Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.1
Keywords
Primary Objective
Objective UTK5.2: Nephrotic Syndrome. Describe the pathophysiology and morphologic features of nephrotic syndrome, and contrast with nephritic syndrome.
Competency 2: Organ System Pathology; Topic: Kidney (UTK); Learning Goal 5: Renal Syndromes.
Secondary Objective
Objective UTK5.5: Dysproteinemic Nephropathies. Describe the pathogenesis of the nephropathies associated with dysproteinemia.
Competency 2: Organ System Pathology; Topic: Kidney (UTK); Learning Goal 5: Renal Syndromes.
Patient Presentation
A 71-year-old African-American male presented to his primary care physician with a long-standing history of asthma, chronic obstructive pulmonary disease, a new gradual onset of generalized edema, and increased fatigue.
Diagnostic Findings, Part 1
The patient’s vital signs showed blood pressure of 125/76 mm Hg, heart rate of 69 beats per minute, respiratory rate of 15 breaths per minute, oxygen saturation of 98% on room air, and temperature of 98.4 °F. Routine physical examination was remarkable for pitting edema, predominately located around the ankles and feet and puffiness around the eyes. The lungs were clear on auscultation. The abdomen was distended and fluid wave test was positive. The liver was normal in size. Electrocardiogram was normal.
Questions/Discussion Points, Part 1
What Is the Differential Diagnosis of a New Onset of Generalized Edema?
Edema refers to the expansion of interstitial fluid volume leading to palpable swelling of the tissue. When edema is massive and generalized, the excess fluid accumulation is called anasarca. Several clinical conditions are associated with the development of generalized edema, the most important being heart failure, liver cirrhosis, nephrotic syndrome, and venous and lymphatic diseases.
What Laboratory Studies Would be Helpful in the Initial Evaluation of This Patient?
The initial laboratory studies should include complete blood count, general chemistry, liver function tests, and lipid profile.
Diagnostic Findings, Part 2
Selected laboratory results are presented in Table 1.
Selected Laboratory Results.
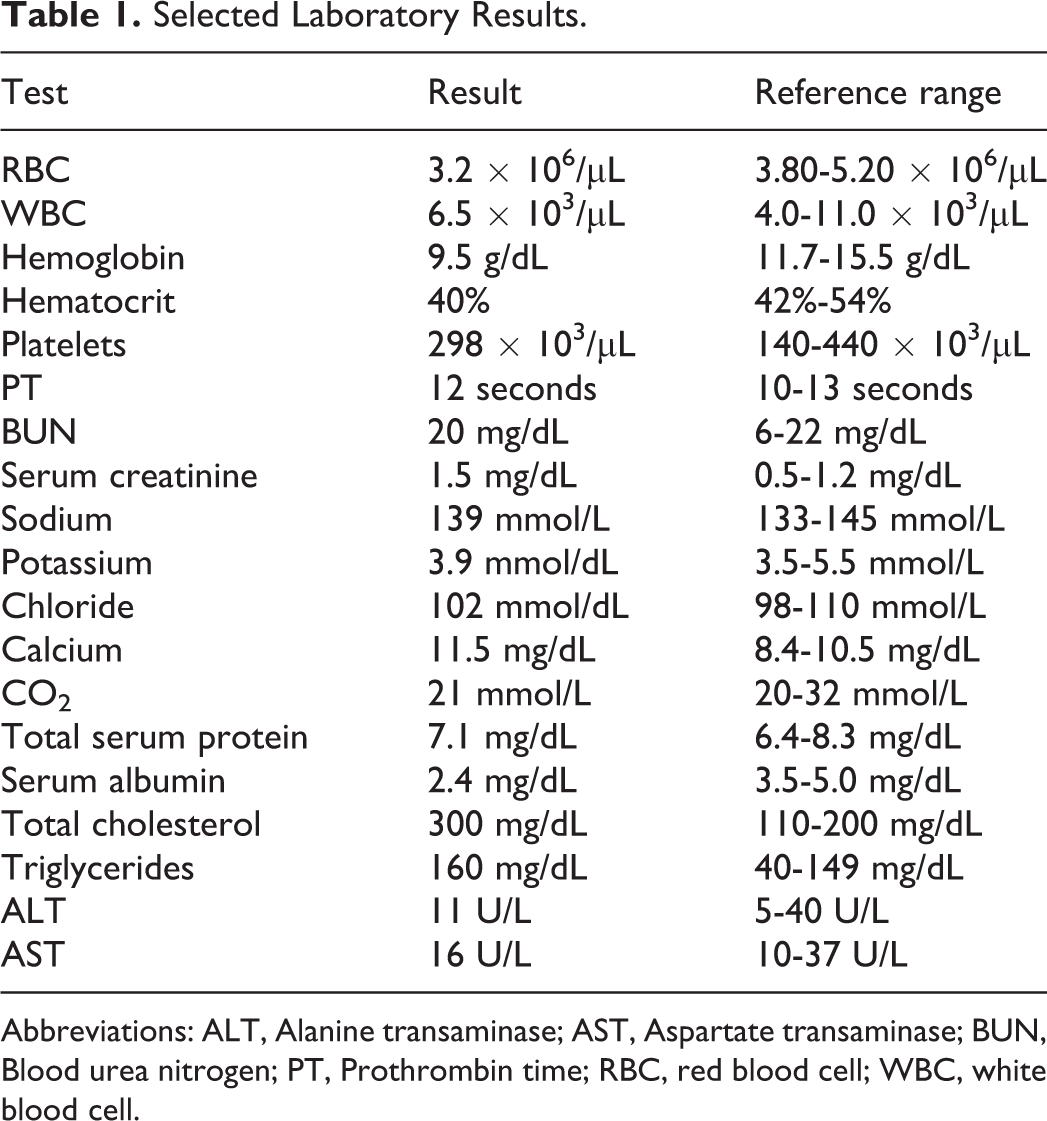
Abbreviations: ALT, Alanine transaminase; AST, Aspartate transaminase; BUN, Blood urea nitrogen; PT, Prothrombin time; RBC, red blood cell; WBC, white blood cell.
