemic lupus erythematosus, and radiation. 7,9,10 Dystrophic calcification of a tricuspid or bicuspid aortic valve likely occurs in many of the same ways as atherosclerosis in the vessels. 10 Damage to the endothelium, through stress or radiation for example, allows the accumulation and oxidation of lipids in the fibrosa, which induces inflammation and mineralization. Also, inflammatory cytokines and oxidized lipid derivatives may induce expression of bone through the transition of interstitial cells into osteoblasts. 7 Apoptosis of interstitial cells can also lead to the unstructured deposition of calcium and phosphorus. 7 Given that aortic stenosis due to calcification of a bicuspid aortic valve presents sooner than aortic stenosis due to calcification of a tricuspid aortic valve, that the abnormal flow through the bicuspid aortic valve contributes to the pathogenesis is likely. Chronic rheumatic heart disease involving the aortic valve is typically the end result of immunologically mediated damage following group A streptococcal pharyngitis. A response of both antibodies and CD4+ T cells against streptococcal virulence factor M protein can recognize cardiac self-antigens, which further results in inflammation of the endocardium and fibrinoid necrosis of the valves. These lesions are characterized by the formation of Aschoff bodies consisting of T lymphocytes, plasma cells, and macrophages called Anitschkow cells, which are characteristic of acute rheumatic fever, but are not found in chronic rheumatic valvulitis. 10 Radiation has also been shown to lead to fibrous formations causing stenosis with and without the presence of calcium deposits or vascularization. 11
As would be expected due to the similar pathogenic mechanisms between atherosclerosis and aortic valve stenosis, many of the same risk factors are shared between the 2 diseases. Older age, metabolic syndrome, elevated lipoprotein A, male sex, smoking, hypertension, hypercholesterolemia, obesity, and diabetes have all shown to be risk factors for aortic valve stenosis. 7 A single nucleotide polymorphism rs10455872 in lipoprotein A locus and congenital conditions like bicuspid valves have also been correlated to a significantly elevated risk. 12 -15 Aortic valve thickening has also been correlated to aortic valve stenosis and may even progress to stenosis as 1 study showed that 15.9% of patients with valve thickening will be found to have stenosis at repeat echocardiogram one year later. 12
What Morphologic Features Help Distinguish the 3 Main Forms of Aortic Stenosis
With calcific aortic stenosis associated with either a tricuspid or a bicuspid valve, examination of the valve will reveal nodules of calcium on the sinus surface of the cusps associated with thickening and fibrosis of the cusps. 10 The commissures will usually be relatively uninvolved. A congenitally bicuspid aortic valve will have 2 cusps, usually one is larger than the other, with the large cusp having a midline raphe, a ridge of fibrous tissue in the center of the cusp extending between the cusp and the sinus wall, which is due to incomplete separation at the commissure. Occasionally, both cusps will be the same size and neither will have a midline raphe. With chronic rheumatic aortic valvulitis, the aortic valve will have fusion at both commissures, without the nodules of calcification being present as in calcific aortic stenosis. 10
What Are the Complications of Aortic Stenosis?
The increased afterload due to the narrowed aortic valve and subsequent reduced outflow will lead to hypertrophy of the left ventricle, an adaptation to the stress induced by the stenotic valve. Typically, in such a pressure-overload driven hypertrophy, sarcomeres are added in parallel to existing cells leading to concentric increase in wall thickness, which leads to its own complications such as increased oxygen demand and impaired ventricular filling. Initially, the increases in generated pressure allow the heart to overcome the increased resistance and maintain cardiac output, but the abnormal metabolic demands and genetic expression lead to increasing stiffness of the ventricle and decreased cardiac output. 10,16
The reduced cardiac output from the left ventricle will eventually lead to fluid buildup in the pulmonary veins and capillaries, which could cause fluid accumulation in the lungs, as well as increased afterload on the right heart. The increased demand on the right side of the heart in addition to the left can quickly lead to congestive heart failure. 10,16
Because compensatory adaptation (ie, hypertrophy) can allow for normal function for a prolonged period of time, aortic valve stenosis often leads to patients not being diagnosed until after symptoms like dyspnea, syncope, and angina develop, which can be late in the disease course. This is also reinforced by the fact that without definite treatment by surgery, the condition will continue to deteriorate with a >50% mortality within 2 years of diagnosis in severe symptomatic patients. 10,16
Without intervention, aortic valve stenosis quickly progresses to severe disease with many complications including left ventricular failure, sudden death, arrhythmia, heart block, systemic embolization, myocardial infarct, stroke, and acute respiratory failure. 12,17 Individuals with calcific aortic stenosis of a tricuspid aortic valve often present in their 70s or 80s, whereas individuals with calcific aortic stenosis of a congenitally bicuspid aortic valve often present in their 40s or 50s. The time course for presentation with chronic rheumatic aortic valvulitis is highly variable. 10 Advanced disease has also been shown to cause von Willebrand factor deficiency and lower gastrointestinal tract bleeding due to angiodysplasia. 12,17 In addition to the direct effects on the left ventricle (ie, cardiac hypertrophy), individuals with aortic stenosis are also at increased risk of endocarditis.
Teaching Points
Careful history taking, physical examination, and diagnostic tests are important in the diagnosis of aortic stenosis.
Three main forms of aortic stenosis are calcification of a normal tricuspid aortic valve, calcification of a congenital bicuspid aortic valve, and chronic rheumatic valvulitis. The morphology of the first 2 is distinguished by the number of cusps, whereas the morphology of the third is distinguished most characteristically by fusion at all 3 commissures.
The long-term consequences of aortic stenosis included left ventricle hypertrophy, leading to reduced ejection fraction and stroke volume, diastolic dysfunction, and pulmonary hypertension, which can lead to heart failure and risk for sudden death.
There are no known therapies proven to affect mortality, delay surgery, or prevent valve dysfunction in patients at risk of aortic stenosis, which includes patients with congenital bicuspid valve or elevated lipoprotein A levels, so managing these risk factors is the primary concern.
The primary treatment available is aortic valve replacement, which has the most benefit in terms of reduced mortality for symptomatic patients. It has also been advocated for in asymptomatic patients that meet ACC/AHA criteria, which consider symptoms as well as objective findings like pressure gradients, flow velocity across the valve, and ejection fraction for making treatment recommendations.
After symptoms arise, the average survival without valve replacement is only 2 to 3 years.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: W. Kemp receives royalties from McGraw-Hill for a pathology review book and from Thieme for a pathology question and answer book.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