Questions/Discussion Points, Part 2
What Are the Implications of the Blood Analysis?
The presence of low albumin level, hypercholesterolemia, and hypertriglyceridemia in a patient with anasarca is strongly suggestive of nephrotic syndrome. Mild anemia and hypercalcemia point to a possibility of hematologic disorder. There is also indication of renal insufficiency.
What Other Laboratory Studies Would Be Helpful in Evaluation of This Patient?
Urinalysis with urine protein excretion quantification, and serum and urine electrophoresis.
Diagnostic Findings, Part 3
The urinalysis was significant for the presence of protein >500 mg/dL (reference range: negative-trace). There was no glucose, ketones, bilirubin, bacteria, or red or white blood cells. Urine sediment contained oval fat bodies, but no cellular casts were identified. A 24-hour urine collection revealed 4.0 g/24 h protein excretion (normal total protein excretion is <150 mg, usually 40-80 mg/24 h). Serum electrophoresis showed abnormal M spike, and urine electrophoresis showed the presence of Bence–Jones protein. Subsequent serum immunofixation confirmed the presence of λ light chain restriction.
Questions/Discussion Points, Part 3
What Is the Most Likely Clinical Diagnosis?
Nephrotic syndrome with an evidence of abnormal serum paraprotein (λ light chains).
What Is Nephrotic Syndrome?
The term “nephrotic syndrome”(NS) refers to a combination of laboratory and clinical findings including heavy proteinuria (protein excretion greater than 3.5 g/24 h), hypoalbuminemia (less than 3 g/dL), and peripheral edema, often accompanied by hyperlipidemia, lipiduria, and less commonly by thrombosis. Typically, the urinary sediment contains no significant number of white or red blood cells, but it may contain lipid, either entrapped in casts (fatty casts), enclosed by the plasma membrane of degenerative epithelial cells (oval fat bodies), or free in the urine. 2
What Are the Mechanisms of Proteinuria, Hypoalbuminemia, Edema, Hyperlipidemia, and Hyperlipiduria in Nephrotic Syndrome?
The underlying mechanism of nephrotic syndrome is related to the renal loss of proteins due to altered structural integrity of the glomerular filtration barrier. The increased permeability to plasma proteins, which are typically selected against filtration, results in marked proteinuria. While the mechanism of hypoalbuminemia in nephrotic patients is not completely understood, it appears that the most of albumin loss is due to urinary excretion. It has been proposed that in patients with nephrotic syndrome, a substantial fraction of the filtered albumin is taken up by and catabolized in the proximal tubular cells, resulting in a much greater degree of albumin loss than estimated from the rate of albumin excretion.
The glomerular loss of proteins causes a systemic drop in plasma colloid osmotic pressure, leading to fluid shift into the extravascular space, causing edema. Decreased intravascular volume leads to decreased renal flow, triggers the renin–angiotensin–aldosterone system, and the resulting selection for salt retention exacerbates the systemic fluid extravasation to the point of anasarca.
Another consequence of decreased plasma oncotic pressure is an increased synthesis of hepatic lipoproteins containing apolipoprotein B and cholesterol resulting in hypercholesterolemia. Conversely, nephrotic hypertriglyceridemia results from impaired metabolism, rather than enhanced synthesis. It has been shown that in NS the delipidation cascade, in which very low-density lipoproteins (LDL) are converted to intermediate-density lipoprotein and then to LDL by lipoprotein lipases, is slowed.
Lipiduria is commonly present in NS. Urinary lipid may be present in the sediment, entrapped in casts (fatty casts), enclosed by the plasma membrane of degenerative epithelial cells (oval fat bodies), or free in the urine, where it can be observed under polarized light as “Maltese crosses.” 3
What Is the Differential Diagnosis of Nephrotic Syndrome?
Nephrotic syndrome may be a manifestation of primary glomerular diseases or may develop as a secondary complication of systemic disorder. 2 The most common etiologies of primary NS include minimal change disease (MCD), focal segmental glomerulosclerosis (FSGS), membranous nephropathy (MN), and idiopathic membranoproliferative glomerulonephritis (MPGN). 3 Systemic (secondary) causes of NS include diabetes mellitus causing diabetic nephropathy, amyloidosis, autoimmune mediated disorders (eg, lupus nephritis), malignancy-associated glomerular syndromes, and others. In systemic diseases, heavy proteinuria is usually seen either in advanced stages of glomerular involvement (eg, diabetic nephropathy) or in disease with capillary wall deposits (eg, membranous lupus nephritis or amyloidosis).
Nephrotic syndrome can also be seen in glomerular diseases that typically present with nephritic syndrome but may potentially (eg, between flares or in advanced disease) manifest as nephrotic; examples of such disorders are immunoglobulin (Ig) A nephropathy, infection-related glomerulonephritis, C3 glomerulonephritis, dense deposit disease, and others. Such patients may present with mixed nephrotic and nephritic syndrome.
Is There a Difference in Etiology of Nephrotic Syndrome in Different Age Groups?
When considering differential diagnosis of nephrotic syndrome, the age of the patent needs to be taken into account. The most common cause of NS in children is MCD, followed by MN and FSGS. In the United States the most common cause of NS in adults age 19 to 65 is FSGS, followed by MN and MCD. In older patients (>65), secondary causes, such amyloidosis and diabetic nephropathy, should be considered. 3
Compare the Nephrotic and Nephritic Syndrome
In contrast to nephrotic syndrome, nephritic syndrome varies widely in clinical presentation depending of the involvement of the glomeruli. Patients may present with microscopic or gross hematuria, various degrees of proteinuria, renal insufficiency, and hypertension. Urinalysis may show “active urinary sediment” containing red and/or white cell casts. There are various mechanisms underlying nephritic syndrome, most common include immune complex-mediated diseases (eg, anti-neutrophilic cytoplasmic antibody (ANCA) - associated glomerulonephritis). 2
What Are the Treatment Options in Nephrotic Syndrome?
The treatment of NS depends largely on its pathogenesis. Patients with primary NS, including MCD, FSGS, MN, and idiopathic MPGN are treated with immunosuppressive agents. In patient with secondary NS, such as diabetic nephropathy, lupus nephritis, and malignancy-associated glomerular syndromes, the therapies are targeted at the underlying systemic and neoplastic disorders, respectively.
Treatment of the different types of amyloidosis varies with the cause of fibril precursor production. In AA amyloidosis, the treatment is aimed at the relevant infectious or inflammatory disorders. Patients with amyloid light chain (AL) amyloidosis receive therapies for the underlying plasma cell dyscrasia, including autologous hematopoietic cell transplantation and/or chemotherapy. In patients with dialysis-related amyloidosis, the treatment is targeted at either altering the mode of dialysis or considering renal transplantation. For the hereditary amyloidoses in which the mutant amyloid precursor protein is produced by the liver (eg, transthyretin), liver transplantation may be considered. Novel therapeutic approaches are being developed, including agents that interfere with fibril formation or enhance their degradation.
Diagnostic Findings, Part 4
Kidney biopsy was performed to establish the cause of this patient’s proteinuria. Light microscopic examination revealed renal cortex containing 18 glomeruli, 2 of which were globally sclerosed. None of the glomeruli were segmentally sclerosed. The remaining glomeruli showed mesangial expansion and irregular thickening of capillary walls due to accumulation of acellular material that stained weakly with periodic acid–Schiff (PAS) and was negative for Jones methenamine silver (JMS) stain (Figure 1). Similar acellular material was present in the interstitium and in arterial vessel walls. There was mild patchy interstitial fibrosis and tubular atrophy involving approximately 10% of the sampled parenchyma. Arteries showed mild degree of intimal sclerosis. The acellular material stained salmon pink with the Congo red reagent and gave apple green birefringence when observed under polarized light (Figure 2). Immunofluorescence microscopy showed smudgy staining in the glomeruli and in arterial vessels for λ light chain only (Figure 3). Staining for the remaining immune reactants including IgG, IgA, IgM, C1q, C3, albumin, fibrinogen, and κ light chains was negative. Ultrastructural examination of the acellular material revealed accumulation of randomly arranged nonbranching fibrils measuring in average 10 nm (range 8-12 nm) in thickness (Figure 4). The glomerular capillary basement membranes had normal architectural organization and thickness. The foot processes of the overlying epithelial cells (podocytes) were largely preserved.
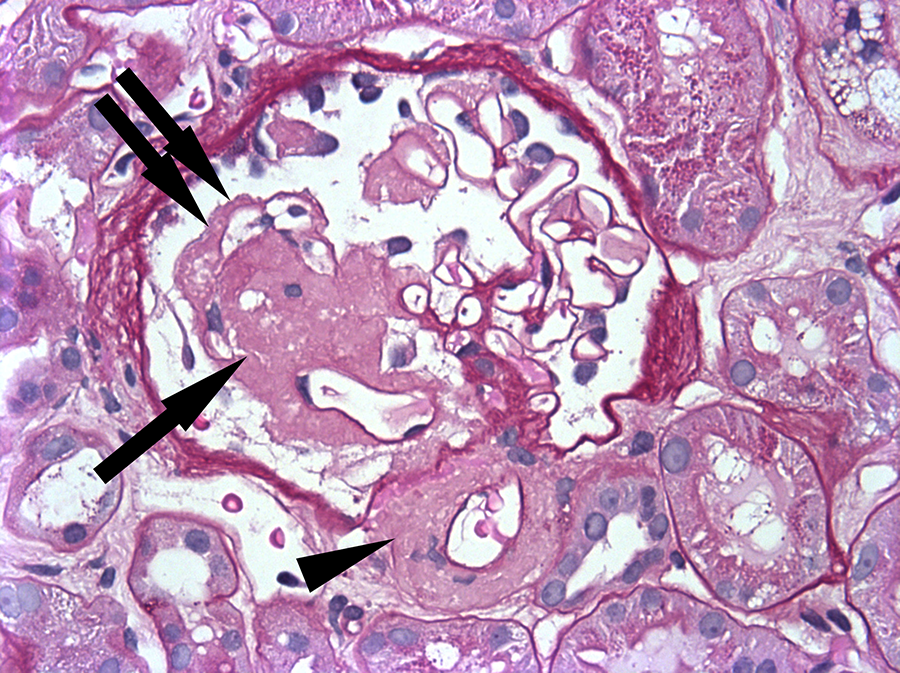
Renal amyloid: accumulation of weakly PAS stained acellular material in glomerular mesangium (arrow) and capillary wall (double arrow), and in hilar arteriole (arrowhead); (PAS, original magnification 400×). PAS indicates periodic acid–Schiff.
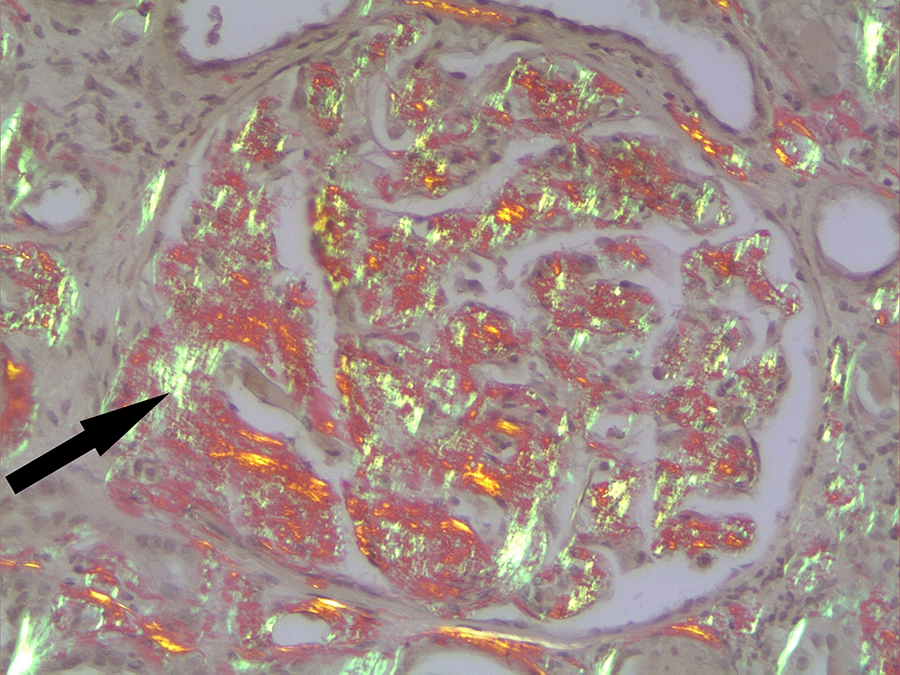
Apple-green birefringence of Congo red–stained amyloid (arrow) observed under polarized light (original magnification 400×).
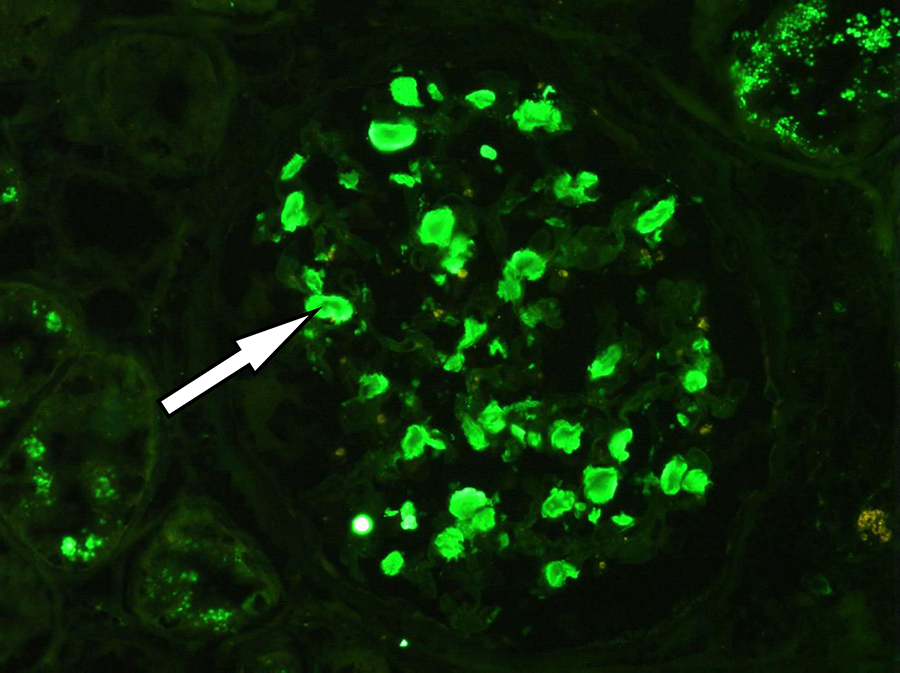
Amyloid: smudgy mesangial (arrow) and capillary wall staining for λ light chain (Immunofluorescence, original magnification 400×).
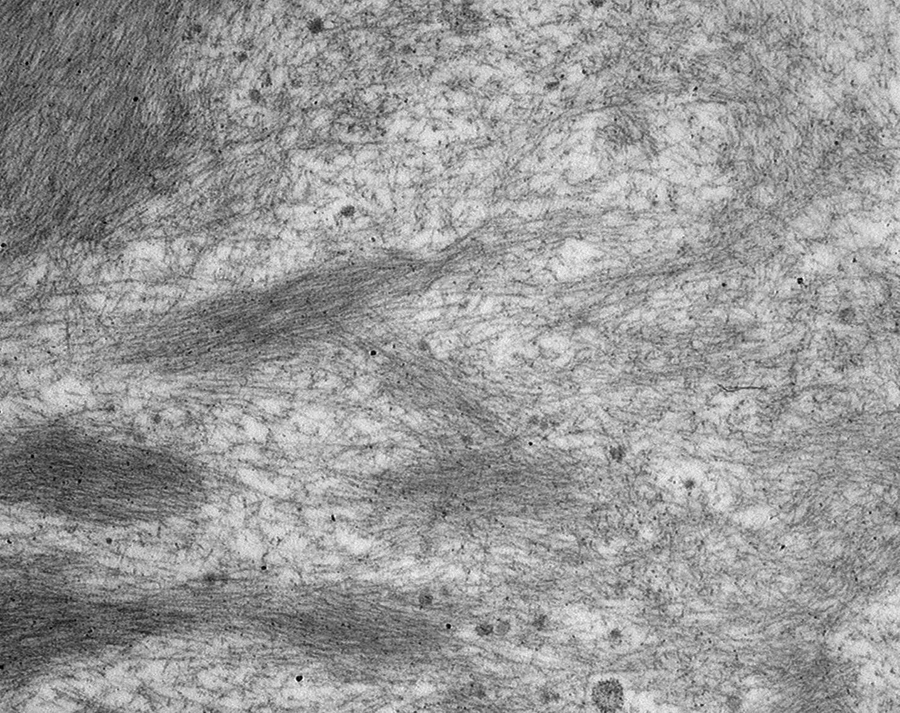
Amyloid: accumulation of randomly arranged nonbranching fibrils (TEM, direct magnification 31 000×). TEM indicates transmission electron microscopy.
Questions/Discussion Points, Part 4
What Is the Pathologic Diagnosis Based on Kidney Biopsy?
Immunoglobulin λ light chain amyloidosis (AL amyloidosis).
Define the terms: amyloid and amyloidosis
In medicine “Amyloid” is defined as mainly extracellular deposits of fibrillary protein with characteristic β-pleated sheet configuration resulting in particular properties, including affinity to Congo red dye with an orange-red appearance by light microscopy, and characteristic apple-green birefringence under polarized light. In histologic sections, amyloid appears as extracellular amorphous material that stains weakly with hematoxylin and eosin and PAS stains, demonstrates a blue-to-gray hue on trichrome stain, and is typically negative on JMS stain. Ultrastructurally, amyloid is composed of randomly arranged nonbranching fibrils measuring in average 10 nm in thickness. 4 While the amyloid has a conserved morphologic appearance, it has a “biochemically heterogeneous” origin, with at least 36 proteins known to form the characteristic non-branching, β-pleated sheet configuration. 5
Amyloidosis is a pathologic process, either systemic or localized, in which there is deposition of amyloid fibrils in the extracellular spaces, leading to the functional impairment of the affected organs.
Discuss different types of amyloidosis
Amyloidosis may be systemic (generalized), involving several organ systems, or it may be localized to a single organ, such as the heart or kidneys. The proteins that form amyloid may be normal proteins that have an inherent tendency to associate and form fibrils, particularly when produced in increased amounts or mutant proteins that are prone to misfolding and aggregation. 4
The current nomenclature of amyloidosis is based on the identification of precursor protein forming the amyloid fibrils. 5 Examples of systemic amyloidosis include AL amyloidosis (precursor protein: immunoglobulin light chain), AA amyloidosis (precursor protein: serum amyloid A), and Aβ2 M amyloidosis (precursor protein: β2-Microglobulin). While AL amyloidosis is frequently associated with plasma cell dyscrasias or other monoclonal B-cell proliferative disorders, the AA amyloidosis follows chronic inflammatory disorders, and both types can involve many organs, except the central nervous system. Aβ2 M amyloidosis is seen in patients with history of long-standing hemodialysis and predominately involves musculoskeletal system. Examples of localized amyloidosis include ACal amyloidosis (precursor protein: (Pro)calcitonin) associated with medullary carcinoma of thyroid, AIAPP amyloidosis (precursor protein: Islet amyloid peptide) in pancreatic Islets of Langerhans, and Aβ amyloidosis (precursor protein: Aβ protein precursor) in central nervous system of patients with Alzheimer disease, and recently recognized leukocyte chemotactic factor 2 amyloid (ALECT2) amyloid (precursor protein: Leukocyte chemotactic factor 2) seen primarily in kidneys of patients of Hispanic origin.
The most common hereditary type of amyloidosis is transthyretin amyloidosis (ATTR) amyloid (precursor protein: Transthyretin, variants) with amyloid deposits in many organs and systems, including peripheral nervous system, heart, eyes, and leptomeninges.
What Diagnostic Procedures Could Be Performed to Determine the Type of Amyloid in the Biopsied Tissue?
Since all types of amyloid stain positively with Congo red, it is critical to characterize the nature of the amyloid protein that is present since each of the various types of amyloidosis would be treated differently. There are a few possible approaches to identify the type of amyloid in the sampled tissue. 6 Immunofluorescence microscopy (as performed in the current case, Figure 3) can identify the type of heavy and/or light chain amyloid. This study is performed as a part of a standard evaluation of kidney and most skin biopsies, but it requires frozen tissue to be completed. Immunohistochemistry can be performed on paraffin-embedded tissue and can be used to identify certain protein such as AA amyloid, Aβ amyloid, apolipoprotein AI, fibrinogen, kerato-epithelin, lactoferrin, lysozyme, β2-microglobulin, immunoglobulin-derived λ-light and κ-light chains, ALECT2, and transthyretin. In cases with inconclusive or negative staining, mass spectrometry may be performed on tissue samples to reliably determine the type of protein present.
What Is the Pathogenesis of the Amyloid Deposits in This Case?
Amyloid light chain amyloidosis (previously referred to as primary amyloidosis) develops due to excess production of immunoglobulin light chains (in 75% of cases λ type) by abnormal population of plasma cells, as seen in plasma cell disorders (eg, monoclonal gammopathy of undetermined significance, multiple myeloma (MM), or Waldenström macroglobulinemia). 4 Light chains are soluble and have sufficiently low molecular weight to pass through the glomerular filter and deposit in the mesangium and in the glomerular capillary walls. In the blood vessels, the light chains accumulate under the endothelium, eventually replacing the smooth muscle wall. Light chains undergo conformational changes that lead to formation of predominantly antiparallel β-pleated sheet configuration. Normally, intracellular misfolded proteins are degraded in proteasomes, and extracellular protein aggregates are taken up and degraded by macrophages. In amyloidosis, these quality control mechanisms fail and fibrillar proteins accumulate outside of cells. 4
Diagnostic Findings, Part 5
Analysis of bone marrow specimens showed an increased percentage of plasma cells (12%) and immunostaining revealed a positive reactivity for a λ light chain.
Questions/Discussion Points, Part 5
What Is the Significance of the Bone Marrow Biopsy Findings in This Patient?
The presence of increased number (>10%) of plasma cells expressing λ light chains is a defining feature of MM.
What Are the Diagnostic Criteria of Multiple Myeloma?
The diagnosis of MM requires findings of clonal bone marrow plasma cells ≥10% or biopsy-proven, bony or extramedullary, plasmacytoma. In addition, one or more of the following myeloma defining events or biomarkers of malignancy must be present fulfill the diagnostic criteria: hypercalcemia (>11 mg/dL), renal insufficiency (creatinine clearance <40 mL/min, or serum creatinine >2 mg/dL), anemia:hemoglobin <10 g/L, and one or more osteolytic lesions on skeletal radiography, computed tomography (CT), or positron emission tomography-CT; clonal bone marrow plasma cell percentage ≥10%, involved/uninvolved serum-free light chain ratio ≥100, and >1 focal lesions on MRI studies. 7
Discuss the renal complications of multiple myeloma
The renal injury in MM is predominately related to the production of increased amounts of monoclonal proteins (paraproteins) and less commonly due to a direct infiltration of kidney by plasma cells. 8 Paraproteins are composed of whole Ig (most commonly IgG) and/or excess of light or heavy chains. It is the biochemical characteristics of the individual immunoglobulins and light or heavy chains that determine which kidney disease (if any) occurs. 7 While the prevalence of different renal lesions varies among the lymphoproliferative disorders, the most common renal complication in MM is myeloma cast nephropathy followed by AL amyloidosis and monoclonal immunoglobulin deposition disease that encompasses the light and heavy chain deposition disease (LCDD and HCDD).
Myeloma cast nephropathy is characterized by the presence of atypical tubular casts composed of precipitated light chains. Typically, the casts have a hard or cracked appearance, sharp, uneven edges, and may be associated with giant cell reaction (Figure 5). Sometimes the light chains precipitate into crystalline structures. The casts are usually associated with acute tubular injury; therefore, the patients typically present with acute kidney injury.
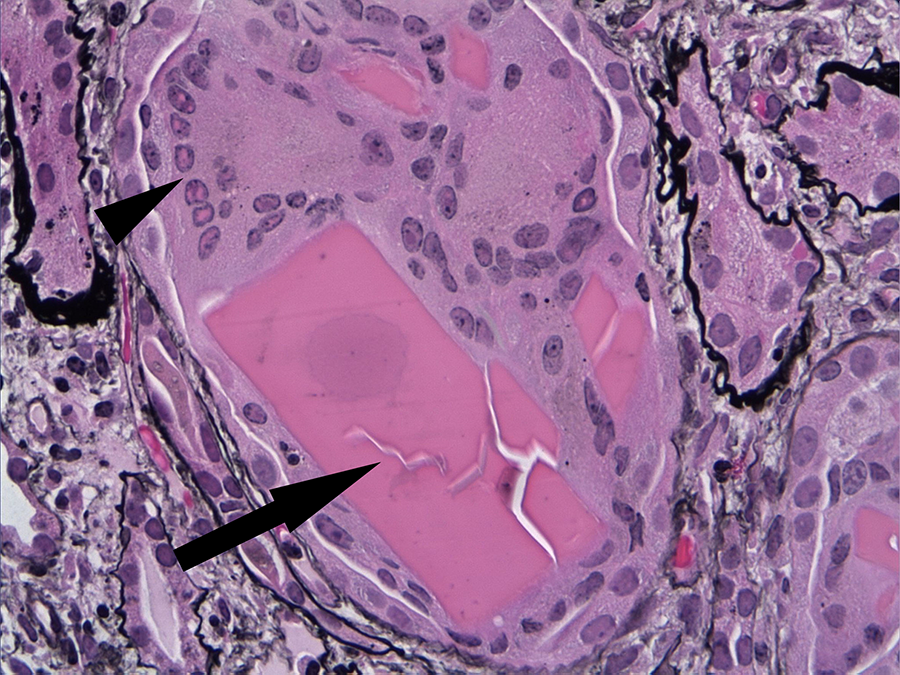
Myeloma cast nephropathy: crystalline fractured cast (arrow) with associated giant cell reaction (arrowhead), (original magnification 400×).
Monoclonal immunoglobulin deposition disease (LCDD and HCDD) are due to accumulation of precipitated light or heavy chains. Due to different physical properties, these light chains do not form amyloid fibrils, but get deposited in various organs in finely granular pattern, and do not stain with Congo red. In LCDD, the deposits are most often composed of κ light chains. In the kidneys, LCDD is characterized by deposition of the light chains in the mesangium, glomerular, and/or tubular basement membranes, and in cell membranes of the smooth muscles. The light chain deposits can be detected by immunofluorescence microscopy by characteristic pattern of linear staining in the glomerular and/or tubular basement membranes and in smooth muscle cell membranes and somewhat smudgy staining in mesangial areas (Figure 6). Electron microscopy confirms the presence of finely granular deposits in the same locations (Figure 7). Heavy chain deposition disease is morphologically similar, but the deposits are made of heavy chains. In some patients with LCDD, routine electrophoretic techniques may not demonstrate a monoclonal protein in the serum or urine, but one can be detected using serum free light chain essay. Patients with predominant glomerular deposition of light or heavy chains may present with nephrotic syndrome (similar to AL amyloidosis), while those with predominant tubular deposits may present with renal insufficiency and relatively mild proteinuria.
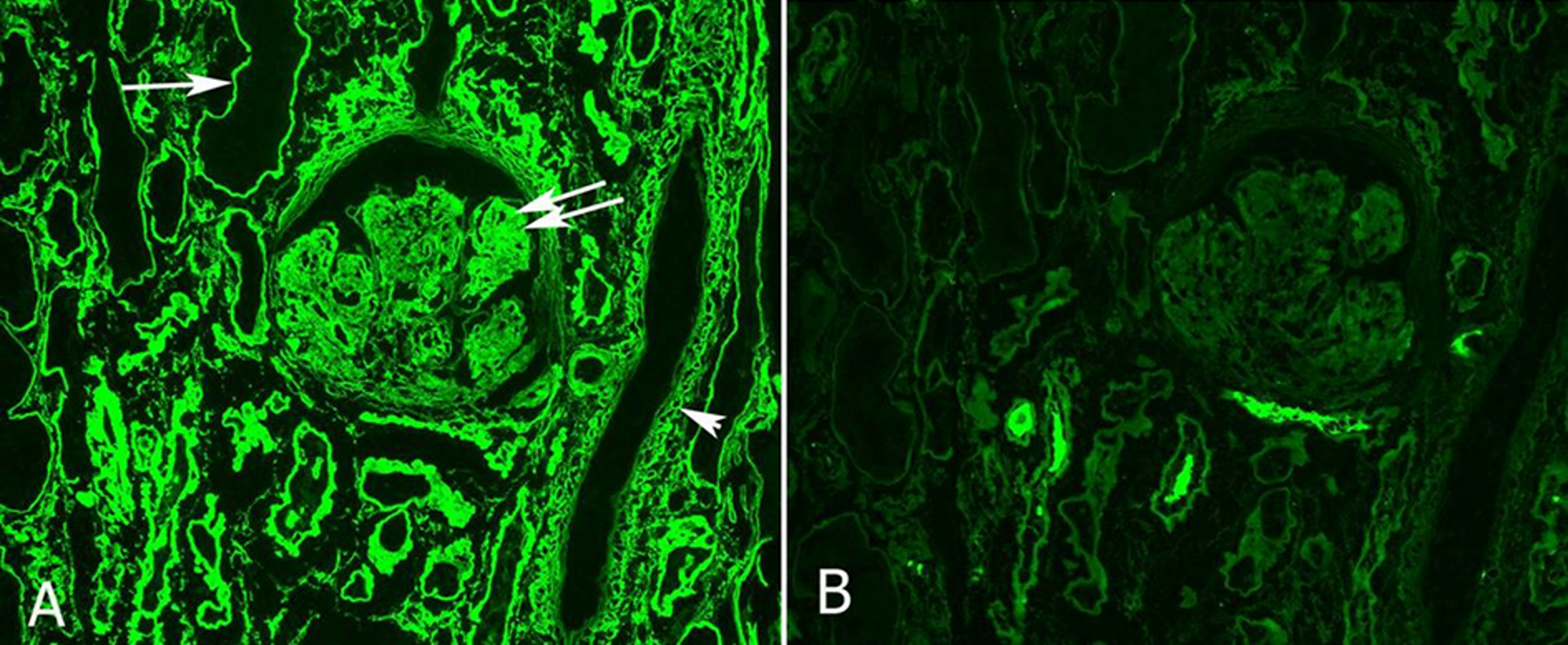
Light chain deposition disease. A, Positive staining for κ light chains in glomerular and tubular basement membranes (arrow), mesangium (double arrow), and in cell membranes of smooth muscle cells (arrowhead). B, Corresponding negative stain for λ light chains (immunofluorescence, original magnification 200×).
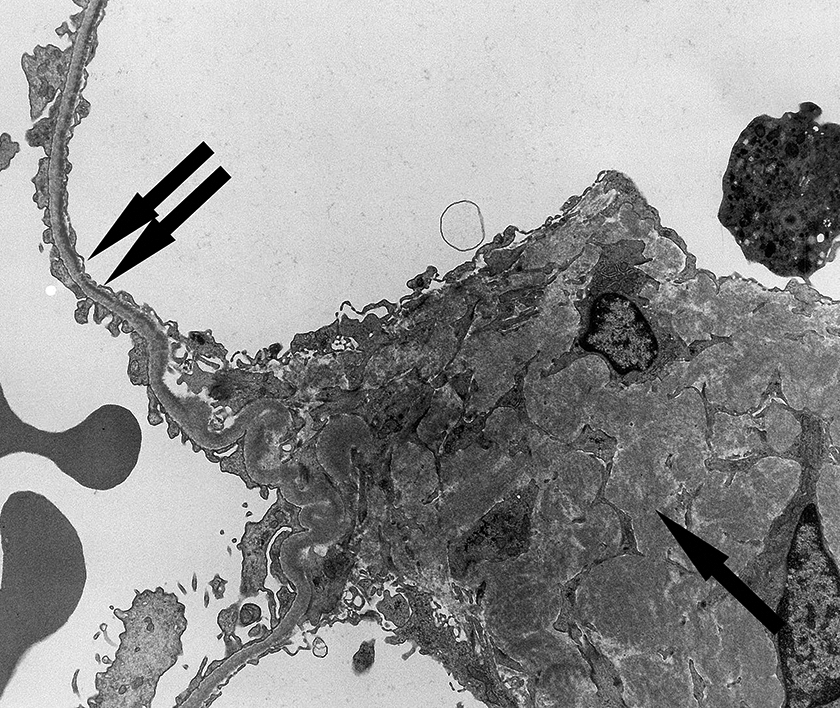
Light chain deposition disease: accumulation of finely granular electron dense deposits in mesangium (arrow) and in subendothelial location of capillary walls (double arrows); (TEM, direct magnification 4400×). TEM indicates transmission electron microscopy.
Other, less common renal complications of MM include proximal light chain tubulopathy (presenting as Fanconi syndrome), immune complex-mediated disorders, such as proliferative glomerulonephritis with monoclonal immunoglobulin deposits and cryoglobulinemic glomerulonephritis (that may present as nephritic syndrome with variable degree of proteinuria), as well as glomerular disorders with organized deposits, such as fibrillary glomerulonephritis and immunotactoid glomerulopathy (that also present with variable proteinuria).
Teaching Points
Nephrotic syndrome is defined as heavy proteinuria (protein excretion greater than 3.5 g/24 h), hypoalbuminemia (less than 3 g/dL), and peripheral edema, often accompanied by hyperlipidemia, lipiduria and less commonly by thrombosis.
Nephritic syndrome varies widely in clinical presentation and includes microscopic or gross hematuria, various degrees of proteinuria, renal insufficiency, and hypertension. Urinalysis may show “active urinary sediment” containing red and/or white cell casts.
The most common cause of nephrotic syndrome in children is minimal change disease, in adults (ages 19-65, in USA) is focal segmental glomerulosclerosis, and in older patients (>65), the systemic causes, such amyloidosis and diabetes, should be considered.
Amyloid is an extracellular fibrillary protein with characteristic β-pleated sheet configuration resulting in particular properties, including affinity to Congo red and apple-green birefringence after such staining.
Amyloidosis is a pathologic process, either systemic or localized, in which there is deposition of amyloid fibrils in the extracellular spaces, leading to the functional impairment of the affected organs.
The current classification of amyloid is based on the type of precursor protein forming the amyloid fibrils, for example, AL amyloidosis (precursor protein: immunoglobulin light chain), AA amyloidosis (precursor protein: serum amyloid A), ATTR amyloid (precursor protein: Transthyretin, variants).
Renal complications of MM are predominately related to the excess production of paraproteins. The most common are myeloma cast nephropathy, AL amyloidosis, and monoclonal immunoglobulin deposition disease.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
